Summary
Background
This case report describes two cases of extralobar pulmonary sequestration in adults with and without torsion/necrosis.
Case Reports
Non-complicated extralobar pulmonary sequestration was found incidentally in a 50-year-old asymptomatic woman (Case 1), diagnosed with the presence of a branching structure in a mass lesion and blood supply from the right inferior phrenic artery. Another case of a 38-year-old woman presented with a sudden onset of back pain caused by extralobar pulmonary sequestration with torsion/necrosis (Case 2). A 4-cm fusiform mass in the paravertebral region showed enhancement in the peripheral rim only, and no feeding artery. These were the same as it had been reported typical findings in extralobar pulmonary sequestration with necrosis. On magnetic resonance imaging, the masses in both cases showed inhomogeneous low signal and branching high signal on T2-weighted images. That was characteristic for a stroma without dilated alveoli as a solid part and dilated alveoli as fluid regions.
Conclusions
By comparing those two cases, we came to a conclusion that only T2-weighted imaging reflects the native structure, even after infarction. Although differentiation from a cystic tumor with hemorrhage or infection can be problematic, inhomogeneous low signal and branching high signal on T2-weighted images may help us distinguish extralobar pulmonary sequestration from other cystic lesions.
Keywords: Bronchopulmonary Sequestration; Necrosis; Torsion, Mechanical
Background
Extralobar pulmonary sequestration is a congenital anomaly resulting in the formation of ectopic lung tissue outside the normal lung with its own pleural investment, but no communication with the normal tracheobronchial tree. Without associated malformations, symptomatic cases are rare, and are usually found incidentally in adulthood. In such cases, differential diagnosis from other mass lesions is needed.
We present a case of necrotic extralobar pulmonary sequestration in a patient who presented with a back pain, which we were unable to diagnose correctly. We also discuss imaging features in comparison with a non-complicated case.
Case Reports
Case 1
A healthy and asymptomatic 50-year-old woman showed suspected right pleural effusion on abdominal ultrasonography carried out for screening. Chest computed tomography (CT) showed a well-circumscribed dome-like solid mass with cysts and branching fluid collection above the right dorsal diaphragm (Figure 1). The abutting lung was displaced and slightly compressed by the mass, and internal structures were not connected to the mass. Laboratory data including tumor markers (carcinoembryonic antigen, 1.9 ng/mL; cytokeratin fragment, 1.4 ng/mL; Pro-gastrin-releasing peptide, 18.2 pg/mL) were all within normal limits. Magnetic resonance imaging showed the solid part of the mass as a region of inhomogeneous low signal intensity on T2-weighted images and enhanced strongly on contrast-enhanced T1-weighted imaging, while cysts and fluid collection within the mass were isointense with water on both T1- and T2-weighted images (Figures 2 and 3). On angiography, the mass was supplied by the dilated right inferior phrenic artery and showed strong enhancement but no drainage vein was detected. Extralobar pulmonary sequestration was suspected because of the presence of branching structures within the mass and the blood supply from the right inferior phrenic artery. Two days after preoperative embolization as operative support, the mass was resected by video-assisted thoracoscopic surgery. On surgical findings, an ectopic lung tissue-like mass was located above the right diaphragm, with its own pleural investment. The mass was connected to the diaphragm by a pedicle, without any connection to the normal lung, and was resected at the pedicle.
Figure 1.

Case 1. Contrast-enhanced chest CT. A solid mass with cyst and branching fluid collection is apparent above the right dorsal diaphragm.
Figure 2.

Case 1. Contrast-enhanced MRI, sagittal image. (A) T2WI; (B) T1WI (FS); (C) contrast-enhanced T1WI (FS). The solid part of the mass showed inhomogeneous low signal intensity on T2WI and strong enhancement on contrast-enhanced T1WI (FS).
Figure 3.

(A–C) Case 1. Unenhanced MRI, sagittal T2 WI. The mass shows an inhomogeneous low signal with cyst and branching signal hyperintensity.
Macroscopically, the mass comprised solid and cystic lesions. Microscopically, various sizes of cystic dilated alveoli and fibrotic stroma were present and extralobar pulmonary sequestration was diagnosed.
Case 2
A 38-year-old healthy woman presented after a sudden onset of back pain that disappeared the next day, then reappeared the day after. Laboratory tests showed slight elevations in C-reactive protein (CRP) (1.91 mg/dL), creatine phosphokinase (221 IU/L), and lactate dehydrogenase (219 IU/L), and other values including white blood cell count were within normal range. Electrocardiography, abdominal ultrasonography, gastrointestinal endoscopy and chest radiography were normal. Contrast-enhanced CT of the chest and abdomen revealed a 4-cm fusiform mass in the azygo-esophageal recess (Figure 4). That mass demonstrated iso-density with muscles, and only the peripheral rim was enhanced. It was abutting the lung, esophagus, vertebrae, and left atrium, but those surrounding structures did not show any abnormalities. A small amount of pleural effusion was seen on the right side, i.e. at the location of the mass. No other lesions that might have caused the symptoms were evident. On magnetic resonance imaging, the mass showed inhomogeneous low signal with branching signal hyperintensity on T2-weighted images, slight hyperintensity on T1-weighted images, and contrast enhancement only at the periphery (Figures 5 and 6). Four days after onset, pleural effusion increased and CRP was elevated to 5.52 mg/dL. A mass was suspected – an acute necrotic solid tumor or complicated (hemorrhagic or infectious) cystic lesion, such as solitary fibrous tumor, neurogenic, foregut cyst or cystic teratoma. Although we could not establish the diagnosis, the mass was considered the cause of the symptoms, and video-assisted thoracoscopic surgery was attempted. Intraoperatively, a dark red-brown mass and small amounts of hemorrhagic pleural effusion were identified in the pleural cavity. Parietal pleura near the mass appeared red, which suggested spreading inflammation. The mass was easily separated from the surrounding tissue, and was connected to the mediastinum by a twisted pedicle. The pedicle passed around the esophagus, but connection to the aorta could not be recognized. The mass was resected at the pedicle. Microscopically, the mass contained circular lining cartilage and fat, and received its blood supply from an artery in the pedicle (Figure 7). That vessel was an elastic artery, with a large diameter compared to the size of the mass. The mass displayed total hemorrhagic necrosis of the remaining native structures without granulation tissue, indicating a pattern of necrosis arising within the past several weeks. We diagnosed necrotic extralobar pulmonary sequestration with torsion based on the clinical course and surgical and pathological findings. Symptom completely resolved following resection.
Figure 4.
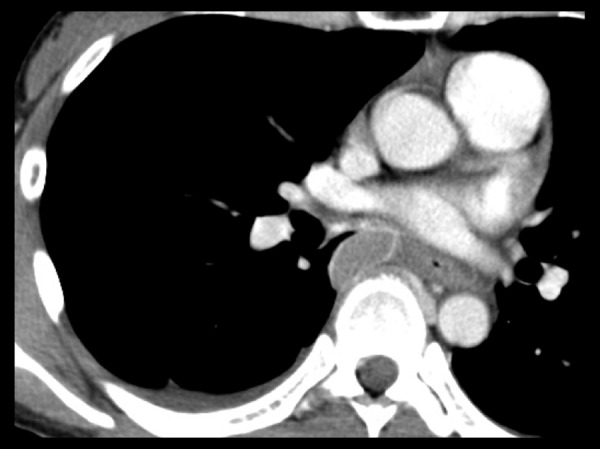
Case 2. Contrast-enhanced chest CT. A mass is present in the azygo-esophageal recess, and only the periphery is enhanced. A small amount of pleural effusion is seen on the right side.
Figure 5.
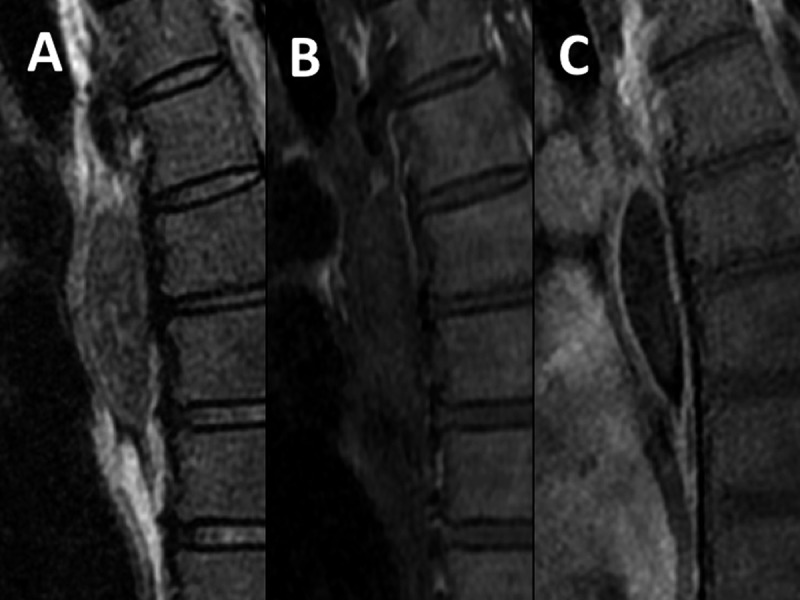
Case 2. Contrast-enhanced MRI, sagittal image. (A) T2WI (FS); (B) T1WI; (C) enhanced T1WI (FS). The mass shows inhomogeneous low signal on T2WI (FS), slightly high signal on T1WI, and enhancement only at the periphery.
Figure 6.

Case 2. Unenhanced MRI, sagittal T2WI (FS). The mass shows an inhomogeneous low signal with branching signal hyperintensity.
Figure 7.

Histopathological study of Case 2. The mass shows total hemorrhagic necrosis and remaining circular lining cartilage.
Discussion
Pulmonary sequestration is defined as ectopic lung tissue lacking normal communication to the tracheobronchial tree and receiving blood supply from systemic vessels [1]. This pathology is classified into intra- and extralobar types.
Intralobar pulmonary sequestration is more frequent than extralobar, and is covered by the visceral pleura in common with the normal lung. Although some cases are congenital, the majority is a result of changes to normal lung tissue after recurrent inflammation [2].
On the other hand, extralobar pulmonary sequestration is an absolutely congenital maldevelopment with its own visceral pleura, and symptomatic cases are rare [3]. Diagnosis is often made in the first year of life because of an associated anomaly such as diaphragmatic hernia, bronchogenic cyst or pericardial defect. In the absence of such malformations, extralobar pulmonary sequestration is usually found incidentally in adulthood, and must be differentiated from other mass lesions including neoplasms. Extralobar pulmonary sequestration in an adult is occasionally symptomatic, without other concomitant anomalies, because of infection, inflammation, hemorrhage, or infarction.
Extralobar pulmonary sequestration is typically found in the thorax on the left side (65–90%), and at the posterior costodiaphragmatic sulcus between the diaphragm and lower lobe of the lung (63–77%). It is sometimes found in the mediastinum, interperitoneally, or in the pericardium. Extralobar pulmonary sequestrations are between 0.5 and 15.0 cm, usually 3 to 6 cm, pyramidal or ovoid in shape, and with a vascular pedicle. Grossly, the tissue resembles lung tissue, and is covered by glistening pleural surface. The feeding artery is often from the aorta (80%), but can be from the splenic, gastric, subclavian, intercostal or internal mammary artery.
Microscopic findings vary depending on the tissue composition and degree of associated changes, from nearly normal lung tissue to tissue resembling cystic adenomatoid malformation, composed of dilated alveoli, thickened stroma, and inflamed fibrotic stroma with mucin-filled airways [4,5]. Because secretions are retained, with no drainage due to the lack of communication with the normal tracheobronchial tree, recurrent inflammation and cystic and fibrotic changes occur over time.
A typical radiological finding is a mass in the lower hemithorax, and it is important for the diagnosis of extralobar pulmonary sequestration to identify the systemic vascular supply using any modality – angiography, ultrasonography, CT, or magnetic resonance imaging. Lee et al. reported that 3-dimensional CT angiography could clearly show the feeding artery and drainage vein [6]. In Case 1, these could not be identified, probably because the vessels were too thin, and arterial-phase scans were not performed. The mass in Case 1 was inflamed, comprising cystic dilated alveoli and fibrotic stroma. Imaging reflected cystic dilated alveoli as cystic lesions, and stroma without dilated alveoli as solid parts, with the latter showing an inhomogeneous low signal on T2WI and contrast enhancement.
Infarcted extralobar pulmonary sequestration was reported in 9 cases (as well as another 6 cases in Japanese literature), and 3 cases showed torsion [7–9]. This pathology manifests with chest or abdominal pain. Huang et al. reported a case with prodromal intermittent pain [8]. Symptoms in the present case were also intermittent over the initial few days, possibly indicating changes in the state of torsion.
Imaging features of extralobar pulmonary sequestration with necrosis reflect the hemorrhagic necrosis, and were reported as: 1) a 4–5-cm ovoid-pyramidal mass in the paravertebral region; 2) a homogeneous high-density mass enhancing only on the periphery on CT; 3) reactive pleural effusion or hemothorax; and 4) no feeding artery identified [7–9].
The lesion in Case 2 presented all these findings. The signal on magnetic resonance imaging reflects acute hemorrhage and necrosis, and would change with the interval from onset. Probably because of thrombosis, no feeding could be detected [7]. Infarcted extralobar pulmonary sequestration without torsion was reported, and we thought this could have taken place because the abnormal vessel easily undergoes atheromatous changes [10].
In Case 2, the problem was differentiating the lesion from cystic tumor with hemorrhage or infection, because the mass was enhanced only on the periphery. The inhomogeneous low signal intensity and branching signal hyperintensity on T2-weighted images reflected the structures contained, and were also similar to the solid and fluid components of the mass in Case 1. This suggests that T2-weighted imaging might reflect the tissue structures before necrosis, to a certain degree, while other sequences cannot. Findings on T2-weighted images might thus help to distinguish these lesions from cystic tumor with hemorrhage or infection, which show homogeneous signal intensity, fluid-fluid level, and have a webbed appearance.
Conclusions
We reported on two cases of extralobar pulmonary sequestration, including one case with torsion/necrosis. Radiographic imaging was able to reflect the state of hemorrhagic necrotizing lung tissue and only T2-weighted imaging reflected native structures even after infarction. Although infarcted extralobar pulmonary sequestration is a rare acute disease, understanding the pathology and its appearance with hemorrhagic necrosis may improve preoperative diagnosis.
Footnotes
Conflict of interest
The authors declare to have no conflict of interest.
References
- 1.Pryce DM. Lower accessory pulmonary artery with intralobar sequestration of lung: a report of seven cases. J Path Bact. 1946;58:457–67. [PubMed] [Google Scholar]
- 2.Stocker JT. Sequestrations of the lung. Semin Diagn Pathol. 1986;3:106–21. [PubMed] [Google Scholar]
- 3.Rosado-de-Christenson ML, Frazier AA, Stocker JT, Templeton PA. Extralobar sequestration: Radiologic pathologic correlation. Radiograpics. 1993;13:425–41. doi: 10.1148/radiographics.13.2.8460228. [DOI] [PubMed] [Google Scholar]
- 4.Hasleton PS. Spencer’s Pathology of the Lung. New York: McGraw-Hill; 2013. pp. 71–73. [Google Scholar]
- 5.Travis WD. AFIP Atlas of nontumor Pathology, Fascicle2-Non-Neoplastic Disorders of the Lower Respiratory Tract. 2002. pp. 484–86. [Google Scholar]
- 6.Lee EY, Dillon JE, Callahan MJ, Voss SD. 3D multiditector CT angiographic evaluation of extralobar pulmonary sequestration with anomalous venous drainage into the left internal mammary vein in pediatric patient. Br J Radiol. 2006;79(945):99–102. doi: 10.1259/bjr/45058144. [DOI] [PubMed] [Google Scholar]
- 7.Chun EJ, Goo JM, LEE HJ, et al. Extralobar pulmonary sequestration with hemorrhagic infarction in an adult. J Thorac Imaging. 2007;22(2):166–68. doi: 10.1097/01.rti.0000213561.55914.41. [DOI] [PubMed] [Google Scholar]
- 8.Huang EY, Monforte HL, Shaul DB. Extralobar pulmonary sequestration presenting with torsion. Pediatr Surg Int. 2004;20(3):218–20. doi: 10.1007/s00383-004-1156-0. [DOI] [PubMed] [Google Scholar]
- 9.Chong NV, Polley TZ, Jr, Gelger JD. Infarction of extralobar pulmonary sequestration after blunt trauma. J Pediatr Surg. 2007;42(6):1127–29. doi: 10.1016/j.jpedsurg.2007.01.069. [DOI] [PubMed] [Google Scholar]
- 10.Yajima H, Tomoyasu H, Tanimura S, et al. A case of extralobar pulmonary sequestration with pulmonary sequestration infarction-A collective review of 142cases reported in Japan. The Journal of the Japanese Associarion for Chest Surgery. 1999;13(2):181–90. [Google Scholar]


