Abstract
Background
Pulmonary arterial hypertension is a severe and progressive disease. Its early diagnosis is the greatest clinical challenge.
Objective
To evaluate the presence and extension of the delayed myocardial contrast-enhanced cardiovascular magnetic resonance, as well as to verify if the percentage of the myocardial fibrosis mass is a severity predictor.
Methods
Cross-sectional study with 30 patients with pulmonary arterial hypertension of groups I and IV, subjected to clinical, functional and hemodynamic evaluation, and to cardiac magnetic resonance.
Results
The mean age of patients was 52 years old, with female predominance (77%). Among the patients, 53% had right ventricular failure at diagnosis, and 90% were in functional class II/III. The mean of the 6-minute walk test was 395m. In hemodynamic study with right catheterism, the mean average pulmonary arterial pressure was 53.3mmHg, of the cardiac index of 2.1L/ min.m2, and median right atrial pressure was 13.5 mmHg. Delayed myocardial contrast enhanced cardiovascular magnetic resonance was found in 28 patients. The mean fibrosis mass was 9.9 g and the median percentage of fibrosis mass was 6.17%. The presence of functional class IV, right ventricular failure at diagnosis, 6-minute walk test < 300 meters and right atrial pressure ≥ 15 mmHg, with cardiac index < 2.0 L/ min.m2, there was a relevant association with the increased percentage of myocardial fibrosis.
Conclusion
The percentage of the myocardial fibrosis mass indicates a non-invasive marker with promising perspectives in identifying patients with high risk factors for pulmonary hypertension.
Keywords: Hypertension, Pulmonary / diagnosis; Risk Assessment; Endomyocardial Fibrosis; Magnetic Resonance Imaging
Introduction
Pulmonary Hypertension (PH) is a syndrome caused by different conditions affecting pulmonary circulation. The causes of PH that involve the pulmonary arterial bed include Pulmonary Arterial Hypertension (PAH) and Chronic Thromboembolic Pulmonary Hypertension (CTPH), known as group I and IV of the current PH classification (Dana Point, 2008)1.
PAH is defined as a Mean Pulmonary Arterial Pressure (mPAP) > 25mmHg at rest, in a situation of Pulmonary Artery Wedge Pressure (PAWP) ≤ 15mmHg, with Pulmonary Vascular Resistance (PVR) > 3 units Wood2. This condition has a poor prognosis, with mortality of approximately 15% in 1 year for those treated with modern therapy 3. Right Ventricular Failure (RVF) is the main cause of death4.
Efforts are made in order to obtain an early diagnosis. Recently, prognostic factors that have an impact on survival were reviewed5. Mortality is higher for patients with Functional Class (FC) III and IV6. The rapid progression of symptoms, as well as signs of RVF at diagnosis, indicates a worse prognosis. The result of the 6-Minute Walk Test (6MWT) is an independent predictor of survival7. The presence of pericardial effusion, increased right atrium and signs of right ventricular failure, at least moderate, on transthoracic echocardiogram are consistent predictors of mortality8. Patients with increased Right Atrial Pressure (RAP) and decreased Cardiac Index (CI) had a higher death risk9.
Study including 64 patients stated that Cardiac Magnetic Resonance (CMR) imaging revealing an impaired function of Right Ventricle (RV) - systolic volume ≤ 25mL/m2, end-diastolic volume of RV ≥ 84mL/m2 and end-diastolic volume of Left Ventricle (LV) ≤ 40mL/m2 - are independent predictors of mortality and therapeutic failure10.
The Delayed Myocardial Enhancement (DME) obtained by CMR is a common finding in patients with PAH. Studies have shown its relation with right ventricular function and abnormalities in hemodynamics of pulmonary circulation11.
The objective of this study was to evaluate the presence and extension of DME by CMR and verify if the percentage of fibrosis mass is an indicator of severity in PAH.
Methods
Study design and patients screening
Cross-sectional study of patients in PAH outpatient clinic at the Clementino Fraga Filho University Hospital (CFFUH), who underwent CMR between May 2010 and April 2012.
This study selected and included 30 patients over 18 years old, irrespective of gender, class, social group or ethnicity, followed-up at the outpatient clinic of PH and CFFUH, and patients with PAH from groups I and IV, who underwent clinical evaluation and hemodynamics for starting a specific treatment.
These patients underwent all exams in the PH evaluation protocol: specific laboratory, chest X-ray, pulmonary function test, ventilation/perfusion (V/P) scintigraphy, chest angiotomography, Right Catheterization (RC) and 6MWT.
Considering an interval of up to 72 hours from RC, these patients were subjected to CMR, with a protocol specific for PH.
Patients with claustrophobia or any type of ferromagnetic implants (pacemaker, orthopedic prosthesis, etc.), patients who have renal dysfunction with creatinine clearance < 30 mL / minute and contraindication for RC due to increased risk of bleeding as a result of severe blood dyscrasia (PTA < 60%, PTT rel > 2.0 and platelets < 50,000cel/mm3).
All patients who participated in this study received an informed consent, and the study was approved by the Research Ethics Committee of CFFUH.
Hemodynamic evaluation by RC
RC was carried out in CFFUH hemodynamic laboratory by an interventional cardiologist from the PH group, inserting a Swan-Ganz catheter through the internal jugular vein (model 131F7, Edwards Baxter, Irvine, CA, USA).
The catheter was positioned in West zone III. Confirmation of its proper position was performed by scan and comparison of the variation of Diastolic Pulmonary Artery Pressure (dPAP) in relation to that of the Pulmonary Artery Occlusion Pressure (PAOP) with respiratory cycle.
Monitoring of pressure, CO (Cardiac Output), and of electrocardiographic tracing was performed with Hewllet-Packard device, model M1176-A (Hewlett-Packard, Walthan, MA, EUA).
After the proper monitor calibration, we directly obtained data from RAP, Systolic Pulmonary Artery Pressures (sPAP), dPAP, mPAP, PAOP and Cardiac Frequency (CF). Blood samples were collected, from the distal lumen of Swan-Ganz catheter, for analyzing the Mixed Venous Oxygen Saturation (MVO2S).
CO measurement was performed by thermodilution technique. Measurements with 10mL physiological saline injection were carried out, at ambient temperature, in any phase of the respiratory cycle, until we obtained three measurements with <10% variation. The mean value was then used in the study.
Using the CO result, we calculated PVR.
Hemodynamic evaluation by CMR
Exams were performed by a radiologist with 10 years of experience, using a 1.5-T magnetic resonance device (Siemens Magneton Avanto, Erlangen, Germany), operating at 45mTm-1 maximum power gradient and 200Tm-1s-1 slew rate, using six anterior canals and six posterior canals for data collection. Patients were requested to exhale at maximum and hold their breath during sequences. TRUE FISP (free steady-state precession) cine resonance sequence was used with ARGUS software, in order to evaluate cardiac function and calculate right and left systolic function indexes (ejection fraction through Simpson's volumetric method), end-diastolic and systolic volumes, ejective volume, cardiac output, and ventricular mass in short axis and horizontal axis of ventricles, with parallel acquisition technique (TR: 200ms; FOV: 400mm; matrix: 128 x 88; flip angle: 15°; slice thickness: 6mm). Moreover, 10 minutes after administrating 0.2mmoL/kg-1 gadodiamide (Dotaren TM, Gerbet, France), and after requesting the patient to take a deep breath and hold it, PSIR (phase-sensitive inversion recovery - TR: 700ms; TE: 4.18ms; TI: 300ms; FOV: 340mm; matrix: 156 x 256; flip angle 25°; slice thickness: 8mm) sequence, using same position and number of cuts from TRUE FISP cine sequence for evaluation of delayed myocardial enhancement.
Fibrosis volume was manually calculated based on the outline of hypertensive myocardial areas obtained with CMR after administrating gadolinium, in the short axis of the heart, in each cut (Figure 1). Mass was obtained by multiplying total fibrosis volume by 1.05 (myocardial density).
Figure 1.
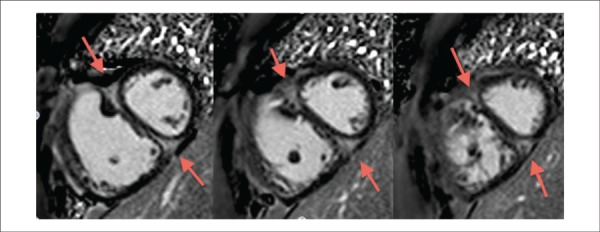
Cardiac magnetic resonance in short axis, two chambers (heart mid section), after injecting gadolinium to evaluate the delayed myocardial enhancement in a 26-year old patient with idiopathic pulmonary arterial hypertension at Clementino Fraga Filho University Hospital. It was observed a delayed myocardial enhancement in the anterior and posterior portions of the interventricular septum, on the right ventricular insertion points (red arrows).
Functional evaluation
Each patient was evaluated by CFFUH physiotherapy team with 6MWT performed in accordance with American Thoracic Society12 guidelines. In this test, the patient is requested to walk as fast as possible for 6 minutes, on a 30 m flat surface, and the distance is registered in meters. In this test, the patient may reduce the intensity, stop and rest, if necessary, without interrupting the timing process. Running or jogging was not allowed.
FC was evaluated according to World Health Organization (WHO). RVF clinical signs were defined as: jugular turgency, increased RV at palpation, hepatomegaly, ascite and edema on lower limbs.
Statistical analysis
Demographic, clinical, functional and hemodynamic variables of CMR and RC were tested in order to verify the type of distribution through histograms and Shapiro-Wilk test. Variables that showed normal distribution were expressed in mean ± standard or median deviation, with minimum and maximum values in case of abnormal distribution.
Presence and extension of DME were evaluated in patients with PAH subjected to CMR, and the percentage of myocardial fibrosis was calculated based on data regarding fibrosis mass and ventricular masses.
The relation between the percentage of myocardial fibrosis to clinical (signs of RVF), functional (CF and 6MWT) and hemodynamic parameters, through Mann-Whitney test, performing an attempt to identify "high risk" patients according to ROC curve, an optimal point (cutoff) of fibrosis percentage.
FC variable was categorized in FC IV ("high risk") and FC II/III ("low risk"). 6MWT variable was categorized in distance walked <300m ("high risk") and distance walked >300m ("low risk"). Hemodynamic variable was categorized in patients who had, at invasive hemodynamic study: RAP ≥ 15mmHg and CI < 2.0L/min.m2 ("high risk") and RAP < 15mmHg and CI ≥ 2.0L/min.m2 ("low risk").
Fifteen exams of CMR were randomized, then examined by the same radiologist and another radiologist with experience in cardiovascular medicine, in different occasions. Examiners were blinded in relation to clinical, functional and hemodynamical evaluation parameters. Intraclass Correlation Coefficient (ICC) was applied to evaluate the intra and interobserver reproducibility in DME research and calculation of myocardial fibrosis percentage.
Statistical analysis was processed by the program Statistical Package for Social Sciences (SPSS) 20.0 for MAC.
The criterion for significance determination was the level of 5%.
Results
Study population was composed of 30 patients, of which 44% had idiopathic PAH. The remaining patients were from Group I of Dana Point Classification, being composed of collagenosis (5 patients), portopulmonary hypertension (3 patients), schistosomiasis (1 patient) and HIV (1 patient). Other 7 patients have CTEPH.
In relation to demographic and functional data (Table 1), patients had a mean age of 52 years and most of them were female (23%). The median onset period for symptoms at diagnosis was 18 months. Among them, 53% had signs of RVF at clinical exam during the diagnosis and most of them had CF II or III (90%). The mean 6MWT was 395m among the patients.
Table 1.
Demographic and functional evaluation data
| Mean ± SD (%) | Median (min.-max.) | P25 | P75 | Shapiro (p) | |
|---|---|---|---|---|---|
| Age | 52.5 ± 16.5 | 45 | 62 | 0.246 | |
| Gender (M/F) | 7 (23%)/23 (77%) | ||||
| Weight (kg) | 65.5 ± 12.5 | 57 | 76 | 0.299 | |
| Height (meters) | 1.57 (1.49-1.82) | 1.54 | 1.67 | 0.001 | |
| BSA (m2/kg) | 1.61 (1.36-2.04) | 1.55 | 1.87 | 0.075 | |
| Time of symptoms onset (months) | 18 (6-36) | 12 | 24 | 0.074 | |
| Presence of RVF (Y/N) | 16 (53%)/14 (47%) | ||||
| FC WHO (II-III/IV) | 27 (90%)/3 (10%) | ||||
| 6MWT (meters) | 395.8 ± 109.8 | 350 | 465 | 0.434 |
Values stated in mean ± standard deviation (SD) or median, with minimum and maximum values with the respective percentiles 25 (P25) and 75% (P75), in Shapiro normality test. Min.: minimum; max.: maximum; M: male; F: female; BSA: body mass area; RVF: right ventricular failure; S: yes; N: no; FC WHO: Functional Class of the World Health Organization; 6MWT: 6-minute walk test
On Table 2 is shown the morphological and functional data obtained from CMR. It is noted that the mean diameter of RV was 76mm and of pulmonary artery was 38.5mm, and the mean mass index of RV was 46.4g/m2 - all parameters indicate increased resistance to blood flow and overload of RV. Pericardial effusion (PE) was detected in nine patients (30%). The mean ejection fraction of RV obtained in CMR was 31.8%, as well as the mean of LV CI of 2.1L/min.m2, which presupposes that most patients had right ventricular dysfunction at diagnosis.
Table 2.
Morphological and functional data, and information on mass/percentage of myocardial fibrosis by cardiac magnetic resonance
| Mean ± SD (%) | Median (min.-max.) | P25 | P75 | Shapiro (p) | |
|---|---|---|---|---|---|
| RV long diam (mm) | 76.4 ± 10.9 | 70 | 84 | 0.842 | |
| RV thickness (mm) | 6.0 (3.0-8.0) | 5.0 | 6.0 | 0.011 | |
| PA trunk diam (mm) | 38.5 (27-72) | 36 | 42 | 0.01 | |
| RV massi (g/m2) | 46.4 ± 10.0 | 36.8 | 51.3 | 0.208 | |
| LV massi (g/m2) | 56.5 (46.5-80.4) | 51.7 | 62.1 | 0.088 | |
| Pericardial effusion (Y/N) | 9 (30%)/27 (90%) | ||||
| CF (BPM) | 76.2 ± 12.4 | 68 | 86 | 0.669 | |
| RV EF (%) | 31.8 ± 12.2 | 24 | 38 | 0.609 | |
| RV EDVI (mL/m2) | 100.8 ± 26.7 | 80.2 | 122.7 | 0.676 | |
| RV ESVI (mL/m2) | 70.7 ± 26.9 | 51 | 88.9 | 0.343 | |
| CI CMR (L/min.m2) | 2.1 (1.4-3.4) | 1.7 | 2.4 | 0.089 | |
| LV EDVI (mL/m2) | 53.1 ± 12.8 | 43.4 | 60.2 | 0.731 | |
| LV ESVI (mL/m2) | 19.7 (8.9-49.4) | 15.6 | 32.1 | 0.037 | |
| LV SVI (mL/m2) | 28.2 (13.7-53.9) | 25.5 | 36.1 | 0.025 | |
| Presence of fibrosis (Y/N) | 28 (93%)/2 (7%) | ||||
| Fibrosis on anterior septum (Y/N) | 25 (83%)/5 (17%) | ||||
| Fibrosis on inferior septum (Y/N) | 28 (93%)/2 (7%) | ||||
| Fibrosis mass (g) | 9.9 (0-20.6) | 8.1 | 12.8 | 0.095 | |
| Percentage of myocardial fibrosis (%) | 6.17 (0-10.52) | 4.79 | 7.10 | 0.14 |
Values stated in mean ± standard deviation (SD) or median, with minimum and maximum values with the respective percentiles 25 (P25) and 75% (P75), in Shapiro normality test. Min.: minimum; max.: maximum; RV long diam: right ventricle longitudinal diameter; RV thickness: right ventricle thickness; PA trunk diam: pulmonary artery trunk diameter; RV mass: right ventricle mass index; LV massi: left ventricle mass index; Y: yes; N: no; CF (BPM): cardiac frequency (beats per minute); RV EF: right ventricle ejection fraction; RV EDVI: right ventricle end-diastolic volume index; RV ESVI: right ventricle end-systolic volume index; CI CMR: cardiac index by cardiac magnetic resonance; LV EDVI: left ventricle end-diastolic volume index; LV ESVI: left ventricle end-systolic volume index; LV SVI: left ventricle systolic volume index.
On Table 3 is the shown hemodynamic data obtained with RC. We noticed that mPAP was 53.3mmHg, PVR index (PVRI) of 22.2 Woods Units (WU) and the RAP median was of 13.5mmHg. The mean CI was 2.1L/min.m2, exactly the same as that obtained with CMR.
Table 3.
Hemodynamic data by right catheterism
| Mean ± SD (%) | Median (min.-max.) | P25 | P75 | Shapiro (p) | |
|---|---|---|---|---|---|
| CF R CAT (BPM) | 73.5 (53-108) | 70 | 80 | 0.016 | |
| mPAP (mmHg) | 53.3 ± 9 | 49 | 60 | 0.990 | |
| sPAP (mmHg) | 85.4 ± 18.8 | 75 | 96 | 0.745 | |
| dPAP (mmHg) | 32.4 ± 6.9 | 26 | 36 | 0.674 | |
| RAP (mmHg) | 13.5 (3-24) | 7 | 20 | 0.045 | |
| CI R CAT (L/min.m2) | 2.14 ± 0.56 | 1.6 | 2.5 | 0.308 | |
| PVRi (WU) | 22.2 ± 8.2 | 16.9 | 28.3 | 0.370 | |
| PCAP (mmHg) | 10.3 ± 3.6 | 8 | 14 | 0.467 | |
| SVM O2 (%) | 57.8 (42-77) | 48.6 | 68 | 0.042 | |
| GT (mmHg) | 43.2 ± 8.6 | 37 | 50 | 0.948 |
Values stated in mean ± standard deviation (SD) or median, with minimum and maximum values with the respective percentiles 25 (P25) and 75% (P75), in Shapiro normality test. Min.: minimum; max.: maximum; CF R CAT (BPM): cardiac frequency by right catheterism (beats per minute); mPAP: mean pulmonary artery pressure; sPAP: systolic pulmonary arterial pressure; dPAP: diastolic pulmonary arterial pressure; RAP: right atrium pressure; CI R CAT: cardiac index by right catheterism; PVRi: pulmonary vascular resistance index; WU: Wood unit; PCAP: pulmonary capillary pressure; MVO2S: mixed venous oxygen saturation; TG: transpulmonary gradient.
DME was present in 28 patients (93%), distributed in the anterior and inferior septal, and insertion areas of RV free-wall. The mean fibrosis mass was 9.9g and the myocardial fibrosis percentage was 6.17% (Table 2).
High risk factors, such as FC IV, RVF at diagnosis, 6MWT < 300 m, RAP ≥ 15 mmHg and CI < 2.0 L/min.m2 had significant association with higher fibrosis percentage (Table 4).
Table 4.
Distribution of patients, according to risk factors, the clinical, functional and hemodynamical evaluations, and percentage of myocardial fibrosis
| Risk factors - percentage of myocardial fibrosis | p | ||
|---|---|---|---|
| RVF | None (n = 13) | Present (n = 17) | |
| Percentage of myocardial fibrosis | 4.30 (3.93-5.55) | 6.83 (6.28-7.19) | < 0.0001 |
| FV IV | None (n = 27) | Present (n = 3) | |
| Percentage of myocardial fibrosis | 5.54 (4.78-6.67) | 7.51 (7.18-7.67) | 0.034 |
| TC6M < 300m | None (n = 24) | Present (n = 6) | |
| Percentage of myocardial fibrosis | 5.38 (4.62-6.78) | 7.18 (6.58-8.16) | 0.021 |
| Hemodynamic (CI < 2.0L/min.m2 and RAP > 15mmHg) | None (n = 17) | Present (n = 13) | |
| Percentage of myocardial fibrosis | 5.12 (4.49-6.28) | 6.55 (6.26-7.18) | 0.039 |
Values of the percentage of myocardial fibrosis stated in mean, with the respective percentiles 25 and 75 in parenthesis. RVF: right ventricular failure; FC: functional class; 6MWT: 6-minute walk test; CI: cardiac index; RAP: right atrium pressure.
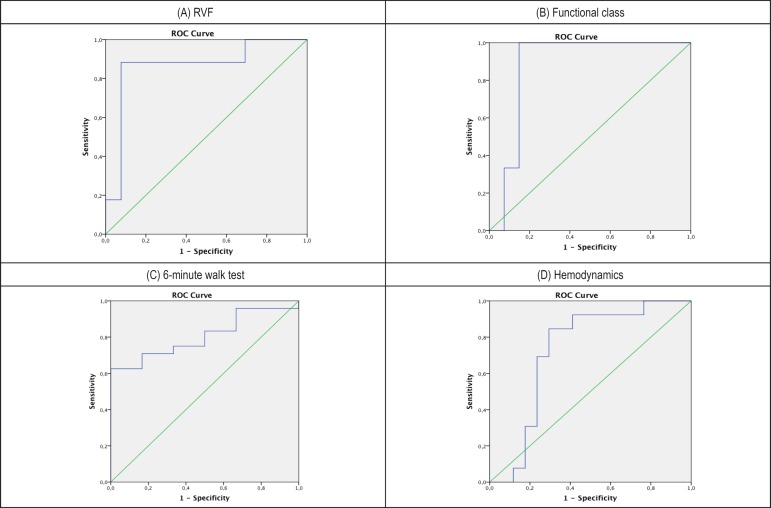
Based on ROC curve analysis (Graph 1), it was observed that a percentage of myocardial fibrosis > 5.99% is capable of identifying patients with RVF (i.e., high risk) with 82% Sensitivity (S), 92% Specificity (E), 99% Positive Predictive Value (PPV) and 28% Negative Predictive Value (NPV).
Graph 1.
ROC curve between clinical and hemodynamical severity stratifiers for patients with pulmonary arterial hypertension and percentage of myocardial fibrosis obtained by cardiac magnetic resonance. A) right ventricular failure (RVD): area under a curve (AUC) of 0.864 (confidence interval of 95% - CI95% = 0.714 - 1.0). The percentage of myocardial fibrosis > 5.99% was capable to identify the patient without signs of RVF (high risk) with 85% sensitivity (S), 92% specificity (E), 99% positive predictive value (PPV) and 28% negative predictive value (NPV). B) Functional class: 0.877 (AUC) (CI95% = 0.753-1.0). The percentage of myocardial fibrosis > 6.67% was capable to identify patients with functional class (FC) IV (high risk) with 98% S, 75% E, 98% PPV and 73% NPV. C) 6-minute walk test (6MWT): 0.806 AUC (CI95% = 0.647-0.965). The percentage of myocardial fibrosis > 6.49% was capable to identify patients who walk < 300 meters (high risk), with 71% S, 83% E, 98% PPV and 18% NPV. D) Hemodynamics: 0.724 AUC (CI95% = 0.530-0.918). The percentage of myocardial fibrosis > 5.99% was capable to identify patients with cardiac index (CI) < 2.0L/min.m2 and right atrium pressure (RAP) ≥ 15mmHg (high risk) with 84% S, 71% E, 97% PPV and 18% NPV.
A percentage of myocardial fibrosis > 6.67% is capable of identifying the patient with a more advanced FC (FC IV, i.e., high risk) with 98% S, 75% E, 98% PPV and 73% NPV.
A percentage of myocardial fibrosis > 6.49% is capable of identifying patients with a functional capacity of exercise at 6MWT < 300m (i.e., high risk), with 71% S, 83% E, 98% PPV and 18% NPV.
Percentage of myocardial fibrosis > 5.99% is capable of identifying patients with PAD ≥ 15mmHg and IC < 2.0 L / min.m2 on RC (i.e., high risk), with 84% S, 81% E, 97% PPV and 18% NPV.
Intraobserver ICC for myocardial fibrosis mass was 0.984 (CI95% = 0.953-0.995) and interobserver of 0.966 (CI95% = 0.900-0.989).
Discussion
Right ventricular dysfunction is the main cause of morbimortality in patients with PAH4. Several studies are developed to find clinical and hemodynamical markers that could be used in diagnosis and follow-up. Thus, it is possible to characterize a high risk patient, who will be subjected to early therapeutic interventions.
CMR is the gold standard for evaluating function, volume and mass of the right ventricle13, which can be qualified with excellent intra and interobserver14 variability and good interstudy reproducibility15.
DME was initially described in dysfunctioning areas of LV after gadolinium-based contrast agent administration in patients undergoing CMR with previous infarction history and fibrosis scarring16,17. Descriptions a posteriori have shown the presence of DME in patients with hypertrophic myocardiopathy and myocardiopathies with deposits or inflammatory (viral myocarditis), each one with a characteristic extension pattern18-22.
DME can be explained by the gadolinium capacity of having a different pattern distribution in healthy and unhealthy tissues. Contrast "removal" is more difficult in the unhealthy tissue, creating a signal difference > 1.080% in DME images. Consequently, areas of the unhealthy myocardial tissue can be precisely identified, with high accuracy and reproducibility23,24.
Even though there are several studies of DME evaluated by CMR in conditions that affect the LV, only three of major importance evaluated this finding in patients with PAH11,25,26.
In McCann et al25 study, the presence of DMC was noticed in insertion areas of the RV interventricular septum in all 15 patients with PAH. The mean fibrosis mass was 3.1 ± 1.9g and there was no positive correlation with any clinical or hemodynamical variable - only an inverse correlation with right ventricular dysfunction measured by CMR. Blyth et al11 noticed the presence of DME on 23 of 25 study patients, of 3.3g and there was a positive correlation with several hemodynamic variables, primarily patients with paradoxical interventricular septum deviation. Junqueira et al26 demonstrated the presence of DME in 65% of study patients and only found a positive correlation with the duration of the disease at diagnosis.
Several speculations exist on the physiopathological mechanism related to DME in patients with PAH. In MacCann et al25 study, two patients who died were subjected to autopsy, and fibrosis was present in the right ventricular insertion point, suggesting that this was the DME mechanism selected for these patients. It is speculated that myocardial fibrosis results from an increased right ventricle afterload. The hypertrophy and right cavity dilatation found in these patients led to a mechanical stress in these points, with subsequent hypoxia and ischemia27,28.
In this study, DME was present in 28 of the 30 study patients. The median fibrosis mass was 9.9g (0-20.2g). Due to a large variation of the ventricular mass related to age, gender, body mass index and comorbities very prevalent in our environment, such as systemic arterial hypertension, we chose to calculate the percentage of myocardial fibrosis, which mean was 6.17%.
Apart from other studies, the percentage of myocardial fibrosis has been able to identify patients with PH with high risk factors, based on clinical (signs of RVF), functional (FC IV and 6MWT < 300m) or hemodynamic (RAP ≥ 15mmHg and CI < 2.0L/min.m2) evaluation.
This study had a few limitations. It was carried out with a relatively small number of patients, which, particularly, is reflected by the low prevalence of PAH in the general population. No patient was subjected to cine angiocoronariography to discard an ischemic heart disease, which is an important cause of DME. However, the DME pattern noticed in these patients is not a characteristic of myocardial ischemic injury.
Conclusion
DME is present in most patients with PAH, and the percentage of myocardial fibrosis evaluated by CMR has shown a good non-invasive parameter for severity evaluation in this group with 30 study patients.
These results are interesting, since the greatest clinical challenge, in PH evaluation, is to identify patients who need early interventions, due to the potential for high severity of the disease. Longitudinal studies and with larger number of patients need to be performed in order to assess the real prognostic role of this evaluation parameter for patients with PAH.
Footnotes
Author contributions
Conception and design of the research: Bessa LGP, Junqueira FP, Garcia MI, Xavier SS, Waetge D; Acquisition of data: Bessa LGP, Junqueira FP, Bandeira MLS, Lavall G, Torres D, Waetge D; Analysis and interpretation of the data: Bessa LGP, Bandeira MLS, Garcia MI, Xavier SS; Statistical analysis and Writing of the manuscript: Bessa LGP; Critical revision of the manuscript for intellectual content: Bessa LGP, Junqueira FP, Bandeira MLS, Garcia MI, Xavier SS, Lavall G, Torres D.
Potential Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Sources of Funding
There were no external funding sources for this study.
Study Association
This article is part of the thesis of master submitted by Luiz Gustavo Pignataro Bessa, from Universidade Federal do Rio de Janeiro.
References
- 1.Simonneau G, Galie N, Rubin LJ, Langleben D, Seeger W, Domenighetti G, et al. Clinical classification of pulmonary hypertension. J Am Coll Cardiol. 2004;43(12) Suppl S:5S–12S. doi: 10.1016/j.jacc.2004.02.037. [DOI] [PubMed] [Google Scholar]
- 2.McGoon M, Gutterman D, Steen V, Barst R, McCrory DC, Fortin TA, et al. American College of Chest Physicians Screening, early detection, and diagnosis of pulmonary arterial hypertension: ACCP evidence based clinical practice guidelines. Chest. 2004;126(1) Suppl:14S–34S. doi: 10.1378/chest.126.1_suppl.14S. [DOI] [PubMed] [Google Scholar]
- 3.Thenappan T, Shah SJ, Rich S, Gomberg-Maitland M. A USA-based registry for pulmonary arterial hypertension: 1982-2006. Eur Respir J. 2007;30(6):1103–1110. doi: 10.1183/09031936.00042107. [DOI] [PubMed] [Google Scholar]
- 4.Voelkel NF, Quaife RA, Leinwand LA, Barst RJ, McGoon MD, Meldrum DR, National Heart, et al. Lung and Blood Institute Working Group on Cellular and Molecular Mechanisms of Right Heart Failure Right ventricular function and failure: report of a National Heart, Lung, and Blood Institute working group on cellular and molecular mechanisms of right heart failure. Circulation. 2006;114(17):1883–1891. doi: 10.1161/CIRCULATIONAHA.106.632208. [DOI] [PubMed] [Google Scholar]
- 5.McLaughlin VV, Presberg KW, Doyle RL, Abman SH, McCrory DC, Fortin T, et al. American College of Chest Physicians Prognosis of pulmonary arterial hypertension: ACCP evidence-based clinical practice guidelines. Chest. 2004;126(1) Suppl:78S–92S. doi: 10.1378/chest.126.1_suppl.78S. [DOI] [PubMed] [Google Scholar]
- 6.D'Alonzo GE, Barst RJ, Ayres SM, Bergofsky EH, Brundage BH, Detre KM. Survival in patients with primary pulmonary hypertension: results from a national prospective registry. Ann Intern Med. 1991;115(5):343–349. doi: 10.7326/0003-4819-115-5-343. [DOI] [PubMed] [Google Scholar]
- 7.Barst RJ, Rubin LJ, Long WA, McGoon MD, Rich S, Badesch DB, et al. Primary Pulmonary Hypertension Study Group A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension. N Engl Med. 1996;334(5):296–301. doi: 10.1056/NEJM199602013340504. [DOI] [PubMed] [Google Scholar]
- 8.Eysmann SB, Palevsky HI, Reicken N, Hackney K, Douglas PS. Two-dimensional and Doppler-echocardiographic and cardiac catheterization correlates of survival in primary pulmonary hypertension. Circulation. 1989;80(2):353–360. doi: 10.1161/01.cir.80.2.353. [DOI] [PubMed] [Google Scholar]
- 9.Sitbon O, Humbert M, Nunes H, Parent F, Garcia G, Hervé P, et al. Long-term intravenous epoprostenol infusion in primary pulmonary hypertension: prognostic factors and survival. J Am Coll Cardiol. 2002;40(4):780–788. doi: 10.1016/s0735-1097(02)02012-0. [DOI] [PubMed] [Google Scholar]
- 10.Van Wolferen SA, Marcus JT, Boonstra A, Marques KM, Bronzwaer JG, Spreeuwenberg MD, et al. Prognostic value of right ventricular mass, volume, and function in idiopathic pulmonary arterial hypertension. Eur Heart J. 2007;28(10):1250–1257. doi: 10.1093/eurheartj/ehl477. [DOI] [PubMed] [Google Scholar]
- 11.Blyth KG, Groenning BA, Martin TN, Foster JE, Mark PB, Dargie HJ, et al. Contrast enhanced-cardiovascular magnetic resonance imaging in patients with pulmonary hypertension. Eur Heart J. 2005;26(19):1993–1999. doi: 10.1093/eurheartj/ehi328. [DOI] [PubMed] [Google Scholar]
- 12.ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories ATS statement: guidelines for the six- minute walk test. Am J Respir Crit Care Med. 2002;166(1):111–117. doi: 10.1164/ajrccm.166.1.at1102. [DOI] [PubMed] [Google Scholar]
- 13.Pennel DJ, Sechtem UP, Higgins CB, Manning WJ, Pohost GM, Rademakers FE, et al. Clinical indications for cardiovascular magnetic resonance (CMR): consensus panel report. Eur Heart J. 2004;25(21):1940–1965. doi: 10.1016/j.ehj.2004.06.040. [DOI] [PubMed] [Google Scholar]
- 14.Alfakih K, Plein S, Thiele H, Jones T, Ridgway JP, Sivananthan MU. Normal human left and right ventricular dimensions for MRI as assessed by turbo gradient echo and steady state free precession imaging sequences. J Magn Reson Imaging. 2003;17(3):323–329. doi: 10.1002/jmri.10262. [DOI] [PubMed] [Google Scholar]
- 15.Grothues F, Moon JC, Bellenger NG, Smith GS, Klein HU, Pennel DJ. Interstudy reproducibility of right ventricular volumes, function, and mass with cardiovascular magnetic resonance. AM Heart J. 2004;147(2):218–223. doi: 10.1016/j.ahj.2003.10.005. [DOI] [PubMed] [Google Scholar]
- 16.Kim RJ, Fieno DS, Parrish TB, Harris K, Chen EL, Simonetti O, et al. Relationship of MRI delayed contrast enhancement to irreversible injury, infart age and contractile function. Circulation. 1999;100(19):1992–2002. doi: 10.1161/01.cir.100.19.1992. [DOI] [PubMed] [Google Scholar]
- 17.Kim RJ, Wu E, Rafael A, Chen EL, Parker MA, Simonetti O, et al. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial disfunction. N Engl J Med. 2000;343(20):1445–1453. doi: 10.1056/NEJM200011163432003. [DOI] [PubMed] [Google Scholar]
- 18.Kim RJ, Judd RM. Gadolinium-enhanced magnetic resonance imaging in hypertrophic cardiomyopathy: in vivo imaging of the pathologic substrate for premature cardiac death? J Am Coll Cardiol. 2003;41(9):1568–1572. doi: 10.1016/s0735-1097(03)00190-6. [DOI] [PubMed] [Google Scholar]
- 19.McCrohon JA, Moon JC, Prasad SK, McKenna WJ, Lorenz CH, Coats AJ, et al. Differentiation of heart failure related to dilated cardiomyopathy and coronary artery disease using gadolinium-enhanced cardiovascular magnetic resonance. Circulation. 2003;108(1):54–59. doi: 10.1161/01.CIR.0000078641.19365.4C. [DOI] [PubMed] [Google Scholar]
- 20.Moon JC, McKenna WJ, McCrohon JA, Elliott PM, Smith GC, Pennell DJ. Toward clinical risk assessment in hypertrophic cardiomyopathy with gadolinium cardiovacular magnetic resonance. J Am Coll Cardiol. 2003;41(9):1561–1567. doi: 10.1016/s0735-1097(03)00189-x. [DOI] [PubMed] [Google Scholar]
- 21.Moon JC, Sachdev B, Elkington AG, McKenna WJ, Mehta A, Pennell DJ, et al. Gadolinium enhanced cardiovascular magnetic resonance in Anderson-Fabry disease: evidence for a disease specific abnormality of the myocardial interstitium. Eur Heart J. 2003;24(23):2151–2155. doi: 10.1016/j.ehj.2003.09.017. [DOI] [PubMed] [Google Scholar]
- 22.van Dockum WG, ten Cate FJ, ten Berg JM, Beek AM, Twisk JWR, Vos J, et al. Myocardial infarction after percutaneous transluminal septal myocardial ablation in hypertrophic obstructive cardiomyopathy: evaluation by contrast-enhanced magnetic resonance imaging. J Am Coll Cardiol. 2004;43(1):27–34. doi: 10.1016/j.jacc.2003.08.031. [DOI] [PubMed] [Google Scholar]
- 23.Wagner A, Mahrholdt H, Holly TA, Elliot MD, Regenfus M, Parker M, et al. Contrast-enhanced MRI and routine single photon emission computed tomography (SPECT) perfusion imaging for detection of subendocardial myocardial infarcts: an imaging study. Lancet. 2003;361(9355):374–379. doi: 10.1016/S0140-6736(03)12389-6. [DOI] [PubMed] [Google Scholar]
- 24.Azevedo CF, Filho, Hadlich M, Petriz JL, Mendonca LA, Moll JN, Filho, Rochitte CE. Quantification of left ventricular infarcted mass on cardiac magnetic resonance imaging: comparison between planimetry and the semiquantitative visual scoring method. Arq Bras Cardiol. 2004;83(2):118-24, 111-17. doi: 10.1590/s0066-782x2004001400003. [DOI] [PubMed] [Google Scholar]
- 25.McCann GP, Gan CT, Beek AM, Niessen HW, Vonk Noordegraaf A, van Rossum AC. Extent of MRI delayed enhancement of myocardial mass is related to right ventricular dysfunction in pulmonary artery hypertension. AJR Am J Roentgenol. 2007;188(2):349–355. doi: 10.2214/AJR.05.1259. [DOI] [PubMed] [Google Scholar]
- 26.Junqueira FP, Macedo R, Coutinho AC, Loureiro R, De Pontes PV, Domingues RC, et al. Myocardial delayed enhancement in patients with pulmonary hypertension and right ventricular failure: evaluation by cardiac MRI. Br J Radiol. 2009;82(982):821–826. doi: 10.1259/bjr/28241773. [DOI] [PubMed] [Google Scholar]
- 27.Marcus JT, Vonk Noordegraaf A, Roeleveld RJ, Postmus PE, Heethaar RM, Van Rossum AC, et al. Impaired left ventricular filling due to right ventricular pressure overload in primary pulmonary hypertension: noninvasive monitoring using MRI. Chest. 2001;119(6):1761–1765. doi: 10.1378/chest.119.6.1761. [DOI] [PubMed] [Google Scholar]
- 28.Vonk Noordegraaf A, Gan T, Marcus J, Boonstra A, Postmus P. Interventricular mechanical asynchrony is an important cause of cardiac dysfunction in pulmonary hypertension. Eur Respir Soc. 2004;24(Suppl. 48):S203–S203. [Google Scholar]