Abstract
Background
There is controversy regarding whether the use of selective serotonin reuptake inhibitors (SSRIs) and other antidepressants in pregnancy is associated with increased risks for congenital cardiac defects. In particular, concerns exist about a possible association between paroxetine and right ventricular outflow tract obstruction (RVOTO), and between sertraline and ventricular septal defects (VSD).
Methods
We performed a cohort study nested in the 2000–2007 nationwide Medicaid Analytic eXtract. The study included 949,504 pregnant women enrolled in Medicaid from three months before conception through one month post delivery, and their live-born infants. We compared the risk of major cardiac defects in women with antidepressant medication use during the first trimester versus no use, restricting the cohort to women with depression and using propensity score adjustment to control for depression severity and other potential confounders.
Results
64,389 women (6.8%) used antidepressants during the first trimester. Overall, 6,403 infants not exposed to antidepressants were born with a cardiac defect (72.3 per 10,000), compared with 580 infants exposed (90.1 per 10,000). Associations between antidepressant use and cardiac defects were attenuated with increasing levels of adjustment for confounding. For SSRIs, relative risks for any cardiac defect were 1.25 (95%CI, 1.13–1.38) unadjusted, 1.12 (1.00–1.26) depression-restricted, and 1.06 (0.93–1.22) depression-restricted and fully-adjusted. We found no significant associations between the use of paroxetine and RVOTO (1.07, 0.59–1.93), or the use of sertraline and VSD (1.04, 0.76–1.41).
Conclusions
Results of this large population-based cohort study suggest no substantial increased risk of cardiac malformations attributable to SSRIs.
Clinical depression occurs in 10–15% of pregnant women.1 The use of antidepressant medications in pregnancy has increased steadily over time, with reported prevalences up to 8–13% in the US.2–4 Selective serotonin reuptake inhibitors (SSRIs) are the most commonly prescribed antidepressants during pregnancy.4 In 2005, based on early results of two epidemiologic studies, the US Food and Drug Administration (FDA) warned healthcare professionals that early prenatal exposure to paroxetine may increase the risk of congenital cardiac malformations and reclassified it to pregnancy category D.5 Most malformations in the early reports leading to the FDA warning were septal defects. Since then, several studies have evaluated the teratogenicity of SSRIs and other antidepressants6–19, but considerable controversy remains as to whether this is a “serious concern or much ado about little” as noted in an editorial published with two of the reports.13,14,20
Existing studies have reported different associations, often in the context of multiple comparisons. Yet, at least two studies reported a 2–3 fold increased risk of right ventricular outflow tract obstruction defects associated with paroxetine use13,14, and of ventricular septal defects associated with sertraline use.13,19 A meta-analysis estimated a 50% increased prevalence of cardiac defects overall with first trimester paroxetine use.21 It has remained unclear, however, whether these associations are causal, or due to systematic error or chance.
We conducted a study using a large national cohort of publicly-insured pregnant women in the US to assess the risk of congenital cardiac defects following use of specific antidepressants, with attention to the potential for confounding by the underlying depression and associated factors.
METHODS
Data Source and Study Cohort
The study cohort was drawn from the Medicaid Analytic eXtract (MAX) for 46 US states and Washington, DC for 2000–2007. Montana and Connecticut were excluded because of difficulty in linking mothers and infants, Michigan because of incomplete data, and data from Arizona were not available. The MAX dataset contains individual-level demographic and Medicaid enrollment information, as well as all physician services and hospitalizations and their accompanying diagnoses and procedures, and all filled outpatient medication prescriptions.
The development of our study cohort has previously been described.22 Briefly, we identified all completed pregnancies in women aged 12 to 55 years and linked these pregnancies to live-born infants. We estimated the date of last menstrual period (LMP) based on the delivery date combined with diagnostic codes indicative of pre-term delivery using a validated algorithm.23 Finally, we required all women to be Medicaid eligible, without supplementary private insurance or restricted benefits, from three months before the LMP through one month post delivery. We excluded pregnancies in which the baby had been diagnosed with a chromosomal abnormality (N=1,609) and pregnancies in which the mother had been treated with known teratogens during the first trimester (i.e., lithium, antineoplastic agents, retinoids, and thalidomide) (N=2,476).
Antidepressant Medications
The etiologically relevant window for exposure extended from the LMP through day 90 of pregnancy (first trimester). We determined maternal use of antidepressants through pharmacy dispensing records, using the dispensing date and number of days supply. Women were considered exposed if days supply overlapped with the first trimester. We defined the following exposure categories: all SSRIs; paroxetine; sertraline; fluoxetine; tricyclic antidepressants (TCAs); serotonin-norepinephrine reuptake inhibitors (SNRIs); bupropion; and other antidepressants (Table S1). The reference group consisted of women without exposure to antidepressants during the first trimester.
Cardiac Malformations
Congenital cardiac malformations were defined based on the presence of in- or outpatient ICD-9 diagnostic codes in the maternal or infant records during the first 90 days post delivery. We used both maternal and infant codes because an infant’s claims may be recorded under the mother’s identification number for the first several months after birth.24 Informed by the previously reported associations, outcomes were grouped as: (i) any cardiac malformation, (ii) right ventricular outflow tract obstruction (RVOTO), (iii) ventricular septal defect (VSD), and (iv) other cardiac malformation (Table S2). We excluded prematurity related anomalies (e.g., patent ductus arteriosus, pulmonary valve stenosis and anomalies of pulmonary artery in preterm infants).25 A given outcome was considered present if (i) there was >1 date with the respective diagnostic codes recorded, or (ii) there was one diagnostic code and a code for a cardiac procedure or surgery (Table S3). The positive predictive values for these outcome definitions were non-differential by exposure, and ranged from 66.7% to 79.5% in a conservative validation study based on primary medical record review in a subset of cases.26
Covariates
Information on covariates considered for confounding adjustment or stratification was obtained during the baseline period (from pre-LMP through the end of the first trimester). In addition to socio-demographic information (year of delivery, state, age, race, parity), we considered known or suspected risk factors for congenital malformations and proxies for such risk factors: multiple gestation, chronic maternal illness (hypertension, diabetes, epilepsy, renal disease), suspected teratogenic medications, other psychotropic medication use (anticonvulsants, antipsychotics, anxiolytics, hypnotics, other benzodiazepines, barbiturates), antidiabetic and antihypertensive medications, and number of distinct prescription drugs used, excluding antidepressants, as a general marker of comorbidity27. To address confounding by the underlying indication, we considered proxies for depression severity (number of outpatient and inpatient depression diagnoses), and other indications for antidepressant use (other mental health disorders, pain-related diagnoses, sleep disorders, premenstrual tension syndrome, smoking, chronic fatigue syndrome).
Data Analyses
We compared the distributions of socio-demographic, clinical and healthcare utilization characteristics for the different exposure groups, and calculated absolute risks of cardiac malformations. Logistic regression analysis was used to estimate odds ratios for cardiac malformations and their corresponding 95% confidence intervals (CI). Because the odds ratio is an excellent approximation of the risk ratio in the case of rare outcomes, the results are referred to as relative risks.28 Use of the robust variance estimator to account for correlations within women with multiple pregnancies did not appreciably change the CI so correlation structures were omitted from all analyses. Results are presented for three levels of adjustment: (i) unadjusted, (ii) restricted to women with a depression diagnosis to control for the potential effect of the underlying illness or factors associated with it, (iii) restricted to women with a depression diagnosis, using propensity score stratification to further control for proxies of depression severity and other potential confounders.29 Propensity scores were derived from the predicted probability of treatment estimated in a logistic regression model which contained all covariates listed above without additional variable selection. We created 100 equally-sized propensity score strata, dropped uninformative strata (i.e., those that did not contain at least one treated and one untreated woman), and stratified the outcome models on these propensity score strata. In all cases, <0.5% of treated and <0.1% of untreated women were dropped. In confirmatory analyses, we used a high-dimensional propensity score algorithm, which evaluates thousands of diagnoses, procedures, and pharmacy claim codes to identify and prioritize those covariates that serve as proxies for unmeasured confounders. These empirically identified confounders (n=200) were combined with investigator-identified covariates to improve adjustment for confounding.30 No adjustments were made for multiple comparisons.
Subgroup and Sensitivity analyses
We performed pre-specified subgroup and sensitivity analyses to evaluate the robustness of the primary results (for any cardiac malformation). Because the cohort studied is younger and more racially diverse than cohorts from previous studies13,14, we tested for effect modification by age (<30 vs. ≥ 30) and race/ethnicity (Caucasian vs. non-Caucasian). We conducted dose-response analyses for low, medium and high dose antidepressants using the first and highest dose dispensed (Table S4).31 For SSRIs, we stratified the analysis for use of just that drug class versus use of multiple antidepressant classes. To evaluate the effect of potential exposure misclassification, we redefined exposure status as having filled one or more prescriptions during the first trimester (as opposed to days supply which overlaps with the first trimester); we redefined the reference group as women with no antidepressant exposure throughout pregnancy. To evaluate the effect of potential outcome misclassification, we restricted the outcome to inpatient diagnoses only, and extended infant follow-up to 1-year. We corrected odds ratios for outcome misclassification using sensitivities and specificities consistent with the positive predictive values estimated in the internal validation study.26, 32 To further assess whether outcomes were well captured, we evaluated some well-known associations in our dataset; in particular, between cardiac malformations and maternal diabetes, anticonvulsant use, and multifetal pregnancy.33
RESULTS
We identified 949,504 eligible pregnancies. During the first trimester, 64,389 women (6.8%) used an antidepressant: 46,144 (4.9%) were exposed to an SSRI, 5,954 (0.6%) to a TCA, 6,904 (0.7%) to an SNRI, 8,856 (0.9%) to bupropion, and 7,055 (0.7%) to other antidepressants. Among the SSRIs, sertraline was used most frequently (n=14,040), followed by paroxetine (n=11,126), and fluoxetine (n=11,048).
Compared to women who took no antidepressant, women who filled a prescription for an antidepressant were older, had greater health care utilization, and were more likely to be white, to use other psychotropic medications, to have a chronic illness (in particular hypertension and diabetes), and to use suspected teratogenic medications (Table 1). Baseline characteristics were more homogeneous among users of different antidepressant classes (Table S5–S13).
Table 1.
Selected cohort characteristics for women exposed to SSRIs during the first trimester and women not exposed to antidepressants; overall unadjusted and depression restricted adjusted. Medicaid Analytic eXtract, 2000–2007.
| Characteristic | Overall cohort, unadjusted | Depression restricted cohort, Accounting for PS strata(1) | ||
|---|---|---|---|---|
| SSRI | Unexposed | SSRI | Unexposed | |
| N | 46,144 | 885,115 | 36,778 | 180,564 |
| Age, mean (SD) | 25.6 (5.9) | 23.9 (5.8) | 25.5 (6.0) | 25.3 (53.1) |
| Race, N (%) | ||||
| White | 34,098 (73. 9) | 339,144 (38.3) | 27,299 (74.2) | 136,506 (75.6) |
| Black | 5,438 (11. 8) | 313,369 (35.4) | 4,193 (11.4) | 19,380 (10.7) |
| Hispanic | 4,145 (9.0) | 164,317 (18. 6) | 3,261 (8.9) | 15,017 (8.3) |
| Other or unknown | 2,463 (5.3) | 68,285 (7.7) | 2,025 (5.5) | 9,661 (5.4) |
| Preterm birth(2), N (%) | 6,470 (14.0) | 98,886 (11.2) | 5,250 (14.3) | 25,299 (14.0) |
| Diabetes | 1,288 (2.8) | 10,628 (1.2) | 997 (2.7) | 4,957 (2.8) |
| Antidiabetics | 1,682 (3.7) | 15,364 (1.7) | 1,303 (3.5) | 6,449 (3.6) |
| Depression | 36,783 (79.7) | 180,564 (20.4) | 36,778 (100.0) | 180,564 (100.0) |
| Proxies for depression severity, mean (SD) | ||||
| Number of outpatient depression diagnoses | 2.8 (6.5) | 0.2 (2.0) | 3.5 (7.1) | 3.3 (20.3) |
| Number of inpatient depression diagnoses | 0.1 (0.3) | 0.0 (0.1) | 0.1 (0.3) | 0.1 (1.0) |
| Other psychotropic medications, N (%) | ||||
| Anticonvulsants | 7,353 (15.9) | 31,681 (3.6) | 6,654 (18.1) | 33,599 (18.6) |
| Antipsychotics | 9,534 (20.7) | 48,657 (5.5) | 8,621 (23.4) | 42,987 (23.8) |
| Anxiolytics | 3,148 (6.8) | 8,189 (0.9) | 2,895 (7.9) | 14,320 (7.9) |
| Benzodiazepines | 14,560 (31.6) | 49,063 (5.5) | 12,856 (35.0) | 63,100 (35.0) |
| Other hypnotics | 13,277 (28.8) | 115,608 (13.1) | 11,540 (31.4) | 56,323 (31.2) |
| Barbiturates | 3,764 (8.2) | 26,030 (2.9) | 3,097 (8.4) | 15,756 (8.7) |
| Suspected teratogens(3) | 3,508 (7.6) | 26,967 (3.1) | 2,806 (7.6) | 14,345 (7.9) |
To account for PS, the untreated observations were weighted using the distribution of the treated among PS strata
Relates to current pregnancy
Pregnancies with exposure to known teratogens were removed from the cohort. Suspected teratogens include: angiotensin-converting-enzyme inhibitors, fluconazole, aminoglycosides, folic acid antagonists, methimazole, potassium iodide, tetracycline, danazol, misoprostol, statins, coumadin, and propylthiouracil.
Overall, cardiac malformations were diagnosed in 72.3 per 10,000 infants not exposed to antidepressants during the first trimester, compared with 90.1 per 10,000 infants exposed. This higher unadjusted risk among exposed infants was observed for each of the specific types of malformations considered (Table 2).
Table 2.
Absolute risk for congenital cardiac malformations in women with and without antidepressant exposure by class; overall cohort. Medicaid Analytic eXtract, 2000–2007.
| Exposure groups | Total | Any cardiac malformation | RVOTO | VSD | Other cardiac malformation | ||||
|---|---|---|---|---|---|---|---|---|---|
| Events | Risk (/10,000) | Events | Risk (/10,000) | Events | Risk (/10,000) | Events | Risk (/10,000) | ||
| Unexposed | 885,115 | 6,403 | 72.3 | 1,045 | 11.8 | 3,212 | 36.3 | 3,232 | 36.5 |
| All antidepressants | 64,389 | 580 | 90.1 | 84 | 13.1 | 286 | 44.4 | 318 | 49.4 |
| SSRI | 46,144 | 416 | 90.2 | 61 | 13.2 | 201 | 43.6 | 226 | 49.0 |
| Paroxetine | 11,126 | 93 | 83.6 | 16 | 14.4 | 44 | 39.6 | 48 | 43.1 |
| Sertraline | 14,040 | 129 | 91.9 | 17 | 12.1 | 63 | 44.9 | 71 | 50.6 |
| Fluoxetine | 11,048 | 99 | 89.6 | 16 | 14.5 | 48 | 43.5 | 55 | 49.8 |
| TCA | 5,954 | 42 | 70.5 | 8 | 13.4 | 24 | 40.3 | 18 | 30.2 |
| SNRI | 6,904 | 75 | 108.6 | 12 | 17.4 | 39 | 56.5 | 38 | 55.0 |
| Other | 7,055 | 74 | 104.9 | 8 | 11.3 | 31 | 43.9 | 46 | 65.2 |
| Bupropion | 8,856 | 76 | 85.8 | 11 | 12.4 | 39 | 44.0 | 49 | 55.3 |
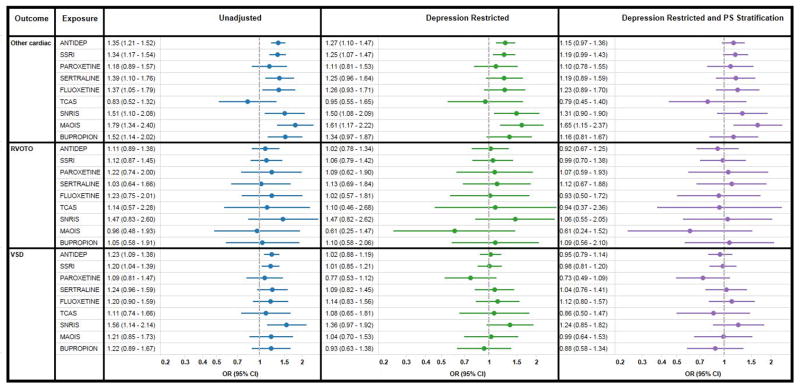
In unadjusted analyses, the relative risk (RR) for any cardiac malformation was 1.25 (95% CI 1.13–1.38) for SSRIs, 0.98 (0.72–1.32) for TCAs, 1.51 (1.20–1.90) for SNRIs, 1.46 (1.16–1.83) for other antidepressants, and 1.19 (0.95–1.49) for bupropion (Figure 1). Numerically increased unadjusted risks were observed for all three subtypes of cardiac malformations in most exposure groups (Figure 2).
Figure 1.
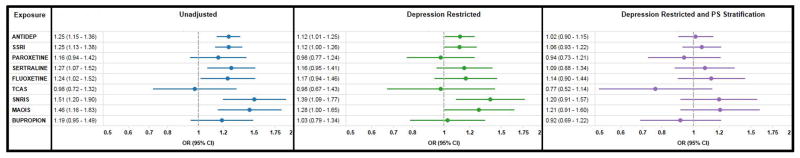
Relative risks and 95% confidence intervals comparing the risk of any cardiac malformation for women with and without antidepressant exposure during the first trimester; unadjusted, restricted to women with depression, and using PS stratification for confounding adjustment. Medicaid Analytic eXtract 2000–2007.
Figure 2.
Relative risks and 95% confidence intervals comparing the risk of specific cardiac malformations for women with and without antidepressant exposure during the first trimester; unadjusted, restricted to women with depression, and using PS stratification for confounding adjustment. Medicaid Analytic eXtract 2000–2007.
Restricting the cohort to women with a depression diagnosis markedly attenuated the associations (Figures 1 and 2). The c-statistic for the propensity score models ranged from 0.84 to 0.91, indicating that substantial differences in patient characteristics remained after restricting the cohort to women with depression. Stratification by propensity score ensured comparisons were made between groups with nearly identical characteristics (Table 1, Table S5–S13). Adjustment for the propensity score further attenuated the remaining positive associations. The adjusted RR for any cardiac malformation was 1.06 (0.93–1.22) for women exposed to SSRIs, 0.77 (0.52–1.14) for TCAs, 1.20 (0.91–1.57) for SNRIs, 1.21 (0.91–1.60) for other antidepressants, and 0.92 (0.69–1.22) for bupropion (Figures 1 and 2). We found no significant associations between paroxetine and RVOTO (1.07, 0.59–1.93), or between sertraline and VSD (1.04, 0.76–1.41). Analyses using high dimensional propensity scores yielded essentially the same results (Table S15).
Subgroup and sensitivity analyses
There was no evidence of effect modification by age or race (Table S16). We did not observe a dose-response relationship either for the first or for the highest dose dispensed (Table S17). The relative risk for cardiac malformations associated with the use of SSRIs appeared similar for users of antidepressant monotherapy (RR=1.04, 0.90–1.21) and polytherapy (RR=1.17, 0.90–1.53). Overall findings were not qualitatively affected when we varied the exposure and outcome definitions (Table S18–S19). The shift in effect estimates resulting from correction for predicted outcome misclassification ranged from 1.3% to 9.6% (Table S20).
We replicated the well-known associations between cardiac malformations and maternal diabetes (RR=3.7, 3.4–4.0), anticonvulsant use (RR=1.6, 1.3–1.8), and multifetal pregnancy (RR=2.9, 2.8–3.1).
DISCUSSION
In this cohort of 949,504 pregnancies in the Medicaid program, after adjustment for depression and other potential confounding factors, we found no significant increase in the risk of cardiac malformations in infants born to women who took antidepressants during the first trimester, as compared with unexposed women. Further, no significantly increased risks were observed for specific cardiac defects previously hypothesized to be associated with such drug use, for specific antidepressant medication classes, or for the most commonly used SSRIs.
Our results do not support earlier findings of an association between antidepressants and cardiac anomalies, in particular for paroxetine and sertraline.13,14,19 In contrast to earlier studies, our analysis restricted the cohort to women with a recorded diagnosis of depression, in order to mitigate potential confounding by the underlying psychiatric illness and associated conditions and behaviors, which might increase the risk for structural cardiac malformations through several mechanisms. Smoking, alcohol and drug use, poor maternal diet, obesity, and chronic conditions such as diabetes and hypertension are all more common in depressed patients and are potential risk factors for congenital cardiac anomalies.34 In addition, depressed, anxious women utilize more health care resources, including ultrasounds, amniocentesis and infant echocardiograms than their healthy counterparts.35,36 Hence, there is a higher chance of detecting an infant with a cardiac malformation that might have gone clinically undetected in other women, particularly milder defects such as muscular VSDs which often close during early childhood. In addition to restricting analyses to women with a depression diagnosis, we adjusted for a large set of predefined and empirical potential confounding variables through the use of propensity scores. Although this approach cannot eliminate all potential confounding, it resulted in exposure groups with virtually identical measured characteristics, and tended to move the risk estimates further downward.
Our crude associations were weaker than what has been reported in some prior studies. A potential concern is misclassification of the exposure and/or the outcome, since non-differential misclassification will tend to bias results towards the null.28 Filling a prescription does not guarantee that the medication was actually taken as prescribed. However, secondary analyses in which we required women to have filled or refilled a prescription during the first trimester did not substantially alter the findings, but estimates were less precise due to the reduced cohort size. We used validated definitions for outcomes based on ICD coding, but a non-trivial proportion of cases were not confirmed on record review. However, an analysis which corrected the relative risk using conservative estimates for the positive predictive values yielded similar results. Finally, the strength of the association between some well-known risk factors (diabetes, anticonvulsant use, multifetal pregnancy) and cardiac malformations estimated in our dataset is consistent with prior reports, supporting the premise that the outcomes of interest were well captured in our study.
Strengths of the Medicaid Analytic eXtract include its very large population-based cohort, objective assessment of drug exposure, linkable clinical information, access to medical records, and the availability of information on multiple pregnancy outcomes and on a wide range of potential confounders. However, our study also has some important limitations. The cohort included live births only. Severe cardiac malformations that result in spontaneous abortions, stillbirths or pregnancy terminations for cardiac anomalies will therefore be missed. Although this could result in a bias that would underestimate the strength of the associations, our study shares this limitation with the studies that identified the potential associations, so this cannot explain the discrepant findings. Moreover, differences in the proportion of terminations among women with depression treated with antidepressants versus those untreated within levels of covariates used in the adjustment would have to be greater than seems plausible to fully account for our findings (see SupplementaryAppendix). Second, there is potential for misclassification. Information on lifestyle factors contained in administrative data is incomplete (e.g., smoking, obesity, alcohol and drug abuse/dependance) or absent (e.g., BMI), as is information on the severity of the underlying condition (proxies only). However, residual confounding by such factors would be unlikely to explain the null findings; data from the National Health and Nutrition Examination Survey indicate, for example, that women of childbearing age using antidepressants are more likely to smoke and to be obese than non-users, with similar distributions for different antidepressants.37
Medicaid covers the medical expenses for over 40% of births in the US.38 Medicaid-eligible pregnant women are a young, racially diverse vulnerable population that is traditionally understudied. However, we found no evidence of effect modification by socio-demographic characteristics. Therefore, unless there are other distinguishing factors which affect the biologic relations studied, our results should be generalizable to other populations.28
Decisions by clinicians and women about whether to continue or discontinue treatment with antidepressants during pregnancy must balance potential risks of treatment with the risks of not treating women with severe depression.39 In conclusion, our results suggest that first trimester use of antidepressants does not substantively increase the risk of specific cardiac defects. The accumulated evidence implies low absolute risks and argue against the existence of important cardiac teratogenic effects for the most commonly used antidepressant medications.
Supplementary Material
Acknowledgments
The authors would like to thank Dr. Robert J. Glynn, Sc.D., Ph.D. for helpful comments on an earlier version of this manuscript.
Funding:
This study was supported by AHRQ Award R01 HSO18533. The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript. Krista Huybrechts was supported by a career development grant K01MH099141 from the National Institute of Mental Health. Kristin Palmsten was supported by Training Grant T32HD060454 in Reproductive, Perinatal and Pediatric Epidemiology from the National Institute of Child Health and Human Development, National Institutes of Health.
Footnotes
Disclosure:
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org.
References
- 1.Evans J, Heron J, Francomb H, Oke S, Golding J. Cohort study of depressed mood during pregnancy and after childbirth. BMJ. 2001;323:257–60. doi: 10.1136/bmj.323.7307.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Andrade SE, Raebel MA, Brown J, et al. Use of antidepressant medications during pregnancy: a multisite study. Am J Obstet Gynecol. 2008;198:194, e1–5. doi: 10.1016/j.ajog.2007.07.036. [DOI] [PubMed] [Google Scholar]
- 3.Mitchell A, Gilboa S, Werler M, Kelley K, Louik C, Hernandez-Diaz S. Medication use during pregnancy, with particular focus on prescription drugs: 1976–2008. American Journal of Obstetrics & Gynecology. 2011;205:51, e1–8. doi: 10.1016/j.ajog.2011.02.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huybrechts K, Palmsten K, Mogun H, et al. National trends in antidepressant medication treatment among publicly-insured pregnant women. General Hospital Psychiatry. 2013;35:265–71. doi: 10.1016/j.genhosppsych.2012.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.US Food and Drug Administration. [last accessed September 30, 2013];FDA Advising of Risk of Birth Defects with Paxil. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2005/ucm108527.htm.
- 6.Diav-Citrin O, Shechtman S, Bar-Oz B, Cantrell D, Arnon J, Ornoy A. Pregnancy outcome after in utero exposure to valproate : evidence of dose relationship in teratogenic effect. CNS Drugs. 2008;22:325–34. doi: 10.2165/00023210-200822040-00004. [DOI] [PubMed] [Google Scholar]
- 7.Kallen B, Otterblad Olausson P. Antidepressant drugs during pregnancy and infant congenital heart defect. Reprod Toxicol. 2006;21:221–2. doi: 10.1016/j.reprotox.2005.11.006. [DOI] [PubMed] [Google Scholar]
- 8.Kallen BA, Otterblad Olausson P. Maternal use of selective serotonin re-uptake inhibitors in early pregnancy and infant congenital malformations. Birth Defects Res A Clin Mol Teratol. 2007;79:301–8. doi: 10.1002/bdra.20327. [DOI] [PubMed] [Google Scholar]
- 9.Malm H, Klaukka T, Neuvonen PJ. Risks associated with selective serotonin reuptake inhibitors in pregnancy. Obstet Gynecol. 2005;106:1289–96. doi: 10.1097/01.AOG.0000187302.61812.53. [DOI] [PubMed] [Google Scholar]
- 10.Wogelius P, Norgaard M, Gislum M, et al. Maternal use of selective serotonin reuptake inhibitors and risk of congenital malformations. Epidemiology. 2006;17:701–4. doi: 10.1097/01.ede.0000239581.76793.ae. [DOI] [PubMed] [Google Scholar]
- 11.Cole JA, Ephross SA, Cosmatos IS, Walker AM. Paroxetine in the first trimester and the prevalence of congenital malformations. Pharmacoepidemiol Drug Saf. 2007;16:1075–85. doi: 10.1002/pds.1463. [DOI] [PubMed] [Google Scholar]
- 12.Berard A, Ramos E, Rey E, Blais L, St-Andre M, Oraichi D. First trimester exposure to paroxetine and risk of cardiac malformations in infants: the importance of dosage. Birth Defects Res B Dev Reprod Toxicol. 2007;80:18–27. doi: 10.1002/bdrb.20099. [DOI] [PubMed] [Google Scholar]
- 13.Louik C, Lin AE, Werler MM, Hernandez-Diaz S, Mitchell AA. First-trimester use of selective serotonin-reuptake inhibitors and the risk of birth defects. N Engl J Med. 2007;356:2675–83. doi: 10.1056/NEJMoa067407. [DOI] [PubMed] [Google Scholar]
- 14.Alwan S, Reefhuis J, Rasmussen SA, Olney RS, Friedman JM. Use of selective serotonin-reuptake inhibitors in pregnancy and the risk of birth defects. N Engl J Med. 2007;356:2684–92. doi: 10.1056/NEJMoa066584. [DOI] [PubMed] [Google Scholar]
- 15.Einarson A, Pistelli A, DeSantis M, et al. Evaluation of the risk of congenital cardiovascular defects associated with use of paroxetine during pregnancy. Am J Psychiatry. 2008;165:749–52. doi: 10.1176/appi.ajp.2007.07060879. [DOI] [PubMed] [Google Scholar]
- 16.Wichman CL, Moore KM, Lang TR, St Sauver JL, Heise RH, Jr, Watson WJ. Congenital heart disease associated with selective serotonin reuptake inhibitor use during pregnancy. Mayo Clin Proc. 2009;84:23–7. doi: 10.4065/84.1.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nordeng H, van Gelder MM, Spigset O, Koren G, Einarson A, Eberhard-Gran M. Pregnancy outcome after exposure to antidepressants and the role of maternal depression: results from the Norwegian Mother and Child Cohort Study. J Clin Psychopharmacol. 2012;32:186–94. doi: 10.1097/JCP.0b013e3182490eaf. [DOI] [PubMed] [Google Scholar]
- 18.Oberlander TF, Warburton W, Misri S, Riggs W, Aghajanian J, Hertzman C. Major congenital malformations following prenatal exposure to serotonin reuptake inhibitors and benzodiazepines using population-based health data. Birth Defects Res B Dev Reprod Toxicol. 2008;83:68–76. doi: 10.1002/bdrb.20144. [DOI] [PubMed] [Google Scholar]
- 19.Pedersen LH, Henriksen TB, Vestergaard M, Olsen J, Bech BH. Selective serotonin reuptake inhibitors in pregnancy and congenital malformations: population based cohort study. BMJ. 2009;339:b3569. doi: 10.1136/bmj.b3569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Greene MF. Teratogenicity of SSRIs--serious concern or much ado about little? N Engl J Med. 2007;356:2732–3. doi: 10.1056/NEJMe078079. [DOI] [PubMed] [Google Scholar]
- 21.Wurst KE, Poole C, Ephross SA, Olshan AF. First trimester paroxetine use and the prevalence of congenital, specifically cardiac, defects: a meta-analysis of epidemiological studies. Birth Defects Res A Clin Mol Teratol. 2010;88:159–70. doi: 10.1002/bdra.20627. [DOI] [PubMed] [Google Scholar]
- 22.Palmsten K, Huybrechts K, Mogun H, et al. Harnessing the Medicaid Analytic eXtract (MAX) to Evaluate Medications in Pregnancy: Design Considerations. PLoS ONE. 2013;8:e67405. doi: 10.1371/journal.pone.0067405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Margulis AV, Setoguchi S, Mittleman MA, Glynn RJ, Dormuth CR, Hernandez-Diaz S. Algorithms to estimate the beginning of pregnancy in administrative databases. Pharmacoepidemiol Drug Saf. 2012;22:16–24. doi: 10.1002/pds.3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Centers for Medicare & Medicaid Services. [Accessed March 20, 2013.];Medicaid Analytic eXtract (MAX) General Information. MAX 1999–2005 state claims anomalies from the “2005 Files” zipped file within the “MAX Data 2005 to 2008 General Information, Data Dictionaries, Data Element Lists, Data Anomalies, Validation Table Measures and SAS Loads zipped file. Available: < http://www.cms.gov/Research-Statistics-Data-and-Systems/Computer-Data-and-Systems/MedicaidDataSourcesGenInfo/MAXGeneralInformation.html>.
- 25.Holmes LB, Westgate MN. Using ICD-9 codes to establish prevalence of malformations in newborn infants. Birth Defects Res A Clin Mol Teratol. 2012;94:208–14. doi: 10.1002/bdra.23001. [DOI] [PubMed] [Google Scholar]
- 26.Palmsten K, Huybrechts K, Kowal M, Hernandez-Diaz S. Validity of Maternal and Infant Outcomes within Medicaid Data. Society for Perinatal Epidemiological Research; Boston, MA: 2013. [Google Scholar]
- 27.Schneeweiss S, Seeger JD, Maclure M, Wang PS, Avorn J, Glynn RJ. Performance of comorbidity scores to control for confounding in epidemiologic studies using claims data. Am J Epidemiol. 2001;154:854–64. doi: 10.1093/aje/154.9.854. [DOI] [PubMed] [Google Scholar]
- 28.Rothman KJ, Greenland S, Lash TL. Modern Epidemiology. 3. Philadelphia: Lippincott Williams & Wilkins; 2008. [Google Scholar]
- 29.Braitman LE, Rosenbaum PR. Rare outcomes, common treatments: Analytic strategies using propensity scores. Annals of Internal Medicine. 2002;137:693–5. doi: 10.7326/0003-4819-137-8-200210150-00015. [DOI] [PubMed] [Google Scholar]
- 30.Schneeweiss S, Rassen J, Glynn R, Avorn J, Mogun H, Brookhart M. High-dimensional propensity score adjustment in studies of treatment effects using health care claims data. Epidemiology. 2009;20:512–22. doi: 10.1097/EDE.0b013e3181a663cc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.O’Donnell J, Shelton R. Drug therapy of depression and anxiety disorders. In: Brunton L, Chabner B, Knollmann B, editors. Goodman & Gilman’s The Pharmacological Basis of therapeutics. 12. New York: McGraw-Hill; 2011. [Google Scholar]
- 32.Fox MP, Lash TL, Greenland S. A method to automate probabilistic sensitivity analyses of misclassified binary variables. Int J Epidemiol. 2005;34:1370–6. doi: 10.1093/ije/dyi184. [DOI] [PubMed] [Google Scholar]
- 33.Holmes L. Common Malformations. New York: Oxford University Press, Inc; 2012. Chapter 11: Heart Defects; p. 459. [Google Scholar]
- 34.Jenkins KJ, Correa A, Feinstein JA, et al. Noninherited Risk Factors and Congenital Cardiovascular Defects: Current Knowledge: A Scientific Statement From the American Heart Association Council on Cardiovascular Disease in the Young: Endorsed by the American Academy of Pediatrics. Circulation. 2007;115:2995–3014. doi: 10.1161/CIRCULATIONAHA.106.183216. [DOI] [PubMed] [Google Scholar]
- 35.Bar-Oz B, Einarson T, Einarson A, et al. Paroxetine and congenital malformations: meta-Analysis and consideration of potential confounding factors. Clin Ther. 2007;29:918–26. doi: 10.1016/j.clinthera.2007.05.003. [DOI] [PubMed] [Google Scholar]
- 36.Koren G. The effect of ascertainment bias in evaluating gestational antidepressant exposure. J Popul Ther Clin Pharmacol. 2011;18:e174–5. [PubMed] [Google Scholar]
- 37.Centers for Disease Control and Prevention (CDC). National Center for Health Statistics (NCHS) National Health and Nutrition Examination Survey Data. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention; 2005. [ www.cdc.gov/nchs/data/nhanes/nhanes_03_04/nhanes_analytic_guidelines_dec_2005.pdf] [Google Scholar]
- 38.Garcia G. [Accessed March 5, 2012.];Maternal and Child Health (MCH) Update: States Increase Eligibility for Children’s Health in 2007. Electronic citation Available at: http://www.nga.org/files/live/sites/NGA/files/pdf/0811MCHUPDATE.PDF;jsessionid=7B47A647247DD4E5CB9B709C8F9797AE.
- 39.Cohen LS, Altshuler LL, Harlow BL, et al. Relapse of major depression during pregnancy in women who maintain or discontinue antidepressant treatment. JAMA. 2006;295:499–507. doi: 10.1001/jama.295.5.499. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.