Abstract
With aging, abnormal benign growth of the prostate results in benign prostate hyperplasia (BPH) with concomitant lower urinary tract symptoms (LUTS). Because the prostate is an androgen target tissue, and transforms testosterone into 5α-dihydrotestosterone (5α-DHT), a potent androgen, via 5α-reductase (5α-R) activity, inhibiting this key metabolic reaction was identified as a target for drug development to treat symptoms of BPH. Two drugs, namely finasteride and dutasteride were developed as specific 5α-reductase inhibitors (5α-RIs) and were approved by the U.S. Food and Drug Administration for the treatment of BPH symptoms. These agents have proven useful in the reducing urinary retention and minimizing surgical intervention in patients with BPH symptoms and considerable literature exists describing the benefits of these agents. In this review we highlight the adverse side effects of 5α-RIs on sexual function, high grade prostate cancer incidence, central nervous system function and on depression. 5α-Rs isoforms (types 1-3) are widely distributed in many tissues including the central nervous system and inhibition of these enzymes results in blockade of synthesis of several key hormones and neuro-active steroids leading to a host of adverse effects, including loss of or reduced libido, erectile dysfunction, orgasmic dysfunction, increased high Gleason grade prostate cancer, observed heart failure and cardiovascular events in clinical trials, and depression. Considerable evidence exists from preclinical and clinical studies, which point to significant and serious adverse effects of 5α-RIs, finasteride and dutasteride, on sexual health, vascular health, psychological health and the overall quality of life. Physicians need to be aware of such potential adverse effects and communicate such information to their patients prior to commencing 5α-RIs therapy.
Keywords: 5α-Reductase inhibitors, Depression, Physiological sexual dysfunction, Prostate neoplasms
INTRODUCTION
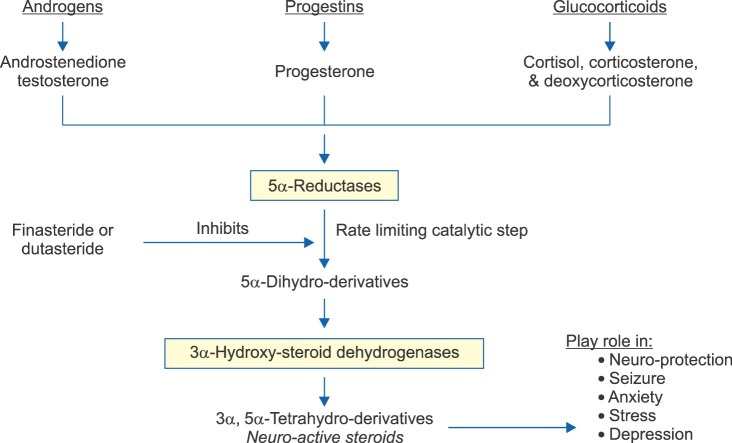
5α-Reductases (5α-Rs) are a family of enzymes, widely distributed in many tissues including the central nervous system [1,2]. 5α-Rs transform steroid precursors into active hormones and neurosteroids [1,2]. The 5α-Rs catalyzed reaction is the rate limiting step in the conversion of testosterone (T) to 5α-dihydroprogesterone (5α-DHP), progesterone to 5α-DHP, deoxycorticosterone (DOC) to 5β-dihydrodeoxycorticosterone, cortisol to 5α-dihydrocortisol, aldosterone to 5α-dihydro-aldosterone [1,2]. The steroid products of the 5α-Rs pathways undergo further metabolism by the 3α-hydroxy-steroid dehydrogenase (3α-HSD) to produce a host of active neurosteroids with important physiological function, in many tissues including the central nervous system (CNS) (Fig. 1) [1,2]. In addition, synthesis of dolichol from polyprenol was recently shown to require the activity of the 5α-R type 3 [3]. Since dolichol phosphate is critical in N-glycosylation of membrane proteins, inhibition of the activity of 5α-R type 3 may have undesirable effects as a result of attenuation of this and other biochemical pathways important in modulating cellular function [4].
FIG. 1.
Transformation of androgens, progestins and glucocorticoids into 5α-dihydroderivatives by 5α-reductases and tetrahydro-metabolites (neurosteroids) by 3α-hydroxysteroid dehydrogenases. The rate limiting step in this pathway is the catalysis by 5α-reductases.
The prostate is an androgen-dependent target organ and requires transformation of T to 5α-dihydrotestosterone (5α-DHT) for development, growth and maintenance of function [5]. In the adult male, once the prostate reaches its normal size, its growth plateaus and remains fairly constant, even in the presence of continuous androgen milieu [5]. Interestingly, with advancing age, circulating androgen levels decrease; however, BPH occurs when androgen levels are relatively low. This would suggest that the pathogenesis of BPH may not be solely dependent on androgens for growth [6,7]. Other growth factors, such as epidermal growth factor, and transforming growth factor-β may come into play in the pathogenesis of BPH; however, this concept has yet to be fully appreciated or understood. Since, the prostate remains responsive to androgens, it was believed that inhibition of 5α-DHT formation will halt or reverse BPH growth and alleviate the symptoms of BPH. Therefore, 5α-Rs were targeted for development of specific inhibitors to treat the symptoms of BPH and lower urinary tract symptoms (LUTS).
The American Urological Association (AUA) and the European Association of Urology guidelines for the treatment of symptomatic BPH recommend use of either alpha blockers (ABs), 5α-reductase inhibitors (5α-RIs) alone or in combinations. ABs are most commonly prescribed followed by 5α-RIs. However, many patients treated with either ABs or 5α-RI monotherapy do not experience significant resolution of their chief complaint and are subsequently treated with combination therapy. The approval of tadalafil, a phosphodiesterase type 5 inhibitor, for combined therapy provided new strategy for management of symptoms of BPH. Furthermore, while 5α-RIs therapy was shown to be effective in reducing prostate volume by approximately 20% in many patients. However, about 25%-30% of patients do not experience any improvement in their urinary symptoms and another 5%-7% developed worsening symptoms and may ultimately require surgery [8]. It should be noted that resistance to therapy with finasteride may occur through silencing of the 5α-R type 2 gene by DNA methylation, leading to a state in which approximately 30% of patients do not express 5α-R type 2 in the prostate [8].
Until recently, the two U.S. Food and Drug Administration (FDA)-approved 5α-RIs, finasteride and dutasteride, were deemed safe and effective in treatment of BPH. However, the adverse effects of 5α-RIs were known for some time, but were deemed not clinically significant and were often dismissed with the arguments that the adverse effects are observed in a small number of patients [1,2,9,10,11,12].
In an attempt by the pharmaceutical industry to repurpose these drugs as chemo-preventive agents for prostate cancer (PCa), under the premise that PCa is androgen-dependent and inhibition of 5α-Rs activity prevents development of PCa, approval was sought for these two drugs as chemo-preventive agents for PCa [13,14]. Although there is no scientific evidence to support the hypothesis that targeting these enzymes will prevent development of Pica, several clinical trials were initiated and the results were presented to the FDA in December of 2010 with the hope to gain approval for these agents, as chemo-prevention for PCa. Interestingly, both finasteride and dutasteride reduced the incidence of lower grade PCa tumors, but significantly increased the incidence of Gleason high grade PCa tumors [15,16] without a significant difference in survival rates [17]. The issues concerning safety and the lack of evidence that either finasteride or dutasteride reduced incidence of high grade PCa led to strong and overwhelming disapproval of these drugs for chemoprevention of PCa by the panel of scientists and medical experts [16].
Finasteride is also approved for the treatment of male pattern hair loss (MPHL; otherwise known as genetic alopecia). While many subjects treated with finasteride for MPHL had experienced minimal or no obvious side effects, for some patients the adverse side effects were manifested in loss of libido, diminished libido, erectile dysfunction, ejaculatory dysfunction, anxiety, depression, and in some cases contemplating suicide [1,2,11,12,18].
In this review, we summarize the findings in the contemporary literature on the adverse side effects of 5α-RIs therapy on sexual function, Gleason high grade PCa, central nervous system function and depression. Whenever possible, we attempted to provide potential biochemical and physiological mechanisms that may explain the pathophysiology underlying these serious, and in some instances, persistent or irreversible side effects.
1. Adverse effects of 5α-RIs' therapy on sexual function
To date, the adverse side effects of 5α-RIs on sexual function and the impact on the overall health have received minimal attention [1,2]. This is attributed, in part, to the fact that the adverse side effects of 5α-RIs were deemed clinically less important [13,14,19]. However, in some patients, these sexual side effects are persistent or irreversible [1,2,11,18] with concomitant negative emotional toll and reduced quality of life.
The importance of 5α-DHT in development and maintenance of male sexual organs was noted in males with 5α-reductase deficiency [20]. MacLaughlin and Donahoe [21] pointed out that loss of 5α-DHT due to congenital defect in 5α-Rs leads to development of ambiguous genitalia including the erectile tissue. Baldinotti et al. [20] provided some key information on the ambiguous genitalia in infants with varying degree of 5α-Rs deficiency.
1) Evidence from preclinical studies
Considerable evidence exists suggesting that effects of T on erectile physiology were mediated by the 5α-reduced metabolite, 5α-DHT [22]. During the past 4 decades, animal studies provided evidence for a key role of 5α-DHT in erectile physiology [23,24,25,26] (Table 1).
TABLE 1.
Effects of 5α-reductase inhibitors on nitric oxide synthase expression and activity, trabecular smooth muscle content and erections, in vivo, as assessed by intracavernosal pressure or by behavioral observations
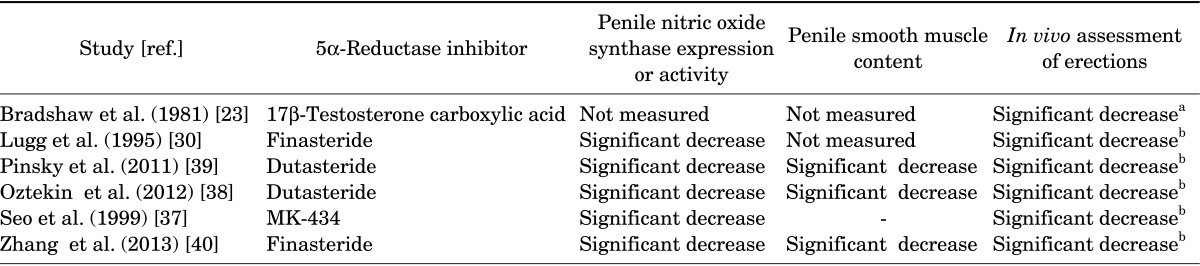
a:"The incidence of adverse events related to sexual dysfunction were significantly higher in the finasteride group than in the placebo group (ejaculation disorder 7.7% vs. 1.7% and impotence 15.8% vs. 6.3%; p<0.01 for both parameters)." [42]. b:"Finasteride treatment led to a small but significant increase in plasma high-density lipoprotein cholesterol and apolipoprotein A1 levels with a concomitant increase in total cholesterol levels, but had no significant effect on plasma low-density lipoprotein and very low-density lipoprotein cholesterol, apolipoprotein B, or triglyceride levels." [43].
Mantzoros et al. [27] reported that castration of mature male animals resulted in reduced erectile function and treatment of castrated animals with T or its 5α-reduced metabolite, 5α-DHT, reversed this effect. Administration of 5α-RI, 17β-testosterone carboxylic acid (17βTC) blocked the stimulatory effects of T propionate (TP) on erection in castrated rats [23,28]. Administration of 5α-DHT with or without the 5α-RIs, 17βTC, restored sexual behavior in long-term castrated male rats and mice suggesting a critical role for 5α-DHT in erectile physiology [29]. Further animal studies showed castration resulted in 50% reduction in erectile response, which was reversed by T treatment [30]. However, treatment with T together with finasteride did not restore the erectile response in castrated animals, suggesting that 5α-DHT is an important hormone in erectile physiology. Administration of 5α-DHT together with finasteride, however, restored nitric oxide synthase (nNOS) expression and activity and also restored the erectile response to electric field stimulation, confirming the role of this metabolite in erectile physiology [30]. In castrated and adrenalectomized rats, treatment with 5α-DHT for 7 days restored erectile function to levels similar to that of control animals [31]. Other studies demonstrated that 5α-DHT treatment in castrated rats improved the erectile response to electrical field stimulation (EFS) [32,33]. Castration in male rats eliminates noncontact erections and this response was restored by 5α-DHT implantation [34,35]. Noncontact erections in animals are thought to be similar to human psychogenic erections [36]. Reduced erectile response and reflex erections were observed in castrated animals [37] and treatment of castrated animals with T or 5α-DHT restored the number of erectile responses and reflex erections. However, only 5α-DHT restored erectile responses and reflex erections, when animals were treated with daily injections of the 5α-R inhibitor MK-434 (1 mg/kg) together with T or 5α-DHT [37]. These findings suggest that 5α-DHT plays a key physiological role in erectile physiology in the animal model and 5α-RIs treatment are likely to produce sexual adverse effect on the erectile response.
In more recent studies by Oztekin et al. [38] and Pinsky et al. [39] it was demonstrated that treatment of mature animals with dutasteride resulted in reduced serum DHT levels by ~86.5% after 30 days. The intracavernosal pressure (ICP) decreased significantly in animals treated with dutasteride. Similarly, EFS-induced and acetylcholine-induced penile smooth muscle relaxations were also significantly attenuated. More profoundly, connective tissue deposition was markedly increased in the corpus cavernosum of the dutasteride-treated animals. Concomitant with these changes are the markedly reduced expression of neuronal nNOS and increased expression of the inducible NOS, suggesting that dutasteride induces altered gene expression in the corpus cavernosum. Oztekin et al. [38] investigated the effects of dutasteride on erectile function after 6 weeks of treatment followed by a washout period of 2 weeks. The ratio of ICP over mean arterial pressure [MAP] and total ICP in the dutasteride treated animals were significantly decreased when compared with control animals (Fig. 2). Even after two-week washout period the effects persisted and the ICP/MAP was significantly reduced in the dutasteride treated animal, suggesting a persistent effect of the drug on erectile physiology (Fig. 2). Also the endothelium-dependent smooth muscle relaxations were also diminished in the dutasteride treated animals. The authors suggested that discontinuation of dutasteride did not restore erectile function, indicating a time-dependent detriment of dutasteride on erectile physiology (Fig. 2).
FIG. 2.
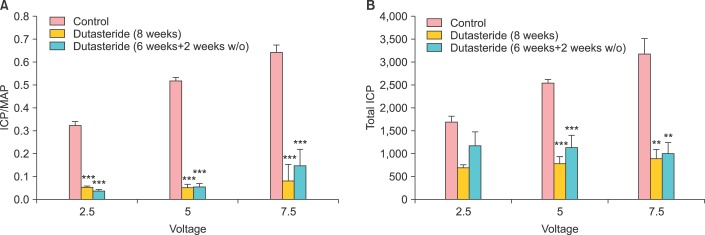
Effects of dutasteride on erectile physiology in the animal model. Graphs showing: (A) intracavernosal pressure (ICP)/mean arterial pressure (MAP) and (B) total ICP at 2.5, 5, and 7.5 voltage levels in control, 8-week dutasteride and 6-week dutasteride plus 2-week washout groups. Data are mean±standard error of the mean (n=8-10). **p<0.01 and ***p<0.001 vs. control group (analysis of variance, Bonferroni post hoc). Adapted from Oztekin CV, et al. J Sex Med 2012;9:1773-81, with permission of John Wiley & Sons, Inc. [38].
A recent study by Zhang et al. [40] reported that treatment of male mature animals for 16 weeks with a daily oral dose of 4.5 mg/kg finasteride significantly reduced 5α-DHT levels and attenuated penile erectile response to EFS of the cavernous nerve. This treatment also reduced the trabecular smooth muscle content and increased connective tissue deposition, reminiscent of the data reported by Traish et al. [41], in the castrated animal model. In addition, finasteride treatment reduced endothelial nitric oxide synthase expression. More importantly, this treatment decreased autophagy and produced deterioration in the ultrastructure of the corpus cavernosum, including mitochondria injury, and increased trabecular smooth muscle cell death (Fig. 3) [40]. These findings, together with the previously reported data from pre-clinical studies point out to serious adverse effects of finasteride and dutasteride on the anatomy, physiology and cell biology of erectile function (Table 1) and suggest that the effects of these drugs may be persistent or irreversible.
FIG. 3.
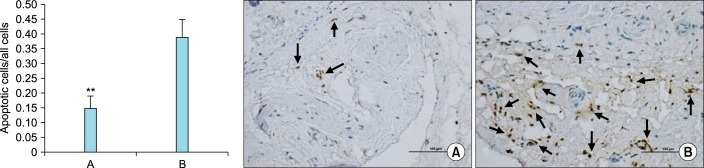
Effects of long-term treatment with finasteride on corpus cavernosum smooth muscle death. Terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling quantification of apoptotic index of cavernous smooth muscle cells in rats with or without 5α-reductase inhibitor (5α-RI) treatment. (Left) Apoptotic indexes (ratio of apoptotic cells to all cells) of 2 groups were assessed. For each corpus cavernosum sample, 5 randomly obtained fields were selected, and the mean ratio of apoptotic cells to all cells was used to calculate the apoptotic index. Values for 5 samples in each group presented as ratio±standard deviation. **p<0.001 compared with 5α-RI-treatment group (unpaired t-test); (A) control group and (B) 5α-RI-treated group. Black arrows indicate apoptotic cells with dark brown-stained nuclei: (A) control group and (B) 5α-RI-treated group (ApopTag Peroxidase In Situ Apoptosis Detection Kit, Scale bar=100 µm, ×200). Adapted from Zhang MG, et al. Urology 2013;82:743.e9-15, with permission of Elsevier Inc. [40].
2) Evidence from clinical studies
Nickel et al. [42], evaluated the efficacy and safety of 2 years treatment of moderate BPH with finasteride. In the safety analysis, the incidence of adverse events related to sexual dysfunction were significantly higher in the finasteride group than in the placebo group (ejaculation disorder, 7.7% vs. 1.7%; erectile dysfunction (ED), 15.8% vs. 6.3%, respectively; p<0.01 or p=0.01 for both parameters). Tenover et al. [43] also showed that the overall sexual adverse events were greater with finasteride (12.3%) compared with placebo (6%). Finasteride increased the incidence of ED by 7.4% compared to 3.3% in the placebo arm. Similarly, finasteride resulted in loss of libido (4.9%) compared to 2.9% in the placebo arm. In a follow-up to the phase III North American Study, Hudson et al. [44] reported that all patients who completed a 12-month period with 5-mg finasteride were invited for an open-label extension of 4 years. The open-label results showed that ejaculation disorder for years 2, 3, 4 and 5, respectively, were 3.2%, 2.2%, 1.9%, 2.7%, respectively; ED 10.4%, 6.7%, 7.3%, 9.6%, respectively, and decreased libido was 4.8%, 2.7%, 3.9%, 3.7%, respectively. The AUA BPH guideline committee meta-analysis, reported that sexual adverse events due to finasteride and placebo were as follows: libido, 5%, 3%; erectile problems, 8%, 4%; and ejaculation, 4%, 1%, respectively. The PLESS trial [45] enrolled 3,040 men with BPH. Overall maintenance of erection diminished in both groups, but was greater in the finasteride treatment group. In an observational cohort of 14,772 men taking finasteride, ED was the most common adverse event leading to withdrawal from the trial [46]. The AUA clinical practice guideline reported erectile problems in 8% and 4% of patients taking finasteride and placebo, respectively [47].
Edwards and Moore [48] reviewed several clinical trials, three of these trials contained active controls, and 19 trials with placebo groups and reported significantly more men with sexual dysfunction (14%) with finasteride than with placebo at 12 months of treatment (Reduced libido, 5%; ED, 8%; ejaculation disorder, 2%). Incidence rates of sexual dysfunction ranged between 2%-14% with finasteride and 0.6%-7% with placebo. The data from the REDUCE trial [13] also reported that dutasteride resulted in loss of libido/decreased libido of 5.2% and ED of 9% compared to 2.9% and 5.7% in the placebo arm. Similarly in the PCPT trial [14] the incidence of loss of libido (5.8%), ED (5.9%) and ejaculatory disorders (13.1%) higher than that observed in the placebo arm. Similar data was reported by Wessells et al. [19]. The authors suggested that in men who discontinued treatment with finasteride approximately 50% of the adverse sexual events were resolved. However, the authors do not address what happened to the remaining 50% whose adverse sexual events were not resolved after drug discontinuation nor do they address the loss to follow up. Gur et al. [10] in a recent review suggested that decreased circulating 5α-DHT resulting from 5α-RIs use is associated with diminished sexual desire and/or orgasm. The authors suggested that the data from clinical trials with 5α-RIs report prevalence rates of erectile dysfunction of 5%-9%. They further noted that the adverse sexual effects are associated with decreased self-esteem, quality of life, and ability to maintain an intimate relationship.
ED was consistently noted in observational studies as well as in double-blind, randomized, placebo controlled trials (Table 2). Roehrborn et al. [49] and Siami et al. [50] reported that approximately six percent of the patients reported ED in a 2-year follow-up to the CombAT trial [49,50]. Hudson et al. [44] reported that ED occurred in 6.7% and 4.0% of patients treated with dutasteride or placebo, respectively, in a trial for treatment of BPH [44]. Desgrandchamps et al. [51] reported 7% of the drug related adverse effects were ED. In the PROSPECT study, ED was established but determined subjectively in an open-ended interview [52].
TABLE 2.
Effects of 5α-reductaste inhibitors on sexual function
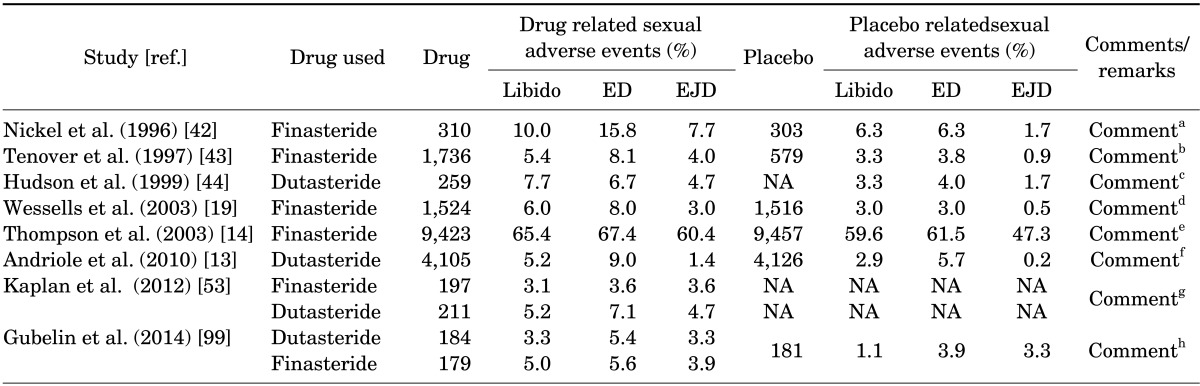
ED, erectile dysfunction; EJD, ejaculatory dysfunction; NA, not applicable.
a:"The incidence of adverse events related to sexual dysfunction were significantly higher in the finasteride group than in the placebo group (ejaculation disorder 7.7% vs. 1.7% and impotence 15.8% vs. 6.3%; p<0.01 for both parameters)." [42]. b:"Finasteride treatment led to a small but significant increase in plasma high-density lipoprotein cholesterol and apolipoprotein A1 levels with a concomitant increase in total cholesterol levels, but had no significant effect on plasma low-density lipoprotein and very low-density lipoprotein cholesterol, apolipoprotein B, or triglyceride levels." [43]. c:"The overall dropout rate was 37.4%, and only 8.1% of patients discontinued therapy because of treatment failure. These rates are consistent with the dropout rates for nonresponders in previous 2-year finasteride studies, which ranged from 3.7% to 5.2%." [44]. d:"In men who discontinued with a sexual adverse events (AE), 50% and 41% experienced resolution of their sexual AE after discontinuing finasteride or placebo therapy, respectively." [19]. The authors did not discuss what happened to those whose sexual AE were not resolved nor did they indicate how many patients were lost to follow up? e:"High-grade disease was noted in 6.4% of the men in the finasteride group, as compared with 5.1 percent of those in the placebo group. A difference in the rate of high-grade disease was seen within the first year of the study." [14]. f:"There was an unexpected imbalance in a composite event termed "cardiac failure," which included conditions such as congestive heart failure, cardiac failure, acute cardiac failure, ventricular failure, cardiopulmonary failure, and congestive cardiomyopathy." [13]. g:"Compared with finasteride, treatment with dutasteride resulted in significantly greater erectile dysfunction, more sexual side effects leading to discontinuation and a greater incidence of breast complications…In addition, at year 5, benign prostate hyperplasia patients on dutasteride had significantly worsened International Index of Erectile Function scores relative to baseline than did those on finasteride." [53]. h:Reported results occurred in less than 24 weeks. [99].
In a retrospective analysis of data from consecutive patients treated at a single clinic, Kaplan et al. [53] showed that both finasteride and dutasteride resulted in sexual adverse effects. Approximately 42.6% discontinued treatment with finasteride and 57.5% discontinued treatment with dutasteride and 29.4% of the patients were lost to follow up in the finasteride group and 36.5 lost to follow up in the dutasteride group, respectively. ED was reported in 3.6% in the finasteride and 7.1% in the dutasteride, respectively. Ejaculatory dysfunction was reported in 3.6% in the finasteride group and 4.7% in the dutasteride group respectively. Decreased libido was approximately 3.1% in the finasteride and 5.2% in the dutasteride group, respectively. The authors suggested that dutasteride elicits more sexual side effects and breast complications than finasteride [53]. Chi and Kim [54] investigated the effects of dutasteride treatment during a 1-year follow-up period in Korean men. The erectile function domain (EF) of the International Index of Erectile Function decreased significantly after 1 month and remained significantly reduced even after 12 months of treatment. Orgasmic function and sexual desire were significantly reduced but slowly recovered after six months. The authors concluded that "After 1 month of treatment, dutasteride therapy resulted in a significant reduction in all investigated sexual functions. Overall, recovery in sexual function was noted at 3 months, and orgasmic function and sexual desire were restored to baseline levels at 6 months. However, EF was still significantly reduced at 12 months". In a recent study Fwu et al. [55] investigated the effects of finasteride with or without combined therapy with ABs on sexual function and found that men assigned to finasteride or finasteride combined with doxazosin experienced a worsening of several domains of sexual function compared to placebo [55].
To this end, the data from a number of clinical studies (Table 2) clearly showed that in some patients, treatment with 5α-RIs diminished libido, erectile, and ejaculatory function. These adverse effects may not be significant in the realm of the overall studies, but for the individual patient this is a serious loss of quality of life and should be given serious considerations prior to commencing therapies with these drugs. 5α-RIs' therapy, while improves urinary symptoms in patients with BPH and may prevent hair loss in patients with MPHL, this therapy produces significant adverse effects in some individuals including loss of libido, ED, ejaculatory dysfunction, and potential depression. These adverse events are serious enough to preclude them from pursuing such therapy. The effects of these agents on vascular health should also be noted in light of recent findings that patients treated with 5α-RIs therapy had significant adverse cardiovascular events [13].
2. 5α-RIs therapy contributes to high Gleason grade PCa
The premise put forth by Huggins (1941) suggested that PCa is androgen-dependent and androgen deprivation results in tumor shrinkage [56]. Androgen-dependency of PCa is predictable because tumor cells are derived from normal prostate cells, which retain expression of the androgen receptor (AR). Androgen deprivation therapy (ADT) of PCa initially results in tumor regression, but the cancer often relapses into androgen-independent cancer within 18-24 months and tumors no longer require androgens for growth [5,6].
Because conversion of T to 5α-DHT is a critical pathway in normal prostate growth and function, inhibition of 5α-R activity in the prostate results in a reduction of intraprostatic 5α-DHT levels and a decrease in prostate growth and size [57]. There is no evidence that T or 5α-DHT causes initiation, promotion or development of PCa [5,6,58,59]. This hypothesis was further supported by the recent study of Muller et al. [59] in which patients enrolled in the Placebo arm of the REDUCE trial [13] showed no association between baseline total T nor 5α-DHT and PCa. The authors concluded that "baseline serum T and 5α-DHT levels were unrelated to prostate cancer detection or grade" [13,59]. This raises the question-how could 5α-RIs be useful as chemo-preventative agents if PCa development is unrelated to T or 5α-DHT? As stated by Walsh (2010) in the FDA panel discussion in 2010, "No clinical benefit has been demonstrated in patients with prostate cancer treated with PROSCAR" [16]. In addition, the potential reduction in the incidence of PCa in the general population with the use of these agents may not exceed 10%, which is not statistically significant [16]. Furthermore, the documented increase in Gleason high-grade PCa tumors in response to treatment with these agents proves that these drugs do not prevent development and growth of PCa; they merely prevent biopsies due to reduction in prostate volume. Most importantly, if 5α-RIs indeed prevent PCa development, then how come they are not approved for treatment of PCa?
As suggested by Crawford et al. [60], Justman [61], and Walsh [62,63] four important points need to be considered before the decision is made to use 5α-RIs for chemo-prevention of PCa. These are: (1) 5α-RIs do not prevent PCa development and therefore, these drugs have no significant effect on reducing the real risk of metastatic PCa in men; (2) 5α-RIs reduce the risk of detecting low-grade tumors, but do not increase survival or reduced mortality [17]; (3) if these drugs indeed chemo-prevent PCa, then why aren't they used in the treatment of PCa?; and (4) to reduce the overall risk of PCa in the population, 5α-RIs as chemo-preventative agents must be entirely free of any side effects [60,61,62,63]. Theoret et al. [15] reported that even after re-evaluation of the data from the PCPT [14] and the REDUCE trial [13] with the revised Gleason scoring system, these drugs were shown to increase the incidence of Gleason high-grade PCa tumors. The authors concluded that "the trade-off inherent in using a 5αRI for prostate cancer prevention is the acceptance of one additional high-grade cancer in order to avert three to four potentially clinically relevant low-grade cancers" [15]. Put more simply, when using 5α-RIs as chemo-preventive agents - the risks outweigh the benefits.
This is not surprising since the recent follow-up data on the PCPT have clearly shown that there are no significant benefits in terms of reducing mortality and increasing survival [17]. As Ehdaie and Touijer [64] pointed out, while the medical community is eager to provide early intervention for patients with high prostate specific antigen levels after therapy, it remains critical to weigh the harm of treatments against the uncertain benefits to the patients. It is incumbent that physicians critically evaluate the current evidence and weigh the risks and benefits of such interventions [64]. Since the PCPT [14] and REDUCE trials [13], used PCa incidence as an end point instead of mortality, there is little that one can conclude about the chemo-preventive nature of these agents and their ability to prolong survival and improve quality of life [65].
Data from a large, long-term Randomized Control Trial (RCT ARI40005) raised a serious concern regarding the safety of 5α-RIs, dutasteride, because of significant increase in the risk of "cardiac failure" [13,66,67]. As pointed out by Justman [61] some of the unidentified risks of 5α-RIs may be attributed to the limitations of the clinical trials. For example, these trials had no primary or secondary endpoints to detect cardiovascular events; therefore, some of the events may have been overlooked or underestimated. Moreover, since cardiovascular events were not investigated as part of the primary or secondary endpoint, clinical trial investigators may have had different interpretations and may have not given them serious consideration [16,61,67].
Several studies have attempted to evaluate the efficacy of 5α-RIs in reducing tumor growth in the animal models. Interestingly, the study by Umekita et al. [68] showed that various PCa tumor cell lines respond differently to androgen treatment. For instance, the tumor cell line, LNCaP 104-R2, when implanted in castrated, nude, male mice continued to grow. However, treatment with TP resulted in tumor regression not tumor growth. Remarkably, treatment with TP together with finasteride resulted in increased tumor growth. These findings suggest that ADT may not necessarily be beneficial for treatment of all forms of PCa and that androgen replacement therapy may be a potential treatment for some forms but not all forms of PCa [68]. Recently, several small clinical trials have reported using testosterone therapy in patients with localized PCa, after radical prostatectomy or brachytherapy. These studies found positive results based on lack of biochemical recurrence in the treatment arms [69].
Since PCa is a heterogeneous disease, different tumors possess different metabolic properties. In a study using androgen-insensitive PCa cell lines in an animal model, it was reported that the 5α-RIs, finasteride and dutasteride, did not inhibit tumor incidence or tumor growth suggesting the limited potential benefits of 5α-RIs as chemo-preventive agents in all forms of PCa.
It should be noted that 5α-RIs alter cellular biology with uncertain outcomes. For example, treatment of animals with 5α-RIs resulted in marked increase in the expression of the AR [70]. The implication of such dysfunctional metabolism may contribute to loss of androgen dependence and to acquisition of high-grade tumors. In addition, Yun et al. [71] postulated that finasteride increases expression of hemoxygenase-1 and other related factors in PCa cell lines (PC-3). The authors suggested that finasteride-induced alteration in gene expression may be in part responsible for finasteride-induced high grade prostate tumors [71]. It is not surprising that Theoret et al. [15] reported a significantly number of high grade tumors (Fig. 4), with finasteride and dutasteride in the data from the PCPT [14] and REDUCE trials [13] respectively, even after using the revised Gleason scoring system [15].
FIG. 4.
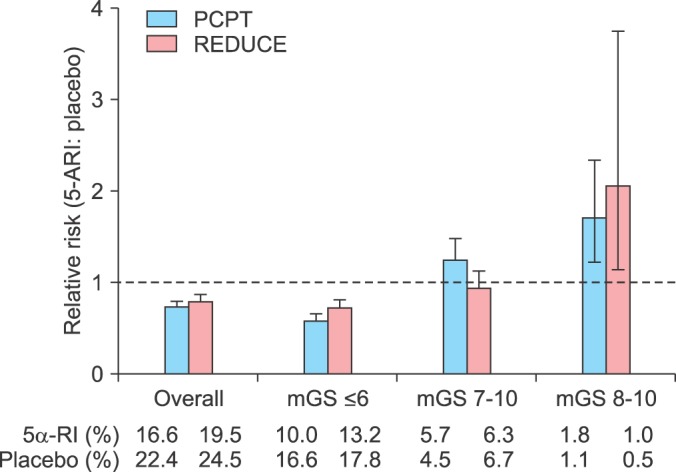
Relative and absolute risk of prostate cancer according to modified Gleason score (mGS), PCPT and REDUCE trial. 5α-RI, 5α-reductase inhibitors. I bars indicate 95% confidence intervals. Adapted from Theoret MR, et al. N Engl J Med 2011;365:97-9, with permission of Massachusetts Medical Society [15].
The overwhelmingly negative decision by the FDA panel of scientists and medical experts [16] speaks volumes and raises a significant concern about the safety and the efficacy of these drugs as chemo-preventive agents for PCa, given that they elicit many serious adverse side effects [16]. With the postmarketing increase in reported adverse events, the FDA has mandated a revision to the labeling for all 5α-RIs [16]. The labeling changes included a warning of increased sexual dysfunction, depression, and increased risk of high-grade PCa.
3. Potential adverse effects of 5α-R therapy on the CNS
The CNS has the capacity and the machinery to synthesize a host of neurosteroids [72]. Neurosteroids play a pivotal role in modulating neural activity through interaction with neurotransmitter receptors and neurotransmitter-gaited ion-channels [73]. These neurosteroids interact with a host of neurotransmitter receptors and modulate seizure susceptibility, anxiety, stress, and depression [74]. 5α-R reaction is the rate limiting step in the conversion of testosterone, progesterone, cortisol, corticosterone, and DOC into their respective 5α-dihydro-deratitves, which serve as precursors for 3α-hydroxysteroid dehydrogenase which transfroms such precursors into their respective neurosteroids (androstanediol, allopregnanolone [AP], tetrahydrocortisol, tetrahdyrocorticosterone, and tetrahydrodeoxycorticosterone) (Fig. 1) [1,2]. All three isoforms of 5α-R are expressed in the various regions of brain and are thought to be critical for brain development since fetal brain express high concentrations of 5α-R [1,2].
It has recently been shown that patients who had been treated with finasteride have reduced or undetectable levels of neuroactive steroids in their cerebro-spinal fluid and plasma, and exhibited higher levels of precursor steroids [75]. This observation strongly suggests that 5α-RIs have a deleterious effect on the biosynthesis and function of neurosteroids in the central nervous system. Finasteride treatment resulted in decreased levels of 5α-DHT and 3α, 5α-tetrahydroprogesterone (AP) and increased levels of testosterone supporting the hypothesis that deleterious effects of finasteride may be persistent or irreversible. This may explain some of the noted symptoms such as anxiety, depression and suicide in patients who have been treated with finasteride [76].
NEUROPROTECTIVE EFFECTS OF NEUROSTEROIDS
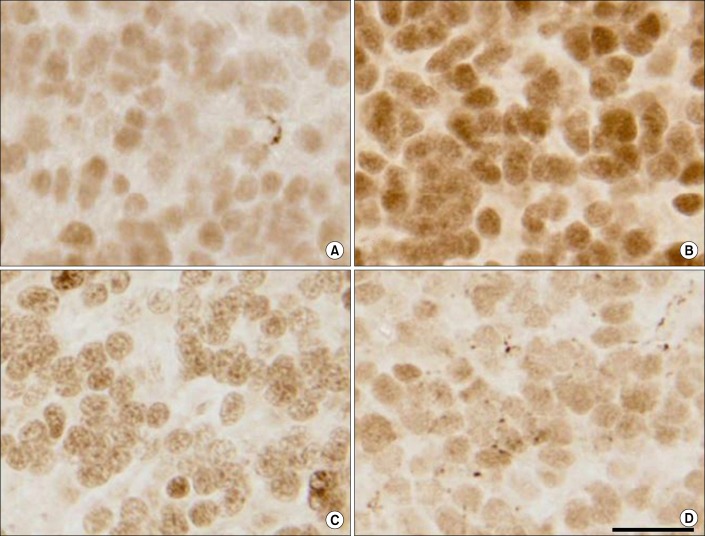
Neuroactive steroids elicit important neuroprotective effects during trauma and injury to the central nervous system [75]. AP is shown to be beneficial in the treatment of traumatic brain injury, attenuating edema, trauma, stress, inflammation, apoptosis, and reducing oxidative stress [76,77,78]. AP is not only a protective agent in ischemia, but also in maintaining blood brain barrier integrity, and in memory and learning [78,79,80,81]. Studies on CNS injury in which asphyxiation was induced in fetal sheep to stimulate neurological stressors, in the presence or absence of finasteride, showed an increase in apoptotic cell death in the cerebellum and hippocampus in the animals treated with finasteride (Fig. 5) [82]. Furthermore, treatment with an AP analogue, alfaxalone, prevented cell death as assessed by the increase in activity of caspace-3 and expression of ki-67 protein. This observation suggests that AP exerts a neuroprotective role in the brain, which is inhibited by finasteride.
FIG. 5.
Effects of finasteride on the number of activated capsase-3 positive cells present in the cerebellar molecular and granular layers. Photomicrographs showing activated caspase-3 immunoreactivity in the granular layer of the cerebellum of a fetus at 24 hours after infusion with vehicle (control, A), finasteride (B), finasteride+alfaxalone (C), and alfaxalone (D). All fetuses were 131±3 days of gestation at the time of autopsy and tissue collection. (A-D: Scale bar, 10 µm). Color was visualized using streptavidin horseradish peroxidase conjugated to diaminobenzidine. Adopted from Yawno T, et al. Neuroscience 2009;163:838-47, with permission of Elsevier Inc. [82].
The neuroprotective effect of AP was further illustrated during injury to the rat hippocampus slices induced by tributyltin treatment, which resulted in significant cell death. Administration of progesterone (P) with finasteride showed similar cell death to that induced by tributyltin treatment, in the various regions of the hippocampus. In contrast, P treatment without finasteride provided a protective effect. This is attributed to the conversion of P to 5α-DHP by 5α-Rs and to AP by 3α-HSD. To confirm that this is due to the neuroprotective effect of AP, the latter was administered with or without finasteride. While the tributyltin induced cell death was significant, administration of AP with and without finasteride produced a markedly protective effect as assessed by the reduced cell death [83]. These findings suggest that 5α-Rs play a pivotal role in neuroprotection.
Neurosteroid synthesis in the hippocampus is suggested to be critical for neuroplasticity in the brain [84]. Inhibition of 5α-R by finasteride is thought to contribute to reduced neuroplasticity due to structural changes resulting from inhibition of neurogenesis in the hippocampus. Finasteride treatment in mice showed decreased cell proliferation in the hippocampus, suggesting that inhibitors of 5α-R blocks neurogenesis [84].
As reported by Mellon [85] in an adult animal model of Niemann-Pick disease type C the biosynthesis of AP is significantly reduced, and this was supported by significant reduction in the activity of 5α-R and 3α-HSD in the brain of these animals [85]. The activity of 5α-R was markedly reduced with the progression of the disease. This reduced 5α-R activity may contribute to the observed accumulation of cholesterol in neurons and gangliosides, cerebro, perkinje cell degeneration, dysmyelination and neurodegeneration [85]. Thus, loss of 5α-R and 3α-HSD activity is attributed to loss of neurons expressing these enzymes. Treatment with AP early at postanatal day 7 was found most effective in preventing neurodegeneration and correlates with reduced tremor, ataxia, and increased lifespan. In the animal model, these findings indicate that the loss of neurosteroid biosynthesis may be responsible for the disease state and its progression. Therefore inhibition of 5α-R by finasteride and dutasteride in the course of treatment of nonlife threatening conditions, such as male pattern baldness (alopecia) or BPH may have detrimental effects on the CNS.
It has been reported that AP levels were significantly decreased in postmortem human brains of Alzheimer disease (AD) patients [86]. An inverse correlation was noted between AP levels and the degree of neurological degeneration in pathological section of AD patient [86]. One of the interesting findings was that pregnenolone levels were greater in the temporal cortex of AD patients suggesting that this may be a compensating mechanism for reduced 5α-R activity. We speculate that 5α-RIs may contribute to reduced levels of neurosteroids in the CNS and this may enhance the progression of neurodegenerative disease, such as AD.
Conversion of polyprenol into dolichol is critical for N-glycosylation of membrane proteins [3]. 5α-R type 3 was shown to be critical for catalyzing this reaction [3]. A mutation in this enzyme lead to mental retardation and opthamologic and cerebral defects [3]. Intraoperative Floppy Iris Syndrome (IFIS) is also a complication experienced during cataract extraction. The syndrome is defined by floppy, or flaccid iris. Studies have indicated a correlation between finasteride and the occurrence of cataracts and IFIS indicating inhibition of glycosylation may contribute to this pathology [87].
EPILEPSY/CONVULSION
P is an effective anticonvulsing agent in humans [88]. The anticonvulsive properties of P diminished when animals were treated with finasteride. In a mouse model of pentylenetetrazol-induced seizures, there was an approximately 50% decrease in the protective effect of P in mice when treated with finasteride [89]. Higher dosages of finasteride produced more persistent symptoms of pentylenetetrazol-induced seizures [89]. However, when AP was administered together with finasteride, the pentylenetetrazol seizure activity was reversed. This finding indicates that the antiseizure properties of P are attributed to its metabolite AP emphasizing the critical role of 5α-Rs [89,90,91].
DEPRESSION
Anxiety is often found as a comorbidity of depression. The administration of AP produces antidepressant and anxiolytic effects [92,93]. Coadministration of finasteride and P blocked P's anxiolytic effect [94]. This finding suggests that a metabolite of progesterone is responsible for the anxiety reducing effect of P. An inverse relationship between levels of AP and depression has been demonstrated in male patients with depression [95]. Preclinical studies have suggested that reduction in AP levels by 5α-RIs may contribute to depressive symptoms [96]. Increased depressive symptoms are thought to be linked to finasteride treatment [97]. A statistically significant correlation was observed between use of finasteride and depressive symptoms [98]. Persistent side effects have been noted even after discontinuation of finasteride treatment [76] from 3 months to 11 years, suggesting that the adverse effects of finasteride may be permanent [97].
SUMMARY
We wish to point out from the outset that 5α-RI therapy was FDA-approved for treatment for LUTS related to BPH. Treatment of symptomatic men with finasteride or dutasteride results in approximately 17%-19% reduction in prostate volume, thus alleviating urinary tract symptoms [8,13,14,53]. Furthermore, 5α-RI therapy reduces urinary tract infection and minimizes the risk of potential acute urinary retention and the need for surgical intervention related to BPH. These positive effects of finasteride and dutasteride contribute to improvement in the quality of life in men suffering from BPH symptoms [8,53]. However, a substantial body of evidence exists which points to serious and potentially ill-health effects of 5α-RIs' therapy. These include loss or reduced libido, erectile dysfunction, orgasmic and ejaculatory dysfunction (Table 2) [99], development of high grade PCa tumors (Fig. 4), potential negative cardiovascular events, and depression. The side effects are potentially harmful in some individuals and in young men may be persistent or irreversible [100]. The argument that the benefits of these drugs outweighs the risks is slowly eroding in the face of new emerging scientific evidence from preclinical (Figs. 2, 3; Table 1) and clinical studies (Table 2). The available data demonstrate that such drugs do pose serious adverse effects, especially in a subset of men who may have the predisposition to be affected severely. This is also corroborated by the overwhelming FDA disapproval of these drugs for the chemoprevention of PCa. The FDA mandated relabeling issued in 2011 equally points to the dark side of these drugs on human health. Physicians need to be aware of the adverse side effects of these drugs and are encouraged to share this information with their patients prior to commencing therapy with finasteride or dutasteride.
Footnotes
The authors have nothing to disclose.
References
- 1.Traish AM. 5α-reductases in human physiology: an unfolding story. Endocr Pract. 2012;18:965–975. doi: 10.4158/EP12108.RA. [DOI] [PubMed] [Google Scholar]
- 2.Traish AM, Hassani J, Guay AT, Zitzmann M, Hansen ML. Adverse side effects of 5α-reductase inhibitors therapy: persistent diminished libido and erectile dysfunction and depression in a subset of patients. J Sex Med. 2011;8:872–884. doi: 10.1111/j.1743-6109.2010.02157.x. [DOI] [PubMed] [Google Scholar]
- 3.Cantagrel V, Lefeber DJ, Ng BG, Guan Z, Silhavy JL, Bielas SL, et al. SRD5A3 is required for converting polyprenol to dolichol and is mutated in a congenital glycosylation disorder. Cell. 2010;142:203–217. doi: 10.1016/j.cell.2010.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stiles AR, Russell DW. SRD5A3: a surprising role in glycosylation. Cell. 2010;142:196–198. doi: 10.1016/j.cell.2010.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Morgentaler A, Traish AM. Shifting the paradigm of testosterone and prostate cancer: the saturation model and the limits of androgen-dependent growth. Eur Urol. 2009;55:310–320. doi: 10.1016/j.eururo.2008.09.024. [DOI] [PubMed] [Google Scholar]
- 6.Traish AM, Morgentaler A. Epidermal growth factor receptor expression escapes androgen regulation in prostate cancer: a potential molecular switch for tumour growth. Br J Cancer. 2009;101:1949–1956. doi: 10.1038/sj.bjc.6605376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Traish AM, Wotiz HH. Prostatic epidermal growth factor receptors and their regulation by androgens. Endocrinology. 1987;121:1461–1467. doi: 10.1210/endo-121-4-1461. [DOI] [PubMed] [Google Scholar]
- 8.Bechis SK, Otsetov AG, Ge R, Olumi AF. Personalized medicine for the management of benign prostatic hyperplasia. J Urol. 2014 Feb 25; doi: 10.1016/j.juro.2014.01.114. [Epub]. http://dx.doi.org/10.1016/j.juro.2014.01.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Erdemir F, Harbin A, Hellstrom WJ. 5-alpha reductase inhibitors and erectile dysfunction: the connection. J Sex Med. 2008;5:2917–2924. doi: 10.1111/j.1743-6109.2008.01001.x. [DOI] [PubMed] [Google Scholar]
- 10.Gur S, Kadowitz PJ, Hellstrom WJ. Effects of 5-alpha reductase inhibitors on erectile function, sexual desire and ejaculation. Expert Opin Drug Saf. 2013;12:81–90. doi: 10.1517/14740338.2013.742885. [DOI] [PubMed] [Google Scholar]
- 11.Irwig MS, Kolukula S. Persistent sexual side effects of finasteride for male pattern hair loss. J Sex Med. 2011;8:1747–1753. doi: 10.1111/j.1743-6109.2011.02255.x. [DOI] [PubMed] [Google Scholar]
- 12.Romer B, Gass P. Finasteride-induced depression: new insights into possible pathomechanisms. J Cosmet Dermatol. 2010;9:331–332. doi: 10.1111/j.1473-2165.2010.00533.x. [DOI] [PubMed] [Google Scholar]
- 13.Andriole GL, Bostwick DG, Brawley OW, Gomella LG, Marberger M, Montorsi F, et al. Effect of dutasteride on the risk of prostate cancer. N Engl J Med. 2010;362:1192–1202. doi: 10.1056/NEJMoa0908127. [DOI] [PubMed] [Google Scholar]
- 14.Thompson IM, Goodman PJ, Tangen CM, Lucia MS, Miller GJ, Ford LG, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003;349:215–224. doi: 10.1056/NEJMoa030660. [DOI] [PubMed] [Google Scholar]
- 15.Theoret MR, Ning YM, Zhang JJ, Justice R, Keegan P, Pazdur R. The risks and benefits of 5α-reductase inhibitors for prostate-cancer prevention. N Engl J Med. 2011;365:97–99. doi: 10.1056/NEJMp1106783. [DOI] [PubMed] [Google Scholar]
- 16.U.S. Food and Drug Administration. 2010 Meeting Materials, Oncologic Drugs Advisory Committee [Internet] Silver Spring: U.S. Food and Drug Administration; c2014. [cited 2010 Dec 1]. Available from: http://www.fda.gov/advisorycommittees/committeesmeetingmaterials/drugs/oncologicdrugsadvisorycommittee/ucm195226.htm. [Google Scholar]
- 17.Thompson IM, Jr, Goodman PJ, Tangen CM, Parnes HL, Minasian LM, Godley PA, et al. Long-term survival of participants in the prostate cancer prevention trial. N Engl J Med. 2013;369:603–610. doi: 10.1056/NEJMoa1215932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Irwig MS. Persistent sexual side effects of finasteride: could they be permanent? J Sex Med. 2012;9:2927–2932. doi: 10.1111/j.1743-6109.2012.02846.x. [DOI] [PubMed] [Google Scholar]
- 19.Wessells H, Roy J, Bannow J, Grayhack J, Matsumoto AM, Tenover L, et al. Incidence and severity of sexual adverse experiences in finasteride and placebo-treated men with benign prostatic hyperplasia. Urology. 2003;61:579–584. doi: 10.1016/s0090-4295(02)02401-9. [DOI] [PubMed] [Google Scholar]
- 20.Baldinotti F, Majore S, Fogli A, Marrocco G, Ghirri P, Vuerich M, et al. Molecular characterization of 6 unrelated Italian patients with 5alpha-reductase type 2 deficiency. J Androl. 2008;29:20–28. doi: 10.2164/jandrol.107.002592. [DOI] [PubMed] [Google Scholar]
- 21.MacLaughlin DT, Donahoe PK. Sex determination and differentiation. N Engl J Med. 2004;350:367–378. doi: 10.1056/NEJMra022784. [DOI] [PubMed] [Google Scholar]
- 22.Mainwaring WI. The mechanism of action of androgens. Monogr Endocrinol. 1977;10:1–178. [PubMed] [Google Scholar]
- 23.Bradshaw WG, Baum MJ, Awh CC. Attenuation by a 5 alpha-reductase inhibitor of the activational effect of testosterone propionate on penile erections in castrated male rats. Endocrinology. 1981;109:1047–1051. doi: 10.1210/endo-109-4-1047. [DOI] [PubMed] [Google Scholar]
- 24.Gray GD, Smith ER, Davidson JM. Hormonal regulation of penile erection in castrated male rats. Physiol Behav. 1980;24:463–468. doi: 10.1016/0031-9384(80)90237-1. [DOI] [PubMed] [Google Scholar]
- 25.Hart BL. Effects of testosterone propionate and dihydrotestosterone on penile morphology and sexual reflexes of spinal male rats. Horm Behav. 1973;4:239–246. doi: 10.1016/0018-506x(73)90008-1. [DOI] [PubMed] [Google Scholar]
- 26.Hart BL. Activation of sexual reflexes of male rats by dihydrotestosterone but not estrogen. Physiol Behav. 1979;23:107–109. doi: 10.1016/0031-9384(79)90129-x. [DOI] [PubMed] [Google Scholar]
- 27.Mantzoros CS, Georgiadis EI, Trichopoulos D. Contribution of dihydrotestosterone to male sexual behaviour. BMJ. 1995;310:1289–1291. doi: 10.1136/bmj.310.6990.1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Saksena SK, Lau IF, Chang MC. The inhibition of the conversion of testosterone into 5alpha-dihydrotestosterone in the reproductive organs of the male rat. Steroids. 1976;27:751–757. doi: 10.1016/0039-128x(76)90135-5. [DOI] [PubMed] [Google Scholar]
- 29.Baum MJ. A comparison of the effects of methyltrienolone (R1881) and 5 alpha-dihydrotestosterone on sexual behavior of castrated male rats. Horm Behav. 1979;13:165–174. doi: 10.1016/0018-506x(79)90055-2. [DOI] [PubMed] [Google Scholar]
- 30.Lugg JA, Rajfer J, Gonzalez-Cadavid NF. Dihydrotestosterone is the active androgen in the maintenance of nitric oxide-mediated penile erection in the rat. Endocrinology. 1995;136:1495–1501. doi: 10.1210/endo.136.4.7534702. [DOI] [PubMed] [Google Scholar]
- 31.Penson DF, Ng C, Rajfer J, Gonzalez-Cadavid NF. Adrenal control of erectile function and nitric oxide synthase in the rat penis. Endocrinology. 1997;138:3925–3932. doi: 10.1210/endo.138.9.5402. [DOI] [PubMed] [Google Scholar]
- 32.Garban H, Marquez D, Cai L, Rajfer J, Gonzalez-Cadavid NF. Restoration of normal adult penile erectile response in aged rats by long-term treatment with androgens. Biol Reprod. 1995;53:1365–1372. doi: 10.1095/biolreprod53.6.1365. [DOI] [PubMed] [Google Scholar]
- 33.Park KH, Kim SW, Kim KD, Paick JS. Effects of androgens on the expression of nitric oxide synthase mRNAs in rat corpus cavernosum. BJU Int. 1999;83:327–333. doi: 10.1046/j.1464-410x.1999.00913.x. [DOI] [PubMed] [Google Scholar]
- 34.Bialy M, Sachs BD. Androgen implants in medial amygdala briefly maintain noncontact erection in castrated male rats. Horm Behav. 2002;42:345–355. doi: 10.1006/hbeh.2002.1821. [DOI] [PubMed] [Google Scholar]
- 35.Manzo J, Cruz MR, Hernandez ME, Pacheco P, Sachs BD. Regulation of noncontact erection in rats by gonadal steroids. Horm Behav. 1999;35:264–270. doi: 10.1006/hbeh.1999.1519. [DOI] [PubMed] [Google Scholar]
- 36.Sachs BD. Placing erection in context: the reflexogenic-psychogenic dichotomy reconsidered. Neurosci Biobehav Rev. 1995;19:211–224. doi: 10.1016/0149-7634(94)00063-7. [DOI] [PubMed] [Google Scholar]
- 37.Seo SI, Kim SW, Paick JS. The effects of androgen on penile reflex, erectile response to electrical stimulation and penile NOS activity in the rat. Asian J Androl. 1999;1:169–174. [PubMed] [Google Scholar]
- 38.Oztekin CV, Gur S, Abdulkadir NA, Lokman U, Akdemir AO, Cetinkaya M, et al. Incomplete recovery of erectile function in rat after discontinuation of dual 5-alpha reductase inhibitor therapy. J Sex Med. 2012;9:1773–1781. doi: 10.1111/j.1743-6109.2012.02759.x. [DOI] [PubMed] [Google Scholar]
- 39.Pinsky MR, Gur S, Tracey AJ, Harbin A, Hellstrom WJ. The effects of chronic 5-alpha-reductase inhibitor (dutasteride) treatment on rat erectile function. J Sex Med. 2011;8:3066–3074. doi: 10.1111/j.1743-6109.2011.02425.x. [DOI] [PubMed] [Google Scholar]
- 40.Zhang MG, Wang XJ, Shen ZJ, Gao PJ. Long-term oral administration of 5α-reductase inhibitor attenuates erectile function by inhibiting autophagy and promoting apoptosis of smooth muscle cells in corpus cavernosum of aged rats. Urology. 2013;82:743.e9–743.e15. doi: 10.1016/j.urology.2013.02.045. [DOI] [PubMed] [Google Scholar]
- 41.Traish AM, Park K, Dhir V, Kim NN, Moreland RB, Goldstein I. Effects of castration and androgen replacement on erectile function in a rabbit model. Endocrinology. 1999;140:1861–1868. doi: 10.1210/endo.140.4.6655. [DOI] [PubMed] [Google Scholar]
- 42.Nickel JC, Fradet Y, Boake RC, Pommerville PJ, Perreault JP, Afridi SK, et al. Efficacy and safety of finasteride therapy for benign prostatic hyperplasia: results of a 2-year randomized controlled trial (the PROSPECT study). PROscar Safety Plus Efficacy Canadian Two year Study. CMAJ. 1996;155:1251–1259. [PMC free article] [PubMed] [Google Scholar]
- 43.Tenover JL, Pagano GA, Morton AS, Liss CL, Byrnes CA Primary Care Investigator Study Group. Efficacy and tolerability of finasteride in symptomatic benign prostatic hyperplasia: a primary care study. Clin Ther. 1997;19:243–258. doi: 10.1016/s0149-2918(97)80113-0. [DOI] [PubMed] [Google Scholar]
- 44.Hudson PB, Boake R, Trachtenberg J, Romas NA, Rosenblatt S, Narayan P, et al. The North American Finasteride Study Group. Efficacy of finasteride is maintained in patients with benign prostatic hyperplasia treated for 5 years. Urology. 1999;53:690–695. doi: 10.1016/s0090-4295(98)00666-9. [DOI] [PubMed] [Google Scholar]
- 45.Bruskewitz R, Girman CJ, Fowler J, Rigby OF, Sullivan M, Bracken RB, et al. Effect of finasteride on bother and other health-related quality of life aspects associated with benign prostatic hyperplasia. PLESS Study Group. Proscar Long-term Efficacy and Safety Study. Urology. 1999;54:670–678. doi: 10.1016/s0090-4295(99)00209-5. [DOI] [PubMed] [Google Scholar]
- 46.Wilton L, Pearce G, Edet E, Freemantle S, Stephens MD, Mann RD. The safety of finasteride used in benign prostatic hypertrophy: a non-interventional observational cohort study in 14,772 patients. Br J Urol. 1996;78:379–384. doi: 10.1046/j.1464-410x.1996.00091.x. [DOI] [PubMed] [Google Scholar]
- 47.AUA Practice Guidelines Committee. AUA guideline on management of benign prostatic hyperplasia (2003). Chapter 1: Diagnosis and treatment recommendations. J Urol. 2003;170(2 Pt 1):530–547. doi: 10.1097/01.ju.0000078083.38675.79. [DOI] [PubMed] [Google Scholar]
- 48.Edwards JE, Moore RA. Finasteride in the treatment of clinical benign prostatic hyperplasia: a systematic review of randomised trials. BMC Urol. 2002;2:14. doi: 10.1186/1471-2490-2-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Roehrborn CG, Siami P, Barkin J, Damiao R, Major-Walker K, Morrill B, et al. The effects of dutasteride, tamsulosin and combination therapy on lower urinary tract symptoms in men with benign prostatic hyperplasia and prostatic enlargement: 2-year results from the CombAT study. J Urol. 2008;179:616–621. doi: 10.1016/j.juro.2007.09.084. [DOI] [PubMed] [Google Scholar]
- 50.Siami P, Roehrborn CG, Barkin J, Damiao R, Wyczolkowski M, Duggan A, et al. Combination therapy with dutasteride and tamsulosin in men with moderate-to-severe benign prostatic hyperplasia and prostate enlargement: the CombAT (Combination of Avodart and Tamsulosin) trial rationale and study design. Contemp Clin Trials. 2007;28:770–779. doi: 10.1016/j.cct.2007.07.008. [DOI] [PubMed] [Google Scholar]
- 51.Desgrandchamps F, Droupy S, Irani J, Saussine C, Comenducci A. Effect of dutasteride on the symptoms of benign prostatic hyperplasia, and patient quality of life and discomfort, in clinical practice. BJU Int. 2006;98:83–88. doi: 10.1111/j.1464-410X.2006.06241.x. [DOI] [PubMed] [Google Scholar]
- 52.Canguven O, Burnett AL. The effect of 5 alpha-reductase inhibitors on erectile function. J Androl. 2008;29:514–523. doi: 10.2164/jandrol.108.005025. [DOI] [PubMed] [Google Scholar]
- 53.Kaplan SA, Chung DE, Lee RK, Scofield S, Te AE. A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has comparable urinary symptom efficacy and prostate volume reduction, but less sexual side effects and breast complications than dutasteride. Int J Clin Pract. 2012;66:1052–1055. doi: 10.1111/j.1742-1241.2012.03010.x. [DOI] [PubMed] [Google Scholar]
- 54.Chi BH, Kim SC. Changes in sexual function in benign prostatic hyperplasia patients taking dutasteride: 1-year follow-up results. Korean J Urol. 2011;52:632–636. doi: 10.4111/kju.2011.52.9.632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Fwu CW, Eggers PW, Kirkali Z, McVary KT, Burrows PK, Kusek JW. Change in sexual function in men with lower urinary tract symptoms/benign prostatic hyperplasia associated with long-term treatment with doxazosin, finasteride and combined therapy. J Urol. 2013 Dec 14; doi: 10.1016/j.juro.2013.12.014. [Epub]. http://dx.doi.org/10.1016/j.juro.2013.12.014. [DOI] [PubMed] [Google Scholar]
- 56.Huggins C, Hodges CV. Studies on prostatic cancer. I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. 1941. J Urol. 2002;167(2 Pt 2):948–951. [PubMed] [Google Scholar]
- 57.Olsson M, Ekstrom L, Guillemette C, Belanger A, Rane A, Gustafsson O. Correlation between circulatory, local prostatic, and intra-prostatic androgen levels. Prostate. 2011;71:909–914. doi: 10.1002/pros.21307. [DOI] [PubMed] [Google Scholar]
- 58.Morgentaler A. Goodbye androgen hypothesis, hello saturation model. Eur Urol. 2012;62:765–767. doi: 10.1016/j.eururo.2012.06.027. [DOI] [PubMed] [Google Scholar]
- 59.Muller RL, Gerber L, Moreira DM, Andriole G, Castro-Santamaria R, Freedland SJ. Serum testosterone and dihydrotestosterone and prostate cancer risk in the placebo arm of the Reduction by Dutasteride of Prostate Cancer Events trial. Eur Urol. 2012;62:757–764. doi: 10.1016/j.eururo.2012.05.025. [DOI] [PubMed] [Google Scholar]
- 60.Crawford ED, Fair WR, Kelloff GJ, Lieber MM, Miller GJ, Scardíno PT, et al. Chemoprevention of prostate cancer: guidelines for possible intervention strategies. J Cell Biochem Suppl. 1992;16H:140–145. doi: 10.1002/jcb.240501232. [DOI] [PubMed] [Google Scholar]
- 61.Justman S. What's wrong with chemoprevention of prostate cancer? Am J Bioeth. 2011;11:21–25. doi: 10.1080/15265161.2011.623932. [DOI] [PubMed] [Google Scholar]
- 62.Walsh PC. Three considerations before advising 5-alpha-reductase inhibitors for chemoprevention. J Clin Oncol. 2009;27:e22. doi: 10.1200/JCO.2009.22.9070. author reply e23. [DOI] [PubMed] [Google Scholar]
- 63.Walsh PC. Chemoprevention of prostate cancer. N Engl J Med. 2010;362:1237–1238. doi: 10.1056/NEJMe1001045. [DOI] [PubMed] [Google Scholar]
- 64.Ehdaie B, Touijer KA. 5-alpha Reductase inhibitors in prostate cancer: from clinical trials to clinical practice. Eur Urol. 2013;63:788–789. doi: 10.1016/j.eururo.2012.12.035. [DOI] [PubMed] [Google Scholar]
- 65.Hoffman RM, Roberts RG, Barry MJ. Battling prostate cancer with 5-alpha-reductase inhibitors: a pyrrhic victory? J Gen Intern Med. 2011;26:798–801. doi: 10.1007/s11606-010-1622-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Andriole GL, Kirby R. Safety and tolerability of the dual 5alpha-reductase inhibitor dutasteride in the treatment of benign prostatic hyperplasia. Eur Urol. 2003;44:82–88. doi: 10.1016/s0302-2838(03)00198-2. [DOI] [PubMed] [Google Scholar]
- 67.Roehrborn CG, Andriole GL, Wilson TH, Castro R, Rittmaster RS. Effect of dutasteride on prostate biopsy rates and the diagnosis of prostate cancer in men with lower urinary tract symptoms and enlarged prostates in the Combination of Avodart and Tamsulosin trial. Eur Urol. 2011;59:244–249. doi: 10.1016/j.eururo.2010.10.040. [DOI] [PubMed] [Google Scholar]
- 68.Umekita Y, Hiipakka RA, Kokontis JM, Liao S. Human prostate tumor growth in athymic mice: inhibition by androgens and stimulation by finasteride. Proc Natl Acad Sci U S A. 1996;93:11802–11807. doi: 10.1073/pnas.93.21.11802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Khera M, Crawford D, Morales A, Salonia A, Morgentaler A. A new era of testosterone and prostate cancer: from physiology to clinical implications. Eur Urol. 2014;65:115–123. doi: 10.1016/j.eururo.2013.08.015. [DOI] [PubMed] [Google Scholar]
- 70.Hsieh JT, Chen SC, Yu HJ, Chang HC. Finasteride upregulates expression of androgen receptor in hyperplastic prostate and LNCaP cells: implications for chemoprevention of prostate cancer. Prostate. 2011;71:1115–1121. doi: 10.1002/pros.21325. [DOI] [PubMed] [Google Scholar]
- 71.Yun DK, Lee J, Keum YS. Finasteride increases the expression of hemoxygenase-1 (HO-1) and NF-E2-related factor-2 (Nrf2) proteins in PC-3 cells: implication of finasteride-mediated high-grade prostate tumor occurrence. Biomol Ther (Seoul) 2013;21:49–53. doi: 10.4062/biomolther.2012.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mensah-Nyagan AG, Do-Rego JL, Beaujean D, Luu-The V, Pelletier G, Vaudry H. Neurosteroids: expression of steroidogenic enzymes and regulation of steroid biosynthesis in the central nervous system. Pharmacol Rev. 1999;51:63–81. [PubMed] [Google Scholar]
- 73.Akk G, Covey DF, Evers AS, Steinbach JH, Zorumski CF, Mennerick S. Mechanisms of neurosteroid interactions with GABA(A) receptors. Pharmacol Ther. 2007;116:35–57. doi: 10.1016/j.pharmthera.2007.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Reddy DS. Role of anticonvulsant and antiepileptogenic neurosteroids in the pathophysiology and treatment of epilepsy. Front Endocrinol (Lausanne) 2011;2:38. doi: 10.3389/fendo.2011.00038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Melcangi RC, Panzica GC. Allopregnanolone: state of the art. Prog Neurobiol. 2014;113:1–5. doi: 10.1016/j.pneurobio.2013.09.005. [DOI] [PubMed] [Google Scholar]
- 76.Melcangi RC, Caruso D, Abbiati F, Giatti S, Calabrese D, Piazza F, et al. Neuroactive steroid levels are modified in cerebrospinal fluid and plasma of post-finasteride patients showing persistent sexual side effects and anxious/depressive symptomatology. J Sex Med. 2013;10:2598–2603. doi: 10.1111/jsm.12269. [DOI] [PubMed] [Google Scholar]
- 77.He J, Evans CO, Hoffman SW, Oyesiku NM, Stein DG. Progesterone and allopregnanolone reduce inflammatory cytokines after traumatic brain injury. Exp Neurol. 2004;189:404–412. doi: 10.1016/j.expneurol.2004.06.008. [DOI] [PubMed] [Google Scholar]
- 78.VanLandingham JW, Cutler SM, Virmani S, Hoffman SW, Covey DF, Krishnan K, et al. The enantiomer of progesterone acts as a molecular neuroprotectant after traumatic brain injury. Neuropharmacology. 2006;51:1078–1085. doi: 10.1016/j.neuropharm.2006.07.015. [DOI] [PubMed] [Google Scholar]
- 79.Ishrat T, Sayeed I, Atif F, Hua F, Stein DG. Progesterone and allopregnanolone attenuate blood-brain barrier dysfunction following permanent focal ischemia by regulating the expression of matrix metalloproteinases. Exp Neurol. 2010;226:183–190. doi: 10.1016/j.expneurol.2010.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Moralí G, Montes P, González-Burgos I, Velázquez-Zamora DA, Cervantes M. Cytoarchitectural characteristics of hippocampal CA1 pyramidal neurons of rats, four months after global cerebral ischemia and progesterone treatment. Restor Neurol Neurosci. 2012;30:1–8. doi: 10.3233/RNN-2011-0605. [DOI] [PubMed] [Google Scholar]
- 81.Sayeed I, Guo Q, Hoffman SW, Stein DG. Allopregnanolone, a progesterone metabolite, is more effective than progesterone in reducing cortical infarct volume after transient middle cerebral artery occlusion. Ann Emerg Med. 2006;47:381–389. doi: 10.1016/j.annemergmed.2005.12.011. [DOI] [PubMed] [Google Scholar]
- 82.Yawno T, Yan EB, Walker DW, Hirst JJ. Inhibition of neurosteroid synthesis increases asphyxia-induced brain injury in the late gestation fetal sheep. Neuroscience. 2007;146:1726–1733. doi: 10.1016/j.neuroscience.2007.03.023. [DOI] [PubMed] [Google Scholar]
- 83.Ishihara Y, Kawami T, Ishida A, Yamazaki T. Allopregnanolone-mediated protective effects of progesterone on tributyltin-induced neuronal injury in rat hippocampal slices. J Steroid Biochem Mol Biol. 2013;135:1–6. doi: 10.1016/j.jsbmb.2012.12.013. [DOI] [PubMed] [Google Scholar]
- 84.Romer B, Pfeiffer N, Lewicka S, Ben-Abdallah N, Vogt MA, Deuschle M, et al. Finasteride treatment inhibits adult hippocampal neurogenesis in male mice. Pharmacopsychiatry. 2010;43:174–178. doi: 10.1055/s-0030-1249095. [DOI] [PubMed] [Google Scholar]
- 85.Mellon SH. Neurosteroid regulation of central nervous system development. Pharmacol Ther. 2007;116:107–124. doi: 10.1016/j.pharmthera.2007.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Naylor JC, Kilts JD, Hulette CM, Steffens DC, Blazer DG, Ervin JF, et al. Allopregnanolone levels are reduced in temporal cortex in patients with Alzheimer's disease compared to cognitively intact control subjects. Biochim Biophys Acta. 2010;1801:951–959. doi: 10.1016/j.bbalip.2010.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wong AC, Mak ST. Finasteride-associated cataract and intraoperative floppy-iris syndrome. J Cataract Refract Surg. 2011;37:1351–1354. doi: 10.1016/j.jcrs.2011.04.013. [DOI] [PubMed] [Google Scholar]
- 88.Singh S, Hota D, Prakash A, Khanduja KL, Arora SK, Chakrabarti A. Allopregnanolone, the active metabolite of progesterone protects against neuronal damage in picrotoxin-induced seizure model in mice. Pharmacol Biochem Behav. 2010;94:416–422. doi: 10.1016/j.pbb.2009.10.003. [DOI] [PubMed] [Google Scholar]
- 89.Kokate TG, Banks MK, Magee T, Yamaguchi S, Rogawski MA. Finasteride, a 5alpha-reductase inhibitor, blocks the anticonvulsant activity of progesterone in mice. J Pharmacol Exp Ther. 1999;288:679–684. [PubMed] [Google Scholar]
- 90.Lonsdale D, Burnham WM. The anticonvulsant effects of progesterone and 5alpha-dihydroprogesterone on amygdala-kindled seizures in rats. Epilepsia. 2003;44:1494–1499. doi: 10.1111/j.0013-9580.2003.59402.x. [DOI] [PubMed] [Google Scholar]
- 91.Reddy DS, Castaneda DC, O'Malley BW, Rogawski MA. Anticonvulsant activity of progesterone and neurosteroids in progesterone receptor knockout mice. J Pharmacol Exp Ther. 2004;310:230–239. doi: 10.1124/jpet.104.065268. [DOI] [PubMed] [Google Scholar]
- 92.Kenny B, Ballard S, Blagg J, Fox D. Pharmacological options in the treatment of benign prostatic hyperplasia. J Med Chem. 1997;40:1293–1315. doi: 10.1021/jm960697s. [DOI] [PubMed] [Google Scholar]
- 93.Khisti RT, Chopde CT, Jain SP. Antidepressant-like effect of the neurosteroid 3alpha-hydroxy-5alpha-pregnan-20-one in mice forced swim test. Pharmacol Biochem Behav. 2000;67:137–143. doi: 10.1016/s0091-3057(00)00300-2. [DOI] [PubMed] [Google Scholar]
- 94.Bitran D, Shiekh M, McLeod M. Anxiolytic effect of progesterone is mediated by the neurosteroid allopregnanolone at brain GABAA receptors. J Neuroendocrinol. 1995;7:171–177. doi: 10.1111/j.1365-2826.1995.tb00744.x. [DOI] [PubMed] [Google Scholar]
- 95.Uzunova V, Sheline Y, Davis JM, Rasmusson A, Uzunov DP, Costa E, et al. Increase in the cerebrospinal fluid content of neurosteroids in patients with unipolar major depression who are receiving fluoxetine or fluvoxamine. Proc Natl Acad Sci U S A. 1998;95:3239–3244. doi: 10.1073/pnas.95.6.3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Le Melledo JM, Baker G. Role of progesterone and other neuroactive steroids in anxiety disorders. Expert Rev Neurother. 2004;4:851–860. doi: 10.1586/14737175.4.5.851. [DOI] [PubMed] [Google Scholar]
- 97.Irwig MS. Depressive symptoms and suicidal thoughts among former users of finasteride with persistent sexual side effects. J Clin Psychiatry. 2012;73:1220–1223. doi: 10.4088/JCP.12m07887. [DOI] [PubMed] [Google Scholar]
- 98.Rahimi-Ardabili B, Pourandarjani R, Habibollahi P, Mualeki A. Finasteride induced depression: a prospective study. BMC Clin Pharmacol. 2006;6:7. doi: 10.1186/1472-6904-6-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Gubelin Harcha W, Barboza Martinez J, Tsai TF, Katsuoka K, Kawashima M, Tsuboi R, et al. A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. J Am Acad Dermatol. 2014;70:489–498.e3. doi: 10.1016/j.jaad.2013.10.049. [DOI] [PubMed] [Google Scholar]
- 100.Irwig MS. Persistent sexual and nonsexual adverse effects of finasteride in younger men. Sex Med Rev. 2014;2:24–35. doi: 10.1002/smrj.19. [DOI] [PubMed] [Google Scholar]