Summary
The ability of bone to resist fracture depends on the intrinsic properties of the materials that comprise the bone matrix mineralization, the amount of bone (i.e. mass), and the spatial distribution of the bone mass (i.e. microarchitecture). Antiresorptive agents may prevent the decay of cancellous bone and cortical thinning, with no improvement of bone microstructure, leading to a partial correction of the principal bone quality defect in osteoporosis, the disruption of trabecular microarchitecture. Anabolic agents promote bone formation at both trabecular and endocortical surfaces, resulting in an increase of cancellous bone volume and cortical thickness. The improvement of cortical bone strength may be limited by an increase in cortical porosity. strontium ranelate improves trabecular network and cortical thickness that will contribute to anti-fracture efficacy at both vertebral and non-vertebral sites.
The results of clinical and experimental studies are consistent with the mode of action of strontium involving dissociation between bone formation and resorption leading to a stimulation both trabecular and cortical bone formation without increasing cortical porosity.
Keywords: strontium ranelate, bone quality, cortical bone
Background
The aim of the pharmacological treatment of osteoporosis is to balance bone formation and bone resorption. Unbalanced bone metabolism, resulting in bone loss, starts during the transition to menopause (perimenopause). Even bone quality degenerates during perimenopause and subsequent menopause (1).
Bone mass builds up during the first twenty years of life (2). Once skeletal growth has been completed, bone health is preserved by the coupled processes of old bone resorption by osteoclasts and of new bone formation led by osteoblasts, together called bone remodeling, taking place throughout life (3).
This process requires a constant flow of communication between osteoclast and osteoblast cells, in order to keep constantly synchronized bone resorption and bone formation (3). A disproportion, resulting in a greater osteoclast activity, would end up causing bone loss, decreased bone mass and subsequent increased fracture risk (4).
Fractures occur when load exceeds bone strength (applied load/bone strength > 1). Therefore therapeutic strategies in order to reduce the risk of fracture would consist in reducing applied load or increasing bone strength.
Strategies to reduce bone failure load consist in the prevention of falls through physiatric approach, nutritional approach, vitamin D supplementation and in biomechanical interventions such as hip pads, preventive hip surgery, vertebral body augmentation.
On other hand, therapeutic strategies to increase bone strength consist in improving both bone quantity and quality through physiatric interventions (e.g. physical exercises), nutritional approach, vitamin D and calcium supplementation, micronutrients supplementation, biophysics stimulation and drug therapy.
The ability of bone to resist fracture (or “whole bone strength”) depends on the intrinsic properties of the materials that comprise the bone matrix mineralization, the amount of bone (ie, mass), and the spatial distribution of the bone mass (ie, microarchitecture).
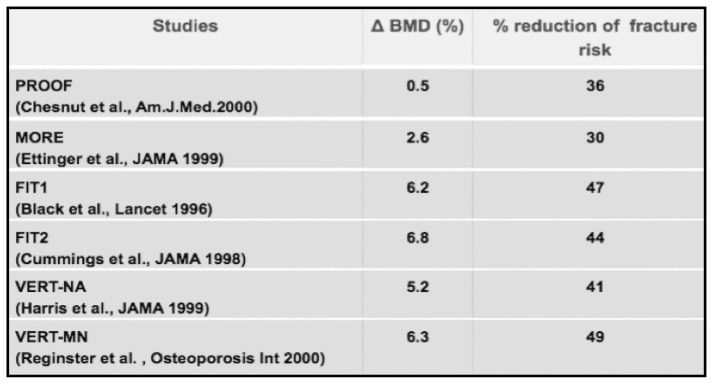
Bone quality has been defined as “an umbrella term to describe a set of characteristics of bone tissue that, in addition to density, contribute to bone strength” (5). The term “bone quality” has become common since the late 90’s when it was clear from the results of clinical trials upon efficacy of several anti-osteoporotic drugs that BMD increase did not reflect a proportional reduction of relative risk of fracture (Figure 1).
Figure 1.
Pivotal studies of antiresorptive drugs.
The role of antiresorptive drugs
Antiresorptive therapies reduce the rate of bone remodeling lowering the fracture rate by 30 to 50%. Antiresorptive agents, however, do not increase bone tissue mass. The gain in bone mineral density observed in clinical trials of antiresorptive drugs is the result of a more complete secondary mineralization of the existing (but reduced) bone tissue mass. Bisphosphonates may prevent the decay of cancellous bone and cortical thinning, although an improvement of bone microstructure above baseline has never been demonstrated with these drugs. Antiresorptive treatment results in partial correction of the principal bone quality defect in osteoporosis, the disruption in bone microarchitecture, but cannot completely restore mechanical integrity because of the absence of an anabolic effect. The main goal of antiosteoporotic treatments is to improve bone quality and strength, thereby reducing the risk of fracture. Antifracture efficacy would therefore be expected to be linked to the effect of treatment on bone microarchitecture. The μCT studies add today more evidences to the idea that both trabecular and cortical microarchitecture are major and independent determinants of osteoporotic fracture, as important as bone mineral density, bone turnover and mineralization (6). Restoration of bone tissue mass and bone structure requires the use of anabolic agents.
The role of the anabolic drugs
Anabolic agents, like PTH or Teriparatide, promote bone formation at both trabecular and endocortical surfaces, resulting in an increase of cancellous bone volume and cortical thickness. The improvement of cortical bone strength may be limited by an increase in cortical porosity.
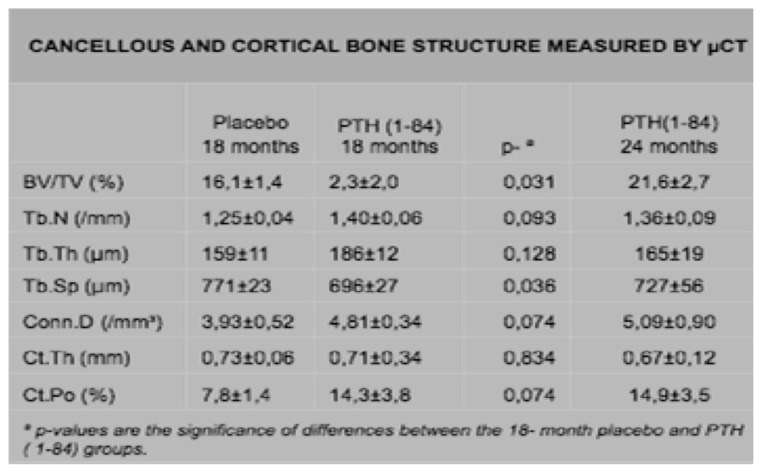
Recker et al. compared PTH (1–84) treatment to placebo in postmenopausal osteoporotic women for 18 months. PTH treatment was associated with higher cancellous bone formation and bone volume in the iliac crest without affecting bone resorption. There was tendency for cortical porosity to be higher and there was no effect on cortical thickness (7) (Figure 2).
Figure 2.
Cancellous and cortical bone structure in POMP women randomized to PTH and placebo treatment.
An 18-month randomized double-blind study was conducted in postmenopausal women with osteoporosis to compare the effects of once-daily teriparatide 20 μg with alendronate 10 mg on bone histomorphometry. Biopsies were obtained from 42 patients. Histomorphometry indices of bone formation were significantly higher after 6 or 18 months of teriparatide compared with alendronate treatment (8).
Treatment with an antiresorptive agent appears to preserve baseline trabecular microarchitecture, to have no impact on cortical bone, and to increase mineralization of bone. On the other hand, the bone-forming agent teriparatide produce an overall improvement in trabecular microarchitecture and increases cortical thickness, even if it tends to decrease bone mineralization and to increase cortical porosity.
The role of strontium ranelate
Strontium ranelate is an orally active agent consisting of two atoms of stable strontium and an organic moiety (ranelic acid), now considered effective in managing osteoporosis and reducing fracture risk in postmenopausal women. By its dual mode of action, strontium ranelate stimulates bone formation and decreases bone resorption, and therefore rebalances bone turnover in favor of new bone formation (9). In numerous studies this drug has demonstrated to improve all parameters linked to bone quality and bone strength (10–13).
Bone mineral density
Strontium ranelate increases lumbar spine BMD by 14.4% compared with placebo at 3 years of treatment (P<0.001). A similar effect was measured at the level of hip, with an increase of femoral neck BMD by 8.3% compared with placebo (P=0.001) (13). Results from the logistic regression analysis showed that in patients treated with strontium ranelate for each increase of 1% in femoral neck BMD, the risk of experiencing a new vertebral fracture after 3 years decreased by 3%. The 3-year changes in femoral neck BMD explained 74% of the reduction in vertebral fractures observed during treatment with strontium ranelate (14).
Degree of mineralization
A higher degree of mineralization produces stiffer but more brittle bone, while a lower degree of mineralization produces less stiff but tougher bone. Strontium ranelate preserves the physiological mineralization process during and after treatment. In postmenopausal women treated for 3 years, analysis of bone biopsies by microradiography showed that the mean degree of mineralization of bone did not differ between groups (15). Moreover, bone strontium content reached a plateau between year 2 and 3 of treatment, thus illustrating that strontium does not replace calcium ions, but is adsorbed onto the bone mineral surface. Based on X-ray cartography, strontium is only distributed in recent bone. These results suggest that the increased BMD noted during strontium ranelate therapy is due to increased bone mass, but not to a modulation of mineralization as it occurs with antiresorptive therapies. The quality of bone mineral is preserved, supporting the safety of this treatment at the bone tissue level (16).
Bone microarchitecture
For the same decline in bone mass, loss of trabecular elements was 2 to 5 times more deleterious to bone strength than thinning of the trabecular struts, implying that maintaining connectivity of the trabecular network is critical. The relationship between femoral geometry and hip fracture risk has also been extensively studied. The odds ratios suggest that for each standard deviation difference in a geometric variable, hip fracture risk is increased by 1.2-fold to 5-fold. Three-dimensional assessment (microCT) of bone biopsies in patients treated with strontium ranelate for three years showed a significant effect on both trabecular and cortical bone. At the trabecular level strontium ranelate improves bone microarchitecture. Compared with placebo, it increased trabecular number by +14% (P=0.05), decreased trabecular separation by −16% (P=0.04), and improved trabecular structural model index by 22% (P=0.01) shifting trabeculae from rod-like structure to plate-like pattern. Strontium ranelate also enlarged cortical bone dimensions increasing cortical thickness by 18% (P=0.008), which represents the basis of resistance of long bones, such as femur (17).
Moreover, using 5-year data from the TROPOS study, 486 hip DXA scans of postmenopausal osteoporotic women with similar baseline characteristics were examined. Over 5 years, strontium ranelate has positive effects on hip geometry: the mean increase in cross-sectional area (CSA) was 5.8 ± 9.1% for strontium ranelate compared with placebo (−3.2 ± 7.2%, P<0.01). A similar effect was seen with the increase in section modulus (8.6 ± 14.3% vs −2.3 ± 11.6%, P<0.001) and the decreases in the buckling ratio (by −3.1 ± 13.2%, P<0.001 in SR group) (18).
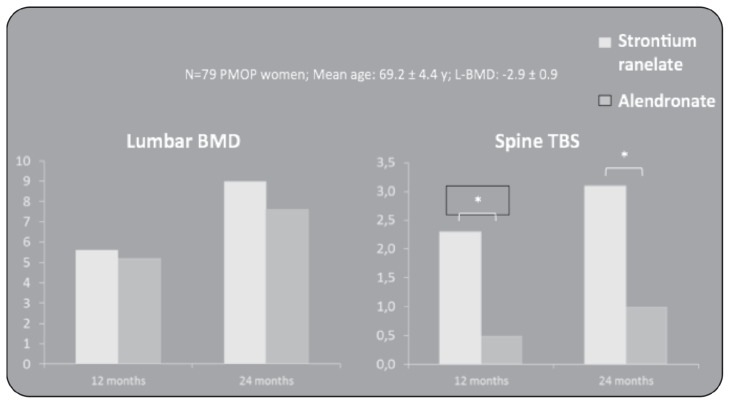
The effects of strontium ranelate on bone microarchitecture have been compared also with another antiosteoporotic treatment (alendronate once a week) in a randomized, double-blind trial. Bone microstructure at the distal tibia was assessed by the noninvasive technique high-resolution peripheral computerized tomography (HR-pQCT) in 88 women with postmenopausal osteoporosis (19). The patients’ baseline characteristics were similar in both groups with a mean age of 63.7 years, a BMD T-score of −2.7 and −2.0, respectively at lumbar and hip site. Within the constraints of HR-pQCT method, strontium ranelate appeared to have greater effects than alendronate on distal tibia cortical thickness, trabecular and cortical bone densities, improving bone microstructure since the 3rd month of treatment and increasing up to 2 years. In this study, to assess the influence of treatments on bone strength, was also used the finite element analysis method (FEA) originated from the need for solving complex elasticity and structural analysis problems in civil and aeronautical engineering. Over 2 years, estimated failure load increased with strontium ranelate by 2.1% (P<0.005), whereas no significant change was observed with alendronate (−0.6%, NS), leading to a significant between-group difference (P<0.01). Treatment with strontium ranelate was also associated with lower trabecular and cortical stresses, with relative changes of −2.4% (P<0.005) and −1.6% (P<0.05), respectively. Recently it has been published a new quantitative index of bone architecture, named Trabecular Bone Score (TBS). This index used DXA method but is independent of BMD. pQCT Stratec Study compares the efficacy of strontium ranelate and alendronate on bone mass, bone geometry and bone strength (mechanical parameters) at level of spine architecture patterns in POMP women using a Trabecular Bone Score (TBS) (20). A post-hoc analysis was performed on DXAs from 79 women out of 189 included in a double blind, double dummy study and randomized to strontium ranelate 2g/day or alendronate 70mg/week during 2 years. The study shows that there is an increase of Spine BMD and TBS on both treatments but a differential effect of alendronate and strontium ranelate on bone microarchitecture in particular SrRan has greater effects on bone architecture; index at the spine compared to ALN as already described in pQCT and HR-pQCT studies (Figure 3).
Figure 3.
Effects of SrRan and ALD on Lumbar BMD and Spine TBS in POMP women.
Conclusion
Increasing knowledge about the cellular and molecular pathways involved in the maintenance of bone homeostasis and about the role of disturbances in these pathways in the pathogenesis of osteoporosis has paved the way for a better understanding of the putative mechanisms responsible for the beneficial effects of strontium ranelate on bone-forming approach to this condition. A decade of extensive in vitro and in vivo pre-clinical evaluation followed by a comprehensive clinical program in postmenopausal women has led to confirm that strontium ranelate represents a unique approach in the management of osteoporosis by its ability to restore the imbalance between bone resorption and formation in favor of bone formation. Its beneficial effects on bone microarchitecture improve the biomechanical properties of bone to resist to fragility fractures.
References
- 1.Watts NB, Bilezikian JP, Camacho PM, et al. American Association of Clinical Endocrinologists medical guidelines for clinical practice for diagnosis and treatment of postmenopausal osteoporosis. Endocr Pract. 2010;16:1–37. doi: 10.4158/ep.16.s3.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Heaney RP, Abrams S, Dawson-Hughes B, Looker A, Marcus R, Matkovic V, Weaver C. Peak bone mass. Osteoporos Int. 2000;11:985–1009. doi: 10.1007/s001980070020. [DOI] [PubMed] [Google Scholar]
- 3.Raisz LG. Pathogenesis of osteoporosis: concepts, conflicts, and prospects. J Clin Invest. 2005;115:3318–3325. doi: 10.1172/JCI27071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Robbins JA, Schott AM, Garnero P, Delmas PD, Hans D, Meunier PJ. Risk factors for hip fracture in women with high BMD: EPIDOS study. Osteoporos Int. 2005;16:149–154. doi: 10.1007/s00198-004-1661-y. [DOI] [PubMed] [Google Scholar]
- 5.Bouxsein M. Bone Quality & Osteoporotic Fracture. National Osteoporosis Foundation. 2007 [Google Scholar]
- 6.Seeman E, Delmas PD. Bone quality - the material and structural basis of bone strength and fragility. N Engl J Med. 2006;354:2250–61. doi: 10.1056/NEJMra053077. [DOI] [PubMed] [Google Scholar]
- 7.Recker RR, Bare SP, Smith SY, Varela A, Miller MA, Morris SA, Fox J. Cancellous and cortical bone architecture and turnover at the iliac crest of postmenopausal osteoporotic women treated with parathyroid hormone 1–84. Bone. 2008;44:113–119. doi: 10.1016/j.bone.2008.09.019. [DOI] [PubMed] [Google Scholar]
- 8.Arlot M, Meunier PJ, Boivin G, et al. Differential Effects of Teriparatide and Alendronate on Bone Remodeling in Postmenopausal Women Assessed by Histomorphometric Parameters. Journal of Bone and Mineral Research. 2005 Jul;20 doi: 10.1359/JBMR.050309. [DOI] [PubMed] [Google Scholar]
- 9.Strontium ranelate. Summary of product characteristics. http://www.emea.europa.eu. Cited 3 June 2010.
- 10.Meunier PJ, Roux C, Ortolani S, Diaz-Curiel M, Compston J, Marquis P, et al. Effects of long-term strontium ranelate treatment on vertebral fracture risk in postmenopausal women with osteoporosis. Osteoporos Int. 2009;20:1663–73. doi: 10.1007/s00198-008-0825-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Meunier PJ, Roux C, Seeman E, Ortolani S, Badurski JE, Spector TD, et al. The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med. 2004;350:459–6812. doi: 10.1056/NEJMoa022436. [DOI] [PubMed] [Google Scholar]
- 12.Meunier PJ, Slosman DO, Delmas PD, Sebert JL, Brandi ML, Albanese C, et al. Strontium Ranelate: dose-dependent effects in established postmenopausal vertebral osteoporosis – a 2-year randomized placebo controlled trial. J Clin Endocrinol Metab. 2002;87:2060–6. doi: 10.1210/jcem.87.5.8507. [DOI] [PubMed] [Google Scholar]
- 13.Reginster JY, Seeman E, De Vernejoul MC, et al. Strontium ranelate reduces the risk of non vertebral fractures in postmenopausal women with osteoporosis: Treatment of Peripheral Osteoporosis (TROPOS) study. J Clin Endocrinol Metab. 2005;90:2816–2822. doi: 10.1210/jc.2004-1774. [DOI] [PubMed] [Google Scholar]
- 14.Bruyere O, Roux C, Detilleux J, Slosman DO, Spector TD, Fardellone P, Brixen K, Devogelaer JP, Diaz-Curiel M, Albanese C, Kaufman JM, Pors-Nielsen S, Reginster JY. Relationship between bone mineral density changes and fracture risk reduction in patients treated with strontium ranelate. J Clin Endocrinol Metab. 2007 Aug;92(8):3076–81. doi: 10.1210/jc.2006-2758. [DOI] [PubMed] [Google Scholar]
- 15.Doublier A, Farlay D, Khebbab MT, Jaurand X, Meunier PJ, Boivin G. Distribution of strontium and mineralization in iliac bone biopsies from osteoporotic women long-term treated with strontium ranelate. Eur J Endocrinol. 2011;165:469–76. doi: 10.1530/EJE-11-0415. [DOI] [PubMed] [Google Scholar]
- 16.Boivin G, Farlay D, Simi C, Meunier PJ. In osteoporotic women treated with strontium ranelate, strontium is located in bone formed during treatment with a maintained degree of mineralization. Osteoporos Int. 2010;21:667–77. doi: 10.1007/s00198-009-1005-z. [DOI] [PubMed] [Google Scholar]
- 17.Arlot ME, Jiang Y, Genant HK, Zhao J, Burt-Pichat B, Roux JP, Delmas PD, Meunier PJ. Histomorphometric and microCT analysis of bone biopsies from postmenopausal osteoporotic women treated with strontium ranelate. J Bone Miner Res. 2008;23:215–22. doi: 10.1359/jbmr.071012. [DOI] [PubMed] [Google Scholar]
- 18.Briot K, Benhamou CL, Roux C. Effect of strontium ranelate on hip structural geometry. Abstract SAT0375. Ann Rheum Dis. 2009;68(3):665. [Google Scholar]
- 19.Rizzoli R, Felsenberg D, Laroche M, Seeman E, Krieg MA, Frieling I, Thomas T, Delmas PD. Superiority of strontium ranelate as compared to alendronate on microstructural determinants of bone strength at the distal tibia in women with postmenopausal osteoporosis. Abstract SAT0388. Ann Rheum Dis. 2009;68(3):669. [Google Scholar]
- 20.Hans D, Krieg MA, Lamy O, Felsenberg D. Beneficial effects of Strontium Ranelate compared to Alendronate on trabecular bone score in postmenopausal osteoporotic women: a 2-year study. J Bone Mineral Res. 2011;26(11):2762–9. [Google Scholar]