Abstract
Objective
To hypothesize whether the association between high forced expiratory volume in 1 second (FEV1) and increased rate of decline in FEV1 in children with cystic fibrosis could be due to less frequent intervention after acute drops (sudden decline events) in FEV1.
Study design
Patients with CF aged 6-17 years enrolled in ESCF were assessed for a sudden decline event, defined as a 10% relative drop in FEV1 % predicted from an average of 3 consecutive stable baseline spirometries. The likelihood of therapeutic intervention within 14 days before and 56 days after this event was then related to their baseline FEV1 % predicted age-specific decile using a logistic regression adjusting for age group (6-12y, 13-17y) and presence of Pseudomonas aeruginosa on respiratory culture.
Results
10,888 patients had at least one sudden decline event in FEV1. Patients in the highest FEV1 decile were significantly less likely than those in the lowest decile to receive intravenous antibiotics (odds ratio [OR], 0.14; 95% confidence interval [CI], 0.11-0.18; P<.001) or be hospitalized (OR, 0.18; 95% CI, 0.14-0.23; P<.001) following decline.
Conclusions
Children and adolescents with high baseline lung function are less likely to receive a therapeutic intervention following an acute drop in FEV1, which may explain their greater rate of FEV1 decline.
Keywords: cystic fibrosis, child, adolescent, lung, epidemiology, respiratory function tests, spirometry, forced expiratory volume, hospitalization, antibacterial agents
Cystic fibrosis (CF) lung disease is characterized by airway obstruction, chronic infection, and inflammation leading to progressive damage with irreversible obstructive airway disease.1 The forced expiratory volume in 1 second (FEV1) has been studied in patients with CF to better understand the progression of CF lung disease.2-6 Many factors have been associated with the rate of FEV1 decline, including high lung function.2,7,8 More recently, we used data from the Epidemiologic Study of Cystic Fibrosis (ESCF) to show that baseline FEV1 values of at least 100% of predicted were associated with a subsequent relative increase in the rate of decline ranging from 12% in adolescents to 46% in children ages 6 to 8 years when compared with the overall rate of decline.9 We reasoned that this increased risk of decline for those with the highest FEV1 values could have been due to a lack of a therapeutic response following a clinically relevant drop in lung function in these children. In other words, we suspected that a disparity in health care may exist in children with high FEV1 values due to a bias to be less therapeutically aggressive in these apparently healthy children.
We have previously shown that pulmonary exacerbations are associated with progressive lung function decline.9 This has been confirmed and there is evidence that patients frequently fail to return to baseline following an exacerbation.10,11 One factor that predicted a failure to recover lung function after treatment with intravenous (IV) antibiotics was a delay in treatment.11 This suggests that the increased rate of decline we have observed in children with high lung function could be due to delayed or deferred response to an acute progression of their disease. We hypothesized that following a meaningful relative decline in FEV1, children with high lung function at baseline would be less likely to be treated with either hospitalization or IV antibiotics.
METHODS
ESCF is a large multicenter, longitudinal, prospective observational study of the clinical course of patients with CF in the United States and Canada from 1994 through 2005.12 Data collected on each patient included clinical demographics, signs and symptoms of lung disease, information on growth and nutrition, results of spirometry, results of respiratory tract cultures, use of routine therapies, and information on pulmonary exacerbations and their treatment. Informed consent was obtained based on decisions by local human subjects review boards.
To be included in the current analysis, patients had to be enrolled in ESCF and have at least four FEV1 values meeting certain criteria. Patients had to have a baseline FEV1 % predicted established by a moving average of three spirometries obtained when the patient was clinically stable and that spanned 30-365 days. At least one recorded clinical encounter must have occurred in this period. If within 1 year from the last stable spirometry there was a relative decline in FEV1 % predicted of at least 10% from baseline, this was considered a sudden decline event (Figure 1). To be included, patients with a sudden decline event were required to have a respiratory tract culture recorded in the year prior and not have been hospitalized between the last stable spirometry and the sudden decline event. For each patient, only the first qualifying sudden decline event was analyzed.
Figure 1.
Analytical schema showing the baseline FEV1 averaged from three values obtained at a time of clinical stability (open circles) followed by a decline in FEV1 below the threshold of a 10% relative decline (filled square). Therapeutic interventions included new antibiotic therapy (evaluated from 14 days before until 56 days following the decline) and hospitalization (evaluated only for 56 days following the decline).
Clinical interventions were then assessed around the time of the sudden decline event. Hospitalization was defined as an intervention if it occurred on the day of, or up to 56 days following, the sudden decline event. Antibiotic interventions were assessed between 14 days before and 56 days following the sudden decline event. This window was chosen to give time for therapeutic interventions to be started, but prior to the next routine clinic visit. Any IV antibiotic therapy during this period was considered a clinical intervention. Other antibiotic therapies were considered new interventions provided they had not been reported at the last encounter during the baseline period. When the last stable spirometry occurred within 14 days prior to decline, therapies started in this time frame were also considered new interventions. These interventions were categorized as either inhaled antibiotics, oral quinolone antibiotics, or other oral antibiotics.
Due to the progressive decline in lung function with age in patients with CF, we reasoned that baseline severity of lung disease would be better characterized relative to children with CF of the same age rather than by comparison with the general CF population. We therefore developed reference values by calculating deciles of FEV1 % predicted for each integer year of age from 6 to 17 years using all spirometry data recorded in ESCF. We used these deciles to evaluate treatment response by lung disease severity. Values for FEV1 % predicted were calculated from Wang13 for males through age 17 years and for females through age 15 years and from the equations of Hankinson14 for patients above these ages.
For each intervention, patient counts and percentages were produced for each FEV1 decile and age group (children aged 6-12 years and adolescents aged 13-17 years). Differences across deciles but within age groups were assessed by the Mantel-Haenszel χ2 statistic. We evaluated the relationship between baseline FEV1 age-specific decile and the likelihood of therapeutic intervention using a logistic regression adjusting for the presence of Pseudomonas aeruginosa on respiratory culture, weight-for-age percentile category, and age category. Because of the importance of symptoms for defining a CF-related pulmonary exacerbation,15-17 we repeated the analysis for only those patients who had daily cough, daily sputum production, and crackles on physical examination at the visit closest to the decline event. Furthermore, the analysis was repeated for only those patients with positive Pseudomonas aeruginosa on respiratory culture. Odds ratios (ORs) and associated 95% confidence intervals (CIs) are reported, as is the P value for the linear trend of OR by FEV1 decile.
Statistical analyses were performed using SAS 9.1 (SAS Institute, Inc., Cary, NC). P values <.05 were considered significant. No adjustments were made for multiple comparisons.
RESULTS
Of all children and adolescents enrolled in ESCF between 1994 and 2005, 16,524 had sufficient FEV1 data. Of these, 11,276 (68%) patients had three stable baseline FEV1 values, were 6-17 years of age at the first of these values, and demonstrated at least one sudden decline event. We excluded 388 patients because either (1) during the moving average period, they had no respiratory cultures or no encounters, or (2) they were hospitalized before or during all their sudden decline events. This left 10,888 patients for this analysis.
Baseline demographic data and clinical status by age group at the time of the decline event are presented in Table I (available at www.jpeds.com). As expected, the adolescents demonstrated lower FEV1 % predicted and weight-for-age percentile values than the children and had higher prevalence of Pseudomonas aeruginosa infection and of respiratory signs and symptoms. The median span of the baseline spirometries was approximately 6 months in both age groups. In both age groups, the median time from the last baseline spirometry to the sudden decline event was 91 days (range, 1-364).
Table I.
Demographic and Clinical Characteristics by Age Group*
| Characteristic | Ages 6-12 (N = 7695) | Ages 13-17 (N = 3193) |
|---|---|---|
| Age, mean ± SD | 8.8 ± 2.1 | 15.3 ± 1.4 |
| Female, n (%) | 3847 (50.0) | 1492 (46.7) |
| ΔF508 Homozygous† n (%) | 3030 (55.2) | 1050 (54.0) |
| Weight for age (percentile), mean ± SD | 30.6 ± 26.3 | 28.5 ± 26.7 |
| FEV1 % predicted at moving average, mean ± SD | 88.6 ± 19.9 | 77.4 ± 22.5 |
| FEV1 % predicted at decline, mean ± SD | 71.5 ± 19.3 | 62.5 ± 20.8 |
| FEV1 % predicted % change, mean ± SD | –19.7 ± 9.8 | –19.8 ± 9.9 |
| Pseudomonas positive, n (%) | 4375 (56.9) | 2399 (75.1) |
| Pulmonary signs and symptoms positive‡ n (%) | ||
| Daily cough | 4583 (60.1) | 2262 (71.5) |
| Daily sputum production | 2652 (35.0) | 1701 (54.2) |
| Clubbing | 4300 (56.2) | 2074 (65.2) |
| Crackles | 1937 (25.3) | 1124 (35.3) |
| Wheeze | 687 (9.0) | 312 (9.8) |
| Hospitalized within 56 days of decline, n (%) | 1406 (18.3) | 834 (26.1) |
| Therapies received within –14 and +56 days of decline, n (%) | ||
| IV antibiotics | 1745 (22.7) | 986 (30.9) |
| New inhaled antibiotics | 1684 (21.9) | 812 (25.4) |
| New quinolone antibiotic | 1616 (21.0) | 1037 (32.5) |
| New oral non-quinolone antibiotics | 1951 (25.4) | 701 (22.0) |
P values for age group comparisons were obtained from t test or χ2 test statistic, as appropriate. All P <.001 with the exception of female (P = .002), ΔF508 homozygous (P = .35), FEV1 (% pred.) % change (P = .61), and wheeze (P = .15).
Includes patients with nonmissing data at baseline. 2206 patients aged 6-12 years and 1,248 patients aged 13-17 years were missing data for ΔF508 homozygous.
Symptom data are unavailable for 24 to 122 patients 6-12 years of age and 11 to 55 patients 13-17 years of age, depending on the symptom.
Although the threshold for sudden decline was a 10% relative drop, the actual drops in FEV1 were much larger, with the mean decline being a relative drop of 19.7% ± 9.8 (median, 16.5) in the children and 19.8% ± 9.9 (median, 16.4) in the adolescents. Among the children with a sudden decline event, over 85% had an absolute drop of at least 10 points in FEV1 % predicted, and over 25% had a drop of at least 20 points. For the adolescents, who have a lower average starting value, nearly 73% had an absolute drop of at least 10 points and over 18% at least 20 points. Yet, no new intervention was reported in a substantial proportion of these patients. Even among those with absolute drops of at least 20 points, nearly one-third of the children and one-fourth of the adolescents were neither hospitalized nor treated with a new course of antibiotic therapy (data not shown).
Therapeutic Intervention by Baseline Lung Function Decile
Hospitalizations were recorded following sudden decline for 18.3% of children and 26.1% of adolescents with any IV antibiotic therapy reported for 22.7% and 30.9%, respectively (Table I). The likelihood of IV antibiotic therapy and hospitalization were both significantly lower in patients with higher lung function in both age groups (linear trend P <.001; Figure 2). Although over half of the children and adolescents in the lowest decile received IV antibiotic therapy, less than 20% of patients in the top four deciles received such therapy.
Figure 2.
Therapeutic intervention by baseline lung function decile by age group. P <.001 for linear trend by severity deciles for both age groups and for both intravenous antibiotic therapy (A) and hospitalization (B).
The likelihood of new inhaled antibiotics was also lower in patients with higher lung function (linear trend P <.001 for both age groups). Similar findings were observed for new quinolone antibiotics for children. However, the linear trend was weaker for children receiving other new oral antibiotics and for adolescents receiving new quinolone antibiotics or other new oral antibiotics (P >.05) (data not shown).
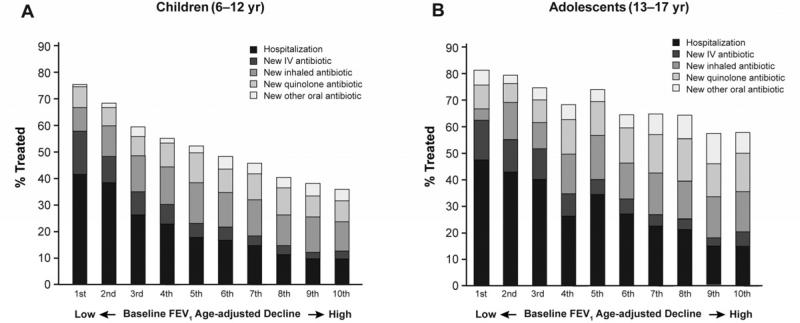
Figure 3 relates antibiotic interventions to FEV1 decile. Although approximately 80% of patients in the lowest decile received some acute intervention, only 49% of children and 58% of adolescents in the highest decile were treated.
Figure 3.
Frequency distribution of new antibiotic interventions (evaluated from 14 days before until 56 days following the decline) and hospitalizations (evaluated only for 56 days following the decline), by baseline lung function decile by age group.
The multivariate logistic regression (Table II) demonstrated that after adjusting for age category, ,respiratory culture status, and weight for age (≤ 25th percentile vs > 25th percentile), high lung function at baseline still predicted a low rate of therapeutic intervention with antibiotics following a sudden decline in FEV1. As compared with patients in the lowest FEV1 decile at baseline, those in the highest decile were significantly less likely to be hospitalized (OR, 0.20; 95% CI, 0.16-0.26; P <.001) or receive IV antibiotics (OR, 0.15; 95% CI, 0.12-0.20; P <.001). Adolescents and patients with a positive culture for Pseudomonas aeruginosa were more likely to be hospitalized, receive IV antibiotics, or receive new antibiotic therapy. Those with low weight for age were also more likely to be hospitalized or receive IV antibiotics (Table II).
Table II.
Adjusted Logistic Regression Predicting Treatment Associated with Sudden Decline
| Adjustment | N | Hospitalizations | IV Antibiotics | Any Treatment* | |||
|---|---|---|---|---|---|---|---|
| n (%) Treated | OR (CI) | n (%) Treated | OR (CI) | n (%) Treated | OR (CI) | ||
| Pseudomonas negative (ref. group) | 4114 | 567 (13.8) | -- | 669 (16.3) | -- | 2099 (51.0) | -- |
| Pseudomonas positive | 6774 | 1673 (24.7) | 1.54 (1.38-1.72) | 2062 (30.4) | 1.68 (1.51-1.86) | 4607 (68.0) | 1.76 (1.62-1.91) |
| Weight-for-age percentile ≥25 (ref. group) | 5202 | 835 (16.1) | -- | 1030 (19.8) | -- | 3039 (58.4) | -- |
| Weight-for-age percentile <25 | 5686 | 1405 (24.7) | 1.22 (1.10-1.35) | 1701 (29.9) | 1.17 (1.06-1.29) | 3667 (64.5) | 1.04 (0.95-1.13) |
| Age 6-12 years (ref. group) | 7695 | 1406 (18.3) | -- | 1745 (22.7) | -- | 4579 (59.5) | -- |
| Age 13-17 years | 3193 | 834 (26.1) | 1.55 (1.40-1.72) | 986 (30.9) | 1.49 (1.35-1.65) | 2127 (66.6) | 1.26 (1.15-1.37) |
| 1st FEV1 decile (ref. group) | 492 | 212 (43.1) | -- | 280 (56.9) | -- | 396 (80.5) | -- |
| 2nd FEV1 decile | 757 | 299 (39.5) | 0.89 (0.70-1.12) | 366 (48.4) | 0.73 (0.58-0.91) | 576 (76.1) | 0.78 (0.59-1.04) |
| 3rd FEV1 decile | 868 | 261 (30.1) | 0.60 (0.48-0.76) | 332 (38.3) | 0.50 (0.40-0.62) | 598 (68.9) | 0.57 (0.44-0.74) |
| 4th FEV1 decile | 1048 | 250 (23.9) | 0.46 (0.36-0.58) | 312 (29.8) | 0.35 (0.28-0.44) | 688 (65.7) | 0.51 (0.40-0.66) |
| 5th FEV1 decile | 1162 | 264 (22.7) | 0.43 (0.34-0.55) | 310 (26.7) | 0.31 (0.24-0.38) | 768 (66.1) | 0.53 (0.41-0.68) |
| 6th FEV1 decile | 1239 | 242 (19.5) | 0.37 (0.30-0.47) | 292 (23.6) | 0.27 (0.22-0.34) | 755 (60.9) | 0.44 (0.34-0.56) |
| 7th FEV1 decile | 1335 | 226 (16.9) | 0.31 (0.25-0.39) | 261 (19.6) | 0.21 (0.17-0.27) | 785 (58.8) | 0.40 (0.31-0.51) |
| 8th FEV1 decile | 1354 | 191 (14.1) | 0.26 (0.20-0.33) | 222 (16.4) | 0.18 (0.14-0.22) | 759 (56.1) | 0.36 (0.28-0.47) |
| 9th FEV1 decile | 1384 | 156 (11.3) | 0.20 (0.16-0.26) | 176 (12.7) | 0.13 (0.10-0.17) | 732 (52.9) | 0.33 (0.25-0.42) |
| 10th FEV1 decile | 1249 | 139 (11.1) | 0.20 (0.16-0.26) | 180 (14.4) | 0.15 (0.12-0.20) | 649 (52.0) | 0.31 (0.24-0.40) |
| Decile trend (P value) | -- | -- | <.001 | -- | <.001 | -- | <.001 |
“Any treatment” is defined as any new antibiotic therapy, evaluated from 14 days before until 56 days following the decline, or hospitalization, evaluated only for 56 days following the decline.
Importantly, the inverse relationship between baseline lung function and likelihood of treatment persisted even when limited to patients who had daily cough, daily sputum, and crackles on lung examination at the time of decline. At the visit nearest the sudden decline event, 13.8% of children (n = 7544) and 24.5% of adolescents (n = 3126) had all three of these signs and symptoms. Even in the presence of cough, sputum, and crackles, patients in the highest decile were less likely to receive IV antibiotics (OR, 0.47; 95% CI, 0.27-0.80) or be hospitalized (OR, 0.62; 95% CI, 0.36-1.08) (data not shown). Similarly, when the analysis was limited to those individuals who were positive for Pseudomonas aeruginosa, patients in the highest decile were still less likely to receive IV antibiotics (OR 0.17; 95% CI, 0.13-0.22) compared with those in the lowest FEV1 decile. However, although there was a trend for a lower rate of hospitalization, it was no longer significant (OR 0.23; 95% CI, 0.17-1.31).
DISCUSSION
This study demonstrates that the likelihood of therapeutic intervention after a substantial and clinically meaningful decline in lung function is inversely related to the level of baseline lung function. Overall, the rate of IV antibiotic therapy or hospitalization for treating patients with CF was surprisingly low when using a threshold of a 10% relative drop in FEV1 % predicted. In fact, these patients experienced large absolute declines in lung function in less than a single year. The mean absolute drop in FEV1 % predicted in children and adolescents was 17.1 (standard deviation [SD], 8.9; median, 14.5) and 14.9 (SD, 8.3; median, 12.8) points, respectively. These are strikingly greater than the average annual decline in FEV1 % predicted of 1.66 points for children and 2.34 points for adolescents reported by Konstan et al in 2007.9
Despite this marked loss in lung function, only 12% of children and 19% of adolescents in the highest FEV1 decile at baseline received IV antibiotics. Furthermore, only 52% of these patients with high lung function received any new antibiotic intervention. After adjusting for age group and Pseudomonas aeruginosa infection, patients in the highest FEV1 decile were about one-sixth as likely to be hospitalized and slightly less likely to receive IV antibiotics when compared with patients in the lowest decile. Although it may be understandable that sicker patients are treated more aggressively, in a disease such as CF where progressive loss of lung function is the rule, failure to respond with hospitalization or new antibiotic therapy for those with potentially more controllable disease is disheartening.
Pulmonary exacerbations in CF are commonly characterized by worsening signs and symptoms including reduced exercise tolerance and increased cough and sputum,16 and also by loss in lung function and increased physical findings such as crackles on chest examination.17 Even when we limited our analysis to patients with daily cough, daily sputum, and crackles, the negative impact of high lung function on the likelihood of clinical intervention was still apparent.
The fact that more rapid lung function decline occurs in patients with CF with higher lung function seems counterintuitive, as these patients appear clinically healthier. In contrast to another chronic inflammatory lung disease, chronic obstructive pulmonary disease (COPD), the rate of FEV1 decline in CF seems to be proportional to the remaining lung function. In COPD, lung function decline is not linear, but seems to accelerate (not decelerate) with age and disability.18,19 The pattern of decline in lung function seen in patients with CF may be inherent to the biology of the disease. However, our findings suggest that caregivers may be less aggressive in treating patients who appear to be healthy.
As with any epidemiologic study we were only able to evaluate the information provided by study sites. Underreporting of therapies, particularly oral antibiotics, would result in underestimating the true frequency of intervention. Most likely, any underreporting would apply to all patients and furthermore is unlikely to explain the large differences between the highest and lowest deciles for major interventions such as hospitalization and IV antibiotic therapy. However, underreporting of oral antibiotics could preferentially affect the highest deciles. Also, we did not assess changes in nonantibiotic therapies. Another potential limitation is that these data were collected from 1994 to 2005 and it is possible that practice patterns have improved since 2005 with the recent focus on early intervention. Our analysis was limited to consideration of a single event and therefore we did not assess the long-term impact of multiple events. To evaluate the long-term effect on rate of lung function decline of a series of treatment decisions associated with exacerbations would require a different analytic approach.
In conclusion, children and adolescents with high baseline lung function are less likely to be hospitalized or receive IV antibiotic therapy following a substantial and clinically meaningful decline in FEV1 than those with low lung function. This disparity in care may help explain the greater risk of FEV1 decline seen in children with high lung function and should challenge all providers to be more vigilant in the care of apparently healthy children with cystic fibrosis.
Acknowledgments
Sponsored by Genentech, Inc (San Francisco, CA). W.M., J.W., and M.K. have received honoraria from Genentech for serving as members of the Scientific Advisory Group for the Epidemiologic Study of Cystic Fibrosis and have served as consultants to Genentech; no compensation was provided to these authors in exchange for creation of this manuscript. D.P. and S.M. are employees of ICON Late Phase & Outcomes Research, which was paid by Genentech, for providing analytical services for this study. A.Y. is and Jeffrey Wagener was employees of Genentech.
Abbreviations used
- CF
cystic fibrosis
- COPD
chronic obstructive pulmonary disease
- ESCF
Epidemiologic Study of Cystic Fibrosis
- FEV1
forced expiratory volume in 1 second
- IV
intravenous
Appendix
Epidemiologic Study of Cystic Fibrosis Investigators included: Alabama: Dana Brasfield, Raymond Lyrene, Lawrence Sindel; Alaska: Dion Roberts; Arkansas: John Carroll, Robert Warren, LouayNassri, Paula Anderson; Arizona: Mark Brown, Amy Silverthorn, Peggy Radford, Gerald Gong, Gregory Legris; California: Gerald Greene, Reddivalam Sudhakar, Arnold Platzker, Bruce Nickerson, Karen Hardy, Ivan Harwood, Gregory Shay, Bryon Quick, Allan Lieberthal, Richard Moss, Chris Landon, Yvonne Fanous, Jay Lieberman, Eugene Spiritus, Bradley Chipps, Ruth McDonald, Mark Pian, Gerd Cropp, Nancy Lewis, Dennis Nielson, Bertrand Shapiro; Colorado: Jeff Wagener, Frank Accurso, Milene Saavedra; Connecticut: Karen Daigle, Jacob Hen, Regina Palazzo; Delaware: Kathryn Dodds, Raj Pad-man, John Goodill; District of Columbia: Glenna Winnie, Lea Davies; Florida: Tony Kriseman, Jorge Sallent, Joseph Chiaro, Martin Kubiet, Sue Goldfinger, Morton Schwartzman, Carlosenrique Diaz, Kevin Maupin, Eduardo Riff, David Geller, Floyd Livingston, Kunjana Mavunda, Jose Birriel Jr, Luis Faverio, David Rosenberg, David Schaeffer, James Sherman, Mary Wagner, Michael Light, Bruce Schnapf; Georgia: Gary Montgomery, Kevin Kirchner, Mark Weatherly, Daniel Caplan, Margaret Guill, Valera Hudson; Illinois: Javeed Akhter, Donald Davison, Steven Boas, Susanna McColley, Youngran Chung, Rennee Latner, Gabriel Aljadeff, Youngran Chan, Jerome Kraut, Arvey Stone, John Lloyd Still, Girish Sharma, Lanie Eagleton, Patricia Hopkins, Umesh Chatrath, Lucille Lester, Young-Jee Kim; Indiana: Veena Anthony, Howard Eigan, Michelle Howenstine, Pushpom James, Edward Gergesha, James Harris, Robert Plant; Iowa: Veljko Zivkovich, Angela Collins, Edward Nassif, Richard Ahrens; Kansas: Daniel Doornbos, Joseph Kanarek, Richard Leff, Pamela Shaw, Elanor Demoss, Maria Riva, Leonard Sullivan; Kentucky: Michael Anstead, Jamshed Kanga, Nemr Eid, Ron Morton; Louisana: Bettina Hilman, Kim Jones, Scott Davis; Maine: Ralph Harder, Tom Lever, Anne Marie Cairns, Edgar Caldwell, Jonathan Zuckerman; Maryland: Peter Mogayzel, Beryl Rosenstein, John McQuestion, Donna Perry, Samuel Rosenberg; Massachusetts: Robert Gerstle, Andrew Colin, Mary Ellen Wohl, Allan Lapey, William Yee, Brian O'Sullivan, Robert Zwerdling; Michigan: Ibrahim Abdulhamid, Adrian O'Hagan, John Schuen, Lawrence Kurlandsky, Richard Honicky, Douglas Homnick, John Marks, Bohdan Pichurko, Norma Maxvold, Samya Nasr, Richard Simon, Wan Tsai, Dana Kissner; Minnesota: John Mc Namara, Nancy Henry, Stephen Marker, Michael Pryor, Warren Regelmann, Lynn Walker; Mississippi: Jim Woodward, Louis Mizell, Suzanne Miller; Missouri: Daniel Rosenbluth, Philip Black, Michael McCubbin, Alan Cohen, Thomas Ferkol, George Mallory, Anthony Rejent, Bruce Rubin, Gavin Graff, Peter Konig; Nebraska: John Colombo, Peter Murphy; New Hampshire: William Boyle, Worth Parker; New Jersey: Chandler Patton, Robert Zanni, Arthur Atlas, Nelson Turcios, Lourdes Laraya-Cuasay, Dorothy Bisberg, Helen Aguila; New Mexico: Sarah Allen, David James, Elizabeth Perkett, Marsha Thompson; Nevada: Sonia Budhecha, Ruben Diaz; New York: Jonathan Rosen, Robert Kaslovsky, Ronald Percciacante, Drucy Borowitz, Joseph Cronin, Colin McMahon, Lynne Quittell, Robert Giusti, Rubin Cohen, Joan DeCelie-Germana, Jack Gorvoy, Kalpan Patel, Meyer Kattan, Allen Dozor, Emily DiMango, Maria Berdella, Ran Anbar, Debra Ianuzzi, James Sexton, Catherine Tayag-Kier, John McBride, Clement Ren, Karen Voter, Mary Dimaio; North Carolina: John Georgitis, Joseph Marc Majure, Maria Martinez, Clarke McIntosh, Margaret Leigh, Michael Schechter, Hugh Black; North Dakota: James Hughes, Anand Kantak; Ohio: Robert Wilmott, Gregory Omlor, Robert Stone, Karen McCoy, James Acton, Carl Doershuk, Michael Konstan, Robert Fink, Michael Steffan, Pierre Vauthy, Patricia Joseph; Oklahoma: Santiago Reyes, John Kramer, James Royall; Oregon: Jay Eisenberg, Michael Wall; Pennsylvania: Stanley Fiel, Thomas Scanlin, Shroti Phadke, Glenna Winnie, Joel Weinberg, William Sexauer, Stephen Wolf, Douglas Holsclaw, Debra Klein, Stuart Warren, Robert Kinsey, Carlos Perez, Muttiah Ganeshanathan, James Shinnick, Howard Panitch, Laurie Varlotta, Cynthia Robinson; Puerto Rico: Jose Rodriguez Santana; Rhode Island: Mary Ann Passero; South Carolina: Jane Gwinn, Robert Baker, Michael Bowman, Patrick Flume, Daniel Brown, Roxanne Marville; South Dakota: James Wallace, Rodney Parry; Tennessee: Don Ellenburg, John Rogers, Ricky Mohon, Joel Ledbetter, Aram Hanissian, Robert Schoumacher, Preston Campbell, Christopher Harris, Bonnie Slovis, Dennis Stokes; Texas: Kathryn Hale, Marcia Katz, Dan Seilheimer, Marianne Sockrider, Allan Frank, James Daniel, James Cunningham, Iley Browning, John Bray, Amanda Dove, Fernando Mandujano, Larry Tremper, Martha Morse, Donna Willey-Courand, Steven Copenhaver, John Pohl, Bennie McWilliams, Marie Martine-Logvinoff, Marsh Wallace, Robert Klein, Rodolfo Amaro, Leslie Couch, Michael Brown, Claude Prestidge, Stephen Inscore, Andrew Lipton; Utah: Barbara Chatfield, Theodore Liou, Bruce Marshall; Virginia: Karl Karlson, Ignacio Ropoll, Thomas Rubio, Joel Schmidt, David Thomas, John Osborn, Deborah Froh, Benjamin Gaston, Greg Elliott; Vermont: Thomas Lahiri, Donald Swartz, Laurie Whittaker; Washington: Ronald Gibson, Bonnie Ramsey, Michael McCarthy, Lawrence Larson, David Ricker, Mark Robbins, Moira Aitken, Julia Emerson;
Wisconsin: Julie Biller, Mark Splaingard, Bradley Sullivan, Paul Pritchard, Stu Adair, Peter Holzwarth, Guillermo Dopico, Keith Meyer, Christopher Green, Michael Rock; West Virginia: Stephen Aronoff, Kathryn Moffett.
Scientific Advisory Group members include: Wayne Morgan (chair), Michael Konstan (co-chair), Ted Liou, Susanna McColley, Ann McMullen, Alexandra Quittner, Warren Regelmann, Clement Ren, Margaret Rosenfeld, Greg Sawicki, Michael Schechter, Donald VanDevanter, Jeffrey Wagener, Marlyn Woo.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.O'Sullivan BP, Freedman SD. Cystic fibrosis. Lancet. 2009;373:1891–904. doi: 10.1016/S0140-6736(09)60327-5. [DOI] [PubMed] [Google Scholar]
- 2.Corey M, Levison H, Crozier D. Five- to seven-year course of pulmonary function in cystic fibrosis. Am Rev Respir Dis. 1976;114:1085–92. doi: 10.1164/arrd.1976.114.6.1085. [DOI] [PubMed] [Google Scholar]
- 3.Kerem E, Corey M, Gold R, Levison H. Pulmonary function and clinical course in patients with cystic fibrosis after pulmonary colonization with Pseudomonas aeruginosa. J Pediatri. 1990;116:714–9. doi: 10.1016/s0022-3476(05)82653-8. [DOI] [PubMed] [Google Scholar]
- 4.Corey M, Edwards L, Levison H, Knowles M. Longitudinal analysis of pulmonary function decline in patients with cystic fibrosis. J Pediatr. 1997;131:809–14. doi: 10.1016/s0022-3476(97)70025-8. [DOI] [PubMed] [Google Scholar]
- 5.Schluchter MD, Konstan MW, Davis PB. Jointly modelling the relationship between survival and pulmonary function in cystic fibrosis patients. Stat Med. 2002;21:1271–87. doi: 10.1002/sim.1104. [DOI] [PubMed] [Google Scholar]
- 6.Schluchter MD, Konstan MW, Drumm ML, Yankaskas JR, Knowles MR. Classifying severity of cystic fibrosis lung disease using longitudinal pulmonary function data. Am J Respir Crit Care Med. 2006;174:780–6. doi: 10.1164/rccm.200512-1919OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Davis PB, Byard PJ, Konstan MW. Identifying treatments that halt progression of pulmonary disease in cystic fibrosis. Pediatr Res. 1997;41:161–5. doi: 10.1203/00006450-199702000-00001. [DOI] [PubMed] [Google Scholar]
- 8.Zemel BS, Jawad AF, FitzSimmons S, Stallings VA. Longitudinal relationship among growth, nutritional status, and pulmonary function in children with cystic fibrosis: analysis of the Cystic Fibrosis Foundation National CF Patient Registry. J Pediatr. 2000;137:374–80. doi: 10.1067/mpd.2000.107891. [DOI] [PubMed] [Google Scholar]
- 9.Konstan MW, Morgan WJ, Butler SM, Pasta DJ, Craib ML, Silva SJ, Stokes DC, Wohl ME, Wagener JS, Regelmann WE, Johnson CA. Risk factors for rate of decline in forced expiratory volume in one second in children and adolescents with cystic fibrosis. J Pediatr. 2007;151:134–9. doi: 10.1016/j.jpeds.2007.03.006. [DOI] [PubMed] [Google Scholar]
- 10.Waters V, Stanojevic S, Atenafu EG, Lu A, Yau Y, Tullis E, Ratjen F. Effect of pulmonary exacerbations on long-term lung function decline in cystic fibrosis. Eur Respir. 2011;40:61–66. doi: 10.1183/09031936.00159111. [DOI] [PubMed] [Google Scholar]
- 11.Sanders DB, Bittner RCL, Rosenfeld M, Hoffman LR, Redding GJ, Goss CH. Failure to recover to baseline pulmonary function after cystic fibrosis pulmonary exacerbation. Am J Respir Crit Care Med. 2010;182:627–632. doi: 10.1164/rccm.200909-1421OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Morgan WJ, Butler SM, Johnson CA, Colin AA, FitzSimmons SC, Geller DE, Konstan MW, Light MJ, Rabin HR, Regelmann WE, Schidlow DV, Stokes DC, Wohl MEB, Kaplowitz H, Wyatt MM, Stryker S. Epidemiologic study of cystic fibrosis: design and implementation of a prospective, multicenter, observational study of patients with cystic fibrosis in the U.S. and Canada. Pediatr Pulmonol. 1999;28:231–41. doi: 10.1002/(sici)1099-0496(199910)28:4<231::aid-ppul1>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 13.Wang X, Dockery DW, Wypij D, Fay ME, Ferris BG., Jr. Pulmonary function between 6 and 18 years of age. Pediatr Pulmonol. 1993;15:75–88. doi: 10.1002/ppul.1950150204. [DOI] [PubMed] [Google Scholar]
- 14.Hankinson JL, Odencrantz JR, Fedan KB. Spirometric reference values from a sample of the general U.S. population. Am J Respir Crit Care Med. 1999;159:179–87. doi: 10.1164/ajrccm.159.1.9712108. [DOI] [PubMed] [Google Scholar]
- 15.Fuchs HJ, Borowitz DS, Christiansen DH, Morris EM, Nash ML, Ramsey BW, et al. Effect of aerosolized recombinant human DNase on exacerbations of respiratory symptoms and on pulmonary function in patients with cystic fibrosis. The Pulmozyme Study Group. N Engl J Med. 1994;331:637–42. doi: 10.1056/NEJM199409083311003. [DOI] [PubMed] [Google Scholar]
- 16.Rosenfeld M, Emerson J, Williams-Warren J, Pepe M, Smith A, Montgomery AB, et al. Defining a pulmonary exacerbation in cystic fibrosis. J Pediatr. 2001;139:359–65. doi: 10.1067/mpd.2001.117288. [DOI] [PubMed] [Google Scholar]
- 17.Rabin HR, Butler SM, Wohl ME, Geller DE, Colin AA, Schidlow DV, Johnson CA, Konstan MW, Regelmann WE. Epidemiologic Study of Cystic Fibrosis. Pulmonary exacerbations in cystic fibrosis. Pediatr Pulmonol. 2004;37:400–6. doi: 10.1002/ppul.20023. [DOI] [PubMed] [Google Scholar]
- 18.Fletcher C, Peto R. The natural history of chronic airflow obstruction. Br Med J. 1977;1:1645–1648. doi: 10.1136/bmj.1.6077.1645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wise RA. The value of forced expiratory volume in 1 second decline in the assessment of chronic obstructive pulmonary disease progression. Am J Med. 2006;119:S4–11. doi: 10.1016/j.amjmed.2006.08.002. [DOI] [PubMed] [Google Scholar]