Abstract
Actinic keratoses (AKs) are common skin lesions heralding an increased risk of developing squamous cell carcinoma (SCC) and other skin malignancies, arising principally due to excessive ultraviolet (UV) exposure. They are predominantly found in fair-skinned individuals, and increasingly, are a problem of the immunosuppressed. AKs may regress spontaneously, remain stable or transform to invasive SCC. The risk of SCC increases for those with more than 5 AKs, and the majority of SCCs arise from AKs. The main mechanisms of AK formation are inflammation, oxidative stress, immunosuppression, impaired apoptosis, mutagenesis, dysregulation of cell growth and proliferation, and tissue remodeling. Human papilloma virus has also been implicated in the formation of some AKs. Understanding these mechanisms guides the rationale behind the current available treatments for AKs. One of the main principles underpinning the management of AKs is that of field cancerization. Wide areas of skin are exposed to increasing amounts of UV light and other environmental insults as we age. This is especially true for the head, neck and forearms. These insults do not target only the skin where individual lesions develop, but also large areas where crops of AKs may appear. The skin between lesions is exposed to the same insults and is likely to contain as-yet undetectable preclinical lesions or areas of dysplastic cells. The whole affected area is known as the ‘field’. Management is therefore divided into lesion-directed and field-directed therapies. Current therapies include lesion-directed cryotherapy and/or excision, and topical field-directed creams: 5-fluorouracil, imiquimod, diclofenac, photodynamic therapy and ingenol mebutate. Combining lesion- and field-directed therapies has yielded good results and several novel therapies are under investigation. Treatment is variable and tailored to the individual making a gold standard management algorithm difficult to design. This literature review article aims to describe the rationale behind the best available therapies for AKs in light of current understanding of pathophysiology and epidemiology. A PubMed and MEDLINE search of literature was performed between January 1, 2000 and September 18, 2013. Where appropriate, articles published prior to this have been referenced. This is not a systematic review or meta-analysis, but aims to highlight the most up to date understanding of AK disease and its management.
Electronic supplementary material
The online version of this article (doi:10.1007/s13555-014-0049-y) contains supplementary material, which is available to authorized users.
Keywords: Actinic keratosis, Cryosurgery, Dermatology, Diclofenac, Field cancerization, 5-Fluorouracil, Imiquimod, Ingenol mebutate, Photodynamic therapy
Introduction
Pathophysiology
Actinic keratoses (AKs) are defined clinically as erythematous, scaly plaques that occur on sun-damaged skin [1] and are a result of exposure to ultraviolet (UV) radiation. They are typically located on the face, scalp, neck and extremities [2–4]. Their potential for malignant transformation is well documented: they are known to be precursors of squamous cell carcinoma in situ (Bowen’s disease) and squamous cell carcinoma (SCC) [5].
Histology
Actinic keratosis is a proliferation of neoplastic keratinocytes limited to the epidermis, characterized by architectural disorder [6, 7]. These features include abnormal keratinocytes of the basal layer that are variable in size and shape, nuclear atypia and hyperkeratosis of the epidermis. Atypical nuclei are enlarged, irregular, and hyperchromatic.
Any single AK lesion may have one of three outcomes. It will enter spontaneous remission [8], remain stable, without further progression, or transform to invasive squamous cell carcinoma (SCC) which may rarely metastasize [7].
The risk of metastasis in invasive SCC cannot be underestimated, despite variable reporting of rates. For example, in 2012, a study was published by Brougham et al. [9] who examined pathology results from 1997 to 2007 in a large New Zealand retrospective cohort. Of 6,164 patients, they found that cutaneous SCC (cSCC) metastasizes in 1.9–2.6% of cases. Certain factors such as larger diameter, location on the ear and retro-auricular area, poor differentiation and perineural invasion of the primary lesion are indicators of increased risk and should be identified. In this study, 87% of metastases are to regional lymph nodes, which is reflective of general statistics. The risk of nodal metastases ranges from 2.0 to 5.8% [10, 11] in a recent US single-institution 10-year cohort, and the risk of disease specific death is 2.1% in anyone diagnosed with cSCC [12].
The risk of progression of AKs is reported in widely variable figures. Some studies estimated the risk at 0.075–0.096% per lesion per year, or about 1% over 10 years [13], with some estimates as high as 10% over 10 years [14]. However, the Veterans Affairs Topical Tretinoin Chemoprevention Trial found that the risk of progression of AK to primary SCC (invasive or in situ) was 0.6% at 1 year and 2.57% at 4 years [5]. This study found that the majority of AKs regressed and, at the end of 5 years, 70% were no longer present. Other earlier studies have found that the risk of progression to invasive disease is approximately 0.025–16% per year for any single lesion [14]. For a patient affected by multiple AK lesions, it has been suggested that the annual risk of developing invasive cutaneous SCC lies between 0.15% and 80% [15]. The relative risk of SCC increases for those with more than five AK [16]. Whilst it is not possible to predict which AK lesions will go on to become SCC, histologic evidence shows that the majority of SCC arises from AK lesions [17].
Genetics
Actinic keratosis results from the adverse effects of UV radiation on keratinocyte DNA. The changes that occur reduce skin immunity and allow the development of AK [18, 19]. Some important mutations associated with an increased risk of progression to SCC are p16 (INK4a) (on chromosome 9p21) [20], p14 (ARF), p15 (INK4b) and p53. These are implicated in the development of AK and in progression of AK to SCC [21]. UV-A (320–400 nm) light is the most abundant and penetrates skin more deeply than UV-B. Here it causes oxidative damage to nucleic acids, membrane lipids and cell proteins through production of reactive oxygen species (ROS) [22]. These ROSs interrupt normal cellular transduction pathways and cell–cell signaling, causing altered proliferation [23]. The signature mutations of UV-A are thymine (T) → guanine (G), due to the formation of 8-hydroxyguanine adducts. UV-B (290–320 nm) irradiation directly causes the formation of cyclobutane pyrimidine dimers and 6-4 photoproducts, which in turn give rise to the characteristic cytosine (C) → T and CC → TT mutations [24].
Inactivation of p53, a gene coding for a tumor suppressor protein, by UV-B light is a crucial step in the path of creating genetically unstable keratinocytes [12]. Mutations in the p53 gene have been found in >90% of human cutaneous SCCs [12]. P53 in its functional form has been found to protect against skin cancer induction by UV light in mice [25]. In the absence of functional repair genes, such as p53, other DNA mutations go on to promote carcinogenesis. Absorbed UV light increases the production of arachidonic acid and its metabolites, and other proinflammatory cytokines. Reactive oxygen species induce lipid peroxidation and cellular destruction [26, 27].
The main mechanisms in the formation of AK, therefore, are inflammation, oxidative stress, immunosuppression, impaired apoptosis, mutagenesis, dysregulation of cell growth and proliferation, and tissue remodeling [6]. This knowledge is the basis of medical management for AK.
Due to the ability of AK to regress or lead to SCC it has traditionally been viewed as premalignant, but there have been publications suggesting that reclassification as ‘cancerous’ would be appropriate [28, 29]. Cockerell proposed using the ‘keratinocyte intraepidermal neoplasia’ (KIN) nomenclature, in keeping with the classification used for cervical intraepithelial neoplasia [32].
More recently, infection with cutaneous human papilloma virus (HPV) has been associated with the formation of AK [30]. The exact mechanism is incompletely understood, but it has been found that the E6 protein of cutaneous HPV can contribute to reduced levels of Bak protein. This protein has pro-apoptotic effects and is usually activated as a protective mechanism in keratinocytes upon exposure to UV light [31]. Further discussion is beyond the scope of this article, but it is an important developing area for research into preventative measures.
It is a challenge to distinguish between AK and early SCC especially, as they share many cellular and histopathological features. A commonly used definition is based on the relative depth of dysplastic cells within the skin. AKs can be defined anatomically as an epidermal lesion of the basal layer that may extend upwards to involve the granular and cornified layers [32]. Disease begins in the basal layer of the epidermis, as this is the site of dividing cells. Bowen’s disease (squamous cell carcinoma in situ) lies at the more extensive end of the epidermal involvement spectrum, but remains cellularly largely indistinguishable from AK. Invasive SCC is defined by extension beyond the epidermis, through the basement membrane and into the dermis. However, the diagnosis of Bowen’s disease must be considered as this is a malignant tumor contained within the epidermis with potential for significant lateral spread and of becoming invasive. It is also a challenge to distinguish between Bowen’s disease and SCC histologically, especially when the SCC is well differentiated and dermal protrusions are smooth edged.
In more recent advances, using a technique of gene expression profiling, Padilla et al. [33] showed convincingly that AK is a precursor lesion of SCC, and that they are closely related genetically. This study lends credence to the general consensus that AK lies on a spectrum between normal skin and SCC. It has also been shown that reduced p53 staining in immunohistochemical analysis of AK is associated with a greater probability of those lesions developing into SCC [34]. This may help in future to predict which biopsied lesions are more likely to require early or surgical intervention.
Epidemiology
AKs are common among fair-skinned individuals, and are particularly prevalent in areas with high levels of sun exposure. In Australia, AKs are found in 40–50% of the Caucasian population over the age of 40 years [35], and reported as 40–60% by Frost and Green [36]. The prevalence in the United States ranges from 11 to 26% [37], whilst in Europe 15% of men and 6% of women have been reported to be affected [37].
Those who sunburn easily and have difficulty tanning (Fitzpatrick skin type I and II) are most at risk of the damaging effects of UV irradiation. Other risk factors include older age, male gender, cumulative UV exposure, living closer to the equator and immunosuppression [38]. Prevalence of AKs is very much age related [39], reflecting the incidence and recurrence rates that exceed the rate of regression as people age [40].
Individuals with AKs tend to have 6–8 lesions on average [15]. AKs develop most commonly on the head, balding scalp, face, dorsal forearms and hands [41]. It has been found in one UK study that 75% of all reported lesions occur on the head, neck and forearms [42]. Immunocompromised patients notably have a greatly increased risk of AK. Organ transplant recipients who are on immunosuppressive medications are up to 250 times more likely to develop AK [43].
Due to the high prevalence of AK and the risk for malignant change, AK is an important subject to address for any physician, but especially those in primary care and dermatology. It is likely that most physicians will be in a position to help prevent either the serious, or disfiguring sequelae of AK at some point in their career, be it by referral or treatment.
Discussion
Management
The most important issue in the management of AKs is to distinguish between isolated lesions and multiples, or the occurrence of ‘field cancerization’. There is no current method to predict which AK within any given cluster will progress to invasive malignancy. Due to the tendency of AKs to develop in certain areas of skin such as the head and neck, the term ‘field’ has been coined to describe the area of skin affected. Treatment of the whole ‘field’ targets visible lesions and also skin which is AK-adjacent, which is therefore at an increased risk of developing AKs having been exposed to similar risk factors.
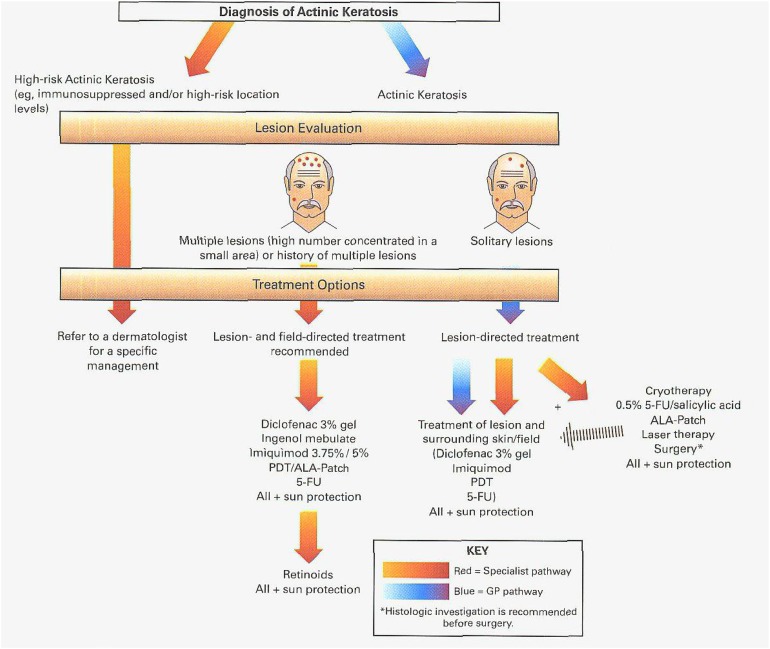
‘Field cancerization’ is a term that was originally coined by Slaughter et al. in 1953 [44, 45] referring to histologically abnormal epithelium adjacent to tumor tissue (within the aerodigestive region) and was designed to explain the occurrence of multiple primary tumors as well as locally recurrent cancer. In AK it is thought that preclinical damage to the epidermis should be treated to prevent progression to more advanced disease. Once a patient is diagnosed with AK, it is necessary to decide the first steps in management. An algorithm showing recommended treatment options is shown in Fig. 1.
Fig. 1.
Decision-making steps in the management of actinic keratosis (AK) [2]. Copied and modified with permission from, Stockfleth [2], and from original publication Stockfleth et al. [133]. 5-FU 5-fluorouracil, ALA 5-aminolevulinic acid, PDT photodynamic therapy
A Comparison of International Guidelines for Management of AKs
In a comparison of international recommendations [46–49] on the management of AKs from Australia, the US, the UK and Europe, it was found that for single lesions Australian, European and American guidelines recommend cryotherapy; whereas, the British guidelines were notably the only set that specify that no therapy or sunblock alone would be an appropriate course of action for mild or thin lesions. The British guidelines recommend that if there are patient or clinical concerns for single lesions then salicylic acid followed by 5-fluorouracil (5-FU), imiquimod, diclofenac, tretinoin or cryotherapy is the treatment of choice. Australian and European guidelines for single lesions also include 5-FU and photodynamic therapy (PDT), respectively.
For hyperkeratotic lesions, Americans and Europeans recommend dermabrasion alone, but Australians and British suggest curettage, with Australians going on to include double-freeze cryotherapy and surgery.
For multiple lesions, there appears to be wide consensus between the Americans, British, Europeans and Australians on the use of the field therapy 5-FU. Australia, US and Europe include imiquimod for multiple lesions while the UK recommends PDT and diclofenac. PDT is also recommended by the Australians. Resistant AKs are treated with 5-FU, imiquimod, PDT or a diclofenac/cryotherapy combination in Australia, whilst this is dealt with by surgical excision or curettage in the UK and imiquimod in the US. For situations where surgery or other therapies are inappropriate, Australian guidelines recommend imiquimod or PDT, Europeans suggest retinoids and the US recommends persisting with 5-FU.
It is currently not considered appropriate to make direct comparisons of efficacy between different therapies for AKs due to the wide variability of the studies performed. Numbers of patients and study designs are not currently standardized, making valid comparisons very difficult. With current knowledge, a chart or table aiming to make this comparison may be misleading until further studies are performed using standardized trial structures. The authors of this paper recommend this as a valuable direction for future research. It is noted, however, that a meta-analysis of 8 interventions for AKs by Gupta and Paquet [50] was published in August 2013. This is a follow-up on a prior Cochrane review of the same treatments. The results indicate that 5-FU is the most efficacious treatment followed by a combination treatment of 5-aminolevulinic acid (ALA) PDT; imiquimod; ingenol mebutate and 5-methylaminolaevulinate (MAL) PDT. The remaining treatments of cryotherapy, diclofenac with hyaluronic acid and placebo gave a lower efficacy ranking, respectively.
As seen in the previous discussion, AK can present as a single lesion, or multiple lesions within a field. Treatment can therefore be divided up into lesion-specific therapy, or field-directed therapy. Surgical solutions are aimed at individual AK lesions (or small clusters of identifiable AKs), and field therapy is most usefully delivered topically, or orally in very advanced cases that include the development of multiple SCCs. Often, a combination of lesion-specific and field-directed therapies is the most effective approach, when tailored appropriately to the patient’s clinical picture. The advantage of topical field-directed therapy is the ability to treat subclinical lesions that are not visible to the naked eye.
Lesion-Directed Therapy
Cryotherapy
Cryotherapy or cryosurgery with liquid nitrogen for individual lesions is quick, and easily performed by the physician [51]. This is the most common ablative therapy for AKs [52]. However, at present there is no widely accepted standard method of application and further studies are required to guide recommendations more effectively.
The treatment is delivered by either spray or contact, destroying the epidermal keratinocytes by freezing. Keratinocytes die at approximately −40 to −50 °C, liquid nitrogen is delivered at −195.8 °C. Dermal structures such as collagen, nerves and blood vessels are preserved due to their relative tolerance of low temperatures.
Success of treatment is operator dependent. The correct method is to freeze the epidermis, creating an ice ball. A blister should then form, providing evidence that the basement membrane has separated from the dermis. This technique, on thinner lesions, has been shown to yield 90% clearance at 6 months [53].
Thai et al. [52] performed a prospective, multi-center study of 90 patients, evaluating the efficacy of cryosurgery for AK. They found that the overall complete response rate (100% lesion clearance) was 67.2%. This was relative to freeze time (from formation of ice ball to commencement of thawing), and revealed that there was 39% response for <5 s of freezing. The rate was up to 83% for freezing periods >20 s. Most physicians would suggest that a freezing time between 5 and 10 s is sufficient to optimize the tradeoff between efficacy and side effects.
The cost is low and therapy is generally well tolerated by patients. The procedure is uncomplicated and adequately clears abnormal tissue for identifiable lesions [54].
Disadvantages include pain and erythema, and reduced efficacy in hyperkeratotic lesions [54]. Aggressive therapy may cause depigmentation and scarring [52, 55]. Hypopigmentation occurs in 29% of cleared lesions, while hyperpigmentation may occur in up to 6% of cases [52]. This is due to the susceptibility of melanocytes to freezing. Individual lesions are treated, without consideration for subclinical epidermal changes in between lesions, and it cannot be used for large areas. Repeat treatment is therefore often necessary as recurrence rates range from 1.2 to 12% within 12 months [47].
Surgical management such as curettage, shave excision or conventional excision are normally reserved for hyperkeratotic lesions or suspected invasive SCC. Surgical field-directed therapies such as laser resurfacing, chemical peels and dermabrasion are effective for eradication of AK. However, they carry a small risk of infection and widespread scarring, and are not frequently performed solely for the management of AK [56, 57].
There is currently no clear guideline regarding the specific density of AK lesions per unit skin area that would indicate a move from lesion-directed to field-directed therapy, and this is usually a decision made on clinical judgment. Moving from field-directed topical therapies to lesion-directed surgical excision would be indicated if there was evidence of invasive SCC.
Field-Directed Therapy
Currently available agents are 5-FU, imiquimod, diclofenac, PDT and ingenol mebutate.
5-Fluorouracil Cream: Anti-neoplastic Agent
Topical 5-fluorouracil (5-FU) has had a place in the treatment of AK for many decades [58]. It is a pyrimidine analog, which disrupts DNA formation by stopping the conversion of deoxyuradilic acid to thymidylic acid [59]. This prevents cell proliferation preferentially in rapidly dividing cells, especially those of AKs and basal layers of the epithelium. 5-FU is available as a cream in 5, 1 and 0.5% concentrations, and as a solution in 5% and 2% concentrations. It is the most established field treatment for AK, and is considered by some the traditional gold standard to which all other topical agents are compared [1]. The typical treatment regime is either 5% cream twice daily or 0.5–1% cream daily for 2–4 weeks. The widespread application has the advantage of treating clinically undetectable AKs.
5-FU causes inflammation, erosion, and ulceration during treatment, which is necessary for therapeutic success. For a standard 3- to 4-week period of twice daily application, these side effects arise after the first week and subside approximately 2 weeks after application has ceased, when re-epithelialization has occurred. These transient side effects can result in non-compliance.
0.5% 5-FU cream was developed in an attempt to reduce the inflammation associated with application. It is applied once daily for a month. In 2010, Kaur et al. [60] analyzed clinical trials from 1965 to 2009 to compare the efficacy of 5% and 0.5% 5-FU cream in treating multiple AKs of the face and scalp. They found that after 4 weeks of treatment, complete clearance for 0.5% 5-FU ranged from 16.7% to 57.8%, and for 5% 5-FU clearance ranged from 43% to 100%. The 5% 5-FU cream had a higher rate of adverse events. It was also stated that there is lack of high-powered clinical trials comparing both groups. However, it appears that 5% 5-FU gives the greater chance of complete clearance despite side effects and should therefore be the preferred treatment.
In 2012, Rhavar et al. [61] performed a systematic review of 103 studies into the efficacy of 0.5% 5-FU. Only four studies were found to be suitable randomized vehicle-controlled trials. Of 668 patients, the percentage achieving complete clearance of their AKs in the 5-FU group was 19, 28.2 and 52.6% in the 1-, 2- and 4-week-treatment groups. Only 0.85% of those in the vehicle-treated group reached complete clearance. The mean lesion count reduction was 90.2% and 28.3% in the 5-FU and vehicle groups, respectively. It was found by Yentzer et al. [62] that 0.5% 5-FU cream had a high adherence rate of 86% to the once daily application over a 4-week period. After 4 weeks of twice daily application of 5% 5-FU, sustained complete field clearance at 12 months was seen in 33% of patients [63].
A number of different methods of reducing side effects have been tried. The application of topical corticosteroids 15 min after 5-FU cream can be helpful to reduce the inflammatory response, and intermittent use of 5-FU cream can cause reduced side effects.
Intermittent, or pulsed 5-FU application is not as widely used currently as previously, but has been shown to reduce side effects, whilst maintaining efficacy for the treatment of AK [64, 65]. Labandeira et al. [64] studied the effects of four applications per week for the first week, followed by two applications per week if irritation was intolerable. One disadvantage to this regime is that the time to complete healing of lesions is prolonged with reduced frequency of application, but remains moderately effective in treating AKs.
The use of corticosteroid creams and pulsed therapy may reduce the efficacy of 5-FU treatment [66]. Side effects can also be minimized in practice by choosing to treat smaller areas only, or visible lesions only after the first week. Exposure to sunlight should be minimal as the treated area is photosensitive and exposure can cause pain.
Imiquimod: Immunomodulator Agent
Imiquimod is an imidazoquinolone immune response modifier that acts on both innate and acquired immunity. It is a toll-like receptor-7 agonist that modifies the immune response in the skin and stimulates apoptosis, thereby disrupting tumor proliferation. Additionally, it induces E-selectin on tumor vessels and consequent infiltration by cutaneous lymphocyte-associated antigen-positive skin-homing cluster of differentiation (CD8+) cytotoxic T cells, and results in histological evidence of tumor regression [67] and a reduction in tumor cell numbers [68]. It triggers a range of proinflammatory cytokines including interferon alpha, tumor necrosis factor alpha (TNF-α), and interleukin-12 (IL-12) [69].
It was first approved as a treatment for AKs in 2004 as a 3 times per week application, on the bald scalp and face, for up to 16 weeks. This has been more recently modified to a twice weekly application, in 4-week application cycles, with a 4-week rest period between these cycles to minimize treatment times and adverse events. A 3.75% cream was first approved in 2010 for once-nightly application for 2 weeks, followed by a 2-week rest period without application, which is then followed by another 2-week treatment period [70].
Available preparations are 5% and 3.75%. The 3.75% preparation allows for a shorter duration of treatment over a larger skin surface area (200 cm2 vs. 25 cm2 for 5% cream) [71]. Absolute clearance rate is higher for the 5% cream, at 45%, compared to 35% for daily use of the 3.75% preparation [70], and the median reduction in AK is 83% [72]. Use of the 5% cream can give up to 57% clearance if used three times per week for 16 weeks [73–75].
Cycle therapy is aimed at reducing local skin reactions. In some studies, 5% Imiquimod was applied 2–3 times weekly for 3–4 weeks, and then reviewed after 4 weeks, with repeated treatment if there were any residual lesions [76, 77]. Complete clearance was achieved in up to 82% of treatment areas. Imiquimod has also been used in immunocompromised patients with good results, and is generally regarded as safe to use in this sub-group [78].
In 2010, Hanke et al. [79] performed two placebo-controlled studies of daily application of imiquimod 2.5% and 3.75% for the treatment of AKs, for two 3-week cycles. This looked at the balding scalp and face only. Up to two packets of 250 mg each, were applied per dose once daily, for two 3-week treatment cycles, with a 3-week no-treatment interval. Efficacy was assessed at 8 weeks post-treatment and found that clearance rates for both treatment options were superior to placebo and had an acceptable safety profile. Complete clearance in the 2.5% cream group was 25%, and in the 3.75% cream group was 34%. Partial clearance rates were higher.
A similar study by Swanson et al. [80] was done to compare efficacy of 2.5% and 3.75% cream in two 2-week cycles. Complete clearance rate with daily application was 30.6% for 2.5% cream and 35.6% for 3.75%, which would suggest that a shorter interval between applications improves outcome. Side effects include skin irritation and erythema, with rarer consequences being flu-like symptoms and lymphadenopathy [81].
In a randomized trial comparing imiquimod against cryotherapy in the treatment of AK over a 12-month period, it was found that repeated cryotherapy (up to 4 sessions) achieved a higher complete lesion clearance rate (85% vs. 66.9%), while cosmetic outcome was better with imiquimod (likely related to lower incidence of hypopigmentation) [82]. In another split-face study comparing imiquimod against PDT, the authors found that there was no significant difference in 100% or 75% response to either treatment regimen, but mean lesion reduction rate was superior in the PDT group [83].
Tolerability of 5% imiquimod was compared to that of MAL–PDT in a randomized controlled trial (n = 58) [84]. The patients were asked for example, about pain, side effects and overall satisfaction. Overall, the two treatments were approximately equitable with the PDT group scoring themselves as ‘very satisfied’ slightly more frequently.
Recommended therapy for the face and scalp, or large areas up to 200 cm2, could therefore usefully be suggested as a short course of 3.75% imiquimod cream daily for 2-week cycles, twice, to give 35.6% of patients complete clearance. Licensed use of 5% cream is for small skin areas up to 25 cm2, 2–3 times per week for up to 4 months, although 1 month is usually sufficient [85]. Evidence suggests 12- to 16-week treatment will give complete clearance in 50% of patients [86]. If inflammation is intolerable, frequency of application can be reduced to once or twice per week with preservation of efficacy. The addition of lesion-directed cryosurgery prior to application gives greater clearance than either therapy alone [79].
There is evidence to suggest that patients may develop T cell memory after treatment with imiquimod, which is likely to reduce the risk of developing further AKs. The 5% preparation has also been shown to be effective and safe in solid organ transplant patients covering up to 100 cm2 skin surface area [78, 87].
A sister drug called resiquimod, which is currently an investigational product, is 10–100 times more potent than imiquimod. A European phase II study of daily applications 3 times per week for 4 weeks found that clearance rates ranged from 40 to 74.2%. The lower concentrations were better tolerated and as effective as the higher concentrations [88].
Diclofenac Sodium 3% Gel: Anti-inflammatory Agent
Diclofenac 3% gel is a nonsteroidal anti-inflammatory formulated with 2.5% hyaluronic acid. It is a popular treatment in Germany, but not widely used in Australia. Diclofenac is a cyclooxygenase (COX) inhibitor exerting its antitumor effects via inhibition of the COX-2 pathway and by inhibiting up regulation of the arachidonic acid cascade. The production of prostaglandins from arachidonic acid may play a role in UV-B-induced skin cancer [basal cell carcinoma (BCC) and SCC] [89], and diclofenac’s inhibition of this cascade may explain its efficacy in AK treatment. This effect may be mediated via inhibition of angiogenesis and induction of apoptosis. There is evidence that diclofenac induces regression of AKs [90, 91].
A 2005 meta-analysis of three randomized trials (n = 364) found that treatment of AKs with diclofenac gel led to complete resolution in approximately 40% of patients, whereas there was only a 12% rate of complete resolution of those treated with placebo [92]. Diclofenac resulted in 100% AK resolution in 50% of the subjects after 3 months of twice daily application. This extended period of treatment reduces compliance, but a shorter duration is reportedly less efficacious [93]. Diclofenac treatment is well tolerated with minimal irritation and inflammation. Adverse reactions can include itch, xerosis, and contact dermatitis [94].
Recommended dosing is twice per day for 90 days. When it is used after cryotherapy, diclofenac has been shown to give greater complete lesion clearance compared to cryosurgery alone (64% vs. 32%, respectively) [95]. Tolerance is better than the twice daily application of 5-FU cream, but appears to be slightly less effective when used alone. Long-term data are restricted to a single uncontrolled study [96] which shows similar efficacy at 12 months to ingenol mebutate (18% complete clearance compared with 19.5%, respectively).
Photodynamic Therapy (PDT): Photosensitizing Agent
Photodynamic therapy is a two-step procedural field therapy, beginning with the topical application of a photosensitizing agent to the treatment area.
Photosensitizing agents are 5-ALA prescribed in the form of an 8 mg adhesive patch, under red light (630 nm); a 78 mg/g nanoemulsion gel, under red light (630 nm) or a 20% solution under blue light (417 nm) and MAL in the form of a 16.8% cream, under red light (630 nm). Licensing of these topical products varies between countries, for example MAL is widely available worldwide, but is the only preparation available in Australia. The 5-ALA 20% solution is only licensed in the US, Korea, Brazil, Mexico, Argentina, Chile and Columbia. These prodrugs are converted by the heme biosynthetic pathway to protoporphyrin IX (PpIX).
After an incubation period whereby the prodrug accumulates preferentially in dysplastic actinic keratosis cells, the area is then illuminated by an appropriate wavelength light. This causes the activation of PpIX and produces reactive oxygen species. The ROSs produced upon light exposure cause apoptosis and necrosis of target tissue, resulting in cell death [97].
Treatment is currently aimed at patients who have had difficulty adhering to topical field therapies, AK lesions resistant to topical therapies or those with concerns regarding the cosmetic results of treatment [2]. A typical regime for nonhyperkeratotic, non-pigmented AKs would be the direct application of 20% ALA topical solution to lesions on the face or the scalp for 18–24 h, followed by blue light for 16 min and 40 s; or 16.8% MAL application for 3 h followed by red light for 7–10 min [98]. A follow-up treatment is recommended for lesions that have not completely resolved after 8 weeks. A single treatment is used for thin or moderately thick lesions, repeated after 3 months if not clinically cleared [99, 100].
PDT is usually well tolerated and clearance rates of up to 90% after two applications have been reported [101]. MAL is used most commonly internationally as it selects and penetrates dysplastic cells better than other commercially available products [102]. PDT with MAL achieved 100% resolution in up to 82% of subjects and cleared 90% of total AK lesions in a 2008 randomized, double-blind, placebo-controlled study by Pariser et al. [103].
In a recent study evaluating the effect that ablative fractional laser resurfacing (AFXL) has on improving PDT efficacy, it was found that AFXL-assisted PDT was significantly more effective than PDT alone (complete lesion clearance rate of 88% vs. 59% for grade 2–3 AK). The authors concluded that AFXL-assisted PDT has a strong potential in treating AK, especially thick lesions in field-cancerized skin [104].
Wiegell et al. 2008 [105], and 2009 [106] demonstrated that natural daylight photodynamic therapy (DL-PDT) provides similar AK clearance to conventional PDT (c-PDT), is almost painless and is much simpler to perform. DL-PDT involves the application of MAL without occlusion, then 30 min later going outside into daylight for 2 h, after which MAL is removed.
New formulations of ALA/MAL are being trialed, and a recent multi-center study of patients with mild-to-moderate AK to the scalp/face found that PDT with BF-200 ALA (Biofrontera, Leverkusen, Germany) (an ALA nanoemulsion that improves ALA stability and skin penetration) was superior to placebo for complete clearance of 78.2%, and lesion complete clearance rate of 90.4% at 3 months post last PDT [107].
A self-adhesive, skin colored, thin 5-ALA patch, applied directly to AK lesions without crust removal was superior to cryotherapy in clearing mild/moderate AK [108]. Side effects of PDT include erythema, itching, edema, exudation, and pain during exposure to light. Local anesthetic nerve blocks or air-cooling for pain relief may be required during PDT if the discomfort is intolerable, with superior analgesia found in those using nerve blocks [109, 110]. The duration of healing rarely exceeds 10 days. The significant advantage of PDT is high satisfaction with respect to cosmetic outcomes [98]. The optimal incubation times for ALA/MAL and optimal light sources for PDT are under ongoing investigation. Despite this, it remains an effective lesion- and field-directed therapy.
Ingenol Mebutate: Anti-neoplastic Agent
Ingenol mebutate is a new, Australian-developed topical therapy, a diterpene ester found in the sap of the Euphorbia peplus (petty spurge) plant. It was approved for the treatment of AK in the USA in 2012 and Australia in early 2013, but has not yet been included in the current guidelines. Ingenol mebutate differs from all other current topical treatments in that it has a dual mechanism of action, which may account for the efficacy after a much shorter treatment period. It is currently available in two preparations of 0.015% and 0.05%.
Firstly, there is rapid cellular necrosis through the disruption of the plasma membrane and mitochondrial swelling. This is detectable within 1 h from onset of treatment [111] and leads to cell death within 24 h in mice [112].
Secondly, there is a specific neutrophil-mediated, antibody-dependent cellular cytotoxicity (ADCC), which targets any remaining dysplastic epidermal cells [111]. Antibodies produced by B cells bind to antigens on dysplastic epidermal cells, and these then bind neutrophils which trigger their cytotoxic mechanisms. ROS are released, amongst other lytic agents, which causes the destruction of the dysplastic epidermal cells.
Ingenol mebutate therefore induces both chemo-ablative and immunostimulatory effects after topical application. The rapid destruction of AK lesions means treatment is necessary for only 2 or 3 days: an unusually short duration of treatment for a topical field therapy. A phase IIa study by Siller et al. [113], with 58 patients was conducted in Australia. Five preselected lesions were treated with ingenol mebutate gel 0.0025, 0.01 or 0.05%, or vehicle gel, on days 1 and 2 (Arm A), or days 1 and 8 (Arm B). The study showed that there were no significant differences in tolerability or efficacy in patients either treated 1 day or 7 days apart. Treatment was well tolerated.
The most common local skin reactions were erythema, scaling, and crusting. The highest dose used (0.05%) achieved highest efficacy, with clinical clearance of 71% of treated lesions. Of all patients treated with 0.05% gel, 67% had clinical clearance of at least four out of five lesions [113].
In another study, a randomized, double-blind, double-dummy, vehicle-controlled trial was conducted across 22 centers in the USA, where ingenol mebutate was assessed at three dosing regimens for non-facial AK. The three different treatment regimens in that trial were significantly more effective than vehicle in clearing AK. The partial clearance rates ranged from 56% to 75.4% (vs. 21.7% for vehicle), complete clearance rates ranged from 40% to 54.4% (vs. 11.7% for vehicle), and median percentage decrease in baseline AK ranged from 75% to 100% (vs. 0% for vehicle). All arms of active treatment were well tolerated [114].
Lebwohl et al. [115] found that complete clearance of AK on the face and scalp treated with ingenol mebutate 0.015% gel for 3 days was 42.2% at the 8-week follow-up visit, and 34.1% for AKs on the trunk and extremities using ingenol mebutate 0.05% gel for 2 days. Local reactions of erythema, scaling, vesiculation, depigmentation, swelling, pruritus and crusting peaks at 4–8 days, and may rarely last up to 30–55 days [115]. The observational follow-up trial 12 months later showed that a mean of 86% of the number of lesions in the treatment area at baseline were still clear. Approximately half the patients developed a recurrence of one or more lesions in the treated field.
A long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses by Lebwohl et al. [116] was completed in 2013. The results showed that sustained clearance after 12 months was 46.1% for patients treated on the face or scalp with 0.015% gel for 3 consecutive days, and 44.0% for those treated on the trunk/extremities with 0.05% gel for 2 consecutive days. This is considerably better than the 12-month cryotherapy complete clearance rate of 4% as described by Krawtchenko et al. [63]. The estimated median times to new or recurrent lesions in the treatment area are: 365 days for the face or scalp and 274 days for the trunk or extremities [116]. The recommended approved therapy is therefore 0.015% gel to the face or scalp for 3 consecutive days, and/or 0.05% gel to the trunk or extremities for 2 consecutive days. Follow-up can usually be recommended at 12 and 9 months, respectively, due to recurrence rates.
Combining Therapeutic Modalities
Until more recently, there had been little research into combined therapies for AK. However, due to the increasing number of treatment modalities, more effective combinations are being studied [117–119].
An obvious choice would be to combine lesion-directed cryotherapy and a field-directed topical therapy. The use of 0.5% 5-FU cream 1 week prior to cryosurgery has been shown to produce complete lesion clearance in a greater proportion of patients than those treated with cryosurgery alone (32.4% vs. 15%, respectively) [120].
In a multi-center North American study, cryosurgery followed by two cycles of 3.75% imiqumod applied daily for 2 weeks was evaluated for the treatment of facial AK. This was compared against a cryosurgery/placebo group. The results showed that the median total AK reductions were 86.5% and 50%, and the complete clearance rates were 30.2% and 3.3% for the cryosurgery/imiquimod and cryosurgery/placebo groups, respectively. Less than 5% of subjects in each arm discontinued the study due to adverse events. The authors concluded that a short cyclical field treatment of imiquimod following cryosurgery was well tolerated and proven to be more effective than cryosurgery alone [118]. In another study, more patients treated with cryotherapy plus imiquimod than cryotherapy plus vehicle achieved complete clearance [121].
Another method is to combine topical modalities. For example, 0.5% 5-FU with 10% salicylic acid has been shown to provide more patients with complete clearance than diclofenac (55.4% vs. 32%, respectively) [122].
In a study conducted in Spain, PDT followed by three times weekly of imiquimod for 4 weeks (with a 1-month interval between the two treatments) was found to provide better clinical and histologic response than either treatment as monotherapy for facial/scalp AK. However, the difference in efficacy results between the PDT and imiquimod against imiquimod monotherapy arm are not significant, while those between PDT and imiquimod against PDT monotherapy were significant. Combination PDT and imiquimod was also found to elicit less intense local reactions and better tolerance and satisfaction than imiquimod monotherapy [117].
In a small pilot study conducted in the Netherlands, pre-treatment of AK on dorsum of hands with twice daily diclofenac 3% gel for 4 weeks followed by PDT 2 weeks later was compared to a placebo gel/PDT group. There were significant reductions in lesion numbers in both groups. Between both groups, a significant difference was only seen at 12 months after PDT, whereby the diclofenac/PDT group has a mean lesion score of 1.5 vs. 5.4 for the placebo/PDT group. Pain during PDT was greater in the diclofenac group. They concluded that both treatments were effective in treating AK, with the diclofenac group resulting in fewer AK at 12 months post-treatment [119].
Combining 70% glycolic acid with 5-FU has shown a 92% reduction in the number of AKs compared to a 20% reduction with chemical peel alone [123]. In a recent study, a combination of imiquimod and 5-FU once daily for up to 12 weeks led to the complete resolution of AK in ten out of ten patients [124], and a similar study found that 5-FU in the morning and imiquimod 5% at night for 1 week, repeated monthly for up to 3 months, gave >90% complete clearance [125]. It was noted that 17% of patients withdrew due to adverse side effects in this study. More combinations of topical therapy are under investigation but are beyond the scope of this discussion.
Oral retinoids are reserved for patients suffering with multiple AKs and where several SCCs are already developing, although evidence for this is scant and use as a monotherapy is most likely suboptimal [126]. It has been shown that retinoids can help decrease the risk of premalignant cells further developing into tumors [127]. The use of topical retinoids in the prevention of keratinocyte carcinoma has been investigated without success so far [128].
Lastly, other products currently under investigation as treatment for AKs include oral nicotinamide [129], betulinic acid [130] and piroxicam [131]. Further discussion is beyond the scope of this article but may yield interesting new adjuncts in the near future.
Conclusion
Actinic keratosis is common amongst fair-skinned patients exposed to significant amounts of UV irradiation. Although the chance of individual AK transforming to SCC is not high, they are useful markers for sun damage and skin cancer risk assessment. There is increasing interest in combining therapies for the treatment of AK, especially as the treatment options are ever increasing. However, the ultimate treatment choice will rest not only on efficacy, but also associated adverse reactions, cosmetic outcomes, accessibility, costs, compliance, and patient choice. The management of multiple AKs is a long-term prospect, with no clear cure. The best approach is the sequential treatment with a lesion-directed and a field-directed therapy. Combination therapies work well and should be adjusted according to patient requirements [132].
It is an area that clearly warrants ongoing research into methods of improving treatment and prevention, in a bid to reduce the burden of such a common disease on individuals and health care services.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
No funding or sponsorship was received for this study or publication of this article. All named authors meet the ICMJE criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Conflict of interest
A. Dodds, A. Chia and S. Shumack declare no conflicts of interest.
Compliance with ethics
This review article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
References
- 1.Samrao A, Cockerell J. Pharmacotherapeutic management of actinic keratosis: focus on newer topical agents. Am J Dermatol. 2013;14:273–277. doi: 10.1007/s40257-013-0023-y. [DOI] [PubMed] [Google Scholar]
- 2.Stockfleth E. The paradigm shift in treating actinic keratosis: a comprehensive strategy. J Drugs Dermatol. 2012;11(12):1462–1467. [PubMed] [Google Scholar]
- 3.Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1 Pt 2):4–7. doi: 10.1067/mjd.2000.103342. [DOI] [PubMed] [Google Scholar]
- 4.Franceschi S, Levi F, Randimbison L, La Vecchia C. Site distribution of different types of skin cancer: new aetiological clues. Int J Cancer. 1996;67(1):24–28. doi: 10.1002/(SICI)1097-0215(19960703)67:1<24::AID-IJC6>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 5.Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin and Chemoprevention Trial. Cancer. 2009;115(11):2523–2530. doi: 10.1002/cncr.24284. [DOI] [PubMed] [Google Scholar]
- 6.Berman B, Cockerell CJ. Pathobiology of actinic keratosis: ultraviolet-dependent keratinocyte proliferation. J Am Acad Dermatol. 2013;68(1 Suppl 1):S10–S19. doi: 10.1016/j.jaad.2012.09.053. [DOI] [PubMed] [Google Scholar]
- 7.Roewert-Huber J, Stockfleth E, Kerl H. Pathology and pathobiology of actinic (solar) keratosis—an update. Br J Dermatol. 2007;157(Suppl 2):18–20. doi: 10.1111/j.1365-2133.2007.08267.x. [DOI] [PubMed] [Google Scholar]
- 8.Marks R, et al. Spontaneous remission of solar keratoses: the case for conservative management. Br J Dermatol. 1986;115(6):649–655. doi: 10.1111/j.1365-2133.1986.tb06644.x. [DOI] [PubMed] [Google Scholar]
- 9.Brougham NDLS, Dennett ER, Cameron R, Tan ST. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol. 2012;106:811–815. doi: 10.1002/jso.23155. [DOI] [PubMed] [Google Scholar]
- 10.Brantsch KD, Meisner C, Schonfisch B, et al. Analysis of risk factors determining prognosis of cutaneous squamous-cell carcinoma: a prospective study. Lancet Oncol. 2008;9(8):713–720. doi: 10.1016/S1470-2045(08)70178-5. [DOI] [PubMed] [Google Scholar]
- 11.Mourouzis C, Boynton A, Grant J, et al. Cutaneous head and neck SCCs and risk of nodal metastasis: UK experience. J Craniomaxillofac Surg. 2009;37(8):443–447. doi: 10.1016/j.jcms.2009.07.007. [DOI] [PubMed] [Google Scholar]
- 12.Schmults CD, Karia PS, Carter JB, Han J, Qureshi AA. Factors predictive of recurrence and death from cutaneous squamous cell carcinoma: a 10-year, single institution cohort study. JAMA Dermatol. 2013;149(5):541–547. doi: 10.1001/jamadermatol.2013.2139. [DOI] [PubMed] [Google Scholar]
- 13.Marks R, Rennie G, Selwood TS. Malignant transformation of solar keratoses to squamous cell carcinoma. Lancet. 1988;1(8589):795–797. doi: 10.1016/s0140-6736(88)91658-3. [DOI] [PubMed] [Google Scholar]
- 14.Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol. 2000;42(1 Pt 2):23–24. doi: 10.1067/mjd.2000.103339. [DOI] [PubMed] [Google Scholar]
- 15.Ratushny V, Gober MD, Hick R, Ridky TW, Seykora JT. From keratinocyte to cancer: the pathogenesis and modelling of cutaneous squamous cell carcinoma. J Clin Invest. 2012;122(2):464–472. doi: 10.1172/JCI57415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Green A, Battistutta D. Incidence and determinants of skin cancer in a high-risk Australian population. Int J Cancer. 1990;46(3):356–361. doi: 10.1002/ijc.2910460303. [DOI] [PubMed] [Google Scholar]
- 17.Rigel DS, Gold LFS. The importance of early diagnosis and treatment of actinic keratosis. J Am Acad Dermatol. 2013;68(1 Suppl 1):S1. doi: 10.1016/j.jaad.2012.10.001. [DOI] [PubMed] [Google Scholar]
- 18.Simon JC, et al. Ultraviolet B radiation converts Langerhans cells from immunogenic to tolerogenic antigen-presenting cells. Induction of specific clonal anergy in CD4+ T helper 1 cells. J Immunol. 1991;146(2):485–491. [PubMed] [Google Scholar]
- 19.Yoshikawa T, et al. Susceptibility to effects of UVB radiation on induction of contact hypersensitivity as a risk factor for skin cancer in humans. J Invest Dermatol. 1990;95(5):530–536. doi: 10.1111/1523-1747.ep12504877. [DOI] [PubMed] [Google Scholar]
- 20.Mortier L, et al. Progression of actinic keratosis to squamous cell carcinoma of the skin correlates with deletion of the 9p21 region encoding the p16(INK4a) tumor suppressor. Cancer Lett. 2002;176(2):205–214. doi: 10.1016/s0304-3835(01)00757-1. [DOI] [PubMed] [Google Scholar]
- 21.Kanellou P, Zaravinos A, Zioga M, Stratigos A, Baritaki S, Soufla G, Zoras O, Spandidos DA. Genomic instability, mutations and expression analysis of the tumour suppressor genes p14(ARF), p15(INK4b), p16(INK4a) and p53 in actinic keratosis. Cancer Lett. 2008;264(1):145–161. doi: 10.1016/j.canlet.2008.01.042. [DOI] [PubMed] [Google Scholar]
- 22.Timares L, Katiyar SK, Elmets CA. DNA damage, apoptosis and Langerhans cells—activators of UV-induced immune tolerance. Photochem Photobiol. 2008;84:422–436. doi: 10.1111/j.1751-1097.2007.00284.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Berman B, Cockerell CJ. Pathobiology of actinic keratosis: ultraviolet-dependent keratinocyte proliferation. J Am Acad Dermatol. 2010 Jan;68(Iss 1, Supp 1):PA1–A4, S10–S19. [DOI] [PubMed]
- 24.Brash DE, Ziegler A, Jonason AS, Simon JA, Kunala S, Leffell DJ. Sunlight and sunburn in human skin cancer: p53, apoptosis, and tumour promotion. J Investig Dermatol Symp Proc. 1996;1:136–142. [PubMed] [Google Scholar]
- 25.Jiang W, Ananthaswamy HN, Muller HK, Kripke ML. p53 protects against skin cancer induction by UV-B radiation. Oncogene. 1999;18(29):4247–4253. doi: 10.1038/sj.onc.1202789. [DOI] [PubMed] [Google Scholar]
- 26.Wang L, Eng W, Cockerell CJ. Effects of ultraviolet irradiation on inflammation in the skin. Adv Dermatol. 2002;18:247–286. [PubMed] [Google Scholar]
- 27.Hruza LL, Pentland AP. Mechanisms of UV-induced inflammation. J Invest Dermatol. 1993;100:35S–41S. doi: 10.1111/1523-1747.ep12355240. [DOI] [PubMed] [Google Scholar]
- 28.Lober BA, Lober CW, Accola J. Actinic keratosis is squamous cell carcinoma. J Am Acad Dermatol. 2000;43(5 Pt 1):881–882. doi: 10.1067/mjd.2000.108373. [DOI] [PubMed] [Google Scholar]
- 29.Heaphy MR, Jr, Ackerman AB. The nature of solar keratosis: a critical review in historical perspective. J Am Acad Dermatol. 2000;43(1 Pt 1):138–150. doi: 10.1067/mjd.2000.107497. [DOI] [PubMed] [Google Scholar]
- 30.Lebwohl MG, Rosen T, Stockfleth E. The role of human papillomavirus in common skin conditions: current viewpoints and therapeutic options. Cutis. 2010;86(5):Suppl 1–11 (quiz suppl 12). [PubMed]
- 31.Jackson S, Harwood C, Thomas M, et al. Role of Bak in UV-induced apoptosis in skin cancer and abrogation by HPV E6 proteins. Genes Dev. 2000;14:3065–3073. doi: 10.1101/gad.182100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cockerell CJ. Histopathology of incipient intraepidermal squamous cell carcinoma (“actinic keratosis”) J Am Acad Dermatol. 2000;42(1 pt 2):11–17. doi: 10.1067/mjd.2000.103344. [DOI] [PubMed] [Google Scholar]
- 33.Padilla RS, Sebastian S, Jiang Z, Nindl I, Larson R. Gene expression patterns of normal human skin, actinic keratosis, and squamous cell carcinoma: a spectrum of disease progression. Arch Dermatol. 2010;146(3):288–293. doi: 10.1001/archdermatol.2009.378. [DOI] [PubMed] [Google Scholar]
- 34.Neto PD, Alchorne M, Michalany N, Abreu M, Borra R. Reduced P53 staining in actinic keratosis is associated with squamous cell carcinoma: a preliminary study. Indian J Dermatol. 2013;58(4):325. doi: 10.4103/0019-5154.113935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Marks R. Epidemiology of non-melanoma skin cancer and solar keratoses in Australia: a tale of self-immolation in Elysian fields. Australas J Dermatol. 1997;38(Suppl 1):S26–S29. doi: 10.1111/j.1440-0960.1997.tb01004.x. [DOI] [PubMed] [Google Scholar]
- 36.Frost CA, Green AC. Epidemiology of solar keratoses. Br J Dermatol. 1994;131(4):455–464. doi: 10.1111/j.1365-2133.1994.tb08544.x. [DOI] [PubMed] [Google Scholar]
- 37.Stockfleth E, Terhorst D, Braathen L, et al., Guideline Subcommittee of the European Dermatology Forum. Guidelines for the management of actinic keratosis—update 2011. Available from: http://bestpractice.bmj.com/best-practice/monograph/616/diagnosis/guidelines.html. Accessed August 25 2013.
- 38.Harvey I, Frankel S, Marks R, Shalom D, Nolan-Farrell M. Non-melanoma skin cancer and solar keratoses, II: analytical results of the South Wales skin cancer study. Br J Cancer. 1996;74:1308–1312. doi: 10.1038/bjc.1996.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holmes C, Foley P, Freeman M, Chong AH. Solar keratosis: epidemiology, pathogenesis, presentation and treatment. Australas J Dermatol. 2007;48(2):67–74. doi: 10.1111/j.1440-0960.2007.00339.x. [DOI] [PubMed] [Google Scholar]
- 40.Frost C, Williams G, Green A. High incidence and regression rates of solar keratoses in a Queensland community. J Invest Dermatol. 2000;115(2):273–277. doi: 10.1046/j.1523-1747.2000.00048.x. [DOI] [PubMed] [Google Scholar]
- 41.Marks R, Ponsford MW, Selwood TS, Goodman G, Mason G. Non-melanotic skin cancer and solar keratoses in Victoria. Med J Aust. 1983;2:619–622. doi: 10.5694/j.1326-5377.1983.tb122724.x. [DOI] [PubMed] [Google Scholar]
- 42.Harvey I, Frankel S, Marks R, Shalom D, Nolan-Farrell M. Non-melanoma skin cancer and solar keratoses, I: methods and descriptive results of the South Wales skin cancer study. Br J Cancer. 1996;74:1302–1307. doi: 10.1038/bjc.1996.534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Euvrard S, et al. Comparative epidemiologic study of premalignant and malignant epithelial cutaneous lesions developing after kidney and heart transplantation. J Am Acad Dermatol. 1995;33(2 Pt 1):222–229. doi: 10.1016/0190-9622(95)90239-2. [DOI] [PubMed] [Google Scholar]
- 44.Kaufman R. The concept of field cancerization. Melanoma Res. 2010;20:e13–e14. [Google Scholar]
- 45.Slaughter DP, Southwick HW, Smejkal W. Field cancerization in oral stratified squamous epithelium: clinical manifestations of multicentric origin. Cancer. 1953;6(5):963–968. doi: 10.1002/1097-0142(195309)6:5<963::aid-cncr2820060515>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- 46.Basal cell carcinoma, squamous cell carcinoma (and related lesions)—a guide to clinical management in Australia. Cancer Council Australia and Australian Cancer Network, Sydney; 2008.
- 47.Stockfleth E, Karl H, Guideline Subcommittee of the European Dermatology Forum Guidelines for the management of actinic keratosis. Eur J Dermatol. 2006;16(6):599–606. [PubMed] [Google Scholar]
- 48.De Berker D, McGregor JM, Hughes BR, British Association of Dermatologists Therapy Guidelines and Audit Subcommittee Guidelines for the management of actinic keratosis. Br J Dermatol. 2007;156(2):222–230. doi: 10.1111/j.1365-2133.2006.07692.x. [DOI] [PubMed] [Google Scholar]
- 49.Berman B, Bienstock L, Kuritzky L, Mayeaux EJ, Tyring SK, Primary Care Education Consortium. Texas Academy of Family Physicians Actinic keratoses: sequelae and treatments. Recommendations from a consensus panel. J Fam Pract. 2006;55(5):Suppl 1–8. [PubMed] [Google Scholar]
- 50.Gupta AK, Paquet M. Network meta-analysis of the outcome ‘participant complete clearance’ in nonimmunosuppressed participants of eight interventions for actinic keratosis: a follow-up on a Cochrane review. Br J Dermatol. 2013;169(2):250–259. doi: 10.1111/bjd.12343. [DOI] [PubMed] [Google Scholar]
- 51.Balkrishnan R, et al. Predictors of treatment choices and associated outcomes in actinic keratoses: results from a national physician survey study. J Dermatol Treat. 2006;17(3):162–166. doi: 10.1080/09546630600765081. [DOI] [PubMed] [Google Scholar]
- 52.Thai KE, Fergin P, Freeman M, et al. A prospective study of the use of cryosurgery for the treatment of actinic keratoses. Int J Dermatol. 2004;43(9):687–692. doi: 10.1111/j.1365-4632.2004.02056.x. [DOI] [PubMed] [Google Scholar]
- 53.Kafmann R, Spelman L, Weightman W, Reifenberger J, Sziemies RM, Verhaeghe E, et al. Multicenter intraindividual randomized trial of topical methyl aminolevulinate-photodynamic therapy vs cryotherapy for multiple actinic keratoses on the extremities. Br J Dermatol. 2008;158:994–999. doi: 10.1111/j.1365-2133.2008.08488.x. [DOI] [PubMed] [Google Scholar]
- 54.McIntyre WJ, Downs MR, Bedwell SA. Treatment options for actinic keratoses. Am Fam Physician. 2007;76:667–671. [PubMed] [Google Scholar]
- 55.Berman B, Bienstock L, Kuritzky L, Mayeaux EJ, Jr, Tyring SK. Actinic keratoses: sequelae and treatments. Recommendations from a consensus panel. J Fam Pract. 2006;55:1–8. [PubMed] [Google Scholar]
- 56.Hantash BM, Stewart DB, Cooper ZA, Rehmus WE, Koch RJ, Swetter SM. Facial resurfacing for nonmelanoma skin cancer prophylaxis. Arch Dermatol. 2006;142(8):976–982. doi: 10.1001/archderm.142.8.976. [DOI] [PubMed] [Google Scholar]
- 57.Winton GB, Salache SJ. Dermabrasion of the scalp as a treatment for actinic damage. J Am Acad Dermatol. 1986;14(4):661–668. doi: 10.1016/s0190-9622(86)70083-2. [DOI] [PubMed] [Google Scholar]
- 58.Dillaha CJ, et al. Further studies with topical 5-fluorouracil. Arch Dermatol. 1965;92(4):410–417. [PubMed] [Google Scholar]
- 59.Eaglstein WH, Weinstein GD, Frost P. Fluorouracil: mechanism of action in human skin and actinic keratoses. I. Effect on DNA synthesis in vivo. Arch Dermatol. 1970;101(2):132–139. [PubMed] [Google Scholar]
- 60.Kaur RR, Alikhan A, Maibach HI. Comparison of topical 5-fluorouracil formulations in actinic keratosis treatment. J Dermatol Treat. 2010;21(5):267–271. doi: 10.3109/09546630903341937. [DOI] [PubMed] [Google Scholar]
- 61.Rhavar M, Lamel SA, Maibach HI. Randomized, vehicle-controlled trials of topical 5-fluorouracil therapy for actinic keratosis treatment: an overview. Immunotherapy. 2012;4(9):939–945. doi: 10.2217/imt.12.93. [DOI] [PubMed] [Google Scholar]
- 62.Yentzer B, Hick J, Williams L, Inabinet R, Wilson R, Camacho FT, Russell GB, Feldman SR. Adherence to a topical regimen of 5-fluorouracil, 0.5%, cream for the treatment of actinic keratoses. JAMA Dermatol. 2009;145(2):203–205. doi: 10.1001/archdermatol.2008.562. [DOI] [PubMed] [Google Scholar]
- 63.Krawtchenko N, Roewert-Huber J, Ulrich M, Mann I, Sterry W, Stockfleth E. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol. 2007;157(Suppl 2):34–40. doi: 10.1111/j.1365-2133.2007.08271.x. [DOI] [PubMed] [Google Scholar]
- 64.Labandeira J, et al. Intermittent topical 5-fluorouracil is effective without significant irritation in the treatment of actinic keratoses but prolongs treatment duration. Dermatol Surg. 2004;30(4 Pt 1):517–520. doi: 10.1111/j.1524-4725.2004.30167.x. [DOI] [PubMed] [Google Scholar]
- 65.Robins P. Pulse therapy with 5-FU in eradicating actinic keratoses with less than recommended dosage. J Drugs Dermatol. 2002;1(1):25–30. [PubMed] [Google Scholar]
- 66.Berman B, Cohen DE, Amini S. What is the role of field directed therapy in the treatment of actinic keratosis? Part 2: commonly used field-directed and lesion-directed therapies. Cutis. 2012;89(6):294–301. [PubMed] [Google Scholar]
- 67.Clark RA, Huang SJ, Murphy GF, et al. Human squamous cell carcinomas evade the immune response by down-regulation of vascular E-selectin and recruitment of regulatory T cells. J Exp Med. 2008;205(10):2221–2234. doi: 10.1084/jem.20071190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Meyer T, et al. Induction of apoptosis by Toll-like receptor-7 agonist in tissue cultures. Br J Dermatol. 2003;149(Suppl 66):9–14. doi: 10.1046/j.0366-077x.2003.05632.x. [DOI] [PubMed] [Google Scholar]
- 69.Schon MP, Schon M. Imiquimod: mode of action. Br J Dermatol. 2007;157(Suppl 2):8–13. doi: 10.1111/j.1365-2133.2007.08265.x. [DOI] [PubMed] [Google Scholar]
- 70.Uhlenhake EE. Optimal treatment of actinic keratoses. Clin Interv Ageing. 2013;8:29–35. doi: 10.2147/CIA.S31930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gupta AK, Cooper EA, Abramovits W. Zyclara (imiquimod) cream, 3.75% Skinmed. 2010;8:227–229. [PubMed] [Google Scholar]
- 72.Lebwohl M, et al. Imiquimod 5% cream for the treatment of actinic keratosis: results from two phase III, randomized, double-blind, parallel group, vehicle-controlled trials. J Am Acad Dermatol. 2004;50(5):714–721. doi: 10.1016/j.jaad.2003.12.010. [DOI] [PubMed] [Google Scholar]
- 73.Lee PK, Harwell WB, Loven KH, et al. Long term clinical outcomes following treatment of actinic keratosis with imiquimod 5% cream. Dermatol Surg. 2005;31(6):659–664. doi: 10.1111/j.1524-4725.2005.31608. [DOI] [PubMed] [Google Scholar]
- 74.Korman N, et al. Dosing with 5% imiquimod cream 3 times per week for the treatment of actinic keratosis: results of two phase 3, randomized, double-blind, parallel-group, vehicle-controlled trials. Arch Dermatol. 2005;141(4):467–473. doi: 10.1001/archderm.141.4.467. [DOI] [PubMed] [Google Scholar]
- 75.Szeimies RM, et al. Imiquimod 5% cream for the treatment of actinic keratosis: results from a phase III, randomized, double-blind, vehicle-controlled, clinical trial with histology. J Am Acad Dermatol. 2004;51(4):547–555. doi: 10.1016/j.jaad.2004.02.022. [DOI] [PubMed] [Google Scholar]
- 76.Salasche SJ, Levine N, Morrison L. Cycle therapy of actinic keratoses of the face and scalp with 5% topical imiquimod cream: an open-label trial. J Am Acad Dermatol. 2002;47(4):571–577. doi: 10.1067/mjd.2002.126257. [DOI] [PubMed] [Google Scholar]
- 77.Chen K, et al. Short-course therapy with imiquimod 5% cream for solar keratoses: a randomized controlled trial. Australas J Dermatol. 2003;44(4):250–255. doi: 10.1046/j.1440-0960.2003.00003.x. [DOI] [PubMed] [Google Scholar]
- 78.Ulrich C, Busch JO, Meyer T, et al. Successful treatment of multiple actinic keratoses in organ transplant patients with topical 5% imiquimod: a report of six cases. Br J Dermatol. 2006;155(2):451–454. doi: 10.1111/j.1365-2133.2006.07233.x. [DOI] [PubMed] [Google Scholar]
- 79.Hanke CW, Beer KR, Stockfleth E, Wu J, Rosen T, Levy S. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: results of two placebo-controlled studies of daily application to the face and balding scalp for two 3-week cycles. J Am Acad Dermatol. 2010;62(4):573–581. doi: 10.1016/j.jaad.2009.06.020. [DOI] [PubMed] [Google Scholar]
- 80.Swanson N, Abramovits W, Berman B, Kulp J, Rigel DS, Levy S. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: results of two placebo controlled studies of daily application to the face and balding scalp for two 2-week cycles. J Am Acad Dermatol. 2010;62(4):582–590. doi: 10.1016/j.jaad.2009.07.004. [DOI] [PubMed] [Google Scholar]
- 81.Haddican M, Goldenberg G. Update on the treatment of actinic keratoses. Wayne PA: Bryn Mawr Communications III LLC; 2012. Available from: http://bmctoday.net/practicaldermatology/pdfs/PD0612_SF_AKs.pdf. Accessed August 30, 2013.
- 82.Foley P, et al. A comparison of cryotherapy and imiquimod for treatment of actinic keratoses: lesion clearance, safety, and skin quality outcomes. J Drugs Dermatol. 2011;10(12):1432–1438. [PubMed] [Google Scholar]
- 83.Hadley J, et al. Results of an investigator-initiated single-blind split-face comparison of photodynamic therapy and 5% imiquimod cream for the treatment of actinic keratoses. Dermatol Surg. 2012;38(5):722–727. doi: 10.1111/j.1524-4725.2012.02340.x. [DOI] [PubMed] [Google Scholar]
- 84.Serra-Guillen C, Nagore E, Hueso L, Llombart B, Requena C, Sanmartin O, Botella-Estrada R, Guillen C. A randomized comparative study of tolerance and satisfaction in the treatment of actinic keratosis of the face and scalp between 5% imiquimod cream and photodynamic therapy with methyl aminolaevulinate. Br J Dermatol. 2011;164(2):429–433. doi: 10.1111/j.1365-2133.2010.10098.x. [DOI] [PubMed] [Google Scholar]
- 85.Stockfleth E, Sterry W, Carey-Yard M, Bichel J. Multicentre, open-label study using imiquimod 5% cream in one or two 4-week courses of treatment for multiple actinic keratoses on the head. Br J Dermatol. 2007;157(Suppl 2):41–46. doi: 10.1111/j.1365-2133.2007.08272.x. [DOI] [PubMed] [Google Scholar]
- 86.Hadley G, Derry S, Moore RA. Imiquimod for actinic keratosis: systematic review and meta-analysis. J Invest Dermatol. 2006;126(6):1251. doi: 10.1038/sj.jid.5700264. [DOI] [PubMed] [Google Scholar]
- 87.Ulrich C, Bichel J, Euvrard S, et al. Topical immunomodulation under systemic immunosuppression: results of a multicentre, randomized, placebo-controlled safety and efficacy study of imiquimod 5% cream for the treatment of actinic keratoses in kidney, heart, and liver transplant patients. Br J Dermatol. 2007;157(Suppl 2):25–31. doi: 10.1111/j.1365-2133.2007.08269.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Szeimies RM, Bichel J, Ortonne JP, et al. A phase II dose-ranging study of topical resiquimod to treat actinic keratosis. Br J Dermatol. 2008;159(1):205–210. doi: 10.1111/j.1365-2133.2008.08615.x. [DOI] [PubMed] [Google Scholar]
- 89.Higashi Y, Kanekura T, Kanzaki T. Enhanced expression of cyclooxygenase (COX)-2 in human skin epidermal cancer cells: evidence for growth suppression by inhibiting COX-2 expression. Int J Cancer. 2000;86(5):667–671. doi: 10.1002/(sici)1097-0215(20000601)86:5<667::aid-ijc10>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 90.Maltusch A, Rowert-Huber J, Matthies C, Lange-Asschenfeldt S, Stockfleth E. Modes of action of diclofenac 3%/hyaluronic acid 2.5% in the treatment of actinic keratosis. J Dtsch Dermatol Ges. 2011;9(12):1011–1017. doi: 10.1111/j.1610-0387.2011.07700.x. [DOI] [PubMed] [Google Scholar]
- 91.Martin GM, Stockfleth E. Diclofenac sodium 3% gel for the management of actinic keratosis: 10+ years of cumulative evidence of efficacy and safety. J Drugs Dermatol. 2012;11(5):600–608. [PubMed] [Google Scholar]
- 92.Pirard D, Vereecken P, Melot C, Heenan M. Three percent diclofenac in 2.5% hyaluronan gel in the treatment of actinic keratoses: a meta-analysis of the recent studies. Arch Dermatol Res. 2005;297(5):185. doi: 10.1007/s00403-005-0601-9. [DOI] [PubMed] [Google Scholar]
- 93.Nelson C, et al. Phase IV, open-label assessment of the treatment of actinic keratosis with 3.0% diclofenac sodium topical gel (Solaraze) J Drugs Dermatol. 2004;3(4):401–407. [PubMed] [Google Scholar]
- 94.Gebauer K, Brown P, Varigos G. Topical diclofenac in hyaluronan gel for the treatment of solar keratoses. Australas J Dermatol. 2003;44(1):40–43. doi: 10.1046/j.1440-0960.2002.00635.x. [DOI] [PubMed] [Google Scholar]
- 95.Berlin JM, Rigel DS. Diclofenac sodium 3% gel in the treatment of actinic keratoses post cryosurgery. J Drugs Dermatol. 2008;7(7):669–673. [PubMed] [Google Scholar]
- 96.Nelson C, Rigel D. Long-term follow-up of diclofenac sodium 3% in 2.5% hyaluronic acid gel for actinic keratosis: one year evaluation. J Clin Aesthet Dermatol. 2009;2(7):20–25. [PMC free article] [PubMed] [Google Scholar]
- 97.Kennedy JC, Pottier RH, Pross DC. Photodynamic therapy with endogenous protoporphyrin IX: basic principles and present clinical experience. J Photochem Photobiol B. 1990;6:143–148. doi: 10.1016/1011-1344(90)85083-9. [DOI] [PubMed] [Google Scholar]
- 98.Stritt A, Merk HF, Braathen LR, von Felbert V. Photodynamic therapy in the treatment of actinic keratosis. Photochem Photobiol. 2008;84(2):388–398. doi: 10.1111/j.1751-1097.2007.00276.x. [DOI] [PubMed] [Google Scholar]
- 99.Braathen LR, et al. Guidelines on the use of photodynamic therapy for nonmelanoma skin cancer: an international consensus. J Am Acad Dermatol. 2007;56:125–143. doi: 10.1016/j.jaad.2006.06.006. [DOI] [PubMed] [Google Scholar]
- 100.Christensen E, et al. Guidelines for practical use of MAL-PDT in non-melanoma skin cancer. J Eur Acad Dermatol Venerol. 2010;24:505–512. doi: 10.1111/j.1468-3083.2009.03430.x. [DOI] [PubMed] [Google Scholar]
- 101.De Berker D, McGregor JM, Hughes BR, British Association of Dermatologists Therapy Guidelines and Audit Subcommittee Guidelines for the management of actinic keratoses (published correction in Br J Dermatol. 2008;158:873) Br J Dermatol. 2007;156:222–230. doi: 10.1111/j.1365-2133.2006.07692.x. [DOI] [PubMed] [Google Scholar]
- 102.Fritsch C, et al. Preferential relative porphyrin enrichment in solar keratoses upon topical application of delta-aminolevulinic acid methylester. Photochem Photobiol. 1998;68(2):218–221. [PubMed] [Google Scholar]
- 103.Pariser DM, et al. Photodynamic therapy with topical methyl aminolevulinate for actinic keratosis: results of a prospective randomized multicenter trial. J Am Acad Dermatol. 2003;48(2):227–232. doi: 10.1067/mjd.2003.49. [DOI] [PubMed] [Google Scholar]
- 104.Togsverd-Bo K, et al. Intensified photodynamic therapy of actinic keratoses with fractional CO2 laser: a randomized clinical trial. Br J Dermatol. 2012;166(6):1262–1269. doi: 10.1111/j.1365-2133.2012.10893.x. [DOI] [PubMed] [Google Scholar]
- 105.Wiegell SR, Haedersdal M, Philipsen PA, Enk CD, Wulf HC. Continuous activation of PpIX by daylight is as effective as and less painful than conventional photodynamic therapy for actinic keratoses; a randomised, controlled, single-blinded study. Br J Dermatol. 2008;158(4):740–746. doi: 10.1111/j.1365-2133.2008.08450.x. [DOI] [PubMed] [Google Scholar]
- 106.Wiegell SR, Haedersdal M, Eriksen P, Wulf HC. Photodynamic therapy of actinic keratoses with 8% and 16% methyl aminolaevulinate and home-based daylight exposure: a double-blinded randomised clinical trial. Br J Dermatol. 2009;160(6):1308–1314. doi: 10.1111/j.1365-2133.2009.09119.x. [DOI] [PubMed] [Google Scholar]
- 107.Dirschka T, Radny P, Dominicus R, et al. Photodynamic therapy with BF-200 ALA for the treatment of actinic keratosis: results of a multicentre, randomized, observer-blind phase III study in comparison with a registered methyl-5-aminolaevulinate cream and placebo. Br J Dermatol. 2012;166(1):137–146. doi: 10.1111/j.1365-2133.2011.10613.x. [DOI] [PubMed] [Google Scholar]
- 108.Hauschild A, Stockfleth E, Popp G, et al. Optimisation of photodynamic therapy with a novel self-adhesive 5-aminolaevulinic acid patch: results of two randomized controlled phase III studies. Br J Dermatol. 2009;160:1066–1074. doi: 10.1111/j.1365-2133.2009.09040.x. [DOI] [PubMed] [Google Scholar]
- 109.Serra-Guillen C, Hueso L, Nagore E, Vila M, Llombart B, Requena Caballero C, Botell-Estrada R, Sanmartin O, Alfaro-Rubio A, Guillen C. Comparative study between cold air analgesia and supraorbital and supratrochlear nerve block for the management of pain during photodynamic therapy for actinic keratoses of the frontotemporal zone. Br J Dermatol. 2009;161(2):353–356. doi: 10.1111/j.1365-2133.2009.09184.x. [DOI] [PubMed] [Google Scholar]
- 110.Halldin CB, Paoli J, Sandberg C, Gonzalez H, Wennberg AM. Nerve blocks enable adequate pain relief during topical photodynamic therapy of field cancerization on the forehead and scalp. Br J Dermatol. 2009;160(4):795–800. doi: 10.1111/j.1365-2133.2008.09024.x. [DOI] [PubMed] [Google Scholar]
- 111.Rosen RH, Gupta AK, Tyring S. Dual mechanism of action of ingenol mebutate gel for topical treatment of actinic keratoses: rapid lesion necrosis followed by lesion-specific immune response. J Am Acad Dermatol. 2012;66(3):486–493. doi: 10.1016/j.jaad.2010.12.038. [DOI] [PubMed] [Google Scholar]
- 112.Ogbourne SM, Suhrbier A, Jones B, Cozzi SJ, Boyle GM, Morris M, et al. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res. 2004;64:2833–2839. doi: 10.1158/0008-5472.can-03-2837. [DOI] [PubMed] [Google Scholar]
- 113.Siller G, et al. PEP005 (ingenol mebutate) gel, a novel agent for the treatment of actinic keratosis: results of a randomized, double-blind, vehicle-controlled, multicentre, phase IIa study. Australas J Dermatol. 2009;50(1):16–22. doi: 10.1111/j.1440-0960.2008.00497.x. [DOI] [PubMed] [Google Scholar]
- 114.Anderson L, et al. Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol. 2009;60(6):934–943. doi: 10.1016/j.jaad.2009.01.008. [DOI] [PubMed] [Google Scholar]
- 115.Lebwohl M, et al. Ingenol mebutate gel for actinic keratosis. N Engl J Med. 2012;366(11):1010–1019. doi: 10.1056/NEJMoa1111170. [DOI] [PubMed] [Google Scholar]
- 116.Lebwohl M, Shumack S, Stein Gold L, Melgaard A, Larsson T, Tyring SK. Long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses. JAMA Dermatol. 2013;149(6):666–670. doi: 10.1001/jamadermatol.2013.2766. [DOI] [PubMed] [Google Scholar]
- 117.Serra-Guillen C, et al. A randomized pilot comparative study of topical methyl aminolevulinate photodynamic therapy versus imiquimod 5% versus sequential application of both therapies in immunocompetent patients with actinic keratosis: clinical and histologic outcomes. J Am Acad Dermatol. 2012;66(4):e131–e137. doi: 10.1016/j.jaad.2011.11.933. [DOI] [PubMed] [Google Scholar]
- 118.Jorizzo JL, et al. A randomized, double-blinded, placebo-controlled, multicenter, efficacy and safety study of 3.75% imiquimod cream following cryosurgery for the treatment of actinic keratoses. J Drugs Dermatol. 2010;9(9):1101–1108. [PubMed] [Google Scholar]
- 119.Van der Geer S, Krekels GA. Treatment of actinic keratoses on the dorsum of the hands: ALA-PDT versus diclofenac 3% gel followed by ALA-PDT. A placebo-controlled, double-blind, pilot study. J Dermatol Treat. 2009;20(5):259–265. doi: 10.1080/09546630902882048. [DOI] [PubMed] [Google Scholar]
- 120.Jorizzo J, Weiss J, Vamvakias G. One-week treatment with 0.5% fluorouracil cream prior to cryosurgery in patients with actinic keratoses: a double blind, vehicle controlled, long-term study. J Drugs Dermatol. 2006;5(2):133–139. [PubMed] [Google Scholar]
- 121.Gupta AK et al. Interventions for actinic keratoses. Cochrane Database Syst Rev 2012;Issue 12. Art. No:CD004415. doi:10.1002/14651858.CD004415.pub2. [DOI] [PMC free article] [PubMed]
- 122.Stockfleth E, Kerl H, Zwingers T, Willers C. Low-dose 5-fluorouracil in combination with salicylic acid as a new lesion-directed option to treat topically actinic keratoses: histological and clinical study results. Br J Dermatol. 2011;165(5):1101–1108. doi: 10.1111/j.1365-2133.2011.10387.x. [DOI] [PubMed] [Google Scholar]
- 123.Marrero GM, Katz BE. The new fluor-hydroxy pulse peel. A combination of 5-fluorouracil and glycolic acid. Dermatol Surg. 1998;24(9):973–978. [PubMed] [Google Scholar]
- 124.Ondo AL, Padilla S, Miedler JD, et al. Treatment-refractory actinic keratosis successfully treated using simultaneous combination topical 5-fluorouracil cream and imiquimod cream: case control study. Dermatol Surg. 2012;38(9):1469–1476. doi: 10.1111/j.1524-4725.2012.02480.x. [DOI] [PubMed] [Google Scholar]
- 125.Price NM. The treatment of actinic keratoses with a combination of 5-fluorouracil and imiquimod creams. J Drugs Dermatol. 2007;6(8):778–781. [PubMed] [Google Scholar]
- 126.Jeffes EW, 3rd, Tang EH. Actinic keratosis. Current treatment options. Am J Clin Dermatol. 2000;1(3):167–179. doi: 10.2165/00128071-200001030-00004. [DOI] [PubMed] [Google Scholar]
- 127.Amini S, Viera MH, Valins W, Berman B. Nonsurgical innovations in the treatment of nonmelanoma skin cancer. J Clin Aesthet Dermatol. 2010;3(6):20–34. [PMC free article] [PubMed] [Google Scholar]
- 128.Weinstock MA, Bingham SF, Digiovanna JJ, Rizzo AE, Marcolivio K, Hall R, Eilers D, Naylor M, Kirsner R, Kalivas J, Cole G, Vertrees JE, Veterans Affairs Topical Tretinoin Chemoprevention Trial Group Tretinoin and the prevention of keratinocyte carcinoma (basal and squamous cell carcinoma of the skin): a veterans affairs randomized chemoprevention trial. J Invest Dermatol. 2012;132(6):1583–1590. doi: 10.1038/jid.2011.483. [DOI] [PubMed] [Google Scholar]
- 129.Surjana D, Halliday GM, Martin AJ, Moloney FJ, Damian DL. Oral nicotinamide reduces actinic keratoses in phase II double-blinded randomized controlled trials. J Invest Dermatol. 2012;132(5):1497–1500. doi: 10.1038/jid.2011.459. [DOI] [PubMed] [Google Scholar]
- 130.Huyke C, Laszczyk M, Scheffler A, Ernst R, Schempp CM. Treatment of actinic keratoses with birch bark extract: a pilot study. JDDG J Dtsch Dermatologischen Ges. 2006;4:132–136. doi: 10.1111/j.1610-0387.2006.05906.x. [DOI] [PubMed] [Google Scholar]
- 131.Campione E, Diluvio L, Paterno EJ, Chimenti S. Topical treatment of actinic keratoses with piroxicam 1% gel: a preliminary open-label study utilizing a new clinical score. Am J Clin Dermatol. 2010;11(1):45–50. doi: 10.2165/11311170-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 132.Berman B, Amini S. Pharmacotherapy of actinic keratosis: an update. Expert Opin Pharmacother. 2013;13(13):1847–1871. doi: 10.1517/14656566.2012.716039. [DOI] [PubMed] [Google Scholar]
- 133.Stockfleth E, Ferrandiz C, Grob JJ, et al. Development of a treatment algorithm for actinic keratoses: a European Consensus. Eur J Dermatol. 2008;18(6):651–659. doi: 10.1684/ejd.2008.0514. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.