Abstract
First branchial cleft anomaly is a rare disease of the head and neck. Because of its rarity, first branchial cleft anomaly is often misdiagnosed and results in inappropriate management. In this article, we present a case of type II first branchial cleft anomaly. A middle-aged woman who had suffered from swelling on lower jaw visited our department with the chief complaint of a swelling. She underwent complete excision of the lesion with preservation of the facial nerve. The patient recovered well and had no recurrence at 1-year of follow up.
Keywords: Branchial cleft cyst, cervical lymphoepithelial cyst, first branchial anomaly
INTRODUCTION
First branchial cleft anomalies are thought to originate from the branchial apparatus that did not completely obliterate during head and neck embryogenesis. The incidence is estimated to be about one per million population/year.[1,2] The anomalies account for <8% of all branchial anomalies and the patients are diagnosed at the average age of 19 years old. The delay between initial presentation and adequate treatment is 3.5 years. There is no obvious familial tendency. Female patients are twice as frequently affected than male with a tendency to occur on the left side.[3] The anomalies are often distributed in the lateral neck below the external ear canals, above the hyoid bones, anterior to the sternocleidomastoid muscle and posterior to the submandibular angles. The spectrum of developmental abnormalities includes cysts, sinuses, fistulas and various combinations of these entities.[4] The principle of management includes early diagnosis, controlling the infection status and complete excision without facial nerve injury. Prognosis for the patients is generally good.
CASE REPORT
A Fifty year old female patient reported to the hospital with complaint of swelling in the lower jaw since 4-5 months. On clinical examination, a swelling was seen below the left side of the angle of the mandible which was measuring 5 × 3cm and extending 4cm below the base of the mandible and obliquely 5cm from the angle of the mandible. The mass was just anterior and deep to the sternocleidomastoid muscle [Figure 1]. On palpation it was soft in consistency, fluctuant and painful. An The FNAC was performed in a municipal hospital which was diagnosed as mucoepidermoid tumor of parotid and was negativity for Koch bacilli. The FNAC report did not mention the detailed findings. Based on these findings the patient was operated on and the excised tissue was grossed. The gross finding showed that the tissue was measuring 5 × 3 cm and cut section was partially brownish in colour. The specimen showed mucoid material within the cystic cavity, the cyst wall was very thick with papillary projections [Figure 2]. The tissue was formalin fixed, processed, sectioned and stained with haematoxylin and eosin.
Figure 1.

Clinical image showing swelling in the neck
Figure 2.

Gross specimen of the excised lesion
Histopathological findings showed cystic cavity lined by epithelium and the underlying connective tissue showed germinal centres. The cystic epithelium was thrown into folds at various places. The epithelium was stratified squamous epithelium type and was 6-8 cells layered thick [Figure 3]. The rete ridges were flat. At some places the epithelium was separated from the underling connective tissue. The cystic lumen showed some squamous epithelial strands which were desquamated from the cystic epithelium. The underlying connective tissue showed abundant lymphoid tissue with many germinal centres. Some fibrous connective tissue elements with fibroblasts and blood vessels lined by endothelial cells were evident. [Figures 4–6]. To rule out tuberculosis Ziehl–Neelsen stain was previously done and there was no evidence of Koch bacilli. Based on all these findings a final diagnosis of infected branchial cyst was given. She underwent complete excision of the lesion with preservation of the facial nerve. The patient recovered well and had no recurrence at 1-year of follow up.
Figure 3.

Photomicrograph showing cystic lesion lined by stratified squamous epithelium. (H&E stain, ×40)
Figure 4.
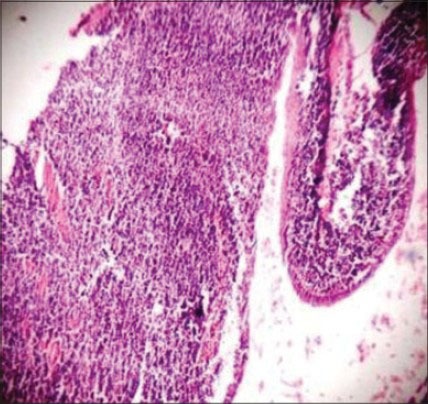
Photomicrograph showing cystic cavity lined by thin stratified squamous epithelium with the underlying connective tissue showing abundant lymphoid tissue aggregate (H&E stain, ×40)
Figure 6.

Photomicrograph showing cystic lumen lined by thin stratified squamous epithelium with flat rete ridges and underlying connective tissue showing abundant lymphoid tissue aggregate (H&E stain, ×100).
Figure 5.

Photomicrograph showing the germinal center within the lymphoid aggregate (H&E stain, ×100)
DISCUSSION
Brachial cleft cyst is a common cause of soft tissue swelling in the neck of a young adult. They generally occur unilaterally and are typically seen in the lateral aspect of the neck. It is clinically apparent in late childhood or early adulthood. In older adults with this presentation, it is important to exclude metastatic lymphadenopathy, lymphoma or tuberculosis.[5]
Etiology of branchial cleft cyst is controversial. There are four main theories of origin of branchial cyst postulated till now. These include incomplete obliteration of branchial mucosa, persistence of vestiges of the pre-cervical sinus, thymo-pharyngeal ductal origin and cystic lymph node origin.[2,6]
The branchial apparatus that begins to form in the second week of fetal life and is completed by the sixth or seventh week; which is probably the structure most widely believed to be the source of branchial cyst. Ascherson in 1832 suggested that branchial cleft cyst arises from incomplete obliteration of branchial cleft mucosa, which remains dormant until stimulated to grow later in life, resulting in cyst formation. Specifically the lack of degeneration of the cervical sinus created by the growth of the second arch over the third and fourth arches is the proposed cause. The third and fourth arches thus overlaid by the second arch persist as small pockets with their ectodermal epithelium. These pockets usually fill in during fetal development; however when they do not, cysts, sinuses and fistulas may arise later. Second theory proposed by him suggests that, branchial fistulas are vestiges of the cervical sinus, rather than of the pharyngeal clefts or pouches.[4] Parallel to the branchial theory, this precervical sinus theory was extended to include lateral cervical cysts. Wenglowski suggested that cystic degeneration of cervical lymph nodes was the mechanism by which lateral cervical cysts were formed. He also suggested that incomplete obliteration of the thymopharyngeal duct resulted in a lateral cervical cyst. Bhaskar and Bernier suggested that cystic alteration of cervical lymph node is stimulated by entrapped epithelium. They suggested three possible sources of these epithelial inclusions: brachial cleft, pharyngeal pouch and parotid gland. At present, to clarify the origin of these cysts, immunohistochemistry is being used to study, at the molecular level, the nature of the epithelial cells and their relationship to other type of epithelium elsewhere in the body.[2,7]
Radiologic Investigations: A CT scan or an MRI scan will show a picture of a fluid-filled cyst and will outline its size and anatomic relationships. The CT scan will also show the characteristic thick wall of a branchial cyst.
Histopathology: The lining of a branchial cyst is generally stratified squamous epithelium but sometimes it is pseudostratified, columnar and ciliated. The lining may be ulcerated. The connective tissue wall contains abundant lymphoid tissue, which shows germinal centers.[3,8]
Differential diagnosis: A possibility of metastatic squamous cell carcinoma may be considered as a differential at this location of the neck. A thyroglossal tract cyst may also be considered as a differential diagnosis. Others differentials to be considered are tuberculosis-related lymphadenitis (scrofula) and HIV-related lymphadenopathy, cat-scratch disease, sarcoidosis and Hodgkin lymphoma.[8,9,10]
Treatment and Prognosis: Branchial cysts are excised and their residual tract ligated. Branchial cysts are permanently eradicated by surgical excision. Rarely, a recurrence or carcinoma within or associated with the cyst may develop.[8]
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Wei YK, Wang KJ, Jou YL, Chang YH, Chou CS. Type II first branchial cleft anomaly - A case report. Tzu Chi Med J. 2005:357–60. [Google Scholar]
- 2.Arndal H, Bonding P. First branchial cleft anomaly. Clin Otolaryngol Allied Sci. 1996;21:203–7. doi: 10.1111/j.1365-2273.1996.tb01725.x. [DOI] [PubMed] [Google Scholar]
- 3.D’Souza AR, Uppal HS, De R, Zeitoun H. Updating concepts of first branchial cleft defects: A literature review. Int J Pediatr Otorhinolaryngol. 2002;62:103–9. doi: 10.1016/s0165-5876(01)00612-7. [DOI] [PubMed] [Google Scholar]
- 4.Frazer JE. The nomenclature of diseased states caused by certain vestigial structures in the neck. Br J Surg. 1923;11:131–6. [Google Scholar]
- 5.McClure MJ, McKinstry CS, Stewart R, Madden M. Late presentation of brachial cyst. Ulster Med J. 1998;67:129–31. [PMC free article] [PubMed] [Google Scholar]
- 6.Glosser JW, Pires CA, Feinberg SE. Branchial cleft or cervical lymphoepithelial cysts: Etiology and management. J Am Dent Assoc. 2003;134:81–6. doi: 10.14219/jada.archive.2003.0020. [DOI] [PubMed] [Google Scholar]
- 7.Little JW, Rickles NH. The histogenesis of the brachial cyst. Am J Pathol. 1967;50:533–47. [PMC free article] [PubMed] [Google Scholar]
- 8.Marx RE, Stern D. 2nd edition. Lllinois: Quintessence Publishing Co, Inc; 2012. Oral and Maxillofacial Pathology A Rational for Diagnosis and Treatment. [Google Scholar]
- 9.Thomaidis V, Seretis K, Tamiolakis D, Papadopoulos N, Tsamis I. Branchial cysts. A report of 4 cases. Acta Dermatovenerol Alp Panonica Adriat. 2006;15:85–9. [PubMed] [Google Scholar]
- 10.Chen MF, Ueng SH, Jung SM, Chen YL, Chang KP. A type ii first branchial cleft cyst masquerading as an infected parotid warthin's tumor. Chang Gung Med J. 2006;29:435–9. [PubMed] [Google Scholar]


