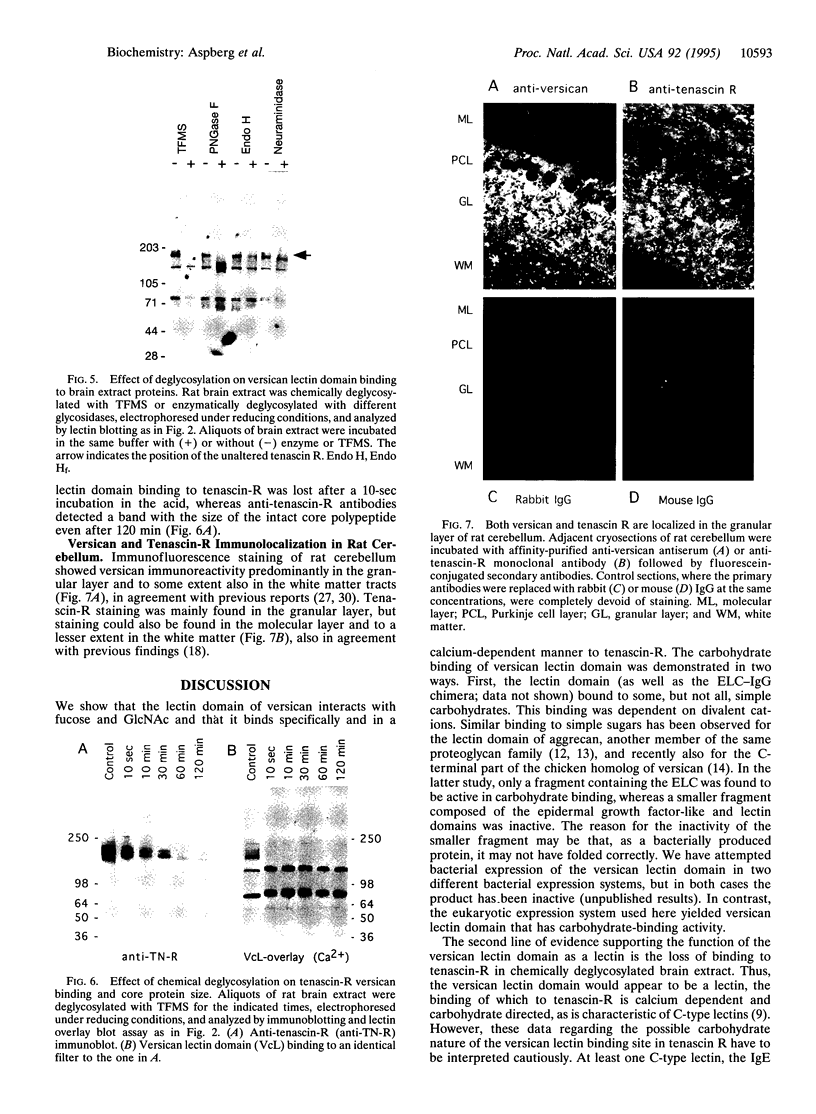
Abstract
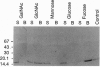
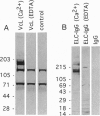
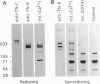
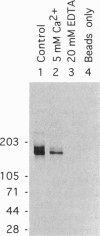
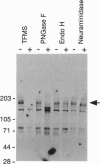
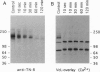
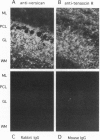
The core proteins of large chondroitin sulfate proteoglycans contain a C-type lectin domain. The lectin domain of one of these proteoglycans, versican, was expressed as a recombinant 15-kDa protein and shown to bind to insolubilized fucose and GlcNAc. The lectin domain showed strong binding in a gel blotting assay to a glycoprotein doublet in rat brain extracts. The binding was calcium dependent and abolished by chemical deglycosylation treatment of the ligand glycoprotein. The versican-binding glycoprotein was identified as the cell adhesion protein tenascin-R, and versican and tenascin-R were both found to be localized in the granular layer of rat cerebellum. These results show that the versican lectin domain is a binding domain with a highly targeted specificity. It may allow versican to assemble complexes containing proteoglycan, an adhesion protein, and hyaluronan.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Argraves W. S., Suzuki S., Arai H., Thompson K., Pierschbacher M. D., Ruoslahti E. Amino acid sequence of the human fibronectin receptor. J Cell Biol. 1987 Sep;105(3):1183–1190. doi: 10.1083/jcb.105.3.1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aruffo A., Stamenkovic I., Melnick M., Underhill C. B., Seed B. CD44 is the principal cell surface receptor for hyaluronate. Cell. 1990 Jun 29;61(7):1303–1313. doi: 10.1016/0092-8674(90)90694-a. [DOI] [PubMed] [Google Scholar]
- Bevilacqua M. P., Nelson R. M. Selectins. J Clin Invest. 1993 Feb;91(2):379–387. doi: 10.1172/JCI116210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bignami A., Dahl D. Brain-specific hyaluronate-binding protein: an immunohistological study with monoclonal antibodies of human and bovine central nervous system. Proc Natl Acad Sci U S A. 1986 May;83(10):3518–3522. doi: 10.1073/pnas.83.10.3518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bignami A., Perides G., Rahemtulla F. Versican, a hyaluronate-binding proteoglycan of embryonal precartilaginous mesenchyma, is mainly expressed postnatally in rat brain. J Neurosci Res. 1993 Jan;34(1):97–106. doi: 10.1002/jnr.490340110. [DOI] [PubMed] [Google Scholar]
- Doege K. J., Sasaki M., Kimura T., Yamada Y. Complete coding sequence and deduced primary structure of the human cartilage large aggregating proteoglycan, aggrecan. Human-specific repeats, and additional alternatively spliced forms. J Biol Chem. 1991 Jan 15;266(2):894–902. [PubMed] [Google Scholar]
- Drickamer K. Two distinct classes of carbohydrate-recognition domains in animal lectins. J Biol Chem. 1988 Jul 15;263(20):9557–9560. [PubMed] [Google Scholar]
- Faissner A., Steindler D. Boundaries and inhibitory molecules in developing neural tissues. Glia. 1995 Apr;13(4):233–254. doi: 10.1002/glia.440130402. [DOI] [PubMed] [Google Scholar]
- Friedlander D. R., Milev P., Karthikeyan L., Margolis R. K., Margolis R. U., Grumet M. The neuronal chondroitin sulfate proteoglycan neurocan binds to the neural cell adhesion molecules Ng-CAM/L1/NILE and N-CAM, and inhibits neuronal adhesion and neurite outgrowth. J Cell Biol. 1994 May;125(3):669–680. doi: 10.1083/jcb.125.3.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuss B., Wintergerst E. S., Bartsch U., Schachner M. Molecular characterization and in situ mRNA localization of the neural recognition molecule J1-160/180: a modular structure similar to tenascin. J Cell Biol. 1993 Mar;120(5):1237–1249. doi: 10.1083/jcb.120.5.1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grumet M., Flaccus A., Margolis R. U. Functional characterization of chondroitin sulfate proteoglycans of brain: interactions with neurons and neural cell adhesion molecules. J Cell Biol. 1993 Feb;120(3):815–824. doi: 10.1083/jcb.120.3.815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grumet M., Milev P., Sakurai T., Karthikeyan L., Bourdon M., Margolis R. K., Margolis R. U. Interactions with tenascin and differential effects on cell adhesion of neurocan and phosphacan, two major chondroitin sulfate proteoglycans of nervous tissue. J Biol Chem. 1994 Apr 22;269(16):12142–12146. [PubMed] [Google Scholar]
- Halberg D. F., Proulx G., Doege K., Yamada Y., Drickamer K. A segment of the cartilage proteoglycan core protein has lectin-like activity. J Biol Chem. 1988 Jul 5;263(19):9486–9490. [PubMed] [Google Scholar]
- Hoffman S., Crossin K. L., Edelman G. M. Molecular forms, binding functions, and developmental expression patterns of cytotactin and cytotactin-binding proteoglycan, an interactive pair of extracellular matrix molecules. J Cell Biol. 1988 Feb;106(2):519–532. doi: 10.1083/jcb.106.2.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman S., Edelman G. M. A proteoglycan with HNK-1 antigenic determinants is a neuron-associated ligand for cytotactin. Proc Natl Acad Sci U S A. 1987 Apr;84(8):2523–2527. doi: 10.1073/pnas.84.8.2523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horvath E., Edwards A. M., Bell J. C., Braun P. E. Chemical deglycosylation on a micro-scale of membrane glycoproteins with retention of phosphoryl-protein linkages. J Neurosci Res. 1989 Nov;24(3):398–401. doi: 10.1002/jnr.490240309. [DOI] [PubMed] [Google Scholar]
- Krusius T., Gehlsen K. R., Ruoslahti E. A fibroblast chondroitin sulfate proteoglycan core protein contains lectin-like and growth factor-like sequences. J Biol Chem. 1987 Sep 25;262(27):13120–13125. [PubMed] [Google Scholar]
- Lasky L. A. Selectins: interpreters of cell-specific carbohydrate information during inflammation. Science. 1992 Nov 6;258(5084):964–969. doi: 10.1126/science.1439808. [DOI] [PubMed] [Google Scholar]
- LeBaron R. G., Zimmermann D. R., Ruoslahti E. Hyaluronate binding properties of versican. J Biol Chem. 1992 May 15;267(14):10003–10010. [PubMed] [Google Scholar]
- Perides G., Rahemtulla F., Lane W. S., Asher R. A., Bignami A. Isolation of a large aggregating proteoglycan from human brain. J Biol Chem. 1992 Nov 25;267(33):23883–23887. [PubMed] [Google Scholar]
- Pesheva P., Spiess E., Schachner M. J1-160 and J1-180 are oligodendrocyte-secreted nonpermissive substrates for cell adhesion. J Cell Biol. 1989 Oct;109(4 Pt 1):1765–1778. doi: 10.1083/jcb.109.4.1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rauch U., Karthikeyan L., Maurel P., Margolis R. U., Margolis R. K. Cloning and primary structure of neurocan, a developmentally regulated, aggregating chondroitin sulfate proteoglycan of brain. J Biol Chem. 1992 Sep 25;267(27):19536–19547. [PubMed] [Google Scholar]
- Rogelj S., Weinberg R. A., Fanning P., Klagsbrun M. Basic fibroblast growth factor fused to a signal peptide transforms cells. Nature. 1988 Jan 14;331(6152):173–175. doi: 10.1038/331173a0. [DOI] [PubMed] [Google Scholar]
- Saleque S., Ruiz N., Drickamer K. Expression and characterization of a carbohydrate-binding fragment of rat aggrecan. Glycobiology. 1993 Apr;3(2):185–190. doi: 10.1093/glycob/3.2.185. [DOI] [PubMed] [Google Scholar]
- Shinomura T., Nishida Y., Ito K., Kimata K. cDNA cloning of PG-M, a large chondroitin sulfate proteoglycan expressed during chondrogenesis in chick limb buds. Alternative spliced multiforms of PG-M and their relationships to versican. J Biol Chem. 1993 Jul 5;268(19):14461–14469. [PubMed] [Google Scholar]
- Springer T. A. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell. 1994 Jan 28;76(2):301–314. doi: 10.1016/0092-8674(94)90337-9. [DOI] [PubMed] [Google Scholar]
- Subramani S., Mulligan R., Berg P. Expression of the mouse dihydrofolate reductase complementary deoxyribonucleic acid in simian virus 40 vectors. Mol Cell Biol. 1981 Sep;1(9):854–864. doi: 10.1128/mcb.1.9.854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ujita M., Shinomura T., Ito K., Kitagawa Y., Kimata K. Expression and binding activity of the carboxyl-terminal portion of the core protein of PG-M, a large chondroitin sulfate proteoglycan. J Biol Chem. 1994 Nov 4;269(44):27603–27609. [PubMed] [Google Scholar]
- Urlaub G., Chasin L. A. Isolation of Chinese hamster cell mutants deficient in dihydrofolate reductase activity. Proc Natl Acad Sci U S A. 1980 Jul;77(7):4216–4220. doi: 10.1073/pnas.77.7.4216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varki A. Selectin ligands. Proc Natl Acad Sci U S A. 1994 Aug 2;91(16):7390–7397. doi: 10.1073/pnas.91.16.7390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vercelli D., Helm B., Marsh P., Padlan E., Geha R. S., Gould H. The B-cell binding site on human immunoglobulin E. Nature. 1989 Apr 20;338(6217):649–651. doi: 10.1038/338649a0. [DOI] [PubMed] [Google Scholar]
- Yamada H., Watanabe K., Shimonaka M., Yamaguchi Y. Molecular cloning of brevican, a novel brain proteoglycan of the aggrecan/versican family. J Biol Chem. 1994 Apr 1;269(13):10119–10126. [PubMed] [Google Scholar]
- Yamagata M., Shinomura T., Kimata K. Tissue variation of two large chondroitin sulfate proteoglycans (PG-M/versican and PG-H/aggrecan) in chick embryos. Anat Embryol (Berl) 1993 May;187(5):433–444. doi: 10.1007/BF00174419. [DOI] [PubMed] [Google Scholar]
- Zimmermann D. R., Ruoslahti E. Multiple domains of the large fibroblast proteoglycan, versican. EMBO J. 1989 Oct;8(10):2975–2981. doi: 10.1002/j.1460-2075.1989.tb08447.x. [DOI] [PMC free article] [PubMed] [Google Scholar]