Abstract
AIM: To establish a novel coculture system for ex vivo expansion of umbilical cord blood(UCB) hematopoietic progenitors using thrombopoietin (TPO)/Flt-3 ligand (FL)-transduced human marrow-derived mesenchymal stem cells (tfhMSCs) as feeder.
METHODS: UCB CD34+ cells were isolated and cultured using four culture systems in serum-containing or serum-free medium. Suitable aliquots of cultured cells were used to monitor cell production, clonogenic activity, and long-term culture-initiating culture (LTC-IC) output. Finally, the severe-combined immunodeficient (SCID) mouse-repopulating cell (SRC) assay was performed to confirm ability of the cultured cells to reconstitute long-term hematopoiesis.
RESULTS: There were no significant differences in the number of total nucleated cells among different culture systems in serum-containing medium during 21-d culture. However, on d 14, the outputs of CD34+ cells, CFU-C and CFU-GEMM in tfhMSCs coculture system were significantly enhanced. LTC-IC assay demonstrated that the tfhMSCs coculture system had the most powerful activity. The severe-combined immunodeficient (SCID) mouse repopulating cell (SRC) assay confirmed extensive ability of the expanded cells to reconstitute long-term hematopoiesis. Furthermore, PCR analysis demonstrated the presence of human hematopoietic cells in the bone marrow and peripheral blood cells of NOD/SCID mice.
CONCLUSION: The TPO/FL-transduced hMSCs, in combination with additive cytokines, can effectively expand hematopoietic progenitors from UCB in vitro and the tfhMSCs coculture system may be a suitable system for ex vivo manipulation of primitive progenitor cells under contact culture conditions.
Keywords: Mesenchymal stem cells, Thrombopoietin, Flt-3 ligand, Hematopoiesis
INTRODUCTION
Hematopoietic stem cells (HSCs) are generally defined as cells having the self-renewing potential and the capacity to give rise to differentiated cells of all hematopoietic lineages[1]. Therefore, HSC transplantation is performed for complete healing of hematologic disorders and as a supportive therapy after high-dose chemotherapy against malignant diseases. HSCs can be collected from peripheral blood (PB), bone marrow (BM), and umbilical cord blood (UCB). Human UCB is thought to contain a high number of primitive hematopoietic cells, because the number of severe combined immunodeficiency (SCID)-repopulating cells (SRCs) in nonobese diabetic/SCID (NOD/SCID) mice that had received transplants from UCB was higher than that in NOD/SCID mice that had received transplants from other sources[2-4]. Moreover, the frequency of graft-versus-host disease, which is a severe side effect of HSC transplantation in patients, is reduced among patients receiving transplants from UCB[5], and UCB can be obtained from the cord-blood bank network. However, the total number of UCB HSCs harvested from one donor’s UCB is limited and is not sufficient for HSC transplantation in an adult patient. To overcome this problem, attention has been increasingly focused on ex vivo expansion of HSCs. Many approaches have been reported during the last decade, and they can be divided into 2 categories. The first category is treatment of HSCs with various combinations of cytokines. Treatment with the following combinations of cytokines increased the progenitor/stem cell population by 2- to 30-fold in the relatively short period of 10 to 14 d: Flt-3 ligand (FL), stem cell factor (SCF), and thrombopoietin (TPO); SCF, granulocyte-colony stimulating factor (G-CSF), and megakaryocyte growth and development factor (MGDF); FL, SCF, G-CSF, interleukin-3 (IL-3), and interleukin-6 (IL-6); and FL, SCF, and IL-6[6-9]. However, it is difficult to maintain HSC activity in long-term cultures even if the total number of hematopoietic cells could be expanded. Hence, these methods could be improved for use in clinical settings. The second category involves using stromal cells. It has been reported that the SCID-repopulating activity (SRA) of human HSCs could be maintained by coculture with murine stromal cells for 7 wk[10], and that the SRA could be maintained by coculture with the AGM-S3 stromal cell line for 4 wk[11]. MS-5 expanded SRCs for 2 wk[12]; FBMD-1 expanded cobblestone area-forming cells by 90-fold[13]; HESS-5 expanded SRCs for only 5 d[14,15]. Contact between HSCs and stromal cells is important for maintaining the function of HSCs[16,17]. However, when human HSCs are cocultured with nonhuman stromal cells, the expanded human HSCs might have a risk of being exposed to an unknown viral contamination in animal stromal cells.
Several methods of ex vivo expansion using human primary stromal cells were reported[18,19]. When HSCs were cocultured with human primary stromal cells, the HSCs were expanded for 2 to 4 wk. However, in general, when human primary somatic cells divide in an in vitro culture, the telomeric DNA at the end of the chromosome shortens at each cell division. Then, the replication of human primary cells slows (aging occurs), and the cells finally cease to divide (crisis phase)[20,21]. To obtain a sufficient number of primary stromal cells for use on a clinical scale, we have to harvest BM many times, and we cannot ignore the burden on the donor. To solve this problem, trials to establish human stromal cell lines using transduction of viral antigens such as human papillomavirus (HPV) E6/E7 and simian virus 40 (SV40) large T have been reported[22-24]. These stromal cells could maintain HSCs, but the possibility of transformation was mentioned[25].
Recently, it was shown that Mesenchymal stem/progenitor cells in human UCB and placenta could support ex vivo expansion of CD34+ hematopoietic stem cells[26,27]. However, no report has investigated whether transduced human marrow-derived mesenchymal stem cells (hMSCs) could be useful in ex vivo expansion of UCB hematopoietic progenitors. Previous research has shown that TPO and FL, the two early-acting cytokines, could lead to significant expansion of HSC populations, including long-term culture-initiating cells (LTC-IC). Our group have introduced TPO and FL genes into cultured hMSCs by retroviral vector transfer and demonstrated long-term expression in vitro and in vivo. In this study, we attempted to establish a coculture system for ex vivo expansion of UCB hematopoietic progenitors using TPO/FL-transduced hMSCs (tfhMSCs) as feeder. As a result, tfhMSCs is capable of expanding UCB hematopoietic cells in synergy with extra cytokines in vitro.
MATERIALS AND METHODS
Analysis of expression of the TPO and FL gene by enzyme-linked immunosorbent assay
Untransduced and TPO/FL-transduced hMSCs (tfhMSCs) were cultured in minimal essential medium α (MEM-α; HyClone, Logan, UT, USA) supplemented with 100 mL/L fetal bovine serum (FBS; GibcoBRL, GrandIsland, NY, USA) at 37 °C with 50 mL/L CO2 in humidified air. For assaying TPO and FL secretion, untransduced hMSCs and/or tfhMSCs were passaged when cells reached 90% confluence by transferring 2.5 ×106 to 5.0 ×106 cells into a 75-cm2 flask with 12 mL of hMSCs medium. Twenty-four hours later, 1 mL of culture supernatant was collected and stored at -80 °C. The assay was performed in triplicate using the TPO and FL ELISA kit (BioSource International, Camarillo, CA, USA). The level of TPO and FL was normalized to the level of endogenously expressed IL-6 measured with an IL-6 ELISA kit (BioSource International) using the procedures suggested by manufacturer. Plates were read on a microplate reader (Bio-Rad Laboratories, Hercules, CA, USA) and the data were analyzed using Microsoft Excel.
UCB sample collection and CD34+ cell purification
UCB were collected from normal full-term pregnancies according to the regulations of the Research Ethics Committee of Women’s Hospital, School of Medicine Zhejiang University. Mononuclear cells (MNC) were isolated using Ficoll-Hypaque (1.077 ± 0.001 Kg/L, Sigma, St. Louis, MO), washed, and resuspended in Iscove’s modified Dulbecco’s medium (IMDM; HyClone, Logan, UT) supplemented with 100 mL/L fetal bovine serum (FBS; GibcoBRL, GrandIsland, NY). CD34+ cell purification utilized positive selection using the miniMACS immunomagnetic separation system (Miltenyi Biotec GmbH, Glodbach, Germany) according to the manufacturer’s instructions. Briefly, MNCs were suspended in buffer containing phosphate-buffered saline (PBS), 5 mL/L bovine serum albumin (BSA; Sigma), and 2 mmol/L EDTA (BSA-EDTA-PBS), and incubated for 15 min with monoclonal hapten-conjugated anti-CD34 antibody (clone: QBEND/10) and human Ig to prevent nonspecific binding. Washed cells were resuspended in BSA-EDTA-PBS and incubated for 15 min with colloidal super-paramagnetic microbeads conjugated to an anti-hapten antibody. After labeling, the cell suspension was passed through a column (VS+ separation column) held within a magnetic field causing CD34+ cells to be retained in the column. CD34+ cells were collected by removal of the column from the magnet and washing with BSA-EDTA-PBS. Ninety-six percent or more of the enriched cells were CD34+ by flow cytometric analysis.
Human cytokines
Recombinant human TPO, granulocyte-macrophage colony-stimulating factor (GM-CSF), and erythropoietin (EPO) were purchased from Peprotech (London, UK). IL-3 and IL-6 was purchased from RELIATech GmbH (Braunschweig, Germany). Recombinant human SCF was a gift from Amgen Biologicals (Thousand Oaks, CA). Recombinant human FL was purchased from R&D Systems (Minneapolis, MN). The final concentrations of cytokines were as follows: TPO, 50 μg/L; FL, 50 μg/L; IL-3, 20 μg/L; IL-6, 20 μg/L; SCF, 50 μg/L; GM-CSF, 10 μg/L; and EPO, 3 000 U/L.
Culture systems
Stroma-free culture and coculture with tfhMSCs or hMSCs were performed in culture media in 24-well microplates (Costar, Bethesda, MD). Serum-containing liquid culture was carried out using a medium containing 125 mL/L horse serum (HS; HyClone), 125 mL/L FBS, 10-4 mol/L 2-mercaptoethanol (Sigma), 2 mmol/L L-glutamine (Sigma) and IMDM supplemented with 10-6 mol/L hydrocortisone (Sigma) with or without feeder layer. In the coculture, tfhMSCs or hMSCs were seeded at 1 × 105 cells per well with MEM-α supplemented with 100 mL/L FBS. After obtaining a confluent feeder layer, cells were washed five times and subjected to γ-irradiation at a dose of 12 Gy. the medium was then changed for coculture. Totally 20 000 UCB CD34+ cells were expanded for 21 d under four conditions: 1) tfhMSCs coculture system (tfhMSCs + SCF + IL-3 + IL-6 + GM-CSF); 2) hMSCs coculture system (hMSCs + TPO + FL + SCF + IL-3 + IL-6 + GM-CSF); 3) cytokines culture system (TPO + FL + SCF + IL-3 + IL-6 + GM-CSF); 4) hMSCs (TPO/FL-free) culture system (hMSCs + SCF + IL-3 + IL-6 + GM-CSF). On d 7 and 14 of culture, the medium in each well was removed and replaced with fresh medium. On d 7, 14 and 21 of culture, aliquots of cultured cells were harvested and subjected to cell count, clonal cell culture, and flow cytometric analysis when contamination of stromal cells in the harvested cells was negligible (< 2%) by microscopic visualization. On d 14, cultured cells were harvested and subjected to LTC-IC assay and SRC assay. Short-term (7 d) serum-free liquid culture was carried out using StemProTM-34SFM (GibcoBRL) supplemented with StemPro™-34 Nutrient Supplement (GibcoBRL), 2 mmol/L L-glutamine, and penicillin/streptomycin (GibcoBRL).
Immunophenotyping by flow cytometry
Aliquots of cells were suspended in EDTA-BSA-PBS and incubated with mouse IgG (InterCell Technologies, Hopewell, NJ) to block nonspecific binding. Cells were then reacted for 15 min with FITC- and PE-conjugated monoclonal antibodies at 4 °C. Unbound antibodies were removed by two washes, and cells were resuspended in EDTA-BSA-PBS. Stained cells were then passed through a nylon mesh filter and subjected to two-color flow cytometric analysis. Cells labeled with FITC- and PE-conjugated mouse isotype-matched antibodies were used as negative controls. The analysis was performed using an FACsort flow cytometer (Becton Dickinson, San Jose, CA) with CELLQUESTTM software (Becton Dickinson). At least 10 000 events were acquired for each analysis. Antibodies used were as follows: FITC-conjugated CD14, CD15, CD19, CD33, CD34, and CD41; PE-conjugated CD38 and CD45 antibodies. Glycophorin A antibodies were from Immunotech (Marseille, France). CD14, CD33, and CD45 antibodies were from Pharmingen (San Diego, CA) and all others were from Becton Dickinson. Furthermore, in some experiments, aliquots of cultured cells were subjected to three-color flow cytometric analysis to assess the lineage commitment of progenitors. Samples were incubated for 15 min with biotin-conjugated anti-CD34 (Immunotech, Marseille, France). Cells labelled with a biotin-conjugated mouse isotype-matched antibody were used as a negative control. After washing, cells were labeled with streptavidin PerCP (Becton Dickinson), PE-conjugated anti-CD38, and various FITC-conjugated monoclonal antibodies. Three-color flow cytometry was performed using an FACSCalibur (Becton Dickinson) with CellQuest software (Becton Dickinson).
Colony-forming cell assay (CFC assay)
Aliquots from initial UCB samples or cultured cells were incubated in methylcellulose media at concentrations of 1-2 × 105 cells/L for purified CD34+ cells and 5-10 × 105 cells/L for cultured cells in 35-mm tissue culture dishes (Costar). One milliliter of culture mixture contained 12 mL/L 1500 cp methylcellulose (Sigma), MEM-α, 10 g/L deionized fraction V BSA (Sigma), 10-4 mol/L 2-mercaptoethanol, 300 mL/L fetal calf serum (JRH Biosciences, Lenexa, KS), EPO, IL-3, SCF, GM-CSF, and cells. Dishes were incubated at 37 °C in a humidified atmosphere with 50 mL/L CO2 in air. All cultures were done in triplicate. Total colony-forming units in culture (CFU-C) and mixed colonies containing erythroid and myeloid cells and megakaryocytes (CFU-GEMM) consisting of 50 or more cells were scored under an inverted microscope at 21 d of culture. To assess the accuracy of in situ identification, individual colonies were lifted with an Eppendorf micropipette under direct microscopic visualization, spread on glass slides using a cytocentrifuge and studied with May-Grunwald-Giemsa staining.
Long-term culture-initiating cell assay (LTC-IC assay)
LTC-IC assay was performed as described by Sutherland et al.[28], with slight modifications. Briefly, bone marrow stromal cells derived from hematologically normal donors were seeded at 105 cells per well in 96-well flat-bottomed plates (Costar) with MEM-α supplemented with 100 mL/L FBS. After obtaining semiconfluent feeder layers, stromal cells were irradiated with 15-Gy using a 60Co γ-irradiator. CD34+ cell subpopulations purified from UCB or those isolated from cultured cells by sorting with an FACSVantage (Becton Dickinson) were seeded at limiting dilution on the feeder layer in serum-containing media. For each evaluation, at least three cell concentrations were used with 24 replicates per concentration. Culture plates were incubated at 37 °C with 50 mL/L CO2 in air and weekly changes of medium. After 5 wk of culture, cells were assayed for CFU-C in methylcellulose medium. Colonies were scored 2 wk later. The frequency of wells in which there were no clonogenic progenitors was determined according to the number of the initial input population. Poisson statistics were applied to the single-hit model and the frequency of LTC-IC was calculated with the maximum likelihood estimator.
SCID-repopulating cells assay (SRC assay)
SRC assay was performed as previously described[29], with slight modifications. Briefly, 8-wk-old male NOD/Shi-scid (NOD/SCID) mice were obtained from the Central Institute for Experimental Animals, Shanghai Institutes for Biological Sciences, CAS. All animals were handled under sterile conditions and maintained under microisolators in the animal facility located at Zhejiang Academy of Medical Sciences. Human hematopoietic cells at the indicated doses were transplanted by tail-vein injection into sublethally irradiated mice (350 cGy using a linear accelerator). Cells were co-transplanted with irradiated (15 Gy using a 60Co γ-irradiator) nonrepopulating CD34- cells as accessory cells. Mice were killed 7 wk after transplantation, and the bone marrow (from the femurs and tibiae) and peripheral blood cells (from the retro-orbital venous plexus using heparin-coated micropipettes) were harvested. The presence of human hematopoietic cells was determined by detection of cells positively stained with FITC-conjugated antihuman CD45 using flow cytometry. Polymerase chain reaction (PCR) analysis using human Alu sequence primers (5’ - GTGGGCGACAGAACGAGATTCTAT; 5’ - CTCACTACTTGGAGACAGGTTCA) was also performed to confirm flow cytometric results.
Statistical analysis
Results are expressed as mean ± SD. Statistical comparisons were performed using the two-sided Student’s t-test. Iterative approximation of Newton’ method was performed using Microsoft Visual Basic 6.0 software.
RESULTS
Analysis of expression of TPO and FL gene from tfhMSCs and untransduced hMSCs
We transduced hMSCs with the secreted cytokine TPO and FL, which allows direct quantitation of the extracellular product. Human MSCs were transduced with a retroviral vector (pLXINTF) expressing TPO, FL and neomycin followed by selection in the presence of G418 for 2 wk. Cultures were maintained and expanded further for up to 8 passages. Twenty-four hours after each replating aliquots of supernatant from tfhMSCs and untransduced hMSCs was removed for measurement of secreted TPO and FL. The TPO and FL secretion value obtained was normalized to the secretion value obtained for endogenous human IL-6 measured in the same sample aliquot. The absolute values for each cytokine are shown in Figure 1. We chose IL-6 as the control cytokine because expression was in a range similar to that of the transduced gene product and previous data showed similar expression of IL-6 from hMSCs over time in hMSC cultures[30].
Figure 1.
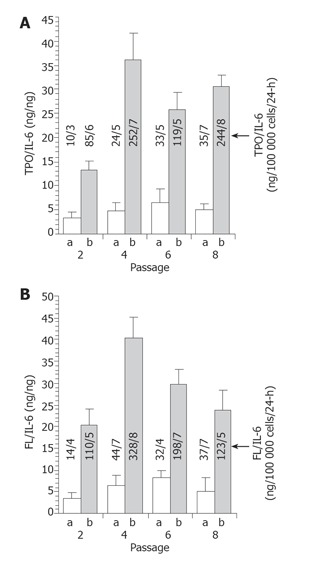
Maintenance of TPO and FL production in vitro. TPO (A) and FL (B) levels secreted from tfhMSCs and untransduced hMSCs and endogenous IL-6 levels were assayed in the medium after 2, 4, 6, and 8 passages in culture. The level of TPO and FL in the supernatant was normalized to the level of endogenous IL-6 and the ratio was plotted for passage number. In addition, the absolute values (ng/100 000 cells/24 h) of TPO, FL and IL-6 are shown (arrow). Data were obtained from triplicate ELISA measurements. a = untransduced hMSCs; b = tfhMSCs.
We observed TPO transgene expression from tfhMSCs and untransduced hMSCs averaging 175 ± 85 and 25 ± 11 ng/105cells/24 h respectively (Figure 1A) and FL transgene expression averaging 190 ± 99 and 31 ± 12 ng/105 cells/24 h respectively (Figure 1B); Endogenous IL-6 protein levels averaging 6 ± 2 ng/105cells/24 h. The cytokine ratio (transduced/endogenous) of tfhMSCs demonstrated a 14- to 36-fold and 22- to 41-fold increase respectively in TPO and FL secretion over endogenous IL-6 expression. The results demonstrate that in vitro transgene expression from tfhMSCs was maintained for at least three months in culture.
Assessment of the supportive effects of tfhMSCs on proliferation of UCB primitive progenitor cells (PPC) in synergy with extra cytokines
To determine whether tfhMSCs was capable of supporting ex vivo expansion of UCB-derived hematopoietic cells, four culture systems were established as shown in materials and methods. In the stroma-containing culture, tfhMSCs or untransduced hMSCs were irradiated and cocultured with CD34+ cells from UCB, and in the stroma-free culture only cytokines were used.for expansion. The total number of nucleated cells, CD34+ cells, CFU-C and CFU-GEMM was evaluated on d 7, 14 and 21. CD34 and CD38 expression among hematopoietic cells in four culture conditions were also determined on d 14.
We first assessed the effect of tfhMSCs on ex vivo expansion of UCB-PPC in the serum-containing culture. As a result, tfhMSCs alone could not effectively support proliferation of UCB hematopoietic progenitors (data not shown). The effects of four culture systems on ex vivo expansion of total nucleated cells and CD34+ cells were then studied in serum-containing medium. During culture, there were no significant differences in the number of total nucleated cells among different culture systems (Figure 2A).The expansion magnitude of CD34+ cells by tfhMSCs coculture system (11.52 ± 1.51 fold) and cytokines culture system (12.32 ± 2.69 fold) was lower than that by hMSCs coculture system (15.40 ± 1.89 fold) on d 7 (P < 0.05), and hMSCs (TPO/FL-free) culture system (6.59 ± 1.26 fold) manifested the lowest expansion capacity (P < 0.01) among the four groups (Figure 2B). However, on d 14, CD34+ cells were generated more by the tfhMSCs coculture system (35.42 ± 2.25 fold) than by hMSCs coculture system (25.24 ± 3.32 fold), cytokines culture system (19.75 ± 2.56 fold) and hMSCs (TPO/FL-free) culture system (11.32 ± 2.48 fold) (P < 0.01)(Figure 2B). Although fold increase of CD34+ cells was largely decreased among all the four systems on d 21, the expansion magnitude of CD34+ cells by tfhMSCs coculture system (20.15 ± 3.16 fold, P < 0.01) was higher than that by the three systems (Figure 2B).
Figure 2.
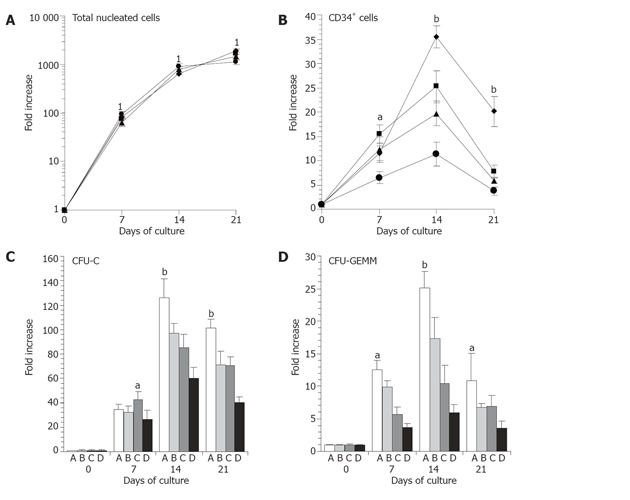
Effects of several culture systems on ex vivo expansion of hematopoietic progenitors. UCB CD34+ cells were cultured in serum-containing medium using four culture systems. A: Total nucleated cells; B: CD34+ cells; C: Total colony-forming units in culture (CFU-C); D: Mixed colonies containing erythroid and myeloid cells and megakaryocytes (CFU-GEMM). The results represent the mean fold increase ± SD of three different experiments on d 7, 14, and 21 of culture. 1No significant differences between different culture systems. aP < 0.05, bP < 0.01 as compared with other three groups. ◆= tfhMSCs coculture system; ▓= hMSCs coculture system; ▲= cytokines culture system; ● = hMSCs (TPO/FL-free) culture system. A = tfhMSCs coculture system; B=hMSCs coculture system; C = cytokines culture system; D = hMSCs (TPO/FL-free) culture system.
Next, we determined the outputs of CFU-C and CFU-GEMM. The expansion magnitude of CFU-C in tfhMSCs coculture system (126.54 ± 15.42 fold), although lower than cytokines culture system on d 7 (P < 0.05), was much higher than other three culture systems on d 14 (P < 0.01). Interestingly, tfhMSCs coculture system significantly stimulated production of CFU-C on d 21 (101.12 ± 7.23 fold, P < 0.01) (Figure 2C). The output of CFU-GEMM in tfhMSCs coculture system on d 7, 14 and 21 was also enhanced (12.65 ± 1.36 fold, 25.16 ± 2.53 fold and 10.93 ± 4.12 fold, respectively) (P < 0.01) (Figure 2D). Analysis for expression of CD34 and CD38 by flow cytometry among the four culture systems on d 14 was shown in Figure 3.
Figure 3.
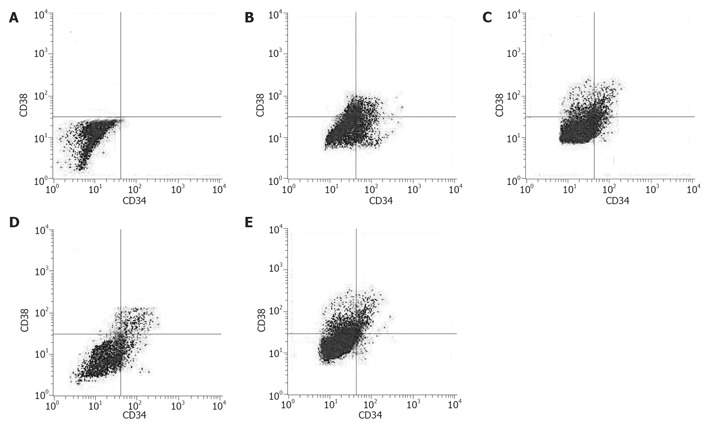
Comparison of CD34 and CD38 expression among hematopoietic cells in different culture systems. CD34+ cells derived from a single delivery were cultured using different culture systems in serum-containing medium. On d 14 of culture, aliquots of harvested cells were subjected to flow cytometric analysis. A = Negative control; B = tfhMSCs coculture system; C = hMSCs coculture system; D = cytokines culture system; E = hMSCs (TPO/FL-free) culture system.
Ex vivo expansion of UCB CD34+ cells in a short-term (7 d) serum-free tfhMSCs coculture system
The synergistic effects of tfhMSCs and additive cytokines on ex vivo expansion of UCB hematopoietic progenitors was also studied in serum-free culture for a short duration. After 7 d of culture, the number of total nucleated cells in tfhMSCs coculture system was remarkably increased (P < 0.01)(Table 1). As a result, the mean number of total nucleated cells was approximately 50 to 90 times the initial input number. CD34+ cells were generated more by tfhMSCs coculture system than by other three groups (P < 0.01); the number of CD34+ cells was over 12 times the initial input number. The outputs of CFU-C and CFU-GEMM in tfhMSCs coculture system were also increased approximately 40-fold (P < 0.05) and 15-fold (P < 0.01), respectively.
Table 1.
Evaluation of effects of four culture systems on ex vivo expansion of hematopoietic progenitors in short-term serum-free culture
| Culture systems | Total nucleated cells | CD34+ cells | CFU-C | CFU-GEMM |
| tfhMSCs coculture system | 87.56 ± 9.51b | 12.39 ± 2.34b | 40.59 ± 6.23a | 15.84 ± 3.96b |
| hMSCs coculture system | 53.23 ± 6.79a | 9.87 ± 2.89a | 38.78 ± 4.28a | 10.57 ± 2.58b |
| Cytokines culture system | 46.25 ± 4.89 | 9.58 ± 1.26a | 27.68 ± 3.32 | 7.19 ± 1.96 |
| hMSCs (TPO/FL-free) culture system | 48.52 ± 6.49 | 6.79 ± 0.62 | 32.85 ± 4.35 | 5.67 ± 1.09 |
P < 0.05,
P < 0.01 vs as compared with hMSCs (TPO/FL-free) culture system.
LTC-IC assay using the CD34+ population isolated from cells cultured in four culture systems
To determine whether cells generated in four culture systems could preserve the ability to sustain long-term hematopoiesis, the LTC-IC frequency in cells cultured by these systems was quantified. Initially, isolated UCB CD34+ cells were cultured for 14 d using four culture systems in the serum-containing culture. Cultured cells were harvested and subjected to a second CD34+ cell purification by sorting. LTC-IC assay was performed using sorted CD34+ cell populations, as well as those initially prepared from UCB (control samples). The LTC-IC frequency was determined as previously described[28]. As shown in Table 2, Although the yields of LTC-IC expansion in hMSCs coculture system (5.32 ± 1.73 fold) was higher than cytokines culture system (3.58 ± 1.48 fold) and hMSCs (TPO/FL-free) culture system (2.79 ± 0.56 fold), the tfhMSCs coculture system demonstrated the most powerful activity (10.23 ± 2.89 fold, P < 0.01). The findings suggest that the tfhMSCs coculture system might be a novel as well as efficient culture system for UCB-derived hematopoietic progenitor cells.
Table 2.
Results of LTC-IC assay using UCB CD34+ cells or those generated by four culture systems
| Culture systems |
LTC-IC frequency |
Fold LTC-IC amplification | |
| Pre-expansion | Post-expansion | ||
| hMSCs (TPO/FL-free) culture system | 1/619.57 ± 120.89 | 1/102 258.56 ± 9 568.49 | 2.79 ± 0.56 |
| Cytokines culture system | 1/625.38 ± 154.26 | 1/84 112.78 ± 10 867.64 | 3.58 ± 1.48 |
| hMSCs coculture system | 1/621.76 ± 138.59 | 1/93 773.42 ± 20 346.23 | 5.32 ± 1.73 |
| tfhMSCs coculture system | 1/617.43 ± 119.76 | 1/24 140.96 ± 7 586.62 | 10.23 ± 2.89b |
P < 0.01 vs calculated by the Student’s t-test, when data of the tfhMSCs coculture system were compared with data in the other groups.
Effects of coculture system using tfhMSCs as feeder layer on human reconstituting hematopoietic progenitors
Accordingly, we studied the SRC assay to determine whether cells cultured in the tfhMSCs coculture system were capable of long-term multilineage reconstitution in vivo. Purified 1 × 105 UCB CD34+ cells were initially cultured for 14 d using the tfhMSCs coculture system; harvested cells were then transplanted into NOD/SCID mice. As controls, uncultured 100 000 UCB CD34+ cells obtained from the same sources were also transplanted into other mice. Seven weeks after transplantation, human CD45+ cells were found in the bone marrow, and peripheral blood cells of mice transplanted with the cultured cells (Figure 4A, and data not shown). There were marked differences in the percentage of chimerism between bone marrow cells in mice transplanted with cultured cells and those transplanted with control samples. Human CD45+ cells in the murine bone marrow were further subjected to flow cytometric analysis to determine multilineage reconstitution. As a result, human CD45+ cells were positive for CD34, CD33, CD14, CD41, glycophorin A, or CD19 (Figure 4B). Furthermore, PCR analysis demonstrated the presence of human hematopoietic cells in the bone marrow and peripheral blood cells of NOD/SCID mice (Figure 5).
Figure 4.
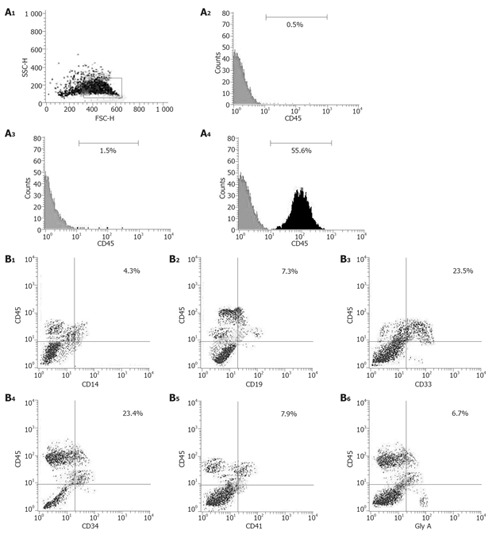
Determination of human hematopoietic reconstitution in NOD/SCID mice 7 wk after transplantation. A: Expression of human CD45 on bone marrow cells (BMC) collected from NOD/SCID mice 7 wk after transplantation; (A1): Cells in the gated area were analyzed by flow cytometry; (A2): Bone marrow cells (BMC) from an untransplanted mouse (negative control); (A3): BMC from a mouse transplanted with 1 ×105 human CD34+ cells before culture; (A4): BMC from a mouse transplanted with cells obtained after 14-d cultivation of those 100 000 human CD34+ cells in the tfhMSCs coculture system; B1-B6: Specific subsets of human CD45+ cells in bone marrow cells of the NOD/SCID mouse transplanted with UCB cells after the coculture. Harvested cells were stained with PE-conjugated CD45 and various FITC-conjugated monoclonal antibodies.
Figure 5.
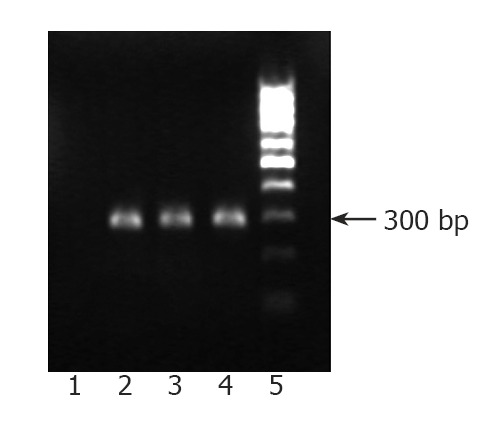
PCR analysis of human Alu sequence in NOD/SCID mice 7 wk after transplantation. DNA was extracted from the bone marrow and peripheral blood cells of NOD/SCID mice 7 wk after transplantation. PCR analysis demonstrated the presence of human hematopoietic cells in the bone marrow (lane 2) and peripheral blood cells (lane 3) of NOD/SCID mice. DNA extracted from human UCB cells was used as a positive control (lane 4). Negative control is shown in lane 1 and DNA marker is shown in lane 5.
DISCUSSION
In this study we demonstrated that TPO/FL–transduced human marrow-derived mesenchymal stem cells (tfhMSCs) could effectively support ex vivo expansion of UCB-PPC in synergy with human cytokines. We assessed the supportive effects of four culture systems on proliferation of PPC in serum-containing liquid medium. Although there were no significant differences in the number of total nucleated cells among the different culture systems during culture, tfhMSCs coculture system could dramatically enhance generation of CFU-C, CFU-GEMM and CD34+ cells, and more importantly was capable of expanding LTC-IC.
In ex vivo expansion of human UCB-PPC, there are several benefits to using the tfhMSCs as the feeder: (1) tfhMSCs can be maintained easily; (2) consistent hematopoietic-supportive effects are repeatedly obtained; (3) additive TPO and FL are not needed in culture system. The effects of tfhMSCs coculture system were comparable to those obtained by classical culture, including cytokines culture and contact culture. Although contamination of tfhMSCs into cultured cells is a problem, and it is difficult to harvest cultured hematopoietic cells completely, since a number of cultured cells migrate under feeder layers, the tfhMSCs coculture system was considered to be a suitable system for ex vivo manipulation of PPC under stroma-contact culture conditions. Moreover, the tfhMSCs, unlike the primary stromal cells[17,18], could be expanded and cryopreserved without transformation. Thus, we can prepare a large quantity of these human tfhMSCs at any time. Taking advantage of this culture system, clinical research on ex vivo expansion could be facilitated. For example, progenitor cells such as CD34+ cells and CFU-C were extensively expanded more than 125-fold in this system in 2 wk, and it may be possible to use these expanded cells as a new source of blood transfusion after differentiation of the expanded cells into megakaryocytes or erythroblast progenitor cells[31].
Although the present study and others[5,8,32] have demonstrated that addition of TPO and FL in culture systems could lead to significant expansion of HSC populations, including LTC-IC, the tfhMSCs coculture system, which have no addition of TPO and FL, manifested higher expansion of LTC-IC. The mechanism of supportive effect of tfhMSCs remains unknown. Previous investigations have shown that the very hematopoietic cell types whose maximum proliferation in vitro depends on stimulation by the highest concentrations of cytokines[32]. The ability that tfhMSCs expressing TPO and FL can maintain a higher concentrations of TPO and FL in medium may be a reason.
Serum-free culture could prevent UCB primitive-cell differentiation[14], and when a serum-free medium is used, the risks of contamination of heterogenic antigen and infectious danger were small[33]. We then assessed the supportive effects of four culture systems on proliferation of PPC in serum-free liquid medium. We attempted ex vivo manipulation for a short duration. As a result, adequate expansion of UCB-PPC was obtained in tfhMSCs coculture system. Furthermore, compared with serum-containing culture the number of CD34+ cells was not increased and the total nucleated cells were not significantly decreased during culture; However, the output of CFU-C and CFU-GEMM was enhanced, suggesting that serum-free conditions prevent primitive-cell differentiation.
Previous findings showed that UCB LTC-IC were present among the CD34+ cell fraction[34,35]. Furthermore, Bhatia et al. identified the SRC that were capable of multilineage reconstitution of human hematopoiesis in the bone marrow of NOD/SCID mice[29]. Therefore, the expanded hematopoietic progenitors were expected to sustain long-term hematopoiesis. As a result, 10.23-fold LTC-IC amplification was observed in cells expanded by the tfhMSCs coculture system, although LTC-IC frequency was decreased during culture. The SRC assay indicated the reconstituting ability of these cultured human PPC. Although we could not perform a quantitative SRC assay, the difference in the percentage of chimerism of human CD45+ cells between bone marrow cells of mice transplanted with cultured cells and those transplanted with control samples strongly suggested the extensive ability of these ex vivo-generated PPC to sustain and reconstitute long-term human hematopoiesis in vivo. Furthermore, PCR analysis confirmed flow cytometric results.
In conclusion, the main obstacle to UCB trans-plantation in adult recipients is the insufficiency of hematopoietic progenitors. A novel tfhMSCs coculture system that could efficiently expand UCB hematopoietic progenitors is successfully established. The extent of LTC-IC expansion shown herein has important practical implications in terms of clinical hematopoietic stem cell transplantation.
ACKNOWLEDGMENTS
The authors thank Drs. En-Ming Zhang, Ke Zheng, Guo-Li Yin for technical assistance and Dr. Jun Liu for critical reading of the manuscript.
Footnotes
Supported by the grants of NIH-Heart, Lung & Blood, No. IR014L70593-01 and Zhejiang Scientific Foundation, No. 2003C23015
S- Editor Wang XL and Guo SY L- Editor Elsevier HK E- Editor Kong LH
References
- 1.Osawa M, Hanada K, Hamada H, Nakauchi H. Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell. Science. 1996;273:242–245. doi: 10.1126/science.273.5272.242. [DOI] [PubMed] [Google Scholar]
- 2.Wang JC, Doedens M, Dick JE. Primitive human hematopoietic cells are enriched in cord blood compared with adult bone marrow or mobilized peripheral blood as measured by the quantitative in vivo SCID-repopulating cell assay. Blood. 1997;89:3919–3924. [PubMed] [Google Scholar]
- 3.Broxmeyer HE, Douglas GW, Hangoc G, Cooper S, Bard J, English D, Arny M, Thomas L, Boyse EA. Human umbilical cord blood as a potential source of transplantable hematopoietic stem/progenitor cells. Proc Natl Acad Sci U S A. 1989;86:3828–3832. doi: 10.1073/pnas.86.10.3828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cairo MS, Wagner JE. Placental and/or umbilical cord blood: an alternative source of hematopoietic stem cells for transplantation. Blood. 1997;90:4665–4678. [PubMed] [Google Scholar]
- 5.Kurtzberg J, Laughlin M, Graham ML, Smith C, Olson JF, Halperin EC, Ciocci G, Carrier C, Stevens CE, Rubinstein P. Placental blood as a source of hematopoietic stem cells for transplantation into unrelated recipients. N Engl J Med. 1996;335:157–166. doi: 10.1056/NEJM199607183350303. [DOI] [PubMed] [Google Scholar]
- 6.Petzer AL, Zandstra PW, Piret JM, Eaves CJ. Differential cytokine effects on primitive (CD34+CD38-) human hematopoietic cells: novel responses to Flt3-ligand and thrombopoietin. J Exp Med. 1996;183:2551–2558. doi: 10.1084/jem.183.6.2551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McNiece I, Kubegov D, Kerzic P, Shpall EJ, Gross S. Increased expansion and differentiation of cord blood products using a two-step expansion culture. Exp Hematol. 2000;28:1181–1186. doi: 10.1016/s0301-472x(00)00520-8. [DOI] [PubMed] [Google Scholar]
- 8.Conneally E, Cashman J, Petzer A, Eaves C. Expansion in vitro of transplantable human cord blood stem cells demonstrated using a quantitative assay of their lympho-myeloid repopulating activity in nonobese diabetic-scid/scid mice. Proc Natl Acad Sci U S A. 1997;94:9836–9841. doi: 10.1073/pnas.94.18.9836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ueda T, Tsuji K, Yoshino H, Ebihara Y, Yagasaki H, Hisakawa H, Mitsui T, Manabe A, Tanaka R, Kobayashi K, et al. Expansion of human NOD/SCID-repopulating cells by stem cell factor, Flk2/Flt3 ligand, thrombopoietin, IL-6, and soluble IL-6 receptor. J Clin Invest. 2000;105:1013–1021. doi: 10.1172/JCI8583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moore KA, Ema H, Lemischka IR. In vitro maintenance of highly purified, transplantable hematopoietic stem cells. Blood. 1997;89:4337–4347. [PubMed] [Google Scholar]
- 11.Xu MJ, Tsuji K, Ueda T, Mukouyama YS, Hara T, Yang FC, Ebihara Y, Matsuoka S, Manabe A, Kikuchi A, et al. Stimulation of mouse and human primitive hematopoiesis by murine embryonic aorta-gonad-mesonephros-derived stromal cell lines. Blood. 1998;92:2032–2040. [PubMed] [Google Scholar]
- 12.Kanai M, Hirayama F, Yamaguchi M, Ohkawara J, Sato N, Fukazawa K, Yamashita K, Kuwabara M, Ikeda H, Ikebuchi K. Stromal cell-dependent ex vivo expansion of human cord blood progenitors and augmentation of transplantable stem cell activity. Bone Marrow Transplant. 2000;26:837–844. doi: 10.1038/sj.bmt.1702634. [DOI] [PubMed] [Google Scholar]
- 13.Kusadasi N, Koevoet JL, van Soest PL, Ploemacher RE. Stromal support augments extended long-term ex vivo expansion of hemopoietic progenitor cells. Leukemia. 2001;15:1347–1358. doi: 10.1038/sj.leu.2402204. [DOI] [PubMed] [Google Scholar]
- 14.Kawada H, Ando K, Tsuji T, Shimakura Y, Nakamura Y, Chargui J, Hagihara M, Itagaki H, Shimizu T, Inokuchi S, et al. Rapid ex vivo expansion of human umbilical cord hematopoietic progenitors using a novel culture system. Exp Hematol. 1999;27:904–915. doi: 10.1016/s0301-472x(99)00012-0. [DOI] [PubMed] [Google Scholar]
- 15.Shimakura Y, Kawada H, Ando K, Sato T, Nakamura Y, Tsuji T, Kato S, Hotta T. Murine stromal cell line HESS-5 maintains reconstituting ability of Ex vivo-generated hematopoietic stem cells from human bone marrow and cytokine-mobilized peripheral blood. Stem Cells. 2000;18:183–189. doi: 10.1634/stemcells.18-3-183. [DOI] [PubMed] [Google Scholar]
- 16.Breems DA, Blokland EA, Siebel KE, Mayen AE, Engels LJ, Ploemacher RE. Stroma-contact prevents loss of hematopoietic stem cell quality during ex vivo expansion of CD34+ mobilized peripheral blood stem cells. Blood. 1998;91:111–117. [PubMed] [Google Scholar]
- 17.Bennaceur-Griscelli A, Tourino C, Izac B, Vainchenker W, Coulombel L. Murine stromal cells counteract the loss of long-term culture-initiating cell potential induced by cytokines in CD34(+)CD38(low/neg) human bone marrow cells. Blood. 1999;94:529–538. [PubMed] [Google Scholar]
- 18.Gan OI, Murdoch B, Larochelle A, Dick JE. Differential maintenance of primitive human SCID-repopulating cells, clonogenic progenitors, and long-term culture-initiating cells after incubation on human bone marrow stromal cells. Blood. 1997;90:641–650. [PubMed] [Google Scholar]
- 19.Yamaguchi M, Hirayama F, Kanai M, Sato N, Fukazawa K, Yamashita K, Sawada K, Koike T, Kuwabara M, Ikeda H, et al. Serum-free coculture system for ex vivo expansion of human cord blood primitive progenitors and SCID mouse-reconstituting cells using human bone marrow primary stromal cells. Exp Hematol. 2001;29:174–182. doi: 10.1016/s0301-472x(00)00653-6. [DOI] [PubMed] [Google Scholar]
- 20.Harley CB, Futcher AB, Greider CW. Telomeres shorten during ageing of human fibroblasts. Nature. 1990;345:458–460. doi: 10.1038/345458a0. [DOI] [PubMed] [Google Scholar]
- 21.Koli K, Keski-Oja J. Cellular senescence. Ann Med. 1992;24:313–318. doi: 10.3109/07853899209147829. [DOI] [PubMed] [Google Scholar]
- 22.Roecklein BA, Torok-Storb B. Functionally distinct human marrow stromal cell lines immortalized by transduction with the human papilloma virus E6/E7 genes. Blood. 1995;85:997–1005. [PubMed] [Google Scholar]
- 23.Loeuillet C, Bernard G, Rémy-Martin J, Saas P, Hervé P, Douay L, Chalmers D. Distinct hematopoietic support by two human stromal cell lines. Exp Hematol. 2001;29:736–745. doi: 10.1016/s0301-472x(01)00644-0. [DOI] [PubMed] [Google Scholar]
- 24.Garrido SM, Appelbaum FR, Willman CL, Banker DE. Acute myeloid leukemia cells are protected from spontaneous and drug-induced apoptosis by direct contact with a human bone marrow stromal cell line (HS-5) Exp Hematol. 2001;29:448–457. doi: 10.1016/s0301-472x(01)00612-9. [DOI] [PubMed] [Google Scholar]
- 25.Novotny JR, Duehrsen U, Welch K, Layton JE, Cebon JS, Boyd AW. Cloned stromal cell lines derived from human Whitlock/Witte-type long-term bone marrow cultures. Exp Hematol. 1990;18:775–784. [PubMed] [Google Scholar]
- 26.Zhang Y, Li C, Jiang X, Zhang S, Wu Y, Liu B, Tang P, Mao N. Human placenta-derived mesenchymal progenitor cells support culture expansion of long-term culture-initiating cells from cord blood CD34+ cells. Exp Hematol. 2004;32:657–664. doi: 10.1016/j.exphem.2004.04.001. [DOI] [PubMed] [Google Scholar]
- 27.Wang JF, Wang LJ, Wu YF, Xiang Y, Xie CG, Jia BB, Harrington J, McNiece IK. Mesenchymal stem/progenitor cells in human umbilical cord blood as support for ex vivo expansion of CD34(+) hematopoietic stem cells and for chondrogenic differentiation. Haematologica. 2004;89:837–844. [PubMed] [Google Scholar]
- 28.Sutherland HJ, Lansdorp PM, Henkelman DH, Eaves AC, Eaves CJ. Functional characterization of individual human hematopoietic stem cells cultured at limiting dilution on supportive marrow stromal layers. Proc Natl Acad Sci U S A. 1990;87:3584–3588. doi: 10.1073/pnas.87.9.3584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bhatia M, Wang JC, Kapp U, Bonnet D, Dick JE. Purification of primitive human hematopoietic cells capable of repopulating immune-deficient mice. Proc Natl Acad Sci U S A. 1997;94:5320–5325. doi: 10.1073/pnas.94.10.5320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Haynesworth SE, Baber MA, Caplan AI. Cytokine expression by human marrow-derived mesenchymal progenitor cells in vitro: effects of dexamethasone and IL-1 alpha. J Cell Physiol. 1996;166:585–592. doi: 10.1002/(SICI)1097-4652(199603)166:3<585::AID-JCP13>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 31.Majka M, Janowska-Wieczorek A, Ratajczak J, Ehrenman K, Pietrzkowski Z, Kowalska MA, Gewirtz AM, Emerson SG, Ratajczak MZ. Numerous growth factors, cytokines, and chemokines are secreted by human CD34(+) cells, myeloblasts, erythroblasts, and megakaryoblasts and regulate normal hematopoiesis in an autocrine/paracrine manner. Blood. 2001;97:3075–3085. doi: 10.1182/blood.v97.10.3075. [DOI] [PubMed] [Google Scholar]
- 32.Piacibello W, Sanavio F, Garetto L, Severino A, Bergandi D, Ferrario J, Fagioli F, Berger M, Aglietta M. Extensive amplification and self-renewal of human primitive hematopoietic stem cells from cord blood. Blood. 1997;89:2644–2653. [PubMed] [Google Scholar]
- 33.Kawano Y, Kobune M, Yamaguchi M, Nakamura K, Ito Y, Sasaki K, Takahashi S, Nakamura T, Chiba H, Sato T, et al. Ex vivo expansion of human umbilical cord hematopoietic progenitor cells using a coculture system with human telomerase catalytic subunit (hTERT)-transfected human stromal cells. Blood. 2003;101:532–540. doi: 10.1182/blood-2002-04-1268. [DOI] [PubMed] [Google Scholar]
- 34.Briddell RA, Broudy VC, Bruno E, Brandt JE, Srour EF, Hoffman R. Further phenotypic characterization and isolation of human hematopoietic progenitor cells using a monoclonal antibody to the c-kit receptor. Blood. 1992;79:3159–3167. [PubMed] [Google Scholar]
- 35.Traycoff CM, Abboud MR, Laver J, Brandt JE, Hoffman R, Law P, Ishizawa L, Srour EF. Evaluation of the in vitro behavior of phenotypically defined populations of umbilical cord blood hematopoietic progenitor cells. Exp Hematol. 1994;22:215–222. [PubMed] [Google Scholar]


