Abstract
In this study we have reprogrammed dermal fibroblasts from an adult female horse into equine induced pluripotent stem cells (equiPSCs). These equiPSCs are dependent only on leukemia inhibitory factor (LIF), placing them in striking contrast to previously derived equiPSCs that have been shown to be co-dependent on both LIF and basic fibroblast growth factor (bFGF). These equiPSCs have a normal karyotype and have been maintained beyond 60 passages. They possess alkaline phosphatase activity and express eqNANOG, eqOCT4, and eqTERT mRNA. Immunocytochemistry confirmed that they produce NANOG, REX1, SSEA4, TRA1-60, and TRA1-81. While our equiPSCs are LIF dependent, bFGF co-stimulates their proliferation via the PI3K/AKT pathway. EquiPSCs lack expression of eqXIST and immunostaining for H3K27me3, suggesting that during reprogramming the inactive X chromosome has likely been reactivated to generate cells that have two active X chromosomes. EquiPSCs form embryoid bodies and in vitro teratomas that contain derivatives of all three germ layers. These LIF-dependent equiPSCs likely reflect a more naive state of pluripotency than equiPSCs that are co-dependent on both LIF and bFGF and so provide a novel resource for understanding pluripotency in the horse.
Introduction
The horse occupies a unique niche in most societies given its varying roles as working animal, companion, and competitor; this diversity in utility is reflected in the horse industry directly contributing around US$300 billion worldwide [1]. A study of the New Zealand thoroughbred racing industry identified a major welfare and economic concern in that around 5% of racing thoroughbreds will suffer musculoskeletal injuries during their careers, with only 25%–50% of these animals recovering sufficiently to race again [2]. Significantly, musculoskeletal injuries accounted for 71% of deaths in North American Quarter Horse racehorses during an 18-year study period [3]. Extensive research efforts have been directed toward the use of mesenchymal stem cells (MSCs) for the treatment of musculoskeletal injuries in horses because of the potential for MSC-based regenerative therapies to improve treatment outcomes, and thus return equine athletes to competitive soundness. However, MSC-based therapies are limited by the restricted proliferative ability of MSCs both in vitro and in vivo, and their scarcity, with MSCs representing only around 0.01%–0.1% of total cells harvested from the patient's adipose tissue or bone marrow [4,5]. Consequently, the development of induced pluripotent stem cells (iPSCs) [6] has significantly advanced the prospects of stem cell-based therapies in horses, allowing for the reprogramming of an adult individual's cells, such as fibroblasts, into pluripotent stem cells, thus providing a source of stem cells for regenerative therapies that are autologous and which can be propagated indefinitely in vitro.
In this report we describe the generation of equine iPSCs (equiPSCs) from adult female dermal fibroblasts, which are dependent only on the cytokine leukemia inhibitory factor (LIF), express key pluripotency markers, and can differentiate into derivatives of all three germ layers. While three other research groups have previously reported on the production of equiPSCs, these differ significantly from ours in their co-dependency on both basic fibroblast growth factor (bFGF) and LIF for the maintenance of pluripotency [7–9]. Thus, our LIF-dependent equiPSCs appear to reflect a more naive state of pluripotency akin to that described for equine embryonic stem cells (eESCs) [10,11].
Materials and Methods
All use of animals, and tissues obtained from animals, was approved by the University of Queensland Animal Ethics Committee.
Generation and maintenance of equiPSCs
Approximately 3.0×106 fibroblasts from an adult female horse were transduced with lentiviruses expressing human OCT4, SOX2, KLF4, and c-MYC (Addgene plasmids 21162 and 21164) as previously described [12]. Forty-eight hours after transduction, fibroblasts were passaged onto six 10-cm culture plates (Costar) coated with Matrigel (BD Biosciences) at a density of 5.0×105 cells per plate and maintained in medium consisting of Knockout Dulbecco's modified Eagle's medium (DMEM) (Gibco), 15% (v/v) ESC-qualified fetal calf serum (ES-FCS; Gibco), 0.1 mM nonessential amino acids (NEAAs; Gibco), 2 mM l-glutamine (Gibco), and 0.1 mM β-mercaptoethanol (Gibco) supplemented with 1,000 U/mL murine LIF (ESGRO; Millipore), 10 ng/mL human bFGF (Invitrogen), 3 μM GSK3β inhibitor (CHIR99021; Stemgent), 0.5 μM MEK inhibitor (PD0325901; Stemgent), 0.25 μM TGF-β antagonist (A83-01; Stemgent), and 2.5 μM ALK receptor inhibitor (SB431542; Stemgent). Cultures were maintained at 37°C with 5% CO2.
Putative equiPSCs were subcultured onto feeder layers of irradiated mouse embryonic fibroblasts (MEFs) once the colonies were large enough to be mechanically isolated. After subculture onto MEFs, equiPSCs were maintained in Knockout DMEM, 15% (v/v) ES-FCS, 0.1 mM NEAAs, 2 mM l-glutamine, and 0.1 mM β-mercaptoethanol with one of five types of supplementation: (1) 1,000 U/mL murine LIF; (2) 1,000 U/mL murine LIF and 10 ng/mL human bFGF; (3) 1,000 U/mL murine LIF, 10 ng/mL human bFGF, and 0.5 μM of the MEK inhibitor PD0325901; (4) 1,000 U/mL murine LIF, 10 ng/mL human bFGF, and 50 μM of the PI3K/AKT inhibitor LY294002 (Cell Signaling Technology); (5) 1,000 U/mL murine LIF, 10 ng/mL human bFGF, 0.5 μM of the MEK inhibitor PD0325901, and 50 μM of the PI3K/AKT inhibitor LY294002.
Karyotyping
EquiPSCs were commercially karyotyped by Sullivan Nicolaides Pathology (Taringa). Fifteen G-banded metaphase spreads were examined for each equiPSC line.
Alkaline phosphatase staining and fluorescence immunocytochemistry
Alkaline phosphatase staining was performed using the Quantitative Alkaline Phosphatase Detection Kit (Millipore) as per the manufacturer's instructions.
Fluorescence immunocytochemistry was performed for the following markers of pluripotency: NANOG, REX1, SSEA4, TRA1-60 and TRA1-81. Immunostaining for the nuclear proteins NANOG and REX1 involved fixation and permeabilization with ice-cold acetone at −20°C for 10 min. For SSEA4, TRA1-60 and TRA1-81, samples were fixed with 4% paraformaldehyde at room temperature for 10 min. All samples were blocked with 5% goat serum (Gibco) in phosphate-buffered saline (PBS; Gibco) and incubated in primary antibody diluted in 3% goat serum in PBS. Primary antibodies, and their dilutions, are as follows: anti-SSEA4 (MAB4304; Millipore), 1:50; anti-TRA1-60 (MAB4360; Millipore), 1:50; anti-TRA1-81 (MAB4381; Millipore), 1:50; anti-REX1 (ab50828-50; Abcam), 1:50; and anti-NANOG (ab80892; Abcam), 1:150. Negative controls were incubated with 3% goat serum in PBS in place of primary antibody. All secondary antibodies were diluted to 1:1,000 in PBS: Alexa Fluor goat anti-mouse IgGH&L (Invitrogen), Alexa Fluor goat anti-mouse IgMμ (Invitrogen), and Alexa Fluor goat anti-rabbit IgGH&L (Invitrogen).
X chromosome inactivation (XCI) was identified by fluorescence immunocytochemistry using an antibody to the trimethylated histone H3K27 (anti-H3K27me3, ab6002; Abcam) which is an XIST-dependent chromatin mark that directly identifies the inactive X chromosome (Xi). Samples were fixed and permeabilized with ice-cold acetone at −20°C for 10 min before blocking with 5% goat serum in PBS and incubation with the primary antibody at a dilution of 1:50. The secondary antibody was applied as described earlier.
The rate of cell proliferation was determined by fluorescence immunocytochemistry using an antibody to phospho-histone H3 (Ser10) [anti-phospho-histone H3 (Ser10), 9708; Cell Signaling Technology] which identifies the phosphorylation of H3 in cells undergoing mitosis. Mitotic figures were counted in 40 high power fields (HPFs;×100 magnification) per sample, with the samples de-identified to the experimenter, and the data represented as the mean number of mitotic figures per HPF±standard error of the mean. Statistical significance was determined by a Student's t-test.
RNA isolation, cDNA synthesis, and polymerase chain reaction
Total RNA was isolated using the Qiagen RNeasy Mini kit (Qiagen) as per the manufacturer's protocol. Complementary DNA was synthesized using the Bio-Rad iScript Reverse Transcriptase kit (Bio-Rad Laboratories) according to the manufacturer's instructions. The polymerase chain reaction (PCR) primers used, and their product size, are listed in Supplementary Table S1 (Supplementary Data are available online at www.liebertpub.com/scd). Negative control reactions incorporated template from cDNA reactions that were performed without reverse transcriptase or using water in place of template in PCRs.
Generation of embryoid bodies and in vitro teratomas
To generate embryoid bodies (EBs), colonies were passaged with trypsin (TrypLE; Gibco) to produce small aggregates of cells that were then cultured in Costar Ultra-low Attachment Plates (Corning Life Sciences) in Knockout DMEM supplemented with 15% FCS (not ESC grade; JRH Biosciences), 0.1 mM NEAAs and 2 mM l-glutamine. After 6 weeks they were harvested for total RNA extraction and their ability to form derivatives of the three germ layers was assessed by reverse-transcription PCR. The PCR primers used, and their product size, are listed in Supplementary Table S1.
Because of the increasing pressure to utilize alternative methods to live-animal experimentation, we have used a modification of the agarose three-dimensional sustained EB/in vitro teratoma assay [13] to assess the ability of the equiPSCs to form derivatives of all three germ layers. While Stenberg et al. [13] demonstrated spontaneous multilineage differentiation in human ESCs (hESCs), our modified protocol has been verified in both hESCs and hiPSCs (Fortuna, unpublished data). Further, our modified protocol supports the in vitro differentiation of hESCs and hiPSCs to a degree of complexity comparable to that seen with in vivo teratomas (Supplementary Fig. S2; Fortuna, unpublished data). Similar results in hESCs have recently been reported by Stachelscheid and colleagues [14] using a three-dimensional perfusion culture system.
EquiPSC colonies were passaged with trypsin (TrypLE) to produce small aggregates of cells. Approximately 4×105 cells were sandwiched between two layers of 10% (w/v) low molecular weight methylcellulose (Sigma-Aldrich) in Knockout DMEM. Cells were maintained in Knockout DMEM supplemented with 20% FCS (not ESC grade), 0.1 mM NEAAs, 2 mM l-glutamine, and 25 mM HEPES (Gibco) for 8 weeks before embedding and sectioning for histological analysis. Because this methodology was developed using human pluripotent stem cells, hiPSCs were used as a positive control.
Results
Generation of equiPSCs
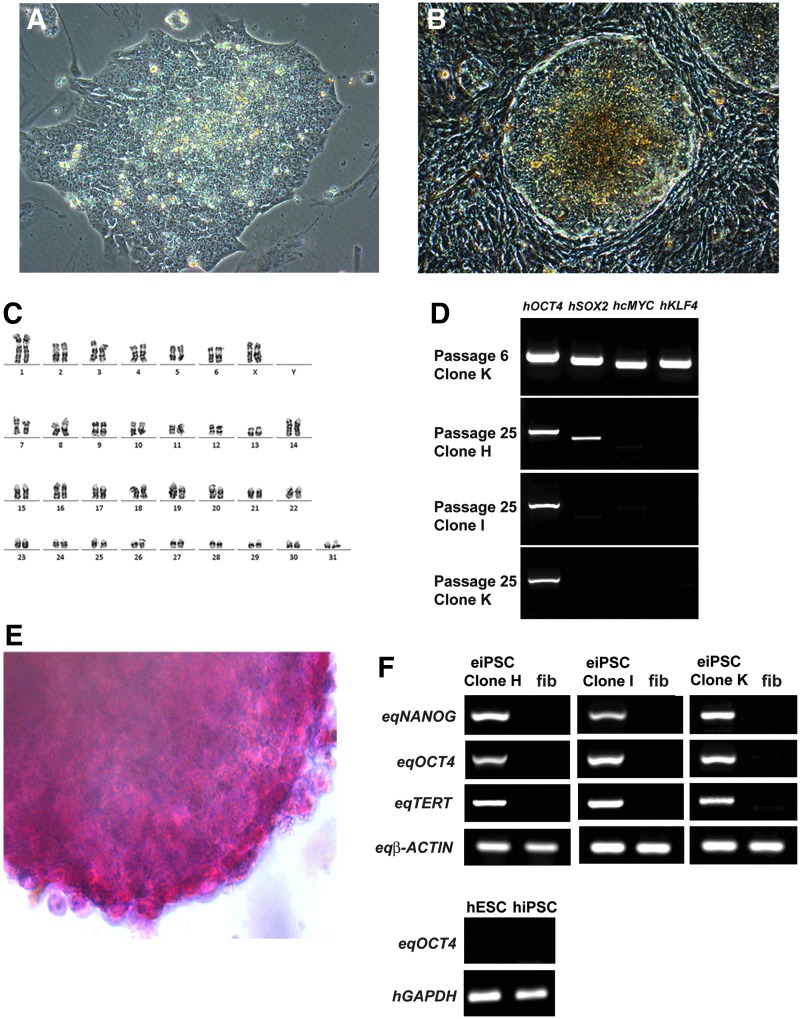
Adult female horse fibroblasts were reprogrammed into iPSCs using lentiviral delivery of human OCT4, SOX2, KLF4, and c-MYC. Fourteen clones of equiPSCs were picked and expanded, seven of which were characterized further. EquiPSCs have a characteristic pluripotent stem cell morphology with a high nuclear-to-cytoplasmic ratio (Fig. 1A). They form compact, domed colonies (Fig. 1B) and are karyotypically normal (64,XX) (Fig. 1C).
FIG. 1.
EquiPSCs exhibit a characteristic pluripotent stem cell morphology and express pluripotency factors. (A) At passage 1, equiPSCs have a characteristic pluripotent stem cell morphology with a high nuclear-to-cytoplasmic ratio. (B) Colonies have a compact, domed appearance. (C) EquiPSCs have a normal 64,XX karyotype. (D) At passage 6, all seven of the selected clones showed robust expression of the four transgenes as demonstrated in this figure with clone K. At passage 25, clones H, I, and K retained expression of the hOCT4 transgene, while Clone H also continued to express hSOX2. (E) EquiPSCs are strongly positive for alkaline phosphatase activity. (F) RT-PCR expression analysis of equine pluripotency factors NANOG, OCT4, and TERT, and positive control β-ACTIN. EquiPSC clones H, I, and K express all three pluripotency factors while equine fibroblasts (fib) do not. hESC and hiPSC (hOCT4 transgene not silenced) samples verified the specificity of the eqOCT4 primers. eiPSC, equine induced pluripotent stem cells; hESC, human embryonic stem cell; hiPSC, human induced pluripotent stem cell; RT-PCR, reverse-transcription polymerase chain reaction.
Transgene expression was examined in the seven selected clones. At passage 6, all clones had robust expression of all four transgenes (hOCT4, hSOX2, hcMYC, and hKLF4) (Fig. 1D). By passage 11, all clones showed silencing of some transgenes but all had continued expression of hOCT4 (data not shown). Based on these results, three clones were selected for further analyses. At passage 25, persistent hOCT4 expression was detected in all three clones (Fig. 1D). In two of these clones the other transgenes were silent, whereas in the third clone expression of the hSOX2 transgene was also still detected (Fig. 1D).
Expression of pluripotency factors by equiPSCs
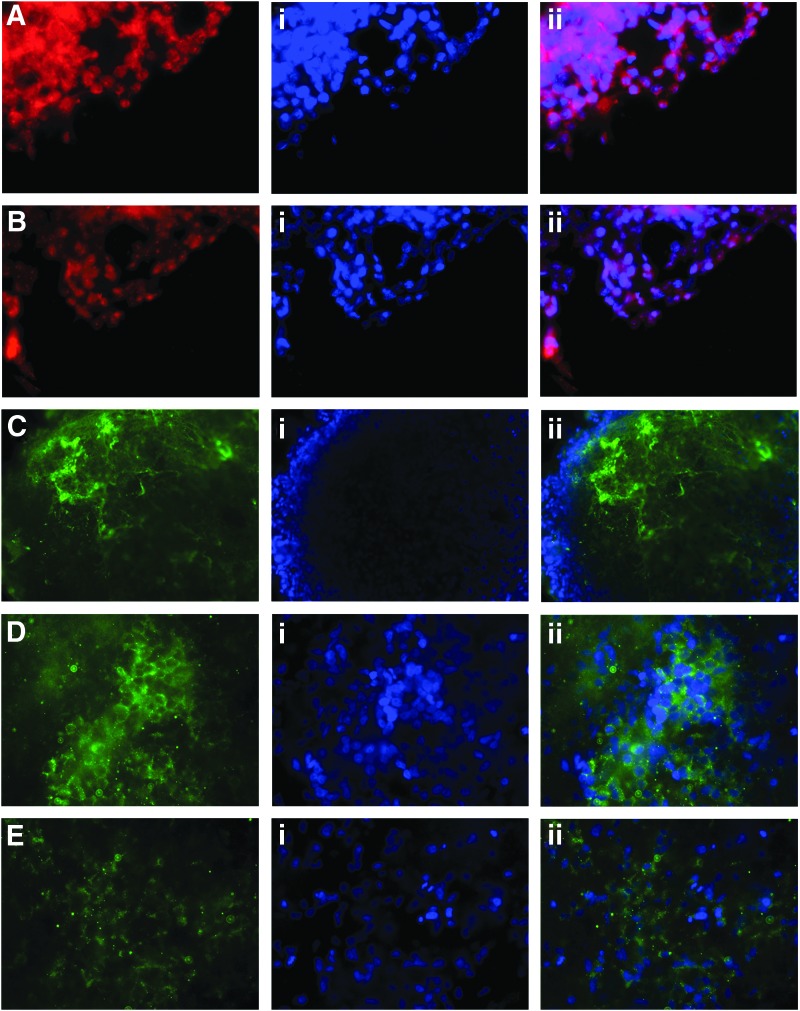
All three of the selected clones are strongly positive for alkaline phosphatase activity (Fig. 1E) and express equine NANOG, OCT4 and TERT mRNA (Fig. 1F). All primers were designed to be specific to the equine mRNA transcripts and so, in the case of OCT4, they do not amplify the human OCT4 transgene (Fig. 1F). Immunocytochemistry confirmed that they also produce the pluripotency markers NANOG, REX1, SSEA4, TRA1-60 and TRA1-81 (Fig. 2A–E). Negative controls that were incubated with 3% goat serum in PBS in place of primary antibody showed no nonspecific staining of the secondary antibody (Supplementary Fig. S1A–E).
FIG. 2.
EquiPSCs express pluripotency factors. (A) Immunocytochemistry for NANOG; staining with DAPI is shown in (i) and the merged image in (ii). (B) REX1. (C) SSEA4. (D) TRA1-60. (E) TRA1-81.
Trilineage differentiation in EBs and in vitro teratomas
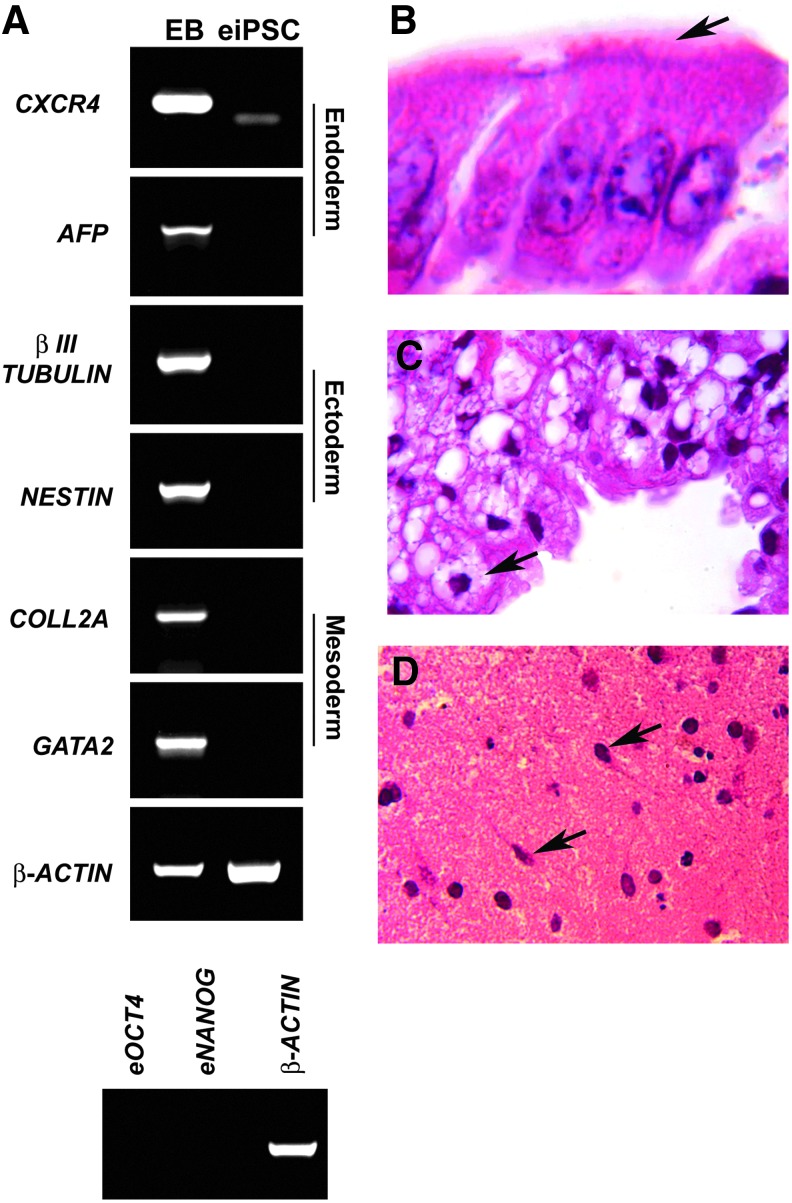
In vitro, equiPSCs readily form EBs that express the endoderm markers CXCR4 and AFP, ectoderm markers βIII-TUBULIN and NESTIN, and the mesoderm markers COLL2A and GATA2 (Fig. 3A). EBs do not express the pluripotency markers OCT4 and NANOG (Fig. 3A). EquiPSCs form teratomas in vitro that contain derivatives of the three germ layers, including ciliated columnar epithelium from endoderm (Fig. 3B), adipose tissue from mesoderm (Fig. 3C), and neural tissue from ectoderm (Fig. 3D). hiPSCs, which were used as a positive control, also formed glandular epithelium (endoderm), cartilage (mesoderm), and neural tissue (ectoderm) (Supplementary Fig. S2A–C).
FIG. 3.
EquiPSCs can differentiate into derivatives of all three germ layers. (A) Expression of differentiation markers by equiPSCs that have undergone spontaneous differentiation as EBs. RT-PCR expression analysis of definitive endoderm markers CXCR4 and AFP, ectoderm markers βIII-TUBULIN and NESTIN, and mesoderm markers COLL2A and GATA2, with β-ACTIN as a positive control. While EBs express differentiation markers from all three embryonic germ layers, the undifferentiated equiPSCs (eiPSC) do not. EBs do not express the pluripotency markers OCT4 and NANOG. (B) EquiPSCs form teratomas in vitro that contain tissue derivatives of the three germ layers including ciliated columnar epithelium from the endoderm (cilia are indicated by the arrow). (C) Adipose tissue consisting of multiloculated adipocytes (arrow) from the mesoderm. (D) Neural tissue that contains neurons (arrows) from the ectoderm. AFP, α-fetoprotein; EBs, embryoid bodies.
bFGF stimulates the proliferation of equiPSCs
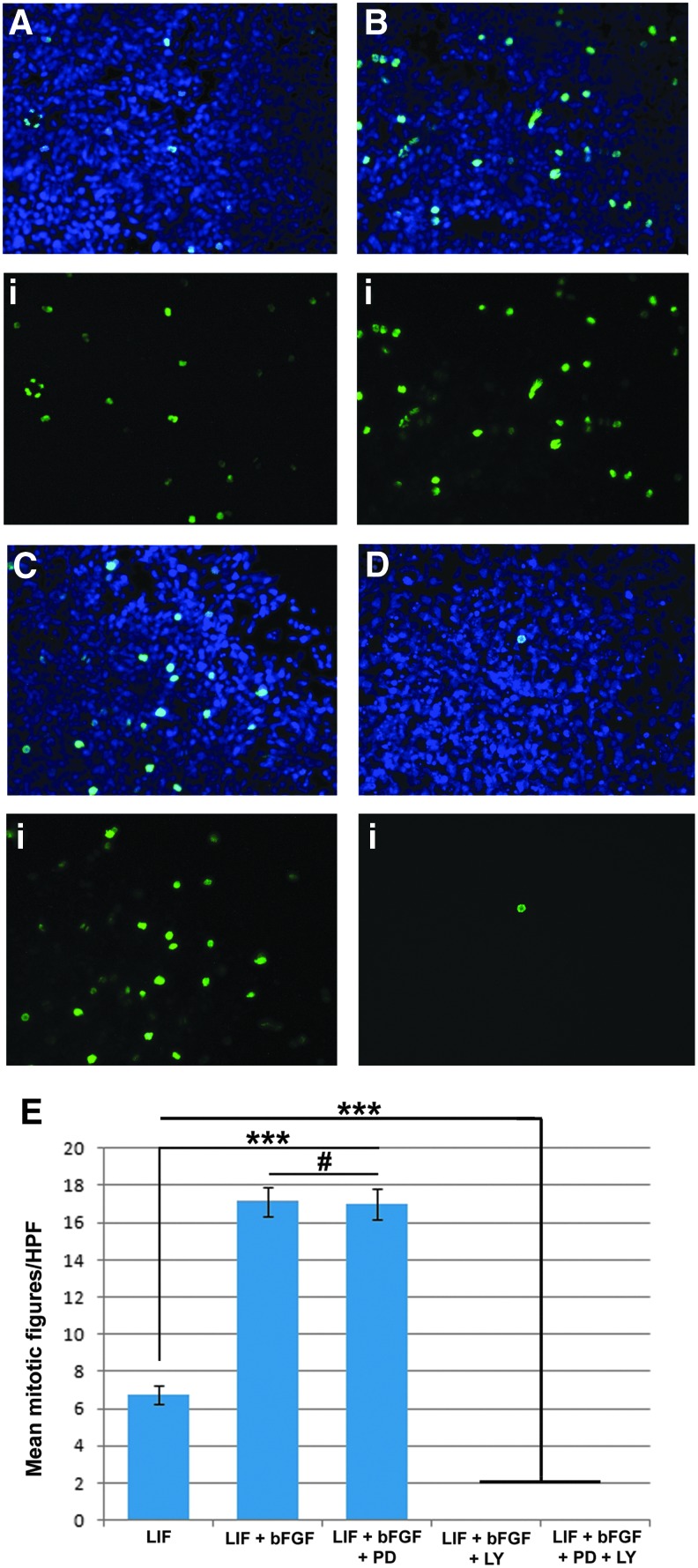
To determine the requirement of both LIF and bFGF for the maintenance of the equiPSCs, we cultured the cells on MEFs in medium containing either LIF or bFGF or both LIF and bFGF. Cultures with bFGF alone, in the absence of LIF, spontaneously differentiated (Supplementary Fig. S3), indicating a dependency on LIF for maintaining pluripotency. This LIF dependency was corroborated by robust expression of the LIFR (Supplementary Fig. S3). While cells cultured with LIF alone maintained their expression of pluripotency markers (data not shown), they did not proliferate as rapidly as cells cultured with both LIF and bFGF. To quantify this effect we performed immunocytochemistry using an antibody to phospho-histone H3 (Ser10) that identifies cells undergoing mitosis. EquiPSCs cultured with both LIF and bFGF had a significantly higher number of mitotic figures per HPF (17.14±0.81) than those cultured with LIF alone (6.74±0.53, P<0.0001) (Fig. 4A, B, E), indicating a role for bFGF in the proliferation of equiPSCs. However, this effect of bFGF signaling must be via an alternative signaling pathway to ERK since proliferation of the equiPSCs cultured with LIF and bFGF was not affected by the presence of the MEK inhibitor PD0325901 (0.5 μM; 17.03±0.83) (Fig. 4C, E).
FIG. 4.
bFGF signaling enhances the proliferation of equiPSCs. (A) EquiPSCs cultured with LIF alone have fewer mitotic figures (green immunofluorescence) than those cultured with both LIF and bFGF. The merged DAPI and immunofluorescence image is shown first with the immunofluorescence image alone in (i). (B) EquiPSCs maintained in medium with LIF and bFGF have significantly more mitotic figures than those cultured with LIF alone. (C) This bFGF-dependent proliferative effect is independent of the MEK signaling pathway since addition of the MEK inhibitor PD0325901 had no effect on the number of mitotic figures. (D) Addition of the PI3K/AKT inhibitor LY294002 abolished nearly all cell division. (E) Graphic representation of the mean number of mitotic figures per HPF±standard error of the mean for equiPSCs maintained in medium with LIF; LIF and bFGF; LIF, bFGF, and the MEK inhibitor PD0325901 (PD); LIF, bFGF, and the PI3K/AKT inhibitor LY294002 (LY); LIF, bFGF, PD, and LY. EquiPSCs cultured with bFGF have a greater number of mitotic figures per HPF than those cultured with LIF alone. This proliferative effect is independent of MEK/ERK signaling but is dependent on PI3K/AKT signaling; ***P<0.0001; #, not significant. LIF, leukemia inhibitory factor. bFGF, basic fibroblast growth factor; HPF, high-power field.
FGF can also signal via the PI3K/AKT pathway and so we reasoned that the proliferative effects of bFGF on the equiPSCs are likely the result of signaling through this pathway. To test this hypothesis we cultured the equiPSCs with the PI3K/AKT inhibitor LY294002 (50 μM). Cells maintained in LIF, bFGF and LY294002 or LIF, bFGF, PD0325901 and LY294002 failed to proliferate with only one mitotic figure detected out of a combined total of 80 HPF under both culture conditions (Fig. 4D, E). Cell viability was 93% in both LY294002-treated cultures and 95% in LIF/bFGF control cultures, indicating that there was no significant loss of cell viability after 3 days of culture in the presence of the PI3K/AKT inhibitor.
EquiPSCs lack immunostaining for H3K27me3 and XIST expression
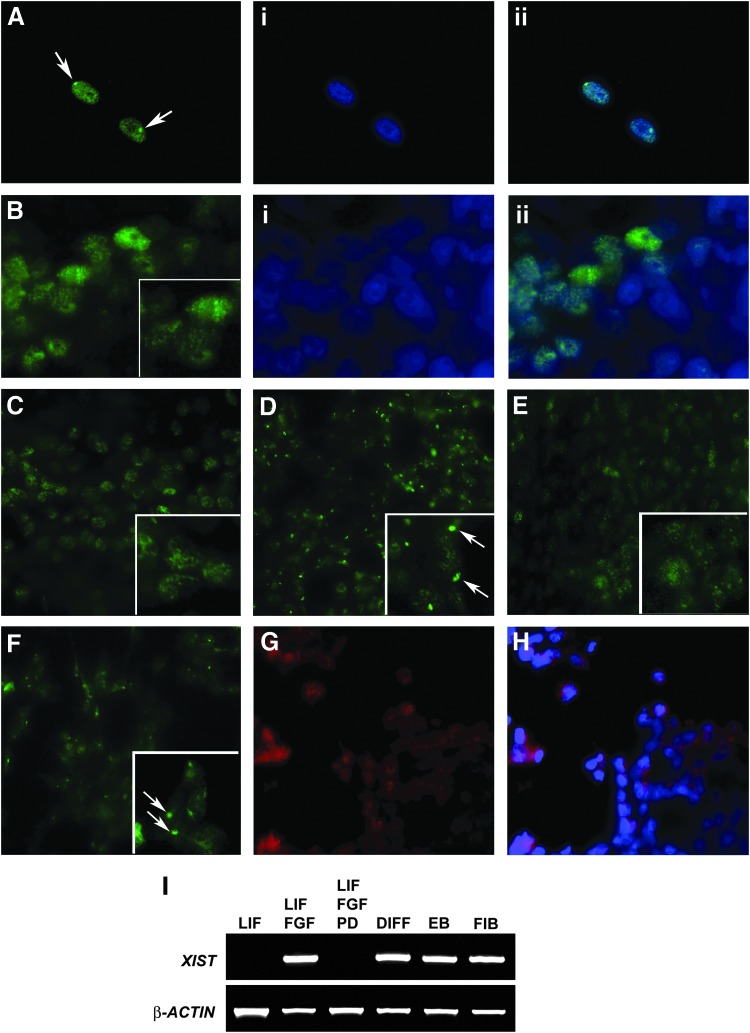
A hallmark of full reprogramming to pluripotency is reactivation of the Xi. Trimethylation of histone H3K27 is an XIST-dependent chromatin mark that directly identifies the Xi [15]. As expected, fluorescence immunocytochemistry for H3K27me3 readily identifies the Xi in the nucleus of the female dermal fibroblasts used for reprogramming (Fig. 5A). In contrast, equiPSCs lack immunostaining for H3K27me3 (Fig. 5B), suggesting that during reprogramming the Xi has likely been reactivated to generate cells that are XaXa.
FIG. 5.
EquiPSCs undergo X chromosome reactivation during reprogramming but bFGF signaling reinstates XCI. (A) Fluorescence immunocytochemistry for H3K27me3 identifies the inactive X chromosome (Xi) (arrows) in the nuclei of the female dermal fibroblasts used for reprogramming; staining with DAPI is shown in (i) and the merged image in (ii). (B) While diffuse immunostaining for H3K27me3 is present in the nuclei of the equiPSCs, they are lacking the dense accumulation of H3K27me3 immunostaining associated with the Xi. The enlarged image within the boxed inset demonstrates both the absence of an Xi and the presence of diffuse nuclear staining. (C) EquiPSCs cultured with LIF alone have two active X chromosomes as indicated by the lack of immunostaining associated with the Xi. (D) When equiPSCs are cultured with both LIF and bFGF, XCI is initiated and immunostaining for the Xi is clearly visible as indicated by the arrows. (E) XCI is due to bFGF signaling via the MEK/ERK pathway since it is abolished when cells are cultured with the MEK inhibitor PD0325901 in addition to bFGF and LIF. (F) Cells that are allowed to spontaneously differentiate in the absence of LIF and bFGF undergo XCI; the Xi is indicated by the white arrows. (G, H) XCI in equiPSCs cultured with LIF and bFGF is not due to the cells differentiating since they still express the pluripotency marker NANOG. (I) In agreement with the immunocytochemistry for H3K27me3, cells cultured with LIF alone do not express XIST, indicating that they are XaXa. In contrast, the addition of bFGF to the medium induces XCI as indicated by the expression of XIST; this effect of bFGF signaling can be abolished by the addition of PD0325901 (PD) to the medium. EquiPSCs that have been allowed to differentiate, either in the absence of LIF and bFGF (DIFF), or as EBs, show XIST expression, as do the fibroblasts (FIB) used for reprogramming. XCI, X chromosome inactivation.
During our experiments to determine the requirement for LIF and bFGF in the maintenance of the equiPSCs, we observed what while cells cultured with LIF alone lacked immunostaining for H3K27me3 (Fig. 5B, C), when bFGF was added to cells that had previously been cultured with LIF alone, immunostaining for H3K27me3 was detected (Fig. 5D). To further confirm that the emergence of positive H3K27me3 immunostaining was due to bFGF signaling, we added the MEK inhibitor PD0325901 in addition to bFGF to cells previously cultured only with LIF, and observed that these cells, like those cultured with LIF alone, lacked immunostaining for H3K27me3 (Fig. 5E). Cells that were allowed to spontaneously differentiate in the absence of LIF and bFGF were positive for H3K27me3 immunostaining (Fig. 5F). To explore the possibility that the cells cultured with LIF and bFGF were similarly positive for H3K27me3 immunostaining because they had moved out of pluripotency and begun to differentiate, we immunostained for NANOG and found that the cells were still highly positive (Fig. 5G, H), suggesting that they had not begun to differentiate.
Trimethylation of histone H3K27 is XIST dependent and so we next examined the expression of XIST in equiPSCs cultured under the previous conditions. EquiPSCs cultured with LIF alone did not express XIST (Fig. 5I). EquiPSCs that were allowed to spontaneously differentiate in the absence of LIF and bFGF express XIST, as do EBs and the fibroblasts used for reprogramming (Fig. 5I). Cells cultured with both LIF and bFGF show robust expression of XIST. However, cells cultured with LIF, bFGF, and the MEK inhibitor PD0325901 lack XIST expression (Fig. 5I), in agreement with our findings with the H3K27me3 immunostaining.
Discussion
In this study we have demonstrated that equine adult dermal fibroblasts can be reprogrammed to LIF-dependent pluripotency. Two studies [10,11] have described the isolation of equine ESCs, both of which used medium containing LIF, without bFGF, to maintain the cells. While both studies describe the expression of a number of pluripotency markers, such as alkaline phosphatase, SSEA1, and OCT4, neither study was successful in generating teratomas. To date, three other laboratories have described the generation and characterization of equiPSCs [7–9] and, significantly, all three point to a co-dependency on both LIF and bFGF. While the reports of Nagy et al. [7] and Khodadadi et al. [8] make no specific mention of the effects of bFGF withdrawal, Breton et al. [9] describe their cells as undergoing differentiation when cultured with LIF alone, indicating a dependency on bFGF for maintaining pluripotency. In the mouse, ESCs, which are isolated from the inner cell mass of the blastocyst, are dependent only on LIF and are considered to exist in a naive, or ground, state of pluripotency [16]. In contrast, a further type of murine stem cell, termed bFGF, activin, and bio-derived stem cells (FAB-SCs), which can also be generated from the blastocyst, is responsive to both LIF and bFGF [17]. While FAB-SCs express pluripotency markers, they are incapable of differentiation unless first converted to ESCs with LIF and bone morphogenetic protein 4 (BMP4) [17]. The epiblast of the postimplantation mouse embryo is the source of yet another population of stem cells, termed epiblast stem cells (EpiSCs) [18]. These cells are exclusively FGF dependent, and LIF independent, and hence are considered to exist in a primed pluripotent state [18]. The status of human ESCs and iPSCs has attracted considerable debate, but generally it is considered that human pluripotent stem cells, by virtue of their FGF dependency, exist in a more primed pluripotent state. Thus, it is possible that our equiPSCs, given their LIF dependency, reflect a more naive state of pluripotency than other equiPSCs that are LIF and bFGF co-dependent.
In our equiPSCs, bFGF signaling is associated with XIST expression and H3K27me3 immunostaining, both of which are suggestive of XCI [15]. When bFGF and the MEK inhibitor PD0325901 are both added to the culture medium, neither XIST expression nor H3K27me3 immunostaining is observed, confirming that both are a consequence of signaling via the FGF/ERK cascade. In the mouse, EpiSCs are FGF dependent and display XCI and hence are considered to exist in a primed pluripotent state [18]. Significantly, when EpiSCs are cultured with PD0325901, in addition to LIF and a GSK3β antagonist, they revert to a naive XaXa pluripotency reminiscent of mESCs [19,20]. Thus, in the mouse, there appears to be a causal relationship between FGF/ERK signaling and XCI. Further support of this relationship can be seen in the mouse embryo where FGF/ERK signaling has been shown to be responsible for the segregation of the hypoblast from the inner cell mass; but if this signaling cascade is blocked with PD0325901, the entire inner cell mass acquires pluripotency [21], further demonstrating the role of FGF/ERK signaling in initiating the transition from a naive to a primed state of pluripotency in the mouse. Thus, our equiPSCs appear to be similar to mouse ESCs and iPSCs in their LIF dependence and potentially also in their response to bFGF signaling. Further studies are necessary to conclusively confirm that both X chromosomes are active in our equiPSCs and to determine the mechanism(s) by which bFGF signaling appears to induce expression of XIST and trimethylation of H3K27, both of which are suggestive of XCI.
FGF signaling has a marked proliferative effect on our equiPSCs. While inhibition of FGF-ERK signaling had no effect on the proliferation of the equiPSCs, inhibition of the PI3K/AKT pathway completely abolished cell division. This pathway can be stimulated by both LIF and bFGF [22–26] and is required for maintaining pluripotency in hESCs [22]. In the mouse, Akt-null ESCs have a significantly reduced rate of proliferation compared with wild-type ESCs [27], and mESCs treated with a PI3K/AKT inhibitor showed a marked increase in the number of cells in the G0/G1 phase, supporting a role for PI3K/AKT in promoting mESC proliferation [28]. In the equiPSCs, proliferation is promoted through both LIF and bFGF signaling, although cells maintained in LIF alone proliferate at a significantly slower rate. Both LIF- and bFGF-derived proliferative effects appear to be mediated through the PI3K/AKT pathway since inhibition of this pathway abolished all proliferation; if only bFGF signaling was via the PI3K/AKT pathway, it would be expected that the equiPSCs would show a similar proliferative rate to those cells maintained in LIF. Thus, joint LIF and bFGF signaling through the PI3K/AKT pathway is critical for promoting proliferation in equiPSCs.
Conclusion
In summary, we have generated LIF-dependent equiPSCs that express a range of pluripotency markers and can differentiate into derivatives of all three germ layers. We also show that both LIF and bFGF signal via the PI3K/AKT pathway to promote proliferation in the equiPSCs. Analyses based on the expression of XIST and trimethylation of H3K27 suggest that when maintained in the absence of bFGF signaling (ie, with LIF alone or in medium supplemented with LIF and both bFGF and the MEK inhibitor PD0325901) both X chromosomes appear to be active. Taken together with their LIF-only dependence, these data suggest that our equiPSCs may exist in a more naive state than previously described equiPSCs that are LIF and bFGF co-dependent.
Supplementary Material
Acknowledgments
The authors are grateful to the Stem Cells Australia Stem Core for cell culture support and to Dr. Lisa Kidd for facilitating the collection of the equine dermal tissue. We are also very grateful to Nilay Thakar and Steven Dingwall for maintaining the cells during D.J.W.'s period of absence. This work was supported by grant 2010000357 from the University of Queensland New Staff Research Start-up Fund to D.J.W.
Author Disclosure Statement
The authors each declare that no competing financial interests exist.
References
- 1.Tecirlioglu RT. and Trounson AO. (2007). Embryonic stem cells in companion animals (horses, dogs and cats): present status and future prospects. Reprod Fertil Dev 19:740–747 [DOI] [PubMed] [Google Scholar]
- 2.Perkins NR, Reid SW. and Morris RS. (2005). Risk factors for injury to the superficial digital flexor tendon and suspensory apparatus in Thoroughbred racehorses in New Zealand. N Z Vet J 53:184–192 [DOI] [PubMed] [Google Scholar]
- 3.Sarrafian TL, Case JT, Kinde H, Daft BM, Read DH, Moore JD, Uzal FA. and Stover SM. (2012). Fatal musculo-skeletal injuries of Quarter Horse racehorses: 314 cases (1990–2007). J Am Vet Med Assoc 241:935–942 [DOI] [PubMed] [Google Scholar]
- 4.Vidal MA, Kilroy GE, Johnson JR, Lopez MJ, Moore RM. and Gimble JM. (2006). Cell growth characteristics and differentiation frequency of adherent equine bone marrow-derived mesenchymal stromal cells: adipogenic and osteogenic capacity. Vet Surg 35:601–610 [DOI] [PubMed] [Google Scholar]
- 5.Borjesson DL. and Peroni JF. (2011). The regenerative medicine laboratory: facilitating stem cell therapy for equine disease. Clin Lab Med 31:109–123 [DOI] [PubMed] [Google Scholar]
- 6.Takahashi K. and Yamanaka S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676 [DOI] [PubMed] [Google Scholar]
- 7.Nagy K, Sung HK, Zhang P, Laflamme S, Vincent P, Agha-Mohammadi S, Woltjen K, Monetti C, Michael IP, Smith LC. and Nagy A. (2011). Induced pluripotent stem cell lines derived from equine fibroblasts. Stem Cell Rev 7:693–702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Khodadadi K, Sumer H, Pashaiasl M, Lim S, Williamson M. and Verma PJ. (2012). Induction of pluripotency in adult equine fibroblasts without cMyc. Stem Cells Int [Epub ahead of print]; DOI: 10.1155/2012/429160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Breton A, Sharma R, Diaz AC, Parham AG, Graham A, Neil C, Whitelaw CB, Milne E. and Donadeu FX. (2013). Derivation and characterization of induced pluripotent stem cells from equine fibroblasts. Stem Cells Dev 22:611–621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Saito S, Ugai H, Sawai K, Yamamoto Y, Minamihashi A, Kurosaka K, Kobayashi Y, Murata T, Obata Y. and Yokoyama K. (2002). Isolation of embryonic stem-like cells from equine blastocysts and their differentiation in vitro. FEBS Lett 531:389–396 [DOI] [PubMed] [Google Scholar]
- 11.Li X, Zhou SG, Imreh MP, Ahrlund-Richter L. and Allen WR. (2006). Horse embryonic stem cell lines from the proliferation of inner cell mass cells. Stem Cells Dev 15:523–531 [DOI] [PubMed] [Google Scholar]
- 12.Whitworth DJ, Ovchinnikov DA. and Wolvetang EJ. (2012). Generation and characterization of LIF-dependent canine induced pluripotent stem cells from adult dermal fibroblasts. Stem Cells Dev 21:2288–2297 [DOI] [PubMed] [Google Scholar]
- 13.Stenberg J, Elovsson M, Strehl R, Kilmare E, Hyllner J. and Lindahl A. (2011). Sustained embryoid body formation and culture in a non-laborious three dimensional culture system for human embryonic stem cells. Cytotechnology 63:227–237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stachelscheid H, Wulf-Goldenberg A, Eckert K, Jensen J, Edsbagge J, Björquist P, Rivero M, Strehl R, Jozefczuk J, et al. (2013). Teratoma formation of human embryonic stem cells in three-dimensional perfusion culture bioreactors. J Tissue Eng Regen Med 7:729–741 [DOI] [PubMed] [Google Scholar]
- 15.Payer B, Lee JT. and Namekawa SH. (2011). X-inactivation and X-reactivation: epigenetic hallmarks of mammalian reproduction and pluripotent stem cells. Hum Genet 130:265–280 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pera MF. and Tam PPL. (2010). Extrinsic regulation of pluripotent stem cells. Nature 465:713–720 [DOI] [PubMed] [Google Scholar]
- 17.Chou YF, Chen HH, Eijpe M, Yabuuchi A, Chenoweth JG, Tesar P, Lu J, McKay RD. and Geijsen N. (2008). The growth factor environment defines distinct pluripotent ground states in novel blastocyst-derived stem cells. Cell 135:449–461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tesar PJ, Chenoweth JG, Brook FA, Davies TJ, Evans EP, Mack DL, Gardner RL. and McKay RD. (2007). New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature 448:196–199 [DOI] [PubMed] [Google Scholar]
- 19.Hanna J, Markoulaki S, Mitalipova M, Cheng AW, Cassady JP, Staerk J, Carey BW, Lengner CJ, Foreman R, et al. (2009). Metastable pluripotent states in NOD-mouse-derived ESCs. Cell Stem Cell 4:513–524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Greber B, Wu G, Bernemann C, Joo JY, Han DW, Ko K, Tapia N, Sabour D, Sterneckert J, Tesar P. and Schöler HR. (2010). Conserved and divergent roles of FGF signaling in mouse epiblast stem cells and human embryonic stem cells. Cell Stem Cell 6:215–226 [DOI] [PubMed] [Google Scholar]
- 21.Nichols J, Silva J, Roode M. and Smith A. (2009). Suppression of Erk signalling promotes ground state pluripotency in the mouse embryo. Development 136:3215–3222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Armstrong L, Hughes O, Yung S, Hyslop L, Stewart R, Wappler I, Peters H, Walter T, Stojkovic P, et al. (2006). The role of PI3K/AKT, MAPK/ERK and NFκβ signalling in the maintenance of human embryonic stem cell pluripotency and viability highlighted by transcriptional profiling and functional analysis. Hum Mol Genet 15:1894–1913 [DOI] [PubMed] [Google Scholar]
- 23.Takahashi K, Murakami M. and Yamanaka S. (2005). Role of phosphoinositide 3-kinase pathway in mouse embryonic stem (ES) cells. Biochem Soc Trans 33:1522–1525 [DOI] [PubMed] [Google Scholar]
- 24.Eiselleova L, Matulka K, Kriz V, Kunova M, Schmidtova Z, Neradil J, Tichy B, Dvorakova D, Pospisilova S, Hampl A. and Dvorak P. (2009). A complex role for FGF-2 in self-renewal, survival, and adhesion of human embryonic stem cells. Stem Cells 27:1847–1857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Niwa H, Ogawa K, Shimosato D. and Adachi K. (2009). A parallel circuit of LIF signalling pathways maintains pluripotency of mouse ES cells. Nature 460:118–122 [DOI] [PubMed] [Google Scholar]
- 26.Kinehara M, Kawamura S, Tateyama D, Suga M, Matsumura H, Mimura S, Hirayama N, Hirata M, Uchio-Yamada K, et al. (2013). Protein kinase C regulates human pluripotent stem cell self-renewal. PLOS One 8:1–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stiles B, Gilman V, Khanzenzon N, Lesche R, Li A, Qiao R, Liu X. and Wu H. (2002). Essential role of AKT-1/protein kinase B alpha in PTEN-controlled tumorigenesis. Mol Cell Biol 22:3842–3851 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jirmanova L, Afanassieff M, Gobert-Gosse S, Markossian S. and Savatier P. (2002). Differential contributions of ERK and PI3-kinase to the regulation of cyclin D1 expression and to the control of the G1/S transition in mouse embryonic stem cells. Oncogene 21:5515–5528 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.