Abstract
Background/Aims
Progressive chronic kidney disease (CKD) is associated with worsening cardiovascular risk not explained by traditional risk factors. Left ventricular hypertrophy (LVH) is an important cardiovascular risk factor, but its progression has not been documented in early CKD. We explored whether progression of LVH in early CKD would occur despite stable kidney function.
Methods
We conducted a post hoc analysis of a 12-m nth study of lanthanum carbonate in stage 3 CKD, which included longitudinal assessments of cardiovascular biomarkers. Primary outcome for the analysis was the change in LV mass indexed to height in meters2.7 (LVM/Ht2.7). Secondary outcomes were changes in blood pressure (BP), pulse-wave velocity, LV systolic/diastolic function, fibroblast growth factor-23 (FGF23), klotho, and eGFR.
Results
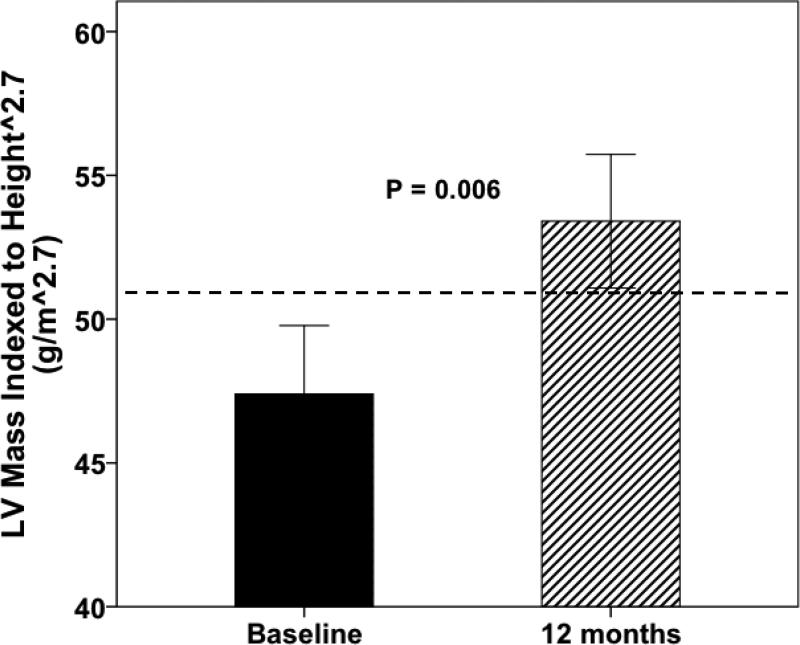
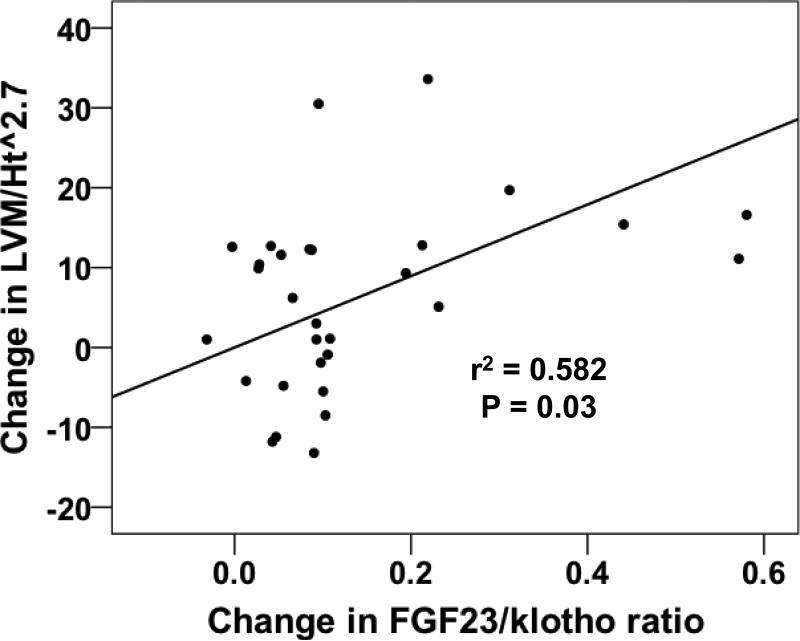
31 of 38 original subjects had sufficient data for analysis. LVM/Ht2.7 increased (47 ± 13 vs. 53 ± 13 g/m2.7, P=0.006) over 12 months despite stable BP, stable eGFR and normal LV systolic function. Vascular stiffness and LV diastolic dysfunction persisted throughout the study. Klotho levels decreased (748 ± 289 to 536 ± 410 pg/ml, P=0.03) but were unrelated to changes in LVM/Ht2.7. The change in FGF23/klotho ratio was strongly correlated with changes in LVM/Ht2.7 (r2 0.582, P=0.03).
Conclusion
Subjects with stage 3 CKD exhibited increasing LV mass, persistent LV diastolic dysfunction and vascular stiffness despite stable kidney function, BP and LV systolic function. Abnormal FGF23 signaling due to reduced klotho expression may be associated with increasing LV mass. These findings deserve further evaluation in a larger population, given the adverse prognostic value of these cardiovascular biomarkers.
Keywords: Cardiovascular, chronic kidney disease, biomarkers, ventricular hypertrophy
INTRODUCTION
Chronic kidney disease (CKD) is associated with increased cardiovascular (CV) risk compared with the general population [1-3]. Traditional CV risk factors such as age, sex, smoking, hypertension, cholesterol, and diabetes do not account for the increased risk in the CKD population [4]. Cardiovascular risk consists of fatal and nonfatal myocardial infarction, congestive heart failure, arrhythmias and sudden death. Biomarkers associated with CV risk include left ventricular mass (LVM), carotid-femoral pulse-wave velocity (PWV), LV systolic and LV diastolic function, and vascular calcification. Circulating biomarkers are also abnormal in CKD, worsen with progressive CKD, and illustrate an inverse correlation between glomerular filtration rate (GFR) and CV risk [3].
Although an estimated 10% of the general population has stage 3 CKD with an estimated GFR (eGFR) between 30-59 ml/min/1.73m2 [5], most of these individuals do not progress to end-stage kidney disease (ESKD). Moreover, patients that do progress to ESKD may do so with variable rate of decline over a period of several months or years. Conceivably, many patients will maintain stable eGFR and remain at the same stage of CKD for years. Despite the apparent stability of kidney function there is ongoing exposure to the pathophysiology of mild to moderate CKD, such as release of substances from the diseased kidney, hypertension, hyperphosphatemia, and vasoactive and hypertrophic signaling from the renin-angiotensin-aldosterone axis. While there are numerous longitudinal studies examining progression of biomarkers of CV risk in stage 5 CKD [6-11], there are few longitudinal studies in patients with stable mild to moderate CKD. Studies in mild to moderate CKD often use progression of CKD as an outcome measure, rather than examine those with stable CKD in a longitudinal fashion to determine changes in biomarkers associated with CV risk [12-16].
Recently, we published a randomized, double-blind, placebo-controlled pilot and feasibility study of the phosphorus binder lanthanum carbonate (LaCO3) in 38 patients with stage 3 CKD [17]. Phosphorus binder therapy did not significantly affect phosphate homeostasis or biomarkers of CV risk after 12 months, but we noted a similar trend for increased LVM/Ht2.7 within the LaCO3 and placebo groups that did not reach statistical significance. Since the cohort was not significantly affected by the intervention compared to placebo, we combined the two groups for a longitudinal post hoc analysis of LVM/Ht2.7 in stable stage 3 CKD.
STUDY POPULATION AND METHODS
Subjects
The original study protocol and post hoc analysis were both approved by the Human Research Protection Office at Washington University in St. Louis. Inclusion criteria, exclusion criteria and methods for stratification and randomization were published previously. Specifically relevant to the post hoc analysis, a history of prior or current congestive heart failure and severe hypertension were each exclusion criteria in the original study [17]. Briefly, 38 subjects with stage 3 CKD were stratified for age, gender, race and diabetes status, and then randomized into 2 groups, allocated 1:1 to receive either LaCO3 or a matching placebo with meals 3 times daily for 12 months. The primary endpoint of the original study was the change in serum phosphorus. Secondary endpoints included the change in mean carotid-femoral PWV, 24-hour urine phosphorus, tubular reabsorption of phosphorus, vascular calcification score, carotid artery intima-media thickness, LVM/Ht2.7, left ventricular ejection fraction (LVEF), plasma fibroblast growth factor 23 (FGF23), plasma Dickkopf-related protein 1 (DKK1) and plasma sclerostin. LaCO3 and placebo groups were analyzed as a single cohort in this post hoc analysis since no differences in outcomes were detected during the original study.
Cardiovascular evaluations
Cardiovascular assessments were performed at baseline and 12 months. PWV was determined by use of applanation tonometry of the carotid and femoral arteries (SphygmoCor, AtCor Medical, Australia) as previously described and validated [17-22]. The applanation tonometry measurements were performed by a research technician who was blinded to clinical data, echocardiographic results and treatment group. Vascular stiffness was defined as a mean PWV greater than the 50th percentile for age (9.8 m/s) using data from The Reference Values for Arterial Stiffness’ Collaboration [23].
Two-dimensional (2D) and M-mode echocardiograms were performed as described previously [17]. LVEF was determined by 2D echocardiography using the modified Simpson's method of disks; LVM was measured by the 2D-guided M-mode-derived cubed method and indexed to both body surface area (LVM/BSA) and height in meters raised to the power of 2.7 (LVM/Ht2.7) [24]. Although LVM/BSA is commonly used in clinical trials, it underestimates the degree of left ventricular hypertrophy (LVH) in overweight and obese individuals. LVM/Ht2.7 enhances the ability to detect LVH in this setting [24-26]. Subjects were considered to have LVH if the LVM/Ht2.7 was greater than 51 g/m2.7 [24]. Diastolic function metrics were obtained using pulsed-wave Doppler and included early peak mitral inflow velocity (E), late peak mitral inflow velocity (A), the E/A ratio, and left atrial volume (LAV) [24,27]. Additional diastolic function metrics were obtained using pulsed-wave tissue Doppler imaging (TDI) and included early diastolic lateral annular velocity (lateral e’) and the ratio of E to lateral e’ (lateral E/e’ ratio), which provides an estimate of left atrial pressure [27]. Normal reference values for these metrics are as follows: LAV < 34 ml/m2, lateral e’ ≥ 10 cm/s, lateral E/e’ ratio ≤ 8 (also normal between 9-12 if normal LAV), E/A ratio 1-2 (along with normal LAV and normal lateral e’) [24,27]. Subjects were considered to have diastolic dysfunction if either of the following patterns were detected by echocardiography: 1) impaired myocardial relaxation: E/A < 0.8, lateral e’ < 10 cm/s, lateral E/e’ < 8, LAV > 34 ml/m2; and/or 2) increased left atrial pressure (LAP): lateral E/e’ > 12 with LAV > 34 ml/m2. The classification scheme for diastolic dysfunction was drawn from a recently published guideline by the American Society of Echocardiography [27]. All measurements were performed in accordance to published guidelines and represent the average of three consecutive cardiac cycles obtained by a single observer blinded to all clinical parameters and treatment group.
Plasma FGF23 and soluble klotho levels
Blood samples were obtained from each subject at the baseline visit and after 12 months of treatment. Plasma levels of FGF23 were measured in duplicate using a commercially available ELISA kit as previously described [17]. Soluble klotho levels were measured in duplicate using a commercially available ELISA kit according to the manufacturer's instructions (Immuno-biological Laboratories, Japan).
Outcome Definitions
The primary outcome was the change in LVM/Ht2.7 (g/m2.7) from baseline to month 12. Secondary outcomes included changes in the non-indexed LVM (g) and LVM indexed to BSA [LVM/BSA (g/m2)], systolic blood pressure (SBP), diastolic blood pressure (DBP), carotid-femoral PWV, LVEF, E velocity, A velocity, E/A ratio, lateral e’ velocity, lateral E/e’ ratio, plasma FGF23, plasma soluble klotho, FGF23/klotho ratio and eGFR (calculated using the Modification of Diet in Renal Disease [MDRD] Study Equation) [28] from baseline to month 12.
Statistical Analysis
Statistical analysis was performed by a statistician who remained blinded to the identity of the subjects and their original study groups (J. M.). The data were analyzed using the software package SAS 9.1 (Cary, NC). All outcomes were analyzed in both original treatment arms combined since there were no significant differences between groups in the original study [17]. The distribution of each outcome variable was evaluated for normality using Kolmogorov-Smirnov Z test. Signed-rank test (non-normally distributed data) and paired t test (normally distributed data) were used to test differences of continuous variables from baseline to the 12-month visit. Bivariate correlations involving normally distributed variables were performed using Pearson's correlation. Independent categorical variables were compared using Chi-square test; paired categorical variables were compared using McNemar's test. Normally distributed data are presented as mean ± standard deviation (SD). Non-normally distributed data are presented as median (range). All tests were two-tailed; statistical significance was considered at p < 0.05.
RESULTS
Baseline Demographics
Of the 38 subjects from the original cohort, 31 had complete demographic, biochemical, and cardiovascular data at the baseline and 12-month visit and were included in the post hoc analysis. Subjects were excluded from the analysis due to either inadequate plasma remaining for measuring circulating klotho levels or incomplete data for assessment of diastolic function, which were both outcomes not analyzed in the original study. There were no significant differences in baseline demographic or clinical data in included versus excluded subjects (data not shown). The demographic data for the combined cohort is presented in Table 1. At baseline, LVH was present in 10/31 (32%) and diastolic dysfunction was present in 15/31 (48%, all with impaired myocardial relaxation: 13 had normal LAP and 2 had increased LAP; Table 2).
Table 1.
Baseline demographic data for all 31 subjects included in the post hoc analysis. Data are presented as mean ± standard deviation or number (%).
| Age (years) | 62 ± 12 |
| Height (cm) | 174 ± 11 |
| Weight (kg) | 96 ± 22 |
| BMI | 31 ± 5 |
| Systolic BP (mmHg) | 131 ± 14 |
| Diastolic BP (mmHg) | 77 ± 10 |
| Heart Rate (beats per minute) | 71 ± 10 |
| Treatment Group (LaCO3/Placebo) | 16/15 |
| Gender (M/F) | 21/10 |
| Diabetes Mellitus (no., %) | 11 (36%) |
| Race (no., %) | |
| African-American | 8 (26%) |
| Caucasian | 21 (68%) |
| Other | 2 (6%) |
| Antihypertensive use (no., %) | 25 (80%) |
Table 2.
Comparison of clinical data at the baseline and 12-month visit for all 31 subjects included in the post hoc analysis. Data are presented as mean ± standard deviation, median (range), or number (%).
| Baseline (n=31) | 12 months (n=31) | P | |
|---|---|---|---|
| Biochemical Data | |||
| BMI | 31 ± 5 | 32 ± 6 | 0.16 |
| Serum creatinine, mg/dl | 1.7 ± 0.3 | 1.7 ± 0.4 | 0.80 |
| eGFR, ml/min/1.73 m2 | 46 ± 12 | 47 ± 15 | 0.95 |
| Serum calcium, mg/dl | 9.2 ± 0.3 | 9.5 ± 0.4 | 0.70 |
| Serum phosphorus, mg/dl | 3.4 ± 0.5 | 3.2 ± 0.5 | 0.80 |
| TRP, % | 77 ± 10 | 73 ± 10 | 0.45 |
| Intact PTH, pg/ml | 67 ± 50 | 71 ± 48 | 0.52 |
| FGF23, pg/ml* | 58 (24, 201) | 55 (33, 367) | 0.95 |
| Soluble klotho, pg/ml | 748 ± 289 | 536 ± 410 | 0.03a |
| FGF23/klotho ratio | 0.09 ± 0.07 | 0.26 ± 0.25 | 0.01a |
| Cardiovascular Data † | |||
| Systolic BP, mmHg | 131 ± 14 | 130 ± 12 | 0.69 |
| Diastolic BP, mmHg | 77 ± 10 | 71 ± 8 | 0.01a |
| Heart rate, beats per minute | 70 ± 10 | 69 ± 10 | 0.25 |
| LVM, g | 203 ± 79 | 223 ± 70 | 0.02a |
| LVM, g/m2 | 100 ± 29 | 106 ± 27 | 0.03a |
| LVM, g/m2.7 | 47 ± 13 | 53 ± 13 | 0.006a |
| LVH, no., % | 10 (32%) | 15 (48%) | 0.33 |
| LVEF, % | 62 ± 10 | 66 ± 9 | 0.04a |
| PWV, m/s | 10.7 ± 1.8 | 10.1 ± 2.1 | 0.08 |
| Diastolic Function † | |||
| E velocity (cm/s) | 0.57 (0.33, 1.23) | 0.58 (0.34, 1.38) | 0.74 |
| A velocity (cm/s) | 0.71 (0.47, 1.24) | 0.69 (0.50, 1.12) | 0.25 |
| E/A ratio | 0.78 (0.43, 1.50) | 0.80 (0.52, 1.75) | 0.60 |
| Lateral e’, cm/s | 8 (5, 18) | 10 (5, 16) | 0.10 |
| Lateral E/e′ ratio | 6.3 (3.3, 22.8) | 6.2 (3.4, 23.6) | 0.10 |
| Left atrial volume, ml/m2 | 51 (21, 128) | 53 (26, 118) | 0.40 |
| Diastolic dysfunction, no., % | 15 (48%) | 14 (45%) | 1.00 |
Please see the Methods section for normal reference values for the cardiovascular data and diastolic function variables.
FGF23 is the only non-normally distributed biochemical variable and is presented as median (range).
Statistically significant difference in means between the baseline and 12-month visit using paired t test.
Primary Outcome of the Combined Cohort Analysis
Mean LVM/Ht2.7 significantly increased from 47.4 ± 13.2 g/m2.7 at baseline to 53.4 ± 12.9 g/m2.7 at 12 months (P = 0.006, Table 2, Figure 1). The proportion of subjects with LVH increased from 32% at baseline to 48% at 12 months, but this change was not statistically significant (Table 2). Of the subjects without LVH at baseline, 29% developed LVH at 12 months; in contrast, only one subject with LVH at baseline demonstrated normal LVM/Ht2.7 at 12 months. The change in LVM/Ht2.7 from baseline to the 12-month visit was similar within the original study groups (LaCO3: 45.6 ± 13.4 to 53.2 ± 13.3 g/m2.7, P = 0.44; Placebo: 49.3 ± 13.2 to 53.6 ± 13.0 g/m2.7, P = 0.51). The between-group comparison of the change in LVM/Ht2.7 was not statistically significant (P = 0.36). Mean eGFR was 46 ml/min/1.73 m2 at baseline and did not significantly change during the study, demonstrating stability of stage 3 CKD during the follow-up period (Table 2).
Figure 1.
Comparison of mean LVM/Ht2.7 between the baseline and 12-month visit using paired t test. Whiskers represent one standard error of the mean for each group. The dotted line represents the threshold value for diagnosing LVH (51 g/m 2.7).
Secondary Outcomes in the Combined Cohort Analysis
Mean non-indexed LVM significantly increased from 203 ± 79 g at baseline to 223 ± 70 g at 12 months (P = 0.02, Table 2). Mean LVM/BSA also significantly increased from 100 ± 29 g/m2 at baseline to 106 ± 27 g/m2 at 12 months (P = 0.03, Table 2). SBP remained stable during the study, but a small yet statistically significant decrease in DBP occurred over 12 months (Table 2). We observed increased PWV reflecting vascular stiffness at the baseline visit and at 12 months (Table 2), with mean PWV greater than the 50th percentile for age (9.8 m/s) using data from The Reference Values for Arterial Stiffness’ Collaboration [23]. Left ventricular ejection fraction (LVEF) remained normal at greater than 60% throughout the study, with a small but statistically significant increase from baseline to the 12-month visit (Table 2).
Diastolic dysfunction was present at baseline and persisted over 12 months in many subjects, although there were no significant changes during the study (Table 2). Specifically, the E velocity (median: 0.57-0.58 cm/s), A velocity (median: 0.69-0.71 cm/s), E/A ratio (median: 0.78-0.80), lateral e’ (median: 8-10 cm/s), lateral E/e’ ratio (median: 6.2-6.3), and left atrial volume (median: 51-53 ml/m2) remained unchanged, all consistent with mild diastolic dysfunction [27] (Table 2).
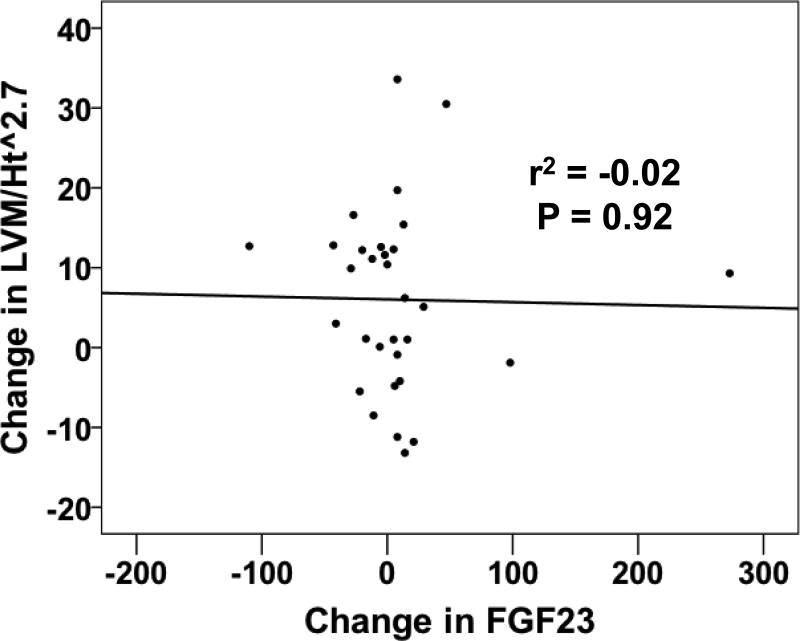
Plasma FGF23 levels were variable at the baseline visit and did not change significantly during the study (Table 2). Furthermore, the change in FGF23 was not associated with the change in LVM/Ht2.7 over 12 months (Pearson's r2 -0.02, P = 0.92, Figure 2). Mean circulating klotho levels were significantly lower at 12 months versus baseline (536 ± 410 versus 748 ± 289 pg/ml, respectively, P = 0.03, Table 2) and overall were 20-50% lower than in previous reports using the same ELISA in patients with stage 3 CKD [29]. There was no correlation between change in soluble klotho levels and change in eGFR or LVM/Ht2.7 over 12 months (data not shown). FGF23 and klotho levels were not correlated with one another at baseline (Pearson's r2 -0.124, P = 0.67) but were strongly correlated at 12 months (Pearson's r2 0.553, P = 0.04). The FGF23/klotho ratio increased significantly over 12 months (0.09 ± 0.07 to 0.26 ± 0.25, P = 0.01, Table 2). Of the secondary outcomes, only the change in FGF23/klotho ratio was significantly associated with the change in LVM/Ht2.7 over 12 months (Pearson's r2 0.582, P = 0.03, Figure 3).
Figure 2.
Scatter plot of the change in FGF23 levels versus the change in LVM/Ht2.7 between the baseline and 12-month visit. The strength (r2) and significance (P-value) of the association were generated using Pearson's correlation.
Figure 3.
Scatter plot of the change in FGF23/klotho ratio versus the change in LVM/Ht2.7 between the baseline and 12-month visit. The strength (r2) and significance (P-value) of the association were generated using Pearson's correlation.
DISCUSSION
In this post hoc analysis, we identified a significant increase in LVM/Ht2.7 over a 12-month follow-up period in 31 adults with stage 3 CKD, despite stable blood pressure, stable kidney function and normal left ventricular systolic function. This increase remained significant across other definitions of LV mass, including non-indexed LVM (g) and LVM indexed to BSA (g/m2). Given that approximately 10% of the general population has stage 3 CKD, there is a pressing need to identify biomarkers associated with poor CV outcomes. While it has been previously shown that biomarkers of CV risk (such as LV mass) progressively worsen in patients with stage 5 CKD on dialysis, to our knowledge this is the first study to demonstrate progression of LV mass (a biomarker that portends adverse CV risk) in an otherwise stable stage 3 CKD population.
We compared our findings to those of a recent study published by Chue et al that followed 109 subjects with stage 3 CKD in a 40-week placebo-controlled study using sevelamer carbonate [29]. In their study, LV mass (non-indexed and indexed to BSA) did not significantly increase in the sevelamer or placebo group over 40 weeks. An important difference between our study and that of Chue et al is their use of cardiac MRI rather than echocardiography to measure LV mass, which precludes a direct comparison of primary outcomes between our two studies. Our two cohorts were similar in age, gender and eGFR, although our cohort had more vascular stiffness, more diastolic dysfunction, and a slightly longer follow-up period (52 versus 40 weeks). Importantly, our cohort had lower baseline levels of circulating klotho that declined significantly over 12 months, versus higher baseline klotho levels that remained stable in Chue et al (using an identical ELISA kit). The decline in klotho levels in our cohort may reflect a decrease in true GFR and subclinical progression of CKD during the study that was not detected using the MDRD-derived eGFR, as suggested by recent studies of klotho levels as a highly sensitive biomarker of kidney injury in early CKD [30-33].
Although we detected a significant increase in LV mass, this was not associated with changes in other biomarkers that have been associated with worsening CV risk such as SBP, DBP, LV systolic function, LV diastolic function, or PWV. However, it is feasible that persistently elevated PWV, a biomarker of vascular stiffness, contributed to cardiac remodeling and increasing LV mass as was seen in the Multiethnic Study of Atherosclerosis [34]. Moreover, we observed persistent, mild diastolic dysfunction over 12 months in stage 3 CKD, which has been previously reported as a correlate of increasing LV mass and a strong predictor of mortality in stage 4-5 CKD [35,36].
To our surprise and in contrast to recent animal and human studies, we did not find an association between plasma FGF23 levels and the change in LVM/Ht2.7 [37,38]. However, we demonstrated a significant decline in circulating klotho levels over 12 months that was not associated with a change in LVM/Ht2.7. We suspect that the decline in circulating klotho levels reflects a loss of membrane-bound klotho from kidney tubules, as was demonstrated in a recent study by Sakan et al in patients with CKD [39]. The loss of membrane-bound klotho should impair canonical FGF23 signaling in the kidney and cardiovascular system since klotho is the required co-receptor for FGF23/FGF receptor (FGFR) interactions [40,41]. However, FGF23/FGFR signaling has been demonstrated in the kidney and cardiovascular system of klotho-deficient mice [37,42]. In a seminal paper by Faul et al, FGF23/FGFR signaling stimulated LVH in a dose-responsive pattern in klotho-haploinsufficient and klotho-knockout mouse models of CKD [37]. However, in these animal models lower expression of klotho was accompanied by higher plasma levels of FGF23, which were not seen in our cohort. In Faul et al, klotho-independent FGF23/FGFR signaling was shown to induce a hypertrophic phenotype in isolated cardiac myocytes but was not tested in other resident cells playing an important role in LVH such as fibroblasts or myofibroblasts [37]. The mechanism of klotho-independent FGF23/FGFR signaling in CKD is unknown, but one potential explanation is the promiscuous binding of FGF23 to FGFR in cardiac myocytes that is further enhanced by reduced klotho expression in the kidney and parathyroid glands [37]. A similar paradigm has been proposed by Hu et al, where a loss of renal klotho and an accompanying decrease in soluble klotho levels stimulated vascular calcification in animal models of CKD [42]. Although highly speculative, our observation that the plasma FGF23/klotho ratio increased significantly over 12 months and was associated with the change in LVM/Ht2.7 could reflect a similar state of klotho-independent FGF23/FGFR signaling at the level of the myocardium in stage 3 CKD. As in the animal models of CKD in Faul et al, this may have contributed to progression of LV mass by activating hypertrophic gene programs in cardiac myocytes.
This study has several limitations inherent to post hoc analyses, the most important of which is the small sample size that limits our ability to draw inferences about the primary outcome. We computed our achieved power for detecting a difference in LVM g/m2.7 from baseline to 12 months. Using the observed mean difference of 6 g/m2.7 and standard deviation of 13 g/m2.7 at each time point, the achieved power in our post hoc analysis was 77% with a two-tailed alpha of 0.05. Thus, despite the small size of our cohort we had modest power to detect the observed small but significant change in LVM g/m2.7. Another limitation is the lack of available plasma samples to generate data in other biomarkers of progression of LVM and heart failure in patients with CKD, such as galectin-3, ST-2, troponin I, and BNP [43]. Exhaustion of the original samples precluded several subjects from inclusion in the post hoc analysis. These biomarkers should be included in future prospective studies of the progression of LV mass in CKD.
In conclusion, subjects with stage CKD 3 exhibited a progressive increase in LV mass with persistent LV diastolic dysfunction and vascular stiffness over 12 months despite stable kidney function, blood pressure and normal LV systolic function. The change in FGF23/klotho ratio was associated with increasing LV mass, which may reflect klotho-independent FGF23 signaling in cardiac myocytes that has been associated with LVH in animal models of CKD. These findings deserve further evaluation in a larger population, given the adverse prognostic value of progression of these cardiovascular biomarkers
ACKNOWLEGMENTS
The authors are grateful to Jingnan Mao for her assistance as a statistician. The LaCO3 and matched placebo in the original study were provided by Shire U.S. Pharmaceuticals, Inc. The study was funded by Shire U.S. Pharmaceuticals, Inc., and by NIH grants DK 070790 (K.A.H.), KL2 RR024994 and UL1 RR024992 (M.E.S. and Washington University), and L40 DK099748-01 (M.E.S.).
Footnotes
DISCLOSURE:
K.A.H. has been a consultant for or the recipient of research funding from Shire, Genzyme and Fresenius.
REFERENCES
- 1.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351:1296–1305. doi: 10.1056/NEJMoa041031. [DOI] [PubMed] [Google Scholar]
- 2.Culleton BF, Larson MG, Wilson PW, Evans JC, Parfrey PS, Levy D. Cardiovascular disease and mortality in a community-based cohort with mild renal insufficiency. Kidney Int. 1999;56:2214–2219. doi: 10.1046/j.1523-1755.1999.00773.x. [DOI] [PubMed] [Google Scholar]
- 3.Foley RN, Murray AM, Li S, Herzog CA, McBean AM, Eggers PW, Collins AJ. Chronic kidney disease and the risk for cardiovascular disease, renal replacement, and death in the united states medicare population, 1998 to 1999. Journal of the American Society of Nephrology. 2005;16:489–495. doi: 10.1681/ASN.2004030203. [DOI] [PubMed] [Google Scholar]
- 4.Baber U, Gutierrez OM, Levitan EB, Warnock DG, Farkouh ME, Tonelli M, Safford MM, Muntner P. Risk for recurrent coronary heart disease and all-cause mortality among individuals with chronic kidney disease compared with diabetes mellitus, metabolic syndrome, and cigarette smokers. Am Heart J. 2013;166:373–380. e372. doi: 10.1016/j.ahj.2013.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, Van Lente F, Levey AS. Prevalence of chronic kidney disease in the united states. Journal of American Medical Association. 2007;298:2038–2047. doi: 10.1001/jama.298.17.2038. [DOI] [PubMed] [Google Scholar]
- 6.Elias MF, Davey A, Dore GA, Gillespie A, Abhayaratna WP, Robbins MA. Deterioration in renal function is associated with increased arterial stiffness. American Journal of Hypertension. 2014;27:207–214. doi: 10.1093/ajh/hpt179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Utescu MS, Couture V, Mac-Way F, De Serres SA, Marquis K, Larivière R, Desmeules S, Lebel M, Boutouyrie P, Agharazii M. Determinants of progression of aortic stiffness in hemodialysis patients: A prospective longitudinal study. Hypertension. 2013;62:154–160. doi: 10.1161/HYPERTENSIONAHA.113.01200. [DOI] [PubMed] [Google Scholar]
- 8.Blacher J, Guerin AP, Pannier B, Marchais SJ, Safar ME, London GrM. Impact of aortic stiffness on survival in end-stage renal disease. Circulation. 1999;99:2434–2439. doi: 10.1161/01.cir.99.18.2434. [DOI] [PubMed] [Google Scholar]
- 9.Blacher J, Guerin AP, Pannier B, Marchais SJ, London GM. Arterial calcifications, arterial stiffness, and cardiovascular risk in end-stage renal disease. Hypertension. 2001;38:938–942. doi: 10.1161/hy1001.096358. [DOI] [PubMed] [Google Scholar]
- 10.Blacher J, Safar ME, Guerin AP, Pannier B, Marchais SJ, London GM. Aortic pulse wave velocity index and mortality in end-stage renal disease. Kidney Int. 2003;63:1852–1860. doi: 10.1046/j.1523-1755.2003.00932.x. [DOI] [PubMed] [Google Scholar]
- 11.Foley RN, Parfrey PS, Kent GM, Harnett JD, Murray DC, Barre PE. Serial change in echocardiographic parameters and cardiac failure in end-stage renal disease. Journal of the American Society of Nephrology. 2000;11:912–916. doi: 10.1681/ASN.V115912. [DOI] [PubMed] [Google Scholar]
- 12.Baek SD, Baek CH, Kim JS, Kim SM, Kim JH, Kim SB. Does stage iii chronic kidney disease always progress to end-stage renal disease? A ten-year follow-up study. Scandinavian Journal of Urology and Nephrology. 2012;46:232–238. doi: 10.3109/00365599.2011.649045. [DOI] [PubMed] [Google Scholar]
- 13.Van Pottelbergh G, Bartholomeeusen S, Buntinx F, Degryse J. The evolution of renal function and the incidence of end-stage renal disease in patients aged ≥50 years. Nephrology Dialysis Transplantation. 2012;27:2297–2303. doi: 10.1093/ndt/gfr659. [DOI] [PubMed] [Google Scholar]
- 14.Nitsch D, Grams M, Sang Y, Black C, Cirillo M, Djurdjev O, Iseki K, Jassal SK, Kimm H, Kronenberg F, Øien CM, Levey AS, Levin A, Woodward M, Hemmelgarn BR. Associations of estimated glomerular filtration rate and albuminuria with mortality and renal failure by sex: A meta-analysis. BMJ. 2013:346. doi: 10.1136/bmj.f324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hallan SI, Matsushita K, Sang Y, et al. Age and association of kidney measures with mortality and end-stage renal disease. JAMA. 2012;308:2349–2360. doi: 10.1001/jama.2012.16817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Isakova T, Xie H, Yang W, et al. Fibroblast growth factor 23 and risks of mortality and end-stage renal disease in patients with chronic kidney disease. JAMA. 2011;305:2432–2439. doi: 10.1001/jama.2011.826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Seifert ME, de las Fuentes L, Rothstein M, Dietzen DJ, Bierhals AJ, Cheng SC, Ross W, Windus D, Dávila-Román VG, Hruska KA. Effects of phosphate binder therapy on vascular stiffness in early-stage chronic kidney disease. American Journal of Nephrology. 2013;38:158–167. doi: 10.1159/000353569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pauca AL, O'Rourke MF, Kon ND. Prospective evaluation of a method for estimating ascending aortic pressure from the radial artery pressure waveform. Hypertension. 2001;38:932–937. doi: 10.1161/hy1001.096106. [DOI] [PubMed] [Google Scholar]
- 19.Van Bortel LM, Balkestein EJ, van der Heijden-Spek JJ, Vanmolkot FH, Staessen JA, Kragten JA, Vredeveld JW, Safar ME, Struijker Boudier HA, Hoeks AP. Non-invasive assessment of local arterial pulse pressure: Comparison of applanation tonometry and echo-tracking. J Hypertens. 2001;19:1037–1044. doi: 10.1097/00004872-200106000-00007. [DOI] [PubMed] [Google Scholar]
- 20.O'Rourke MF, Pauca A, Jiang XJ. Pulse wave analysis. Br J Clin Pharmacol. 2001;51:507–522. doi: 10.1046/j.0306-5251.2001.01400.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.O'Rourke MF, Adji A. An updated clinical primer on large artery mechanics: Implications of pulse waveform analysis and arterial tonometry. Curr Opin Cardiol. 2005;20:275–281. doi: 10.1097/01.hco.0000166595.44711.6f. [DOI] [PubMed] [Google Scholar]
- 22.Wilkinson IB, Fuchs SA, Jansen IM, Spratt JC, Murray GD, Cockcroft JR, Webb DJ. Reproducibility of pulse wave velocity and augmentation index measured by pulse wave analysis. J Hypertens. 1998;16:2079–2084. doi: 10.1097/00004872-199816121-00033. [DOI] [PubMed] [Google Scholar]
- 23.Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: ‘establishing normal and reference values’. Eur heart j. England. 2010;31:2338–2350. doi: 10.1093/eurheartj/ehq165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, Picard MH, Roman MJ, Seward J, Shanewise JS, Solomon SD, Spencer KT, Sutton MS, Stewart WJ, Group CQW, Committee ASoEsGaS, Echocardiography EAo Recommendations for chamber quantification: A report from the american society of echocardiography's guidelines and standards committee and the chamber quantification writing group, developed in conjunction with the european association of echocardiography, a branch of the european society of cardiology. J Am Soc Echocardiogr. 2005;18:1440–1463. doi: 10.1016/j.echo.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 25.de Simone G, Daniels SR, Devereux RB, Meyer RA, Roman MJ, de Divitiis O, Alderman MH. Left ventricular mass and body size in normotensive children and adults: Assessment of allometric relations and impact of overweight. J Am Coll Cardiol. 1992;20:1251–1260. doi: 10.1016/0735-1097(92)90385-z. [DOI] [PubMed] [Google Scholar]
- 26.de Simone G, Devereux RB, Daniels SR, Koren MJ, Meyer RA, Laragh JH. Effect of growth on variability of left ventricular mass: Assessment of allometric signals in adults and children and their capacity to predict cardiovascular risk. J Am Coll Cardiol. 1995;25:1056–1062. doi: 10.1016/0735-1097(94)00540-7. [DOI] [PubMed] [Google Scholar]
- 27.Nagueh SF, Appleton CP, Gillebert TC, Marino PN, Oh JK, Smiseth OA, Waggoner AD, Flachskampf FA, Pellikka PA, Evangelisa A. Recommendations for the evaluation of left ventricular diastolic function by echocardiography. Eur J Echocardiogr. 2009;10:165–193. doi: 10.1093/ejechocard/jep007. [DOI] [PubMed] [Google Scholar]
- 28.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D, Group* ftMoDiRDS A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Annals of Internal Medicine. 1999;130:461–470. doi: 10.7326/0003-4819-130-6-199903160-00002. [DOI] [PubMed] [Google Scholar]
- 29.Chue CD, Townend JN, Moody WE, Zehnder D, Wall NA, Harper L, Edwards NC, Steeds RP, Ferro CJ. Cardiovascular effects of sevelamer in stage 3 ckd. J Am Soc Nephrol. 2013;24:842–852. doi: 10.1681/ASN.2012070719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hu MC, Shi M, Zhang J, Quiñones H, Griffith C, Kuro-o M, Moe OW. Klotho deficiency causes vascular calcification in chronic kidney disease. Journal of the American Society of Nephrology. 22:124–136. doi: 10.1681/ASN.2009121311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hu MC, Shi M, Zhang J, Quinones H, Kuro-o M, Moe OW. Klotho deficiency is an early biomarker of renal ischemia-reperfusion injury and its replacement is protective. Kidney Int. 78:1240–1251. doi: 10.1038/ki.2010.328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pavik I, Jaeger P, Ebner L, Poster D, Krauer F, Kistler AD, Rentsch K, Andreisek G, Wagner CA, Devuyst O, Wüthrich RP, Schmid C, Serra AL. Soluble klotho and autosomal dominant polycystic kidney disease. Clinical Journal of the American Society of Nephrology. 2012;7:248–257. doi: 10.2215/CJN.09020911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Moreno JA, Izquierdo MC, Sanchez-Niño MD, Suárez-Alvarez B, Lopez-Larrea C, Jakubowski A, Blanco J, Ramirez R, Selgas R, Ruiz-Ortega M, Egido J, Ortiz A, Sanz AB. The inflammatory cytokines tweak and tnfα reduce renal klotho expression through nfκb. J Am Soc Nephrol. 2011;22:1315–1325. doi: 10.1681/ASN.2010101073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ix JH, Katz R, Peralta CA, de Boer IH, Allison MA, Bluemke DA, Siscovick DS, Lima JA, Criqui MH. A high ankle brachial index is associated with greater left ventricular mass mesa (multi-ethnic study of atherosclerosis). J Am Coll Cardiol. 2010;55:342–349. doi: 10.1016/j.jacc.2009.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Peterson GE, de Backer T, Contreras G, Wang X, Kendrick C, Greene T, Appel LJ, Randall OS, Lea J, Smogorzewski M, Vagaonescu T, Phillips RA, Investigators AASoKD Relationship of left ventricular hypertrophy and diastolic function with cardiovascular and renal outcomes in african americans with hypertensive chronic kidney disease. Hypertension. 2013;62:518–525. doi: 10.1161/HYPERTENSIONAHA.111.00904. [DOI] [PubMed] [Google Scholar]
- 36.Farshid A, Pathak R, Shadbolt B, Arnolda L, Talaulikar G. Diastolic function is a strong predictor of mortality in patients with chronic kidney disease. BMC Nephrol. 2013;14:280. doi: 10.1186/1471-2369-14-280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, Gutiérrez OM, Aguillon-Prada R, Lincoln J, Hare JM, Mundel P, Morales A, Scialla J, Fischer M, Soliman EZ, Chen J, Go AS, Rosas SE, Nessel L, Townsend RR, Feldman HI, St John Sutton M, Ojo A, Gadegbeku C, Di Marco GS, Reuter S, Kentrup D, Tiemann K, Brand M, Hill JA, Moe OW, Kuro-O M, Kusek JW, Keane MG, Wolf M. Fgf23 induces left ventricular hypertrophy. J Clin Invest. 2011;121:4393–4408. doi: 10.1172/JCI46122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gutiérrez OM, Januzzi JL, Isakova T, Laliberte K, Smith K, Collerone G, Sarwar A, Hoffmann U, Coglianese E, Christenson R, Wang TJ, deFilippi C, Wolf M. Fibroblast growth factor 23 and left ventricular hypertrophy in chronic kidney disease. Circulation. 2009;119:2545–2552. doi: 10.1161/CIRCULATIONAHA.108.844506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sakan H, Nakatani K, Asai O, Imura A, Tanaka T, Yoshimoto S, Iwamoto N, Kurumatani N, Iwano M, Nabeshima Y, Konishi N, Saito Y. Reduced renal α-klotho expression in ckd patients and its effect on renal phosphate handling and vitamin d metabolism. PLoS One. 2014;9:e86301. doi: 10.1371/journal.pone.0086301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Goetz R, Beenken A, Ibrahimi OA, Kalinina J, Olsen SK, Eliseenkova AV, Xu C, Neubert TA, Zhang F, Linhardt RJ, Yu X, White KE, Inagaki T, Kliewer SA, Yamamoto M, Kurosu H, Ogawa Y, Kuro-o M, Lanske B, Razzaque MS, Mohammadi M. Molecular insights into the klotho-dependent, endocrine mode of action of fibroblast growth factor 19 subfamily members. Molecular and Cellular Biology. 2007;27:3417–3428. doi: 10.1128/MCB.02249-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kurosu H, Ogawa Y, Miyoshi M, Yamamoto M, Nandi A, Rosenblatt KP, Baum MG, Schiavi S, Hu MC, Moe OW, Kuro-o M. Regulation of fibroblast growth factor-23 signaling by klotho. J Biol Chem. 2006;281:6120–6123. doi: 10.1074/jbc.C500457200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hu MC, Shi M, Zhang J, Quinones H, Griffith C, Kuro-o M, Moe OW. Klotho deficiency causes vascular calcification in chronic kidney disease. J Am Soc Nephrol. 22:124–136. doi: 10.1681/ASN.2009121311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tang WH, Shrestha K, Shao Z, Borowski AG, Troughton RW, Thomas JD, Klein AL. Usefulness of plasma galectin-3 levels in systolic heart failure to predict renal insufficiency and survival. Am J Cardiol. 2011;108:385–390. doi: 10.1016/j.amjcard.2011.03.056. [DOI] [PMC free article] [PubMed] [Google Scholar]