Abstract
The aim of the present study was to genotype Salmonella enterica serotype paratyphi A (SPA) isolated from Yuxi, China, in a multiple-locus variable number of tandem repeats (VNTRs) analysis (MLVA) and to compare them with isolates from the Chinese Medical Culture Collection Center (CMCC). Potential VNTRs were screened from the genomes of ATCC9150 and AKU_12601 using the Tandem Repeats Finder program. Nine VNTRs were established for MLVA typing of 195 SPA isolates from Yuxi and 20 isolates from CMCC. The dendogram for MLVA profiles and minimum spanning tree (MST) were drawn using the categorical coefficient calculated by BioNumerics software. A total of 23 MLVA types were identified in 215 SPA isolates and were grouped into six distinct cluster groups A, B, C, D, E and F. A total of 195 Yuxi SPA isolates were exclusively grouped into cluster C with nine MLVA genotypes. A total of 20 CMCC isolates were grouped in clusters A B, D, E and F with the other 14 MLVA types. The MLVA with nine VNTR loci, which was exploited in the present study, represents a successful strategy for genotyping SPA. Furthermore, the 195 Yuxi isolates appear to be closely related to each other and distinct from the 20 CMCC strains.
Keywords: Salmonella enterica serotype paratyphi A, multiple-locus variable number of tandem repeat analysis
Introduction
Infectious diseases caused by a variety of Salmonella enterica serotypes are widespread worldwide, representing a severe public health concern (1). Infection with Salmonella enterica serotype paratyphi A (SPA) is an emerging global public health problem due to the increase in enteric fever cases caused by SPA and the lack of protective vaccines (2–4). In Southeast and Southwest China, the infection rate of SPA has increased in the past several decades with the development of tourism, where >80% of the enteric fever outbreaks are caused by SPA (5). In recent years, Yuxi City of Yunnan Province has become one of the most severely endemic areas of SPA in China (6).
Subtyping and tracking individual strains involved in SPA outbreak or sporadic cases are important for the control and prevention of SPA transmission in Yuxi. The technique of pulsed-field gel electrophoresis (PFGE) is currently the standard method for molecular typing and epidemic surveillance of Salmonella spp., including SPA (7,8). However, PFGE is not a routine method for SPA surveillance due to the expense of the equipment and the requirement of highly trained technicians (9). Multi-locus variable number tandem repeat (VNTR) analysis (MLVA), a genotyping method based on polymerase chain reaction (PCR) and sequencing, which distinguishes tandem sequence repeats that vary in copy numbers (10,11), may be practical for subtyping SPA due to the simple operation, low cost, high-speed and weak laboratory-dependence (12). Furthermore, MLVA genotyping is becoming an important DNA-based typing tool for investigating strains that are related or unrelated to outbreaks (13).
Although one study has previously investigated the use of MLVA for subtyping SPA, the information of VNTRs for MLVA of SPA in this investigation is limited as the VNTRs were examined from the genomes of one strain of SPA (ATCC9150) and two strains of S. enterica serovar Typhi (S. Typhi; CT18 and Ty2) (14). Although the genomes of S. Typhi and SPA are closely related (15), their tandem repeats (TRs) are different. The present study searched for TR loci from two SPA genomes, ATCC9150 (NC_006511) and AKU_12601 (NC_011147), and determined nine VNTR loci for MLVA typing of SPA. We aimed to identify the type of epidemic clone in Yuxi and whether the Yuxi SPA isolates were phylogenetically distant from the 20 strains of SPA isolates collected by the Chinese Medical Culture Collection Center (CMCC).
Materials and method
Strains and extraction of bacterial genomic DNA
A total of 215 strains of SPA, including 195 Yuxi isolates and 20 CMCC strains were used in the present study. Among the 20 CMCC strains, one strain was ATCC9150 while the other 19 were collected from various research organizations with limited background information and stored by CMCC (Table I). Among the 195 Yuxi isolates, 48 were separated from the patients of the SPA outbreak in 2007 while the others were isolated from sporadic cases between 2005 and 2009.
Table I.
Information of 20 SPA strains collected by CMCC.
| Strain | Source |
|---|---|
| ATCC9150 | ATCC |
| 50001 | Denmark |
| 50002 | Denmark |
| 50084 | USA |
| 50101 | Former Soviet Union |
| 50154 | France |
| 50433 | Bulgaria |
| 50434 | Bulgaria |
| 50672 | Poland |
| 50674 | Poland |
| 50701 | Czech Republic |
| 50078 | Beijing, China |
| 50501 | Dalian, China |
| 50502 | Dalian, China |
| 50504 | Dalian, China |
| 50505 | Dalian, China |
| 50506 | Dalian, China |
| 50507 | Lanzhou, China |
| 50508 | Guangdong, China |
| 50509 | Guangdong, China |
CMCC, Chinese Medical Culture Collection Center; SPA, Salmonella enterica serotype paratyphi A; ATCC, American Type Culture Collection.
Genomic DNA of SPA was extracted as previously described (16,17). Briefly, the bacteria were streaked on brain heart infusion agar (BHIA) plates and grown at 37°C overnight in 5% CO2 incubator. A loop of typical colonies was removed from the BHIA plates and boiled for 10 min in 200 μl Tris-EDTA buffer (10 mM Tris-Cl and 1 mM EDTA, pH 8.0). The supernatant was obtained by centrifugation at 8,000 × g for 10 min and used directly for PCR (18).
Identification of VNTRs
Potential TRs were first exploited from the genomes of ATCC9150 and AKU_12601 using the Tandem Repeats Finder (TRF) program (19,20) and the http://tandem.bu.edu/trf/trf.htlm website (21). The candidates were scored as match(+2), mismatch(−3) and indel(−5) for pattern alignment (22). The potential TRs were selected by alignment scores ≥80, or homology of repeat locus ≥85%. A total of 51 TRs (TR1-51) were screened from the genomes of ATCC9150 and AKU_12601 (data not shown). Primers flanking >51 TRs were designed using the Primer 5.0 software (Premier Biosoft International, Palo Alto, CA, USA) and synthesized by Sangon Company (Shanghai, China). The polymorphism of PCR fragments amplified with primers of 51 TRs was analyzed by agarose electrophoresis and nine VNTR loci (TR27, TR51, TR41, TR43, TR5, TR40, TR44, TR24 and TR49) were verified to be polymorphic (Fig. 1). The nine VNTRS of 19 CMCC strains except ATCC9150 were sequenced. The repeat numbers for each locus corresponding to 20 CMCC and AKU_12601 are summarized in Table II.
Figure 1.
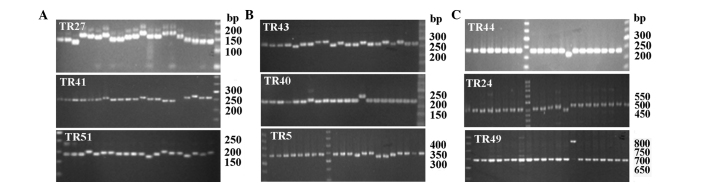
The polymorphisms of nine VNTR loci TR27, TR51, TR41, TR43, TR5, TR40, TR44, TR24 and TR49 analyzed by agarose gel electrophoresis, capillary electrophoresis and sequencing for PCR products amplified from 20 CMCC strains and one Yuxi isolate YN07044. (A) The agarose gel electrophoresis for PCR products of nine VNTR loci. Lanes: left to right, ATCC9150, 50078, 50001, 50502, 50506, 50501, 50505, 50154, 50434, 50101, 50507, 50509,50672, 50674, 50084, 50433, 50508, 50701, 50002, 50504, YN07044. DNA Marker, 50bp DNA Ladder Marker. (B) The representative electropherogram from pooled capillary electrophoresis runs of FAM-labeled or HEX-labeled primers of nine VNTRs. The PCR products for TR27, TR41, TR51, TR43, TR40 and TR5 were amplified from YN07044, demonstrating 143, 259, 194, 276, 217 and 338 bp, respectively. The PCR products for TR44 and TR24 were amplified from 50504 with 254 and 527 bp respectively. (C) The precise numbers of TR40 in three representative sequences amplified from ATCC9150 (3), 50154 (4), and 50674 (6) were analyzed by sequencing. CMCC, Chinese Medical culture Collection Center; SPA, Salmonella enterica serotype paratyphi A; VNTR, variable number of tandem repeats.
Table II.
Characteristics of 9 VNTR loci for 20 CMCC SPA strains and AKU_12601.
| VNTR locus | Primer sequence (5′-3′) | Repeat model | Repeat number |
|---|---|---|---|
| TR27 | F: GGAAAGACTGGCGAACAAAT | ||
| R: TCGCCAATACCATGAGTACG | TACTGG | 9–16 | |
| TR51 | F: CCATGGCTGCAGTTAATTTCT | ||
| R: TGATACGCTTTTGACGTTGC | ACCATG | 1–11 | |
| TR41 | F: TGGGAAACTTATCTTCGA | ||
| R: TAATCAGTCTGGCCTGTG | ACATCTCCT | 1–6 | |
| TR43 | F: TACTGCTTTCGCCATCGG | ||
| R: ATAATCCGGGTAAAGACC | CCGTTAACCG | 3–5 | |
| TR5 | F: GCATACACCGCAGCACTC | ||
| R: TTCCTTTCCCTGCTTATTTGTC | TAGCAGGTAA | 2–4 | |
| TR40 | F: CGGGTGATTCTGTTATCT | ||
| R: ATAGTGTTACGCACCTCA | TTTTTTAAG | 3–6 | |
| TR44 | F: CAGAAGCAGTTCCACCACCT | ||
| R: CATTTCACATCGCCGACTTT | GCAGGAGCTGGTGGGCGA | 1–3 | |
| TR24 | F: GCTGAAGAAGCGGCAAAAC | ||
| R: GTACCGCTATCTTTCGATGGC | 45bpa | 7–8 | |
| TR49 | F: GCTTGCAGCTAAATGGAT | ||
| R: ATCTGACGAAAGCGGAAC | 232bpb | 2–3 |
Model of repeats: TCGGCAGCCGCTTTCTTCTTAGCGTCCGCCGCTGCTTTCGCCGCC;
Model of repeats: TTCCC GCTC CAAAATTTGAAAGTACTTGTTAAGTACAGACCACCAATCGCAGGATTTCGAATTGCGACAA GGCGGCAACTGAATGAGTCCT CAGGAGCTTACTGAAGTAAGTGACTGAGGCGAGTGAAGGCAGCCAACGCAGTAGCGGTTCGAAAGACGAAGATTATGC GGGAATAGCTC AGTTGGTAGAGCACGACCTTGCCAAGGTCGGGGTCGCGAGTTCGAGTCTCGT.
CMCC, Chinese Medical culture Collection Center; SPA, Salmonella enterica serotype paratyphi A; VNTR, variable number of tandem repeats.
PCR and agarose electrophoresis analysis
All selected loci were amplified from the genomic DNA of the 20 CMCC SPA strains by PCR as described previously (23). Briefly, 1 μl bacterial lysate was amplified by a thermal cycler PTC-200 DNA Engine (MJ Search Partners, Inc., Lake Forest, IL, USA) in a 25 μl final reaction volume containing 0.1 μmol/ml dNTPs, 0.2 μmol/ml primers, 0.5 U Taq DNA polymerase (Takara Bio, Inc., Shiga, Japan) under the following conditions: 10 min at 95°C, followed by 30 cycles of three temperatures (15 sec at 95°C, 1 min at 55~60°C, 1 min at 72°C) and then 10 min at 72°C. A total of 5 μl of the PCR products were separated in 1.5% agarose gels in 1X TAE buffer (AppliChem Inc., St. Louis, MO, USA) at a voltage of 6 V/cm for ~3 h. The gels were stained in ethidium bromide for visualization under UV light and were photographed on a Gel Doc 2000 system (Bio-Rad, Hercules, CA, USA). The 50 bp (base pair) DNA Ladder Marker (Takara Bio, Inc.) was loaded in all of the gels to facilitate determining the size of the DNA fragments. To ensure the accuracy of agarose electrophoresis and to compare the results between multiple gels, the PCR products of ATCC9150 in each locus were obtained as a positive control. The TRs were identified to be polymorphic if large differences between their PCR fragments in the agarose gel electrophoresis were observed. The PCR products were purified with the QIAquick PCR Purification kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions.
MLVA typing and data analysis
In order to confirm that any length polymorphism of fragment was due to variations in the VNTR copy number (24), the purified PCR products amplified from 195 Yuxi isolate and 20 CMCC strains were sequenced by the Sangon Company. The numbers of repeats in each allele were analyzed by BioNumerics version 6.0 (Applied Maths, Austin, TX, USA) (25), and the numerical profile for each locus was created according to the copies of VNTR (14,26). The dendogram for MLVA profiles was drawn using the categorical coefficient and the alignment of unweighted pair group method using arithmetic averages (27). A minimum spanning tree (MST) was constructed using the categorical coefficient (10,23,28). The priority rule for constructing MST was set so that the genotypes that had the highest number of single-locus variants would be linked first (23).
Results
MLVA genotyping and phylogenetic analysis for 215 SPA isolates
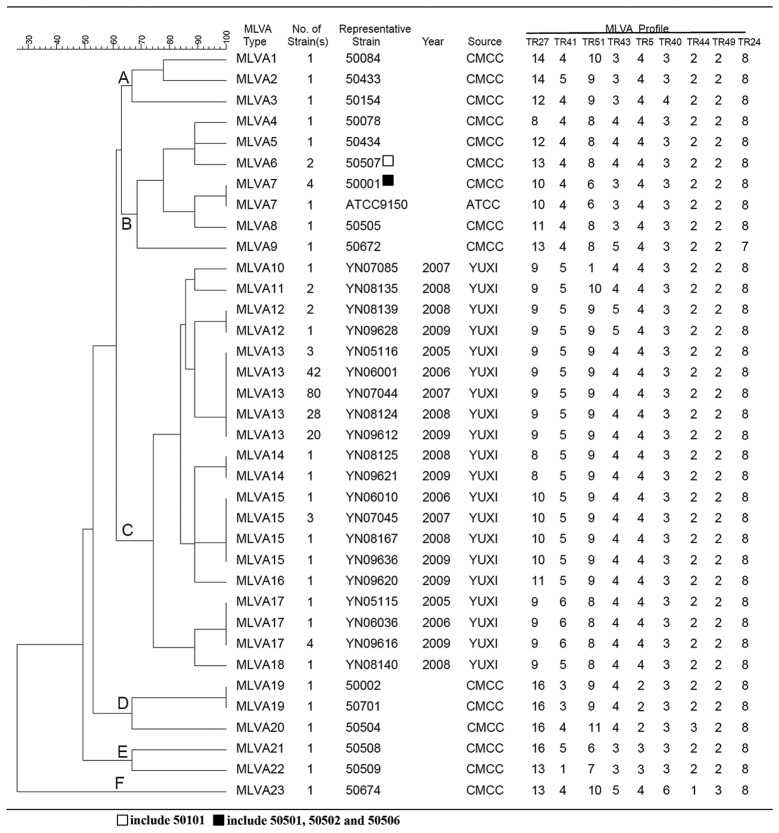
The dendogram for the MLVA types distribution demonstrates that 23 MLVA types (MLVA 1-23) were identified in the 215 SPA isolates and were grouped into six distinct cluster groups A, B, C, D, E and F. All of the Yuxi isolates were exclusively grouped into cluster C with nine MLVA genotypes (MLVA 10-18; Fig. 2). The 20 CMCC isolates were grouped in cluster A B, D, E and F with 14 MLVA types (MLVA 1-9 and MLVA 19-23; Fig. 2). There are two key observations to note among the 20 CMCC strains. Firstly, the same MLVA type of SPA emerged in different countries. For example, 50501, 50502 and 50506 (Dalian, China) and 50001 (Denmark) were typed as MLVA7, ATCC9150, 50002 (Denmark) and 50701 (Czech Republic) were typed as MLVA19, and 50507 (Lanzhou, China) and 50101 (Former Soviet Union) were typed as MLVA6, although they were from different countries. Secondly, there existed different MLVA types in the same region or country. For instance, one strain from Dalian, China (50505) was identified to be MLVA8, which is different from the other three Dalian strains of MLVA7 (50501, 50502 and 50506). Despite the fact that 50672 and 50674 were from Poland their MLVA types were MLVA9 and MLVA23, respectively. The same phenomena were observed in two strains from Bulgaria 50433 (MLVA2) and 50434 (MLVA5).
Figure 2.
MLVA type distribution of 215 SPA isolates using categorical coefficient and unweighted pair group method using arithmetic average. The 215 SPA isolates, including 195 Yuxi isolates and 20 CMCC strains were subtyped into 23 MLVA types (MLVA1~23) and grouped into six distinct clusters (A, B, C, D, E and F). All of the 195 Yuxi isolates were grouped in the C cluster with nine MLVA genotypes (MLVA10~18). The 20 CMCC strains were grouped into A, B, D, E and F clusters with 14 MLVA types (MLVA 1-9 and MLVA 19-23). MLVA, multiple-locus variable number of tandem repeats analysis; SPA, Salmonella enterica serotype paratyphi A; CMCC, Chinese Medical Culture Collection Center.
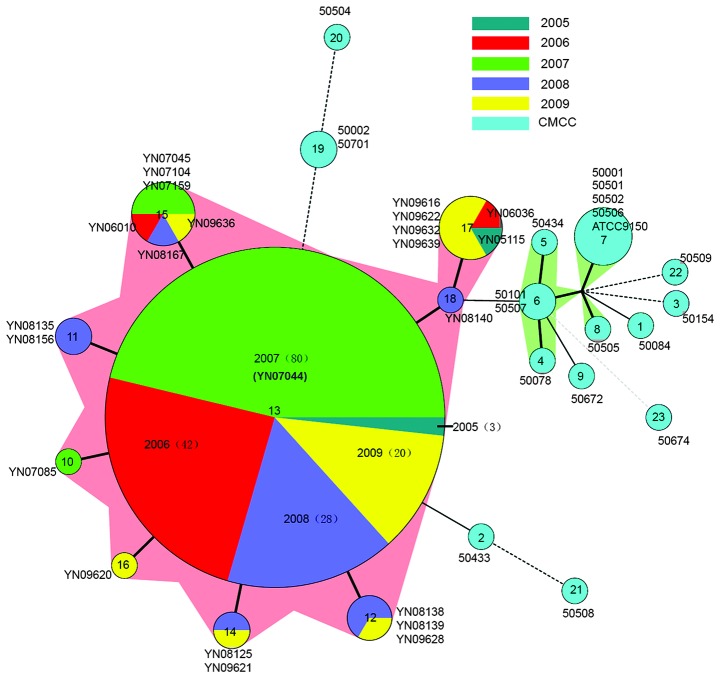
The genetic correlations among the 215 isolates were determined based on the MLVA profiles using the MST algorithm (10,23,28). As demonstrated in Fig. 3, MST offers a more detailed view of the diversity of the 215 isolates and highlights the closer subtypes that differ by few allelic changes (23). Isolates with the same MLVA profiles were clustered in a circle. A total of 173 Yuxi isolates form the MLVA13 circle surrounded by 22 other isolates with 8 MLVA types. It indicates that the 195 Yuxi isolates are closely related with each other. Although the 195 Yuxi isolates are distinct from the 20 CMCC strains, they are relatively close to 50433 (Bulgaria) with MLVA2, and also close to 50002 (Denmark), and 50701 (Czech) with MLVA19. YN08140 (Yuxi) with MLVA18 is closely related to 50101 (Former Soviet Union) and 50507 (Lanzhou, China) with MLVA6, which varied in only two VNTR loci (Fig. 2).
Figure 3.
MST obtained from 215 SPA isolates with 23 MLVA types distinguished by nine VNTRs. Each circle represents a particular MLVA type with the same MLVA profiles. The size of the circle reflects the number of isolates whereas the distance between the circles represents the genetic divergence. The heavy short lines connect single-locus variants, the thin long lines connect double-locus variant and the dotted lines connect those MLVA genotypes with differences in >2 loci. The colors represent the years of the isolates or origin of isolates. The halos surrounding the various types denote the grouping obtained by BioNumerics analysis. MST, minimum spanning trees; SPA, Salmonella enterica serotype paratyphi A.
Epidemiology of SPA in Yuxi
Although 195 Yuxi SPA isolates distribute in nine genotypes (MLVA 10-18), they express only one or two VNTR loci that are different from each other (Fig. 2). The MST demonstrated that they are closely related with each other and separated from the 20 CMCC strains (Fig. 3). Table III reveals the MLVA type distribution of 195 Yuxi isolates collected between 2005 and 2009. In all, MLVA13 accounted for 88.7% (173/195) of the Yuxi isolates. Among the 48 outbreak isolates in 2007, MLVA13 accounted for 91.7% (44/48). Outside of the 2007 outbreak, MLVA13 accounted for 87.8% (129/147) of Yuxi sporadic isolates.
Table III.
The MLVA types distribution of 195 SPA Yuxi isolates between 2005 and 2009.
| MLVA type | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
|
||||||||||
| Year | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | Total |
| 2005 | 3 | 1 | 4 | |||||||
| 2006 | 42 | 1 | 1 | 44 | ||||||
| 2007 | 1 | 80 | 3 | 84 | ||||||
| 2008 | 2 | 2 | 28 | 1 | 1 | 1 | 35 | |||
| 2009 | 1 | 20 | 1 | 1 | 1 | 4 | 28 | |||
| Total | 1 | 2 | 3 | 173 | 2 | 6 | 1 | 6 | 1 | 195 |
MLVA, multiple-locus variable number of tandem repeats analysis; SPA, Salmonella enterica serotype paratyphi A.
Discussion
A reliable method for subtyping bacterial isolates is a prerequisite for the identification of sources and transmission routes of an infectious disease (29). There is no doubt that PFGE is currently the gold-standard technique for subtyping numerous bacteria, including Salmonella serotypes with reproducible patterns and high resolution and is widely used by the CDC PulseNet surveillance program worldwide (30). However, the widespread use of PFGE is limited in the CDC of numerous Chinese cities and counties by the lack of specifically trained personnel, sophisticated and expensive equipment and precise standard protocols (31). Conversely, MLVA, which is based on the evaluation of differences in the number of TRs, is a quick, cheap and simple method for the molecular typing of bacteria (20). In the present study, a MLVA with nine VNTR markers was developed, which exhibited a wide range of variability for subtyping 215 SPA isolates into 23 MLVA types. The phylogenetic association among the 20 CMCC SPA strains with various backgrounds was elucidated clearly with 14 MLVA types. Clonal groups among the 195 Yuxi isolates in the different years were discerned with nine other MLVA types. The outbreak-related isolate was identified to be MLVA13 in 2007. Eight novel SPA isolates separated from patients in 2010 were examined with the MLVA method developed in the present study, and it was identified that six of the isolates were MLVA13 while one was MLVA14 and another was MLVA16. These results indicate that the VNTR markers identified in the present study are applicable to subtype SPA.
Yuxi, a medium-sized city (15,285 km2) with 2,095,532 residents distributed into two districts (Hongta and Eshan) and six counties (Chengjiang, Tonghai, Jiangchuan, Huaning, Xinping and Yuanjiang) has been one of the most severely endemic areas of paratyphoid fever in China since 1999. There was a progressive increase in the number of SPA cases in Yuxi between 2005 and 2009. The results of MLVA typing for Yuxi isolates indicate that the MLVA13 isolate was the epidemic clone in Yuxi in outbreaks and sporadic cases. Consistent with the sources and transmission routes of enteric fever (32,33), contaminated water and food are major sources of SPA in Yuxi. It was identified that contaminated well water in a vegetable market of Hongta was the direct factor leading to the 2007 outbreak of SPA. More than 90% of patients in the 2007 outbreak were retrospectively investigated to have purchased vegetables from the Hongta vegetable market near the infected well, where the vendors watered the vegetables using the well water. Subsequently, the SPA isolates were separated from the water in the well. The sources of SPA from the well water were further confirmed by the result of MLVA typing for SPA in the present study, demonstrating that MLVA13 SPA were the major clones isolated from the well water, vegetables and patients during the epidemic. In Asia, SPA may also be transmitted by consumption of contaminated foods from street vendors (4). The contaminated foods sold by street vendors may be important vectors of the SPA sporadic isolates in Yuxi as it is highly common in Yuxi to eat at street vendors with poor sanitary conditions. From the patients who had eaten at street vendors, a variety of MLVA types were separated with the majority being the MLVA13 type of SPA. The incidence rate of enteric fever has decreased significantly and remained at a low level following 2010 with the strengthened surveillance of stock sold in the markets and by street vendors.
A total of 20 SPA isolates have been collected from different countries and regions during different periods by the CMCC thus far. To investigate the Yuxi SPA isolates, the MLVA type of 20 CMCC SPA strains was analyzed, and revealed a large diversity with 14 MLVA types which are unrelated to the 195 Yuxi isolates.
Acknowledgements
This study was supported by grants from the Foundation of Provincial Education Department of Hunan Province, China (grant no. 05B046), and the Cooperative Foundation of Yuxi Science and Technology Committee, Yunnan, China (grant no. HZ200706).
References
- 1.Smith KP, George J, Cadle KM, Kumar S, Aragon SJ, Hernandez RL, et al. Elucidation of antimicrobial susceptibility profiles and genotyping of Salmonella enterica isolates from clinical cases of Salmonellosis in New Mexico in 2008. World J Microbiol Biotechnol. 2010;26:1025–1031. doi: 10.1007/s11274-009-0265-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Maskey AP, Day JN, Phung QT, Thwaites GE, Campbell JI, Zimmerman M, et al. Salmonella enterica serovar Paratyphi A and S. enterica serovar Typhi cause indistinguishable clinical syndromes in Kathmandu, Nepal. Clin Infect Dis. 2006;42:1247–1253. doi: 10.1086/503033. [DOI] [PubMed] [Google Scholar]
- 3.Sheikh A, Charles RC, Rollins SM, Harris JB, Bhuiyan MS, Khanam F, et al. Analysis of Salmonella enterica serotype paratyphi A gene expression in the blood of bacteremic patients in Bangladesh. PLoS Negl Trop Dis. 2010;4:e908. doi: 10.1371/journal.pntd.0000908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fangtham M, Wilde H. Emergence of Salmonella paratyphi A as a major cause of enteric fever: need for early detection, preventive measures, and effective vaccines. J Travel Med. 2008;15:344–350. doi: 10.1111/j.1708-8305.2008.00237.x. [DOI] [PubMed] [Google Scholar]
- 5.Dong BQ, Yang J, Wang XY, Gong J, von Seidlein L, Wang ML, et al. Trends and disease burden of enteric fever in Guangxi province, China, 1994–2004. Bull World Health Organ. 2010;88:689–669. doi: 10.2471/BLT.09.069310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang SK, Chu CJ, Sun PS, Shan DS, Kong FL, Liu HY, et al. Study on blood cultures and bacteria counts in the blood of paratyphoid fever A patients. Eur J Clin Microbiol Infect Dis. 2009;28:1259–1261. doi: 10.1007/s10096-009-0766-9. [DOI] [PubMed] [Google Scholar]
- 7.Gaind R, Paglietti B, Murgia M, Dawar R, Uzzau S, Cappuccinelli P, et al. Molecular characterization of ciprofloxacin-resistant Salmonella enterica serovar Typhi and Paratyphi A causing enteric fever in India. J Antimicrob Chemother. 2006;58:1139–1144. doi: 10.1093/jac/dkl391. [DOI] [PubMed] [Google Scholar]
- 8.Gal-Mor O, Suez J, Elhadad D, Porwollik S, Leshem E, Valinsky L, et al. Molecular and cellular characterization of a Salmonella enterica serovar Paratyphi A outbreak strain and the human immune response to infection. Clin Vaccine Immunol. 2012;19:146–156. doi: 10.1128/CVI.05468-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Elberse KE, Nunes S, Sá-Leão R, van der Heide HG, Schouls LM. Multiple-locus variable number tandem repeat analysis for Streptococcus pneumoniae: comparison with PFGE and MLST. PLoS One. 2011;6:e19668. doi: 10.1371/journal.pone.0019668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tien YY, Ushijima H, Mizuguchi M, Liang SY, Chiou CS. Use of multilocus variable-number tandem repeat analysis in molecular subtyping of Salmonella enterica serovar Typhi isolates. J Med Microbiol. 2012;61:223–232. doi: 10.1099/jmm.0.037291-0. [DOI] [PubMed] [Google Scholar]
- 11.U’Ren JM, Schupp JM, Pearson T, Hornstra H, Friedman CL, Smith KL, et al. Tandem repeat regions within the Burkholderia pseudomallei genome and their application for high resolution genotyping. BMC Microbiol. 2007;7:23. doi: 10.1186/1471-2180-7-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Heck M. Multilocus variable number of tandem repeats analysis (MLVA) - a reliable tool for rapid investigation of Salmonella typhimurium outbreaks. Euro Surveill. 2009:14. pii: 19177. [PubMed] [Google Scholar]
- 13.Kruy SL, van Cuyck H, Koeck JL. Multilocus variable number tandem repeat analysis for Salmonella enterica subspecies. Eur J Clin Microbiol Infect Dis. 2011;30:465–473. doi: 10.1007/s10096-010-1110-0. [DOI] [PubMed] [Google Scholar]
- 14.Tien YY, Wang YW, Tung SK, Liang SY, Chiou CS. Comparison of multilocus variable-number tandem repeat analysis and pulsed-field gel electrophoresis in molecular subtyping of Salmonella enterica serovars Paratyphi A. Diagn Microbiol Infect Dis. 2011;69:1–6. doi: 10.1016/j.diagmicrobio.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 15.McClelland M, Sanderson KE, Clifton SW, Latreille P, Porwollik S, Sabo A, et al. Comparison of genome degradation in Paratyphi A and Typhi, human-restricted serovars of Salmonella enterica that cause typhoid. Nat Genet. 2004;36:1268–1274. doi: 10.1038/ng1470. [DOI] [PubMed] [Google Scholar]
- 16.Killgore GE, Kato H. Use of arbitrary primer PCR to type Clostridium difficile and comparison of results with those by immunoblot typing. J Clin Microbiol. 1994;32:1591–1603. doi: 10.1128/jcm.32.6.1591-1593.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Queipo-Ortuño MI, De Dios Colmenero J, Macias M, Bravo MJ, Morata P. Preparation of bacterial DNA template by boiling and effect of immunoglobulin G as an inhibitor in real-time PCR for serum samples from patients with brucellosis. Clin Vaccine Immunol. 2008;15:293–296. doi: 10.1128/CVI.00270-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jiang Y, Liu HC, Zheng HJ, Tang B, Dou XF, Zhao XQ, et al. Evaluation of four candidate VNTR Loci for genotyping 225 Chinese clinical Mycobacterium tuberculosis complex strains. Biomed Environ Sci. 2012;25:82–90. doi: 10.3967/0895-3988.2012.01.012. [DOI] [PubMed] [Google Scholar]
- 19.Benson G. Tandem repeats finder: a program to analyze DNA sequences. Nucleic Acids Res. 1999;27:573–580. doi: 10.1093/nar/27.2.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Haguenoer E, Baty G, Pourcel C, Lartigue MF, Domelier AS, Rosenau A, et al. A multi locus variable number of tandem repeat analysis (MLVA) scheme for Streptococcus agalactiae genotyping. BMC Microbiol. 2011;11:171. doi: 10.1186/1471-2180-11-171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mouton L, Nong G, Preston JF, Ebert D. Variable-number tandem repeats as molecular markers for biotypes of Pasteuria ramosa in Daphnia spp. Appl Environ Microbiol. 2007;73:3715–3718. doi: 10.1128/AEM.02398-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.O’Dushlaine CT, Shields DC. Tools for the identification of variable and potentially variable tandem repeats. BMC Genomics. 2007;7:290. doi: 10.1186/1471-2164-7-290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ramisse V, Houssu P, Hernandez E, Denoeud F, Hilaire V, Lisanti O, et al. Variable number of tandem repeats in Salmonella enterica subsp. enterica for typing purposes. J Clin Microbiol. 2004;42:5722–5730. doi: 10.1128/JCM.42.12.5722-5730.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu Y, Lee MA, Ooi EE, Mavis Y, Tan AL, Quek HH. Molecular typing of Salmonella enterica serovar typhi isolates from various countries in Asia by a multiplex PCR assay on variable-number tandem repeats. J Clin Microbiol. 2003;41:4388–4394. doi: 10.1128/JCM.41.9.4388-4394.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guo C, Liao Y, Li Y, Duan J, Guo Y, Wu Y, et al. Genotyping analysis of Helicobacter pylori using multiple-locus variable-number tandem-repeats analysis in five regions of China and Japan. BMC Microbiol. 2011;11:197. doi: 10.1186/1471-2180-11-197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yazdankhah SP, Lindstedt BA. Variable number tandem repeat typing of bacteria. Methods Mol Biol. 2007;396:395–405. doi: 10.1007/978-1-59745-515-2_25. [DOI] [PubMed] [Google Scholar]
- 27.Lavania M, Katoch K, Sharma R, Sharma P, Das R, Gupta AK, et al. Molecular typing of Mycobacterium leprae strains from northern India using short tandem repeats. Indian J Med Res. 2011;133:618–626. [PMC free article] [PubMed] [Google Scholar]
- 28.Witonski D, Stefanova R, Ranganathan A, Schutze GE, Eisenach KD, Cave MD. Variable-number tandem repeats that are useful in genotyping isolates of Salmonella enterica subsp. enterica serovars Typhimurium and Newport. J Clin Microbiol. 2006;44:3849–3854. doi: 10.1128/JCM.00469-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nielsen EM, Engberg J, Fussing V, Petersen L, Brogren CH, On SL. Evaluation of phenotypic and genotypic methods for subtyping Campylobacter jejuni isolates from humans, poultry, and cattle. J Clin Microbiol. 2000;38:3800–3810. doi: 10.1128/jcm.38.10.3800-3810.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Harbottle H, White DG, McDermott PF, Walker RD, Zhao S. Comparison of multilocus sequence typing, pulsed-field gel electrophoresis, and antimicrobial susceptibility typing for characterization of Salmonella enterica serotype Newport isolates. J Clin Microbiol. 2006;44:2449–2457. doi: 10.1128/JCM.00019-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Montesinos I, Salido E, Delgado T, Cuervo M, Sierra A. Epidemiologic genotyping of methicillin-resistant Staphylococcus aureus by pulsed-field gel electrophoresis at a university hospital and comparison with antibiotyping and protein A and coagulase gene polymorphisms. J Clin Microbiol. 2002;40:2119–2125. doi: 10.1128/JCM.40.6.2119-2125.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Korbsrisate S, Sarasombath S, Janyapoon K, Ekpo P, Pongsunk S. Immunological detection of Salmonella paratyphi A in raw prawns. Appl Environ Microbiol. 1994;60:4612–4613. doi: 10.1128/aem.60.12.4612-4613.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Basnyat B, Maskey AP, Zimmerman MD, Murdoch DR. Enteric (typhoid) fever in travelers. Clin Infect Dis. 2005;41:1467–1472. doi: 10.1086/497136. [DOI] [PubMed] [Google Scholar]