Abstract
Supplemental oxygen used to treat infants born prematurely disrupts angiogenesis and is a risk factor for persistent pulmonary disease later in life. Although it is unclear how neonatal oxygen affects development of the respiratory epithelium, alveolar simplification and depletion of type II cells has been observed in adult mice exposed to hyperoxia between postnatal Days 0 and 4. Because hyperoxia inhibits cell proliferation, we hypothesized that it depleted the adult lung of type II cells by inhibiting their proliferation at birth. Newborn mice were exposed to room air (RA) or hyperoxia, and the oxygen-exposed mice were recovered in RA. Hyperoxia stimulated mRNA expressed by type II (Sftpc, Abca3) and type I (T1α, Aquaporin 5) cells and inhibited Pecam expressed by endothelial cells. 5-Bromo-2'-deoxyuridine labeling and fate mapping with enhanced green fluorescence protein controlled statically by the Sftpc promoter or conditionally by the Scgb1a1 promoter revealed increased Sftpc and Abca3 mRNA seen on Day 4 reflected an increase in expansion of type II cells shortly after birth. When mice were returned to RA, this expanded population of type II cells was slowly depleted until few were detected by 8 weeks. These findings reveal that hyperoxia stimulates alveolar epithelial cell expansion when it disrupts angiogenesis. The loss of type II cells during recovery in RA may contribute to persistent pulmonary diseases such as those reported in children born preterm who were exposed to supplemental oxygen.
Keywords: bronchopulmonary dysplasia, neonatal hyperoxia, persistent pulmonary disease, proliferation, type II epithelial cells
Clinical Relevance
It is unclear how supplemental oxygen used to treat preterm infants causes long-lasting changes in lung development and function later in life. This study demonstrates that neonatal hyperoxia stimulates expansion of type II cells when it disrupts angiogenesis and that expanded type II cells are slowly depleted during recovery in room air. The depletion of type II cells observed in adult mice exposed to neonatal hyperoxia may explain why persistent pulmonary disease is seen in children born preterm.
Supplemental oxygen used to treat preterm infants in respiratory distress is a major risk factor for bronchopulmonary dysplasia (BPD), a chronic lung disease seen in infants needing oxygen therapy beyond 36 weeks postconceptual age (1). Although the use of milder ventilation strategies, exogenous surfactant, and antenatal steroids has reduced the incidence of airway inflammation and fibrosis, infants dying from BPD have simplified alveoli that are less vascularized (2). Likewise, alveolar simplification and disorganized vasculature is seen in preterm baboons (3, 4) and in term rodents exposed to hyperoxia at birth (5, 6). Oxidative stress caused by high oxygen exposure or by the inflammatory response to oxygen-induced cell injury promotes alveolar simplification by suppressing expression of angiogenic factors such as vascular endothelial cell growth factor (VEGF) (for review, see Ref. 7). Hyperoxia also reduces the number of circulating endothelial cell precursors in blood and lung of newborn mice (8). Hence, early-life exposure to high oxygen inhibits cell proliferation and angiogenesis, resulting in altered postnatal lung development.
Although advances in the care of neonates have markedly reduced infant mortality, survivors often exhibit nonatopic airway wheezing (9, 10), and many are rehospitalized when infected with respiratory syncytial virus (11, 12). High blood pressure has also been observed in young adolescents who were born preterm (13). It is not known whether some or all of these changes persist into adulthood. However, grossly simplified alveoli have been observed in a former preterm child who died of an acute asthma attack, suggesting that alveolar simplification may persist or worsen into childhood (14). Because lung tissues of former preterm infants are rare, investigators have increasingly relied on animal models to understand how the developing lung repairs after oxygen-induced injury. Similar to the lung function changes reported in humans, long-term changes in airway responsiveness, lung compliance, and alveolar simplification occur in a variety of animals exposed to high oxygen as neonates (15–19). We previously reported that 8-week-old adult mice exposed to ≥ 60% oxygen between postnatal day (pnd)0 and pnd4 have enlarged alveoli attributed in part to increased elastin expression and an imbalance in alveolar epithelial type I and II cells (15, 20, 21). Although vascular changes were not observed at this age, microvessel pruning and rarefaction, cardiac hypertrophy, and reduced survival were observed by 1 year of age (22). Analogous to children born prematurely, young adult mice exposed to hyperoxia as neonates also exhibit an altered host response to influenza A virus infection (21, 23, 24). Because this model of neonatal hyperoxia phenotypically recapitulates human diseases attributed to prematurity, it may provide an opportunity to understand how a high-oxygen environment at birth permanently alters lung development.
It is widely accepted that neonatal hyperoxia promotes alveolar simplification and BPD-like pathology by inhibiting cell proliferation and impairing angiogenesis (7). Although it remains unclear how hyperoxia affects alveolar epithelial development, type II cell hyperplasia has been observed in preterm baboons and adult rats exposed to sublethal levels of oxygen (4, 25). Type II cells expressing high levels of Sftpc mRNA have also been observed lining the alveolar wall of human infants with BPD (2). On the other hand, adult mice exposed to neonatal hyperoxia have fewer type II cells, as defined by a reduction in the number of cells expressing prosurfactant protein C (proSP-C) or by reduced expression of enhanced green fluorescent protein (EGFP), in a small subpopulation of type II cells (15, 20). In addition, the proportion of type I cells may be greater in these mice because they express higher levels of T1α and aquaporin 5 (Aqp5). Here, transgenic mice that statically or conditionally express EGFP in type II cells are used to investigate how oxygen affects development of the alveolar epithelium.
Materials and Methods
Additional details for these procedures are provided in the online supplement.
Mice and Oxygen Exposure
Sftpc-EGFP mice statically express EGFP under control of the 3.7-kb human surfactant protein C (Sftpc) promoter (26). Rat Scgb1a1-rtTA (secretoglobin, family 1A, member 1 or uteroglobin) and (otet)7CMV-cre bitransgenic mice were mated to transgenic mT/mG (Jackson Laboratories, Bar Harbor, ME) mice, which contain a floxed membrane-targeted tomato red (mT) cassette followed by membrane-targeted EGFP, both controlled by a CMV enhancer/chicken β-actin core promoter. Administration of doxycycline to adult mice activates reporter genes in airway cells and in approximately 50% of alveolar epithelial type II cells (27). Administration of doxycline to pregnant dams activates reporter genes in airway and in precursor cells or type II cells that expand postnatally. The tomato red cassette was deleted by adding doxycycline (600 mg/kg) to the chow of pregnant dams between embryonic day (E)14.5 and 18.5 or to adult mice for 48 hours. Newborn mice were exposed to room air (RA) or hyperoxia as previously described (20). Unless specifically defined, hyperoxia represents 100% oxygen. Some mice exposed to high oxygen were returned to RA on pnd4 and analyzed when they were 8 to 10 weeks old. Mice were injected with 5-bromo-2′-deoxyuridine (BrdU) 2 hours before death (15). The University Committee on Animal Resources at the University of Rochester approved the use of mice for these studies.
Histology and Immunohistochemistry
Lungs were inflation fixed with 10% neutral-buffered formalin, embedded in paraffin, and sectioned (15). Tissue sections were stained with antibodies against proSP-C and ATP-binding cassette, subfamily A, member 3 (ABCA3) (Seven Hills Bioreagents, Cincinnati, OH), T1α (Developmental Studies Hybridoma Bank at the University of Iowa, Iowa City, IA), EGFP and BrdU (Abcam, Cambridge, MA), or thyroid transcription factor (TTF)-1 (Dako, Carpinteria, CA) (15, 20).
Real-Time PCR Analysis
RNA was isolated from the right lung lobe and reverse transcribed using the iScript cDNA synthesis kit (Bio-Rad Laboratories, Hercules, CA). cDNA was then amplified with SYBR Green I dye on theCFX96 Touch and CFX384 Touch Real-Time PCR detection system (Bio-Rad Laboratories). PCR products were amplified with sequence-specific primers, or 18S rRNA was used to normalize equal loading of the template cDNAs (Table 1).
Table 1:
RT-PCR Oligonucleotide Sequences
| Genes | Primers | GenBank Accession Number | Product Size (bp) |
|---|---|---|---|
| Sftpc | F: 5′ TGATGGAGAGTCCACCGGATTA 3′; R: 5′ CCTACAATCACCACGACAACGA 3′ | NM_011359.2 | 130 |
| Abca3 | F: 5′ GAGGGTCGGTGCCAGCACAT 3′; R: 5′ GTCGCCTGGCGTCAGCAGTT 3′ | NM_001039581.1 | 158 |
| T1α | F: 5′ AGCAAAGCCAAGACAGTATCGC 3′; R: 5′ TTAGGACTGGGCTGGAATGTGT 3′ | NM_010329.2 | 180 |
| Aqp5 | F: 5′ AGATCTCCATAGCCTTTGGCCT 3′; R: 5′ AGCAGAGAGATCTGGTTGCCTA 3′ | NM_009701.4 | 124 |
| Ttf-1 | F:5′TCCAGCCTATCCCATCTGAACT 3′; R: 5′ CAAGCGCATCTCACGTCTCA 3′ | NM_001146198.1 | 101 |
| Pecam | F:5′ AGCCTCACCAAGCTCTGGGAAC 3′; R: 5′ CGGGACATGGACGACCTCCCA 3′ | NM_008816.2 | 144 |
| 18s | F:5′ CGGCTACCACATCCAAGGAA 3′; R: 5′ GCTGGAATTACCGCGGCT 3′ | NR_003278.1 | 187 |
Definition of abbreviations: F, forward primer; R, reverse primer.
Western Blot Analysis
Frozen mouse lung tissues were homogenized in lysis buffer, and supernatants were collected for Western blotting as previously described (20). Western blot membranes were immunoblotted with rabbit anti–proSP-C, hamster anti-T1α, goat antiplatelet endothelial cell adhesion molecule (PECAM, also called CD31) antibody (Santa Cruz Biotechnology, Santa Cruz, CA), mouse anti-EGFP (Clontech, Palo Alto, CA), or anti–β-actin antibody (Sigma, St. Louis, MO). Immune complexes were detected with horseradish peroxidase–conjugated secondary antibody (SouthernBiotech, Birmingham, AL) and visualized with enhanced chemiluminescence (ECL kit; GE Healthcare Life Sciences, Piscataway, NJ) according to the manufacturer’s protocol.
Statistical Analyses
Values are expressed as means ± SE. Group means were compared by ANOVA followed by a Fisher least significant difference post hoc test. An unpaired t test was used to make comparisons between two treatment groups at a specific point in time. All data were analyzed using StatView software (SAS Institute, Cary, NC), and the means were considered significantly different when P < 0.05.
Results
Neonatal Hyperoxia Alters Expression of Alveolar Genes
Newborn mice were exposed to RA or hyperoxia between birth and pnd4, pnd7, and pnd10. Lung homogenates were immunoblotted for proSP-C, T1α, and PECAM, using β-actin as a loading control. Hyperoxia inhibited proSP-C expression on pnd4, had little effect on pnd7, and stimulated expression on pnd10 (Figure 1A). In contrast, hyperoxia stimulated T1α expression and inhibited expression of PECAM at these times. Changes in proSP-C and T1α expression were confirmed by immunohistochemistry. Hyperoxia reduced the number of proSP-C–positive cells on pnd4 and increased it on pnd10 (Figure 1B). These changes were not specific for proSP-C because they were also observed with ABCA3, a lipid transport protein also expressed exclusively in the lung by type II cells. In contrast to the changes observed in proSP-C and ABCA3, hyperoxia increased T1α staining in squamous alveolar cells, consistent with the morphology of type I cells.
Figure 1.
Neonatal hyperoxia differentially affects alveolar epithelial protein expression. Newborn mice were exposed to room air (RA) or hyperoxia (100% oxygen [O2]) through postnatal days (pnd)4, pnd7, and pnd10. (A) Lung homogenates were immunoblotted with antibodies against prosurfactant protein C (proSP-C), T1α, and platelet endothelial cell adhesion molecule (PECAM), with β-actin used as a loading control. Band intensities in mice exposed to RA (white bar) and hyperoxia (black bar) were graphed as mean ± SE fold over RA (n = 3–4 mice per group). *P < 0.05, **P < 0.01, and ***P < 0.001 when compared with RA. (B) Representative images of pnd4 and pnd10 lungs immunostained with antibody against proSP-C or ABCA3 (red), T1α (green), and 4',6-diamidino-2-phenylindole (DAPI) (blue). Scale bar = 50 μm.
Changes in protein expression did not always correlate with the expression of their corresponding mRNAs. Although hyperoxia initially suppressed and then stimulated expression of proSP-C and ABCA3 protein, it increased expression of Sftpc and Abca3 mRNA on pnd4 and pnd10 (Figure 2). It also stimulated mRNA expression of the type I cell–specific genes T1α and aquaporin (Aqp) 5. In contrast, hyperoxia inhibited mRNA expression of Pecam, which is consistent with its inhibitory effects on angiogenesis (7).
Figure 2.
Neonatal hyperoxia stimulates alveolar epithelial mRNA expression. Newborn mice were exposed to RA or O2 through pnd4, pnd7, and pnd10. The mRNA expression of Sftpc, Abca3, T1α, Aqp5, Pecam, and 18S ribosomal RNA as a loading control were determined by quantitative RT-PCR. The mRNA expression in mice exposed to RA (white bar) and hyperoxia (black bar) were graphed as mean ± SE fold change over RA (n = 4 mice per group). *P < 0.05 and **P < 0.01 when compared with RA.
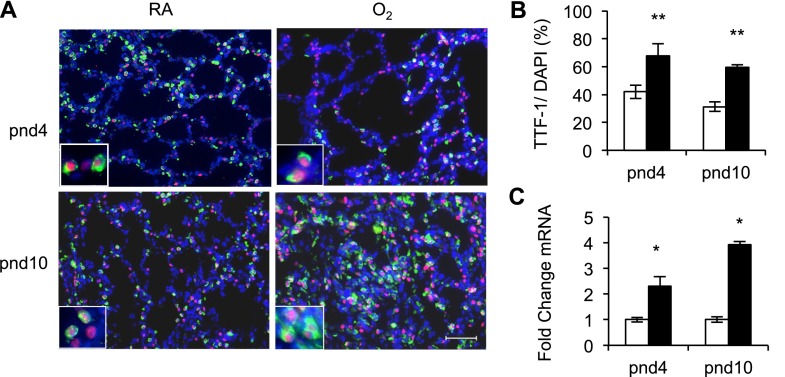
The opposing effects of hyperoxia on proteins and mRNA expressed by type II cells on pnd4 were reconciled by studying expression of TTF-1 (also called Nkx2.1), which stimulates transcription of Sftpc and Abca3 and is expressed by type II but not type I cells (28). Neonatal hyperoxia stimulated the number of alveolar cells expressing TTF-1 on pnd4 and pnd10 (Figures 3A and 3B). TTF-1 was detected in approximately 40% of alveolar cells on pnd4 in mice exposed to RA, as opposed to 70% of alveolar cells in siblings exposed to hyperoxia. At this time, many alveolar cells expressed TTF-1 but not proSP-C. By pnd10, TTF-1 was detected in approximately 30% of alveolar cells from mice exposed to RA and in 60% of alveolar cells from mice exposed to hyperoxia. At this time, most alveolar cells expressing TTF-1 also expressed proSP-C. The increased expression of TTF-1 protein seen on pnd4 and pnd10 correlated with increased expression of Ttf-1 mRNA (Figure 3C). The oxygen-dependent increase in TTF-1 seen on pnd4 suggested that the loss of proSP-C and ABCA3 represented loss of gene expression and not loss of type II cells.
Figure 3.
Neonatal hyperoxia increases the number cells expressing thyroid transcription factor (TTF)-1. Newborn mice were exposed to RA or O2 through pnd4 and pnd10. (A) Lungs were immunostained with antibody against TTF-1 (red) and Sftpc (green) and were counterstained with 4',6-diamidino-2-phenylindole (DAPI) (blue). Scale bar = 50 μm. (B) The percentage of TTF-1 to DAPI–positive cells in mice exposed to RA (white bar) and hyperoxia (black bar) was graphed as mean ± SE fold change over RA (n = 5 mice per group). **P < 0.01 when compared with RA. (C) The mRNA expression of Ttf-1 and 18S ribosomal RNA as a loading control was determined by quantitative RT-PCR. The mRNA expression in mice exposed to RA (white bar) and hyperoxia (black bar) was graphed as mean ± SE fold change over RA (n = 5 mice per group). *P < 0.05 when compared with RA.
Neonatal Hyperoxia Stimulates Expansion of Type II Cells
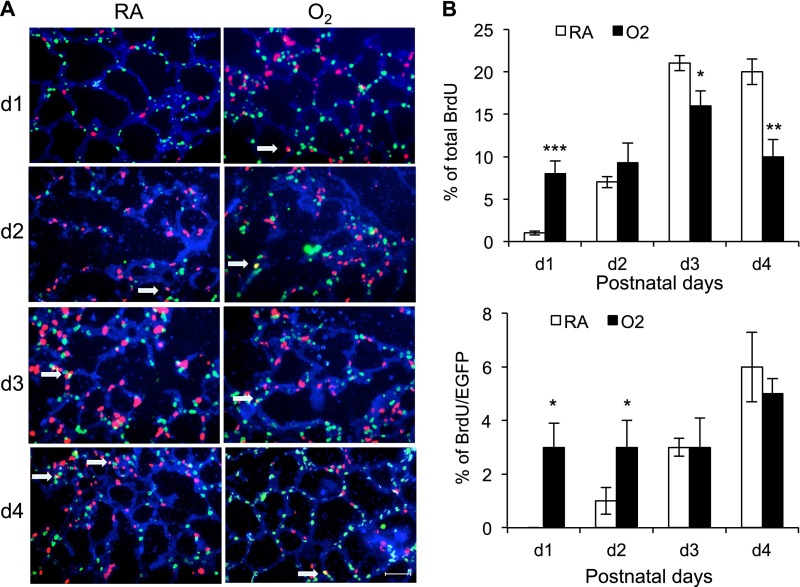
It was difficult to use proSP-C or ABCA3 as markers of type II cells when their expression declined in mice exposed to hyperoxia. We therefore decided to study proliferation of type II cells using transgenic mice expressing EGFP under control of the human Sftpc promoter. EGFP provided a surrogate marker for type II cells because it is not secreted and is resistant to proteasome degradation (29). Sftpc-EGFP mice were exposed to RA or hyperoxia between pnd0 and pnd4. Mice were injected with BrdU on pnd1, pnd2, pnd3, and pnd4. Lungs were harvested 2 hours later and stained for EGFP and BrdU (Figure 4A). In mice exposed to RA, the number of BrdU-positive alveolar cells increased from 1% on pnd1 to approximately 20% on pnd3 and pnd4 (Figure 4B). In contrast to the low 1% labeling index seen on pnd1, approximately 8% of alveolar cells were BrdU positive after 1 day of hyperoxia. It nearly doubled by pnd3 and then declined to approximately 10% by pnd4. To determine whether these were type II cells, BrdU was colocalized to EGFP. In mice exposed to RA, proliferating type II cells were rarely detected on pnd1, occasionally detected on pnd2 and pnd3, and represented 5% of the population by pnd4. In mice exposed to hyperoxia, approximately 3% of type II cells were proliferating on pnd1, pnd2, and pnd3 and then doubled to 6% on pnd4. To confirm hyperoxia stimulates proliferation of type II cells on pnd1, BrdU was colocalized to proSP-C. Although BrdU-positive cells were rare in mice exposed to RA, they were readily detected in mice exposed to hyperoxia and often detected in proSP-C–positive cells (see Figure E1 in the online supplement). These findings confirm older studies showing that proliferation of type II cells is very low at birth and increases postnatally (30, 31). It extends that knowledge by showing how hyperoxia stimulates proliferation of type II cells shortly after birth.
Figure 4.
Neonatal hyperoxia stimulates proliferation of type II cells. Newborn Sftpc-EGFP mice were exposed to RA or O2. (A) Lungs were harvested daily through pnd4 and immunostained with antibody against BrdU (red) and enhanced green fluorescence protein (EGFP) (green) followed by counterstaining with DAPI. (B) The percentage of cells expressing BrdU and BrdU/EGFP on each day was determined and graphed as mean ± SE (n = 5 mice per group). *P < 0.05, **P < 0.01, and ***P < 0.001 when compared with RA on the same day. Scale bar, 30 μm.
The postnatal expansion of type II cells was also mapped by conditionally activating EGFP in precursors of type II cells before birth. Bitransgenic Scgb1a1-rtTA X (otet)7CMV-Cre mice were mated to mT/mG mice that express a floxed myristoylated tomato red (mT) cassette followed a myristoylated EGFP (mG) cassette (Figure E2). Administration of doxycycline to the chow of pregnant dams deletes the tomato red gene, thereby permitting expression of EGFP in airway Clara and alveolar cells that expand postnatally to produce alveolar type II cells (32). To confirm neonatal hyperoxia stimulates expansion of type II cells, doxycycline was administered to pregnant dams containing triple transgenic Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG fetuses between E14.5 and E18.5 (Figure 5A). Doxycycline was removed from the chow at birth and the newborn pups were exposed to RA or hyperoxia. Triple transgenic mice treated with doxycycline and exposed to hyperoxia frequently died after pnd4 through mechanisms that we have yet to discern. However, EGFP was readily detected on pnd4 in the airway of mice exposed to RA and hyperoxia (Figures 5B and 5C). Although EGFP was also detected in some alveolar cells of mice exposed to RA, it was readily detected throughout the alveoli of mice exposed to 100% O2. Control experiments confirmed that hyperoxia did not stimulate expression of the mT/mG reporter (Figure E2) and that it did not activate excision of mT red in the absence of doxycycline. To confirm that the expansion of type II cells was not related to their morbidity, the study was repeated with 60% oxygen, which is still sufficient to disrupt lung development (20, 33). Similar to the effects seen with 100% oxygen, EGFP was readily detected in alveolar cells of mice exposed to 60% oxygen (Figures 5B and 5C). None of the mice died when exposed to this lower level of oxygen.
Figure 5.
Neonatal hyperoxia stimulates lineage expansion of type II cells. (A) Cartoon model depicting administration of doxycycline to triple transgenic Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG mice doxycycline between embryonic day (E)14.5 and E18.5 followed by exposure to RA or hyperoxia between birth and pnd4. (B) The percentage of EGFP to DAPI-positive cells in mice exposed to RA (white bar), 60% oxygen (gray bar), or 100% oxygen (black bar) was quantified and graphed as mean ± SE (n = 5 mice per group). **P < 0.01 when compared with RA. (C) Representative images of EGFP (green) and DAPI (blue) staining in lung tissues of mice exposed to RA, 60% oxygen, or 100% oxygen. Arrows point to airway. Scale bar, 200 μm. Dox, doxycycline.
Expanded Type II Cells Are Pruned during Recovery in RA
Because adult mice exposed to hyperoxia as neonates have fewer type II cells (15), we hypothesized that the expanded type II cells seen on pnd4 must be depleted when mice are recovered in RA. To test this, EGFP expression was evaluated in adult Sftpc-EGFP mice that had been exposed to RA or hyperoxia (100% oxygen) between pnd0 and pnd4. Approximately 25% of alveolar cells in adult mice exposed to RA as neonates expressed EGFP (Figure 6A). In contrast, only 15% of alveolar cells in adult mice exposed to hyperoxia as neonates expressed EGFP. Western blot analysis confirmed that adult mice exposed to hyperoxia as neonates express less EGFP than siblings exposed to RA as neonates (Figure 6B). Similarly, expanded EGFP-positive alveolar cells in Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG transgenic mice exposed to 60% oxygen at birth were depleted when the mice were recovered in RA (Figure 6C). Western blot analysis confirmed that exposure to 60% oxygen was sufficient to reduce proSP-C expression in adult mice (Figure 6D). Reduced expression of proSP-C did not always correlate with increased expression of T1α (Figure 6D). To further define when type II cells were lost, alveolar expression of EGFP was investigated weekly. Although the number of EGFP-positive alveolar cells progressively increased in mice exposed to RA between pnd7 and pnd35, it progressively decreased in mice recovered from hyperoxia, with the greatest change occurring between pnd28 and pnd35 (Figure E3). Some Tunel-positive cells were consistently detected in mice recovered from hyperoxia but only on pnd7 and pnd14 from mice exposed to RA.
Figure 6.
Expanded type II cells are depleted when mice are recovered in RA. (A) Lungs of Sftpc-EGFP mice exposed to RA or 100% O2 between birth and pnd4 were stained with antibody against EGFP (green), Sftpc (red), and DAPI (blue). Note that virtually all EGFP cells express Sftpc. The percentage of EGFP to DAPI-positive cells was quantified in mice exposed to RA (white bar) or hyperoxia (closed bar) and graphed as mean ± SE fold change over RA (n = 4 mice per group). *P < 0.05 when compared with RA. (B) Lung homogenates were immunoblotted for EGFP and β-actin. Band intensities in mice exposed to RA and hyperoxia were quantified and graphed as mean ± SE fold (n = 4 mice per group). *P < 0.05 when compared with RA. (C) Lungs of Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG mice exposed to RA or 60% O2 between birth and pnd4 were stained with antibody against EGFP (green) and DAPI (blue). The percentage of EGFP to DAPI-positive cells was quantified in mice exposed to RA or hyperoxia and graphed as mean ± SE fold change over RA (n = 4 mice per group). **P < 0.01 when compared with RA. (D) Lung homogenates were immunoblotted with antibody against proSP-C, T1α, and β-actin. Band intensities in mice exposed to RA (white bar) and hyperoxia (black bar) were quantified and graphed as mean ± SE fold (n = 4 RA and n = 5 hyperoxia mice). *P < 0.compared with RA. Scale bar, 50 μm.
Because expression of the reverse transcriptional transactivator (rtTA) used to induce Cre expression can cause lung emphysema (34), we confirmed that the oxygen-dependent loss of type II cells was unrelated to the expression of rtTA or the sensitivity of the triple transgenic mice to hyperoxia by activating the reporter after mice were exposed to hyperoxia. Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG mice were exposed to RA or hyperoxia between pnd0 and pnd4, and then the oxygen-exposed mice were recovered in RA until they were 8 weeks old. All of the mice survived. Doxycycline was administered to mice for 48 hours and then removed for 5 days, and the expression of EGFP and proSP-C was evaluated in lung tissue sections (Figure 7A). EGFP was only detected in cuboidal alveolar cells of mice administered doxycycline (Figure 7B). Neonatal hyperoxia significantly reduced the number of proSP-C–positive cells by 50% (Figure 7B, upper graph). EGFP was detected in approximately 50% of type II cells defined by dual expression of EGFP and proSP-C (Figure 7B, lower graph). Neonatal hyperoxia also reduced this population of type II cells by 50%.
Figure 7.
Type II cells are depleted in adult mice exposed to neonatal hyperoxia. (A) Cartoon model showing how Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG mice were exposed to RA or 100% O2 between pnd0 and pnd4 and then administered doxycycline as adults. (B) Lungs of adult Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG mice exposed to RA or O2 as neonates and vehicle or doxycycline as adults were stained with antibody against proSP-C (red), EGFP (green), and DAPI (blue). The percentage of proSP-C to DAPI-positive cells and the percentage of EGFP to proSP-C–positive cells in mice exposed to RA or hyperoxia were graphed as mean ± SE fold (n = 5 mice per group). *P < 0.05 and **P < 0.01 when compared with RA. Scale bar, 50 μm. Dox, doxycycline.
Discussion
Supplemental oxygen is often used to treat infants born prematurely even though it can adversely affect lung development and overall health later in life. It is widely believed that exposure to high levels of oxygen disrupt lung development by concomitantly inhibiting cell proliferation and angiogenesis, resulting in a simplified lung. Here, using an established mouse model of persistent pulmonary disease, we provide evidence that neonatal hyperoxia rapidly stimulate expansion of alveolar type II cells that normally expand slowly in mice birthed into RA (Figure 8). The rapidly expanded population of cells is then depleted during recovery in RA, resulting in an adult lung with proportionally fewer type II cells than those of siblings exposed to RA. Type II cells present in adult lung exposed to neonatal hyperoxia may therefore represent the population of type II cells present at birth. Because type II cells are selectively lost in human emphysema (35), their loss may contribute to the alveolar simplification seen in adult mice exposed to hyperoxia as neonates (20). Type II cells also express innate immune genes (36) and act as progenitor cells after injury (37), which may explain why the mice are also sensitive to influenza A virus infection and bleomycin-induced lung fibrosis (23, 38, 39). Hence, persistent pulmonary diseases attributed to prematurity, and more specifically early life exposure to oxygen, may be related to overexpansion of type II cells at birth that are depleted over time.
Figure 8.
Cartoon model depicting the expansion of type II cells during exposure to hyperoxia followed by pruning during recovery in RA. Because type II cells are defenders of the alveolus, their loss in adult mice exposed to neonatal hyperoxia may impair host defense and ability to effectively repair after epithelial injury.
It is well known that high levels of oxygen inhibit cell proliferation, including that of type II cells (5, 15, 40). Hyperoxia may directly inhibit proliferation through cell cycle checkpoints activated in response to oxidative damage, such as in DNA (41). Hyperoxia may also indirectly inhibit proliferation through oxidative damage created by inflammatory cells recruited to the injured lung (42). Therefore, it was surprising to observe that high levels of oxygen stimulated proliferation and expansion of type II cells shortly after birth and before a time when hyperoxia is known to inhibit proliferation. Hyperoxia might stimulate proliferation by activating mitogen-activated protein kinases or stimulating expression of various growth factors such as keratinocyte growth factor, the same pathways used to block oxygen-induced cell death (43). Consistent with this idea, type II cell hyperplasia has been observed in preterm baboons and in adult rats exposed to sublethal amounts of hyperoxia (4, 25). Growth arrest that occurs by pnd4 may therefore be a response to oxidative damage that occurs with prolonged oxygen exposure. However, by this time, an overexpanded population of type II cells occupies the alveolar space. The alveolar surface of infants with BPD is lined with cells expressing high levels of Sftpc mRNA (2). Type II cell that express high levels of Sftpc mRNA express low levels of VEGF (44). Hence, the loss of VEGF expression and impaired angiogenesis seen in human BPD may be related in part to the rapid expansion of type II cells that occurs when the preterm lung is exposed to high oxygen at birth.
Our findings suggest hyperoxia promotes expansion of type II cells by stimulating their proliferation and the growth of precursor cells that will become type II cells. BrdU labeling revealed that hyperoxia stimulates proliferation of type II cells that normally are not proliferating on pnd1. Our findings are consistent with older studies in which postnatal proliferation of mouse or rat type II cells were evaluated using 3H-thymidine incorporation (30, 45). Fate mapping studies using Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG transgenic mice confirmed the conclusion that hyperoxia stimulates proliferation of type II cells and extended it by suggesting hyperoxia may also stimulate proliferation of precursor cells that become type II cells. Although the endogenous Scgb1a1 gene is expressed exclusively in airway epithelial cells of mice, the rat Scgb1a1 promoter faithfully drives proper expression typically seen in rat lung. Administration of doxycycline to adult rats activated reporter gene expression throughout the airway and in approximately 50% of type II cells (27). On the other hand, activation during embryonic development activates reporter gene expression in airway and a small population of alveolar cells that slowly expand postnatally (27). Consistent with those earlier findings, conditionally activated EGFP was rarely seen in alveoli of pnd4 mice exposed to RA, suggesting minimal expansion of type II cells at this time. In marked contrast, EGFP-labeled cells were readily observed on pnd4 in mice exposed to hyperoxia, suggesting that hyperoxia had stimulated proliferation of type II cell precursors. The source of these precursors remains to be determined but is unlikely to be derived from Scgb1a1-positive airway cells (46).
Hyperoxia also stimulated mRNA levels of T1α and Aqp5, genes expressed by type I cells. Whether this represents increased gene expression and/or increased number of type I cells remains to be determined. However, several pieces of data suggest the elevated expression of type I cell genes reflects an increase in the number of type I cells and that these cells are not derived from the expanded pool of type II cells defined by the Scgb1a1 lineage of epithelial cells. First, hyperoxia appears to have increased the number of type I cells because it increased expression of two different genes expressed by type I cells, implying a global effect on the type I cell phenotype. Second, expression of these genes remains elevated even when mice are returned to RA, suggesting that the effects are persistent and not just a change in gene expression. However, increased expression of T1α did not always correlate with reduced expression of proSP-C (Figure 6D). This supports an earlier study showing that exposure to 40% oxygen at birth is sufficient to increase T1α in adult mice, whereas exposure to 60% or more oxygen is needed to reduce expression of proSP-C (20). Also, EGFP-labeled cells of Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG transgenic mice exposed to hyperoxia were mostly cuboidal and not squamous. The rare squamous cell–expressing EGFP often did not stain with antibody against T1α, suggesting that they were not type I cells (data not shown). This line of mice rarely labels type I cells and only labels approximately 50% of type II cells (27). Taken together, these data suggest that elevated expression of type I genes reflects an expansion of type I cells; however, this expanded population was not derived from the expanded population of type II cells defined by the Scgb1a1-rtTA X (otet)7CMV-Cre X mT/mG reporter mice. Regardless of how hyperoxia affects expansion of type I cells, it clearly exerts a positive effect on the alveolar epithelium because it inhibits angiogenesis represented by expression of PECAM (Figure 1).
Type II cells expanded during hyperoxia were depleted during recovery in RA. This loss appears to be specific for type II cells because genes expressed by type I cells usually remained elevated during recovery. Mechanisms underlying the depletion of type II cells remain to be elucidated. The loss of type II cells seen in 8–week-old mice is unlikely to be related to hypoxia created when newborn mice are returned to RA because high numbers of type II cells are still seen weeks after recovery in RA. Because type II cells are slowly depleted during recovery, an alternative hypothesis is that they were injured during exposure and die out over time. Indeed, proliferating epithelial cells are more susceptible to genotoxic stress, including hyperoxia (47). Catastrophic damage to DNA or other macromolecules created during hyperoxia may be responsible for causing the cells to die. This conclusion is supported by fate mapping studies showing that EGFP-labeled type II cells expanded during hyperoxia are progressively lost with time and are not readily detected by 8 weeks of age. Alternatively, a genetic program that controls proper alveolar development is responsible for the loss of type II cells. Cell death is often required for proper development of several organs and tissues. For example, fingers and toes are created when excess cells produced between digits undergo apoptosis during fetal development (48). Axon overproduction followed by neuronal pruning and death are necessary for proper development of the brain (49). Postlactation involution of the mammary gland involves extensive death of the secretory epithelium and remodeling of the fat pad (50). Vascular pruning is also important for development of the retina, and its disruption in preterm infants receiving oxygen therapies causes retinopathy and blindness (51). Vascular pruning might also be responsible for the loss of microvessels seen in aged mice exposed to neonatal hyperoxia (22). Hence, the loss of type II cells observed in adult mice recovering from exposure to neonatal hyperoxia may be caused by the activation of an apoptotic program designed to ensure proper development of the alveolar epithelium. Consistent with this idea, apoptosis of some type II cells has been observed in rats by the third postnatal week as alveolar development is finalized (52).
The current study emphasizes the importance of using multiple markers to identify cells, particularly when trying to distinguish between changes in gene expression versus the number of cells expressing that gene. This became especially important when we discovered that hyperoxia inhibited proSP-C and ABCA3 protein while increasing expression of their respective mRNAs. Indeed, the literature is replete with studies showing diverse effects of hyperoxia on surfactant protein and mRNA expression. Type II cell hyperplasia in preterm baboons exposed to sublethal amounts of oxygen is associated with increased mRNA expression of Sftpb and Sftpc but not Sftpa (53). However, increased mRNA did not always correlate with increased protein in lavage (54). Increased mRNA for Sftpa, Sftpb, Sftpc, and Sftpd is seen in newborn rats exposed to 85% oxygen (55). However, the pattern of change did not always correlate with protein abundance. Increased mRNA for some surfactant genes was also observed in preterm sheep exposed to hyperoxia, but that too varied by gestational age (56). Hence, it has been difficult to discern whether altered expression of surfactant mRNA or protein reflects a change in gene expression and/or a change in the number of cells expressing the gene (57). Whether differences in mRNA and the respective protein reflect an important biological response to hyperoxia or a general response to oxidative damage remains to be discerned. Because the goal of the current study was to track the fate of type II cells during hyperoxia, it was necessary to use additional markers of type II cells that would be potentially less affected by hyperoxia. Temporal and spatial activation of Cre-mediated excision of reporter genes has proven useful for mapping cell fate during organ development or tissue regeneration. This approach circumvented the need to use endogenous markers to identify cells as they change with time. The rat Scgb1a1 (secretoglobin, family 1A, member 1, also called uteroglobin or clara cell secretory protein) promoter driving a doxycycline-dependent rtTA gene has been especially useful for defining airway and parenchymal lung development because it marks conducting airway epithelial cells before birth and conducting airway and alveolar cells after birth (32). Because the site of transgene integration into the genome can affect gene expression, the effects of hyperoxia on type II cells were also studied in Sftpc-EGFP transgenic mice. Unlike proSP-C, which is lost early during hyperoxia, EGFP recapitulated Sftpc mRNA expression perhaps because it is retained within the cell and is resistant to proteasome degradation (29). Although these approaches do not rule out the possibility that hyperoxia affects gene expression, they allowed us to conclude that hyperoxia does stimulate the expansion of ATII cells that are pruned during recovery in RA.
In summary, we have shown for the first time that neonatal hyperoxia rapidly stimulates proliferation of mouse type II cells within the first day of life. Longer exposure to hyperoxia inhibits expansion of these cells (15), which are then pruned during recovery in RA. Understanding how oxygen affects proliferation and pruning of type II cells or its precursors should help clarify why children born prematurely and treated with supplemental oxygen have increased risk for persistent pulmonary disease later in life.
Acknowledgments
Acknowledgments
The authors thank Brigid Hogan for sharing Sftpc-EGFP transgenic mice (created by John K. Heath, University of Birmingham, UK) and Jeffrey Whitsett for sharing rat CCSP-rtTA and (otet)7CMV-Cre lines of mice with us and Gloria Pryhuber for helpful discussions on how our findings in mice relate to what is known in human BPD.
Footnotes
This work was supported by National Institutes of Health grants HL067392, HL091968, and HL097141; Training Grants HL66988 and ES07026; and Center grant ES01247 and by March of Dimes grant 06-FY08–264.
This article has an online supplement, which is accessible from this issue's table of contents at www.atsjournals.org
Originally Published in Press as DOI: 10.1165/rcmb.2013-0207OC on November 4, 2013
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Eber E, Zach MS. Long term sequelae of bronchopulmonary dysplasia (chronic lung disease of infancy) Thorax. 2001;56:317–323. doi: 10.1136/thorax.56.4.317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bhatt AJ, Pryhuber GS, Huyck H, Watkins RH, Metlay LA, Maniscalco WM. Disrupted pulmonary vasculature and decreased vascular endothelial growth factor, Flt-1, and TIE-2 in human infants dying with bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;164:1971–1980. doi: 10.1164/ajrccm.164.10.2101140. [DOI] [PubMed] [Google Scholar]
- 3.Coalson JJ, Winter VT, Gerstmann DR, Idell S, King RJ, Delemos RA. Pathophysiologic, morphometric, and biochemical studies of the premature baboon with bronchopulmonary dysplasia. Am Rev Respir Dis. 1992;145:872–881. doi: 10.1164/ajrccm/145.4_Pt_1.872. [DOI] [PubMed] [Google Scholar]
- 4.Maniscalco WM, Watkins RH, O’Reilly MA, Shea CP. Increased epithelial cell proliferation in very premature baboons with chronic lung disease. Am J Physiol Lung Cell Mol Physiol. 2002;283:L991–L1001. doi: 10.1152/ajplung.00050.2002. [DOI] [PubMed] [Google Scholar]
- 5.Warner BB, Stuart LA, Papes RA, Wispé JR. Functional and pathological effects of prolonged hyperoxia in neonatal mice. Am J Physiol. 1998;275:L110–L117. doi: 10.1152/ajplung.1998.275.1.L110. [DOI] [PubMed] [Google Scholar]
- 6.Bonikos DS, Bensch KG, Ludwin SK, Northway WH., Jr Oxygen toxicity in the newborn. The effect of prolonged 100 per cent O2 exposure on the lungs of newborn mice. Lab Invest. 1975;32:619–635. [PubMed] [Google Scholar]
- 7.Thébaud B, Abman SH. Bronchopulmonary dysplasia: where have all the vessels gone? Roles of angiogenic growth factors in chronic lung disease. Am J Respir Crit Care Med. 2007;175:978–985. doi: 10.1164/rccm.200611-1660PP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Balasubramaniam V, Mervis CF, Maxey AM, Markham NE, Abman SH. Hyperoxia reduces bone marrow, circulating, and lung endothelial progenitor cells in the developing lung: implications for the pathogenesis of bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol. 2007;292:L1073–L1084. doi: 10.1152/ajplung.00347.2006. [DOI] [PubMed] [Google Scholar]
- 9.Doyle LW, Faber B, Callanan C, Freezer N, Ford GW, Davis NM. Bronchopulmonary dysplasia in very low birth weight subjects and lung function in late adolescence. Pediatrics. 2006;118:108–113. doi: 10.1542/peds.2005-2522. [DOI] [PubMed] [Google Scholar]
- 10.Robin B, Kim YJ, Huth J, Klocksieben J, Torres M, Tepper RS, Castile RG, Solway J, Hershenson MB, Goldstein-Filbrun A. Pulmonary function in bronchopulmonary dysplasia. Pediatr Pulmonol. 2004;37:236–242. doi: 10.1002/ppul.10424. [DOI] [PubMed] [Google Scholar]
- 11.Smith VC, Zupancic JA, McCormick MC, Croen LA, Greene J, Escobar GJ, Richardson DK. Rehospitalization in the first year of life among infants with bronchopulmonary dysplasia. J Pediatr. 2004;144:799–803. doi: 10.1016/j.jpeds.2004.03.026. [DOI] [PubMed] [Google Scholar]
- 12.Weisman LE. Populations at risk for developing respiratory syncytial virus and risk factors for respiratory syncytial virus severity: infants with predisposing conditions. Pediatr Infect Dis J. 2003;22:S33–S37; discussion S37–S39. doi: 10.1097/01.inf.0000053883.08663.e5. [DOI] [PubMed] [Google Scholar]
- 13.Doyle LW, Faber B, Callanan C, Morley R. Blood pressure in late adolescence and very low birth weight. Pediatrics. 2003;111:252–257. doi: 10.1542/peds.111.2.252. [DOI] [PubMed] [Google Scholar]
- 14.Cutz E, Chiasson D. Chronic lung disease after premature birth. N Engl J Med. 2008;358:743–745, author reply 745–746. doi: 10.1056/NEJMc073362. [DOI] [PubMed] [Google Scholar]
- 15.Yee M, Vitiello PF, Roper JM, Staversky RJ, Wright TW, McGrath-Morrow SA, Maniscalco WM, Finkelstein JN, O’Reilly MA. Type II epithelial cells are critical target for hyperoxia-mediated impairment of postnatal lung development. Am J Physiol Lung Cell Mol Physiol. 2006;291:L1101–L1111. doi: 10.1152/ajplung.00126.2006. [DOI] [PubMed] [Google Scholar]
- 16.Schulman SR, Canada AT, Fryer AD, Winsett DW, Costa DL. Airway hyperreactivity produced by short-term exposure to hyperoxia in neonatal guinea pigs. Am J Physiol. 1997;272:L1211–L1216. doi: 10.1152/ajplung.1997.272.6.L1211. [DOI] [PubMed] [Google Scholar]
- 17.McGrath-Morrow SA, Cho C, Soutiere S, Mitzner W, Tuder R. The effect of neonatal hyperoxia on the lung of p21Waf1/Cip1/Sdi1-deficient mice. Am J Respir Cell Mol Biol. 2004;30:635–640. doi: 10.1165/rcmb.2003-0049OC. [DOI] [PubMed] [Google Scholar]
- 18.Denis D, Fayon MJ, Berger P, Molimard M, De Lara MT, Roux E, Marthan R. Prolonged moderate hyperoxia induces hyperresponsiveness and airway inflammation in newborn rats. Pediatr Res. 2001;50:515–519. doi: 10.1203/00006450-200110000-00015. [DOI] [PubMed] [Google Scholar]
- 19.Dauger S, Ferkdadji L, Saumon G, Vardon G, Peuchmaur M, Gaultier C, Gallego J. Neonatal exposure to 65% oxygen durably impairs lung architecture and breathing pattern in adult mice. Chest. 2003;123:530–538. doi: 10.1378/chest.123.2.530. [DOI] [PubMed] [Google Scholar]
- 20.Yee M, Chess PR, McGrath-Morrow SA, Wang Z, Gelein R, Zhou R, Dean DA, Notter RH, O’Reilly MA. Neonatal oxygen adversely affects lung function in adult mice without altering surfactant composition or activity. Am J Physiol Lung Cell Mol Physiol. 2009;297:L641–L649. doi: 10.1152/ajplung.00023.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Buczynski BW, Yee M, Paige Lawrence B, O’Reilly MA. Lung development and the host response to influenza A virus are altered by different doses of neonatal oxygen in mice. Am J Physiol Lung Cell Mol Physiol. 2012;302:L1078–L1087. doi: 10.1152/ajplung.00026.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yee M, White RJ, Awad HA, Bates WA, McGrath-Morrow SA, O’Reilly MA. Neonatal hyperoxia causes pulmonary vascular disease and shortens life span in aging mice. Am J Pathol. 2011;178:2601–2610. doi: 10.1016/j.ajpath.2011.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.O’Reilly MA, Marr SH, Yee M, McGrath-Morrow SA, Lawrence BP. Neonatal hyperoxia enhances the inflammatory response in adult mice infected with influenza A virus. Am J Respir Crit Care Med. 2008;177:1103–1110. doi: 10.1164/rccm.200712-1839OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Giannandrea M, Yee M, O’Reilly MA, Lawrence BP. Memory CD8+ T cells are sufficient to alleviate impaired host resistance to influenza A virus infection caused by neonatal oxygen supplementation. Clin Vaccine Immunol. 2012;19:1432–1441. doi: 10.1128/CVI.00265-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Crapo JD, Barry BE, Foscue HA, Shelburne J. Structural and biochemical changes in rat lungs occurring during exposures to lethal and adaptive doses of oxygen. Am Rev Respir Dis. 1980;122:123–143. doi: 10.1164/arrd.1980.122.1.123. [DOI] [PubMed] [Google Scholar]
- 26.Lo B, Hansen S, Evans K, Heath JK, Wright JR. Alveolar epithelial type II cells induce T cell tolerance to specific antigen. J Immunol. 2008;180:881–888. doi: 10.4049/jimmunol.180.2.881. [DOI] [PubMed] [Google Scholar]
- 27.Perl AK, Tichelaar JW, Whitsett JA. Conditional gene expression in the respiratory epithelium of the mouse. Transgenic Res. 2002;11:21–29. doi: 10.1023/a:1013986627504. [DOI] [PubMed] [Google Scholar]
- 28.Kolla V, Gonzales LW, Gonzales J, Wang P, Angampalli S, Feinstein SI, Ballard PL. Thyroid transcription factor in differentiating type II cells: regulation, isoforms, and target genes. Am J Respir Cell Mol Biol. 2007;36:213–225. doi: 10.1165/rcmb.2006-0207OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu CW, Corboy MJ, DeMartino GN, Thomas PJ. Endoproteolytic activity of the proteasome. Science. 2003;299:408–411. doi: 10.1126/science.1079293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Amy RW, Bowes D, Burri PH, Haines J, Thurlbeck WM. Postnatal growth of the mouse lung. J Anat. 1977;124:131–151. [PMC free article] [PubMed] [Google Scholar]
- 31.Kauffman SL, Burri PH, Weibel ER. The postnatal growth of the rat lung. II. Autoradiography. Anat Rec. 1974;180:63–76. doi: 10.1002/ar.1091800108. [DOI] [PubMed] [Google Scholar]
- 32.Perl AK, Wert SE, Loudy DE, Shan Z, Blair PA, Whitsett JA. Conditional recombination reveals distinct subsets of epithelial cells in trachea, bronchi, and alveoli. Am J Respir Cell Mol Biol. 2005;33:455–462. doi: 10.1165/rcmb.2005-0180OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yi M, Jankov RP, Belcastro R, Humes D, Copland I, Shek S, Sweezey NB, Post M, Albertine KH, Auten RL, et al. Opposing effects of 60% oxygen and neutrophil influx on alveologenesis in the neonatal rat. Am J Respir Crit Care Med. 2004;170:1188–1196. doi: 10.1164/rccm.200402-215OC. [DOI] [PubMed] [Google Scholar]
- 34.Sisson TH, Hansen JM, Shah M, Hanson KE, Du M, Ling T, Simon RH, Christensen PJ. Expression of the reverse tetracycline-transactivator gene causes emphysema-like changes in mice. Am J Respir Cell Mol Biol. 2006;34:552–560. doi: 10.1165/rcmb.2005-0378OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Otto-Verberne CJ, Ten Have-Opbroek AA, Willems LN, Franken C, Kramps JA, Dijkman JH. Lack of type ii cells and emphysema in human lungs. Eur Respir J. 1991;4:316–323. [PubMed] [Google Scholar]
- 36.McCormack FX, Whitsett JA. The pulmonary collectins, SP-A and SP-D, orchestrate innate immunity in the lung. J Clin Invest. 2002;109:707–712. doi: 10.1172/JCI15293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fehrenbach H. Alveolar epithelial type II cell: defender of the alveolus revisited. Respir Res. 2001;2:33–46. doi: 10.1186/rr36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yee M, Buczynski BW, Lawrence BP, O'Reilly MA. Neonatal hyperoxia increases sensitivity of adult mice to bleomycin-induced lung fibrosis. Am J Respir Cell Mol Biol. 2013;48:258–266. doi: 10.1165/rcmb.2012-0238OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.O’Reilly MA, Yee M, Buczynski BW, Vitiello PF, Keng PC, Welle SL, Finkelstein JN, Dean DA, Lawrence BP. Neonatal oxygen increases sensitivity to influenza A virus infection in adult mice by suppressing epithelial expression of Ear1. Am J Pathol. 2012;181:441–451. doi: 10.1016/j.ajpath.2012.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Auten RL, Mason SN, Auten KM, Brahmajothi M. Hyperoxia impairs postnatal alveolar epithelial development via NADPH oxidase in newborn mice. Am J Physiol Lung Cell Mol Physiol. 2009;297:L134–L142. doi: 10.1152/ajplung.00112.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.O’Reilly MA. DNA damage and cell cycle checkpoints in hyperoxic lung injury: braking to facilitate repair. Am J Physiol Lung Cell Mol Physiol. 2001;281:L291–L305. doi: 10.1152/ajplung.2001.281.2.L291. [DOI] [PubMed] [Google Scholar]
- 42.Auten RL, Whorton MH, Nicholas Mason S. Blocking neutrophil influx reduces DNA damage in hyperoxia-exposed newborn rat lung. Am J Respir Cell Mol Biol. 2002;26:391–397. doi: 10.1165/ajrcmb.26.4.4708. [DOI] [PubMed] [Google Scholar]
- 43.Lee PJ, Choi AM. Pathways of cell signaling in hyperoxia. Free Radic Biol Med. 2003;35:341–350. doi: 10.1016/s0891-5849(03)00279-x. [DOI] [PubMed] [Google Scholar]
- 44.Maniscalco WM, Watkins RH, Finkelstein JN, Campbell MH. Vascular endothelial growth factor mRNA increases in alveolar epithelial cells during recovery from oxygen injury. Am J Respir Cell Mol Biol. 1995;13:377–386. doi: 10.1165/ajrcmb.13.4.7546767. [DOI] [PubMed] [Google Scholar]
- 45.Kauffman SL. Kinetics of pulmonary epithelial proliferation during prenatal growth of the mouse lung. Anat Rec. 1975;183:393–403. doi: 10.1002/ar.1091830304. [DOI] [PubMed] [Google Scholar]
- 46.Rawlins EL, Okubo T, Xue Y, Brass DM, Auten RL, Hasegawa H, Wang F, Hogan BL. The role of Scgb1a1+ Clara cells in the long-term maintenance and repair of lung airway, but not alveolar, epithelium. Cell Stem Cell. 2009;4:525–534. doi: 10.1016/j.stem.2009.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rancourt RC, Hayes DD, Chess PR, Keng PC, O’Reilly MA. Growth arrest in G1 protects against oxygen-induced DNA damage and cell death. J Cell Physiol. 2002;193:26–36. doi: 10.1002/jcp.10146. [DOI] [PubMed] [Google Scholar]
- 48.Zuzarte-Luis V, Hurle JM. Programmed cell death in the embryonic vertebrate limb. Semin Cell Dev Biol. 2005;16:261–269. doi: 10.1016/j.semcdb.2004.12.004. [DOI] [PubMed] [Google Scholar]
- 49.Vanderhaeghen P, Cheng HJ. Guidance molecules in axon pruning and cell death. Cold Spring Harb Perspect Biol. 2010;2:a001859. doi: 10.1101/cshperspect.a001859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Watson CJ, Kreuzaler PA. Remodeling mechanisms of the mammary gland during involution. Int J Dev Biol. 2011;55:757–762. doi: 10.1387/ijdb.113414cw. [DOI] [PubMed] [Google Scholar]
- 51.Jenkinson SG. Oxygen toxicity. New Horiz. 1993;1:504–511. [PubMed] [Google Scholar]
- 52.Schittny JC, Djonov V, Fine A, Burri PH. Programmed cell death contributes to postnatal lung development. Am J Respir Cell Mol Biol. 1998;18:786–793. doi: 10.1165/ajrcmb.18.6.3031. [DOI] [PubMed] [Google Scholar]
- 53.Minoo P, Segura L, Coalson JJ, King RJ, DeLemos RA. Alterations in surfactant protein gene expression associated with premature birth and exposure to hyperoxia. Am J Physiol. 1991;261:L386–L392. doi: 10.1152/ajplung.1991.261.6.L386. [DOI] [PubMed] [Google Scholar]
- 54.Awasthi S, Coalson JJ, Crouch E, Yang F, King RJ. Surfactant proteins A and D in premature baboons with chronic lung injury (Bronchopulmonary dysplasia): evidence for an inhibition of secretion. Am J Respir Crit Care Med. 1999;160:942–949. doi: 10.1164/ajrccm.160.3.9806061. [DOI] [PubMed] [Google Scholar]
- 55.White CW, Greene KE, Allen CB, Shannon JM. Elevated expression of surfactant proteins in newborn rats during adaptation to hyperoxia. Am J Respir Cell Mol Biol. 2001;25:51–59. doi: 10.1165/ajrcmb.25.1.4296. [DOI] [PubMed] [Google Scholar]
- 56.Woods E, Ohashi T, Polk D, Ikegami M, Ueda T, Jobe AH. Surfactant treatment and ventilation effects on surfactant SP-A, SP-B, and SP-C mRNA levels in preterm lamb lungs. Am J Physiol. 1995;269:L209–L214. doi: 10.1152/ajplung.1995.269.2.L209. [DOI] [PubMed] [Google Scholar]
- 57.Boggaram V. Regulation of surfactant protein gene expression by hyperoxia in the lung. Antioxid Redox Signal. 2004;6:185–190. doi: 10.1089/152308604771978499. [DOI] [PubMed] [Google Scholar]