Abstract
Electrochemotherapy is a new method for the local treatment of cutaneous and subcutaneous metastases. If surgery and/or radiotherapy are no longer possible, this innovative technology can be used effectively for local tumour control. The minor side effects of the therapy and the low intraoperative duration of treatment make it possible to admit patients to hospital for just a short time. Thus, the repeated use of electrochemotherapy has allowed for an increase in the rate of complete remissions. In fact, in 2013, this form of treatment, which consists of a low-dose cytostatic and electroporation, was also included in the mammary guidelines of the Working Group for Gynaecological Oncology (AGO) and the German Cancer Society (DKG). The favourable cost-benefit ratio makes this method of treatment very interesting in clinical practice and, as a result, it is already being used successfully in many German hospitals.
Key words: breast cancer, metastasis, mammary
Abstract
Zusammenfassung
Die Elektrochemotherapie ist eine neue Methode zur lokalen Behandlung von kutanen und subkutanen Metastasen. Sind Operation und/oder Radiatio nicht mehr möglich, so kann diese innovative Technik effektiv zur lokalen Tumorkontrolle eingesetzt werden. Die geringen Nebenwirkungen der Therapie und die geringe intraoperative Behandlungsdauer machen eine kurze Hospitalisation möglich. Durch wiederholten Einsatz der Elektrochemotherapie kann dabei die Rate an Komplettremissionen erhöht werden, sodass 2013 diese aus einem niedrig dosiertem Zytostatikum und Elektroporation bestehende Therapieform auch in die Mamma-Leitlinien der AGO/DKG aufgenommen wurde. Die günstige Kosten-Nutzen-Relation macht dieses Behandlungsverfahren in der klinischen Praxis sehr interessant, sodass es bereits erfolgreich in zahlreichen deutschen Kliniken eingesetzt wird.
Schlüsselwörter: Mammakarzinom, Metastasierung, Mamma
Fundamentals and Technical Requirements of Electrochemotherapy
Electrochemotherapy is a combination of electroporation and low-dose chemotherapy. The first clinical (phase I/II) studies were already being carried out at the beginning of the 1990s 1. Thus far, this innovative technology has established itself mainly in the dermatological field. New study results, however, have been able to prove the treatmentʼs high degree of effectiveness when used for gynaecological tumours, with electrochemotherapy now being increasingly employed in the field of gynaecology as a result.
Electrochemotherapy utilises the phenomenon of electroporation. By applying an external electric field, the membrane potential of the cell can be changed. At a sufficiently high intensity, this results in changes in the structure of the membrane. This can cause small “pores” in the membrane, which in turn make the membrane permeable to molecules which would otherwise be unable to penetrate it. In this way, the increased permeability can allow cytostatic agents to cross the cell membrane and then enter into the cytosol by diffusion. The increased permeability also allows the cytostatic to reach the cytosol at much higher concentrations, leading to increased cytotoxicity 2. As electroporation is limited to the tumour-affected area, there is no increase in systemic toxicity caused by the chemotherapeutic agent, resulting in the good tolerability of this method. The permeability of the cell membrane, however, does not persist for an unlimited period, and is suspended once again a few minutes after the electrical impulse (Fig. 1) 3.
Fig. 1.
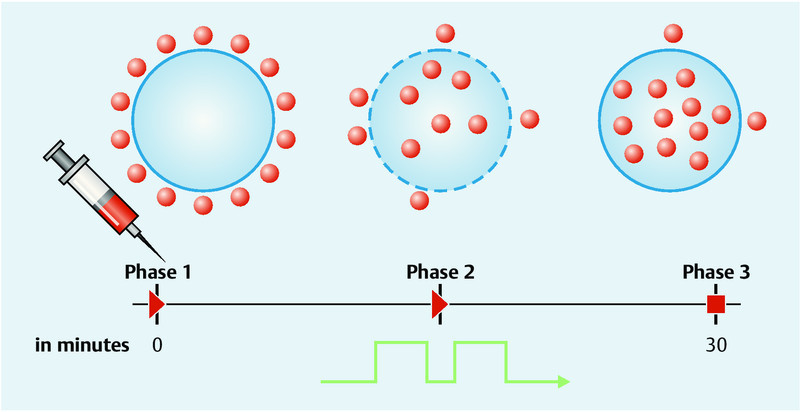
Schematic representation of electrochemotherapy: Phase 1: Injection of the cytostatic: bleomycin (i. v./intratumoural) or cisplatin (intratumoural); Phase 2: high-frequency electroporation results in a permeable cell membrane and the cytostatic agent reaches up to 10 000 times higher concentrations in the tumour cell; Phase 3: reversible poration, the cell membrane closes and the cytostatic drug accumulates in the tumour cell (courtesy of IGEA, Carpi, Italy).
Practical Implementation and Technical Requirements
Electrochemotherapy is performed using an electrical impulse generator, the Cliniporator™ (IGEA, Carpi, Italy). The device is shown in Fig. 1 and consists of a console unit, a power supply unit for supplying the current and an applicator for placement on the skin. The Cliniporator™ allows for electroporation by means of the generation of an electrical low and an electrical high voltage impulse, which are applied to the tumour cells using specific electrodes. This makes possible the transfer of intracellular substances or molecules which would not normally be able to pass through the cell membrane. The emitted impulse lasts 100 µs. The number of impulses varies between 1–20 and their amplitude between 100–1000 volts. The frequency used is between 1–5000 Hz. During implementation, both the applied voltage and the corresponding current curve are shown on the display in real-time, thus allowing for the effectiveness of each individual electroporation to be seen on the monitor.
Prior to the start of the impulse generation, a low-dose cytostatic – usually bleomycin at a dosage of 15 mg/m2 – is administered intravenously over one minute. Following a recommended time interval of 8 minutes, the administration of the electrical impulses begins. Special electrodes are used to generate an electrical field around the tumour. The cell membrane then opens and the cytostatic agent is able to accumulate in the cell at a high concentration. Different needle electrodes and plate electrodes are used depending on the size, depth and shape of the tumour (Fig. 2) 3. The procedure is performed under local anaesthesia, regional anaesthesia or general anaesthesia, depending on the location of the tumour. In the case of large surfaces, painful muscle contractions are to be expected: as a result, it is wise to carry out the procedure under general anaesthesia. The best results are obtained when the impulse is applied within 25 minutes following administration of the chemotherapeutic agent 3, 4, 5.
Fig. 2.
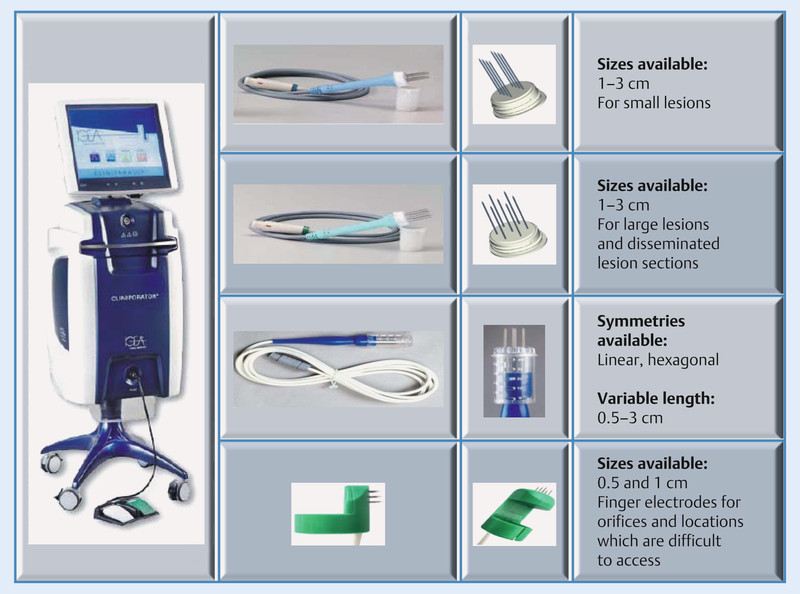
Cliniporator and electrodes for carrying out electrochemotherapy (courtesy of IGEA, Carpi, Italy).
Cytotoxic Agents Used
Several cytotoxic agents have been tested in preclinical studies (e. g. doxorubicin, daunorubicin, paclitaxel, etoposide, bleomycin and cisplatin, etc.). In principle, following electroporation, the anti-tumour effect of cisplatin is increased between 1.1 and 80 times, and is increased several thousand times in the case of bleomycin 6. On the basis of these studies, bleomycin and cisplatin proved to be the optimal substances in terms of their effects 6. Cytotoxic agents can be administered both intravenously and intratumourally 3. With respect to response rates, there was no significant difference found between intratumoural and intravenous administration of cisplatin and bleomycin (Fig. 3).
Fig. 3.
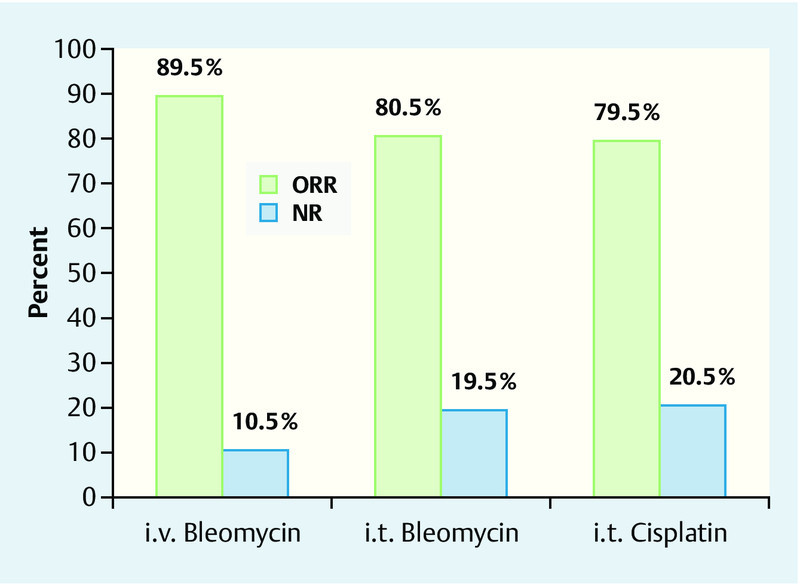
Objective response rate of tumours treated with intravenous or intratumoural (i. v. or i. t.) bleomycin and intratumoural (i. t.) cisplatin. ORR: “overall response”, NR: “no response” (courtesy of IGEA, Carpi, Italy).
Bleomycin
Bleomycin is an anti-cancer antibiotic from the group of glycopeptides with minimal myelotoxic effect 7. In the case of bleomycin, it is important to note that it is primarily excreted renally, and that it is rendered inactive by the enzyme bleomycin hydrolase. This enzyme is present in all cells of the human body, however the concentration of the enzyme is reduced in skin cells and in alveolar epithelial cells in the lung.
The administration of bleomycin can cause subacute or chronic interstitial plasmacytic pneumonia, which can also lead to interstitial fibrosis in the worst case. Pulmonary toxicity is reported in approximately 10 % of patients and approximately 1 % of patients develop a pulmonary fibrosis from the bleomycin-induced nonspecific pneumonitis; this can also be fatal in the worst case 8. Risk factors that promote pulmonary fibrosis include advanced age, pre-existing pulmonary disease, smoking, renal insufficiency, previous radiotherapy of the lung, increased oxygen concentration in the air inhaled, etc. 9 (bleomycin drug information sheet).
A connection has been established between bleomycin administration and pulmonary toxicity. Therefore, a bleomycin dose of 400 IU/m2 should not be exceeded.
It is also important that ⅔ of the bleomycin dose administered is excreted unchanged in the patientʼs urine. In conclusion, patients with impaired renal function have greatly increased plasma concentrations when standard doses are administered. Therefore, patients with moderately reduced renal function (glomerular filtration rate [GFR] 10–50 ml/minute) should receive only 75 % of the usual dose at normal dose intervals, while patients with severe renal impairment (GFR < 10 ml/minute) should receive 50 % of the usual dose at normal dose intervals. No dosage adjustment is necessary in patients with a GFR > 50 ml/minute (bleomycin drug information sheet).
An overdose can cause hypotension, fever, an increase in pulse, difficulty breathing and general symptoms of shock. It is important to note that there is no specific antidote and that bleomycin cannot be dialysed.
In the case of respiratory problems, the patient should be treated with corticosteroids and broad-spectrum antibiotics.
How does bleomycin work at cellular level?
At DNA level, bleomycin leads to DNA strand breaks and inhibits the DNA-dependent polymerase. This leads to the fragmentation of the DNA 10.
The effect of bleomycin remains unchanged despite the effect of electroporation: cell death is induced by means of DNA breakages, similar to diffusion through non-permeable membranes. In the case of non-electroporated cells, the uptake of bleomycin is limited.
In electrochemotherapy, both intratumoural and intravenous bleomycin administration are possible. Intratumoural administration is recommended in cases in which a small number of metastases (< 7) and small nodules (< 20 mm in diameter) are present 3, 6, 11, 12. Dosing in intratumoural administration is dependent on tumour volume. In Table 1, the corresponding doses from the SOP are shown. The number of metastases to be treated must of course be determined in advance.
Table 1 Dosing of intratumoural bleomycin and intratumoural cisplatin according to the tumour volume (Mir et al. 2006).
| Tumour volume (V = ab2π/6) | < 0.5 cm3 | 0.5–1 cm3 | > 1 cm3 |
|---|---|---|---|
| Bleomycin dosage concentration 1 000 IU/ml | 1 ml (1 000 IU)/cm3 of tumour | 0.5 (500 IU)/cm3 of tumour | 0.25 (250 IU)/cm3 of tumour |
| Carboplatin dosage concentration 2 mg/ml | 1 ml (2 mg)/cm3 of tumour | 0.5 ml (1 mg)/cm3 of tumour | 0.25 ml (0.5 mg)/cm3 of tumour |
Studies have demonstrated that the response of cutaneous metastases in the intratumoural administration of bleomycin alone is significantly worse (31 %) than the response in the case of the administration of a combination of intratumoural bleomycin and electroporation (78 %) 12 (Fig. 4).
Fig. 4.
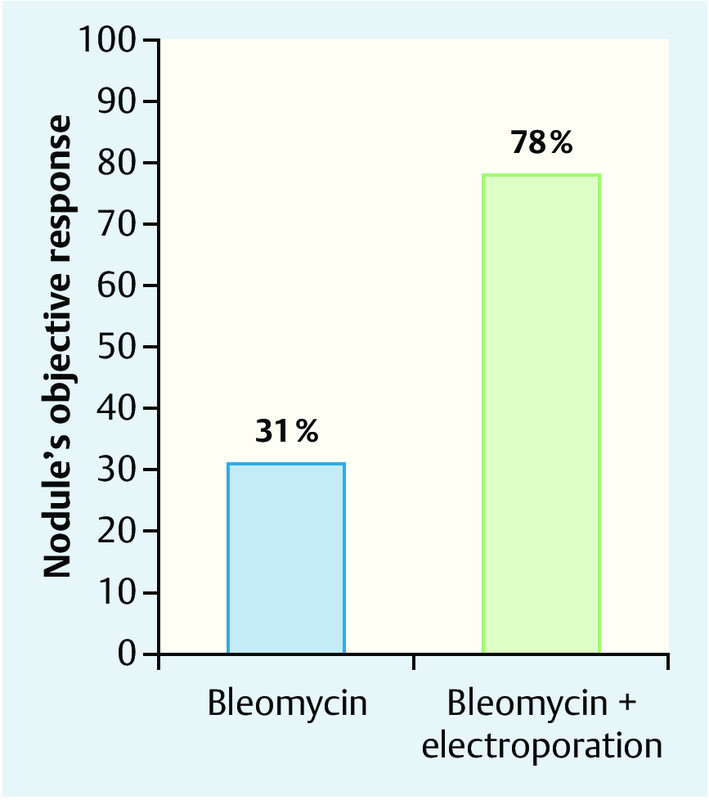
Response rate of cutaneous metastases in intratumoural bleomycin therapy alone vs. combination therapy consisting of intratumoural bleomycin and simultaneous electroporation (courtesy of IGEA, Carpi).
If several, larger metastases (> 20 mm in diameter) are present, the intravenous administration of bleomycin is recommended. In this case, the dosage of bleomycin should be 15 mg/m2 or 18–27 IU/m2. For intravenous administration it should be dissolved in 5 or 10 ml (max. 50 ml), so that it can be administered within one minute. The dosage is comparable to conventional chemotherapy. However, as it is only administered once, the total dose is generally regarded as low. Electrochemotherapy treatment, however, can also be repeated several times if necessary. The cumulative bleomycin dose of 400 IU/m2 must not be exceeded, however, due to the danger of pulmonary fibrosis. It is also important, in the case of conscious sedation or general anaesthesia, that the FiO2 chosen by the anaesthesiologist is as low as possible due to the increased risk of pulmonary fibrosis in the case of hyperoxia.
The intravenous administration of bleomycin has been proven in practice, while intratumoural administration has not.
Cisplatin
Cisplatin works exactly like bleomycin at DNA level. Unlike bleomycin, however, cisplatin leads to the cross-linking of DNA, thus destroying their structure and rendering them inoperative. The result is that the cell subsequently initiates apoptosis.
90 % of cisplatin is excreted renally (tubular and glomerular). Prior to the start of and during treatment renal function, electrolytes, blood count and hearing should be investigated, as well as hepatic and neurological functions. In order to avoid serious kidney damage, attention should be paid to adequate hydration. Forced diuresis should never be performed using loop diuretics, as this increases the risk of renal tubular injury and increased ototoxicity. Should forced fluid retention prove necessary, for example in the case of fluid retention ≥ 1000 ml, mannitol must be administered.
Two hours after administration, more than 90 % of cisplatin will have bound to plasma proteins. Therefore, cisplatin can only be dialysed in the first 2 hours following administration (cisplatin drug information sheet).
Cisplatin may be administered as part of an intravenous chemotherapy treatment in doses up to 100 mg/m2 BSA, depending on the treatment regimen. These high doses cannot be achieved in electrochemotherapy, due to the fact that, in contrast to bleomycin, cisplatin should only be administered intratumourally, and not intravenously. The expected side effects of electrochemotherapy are therefore considered to be minor (Table 1).
Cisplatin should also only be administered in the case of a small number (< 7) of small metastases (< 20 mm in diameter). The dosing is shown in Table 1. The number of metastases to be treated must also be determined in advance in the case of cisplatin use.
Indication Studies
General
The method is suitable for patients with cutaneous or subcutaneous metastases who have already exhausted the possibilities of surgery and radiation therapy and who are no longer strong candidates for surgery or chemotherapy treatment. The method is generally suitable for all solid tumours. The application is palliative, with the objectives of relieving tumour-associated symptoms and improving quality of life. These are ensured by a short treatment time, the rapid action of the electrochemotherapy and simple after-care.
In 2003, the ESOPE project (European Standard Operating Procedures for Electrochemotherapy) 3 was founded. The aim of this project was to confirm the efficacy of electrochemotherapy in cutaneous and subcutaneous metastases as part of an international, multi-centre prospective study, and to develop standardisation of the method. The endpoints of the study were response rate according to WHO criteria, and response rate taking into account the tumour type, size, location, etc. In addition, the side effects and toxicity of the therapy were also to be investigated within the framework of the project. A total of 41 patients with cutaneous and subcutaneous metastases with a tumour size < 3 cm were included in the study. The study showed that electrochemotherapy was effective in both melanomas and non-melanomas, with a complete remission rate of 74 % and a partial remission rate of 11 % according to the WHO criteria. The local tumour control rate 5 months after treatment was between 73–88 % 3, 13. Another study of 52 patients with varying tumour types published in 2008 showed that 50 % of patients were in complete remission 1 month after electrochemotherapy treatment. Partial remission was observed in 46 % of patients and no response was observed in 4 %. The response was objectivised according to RECIST criteria. 94 % of patients surveyed in this study indicated an improvement in their quality of life after receiving electrochemotherapy 14. A further Danish and English study of 52 patients showed a complete remission rate of 68 % and a partial remission rate of 18 % for cutaneous metastases < 3 cm, corresponding to 8 and 23 % for metastases > 3 cm 15.
In 2012, Mali et al. published a review which aimed to further strengthen the effects of electrochemotherapy. A total of 44 previously published studies (1993–2011) of 413 patients and 1894 tumours were analysed. Patients with malignant melanomas, breast cancer and head and neck tumours were all included in the study. It was noted that, regardless of tumour type, electrochemotherapy showed a complete remission rate of 59.4 % and an objective response rate of 84.1 % 16.
Breast cancer
In the case of breast cancer, recurrences in the thoracic wall area following a mastectomy have been reported with an incidence of 5–40 % 17, 18, 19. It is also known that approximately 10–15 % of all breast cancer patients develop a locoregional recurrence within 10 years following a mastectomy and radiation therapy. In fact, in the case of most patients, this occurred after 2–3 years 20, 21, 22 in the form of numerous cutaneous and subcutaneous nodules in the area of the thoracic wall. If these are left untreated, they often lead to ulceration, bleeding, pain, etc. The quality of life of affected patients is thus significantly reduced.
Once the treatment options of surgery and radiation therapy have been exhausted, the possibilities of local tumour control are limited. If the cutaneous metastases do not respond well to chemotherapy, miltefosine or electrochemotherapy are the only remaining options. Topical chemotherapy with miltefosine was not a possibility as this method was withdrawn from the market and is no longer available in Germany. Furthermore, studies also showed only a moderate success rate for this preparation when used for cutaneous metastases. The overall response rate for miltefosine is only 33 %. In addition, topical chemotherapy does not possess a low side effect profile, as a severe grade 3 or grade 4 toxic skin reaction occurs during the therapy in more than ⅓ of patients treated 23. Because of its low side effects, electrochemotherapy is therefore an appropriate therapy for patients who have exhausted all other treatment options.
The studies published to date are very promising: in a phase II study published in 2012, 19 of 35 patients (54.3 %) with an unresectable chest wall recurrence were in complete remission 2 months after electrochemotherapy, while 13 patients (37.1 %) were in partial remission and 3 patients (8.6 %) did not respond to treatment. Electrochemotherapy was performed an average of two times in this study. Using this method, it was possible to achieve a tumour control rate of 81 % over the observation period of 3 years 24. In another phase II study of 12 patients, the data is somewhat more sobering. This study showed complete and partial remission rates of 8 %. Stable disease was noted, however, in 75 % of patients, and a progression was observed in 8 % 25. The Benevento study of 12 elderly patients (mean age 76, 11 females, 1 male) published in 2012 shows significantly better response rates: using electrochemotherapy, it was possible to achieve a complete remission rate of metastases of 75.3 % and a partial remission rate of 17 % 4.
In 2011, Sersa et al. published a review that examined the response rate of electrochemotherapy for breast cancer thoracic wall recurrences. A total of eight studies were analysed. It was noted that the response rate was 89 % and the complete remission rate stood at 59 % 26. These results are encouraging, considering that the treatments were carried out in different centres and the tumours treated were of varying sizes and expansions 26.
Summary of the Advantages and Disadvantages of Electrochemotherapy
Advantages
The method is suitable for patients with severe co-morbidity and/or patients of an advanced age who have already exhausted all other treatments. Electrochemotherapy can be used to treat painful, bleeding and weeping metastases, as well as large lesions up to a depth of 4 cm. Furthermore, the implementation of electrochemotherapy can also result in the prevention of large scars and can also potentially result in preserved function e.g. of the arm in the case of severe lymphedema. On the whole, the quality of life of patients is improved by this procedure. The procedure is associated with a short hospital stay of 2–3 days. The side effects are minor and most patient do not require analgesics. In most cases, only a temporary reddening of the skin and the injection sites of the electrodes were visible in the area of the treated thoracic wall. Muscular fasciculation in terms of muscle aches is rare.
Local tumour control is possible with the aid of electrochemotherapy. Furthermore, in contrast to radiotherapy, for example, repeatability is possible in the case of this method.
The favourable cost-benefit ratio makes this method attractive.
Disadvantages
The procedure is performed under general anaesthesia and is associated with common anaesthesia-related risks. The use of intravenous bleomycin can result in the development of pulmonary fibrosis, particularly as this treatment is administered to patients who have previously received radiation therapy and who are comorbid, older, etc. A pulmonary function test should therefore be carried out prior to the procedure. During anaesthesia, the anaesthetist should select the lowest possible FiO2 in order to minimise the risk of pulmonary fibrosis. The electrode used during electrochemotherapy is a disposable instrument which is not particularly inexpensive. In the case of extensive electrochemotherapy in particular, the formation of large ulcers is possible due to increased tumour decomposition. As the use of electrochemotherapy is not curative but palliative, it is very likely that the treatment will have to be repeated, as the metastases become progressive over time.
Individual Experiences
Fig. 5 shows a 75 year-old patient with breast cancer cutaneous metastases in the area on the right of the thoracic wall which has been extensively pre-treated. The lesions have been present since 2009, and have been growing over the course of time. The patient has already undergone radiotherapy and further radiation therapy is no longer possible. The lesions on the thoracic wall are inoperable and there is no longer a chemotherapeutic option as the patient has already received 7 courses of chemotherapy. Electrochemotherapy was performed on the patient when the patient was experiencing a great deal of suffering caused by pain, itching and bleeding from the ulcerated metastases. Fig. 5 a shows the patient preoperatively. The results 1 week and 1 month after electrochemotherapy are shown in Figs. 5 b and c.
Fig. 5 a.
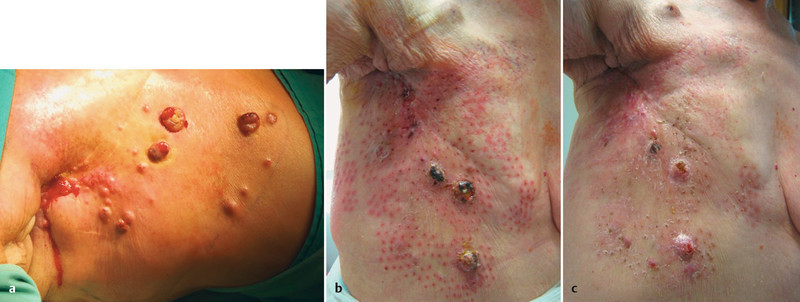
to c Result after electrochemotherapy (individual experience). Patient with numerous cutaneous metastases in the thoracic wall area due to known breast cancer. The patient complained of itching, pain and bleeding from the metastasis shown above. a shows the patient prior to chemotherapy. Figure b shows the patient 1 week after electrochemotherapy. It can clearly be seen that the ulcerating metastases have become necrotic in places, and are also significantly smaller. Figure c shows the patient 4 weeks after the procedure. After 4 weeks, the metastases have regressed further.
Conclusions for Clinical Practice
Electrochemotherapy is a promising method for the treatment of cutaneous and subcutaneous metastases in the local/locoregional recurrence of breast cancer in the palliative situation.
This innovative treatment improves the quality of life of patients and contributes to local tumour control. The side effects are minor, depending on the size of the area being treated. These are redness in the treated area of the thoracic wall, which can also lead to persistent skin changes (hyperpigmentation of the skin) and muscular fasciculation in this area.
Footnotes
Conflict of Interest None.
Supporting Information
German supporting informations for this article
References
- 1.Mir L M, Belehradek M, Domenge C. et al. [Electrochemotherapy, a new antitumor treatment: first clinical trial] C R Acad Sci III. 1991;313:613–618. [PubMed] [Google Scholar]
- 2.Mir L M, Orlowski S, Belehradek J jr.. et al. Electrochemotherapy potentiation of antitumour effect of bleomycin by local electric pulses. Eur J Cancer. 1991;27:68–72. doi: 10.1016/0277-5379(91)90064-k. [DOI] [PubMed] [Google Scholar]
- 3.Lluis M, Gehl J, Sersa G. et al. Standard operating procedures of the electrochemotherapy: instructions for the use of bleomycin or cisplatin administered either systemically or locally and electric pulses delivered by the Cliniporator™ by means of invasive or non-invasive electrodes. EJC Supplements. 2006;4:14–25. [Google Scholar]
- 4.Benevento R, Santoriello A, Perna G. et al. Electrochemotherapy of cutaneous metastastes from breast cancer in elderly patients: a preliminary report. BMC Surg. 2012;12 01:S6. doi: 10.1186/1471-2482-12-S1-S6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Grischke E-M, Wallwiener D, Souchon R. et al. Isolated loco-regional recurrence of breast cancer – established and innovative therapy concepts. Geburtsh Frauenheilk. 2013;73:611–622. [Google Scholar]
- 6.Sersa G Miklavcic D Electrochemotherapy of tumours J Vis Exp 200822pii: 1038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Umezawa H. Bleomycin and other antitumor antibiotics of high molecular weight. Antimicrob Agents Chemother (Bethesda) 1965;5:1079–1085. [PubMed] [Google Scholar]
- 8.Ichikawa T. [A new anti-neoplastic agent, bleomycin–its effect on squamous cell carcinoma] Nihon Ishikai Zasshi. 1969;61:487–497. [PubMed] [Google Scholar]
- 9.Schmidt-Matthiesen H Bastert G Wallwiener DHrsg.Gynäkologische Onkologie: Diagnostik, Therapie und Nachsorge – auf der Basis der AGO-Leitlinien. 7. Aufl. Stuttgart: Schattauer Verlag; 2002 [Google Scholar]
- 10.Lüllmann H, Mohr K, Wehling M. Stuttgart: Thieme; 2003. Pharmakologie und Toxikologie: Arzneimittelwirkungen verstehen – Medikamente gezielt einsetzen. 15. Aufl. p. 476. [Google Scholar]
- 11.Moller M G, Salwa S, Soden D M. et al. Electrochemotherapy as an adjunct or alternative to other treatments for unresectable or in-transit melanoma. Expert Rev Anticancer Ther. 2009;9:1611–1630. doi: 10.1586/era.09.129. [DOI] [PubMed] [Google Scholar]
- 12.Testori A, Tosti G, Martinoli C. et al. Electrochemotherapy for cutaneous and subcutaneous tumor lesions: a novel therapeutic approach. Dermatol Ther. 2010;23:651–661. doi: 10.1111/j.1529-8019.2010.01370.x. [DOI] [PubMed] [Google Scholar]
- 13.Marty M, Sersa G, Garbay J R. et al. Electrochemotherapy – An easy, highly effective and safe treatment of cutaneous and subcutaneous metastases: Results of ESOPE (European Standard Operating Procedures of Electrochemotherapy) study. EJC Supplements. 2006;4:3–13. [Google Scholar]
- 14.Campana L G, Mocellin S, Basso M. et al. Bleomycin-based electrochemotherapy: clinical outcome from a single institutionʼs experience with 52 patients. Ann Surg Oncol. 2009;16:191–199. doi: 10.1245/s10434-008-0204-8. [DOI] [PubMed] [Google Scholar]
- 15.Matthiessen L W, Chalmers R L, Sainsbury D C. et al. Management of cutaneous metastases using electrochemotherapy. Acta Oncol. 2011;50:621–629. doi: 10.3109/0284186X.2011.573626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mali B, Jarm T, Snoj M. et al. Antitumor effectiveness of electrochemotherapy: a systematic review and meta-analysis. Eur J Surg Oncol. 2013;39:4–16. doi: 10.1016/j.ejso.2012.08.016. [DOI] [PubMed] [Google Scholar]
- 17.Spratt J S. Locally recurrent cancer after radical mastectomy. Cancer. 1967;20:1051–1053. doi: 10.1002/1097-0142(196707)20:7<1051::aid-cncr2820200705>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 18.Valagussa P, Bonadonna G, Veronesi U. Patterns of relapse and survival following radical mastectomy. Analysis of 716 consecutive patients. Cancer. 1978;41:1170–1178. doi: 10.1002/1097-0142(197803)41:3<1170::aid-cncr2820410355>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 19.Bedwinek J. Natural history and management of isolated local-regional recurrence following mastectomy. Semin Radiat Oncol. 1994;4:260–269. doi: 10.1053/SRAO00400260. [DOI] [PubMed] [Google Scholar]
- 20.Arriagada R, Le M G, Rochard F. et al. Conservative treatment versus mastectomy in early breast cancer: patterns of failure with 15 years of follow-up data. Institut Gustave-Roussy Breast Cancer Group. J Clin Oncol. 1996;14:1558–1564. doi: 10.1200/JCO.1996.14.5.1558. [DOI] [PubMed] [Google Scholar]
- 21.Buchanan C L, Dorn P L, Fey J. et al. Locoregional recurrence after mastectomy: incidence and outcomes. J Am Coll Surg. 2006;203:469–474. doi: 10.1016/j.jamcollsurg.2006.06.015. [DOI] [PubMed] [Google Scholar]
- 22.van Tienhoven G, Voogd A C, Peterse J L. et al. Prognosis after treatment for loco-regional recurrence after mastectomy or breast conserving therapy in two randomised trials (EORTC 10801 and DBCG-82TM). EORTC Breast Cancer Cooperative Group and the Danish Breast Cancer Cooperative Group. Eur J Cancer. 1999;35:32–38. doi: 10.1016/s0959-8049(98)00301-3. [DOI] [PubMed] [Google Scholar]
- 23.Leonard R, Hardy J, van Tienhoven G. et al. Randomized, double-blind, placebo-controlled, multicenter trial of 6 % miltefosine solution, a topical chemotherapy in cutaneous metastases from breast cancer. J Clin Oncol. 2001;19:4150–4159. doi: 10.1200/JCO.2001.19.21.4150. [DOI] [PubMed] [Google Scholar]
- 24.Campana L G, Valpione S, Falci C. et al. The activity and safety of electrochemotherapy in persistent chest wall recurrence from breast cancer after mastectomy: a phase-II study. Breast Cancer Res Treat. 2012;134:1169–1178. doi: 10.1007/s10549-012-2095-4. [DOI] [PubMed] [Google Scholar]
- 25.Matthiessen L W, Johannesen H H, Hendel H W. et al. Electrochemotherapy for large cutaneous recurrence of breast cancer: a phase II clinical trial. Acta Oncol. 2012;51:713–721. doi: 10.3109/0284186X.2012.685524. [DOI] [PubMed] [Google Scholar]
- 26.Sersa G, Cufer T, Paulin S M. et al. Electrochemotherapy of chest wall breast cancer recurrence. Cancer Treat Rev. 2012;38:379–386. doi: 10.1016/j.ctrv.2011.07.006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
German supporting informations for this article


