Abstract
Objectives: Estrogen is suggested to participate in pathogenesis of irritable bowel syndrome (IBS), but expression of G protein-coupled estrogen receptor (GPER) in the colon of IBS patients has never been investigated. The aim of this study was to investigate the expression of GPER and classical estrogen receptors in the colon of IBS patients and healthy controls. Methods: Colonic biopsies were obtained by endoscopy from patients with IBS (n = 46) and healthy subjects (n = 13). Expression of GPER, estrogen receptor α (ERα) and estrogen receptor β (ERβ) in mast cells were measured by double-labelling immunofluorescence. Quantification of mRNA expression was performed for GPER, ERα and ERβ by real-time polymerase chain reaction. Results: Differential distribution of GPER, ERα and ERβ were detected in human colonic mucosa. The expression of GPER in the cytoplasm of mast cells and GPER-positive cells was significantly higher in diarrhea-predominant IBS (D-IBS) patients than that in constipation-predominant IBS (C-IBS, P < 0.001) patients and healthy subjects (P = 0.005). ERα and ERβ were not detected in majority of mast cells in colonic mucosa and no difference of immunostaining results for ERα and ERβ was found among these three groups. A positive correlation (r = 0.451, P = 0.011) between GPER-positive cell counts and abdominal pain severity was observed in D-IBS group. Relative mRNA expression of GPER in D-IBS was also higher than that in C-IBS (P = 0.018) and healthy subjects (P = 0.011). Conclusions: The present study, for the first time, demonstrated the expression of GPER in human colonic mucosa and its correlation with abdominal pain severity.
Keywords: G protein-coupled estrogen receptor, GPER, estrogen, estrogen receptor, irritable bowel syndrome
Introduction
Irritable bowel syndrome (IBS) is one of the most common functional gastrointestinal diseases, which is characterized by abdominal pain and bowel habits disturbance. Gender differences in prevalence of IBS were reported both in Asian and Western countries in previous studies [1-3], but the reason of female predominance in IBS remains unclear. Symptoms of female patients with IBS such as abdominal pain seemed to be related with menstrual cycle and were always exacerbated in the premenstrual and menstrual phases [4,5]. Sex hormones have been suggested to play important roles in IBS etiology [6,7].
As a common symptom in IBS, abdominal pain is associated with the changes of visceral perception, gastrointestinal motility and immune response. A number of studies conducted in animal models of visceral pain have demonstrated that sex hormones, especially estrogen, participated in modulating visceral sensitivity, but the mechanisms were still unclear [8-10]. By binding to two ligand-activated transcription factors: estrogen receptor α (ERα) and estrogen receptor β (ERβ), the typical role of estrogen on pain modulation is mostly shown in mediating genomic effects [11]. Nevertheless, estrogen is also involved in non-genomic effects. This is achieved by rapid cellular signaling, which is associated with membrane receptors [12].
G protein-coupled estrogen receptor (GPER), also known as GPR30, was first described in 1990s [13] and detected in numerous tissues [14]. GPER could mediate estrogen-dependent rapid signaling events, independent of classical estrogen nuclear receptors [15]. The association of estrogen with IBS etiology has been studied extensively, and most experiments were focusing on classical estrogen receptors, of which the effects need hours or days to be achieved. But estrogen mediated non-genomic effects (within a few minutes) on visceral sensitivity were rarely investigated. There were few reports about the expression of GPER in the colon of human and IBS patients neither.
A number of studies demonstrated mast cell (MC) counts and MC released mediators, such as mast cell tryptase (MCT) and histamine, were increased in colon of IBS patients [16,17]. It has been widely accepted that MC participates in regulating visceral hypersensitivity, intestinal motility and immune response. Estrogen could regulate cytokine release in human mast cells [18] via a non-genomic effect [19]. It is suggested that there are associations among estrogen, mast cells and IBS pathogenesis.
Our hypothesis is that estrogen receptors are expressed in mast cells in human colon. In the present study, we aimed to examine the expression of ERα, ERβ and GPER in the colonic mucosa of IBS patients and healthy controls, and to investigate whether or not these estrogen receptors are located in mast cells of the colon.
Materials and methods
Subjects and tissue samples
The study was approved by the ethics committee of the Second Affiliated Hospital of Xi’an Jiaotong University. As showed in Table 1, 46 IBS patients (D-IBS 31 and C-IBS 15) fulfilling the Rome III criteria were recruited from September 2011 to December 2013 in the Department of Gastroenterology, the Second Affiliated Hospital of Xi’an Jiaotong University. 13 Healthy controls (HCs) were selected in subjects with normal colon and no gastrointestinal symptoms (such as patients with polyps and family history of colon cancer). Exclusion criteria for IBS patients and HCs were as following: use of mast cell stabilizers, NSAIDS, corticosteroids, estradiol and anti-estrogen agents in the past 2 weeks, history of allergic diseases, history of ovariectomy, other gastrointestinal diseases and severe organic disorders. Patients and HCs gave written informed consent before the procedures.
Table 1.
Clinical characteristics of the patients and healthy controls
| Characteristics | D-IBS | C-IBS | HCs |
|---|---|---|---|
| Numbers | 31 | 15 | 13 |
| Gender (F/M) | 20/11 | 10/5 | 8/5 |
| Age (median/range) | 35.5/20-55 | 37/19-58 | 47/35-61 |
| Pain severity (mean ± SD) | 1.94 ± 0.89 | 1.40 ± 0.63 | 0 |
| Duration (median/range) (months) | 48/8-240 | 54/9-180 | 0 |
| Allergic diseases | none | none | none |
| Other GI diseases | none | none | Reexamination for polyps 7; Family history of colorectal cancer 4; none 2 |
| Drugs using within 2 weeks | none | none | none |
F/M = female/male; GI = gastrointestinal, HCs = healthy controls.
Before the colonoscopy, all the subjects were performed with a complete medical history and physical examination. Severity of abdominal pain was scored according to published IBS related studies [20,21]: 0, absent; 1, mild (not influencing activities); 2, relevant (diverting from, but not urging modification of activities); 3, severe (influencing activities markedly enough to urge modifications); 4, extremely severe (precluding daily activities). Other information about gender, age, duration of symptoms were also obtained. After taking polyethylene glycol-electrolyte solution for 4-6 hours, all the subjects underwent colonoscopy. Four mucosal biopsy specimens were taken from the sigmoid colon and immediately put into liquid nitrogen and stored at -70°C until use.
Immunofluorescence
Specimens were fixed in 10% formalin for H&E staining and immunofluorescence staining. Routine H&E staining were performed and analyzed by experienced pathologists to exclude inflammation of the colonic mucosa. After dehydration with 30% sucrose solution, specimens were immersed in OCT embedding medium and snap frozen. Cryostat sections (5 μm) were cut at -27°C onto glass slides and stored at -20°C until use.
For single-labelling immunofluorescence of estrogen receptors, sections were incubated with 10% goat serum for 30 min to block nonspecific binding sites, followed by incubating overnight with antibodies (rabbit polyclonal) against G-protein coupled receptor 30 (Abcam, Hong Kong, China, 1:200), ERα (Santa Cruz Biotechnology, USA, 1:100), ERβ (Santa Cruz Biotechnology, USA, 1:100), respectively at 4°C. After PBS washes, sections were then incubated with anti-rabbit IgG-FITC antibody produced in goat (Sigma, Germany, 1:100) for 1 h at 37°C in the dark.
For double labelling immunofluorescence, primary antibodies of estrogen receptors were as described above. To detect mast cell in the colon, anti-mast cell tryptase antibody (Mouse monoclonal, Abcam, Hong Kong, 1:200) was used as primary antibody. The mixture of antibodies against mast cell tryptase and a single estrogen receptor were incubated overnight at 4°C, washed in PBS, and incubated with goat anti-mouse IgG-TRITC antibody (ZSGB, China, 1:100) and goat anti-rabbit IgG-FITC antibody (Sigma, Germany, 1:100) for 1 h at 37°C in the dark. To identifying the cell morphology of the nuclei, the sections were counterstained with DAPI (Sigma, Germany) for 3 min followed by PBS washes. The negative controls were incubated with PBS instead of the primary antibodies.
All sections were mounted in a fluorescence microscope (Nikon ECLPSE TE2000-U) for examination. Counts of positive stained-cells were performed at 200 × magnification. The number of positive-stained cells were counted by 2 researchers independently in three non-overlapping fields and expressed as average positive-stained per high power field (cells/hpf).
Real-time PCR
Total RNA was extracted from the frozen tissue using the TRIzol reagent (TaKaRa, Japan) according to the manufacturer’s instructions and stored at 4°C. The amount of RNA was estimated by UV spectrophotometry (NanoDrop Technologies, USA) at 260 nm. 2 μg sample of total RNA was used to synthesize cDNA using a PrimeScript RT Master Mix (TaKaRa, Japan).
The quantitative real-time polymerase chain reaction (PCR) was performed with the ABI PRISM 7500 Sequence Detection System (Applied Biosystems) using SYBR® Premix Ex Taq™ (TaKaRa, Japan) in accordance with the manufacturer’s protocol. The primers used in the present study were as follows: GPER (172 bp), forward CAACCTCGCCGCCTTCTC, reverse CTGCTCGGTGCTGTCTGG; ERα (185 bp), forward CCTGATGATTGGTCTCGTCTG, reverse GGCACACAAACTCCTCTCC; ERβ (197 bp), forward ATACCTTCCTCCTATGTAGAC, reverse TGTGATAACTGGCGATGG; β-actin (156 bp), forward ACTTAGTTGCGTTACACCCTT, reverse GTCACCTTCACCGTTCCA. Briefly, The 25 μl PCR reactions consisted of 12.5 μl SYBR green, 1 μl PCR forward and 1 μl PCR reverse primers, 2 μl cDNA and 8.5 μl ddH2O. The amplification protocol was: 3 min at 95°C, followed by denaturing at 94°C for 30 s, annealing at 55 to 56°C (primers dependent) for 30 s and extension at 72°C for 30 s for a total of 45 amplification cycles. All the experiments were repeated triplicate and relative mRNA quantities of GPER, ERα and ERβ were normalized to β-actin expression.
Statistical analysis
All data were analyzed using the SPSS 16.0 software (SPSS Inc., Chicago). Values were presented as mean ± standard deviation (SD). The comparison of positive-stained cell counts were performed using non-parametric Kruskal-Wallis test for group differences and Mann-Whitney U test for within group differences. Correlations between cell counts and symptoms were analyzed using Spearman correlation test. The relative mRNA expression levels were compared using one-way analysis of variance (ANOVA) and Dunnett t test. Two-tailed P value less than 0.05 was considered statistically significant.
Results
Localization of GPER in mast cell of colonic mucosa
All sections for routine H&E staining were reported as normal. GPER was detected in colonic mucosa of all the IBS patients and healthy controls. It distributed mainly in the cells surrounding mucosal glands within lamina propria (Figure 1B). Double staining with antibodies to GPER and tryptase (Figure 1A) showed GPER was present in majority of tryptase-positive mast cells of colonic mucosa (Figure 1C and 1D). By counterstaining with DAPI (Figure 1E), we found GPER was located in the cytoplasm of mast cells and no positive-staining of the nucleus were observed (Figure 1F).
Figure 1.
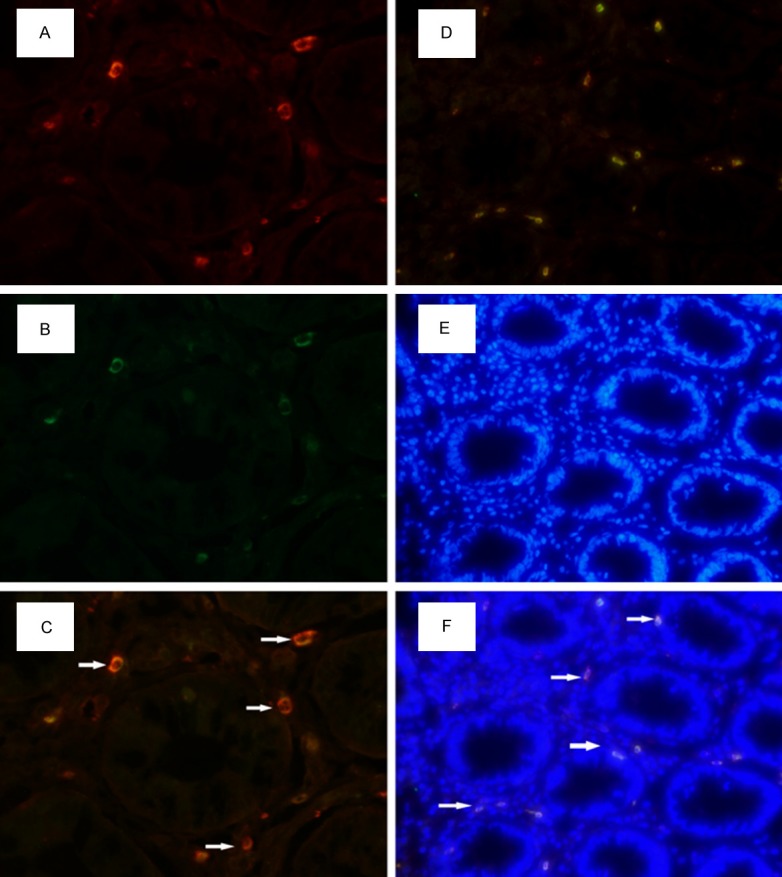
Localization of GPER in mast cells of colonic mucosa. GPER was mainly detected surrounding mucosal glands within lamina propria (B and D). GPER (B, green) and tryptase (A, red) were observed to coexpress (C and D, yellow) in mast cells within lamina propria. With DAPI staining (E, blue), GPER was observed to locate in the cytoplasm (F, yellow). The staining was showed with 400 × magnification (A-C) and 200 × magnification (D-F).
Increased GPER-positive cells in D-IBS patients
As showed in Figure 2A, mean counts of GPER-positive cells in colonic mucosa of D-IBS patients were 23.5 ± 5.2 (cells/hpf), which was higher than that of C-IBS patients (15.0 ± 5.2, P < 0.001) and healthy controls (17.1 ± 6.6, P = 0.005). However, no differences were found between C-IBS patients and healthy controls (P = 0.344). There was also no difference of GPER-positive cell counts between females and males (24.0 ± 4.8 vs. 22.8 ± 6.2, P = 0.847).
Figure 2.
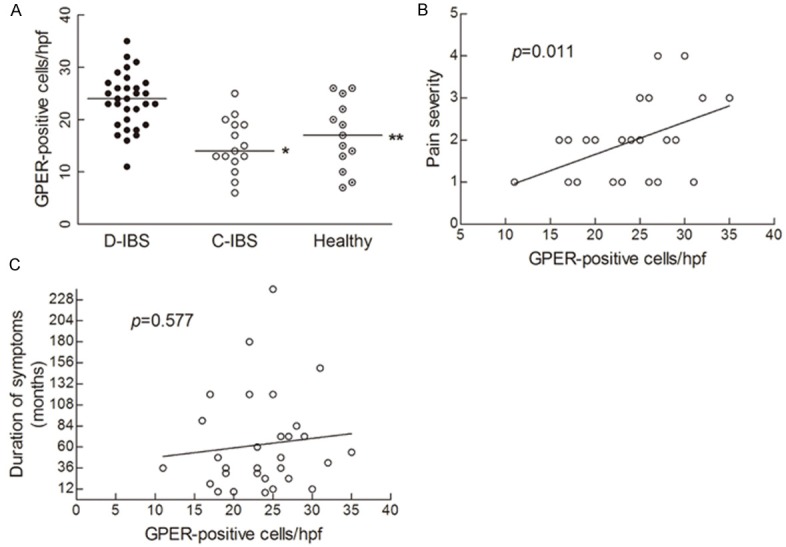
Comparison of GPER-positive cell counts and the correlation with symptoms in D-IBS patients. Mean counts of GPER-positive (A) cells in colonic mucosa of D-IBS patients were significantly higher than that of C-IBS (*P < 0.001) and HCs (**P = 0.005), compared by Mann-Whitney U test. GPER-positive cells counts had positive correlation (B) with pain severity (r = 0.451, P = 0.011) and no correlation (C) with duration of symptoms (r = 0.104, P = 0.577), compared by Spearman correlation test.
Correlation of GPER-positive cell counts with symptoms in D-IBS patients
As showed in Table 1, all patients with D-IBS complained of abdominal pain, with an average score of 1.94 (95% CI 1.61-2.26). Duration of symptoms for D-IBS patients ranged from 6 to 240 with a median of 48 months. Analyzed by Spearman correlation test (Figure 2B and 2C), there was a significant positive correlation (r = 0.451, P = 0.011) between GPER-positive cell counts and abdominal pain severity in the D-IBS group. There was no correlation (r = 0.104, P = 0.577) of GPER-positive cell counts with symptoms duration.
Distribution of ERα and ERβ in colonic mucosa
Immunofluorescence for ERα and ERβ was performed in the same condition. ERα-positive cells (Figure 3B) in the colonic mucosa were detected in 10 of 31 D-IBS patients, 4 of 15 C-IBS patients and 4 of 13 HCs. ERβ-positive cells (Figure 3E) were detected in 13 of 31 D-IBS patients, 6 of 15 C-IBS patients and 7 of 13 HCs. No differences of positive-staining percentages for ERα (P = 0.929) and ERβ (P = 0.722) were found between the IBS patients and HCs. Both ERα (Figure 3B) and ERβ (Figure 3E) were detected mostly in lamina propria and minorly in epithelial layer. By counterstaining with DAPI, ERα and ERβ were mainly detected with a nuclear localization in cells within lamina propria, but we did not detect the colocalization of ERα (Figure 3C) or ERβ (Figure 3F) with MCT (Figure 3A and 3D) in these samples, suggesting ERα and ERβ did not express in mucosal mast cells of human colon.
Figure 3.

Distribution of ERα and ERβ in human colonic mucosa. ERα (B, green) and ERβ (E, green) were detected mostly in lamina propria and minorly in epithelial layer. ERα and ERβ, by double staining with tryptase (A and D; red), were not detected in majority of mast cells (C and F). The staining results were showed with 200 × magnification.
Relative mRNA expression of GPER, ERα and ERβ
We found by real-time quantitative PCR that the mRNA of GPER were detected in colonic mucosa of all the subjects. The average mRNA level (normalized to β-actin) of GPER were 0.839 (95% CI 0.719-0.960) in D-IBS patients, 0.567 (95% CI 0.415-0.721) in C-IBS patients and 0.513 (95% CI 0.333-0.692) in HCs, and mRNA level of GPER was significantly higher than that in C-IBS (P = 0.018) and healthy subjects (P = 0.011). ERα and ERβ had low levels of mRNA expression and there were no differences of mRNA levels for ERα (P = 0.150) and ERβ (P = 0.178) between IBS patients and the healthy controls. The results were showed in Figure 4.
Figure 4.
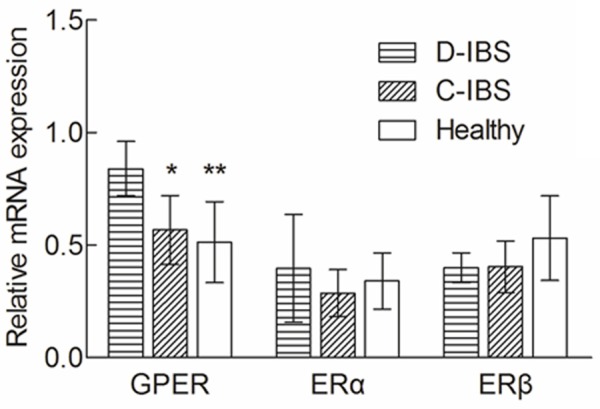
Relative mRNA expression of GPER, ERα and ERβ. Compared with ANOVA and Dunnett t test, relative mRNA expression of GPER in D-IBS was higher than that in C-IBS (*P = 0.018) and healthy subjects (**P = 0.011). No significant differences were found among the three groups for ERα (P = 0.150) and ERβ (P = 0.178).
Discussion
To the best of our knowledge, G protein-coupled estrogen receptor (GPER) has never been investigated in human colonic mucosa. We found that GPER was located in mast cells of the lamina propria in human colonic mucosa. Our results offered an additional site distinct from classical estrogen receptors for estrogen to act on mast cells in human colon.
Recent studies suggested that estrogen could modulate visceral sensitivity in animal models [22,23], and the effects were achieved by binding with classical nuclear estrogen receptors [10] to induce long-term genomic processes. But IBS-related symptoms as abdominal pain and discomfort always appear and disappear rapidly. It is supposed that rapid non-genomic effects of estrogen may be involved in regulating the visceral perception. GPER, an intracellular transmembrane estrogen receptor [24], had been reported to induce rapid pronociceptive estrogenic effects [25] at the spinal level, but association of GPER with colonic mast cells has never been examined. We showed that GPER was located specifically in the cytoplasm of mucosal mast cells of colon, but whether GPER located at plasma membrane or cytoplasm is still controversial [24,26] and need to be clearly resolved.
Mast cells were described in a number of studies to participate in pathogenesis of visceral hypersensitivity [27,28] and its stabiliser could improve symptoms in IBS patients [29]. However, relationship of gender difference in IBS with mast cell behavior was not defined. Previous studies have demonstrated that estrogen nuclear receptors were expressed in mast cells and might regulate mast cell behavior by mediating long-term genomic effects [30]. It was also demonstrated that estrogen activated mast cells via a non-genomic effect mediated by ERα [19], yet in the study design effect of GPER was not excluded and function of estrogen receptors located in colonic mast cells were not investigated. Our findings were not in consistent with these results. We found ERα and ERβ were not detected in majority of mast cells of colonic mucosa and no differences were found of expression for ERα and ERβ in colonic mucosa between the IBS patients and healthy controls, which suggested that effects of estrogen on colonic mast cells were not mediated directly by ERα and ERβ. Furthermore, by immunofluorescene methods, GPER was also detected in samples of colonic mucosa negative-stained with ERα and ERβ. In our study, increased GPER-positive cells were found in D-IBS patients compared to C-IBS patients and healthy subjects, and a positive correlation between GPER expression and abdominal pain severity was observed in D-IBS patients. We suggested that GPER in colonic mucosa might mediate effects of estrogen on mast cells independently from traditional estrogen receptors to influence visceral sensitivity.
Mechanisms underlying overexpression of GPER in the colon of D-IBS patients were not investigated in this study and remain unclear. A possible explanation is that GPER is mostly located in colonic mast cells, and its expression might be increased accompanied with elevated mast cell numbers. Mechanism of estrogenic effects on colonic mast cells via GPER was still unclear. It was suggested in a study [26] that GPER mediated peripheral mechanism for pain modulation might be involved. We supposed that increased mast cells in D-IBS patients provided more binding sites of GPER for estrogen and the occurrence of abdominal pain might be associated with the estrogen level. Females always have higher level of estrogen than males, which can explain why female was easier to be disturbed by IBS related symptoms. In our study we did not found correlation between GPER expression and symptoms duration. An explanation is that GPER mediated estrogenic effects is rapid and not long acting as genomic effects mediated by ERα and ERβ. We did not study GPER expression in IBS patients of mixed type (IBS-M) and undefined type (IBS-U) because of insufficient recruitments (IBS-M 2, IBS-U 0).
In conclusion, we demonstrated that GPER rather than ERα and ERβ was located in mast cells of the human colon and increased expression of GPER in D-IBS patients was correlated with abdominal pain severity. It was suggested that GPER-mediated estrogenic effects on IBS pathogenesis might be associated with activation of mast cells in the colon. Our findings provided a new direction for understanding the pathogenesis of gender differences in IBS and the mechanisms should be investigated by further studies.
Acknowledgements
This study was supported by the National Spark Plan Project of China (No. 2011GAB50001). We thank Ms Ni Lei (Department of Pathology, Medical College of Xi’an Jiaotong University) for advices and excellent technical support.
Disclosure of conflict of interest
None.
References
- 1.Cain KC, Jarrett ME, Burr RL, Rosen S, Hertig VL, Heitkemper MM. Gender differences in gastrointestinal, psychological, and somatic symptoms in irritable bowel syndrome. Dig Dis Sci. 2009;54:1542–1549. doi: 10.1007/s10620-008-0516-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chang FY, Lu CL, Chen TS. The current prevalence of irritable bowel syndrome in Asia. J Neurogastroenterol Motil. 2010;16:389–400. doi: 10.5056/jnm.2010.16.4.389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Anbardan SJ, Daryani NE, Fereshtehnejad SM, Taba Taba Vakili S, Keramati MR, Ajdarkosh H. Gender Role in Irritable Bowel Syndrome: A comparison of irritable bowel syndrome module (ROME III) between male and female patients. J Neurogastroenterol Motil. 2012;18:70–77. doi: 10.5056/jnm.2012.18.1.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jane ZY, Chang CC, Lin HK, Liu YC, Chen WL. The association between the exacerbation of irritable bowel syndrome and menstrual symptoms in young Taiwanese women. Gastroenterol Nurs. 2011;34:277–286. doi: 10.1097/SGA.0b013e3182248708. [DOI] [PubMed] [Google Scholar]
- 5.Kane SV, Sable K, Hanauer SB. The menstrual cycle and its effect on inflammatory bowel disease and irritable bowel syndrome: a prevalence study. Am J Gastroenterol. 1998;93:1867–1872. doi: 10.1111/j.1572-0241.1998.540_i.x. [DOI] [PubMed] [Google Scholar]
- 6.Houghton LA, Lea R, Jackson N, Whorwell PJ. The menstrual cycle affects rectal sensitivity in patients with irritable bowel syndrome but not healthy volunteers. Gut. 2002;50:471–474. doi: 10.1136/gut.50.4.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ouyang A, Wrzos HF. Contribution of gender to pathophysiology and clinical presentation of IBS: should management be different in women? Am J Gastroenterol. 2006;101:S602–609. doi: 10.1111/j.1572-0241.2006.00975.x. [DOI] [PubMed] [Google Scholar]
- 8.Chaloner A, Greenwood-Van Meerveld B. Sexually dimorphic effects of unpredictable early life adversity on visceral pain behavior in a rodent model. J Pain. 2013;14:270–280. doi: 10.1016/j.jpain.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 9.Sanoja R, Cervero F. Estrogen-dependent abdominal hyperalgesia induced by ovariectomy in adult mice: a model of functional abdominal pain. Pain. 2005;118:243–253. doi: 10.1016/j.pain.2005.08.021. [DOI] [PubMed] [Google Scholar]
- 10.Cao DY, Ji Y, Tang B, Traub RJ. Estrogen receptor beta activation is antinociceptive in a model of visceral pain in the rat. J Pain. 2012;13:685–694. doi: 10.1016/j.jpain.2012.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Welboren WJ, Sweep FC, Span PN, Stunnenberg HG. Genomic actions of estrogen receptor alpha: what are the targets and how are they regulated? Endocr Relat Cancer. 2009;16:1073–1089. doi: 10.1677/ERC-09-0086. [DOI] [PubMed] [Google Scholar]
- 12.Rouayrenc JF, Vignon F, Bringer J, Pujol P. [Non-genomic steroid effects: estrogen action revisited] . Ann Endocrinol (Paris) 2000;61:517–523. [PubMed] [Google Scholar]
- 13.Carmeci C, Thompson DA, Ring HZ, Francke U, Weigel RJ. Identification of a gene (GPR30) with homology to the G-protein-coupled receptor superfamily associated with estrogen receptor expression in breast cancer. Genomics. 1997;45:607–617. doi: 10.1006/geno.1997.4972. [DOI] [PubMed] [Google Scholar]
- 14.Kvingedal AM, Smeland EB. A novel putative G-protein-coupled receptor expressed in lung, heart and lymphoid tissue. FEBS Lett. 1997;407:59–62. doi: 10.1016/s0014-5793(97)00278-0. [DOI] [PubMed] [Google Scholar]
- 15.Prossnitz ER, Arterburn JB, Smith HO, Oprea TI, Sklar LA, Hathaway HJ. Estrogen signaling through the transmembrane G protein-coupled receptor GPR30. Annu Rev Physiol. 2008;70:165–190. doi: 10.1146/annurev.physiol.70.113006.100518. [DOI] [PubMed] [Google Scholar]
- 16.O‘Sullivan M, Clayton N, Breslin NP, Harman I, Bountra C, McLaren A, O‘Morain CA. Increased mast cells in the irritable bowel syndrome. Neurogastroenterol Motil. 2000;12:449–457. doi: 10.1046/j.1365-2982.2000.00221.x. [DOI] [PubMed] [Google Scholar]
- 17.Piche T, Saint-Paul MC, Dainese R, Marine-Barjoan E, Iannelli A, Montoya ML, Peyron JF, Czerucka D, Cherikh F, Filippi J, Tran A, Hebuterne X. Mast cells and cellularity of the colonic mucosa correlated with fatigue and depression in irritable bowel syndrome. Gut. 2008;57:468–473. doi: 10.1136/gut.2007.127068. [DOI] [PubMed] [Google Scholar]
- 18.Kim MS, Chae HJ, Shin TY, Kim HM, Kim HR. Estrogen regulates cytokine release in human mast cells. Immunopharmacol Immunotoxicol. 2001;23:495–504. doi: 10.1081/iph-100108596. [DOI] [PubMed] [Google Scholar]
- 19.Zaitsu M, Narita S, Lambert KC, Grady JJ, Estes DM, Curran EM, Brooks EG, Watson CS, Goldblum RM, Midoro-Horiuti T. Estradiol activates mast cells via a non-genomic estrogen receptor-alpha and calcium influx. Mol Immunol. 2007;44:1977–1985. doi: 10.1016/j.molimm.2006.09.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Piche T, Barbara G, Aubert P, Bruley des Varannes S, Dainese R, Nano JL, Cremon C, Stanghellini V, De Giorgio R, Galmiche JP, Neunlist M. Impaired intestinal barrier integrity in the colon of patients with irritable bowel syndrome: involvement of soluble mediators. Gut. 2009;58:196–201. doi: 10.1136/gut.2007.140806. [DOI] [PubMed] [Google Scholar]
- 21.Wilcz-Villega E, McClean S, O’Sullivan M. Reduced E-cadherin expression is associated with abdominal pain and symptom duration in a study of alternating and diarrhea predominant IBS. Neurogastroenterol Motil. 2014;26:316–25. doi: 10.1111/nmo.12262. [DOI] [PubMed] [Google Scholar]
- 22.Tang B, Ji Y, Traub RJ. Estrogen alters spinal NMDA receptor activity via a PKA signaling pathway in a visceral pain model in the rat. Pain. 2008;137:540–549. doi: 10.1016/j.pain.2007.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Moussa L, Bezirard V, Salvador-Cartier C, Bacquie V, Houdeau E, Theodorou V. A new soy germ fermented ingredient displays estrogenic and protease inhibitor activities able to prevent irritable bowel syndrome-like symptoms in stressed female rats. Clin Nutr. 2013;32:51–58. doi: 10.1016/j.clnu.2012.05.021. [DOI] [PubMed] [Google Scholar]
- 24.Revankar CM, Cimino DF, Sklar LA, Arterburn JB, Prossnitz ER. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science. 2005;307:1625–1630. doi: 10.1126/science.1106943. [DOI] [PubMed] [Google Scholar]
- 25.Deliu E, Brailoiu GC, Arterburn JB, Oprea TI, Benamar K, Dun NJ, Brailoiu E. Mechanisms of G protein-coupled estrogen receptor-mediated spinal nociception. J Pain. 2012;13:742–754. doi: 10.1016/j.jpain.2012.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lu CL, Hsieh JC, Dun NJ, Oprea TI, Wang PS, Luo JC, Lin HC, Chang FY, Lee SD. Estrogen rapidly modulates 5-hydroxytrytophan-induced visceral hypersensitivity via GPR30 in rats. Gastroenterology. 2009;137:1040–1050. doi: 10.1053/j.gastro.2009.03.047. [DOI] [PubMed] [Google Scholar]
- 27.Cremon C, Gargano L, Morselli-Labate AM, Santini D, Cogliandro RF, De Giorgio R, Stanghellini V, Corinaldesi R, Barbara G. Mucosal immune activation in irritable bowel syndrome: gender-dependence and association with digestive symptoms. Am J Gastroenterol. 2009;104:392–400. doi: 10.1038/ajg.2008.94. [DOI] [PubMed] [Google Scholar]
- 28.Larauche M. Novel insights in the role of peripheral corticotropin-releasing factor and mast cells in stress-induced visceral hypersensitivity. Neurogastroenterol Motil. 2012;24:201–205. doi: 10.1111/j.1365-2982.2011.01867.x. [DOI] [PubMed] [Google Scholar]
- 29.Klooker TK, Braak B, Koopman KE, Welting O, Wouters MM, van der Heide S, Schemann M, Bischoff SC, van den Wijngaard RM, Boeckxstaens GE. The mast cell stabiliser ketotifen decreases visceral hypersensitivity and improves intestinal symptoms in patients with irritable bowel syndrome. Gut. 2010;59:1213–1221. doi: 10.1136/gut.2010.213108. [DOI] [PubMed] [Google Scholar]
- 30.Zierau O, Zenclussen AC, Jensen F. Role of female sex hormones, estradiol and progesterone, in mast cell behavior. Front Immunol. 2012;3:169. doi: 10.3389/fimmu.2012.00169. [DOI] [PMC free article] [PubMed] [Google Scholar]


