Abstract
Background and Aims
Changes occurring in the macromolecular traits of cell wall components in elm wood following attack by Ophiostoma novo-ulmi, the causative agent of Dutch elm disease (DED), are poorly understood. The purpose of this study was to compare host responses and the metabolic profiles of wood components for two Dutch elm (Ulmus) hybrids, ‘Groeneveld’ (a susceptible clone) and ‘Dodoens’ (a tolerant clone), that have contrasting survival strategies upon infection with the current prevalent strain of DED.
Methods
Ten-year-old plants of the hybrid elms were inoculated with O. novo-ulmi ssp. americana × novo-ulmi. Measurements were made of the content of main cell wall components and extractives, lignin monomer composition, macromolecular traits of cellulose and neutral saccharide composition.
Key Results
Upon infection, medium molecular weight macromolecules of cellulose were degraded in both the susceptible and tolerant elm hybrids, resulting in the occurrence of secondary cell wall ruptures and cracks in the vessels, but rarely in the fibres. The 13C nuclear magnetic resonance spectra revealed that loss of crystalline and non-crystalline cellulose regions occurred in parallel. The rate of cellulose degradation was influenced by the syringyl:guaiacyl ratio in lignin. Both hybrids commonly responded to the medium molecular weight cellulose degradation with the biosynthesis of high molecular weight macromolecules of cellulose, resulting in a significant increase in values for the degree of polymerization and polydispersity. Other responses of the hybrids included an increase in lignin content, a decrease in relative proportions of d-glucose, and an increase in proportions of d-xylose. Differential responses between the hybrids were found in the syringyl:guaiacyl ratio in lignin.
Conclusions
In susceptible ‘Groeneveld’ plants, syringyl-rich lignin provided a far greater degree of protection from cellulose degradation than in ‘Dodoens’, but only guaiacyl-rich lignin in ‘Dodoens’ plants was involved in successful defence against the fungus. This finding was confirmed by the associations of vanillin and vanillic acid with the DED-tolerant ‘Dodoens’ plants in a multivariate analysis of wood traits.
Keywords: Cellulose degradation, crystallinity, Dutch elm disease, Ophiostoma novo-ulmi, syringyl to guaiacyl ratio, lignin, Ulmus
INTRODUCTION
The ascomycetous fungus Ophiostoma novo-ulmi is the causative agent of the current Dutch elm disease (DED) pandemic, which has ravaged both European and North American elm populations. This fungus is polytypic, spreading in the form of two subspecies, subsp. novo-ulmi and subsp. americana, previously referred to as the Eurasian and North American races, respectively (Brasier and Kirk, 2001). Rapid emergence of hybrids between these two subspecies has recently been reported, and it is likely that complex hybrid swarms are now expanding across the European continent (Brasier and Kirk, 2010). Several European elm breeding programmes were initiated in an attempt to combat the disease. Ulmus glabra × U. minor hybrids are frequently included in these programmes as they often show lower susceptibility to the pathogen than that found in U. glabra trees. In addition, Asian elms, particularly U. wallichiana and U. pumila, proved to be a useful additional source of DED-resistance genes with the advent of the second disease pandemic in Europe during the 1970s (Heybroek, 1983; Smalley and Guries, 1993; Santini et al., 2008, 2010). For better control of this disease, current elm breeding programmes also integrate biotechnological approaches (Pijut et al., 1990; Fenning et al., 1996; Newhouse et al., 2007; Shukla et al., 2012).
The pathogenic fungus spreads within the secondary xylem vessels of infected trees, causing the formation of vessel plugs due to tyloses and gels (Ouellette et al., 2004), which ultimately results in foliar wilting and subsequent tree death (Newbanks et al., 1983). The wilt syndrome is apparently a result of interactions between fungal metabolites and the tree (Scheffer et al., 1987). The fungus produces hydrophobin cerato-ulmin (a parasitic fitness factor; Temple et al., 1997), phytotoxic peptidorhamnomannan (Strobel et al., 1978; Sticklen et al., 1991), tissue-invading structures that are thought to be involved in cavitation of the water column and alteration of parenchyma cells (Ouellette et al., 2004), and cell wall-degrading enzymes such as glucanases, glucosidases (Przybył et al., 2006), xylanases, laccases (Binz and Canevascini, 1996a, b), exo-glycanases and glycosidases (Svaldi and Elgersma, 1982). Several anatomical parameters of wood have been found to be related to DED resistance, and their possible use in early screening has been discussed. Unfortunately, the shortage of resistant trees tested has frequently made it difficult to obtain conclusive or reproducible results (Martín et al., 2008).
Plant interactions with pathogens are characterized by the deployment of chemical signals and highly coordinated reprogramming of metabolites (Allwood et al., 2006; Lloyd et al., 2011). Biochemical profiles of biomacromolecules and their metabolic fingerprinting provide rapid classification of plant samples according to their origin and physiological state. The ability to obtain biochemical profiles and fingerprints for elms would be useful in studies to characterize mechanisms of DED or to identify resistant elms (Martín et al., 2005b). Changes in the levels of cell wall components of elm wood after inoculations with O. novo-ulmi isolates have been investigated for the most part by Fourier transform–infrared (FT-IR) spectroscopy to discriminate between resistant and susceptible elms, as well as to identify metabolic profiles related to host resistance (Martín et al., 2005a, 2007, 2008). However, changes in the macromolecular traits of cell wall components of elm wood affected by DED are mostly unknown.
In this study we compared lignin monomer composition, macromolecular traits of cellulose and neutral saccharide composition between two Dutch elm hybrids (‘Groeneveld’ and ‘Dodoens’) that possess contrasting tolerance to DED. We addressed the following specific questions. (1) Is the lignin macromolecular structure, quantified by the ratio of syringyl (S) to guaiacyl (G) subunits, involved in defence against O. novo-ulmi and does the S/G ratio change upon infection? (2) How do the macromolecular traits of cellulose (polydispersity, degree of polymerization, crystallinity) change upon infection? (3) Is the content of neutral saccharides influenced by infection?
MATERIALS AND METHODS
Plant material and study site
The experiments were conducted on clonally micropropagated (Krajňáková and Longauer, 1996) plants of the Dutch elm hybrid cultivars ‘Groeneveld’ [(Ulmus × hollandica 49) × U. minor ssp. minor 1] and ‘Dodoens’ (open-pollinated U. glabra ‘Exoniensis’ × U. wallichiana P39) growing in an experimental field plot at Banská Belá, Slovakia (48°28′N, 18°57′E, 590 m a.s.l.). According to the meteorological station at Arboretum Kysihýbel in Banská Štiavnica (540 m a.s.l.), located 3·6 km south-west of the study site, the climate of the area is characterized by a mean annual temperature of 7·7 °C and a mean annual precipitation of 831 mm. The study site soil, which has a silt loam texture, is identified as an Eutric Cambisol formed from the slope deposits of volcanic rocks (andesite and pyroclastic materials).
Fungus identification and inoculation and wood sampling
Ten-year-old plants of similar size for each cultivar were selected and inoculated with Ophiostoma novo-ulmi ssp. americana × novo-ulmi isolate M3 according to the procedure of Solla et al. (2005). The spore suspension (1 × 107 spores mL–1) was inoculated into the current annual ring, 20 cm above the base of the stem. Hybrid isolate M3 belongs to mating type B and was isolated from an infected elm tree in Brno, Czech Republic. This isolate proved to be ssp. americana in a fertility test and had a cerato-ulmi (cu) gene profile of ssp. americana, but also had a colony-type (col1) gene profile of ssp. novo-ulmi (Konrad et al., 2002; Dvořák et al., 2007). The infected ‘Groeneveld’ plants showed no tolerance to DED and died at the end of the growing season after fungal inoculation (Dvořák et al., 2009). However, the infected ‘Dodoens’ plants showed high tolerance to O. novo-ulmi ssp. americana × novo-ulmi (Ďurkovič et al., 2013). No signs of physiological weakening were found in these plants, and they grew for an additional 3 and a half years following fungal infection.
Discs 4 cm thick were sawn at a height of 1 m from the trunks of both hybrids. For ‘Groeneveld’ plants, the sampling was done half a year after inoculation. For ‘Dodoens’ plants, the sampling was done 3 and a half years after inoculation. The discs were symmetrical and visibly free of reaction wood and did not contain knotwood. The infection zones, concentrated within the sixth annual ring at a height of 1 m, were separated from the discs of infected plants of both hybrids. The seventh and eighth annual rings (taken at a height of 1 m) were separated only from the discs of infected ‘Dodoens’ plants (the two annual rings were mixed together because some portion of the seventh annual ring was damaged and lost during separation of the sixth annual ring). The sixth, seventh and eighth annual rings, respectively, separated from the discs of non-infected ‘Groeneveld’ and ‘Dodoens’ plants, were used as controls. Figure 1A–I presents scanning electron microscopy (SEM) images of plant material responding to the fungal inoculation that was used in this study. The experiments were conducted on wood samples taken from four infected and four non-infected plants per hybrid.
Fig. 1.
Scanning electron microscopy images of wood samples separated from the sixth annual ring of infected ‘Groeneveld’ plants (A–C), the sixth annual ring of infected ‘Dodoens’ plants (D–F) and the seventh annual ring of infected ‘Dodoens’ plants (G–I). (A) Tyloses formed in latewood vessels in response to the Ophiostoma novo-ulmi ssp. americana × novo-ulmi inoculation. Cross-section. (B) Tyloses occluding latewood vessel lumens. Tangential section. (C) Fungal hyphae growing inside latewood vessels. Cross-section. (D) Tyloses formed in earlywood vessels in response to fungal inoculation. Cross-section. (E) Formation of many narrowed latewood vessels in response to fungal inoculation. Cross-section. (F) Narrowed latewood vessels as a possible resistance factor limiting the movement of the fungus in the vascular system. Radial section. (G) Reduced formation of large earlywood vessels in the next growing season after fungal inoculation. Cross-section. (H) Continuation of narrowed latewood vessel formation in the next growing season after fungal inoculation. Cross-section. (I) Occasional occurrence of fungal hyphae (arrows) growing inside earlywood vessel. Radial section. Scale bars: (A, D, E, G, H) = 500 μm; (B) = 200 μm; (C, I) = 50 μm; (F) = 100 μm.
Scanning electron microscopy of wood samples
Wood sections (transverse, radial and tangential surfaces) were mounted on specimen stubs, sputter-coated with gold and observed by high-vacuum SEM using a Vega TS 5130 instrument (Tescan, Brno, Czech Republic) operating at 15 kV.
Preparation and determination of extractives
Separated wood was mechanically disintegrated to sawdust, and a fraction size of 0·5–1·0 mm was extracted in a Soxhlet apparatus with a mixture of ethanol and toluene according to the American Society for Testing and Materials (ASTM) International standard procedure D 1107-96. Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Determinations of lignin content and lignin monomer composition
Lignin content was determined according to the US Department of Energy, National Renewable Energy Laboratory analytical procedure (Sluiter et al., 2010). Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Nitrobenzene oxidation (NBO) was carried out using 2 m NaOH in 10 mL stainless steel vessels at 180 °C for 2 h, and oxidation products were analysed by isocratic high-performance liquid chromatography (HPLC) under the following conditions: LiChrospher 100 RP-18, 5 μm, 100 × 4 mm i.d. column (Merck, Darmstadt, Germany), mobile phase water:methanol:acetic acid (850:150:1), flow rate 1·0 mL min–1, column temperature 35 °C, detection via diode array detector in the 210–360 nm region (Ďurkovič et al., 2012). The S/G ratio in lignin was calculated as (syringaldehyde + syringic acid) / (vanillin + vanillic acid). Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Determinations of cellulose content and macromolecular traits of cellulose
Cellulose content was determined by the method of Seifert (1956). Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Molecular weight distribution analysis of the cellulose samples was performed after their conversion into cellulose tricarbanilates by a modified method described by other authors (Josefsson et al., 2001; Foston and Ragauskas, 2010). Briefly, the cellulose samples were dried over silica gel for several days. Anhydrous pyridine (8·0 mL), cellulose (50 mg) and phenyl isocyanate (1·0 mL) were sealed in a 50-mL dropping flask. The flasks were immersed in an oil bath at 70 °C for 72 h. At the end of the reaction, methanol (2·0 mL) was added to the mixture to eliminate excess phenyl isocyanate. The yellow solutions were then added dropwise into a methanol:water mixture (7:3, 150 mL), stirring rapidly with a magnetic stirrer. Cellulose tricarbanilates were dissolved in tetrahydrofuran and filtered through a Puradisc 25 NYL filter (Whatman International, Maidstone, UK) with a pore size of 0·45 μm. Size exclusion chromatography was performed at 35 °C with tetrahydrofuran at a flow rate of 1 mL min–1 on two PLgel, 10 μm, 7·5 × 300 mm, MIXED-B columns (Agilent Technologies, Santa Clara, CA, USA) preceded by a PLgel, 10 μm, 7·5 × 50 mm, Guard-column (Agilent Technologies) as described by Kačík et al. (2009). Data were acquired with ChemStation software (Agilent Technologies) and calculations were performed with the Clarity GPC module (DataApex, Prague, Czech Republic). Numerical outputs were obtained for Mn (number-average molecular weight), Mw (weight-average molecular weight), Mz (z-average molecular weight), Mz+1 (z + 1-average molecular weight) and Mp (peak molecular weight). The polydispersity index (PDI) of cellulose was calculated as the ratio Mw/Mn. The degree of polymerization (DPw) was calculated by dividing the molecular weight by the monomer equivalent weight of anhydroglucose (DPw = Mw/162). Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Solid-state 13C nuclear magnetic resonance and FT-IR measurements of cellulose crystallinity
The single-pulse 13C magic angle spinning (MAS) nuclear magnetic resonance (NMR) measurements were performed with a 400-MHz solid-state NMR spectrometer (Varian, Palo Alto, CA, USA). High-resolution 13C NMR spectra were recorded at the resonance frequency of ∼100 MHz with the use of 4-mm rotors and an MAS frequency of 10 kHz at a temperature of 30 °C inside the rotor. A free induction decay was recorded after a radio-frequency π/2 pulse of 2·0 μs duration, with high-power proton decoupling at 71 kHz and a recycle delay of 60 s. The 13C MAS NMR spectrum was obtained by Fourier transformation of the free induction decay, which is an average of ∼3500 scans. The chemical shifts were referenced to tetramethylsilane using adamantane as an external standard. Deconvolutions were carried out with Mnova 8·1 software (Mestrelab Research, Santiago de Compostela, Spain). The crystallinity index (CI) of cellulose was determined by separating the C4 region of the spectrum into crystalline (89 p.p.m.) and amorphous (83 p.p.m.) peaks (Matulova et al., 2005; Vane et al., 2006) and calculated according to a modified method described by Park et al. (2010). The height of the crystalline peak was divided by the sum of the crystalline and amorphous peak heights.
FT-IR spectra of wood powder samples were recorded on a Nicolet iS10 FT-IR spectrometer equipped with a Smart iTR attenuated total reflectance sampling accessory (Thermo Fisher Scientific, Waltham, MA, USA). The spectra were acquired by accumulating 64 interferograms at a resolution of 4 cm–1 in an absorbance mode at wavenumbers from 4000 to 400 cm–1. Spectral peaks were measured using OMNIC 8·0 software (Thermo Fisher Scientific). In order to normalize the infrared spectra obtained, we used the 1032 cm–1 band, assigned to the C–O stretching vibration in cellulose (Liang and Marchessault, 1959; Colom et al., 2003). The lateral order index (LOI) of cellulose (A1422/A896) was calculated as described by O’Connor et al. (1958). Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Determinations of polysaccharide content and neutral sugar composition
Total content of polysaccharides (i.e. holocellulose) was determined using the method of Wise et al. (1946). Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Qualitative and quantitative analyses of saccharides were carried out by HPLC according to the ASTM International standard procedure E 1758-01. The samples were hydrolysed in a two-stage process. In the first stage, 72 % (w/w) H2SO4 at a temperature of 30 °C was used for 1 h, and in the second stage the formed oligomers were hydrolysed to monosaccharides after dilution to 4 % (w/w) H2SO4 at 121 °C for 1 h. The analyses were performed with an Agilent 1200 HPLC chromatograph (Agilent Technologies) equipped with an Aminex HPX-87P column (Bio-Rad Laboratories, Hercules, CA, USA) at a temperature of 80 °C and a mobile phase flow rate of 0·6 mL min–1. Measurements were performed on four replicates of infected and four replicates of non-infected plants per hybrid.
Statistical analysis
Data showed a normal distribution and were therefore subjected to one-way analysis of variance. Duncan's multiple range tests were used for pairwise comparisons of means. The Pearson correlation coefficient was calculated for the relationship between cellulose content and DPw. The relationship was considered significant if P < 0·05.
Multivariate association of the 25 examined wood traits was analysed by principal components analysis (PCA) to describe patterns of covariation among the contents of main cell wall components, lignin monomer composition, macromolecular traits of cellulose and the neutral saccharide composition of wood.
RESULTS
Changes in contents of cell wall components
Data on the contents of main cell wall components for the Dutch elm hybrids are presented in Table 1. Within the sixth annual ring separated from non-infected plants, significant differences were found for the contents of lignin, cellulose, holocellulose and extractives. This result predominantly reflects genotypic differences between the hybrids. On the other hand, the two hybrids shared some common trends in host responses to DED infection. Upon infection, in both hybrids, the lignin content increased, while the content of cellulose decreased. In the infected ‘Groeneveld’ plants, a moderate decrease in the content of cellulose was found, whereas in the infected ‘Dodoens’ plants there was a marked decline in cellulose content. Strong dissimilarities between the hybrids were found for the content of extractives. Infected ‘Groeneveld’ plants responded with a significant decrease in the amount of extractives, whereas infected ‘Dodoens’ plants markedly increased their content of these substances. When comparing the sixth annual ring with the seventh and eighth annual rings within the infected 'Dodoens' plants, the seventh and eighth rings contained lower amounts of lignin, cellulose and extractives (Table 1). This implies that the content of hemicelluloses increased at the expense of the above-mentioned components in successive annual rings after fungal inoculation.
Table 1.
Contents of main cell wall components and extractives present in wood of Dutch elm hybrids (%)
| Wood component | ‘Groeneveld’ non-infected (sixth annual ring) | ‘Groeneveld’ infected (sixth annual ring) | ‘Dodoens’ non-infected (sixth annual ring) | ‘Dodoens’ infected (sixth annual ring) | ‘Dodoens’ non-infected (seventh and eighth annual rings) | ‘Dodoens’ infected (seventh and eighth annual rings) |
|---|---|---|---|---|---|---|
| Lignin | 18·24 ± 0·17e | 22·85 ± 0·28b | 22·10 ± 0·27c | 24·05 ± 0·22a | 20·67 ± 0·14d | 22·92 ± 0·23b |
| Cellulose | 41·31 ± 0·22b | 37·03 ± 0·30d | 39·45 ± 0·25c | 28·68 ± 0·12e | 42·27 ± 0·45a | 25·91 ± 0·40f |
| Holocellulose | 80·40 ± 0·23d | 83·74 ± 0·28a | 82·31 ± 0·24b | 75·08 ± 0·24e | 81·58 ± 0·29c | 75·17 ± 0·14e |
| Extractives | 5·84 ± 0·17b | 3·51 ± 0·14e | 4·26 ± 0·10d | 7·68 ± 0·15a | 4·93 ± 0·13c | 4·93 ± 0·09c |
Data are means ± s.d.
Mean values followed by the same letters within the same row across hybrids are not significantly different at P < 0·05.
Changes in lignin monomer composition
Based on the analysis of NBO products, the hybrids showed strong genotypic differences in lignin monomer composition (Table 2). Within the sixth annual ring of the non-infected ‘Groeneveld’ plants, the main constituents of lignin were S units; G units were much less abundant and the representation of p-hydroxyphenyl units was almost negligible. For non-infected ‘Dodoens’ plants, the prevailing constituents of lignin were G units; S units were less abundant and the number of p-hydroxyphenyl units was also very low. Upon infection, the hybrids showed completely different host responses in lignin monomer composition. Within the sixth annual ring of infected ‘Groeneveld’ plants, the S/G ratio decreased significantly but the abundance of S units was still twice as high as that of G units (S/G = 2·06). On the contrary, the sixth annual ring of infected ‘Dodoens’ plants was characterized by a significant increase in S units, which prevailed over G units within the lignin macromolecular structure (S/G = 1·43). When comparing the sixth with the seventh and eighth annual rings within the infected ‘Dodoens’ plants, the seventh and eighth rings contained only a slightly decreased number of S units (S/G = 1·40).
Table 2.
Nitrobenzene oxidation (NBO) products present in wood of Dutch elm hybrids (%)
| NBO product | ‘Groeneveld’ non-infected (sixth annual ring) | ‘Groeneveld’ infected (sixth annual ring) | ‘Dodoens’ non-infected (sixth annual ring) | ‘Dodoens’ infected (sixth annual ring) | ‘Dodoens’ non-infected (seventh and eighth annual rings) | ‘Dodoens’ infected (seventh and eighth annual rings) |
|---|---|---|---|---|---|---|
| p-Hydroxybenzoic acid | 0·01 ± 0·00cd | 0·03 ± 0·01a | 0·02 ± 0·00b | 0·01 ± 0·00d | 0·01 ± 0·00c | 0·01 ± 0·00d |
| p-Hydroxybenzaldehyde | 0·04 ± 0·00c | 0·06 ± 0·01a | 0·06 ± 0·01a | 0·05 ± 0·01b | 0·05 ± 0·00b | 0·03 ± 0·00c |
| Vanillic acid | 0·19 ± 0·01e | 0·22 ± 0·01d | 0·31 ± 0·01a | 0·26 ± 0·01c | 0·29 ± 0·01b | 0·21 ± 0·01d |
| Vanillin | 2·43 ± 0·02f | 2·71 ± 0·03d | 3·96 ± 0·03a | 3·42 ± 0·05c | 3·82 ± 0·01b | 2·65 ± 0·04e |
| Syringic acid | 0·42 ± 0·01a | 0·41 ± 0·02a | 0·29 ± 0·01c | 0·38 ± 0·01b | 0·26 ± 0·01d | 0·29 ± 0·01c |
| Syringaldehyde | 5·61 ± 0·04a | 5·61 ± 0·04a | 3·75 ± 0·08c | 4·89 ± 0·05b | 3·42 ± 0·02d | 3·72 ± 0·03c |
| Total yield in wood | 8·70 ± 0·08b | 9·05 ± 0·11a | 8·39 ± 0·12c | 9·01 ± 0·12a | 7·85 ± 0·06d | 6·92 ± 0·09e |
| S/G ratio | 2·29 ± 0·01a | 2·06 ± 0·02b | 0·94 ± 0·02e | 1·43 ± 0·01c | 0·89 ± 0·01f | 1·40 ± 0·01d |
Data are means ± s.d.
Mean values followed by the same letters within the same row across hybrids are not significantly different at P < 0·05.
Changes in macromolecular traits of cellulose
Changes in the macromolecular traits of cellulose samples are given in Table 3. The hybrids showed genotypic differences for distributions of the molecular weights Mn, Mw, Mz and Mz+1. Upon infection, in both hybrids, the values for Mw, Mz and Mz+1 significantly increased within the sixth annual ring. Figure 2 shows that both hybrids responded to infection with a significant increase in the biosynthesis of high molecular weight macromolecules of cellulose, followed by a shift in the peaks (Mp values) to the high molecular weight area. At the same time, however, a substantial increase in low molecular weight macromolecules of cellulose occurred, which indicates that the medium molecular weight macromolecules were degraded. Biodegradation affected mostly the macromolecules with DPw values of ∼202–206 (infected ‘Groeneveld’ plants) and those with DPw values of ∼235–238 (infected ‘Dodoens’ plants).
Table 3.
Macromolecular traits of cellulose isolated from wood of Dutch elm hybrids
| Trait | ‘Groeneveld’ non-infected (sixth annual ring) | ‘Groeneveld’ infected (sixth annual ring) | ‘Dodoens’ non-infected (sixth annual ring) | ‘Dodoens’ infected (sixth annual ring) | ‘Dodoens’ non-infected (seventh and eighth annual rings) | ‘Dodoens’ infected (seventh and eighth annual rings) |
|---|---|---|---|---|---|---|
| Mn | 13 127 ± 98d | 14 305 ± 86c | 15 156 ± 163a | 13 285 ± 146d | 12 235 ± 72e | 14 784 ± 183b |
| Mw | 59 029 ± 70e | 108 471 ± 327c | 85 287 ± 221d | 110 509 ± 636b | 53 084 ± 76f | 115 541 ± 541a |
| Mz | 185 817 ± 179e | 343 385 ± 915c | 272 344 ± 630d | 348 548 ± 2328 b | 180 184 ± 464f | 357 787 ± 1240a |
| Mz+1 | 392 846 ± 779f | 608 574 ± 4072b | 513 384 ± 1378d | 594 341 ± 5624c | 423 274 ± 2149e | 623 804 ± 4085a |
| Mp | 33 013 ± 218e | 44 585 ± 386c | 38 298 ± 132d | 48 848 ± 721b | 30 492 ± 105f | 53 822 ± 807a |
| PDI | 4·50 ± 0·03e | 7·58 ± 0·03c | 5·63 ± 0·05d | 8·32 ± 0·06a | 4·34 ± 0·02f | 7·82 ± 0·07b |
| DPw | 364 ± 1e | 670 ± 2c | 527 ± 2d | 682 ± 4b | 328 ± 1f | 713 ± 3a |
| LOI (A1422/A896) | 1·08 ± 0·07b | 1·19 ± 0·15b | 1·16 ± 0·12b | 1·39 ± 0·20a | 1·03 ± 0·06b | 1·14 ± 0·09b |
Data are means ± s.d.
Mean values followed by the same letters within the same row across hybrids are not significantly different at P < 0·05.
Fig. 2.
Size exclusion chromatography of molecular weight distributions of cellulose tricarbanilates. (A) Sixth annual ring of ‘Groeneveld’ plants. (B) Sixth annual ring of ‘Dodoens’ plants. (C) Seventh and eighth annual rings of ‘Dodoens’ plants.
In both hybrids, co-occurring biosynthetic and biodegradation processes resulted in a huge increase in the polydispersity index of cellulose in the wood of infected plants (Table 3). Increased values of DPw probably resulted from the biosynthesis of high molecular weight macromolecules of cellulose, which remained unaffected by the fungus. When comparing the sixth annual ring with the seventh and eighth annual rings within the infected ‘Dodoens’ plants, the seventh and eighth rings simultaneously showed continuing biosynthesis of high molecular weight macromolecules together with both the biodegradation of medium molecular weight macromolecules having DPw values of ∼186–189 and low molecular weight macromolecules (Fig. 2C). Biodegradation of medium and low molecular weight macromolecules of cellulose may explain the occurrence of secondary cell wall ruptures (Fig. 3A) and cracks (Fig. 3B) in the vessels but rarely in the fibres of infected plants.
Fig. 3.
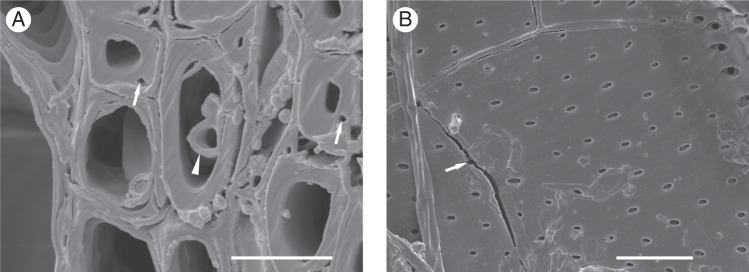
Scanning electron microscopy images of secondary cell wall ruptures and cracks. (A) Ophiostoma novo-ulmi ssp. americana × novo-ulmi hypha inside the vascular tracheid (arrowhead) and ruptures in secondary cell walls of wood fibres (arrows). Cross-section of sixth annual ring of an infected ‘Groeneveld’ plant. Scale bar = 10 μm. (B) Fungal hyphae inside an earlywood vessel and radial cracks in the secondary cell wall (arrow). Radial section of sixth annual ring of an infected ‘Groeneveld’ plant. Scale bar = 20 μm.
For non-infected plants of both hybrids, the mean content of cellulose in these samples was 41·01 % and the mean DPw value was 406·17. Upon infection, the mean content of cellulose in the infected samples for both hybrids decreased to 30·54 %, whereas the mean DPw increased to 688·33. Figure 4A shows the close negative linear relationship between cellulose content and DPw in non-infected plants. Upon infection, the slope of the descending line changed rapidly (Fig. 4B) but the Pearson correlation value remained highly significant. This indicates compensation for loss of cellulose through an increase in DPw, and thus a trade-off between these two traits to ensure the tensile strength of the wood.
Fig. 4.
Relationship of cellulose content with the degree of polymerization (DPw) in (A) non-infected plants and (B) infected plants.
In addition, 13C NMR spectra of the examined wood samples displayed two signals at 83 and 89 p.p.m., corresponding to C4 carbon atoms of amorphous and crystalline cellulose, respectively. In infected plants of both hybrids, the decrease in signal intensities at both resonances revealed that losses of crystalline and non-crystalline cellulose regions occurred in parallel. Within the sixth annual ring of the infected ‘Groeneveld’ plants, loss of the amorphous region was 3·13 % and that of the crystalline region was 5·26 % (Fig. 5A), and the CI of cellulose inferred from the NMR spectra decreased from 37·25 to 35·42 % (i.e. 1·83 % loss). Within the sixth annual ring of the infected ‘Dodoens’ plants, loss of the amorphous region was 5·26 % and that of the crystalline region was 26·09 % (Fig. 5B), and the CI decreased from 37·70 to 30·77 % (i.e. 6·93 % loss). When comparing the sixth annual ring with the seventh and eighth annual rings within the infected ‘Dodoens’ plants, the seventh and eighth rings contained a 2·78 % lower proportion of the amorphous region and a 6·25 % lower proportion of the crystalline region (Fig. 5C), and the CI decreased from 30·77 to 29·41 % (i.e. 1·36 % loss).
Fig. 5.
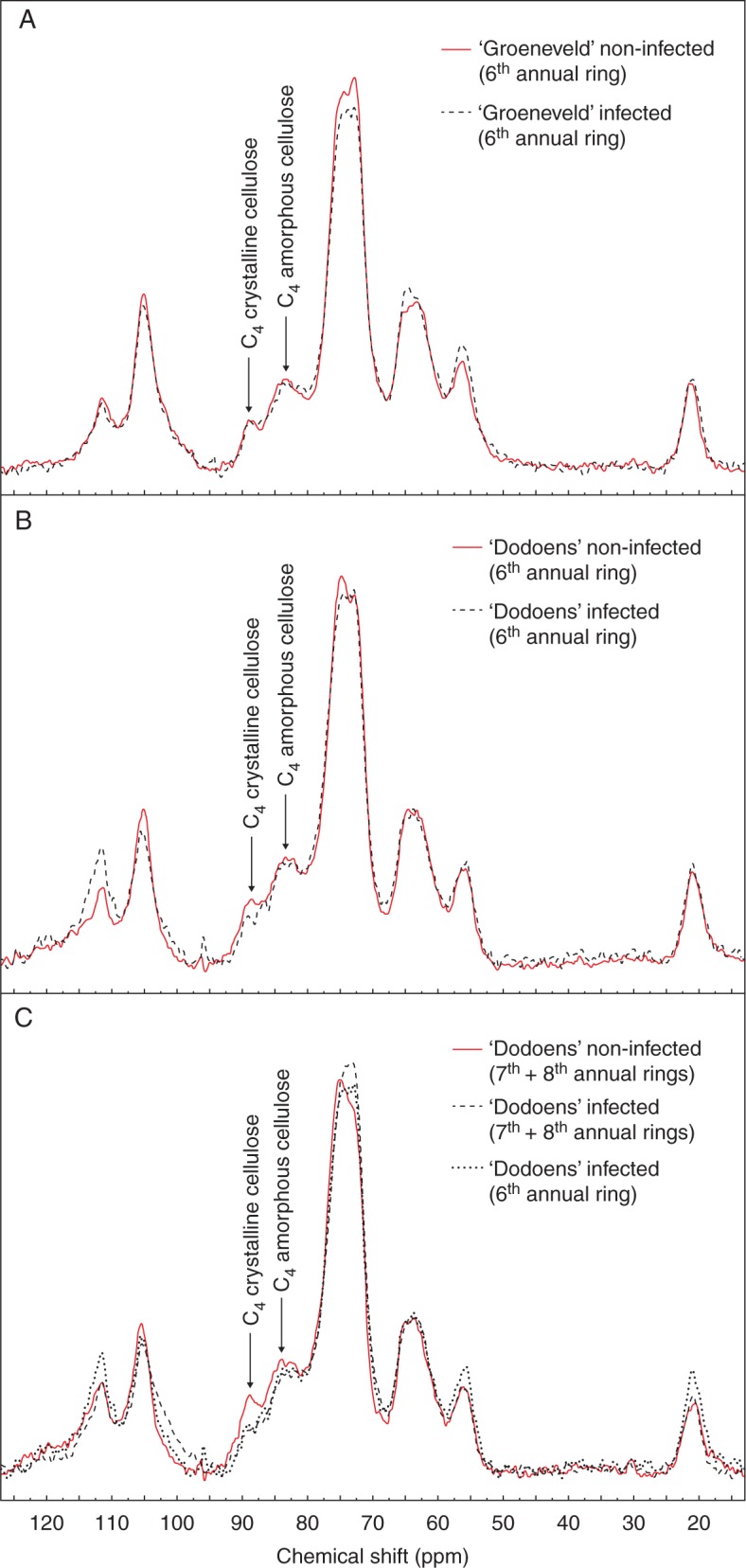
Solid-state 13C MAS NMR spectra of wood sawdust samples. (A) Sixth annual ring of ‘Groeneveld’ plants. (B) Sixth annual ring of ‘Dodoens’ plants. (C) Seventh and eighth annual rings of ‘Dodoens’ plants.
With regard to the LOI of the cellulose crystallinity inferred from the FT-IR spectra, there were non-significant differences between the infected and non-infected plants, except in the sixth annual ring of infected ‘Dodoens’ plants. In this case, LOI increased within the bulk of the cellulose macromolecules (Table 3).
Changes in saccharide contents
The absolute yields of saccharides and their relative contents in the wood of the hybrids are presented in Table 4. We found very small genotypic differences between the hybrids for the contents of d-glucose, l-arabinose and d-galactose. Both hybrids showed common trends in host responses to DED infection. Upon infection, the relative proportions of d-glucose and d-mannose decreased substantially within the sixth annual ring, whereas the proportions of d-xylose, l-arabinose and d-galactose significantly increased. The most substantial changes were related to the contents of d-glucose and d-xylose. When comparing the sixth annual ring with the seventh and eighth annual rings within the infected ‘Dodoens’ plants, the seventh and eighth rings contained higher absolute yields and relative proportions of d-glucose and lower amounts of d-xylose, l-arabinose and d-mannose, while the content of d-galactose remained steady. The excessive occurrence of agglomerates of storage compounds and partially gelatinized starch inside ray parenchyma cells and axial parenchyma cells might contribute to the increased amounts of d-glucose within the seventh and eighth annual rings of the infected ‘Dodoens’ plants.
Table 4.
Absolute yields of saccharides (% of oven-dry weight per unextracted wood) and their relative proportions (%) in wood of Dutch elm hybrids
| Saccharide | ‘Groeneveld’ non-infected (sixth annual ring) | ‘Groeneveld’ infected (sixth annual ring) | ‘Dodoens’ non-infected (sixth annual ring) | ‘Dodoens’ infected (sixth annual ring) | ‘Dodoens’ non-infected (seventh and eighth annual rings) | ‘Dodoens’ infected (seventh and eighth annual rings) |
|---|---|---|---|---|---|---|
| Absolute yields | ||||||
| d-Glucose | 58·45 ± 0·04b | 53·41 ± 0·06e | 57·39 ± 0·05c | 48·73 ± 0·26f | 61·18 ± 0·04a | 54·57 ± 0·08d |
| d-Xylose | 13·92 ± 0·05d | 18·78 ± 0·06a | 14·64 ± 0·05c | 17·42 ± 0·20b | 12·74 ± 0·04e | 14·58 ± 0·07c |
| l-Arabinose | 1·25 ± 0·02c | 1·55 ± 0·04a | 1·22 ± 0·01c | 1·35 ± 0·02b | 1·04 ± 0·01d | 1·24 ± 0·03c |
| d-Mannose | 1·43 ± 0·01a | 0·41 ± 0·02e | 0·86 ± 0·01b | 0·69 ± 0·02c | 0·36 ± 0·01f | 0·52 ± 0·02d |
| d-Galactose | 0·22 ± 0·01d | 0·28 ± 0·04c | 0·22 ± 0·02d | 0·32 ± 0·02b | 0·38 ± 0·02a | 0·32 ± 0·01b |
| Total yield | 75·27 ± 0·12b | 74·42 ± 0·18c | 74·32 ± 0·12c | 68·50 ± 0·44e | 75·70 ± 0·09a | 71·23 ± 0·13d |
| Relative proportions | ||||||
| d-Glucose | 77·65 ± 0·06b | 71·76 ± 0·09e | 77·22 ± 0·08c | 71·14 ± 0·18f | 80·81 ± 0·05a | 76·61 ± 0·04d |
| d-Xylose | 18·49 ± 0·04e | 25·24 ± 0·02b | 19·70 ± 0·04d | 25·43 ± 0·18a | 16·84 ± 0·04f | 20·47 ± 0·06c |
| l-Arabinose | 1·66 ± 0·02d | 2·08 ± 0·04a | 1·64 ± 0·01d | 1·96 ± 0·03b | 1·37 ± 0·02e | 1·75 ± 0·04c |
| d-Mannose | 1·90 ± 0·01a | 0·55 ± 0·02e | 1·15 ± 0·02b | 1·01 ± 0·02c | 0·48 ± 0·01f | 0·73 ± 0·02d |
| d-Galactose | 0·30 ± 0·01d | 0·37 ± 0·05c | 0·29 ± 0·03d | 0·46 ± 0·02ab | 0·50 ± 0·03a | 0·44 ± 0·02b |
Data are means ± s.d.
Mean values followed by the same letters within the same row across hybrids are not significantly different at P < 0·05.
Associations among wood traits
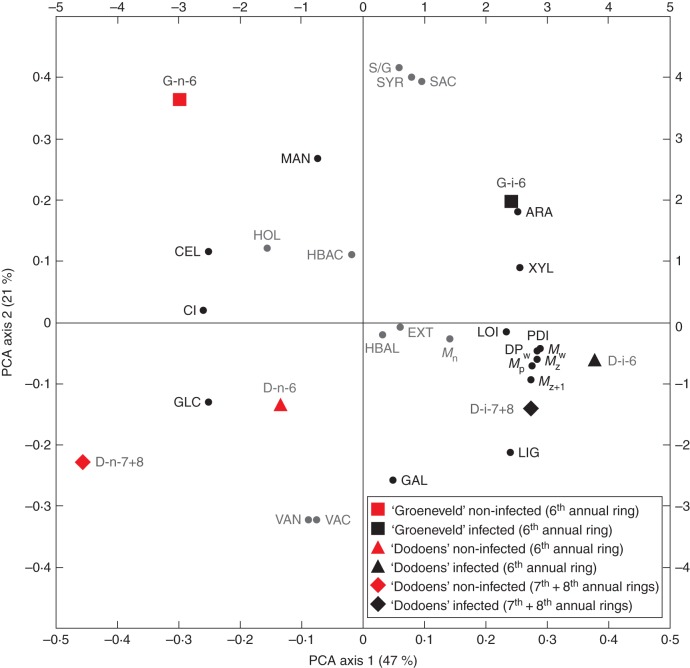
A PCA was done to evaluate how wood traits were associated (Fig. 6). The first axis explained 47 % of the variation and showed strong positive loadings for macromolecular traits of cellulose, such as polydispersity, degree of polymerization, Mw, Mz, Mz+1, Mp and the relative proportions of d-xylose and l-arabinose. The negative side of the axis indicated strong loadings for the CI of cellulose, the cellulose content and the relative proportion of d-glucose. The second axis explained 21 % of the variation and showed strong positive loadings for the S/G ratio in lignin, syringaldehyde, syringic acid and the relative proportion of d-mannose. The negative side of the axis indicated strong loadings for vanillic acid, vanillin, d-galactose and lignin content. The traits associated with differential host responses to DED infection correlated with the second PCA axis, whereas the traits associated with common host responses correlated with the first PCA axis. In addition, PCA showed that infected annual rings in both hybrids were clearly separated from the non-infected control annual rings.
Fig. 6.
Positions of 25 wood traits on the first and second axes of the principal components analysis (PCA). Traits associated with differential host responses are indicated in grey. Bottom and left axes refer to wood traits; top and right axes refer to annual rings of Dutch elm hybrids. Trait abbreviations: ARA, l-arabinose; CEL, cellulose content; CI, crystallinity index of cellulose; DPw, degree of polymerization of cellulose; EXT, extractives content; GAL, d-galactose; GLC, d-glucose; HBAC, p-hydroxybenzoic acid; HBAL, p-hydroxybenzaldehyde; HOL, holocellulose content; LIG, lignin content; LOI, lateral order index; MAN, d-mannose; Mn, number-average molecular weight; Mp, peak molecular weight; Mw, weight-average molecular weight; Mz, z-average molecular weight; Mz+1, z + 1-average molecular weight; PDI, polydispersity index; S/G, syringyl:guaiacyl ratio in lignin; SAC, syringic acid; SYR, syringaldehyde; VAC, vanillic acid; VAN, vanillin; XYL, d-xylose.
DISCUSSION
Metabolic fingerprinting of elm secondary xylem tissues to provide bioindicators of chemical changes caused by DED infection is a recent tool in detecting progressive tree responses to the pathogen. We found common host responses as well as differential host responses to O. novo-ulmi ssp. americana × novo-ulmi infection between the two Dutch elm hybrids examined here. We relate these findings to previously published work, highlighting the novel findings pertinent to biodegradation of medium molecular weight macromolecules of cellulose.
Host responses of lignin metabolism to O. novo-ulmi infection
Previous studies revealed increased levels of lignin in the branches of U. minor (Martín et al., 2005a) and U. minor × U. pumila hybrids (Martín et al., 2007) as an induced host defence response to inoculation with O. novo-ulmi. Our results regarding the changes in lignin content upon infection in Dutch elm hybrids ‘Dodoens’ and ‘Groeneveld’ support the above observations. It is recognized that lignin provides woody tissues with protection against pathogens because of its resistance to biodegradation (Duchesne et al., 1992). Unfortunately, none of the above studies examined the influence of S/G ratio in lignin on cell wall degradation by O. novo-ulmi. The macromolecular structure of lignin depends on the proportions of S and G aromatic subunits, which determine the type and number of cross-links within the macromolecule as well as the reactivity of the lignin (S-rich lignin forms fewer cross-linking structures than G-rich lignin). NBO products originate from the corresponding phenylpropane units and their α(β)-O-4 alkyl-arylethers due to the oxidation of lignin in an alkaline environment. Thus, the S/G ratio provides information regarding the relative amounts of uncondensed guaiacyl- and syringylpropane units constituting the original lignin. In addition, the yield of these aldehydes reflects the degree of condensation of the lignin, because these uncondensed structures are cleaved by nitrobenzene oxidation, leaving condensed lignins as the residue (Kačík et al., 2012). With regard to changes in the S/G ratio in lignin, our results showed differential host responses to fungal infection, but only the G-rich lignin of ‘Dodoens’ was involved in successful defence against O. novo-ulmi ssp. americana × novo-ulmi. This finding was supported by the loadings for vanillin and vanillic acid in the multivariate wood trait analysis (Fig. 6). These two NBO products, typical of G lignin, were associated with the sixth annual ring of the non-infected, DED-tolerant ‘Dodoens’. On the other hand, d-mannose was associated with the sixth annual ring of the non-infected but sensitive ‘Groeneveld’. It is well established that lignin that is rich in G units has a greater degree of carbon–carbon bonding and a more condensed macromolecular structure than lignin rich in S units. Some monolignol precursors (for example ferulates and their dehydrodimers) can cross-link cell wall polysaccharides, forming an enhanced structural barrier to reduce biodegradability of the cell wall (Fry, 1986). Ferulate and monolignol metabolisms were found to be major sources of ethylene-mediated resistance of Arabidopsis thaliana to the necrotrophic fungus Botrytis cinerea (Lloyd et al., 2011). In addition, Quercus alba bark lignin rich in G units showed a greater resistance to white rot fungus (Lentinula edodes) decay compared with wood lignin rich in S units (Vane et al., 2006).
Changes in macromolecular traits of cellulose after attack by O. novo-ulmi
Owing to its high degree of polymerization and crystallinity, cellulose is considered to be primarily responsible for the tensile strength of wood. Therefore, reducing the length of the cellulose molecules (DPw) would cause a reduction in macro-strength properties (Sweet and Winandy, 1999; Vizárová et al., 2012). Upon infection, we observed the occurrence of secondary cell wall ruptures and cracks. From a mechanical point of view, a decrease in cellulose content was partially compensated for by an increase in DPw of the cellulose chain to maintain the tensile strength of the wood in the infected plants. Production of cellulolytic enzymes by O. ulmi and O. novo-ulmi isolates was reported by Przybył et al. (2006). Scheffer and Elgersma (1982) documented that aggressive DED isolates caused severe cell wall erosions in Ulmus americana secondary xylem vessels, whereas non-agressive isolates left cell walls intact. In this study, we used a highly aggressive isolate of the hybrid subspecies americana × novo-ulmi of O. novo-ulmi that is capable of severe cell wall degradation. We found that the primary targets of fungal attack were medium molecular weight macromolecules of cellulose. Kleman-Leyer et al. (1992) observed a difference in the kinetics of cotton cellulose depolymerization by the brown rot fungus Postia placenta and the white rot fungus Phanerochaete chrysosporium. Cellulose attacked by P. placenta showed a broader molecular mass distribution with progressively decreasing values of DPw over time. However, a different molecular mass distribution of cellulose was observed during attack by P. chrysosporium. At first, the molecular mass distribution curve broadened, but as attack progressed the curve narrowed. The DPw decreased during the initial period of fungal attack but then increased gradually to near control values. Simultaneous biodegradation of amorphous and crystalline forms of cellulose, examined using 13C NMR spectra, was also reported for Betula papyrifera wood and Q. alba bark decayed by P. chrysosporium (Davis et al., 1994) and L. edodes (Vane et al., 2006). Seen from the viewpoint of cellulose depolymerization, the mechanism of elm wood cell wall degradation by O. novo-ulmi ssp. americana × novo-ulmi appears to resemble that caused by white rot fungi.
LOI represents the ordered regions of cellulose perpendicular to chain direction and is inferred from the ratio of infrared peak areas at 1422 and 896 cm–1 (Široký et al., 2010). The absorption band at 1422 cm–1 represents CH2 scissoring motion at C6 (Nelson and O'Connor, 1964a, b; Široký et al., 2010). The 896 cm–1 band indicates the vibrational mode involving C1 and the four atoms attached to it, which is characteristic of β-anomers or β-linked glucose polymers (Nelson and O'Connor, 1964a, b; Široký et al., 2010). A significant increase in LOI within the sixth annual ring of the infected ‘Dodoens’ plants may imply that either the crystalline regions parallel to the cellulose chain direction were predominantly degraded or a recrystallization effect accompanied cellulose chain scission during fungal attack (Ibbett et al., 2008).
An interesting point in this study was how the S/G ratio in lignin affected cellulose degradability by O. novo-ulmi cellulolytic enzymes in the infected plants. Recent studies revealed that the S/G ratio has a significant influence on the cross-linking between lignin and other cell wall components, thus modifying the microscopic structure and topochemistry of the cell wall (Jung and Casler, 2006). Differences in S/G ratio affected the efficiency of kraft pulping (Lapierre et al., 1999; Li et al., 2008; Santos et al., 2013), as well as cell wall degradability during chemical and hot water pretreatments, and the subsequent hydrolysis of cellulose to glucose (Li et al., 2010; Studer et al., 2011; Papa et al., 2012). In the present study the lowest rate of cellulose degradation was found in the infected ‘Groeneveld’ plants. Syringyl-rich lignin in non-infected ‘Groeneveld’ plants contained a 2·4-fold higher proportion of methoxyl groups than G-rich lignin in non-infected ‘Dodoens’ plants (or a 1·4-fold higher proportion of methoxyl groups for infected ‘Groeneveld’ plants than for infected ‘Dodoens’ plants). Thus, the ‘Groeneveld’ cellulose microfibrils were provided with denser steric protection by methoxyl groups and consequently with a decreased accessibility of the fungal cellulolytic enzymes, compared with ‘Dodoens’ microfibrils. This might be the reason why the medium molecular weight macromolecules of ‘Groeneveld’ cellulose were degraded to a lesser extent than those of ‘Dodoens’. Similar results with respect to the role of S-rich lignin in the resistance of hybrid poplar trees to degradation by wood decay fungi were recently reported by Skyba et al. (2013). In addition, it is well documented that amorphous regions of cellulose microfibrils are more readily degradable to glucose than crystalline regions (Suchy et al., 2011; Papa et al., 2012). Cellulose degradation rates mediated by fungal cellulases are typically 3- to 30-fold faster for amorphous cellulose substrates than for high crystalline cellulose substrates (Zhang and Lynd, 2004). However, our results revealed that, in both hybrids, crystalline regions of cellulose were degraded to a higher extent than amorphous regions. This suggests that, in both hybrids, readily hydrolysable amorphous regions were mostly provided with steric protection by methoxyl groups of both S-rich and G-rich lignins. Adani et al. (2011) showed that the particular spatial disposition of cell wall polymers determines the presence of microporous structures that modulate the accessibility of enzymes to the cell wall. Steric protection (3-D molecular geometry) is the main mechanism proposed to explain the biochemical recalcitrance of plant tissues (Papa et al., 2012).
To conclude, we have used metabolic profiles of wood components to find that, upon infection with O. novo-ulmi ssp. americana × novo-ulmi, medium molecular weight macromolecules of cellulose were degraded. The 13C NMR spectra revealed that loss of crystalline and non-crystalline cellulose regions occurred in parallel. The rate of cellulose degradation was influenced by the S/G ratio in lignin, but only the G-rich lignin of ‘Dodoens’ plants was involved in successful defence against DED. This finding was confirmed by the associations of vanillin and vanillic acid with DED-tolerant ‘Dodoens’ plants in the multivariate wood trait analysis.
ACKNOWLEDGEMENTS
We thank Drs M. Dvořák, I. Čaňová, M. Mamoňová, R. Lagaňa and M. Moravčík for their technical assistance with fungal inoculation, wood sampling and scanning electron microscopy, and Mrs E. Ritch-Krč for language revision. We also wish to acknowledge the thoughtful comments and suggestions of the anonymous reviewers, which improved the quality of the paper. This work was supported by funding from the Slovak scientific grant agency VEGA (1/0132/12).
LITERATURE CITED
- Adani F, Papa G, Schievano A, Cardinale G, D'Imporzano G, Tambone F. Nanoscale structure of the cell wall protecting cellulose from enzyme attack. Environmental Science & Technology. 2011;45:1107–1113. doi: 10.1021/es1020263. [DOI] [PubMed] [Google Scholar]
- Allwood JW, Ellis DI, Heald JK, Goodacre R, Mur LAJ. Metabolomic approaches reveal that phosphatidic and phosphatidyl glycerol phospholipids are major discriminatory non-polar metabolites in responses by Brachypodium distachyon to challenge by Magnaporthe grisea. Plant Journal. 2006;46:351–368. doi: 10.1111/j.1365-313X.2006.02692.x. [DOI] [PubMed] [Google Scholar]
- Binz T, Canevascini G. Xylanases from the Dutch elm disease pathogens Ophiostoma ulmi and O. novo-ulmi. Physiological and Molecular Plant Pathology. 1996a;49:159–175. [Google Scholar]
- Binz T, Canevascini G. Differential production of extracellular laccase in the Dutch elm disease pathogens Ophiostoma ulmi and O. novo-ulmi. Mycological Research. 1996b;100:1060–1064. [Google Scholar]
- Brasier CM, Kirk SA. Designation of the EAN and NAN races of Ophiostoma novo-ulmi as subspecies. Mycological Research. 2001;105:547–554. [Google Scholar]
- Brasier CM, Kirk SA. Rapid emergence of hybrids between the two subspecies of Ophiostoma novo-ulmi with a high level of pathogenic fitness. Plant Pathology. 2010;59:186–199. [Google Scholar]
- Colom X, Carrillo F, Nogués F, Garriga P. Structural analysis of photodegraded wood by means of FTIR spectroscopy. Polymer Degradation and Stability. 2003;80:543–549. [Google Scholar]
- Davis MF, Schroeder HA, Maciel GE. Solid-state 13C nuclear magnetic resonance studies of wood decay. II. White rot decay of paper birch. Holzforschung. 1994;48:186–192. [Google Scholar]
- Duchesne LC, Hubbes M, Jeng RS. Biochemistry and molecular biology of defense reactions in the xylem of angiosperm trees. In: Blanchette RA, Biggs AR, editors. Defense mechanisms of woody plants against fungi. Berlin: Springer; 1992. pp. 133–146. [Google Scholar]
- Ďurkovič J, Kaňuchová A, Kačík F, Solár R, Lengyelová A. Genotype- and age-dependent patterns of lignin and cellulose in regenerants derived from 80-year-old trees of black mulberry (Morus nigra L.) Plant Cell, Tissue and Organ Culture. 2012;108:359–370. [Google Scholar]
- Ďurkovič J, Čaňová I, Lagaňa R, et al. Leaf trait dissimilarities between Dutch elm hybrids with a contrasting tolerance to Dutch elm disease. Annals of Botany. 2013;111:215–227. doi: 10.1093/aob/mcs274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dvořák M, Tomšovský M, Jankovský L, Novotný D. Contribution to identify the causal agents of Dutch elm disease in the Czech Republic. Plant Protection Science. 2007;43:142–145. [Google Scholar]
- Dvořák M, Jankovský L, Krajňáková J. Characterisation of Czech Ophiostoma novo-ulmi isolates. Süleyman Demirel University Faculty of Forestry Journal. 2009;A:233–237. [Google Scholar]
- Fenning TM, Tymens SS, Gartland JS, Brasier CM, Gartland KMA. Transformation and regeneration of English elm using wild-type Agrobacterium tumefaciens. Plant Science. 1996;116:37–46. [Google Scholar]
- Foston M, Ragauskas AJ. Changes in lignocellulosic supramolecular and ultrastructure during dilute acid pretreatment of Populus and switchgrass. Biomass & Bioenergy. 2010;34:1885–1895. [Google Scholar]
- Fry SC. Cross-linking of matrix polymers in the growing cell walls of angiosperms. Annual Review of Plant Physiology. 1986;37:165–186. [Google Scholar]
- Heybroek HM. Resistant elms for Europe. In: Burdekin DA, editor. Research on Dutch elm disease in Europe. London: Her Majesty's Stationery Office; 1983. pp. 108–113. [Google Scholar]
- Ibbett RN, Domvoglou D, Phillips DAS. The hydrolysis and recrystallisation of lyocell and comparative cellulosic fibres in solutions of mineral acid. Cellulose. 2008;15:241–254. [Google Scholar]
- Josefsson T, Lennholm H, Gellerstedt G. Changes in cellulose supramolecular structure and molecular weight distribution during steam explosion of aspen wood. Cellulose. 2001;8:289–296. [Google Scholar]
- Jung HG, Casler MD. Maize stem tissues: cell wall concentration and composition during development. Crop Science. 2006;46:1793–1800. [Google Scholar]
- Kačík F, Kačíková D, Jablonský M, Katuščák S. Cellulose degradation in newsprint paper ageing. Polymer Degradation and Stability. 2009;94:1509–1514. [Google Scholar]
- Kačík F, Ďurkovič J, Kačíková D. Chemical profiles of wood components of poplar clones for their energy utilization. Energies. 2012;5:5243–5256. [Google Scholar]
- Kleman-Leyer K, Agosin E, Conner AH, Kirk TK. Changes in molecular size distribution of cellulose during attack by white rot and brown rot fungi. Applied and Environmental Microbiology. 1992;58:1266–1270. doi: 10.1128/aem.58.4.1266-1270.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konrad H, Kirisits T, Riegler M, Halmschlager E, Stauffer C. Genetic evidence for natural hybridization between the Dutch elm disease pathogens Ophiostoma novo-ulmi ssp. novo-ulmi and O. novo-ulmi ssp. americana . Plant Pathology. 2002;51:78–84. [Google Scholar]
- Krajňáková J, Longauer R. Culture initiation, multiplication and identification of in vitro regenerants of resistant hybrid elms. Lesnictví. 1996;42:261–270. [Google Scholar]
- Lapierre C, Pollet B, Petit-Conil M, et al. Structural alterations of lignins in transgenic poplars with depressed cinnamyl alcohol dehydrogenase or caffeic acid O-methyltransferase activity have an opposite impact on the efficiency of industrial kraft pulping. Plant Physiology. 1999;119:153–163. doi: 10.1104/pp.119.1.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Weng J-K, Chapple C. Improvement of biomass through lignin modification. Plant Journal. 2008;54:569–581. doi: 10.1111/j.1365-313X.2008.03457.x. [DOI] [PubMed] [Google Scholar]
- Li X, Ximenes E, Kim Y, et al. Lignin monomer composition affects Arabidopsis cell-wall degradability after liquid hot water pretreatment. Biotechnology for Biofuels. 2010;3:27. doi: 10.1186/1754-6834-3-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang CY, Marchessault RH. Infrared spectra of crystalline polysaccharides. II. Native celluloses in the region from 640 to 1700 cm–1 . Journal of Polymer Science. 1959;39:269–278. [Google Scholar]
- Lloyd A, Allwood JW, Winder CL, et al. Metabolomic approaches reveal that cell wall modifications play a major role in ethylene-mediated resistance against Botrytis cinerea. Plant Journal. 2011;67:852–868. doi: 10.1111/j.1365-313X.2011.04639.x. [DOI] [PubMed] [Google Scholar]
- Martín JA, Solla A, Coimbra MA, Gil L. Metabolic distinction of Ulmus minor xylem tissues after inoculation with Ophiostoma novo-ulmi. Phytochemistry. 2005a;66:2458–2467. doi: 10.1016/j.phytochem.2005.08.004. [DOI] [PubMed] [Google Scholar]
- Martín JA, Solla A, Woodward S, Gil L. Fourier transform-infrared spectroscopy as a new method for evaluating host resistance in the Dutch elm disease complex. Tree Physiology. 2005b;25:1331–1338. doi: 10.1093/treephys/25.10.1331. [DOI] [PubMed] [Google Scholar]
- Martín JA, Solla A, Woodward S, Gil L. Detection of differential changes in lignin composition of elm xylem tissues inoculated with Ophiostoma novo-ulmi using Fourier transform-infrared spectroscopy. Forest Pathology. 2007;37:187–191. [Google Scholar]
- Martín JA, Solla A, Coimbra MA, Gil L. Metabolic fingerprinting allows discrimination between Ulmus pumila and U. minor, and between U. minor clones of different susceptibility to Dutch elm disease. Forest Pathology. 2008;38:244–256. [Google Scholar]
- Matulova M, Nouaille R, Capek P, Péan M, Forano E, Delort A-M. Degradation of wheat straw by Fibrobacter succinogenes S85: a liquid- and solid-state nuclear magnetic resonance study. Applied and Environmental Microbiology. 2005;71:1247–1253. doi: 10.1128/AEM.71.3.1247-1253.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson ML, O'Connor RT. Relation of certain infrared bands to cellulose crystallinity and crystal lattice type. Part I. Spectra of lattice types I, II, III and of amorphous cellulose. Journal of Applied Polymer Science. 1964a;8:1311–1324. [Google Scholar]
- Nelson ML, O'Connor RT. Relation of certain infrared bands to cellulose crystallinity and crystal lattice type. Part II. A new infrared ratio for estimation of crystallinity in celluloses I and II. Journal of Applied Polymer Science. 1964b;8:1325–1341. [Google Scholar]
- Newbanks D, Bosch A, Zimmermann MH. Evidence for xylem dysfunction by embolization in Dutch elm disease. Phytopathology. 1983;73:1060–1063. [Google Scholar]
- Newhouse AE, Schrodt F, Liang H, Maynard CA, Powell WA. Transgenic American elm shows reduced Dutch elm disease symptoms and normal mycorrhizal colonization. Plant Cell Reports. 2007;26:977–987. doi: 10.1007/s00299-007-0313-z. [DOI] [PubMed] [Google Scholar]
- O'Connor RT, DuPré EF, Mitcham D. Applications of infrared absorption spectroscopy to investigations of cotton and modified cottons: Part I: Physical and crystalline modifications and oxidation. Textile Research Journal. 1958;28:382–392. [Google Scholar]
- Ouellette GB, Rioux D, Simard M, Cherif M. Ultrastuctural and cytochemical studies of host and pathogens in some fungal wilt diseases: retro- and introspection towards a better understanding of DED. Investigación Agraria: Sistemas y Recursos Forestales. 2004;13:119–145. [Google Scholar]
- Papa G, Varanasi P, Sun L, et al. Exploring the effect of different plant lignin content and composition on ionic liquid pretreatment efficiency and enzymatic saccharification of Eucalyptus globulus L. mutants. Bioresource Technology. 2012;117:352–359. doi: 10.1016/j.biortech.2012.04.065. [DOI] [PubMed] [Google Scholar]
- Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK. Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnology for Biofuels. 2010;3:10. doi: 10.1186/1754-6834-3-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pijut PM, Domir SC, Lineberger RD, Schreiber LR. Use of culture filtrates of Ceratocystis ulmi as a bioassay to screen for disease tolerant Ulmus americana. Plant Science. 1990;70:191–196. [Google Scholar]
- Przybył K, Dahm H, Ciesielska A, Moliński K. Cellulolytic activity and virulence of Ophiostoma ulmi and O. novo-ulmi isolates. Forest Pathology. 2006;36:58–67. [Google Scholar]
- Santini A, La Porta N, Ghelardini L, Mittempergher L. Breeding against Dutch elm disease adapted to the Mediterranean climate. Euphytica. 2008;163:45–56. [Google Scholar]
- Santini A, Pecori F, Pepori AL, Ferrini F, Ghelardini L. Genotype × environment interaction and growth stability of several elm clones resistant to Dutch elm disease. Forest Ecology and Management. 2010;260:1017–1025. [Google Scholar]
- Santos RB, Jameel H, Chang H-M, Hart PW. Impact of lignin and carbohydrate chemical structures on degradation reactions during hardwood kraft pulping processes. BioResources. 2013;8:158–171. [Google Scholar]
- Scheffer RJ, Elgersma DM. A scanning electron microscope study of cell wall degradation in elm wood by aggressive and non-aggressive isolates of Ophiostoma ulmi. European Journal of Forest Pathology. 1982;12:25–28. [Google Scholar]
- Scheffer RJ, Liem JI, Elgersma DM. Production in vitro of phytotoxic compounds by non-aggressive and aggressive isolates of Ophiostoma ulmi, the Dutch elm disease pathogen. Physiological and Molecular Plant Pathology. 1987;30:321–335. [Google Scholar]
- Seifert VK. Über ein neues Verfahren zur Schnellbestimmung der Rein-Cellulose. Papier. 1956;10:301–306. [Google Scholar]
- Shukla MR, Jones AMP, Sullivan JA, Liu C, Gosling S, Saxena PK. In vitro conservation of American elm (Ulmus americana): potential role of auxin metabolism in sustained plant proliferation. Canadian Journal of Forest Research. 2012;42:686–697. [Google Scholar]
- Široký J, Blackburn RS, Bechtold T, Taylor J, White P. Attenuated total reflectance Fourier-transform infrared spectroscopy analysis of crystallinity changes in lyocell following continuous treatment with sodium hydroxide. Cellulose. 2010;17:103–115. [Google Scholar]
- Skyba O, Douglas CJ, Mansfield SD. Syringyl-rich lignin renders poplars more resistant to degradation by wood decay fungi. Applied and Environmental Microbiology. 2013;79:2560–2571. doi: 10.1128/AEM.03182-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sluiter A, Hames B, Ruiz R, et al. Golden, CO: National Renewable Energy Laboratory; 2010. Determination of structural carbohydrates and lignin in biomass. Technical Report NREL/TP-510-42618. [Google Scholar]
- Smalley EB, Guries RP. Breeding elms for resistance to Dutch elm disease. Annual Review of Phytopathology. 1993;31:325–352. [Google Scholar]
- Solla A, Bohnens J, Collin E, et al. Screening European elms for resistance to Ophiostoma novo-ulmi. Forest Science. 2005;51:134–141. [Google Scholar]
- Sticklen MB, Bolyard MG, Hajela RK, Duchesne LC. Molecular and cellular aspects of Dutch elm disease. Phytoprotection. 1991;72:1–13. [Google Scholar]
- Strobel G, Van Alfen N, Hapner KD, McNeil M, Albersheim P. Some phytotoxic glycopeptides from Ceratocystis ulmi, the Dutch elm disease pathogen. Biochimica et Biophysica Acta. 1978;538:60–75. doi: 10.1016/0304-4165(78)90252-0. [DOI] [PubMed] [Google Scholar]
- Studer MH, DeMartini JD, Davis MF, et al. Lignin content in natural Populus variants affects sugar release. Proceedings of the National Academy of Sciences of the USA. 2011;108:6300–6305. doi: 10.1073/pnas.1009252108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suchy M, Linder MB, Tammelin T, Campbell JM, Vuorinen T, Kontturi E. Quantitative assessment of the enzymatic degradation of amorphous cellulose by using a quartz crystal microbalance with dissipation monitoring. Langmuir. 2011;27:8819–8828. doi: 10.1021/la2014418. [DOI] [PubMed] [Google Scholar]
- Svaldi R, Elgersma DM. Further studies on the activity of cell wall degrading enzymes of aggressive and non-aggressive isolates of Ophiostoma ulmi. European Journal of Forest Pathology. 1982;12:29–36. [Google Scholar]
- Sweet MS, Winandy JE. Influence of degree of polymerization of cellulose and hemicellulose on strength loss in fire-retardant-treated southern pine. Holzforschung. 1999;53:311–317. [Google Scholar]
- Temple B, Horgen PA, Bernier L, Hintz WE. Cerato-ulmin, a hydrophobin secreted by the causal agents of Dutch elm disease, is a parasitic fitness factor. Fungal Genetics and Biology. 1997;22:39–53. doi: 10.1006/fgbi.1997.0991. [DOI] [PubMed] [Google Scholar]
- Vane CH, Drage TC, Snape CE. Bark decay by the white-rot fungus Lentitula edodes: polysaccharide loss, lignin resistance and the unmasking of suberin. International Biodeterioration & Biodegradation. 2006;57:14–23. [Google Scholar]
- Vizárová K, Kirschnerová S, Kačík F, Briškárová A, Šutý Š, Katuščák S. Relationship between the decrease of degree of polymerisation of cellulose and the loss of groundwood pulp paper mechanical properties during accelerated ageing. Chemical Papers. 2012;12:1124–1129. [Google Scholar]
- Wise LE, Murphy M, D'Addieco AA. Chlorite holocellulose, its fractionation and bearing on summative wood analysis and on studies on the hemicelluloses. Paper Trade Journal. 1946;122:35–43. [Google Scholar]
- Zhang Y-HP, Lynd LR. Toward an aggregated understanding of enzymatic hydrolysis of cellulose: noncomplexed cellulase systems. Biotechnology and Bioengineering. 2004;88:797–824. doi: 10.1002/bit.20282. [DOI] [PubMed] [Google Scholar]