Abstract
Allocation of limiting resources, such as nutrients, is an important adaptation strategy for plants. Plants may allocate different nutrients within a specific organ or the same nutrient among different organs. In this study, we investigated the allocation strategies of nitrogen (N) and phosphorus (P) in leaves, stems and roots of 126 shrub species from 172 shrubland communities in Northern China using scaling analyses. Results showed that N and P have different scaling relationships among plant organs. The scaling relationships of N concentration across different plant organs tended to be allometric between leaves and non-leaf organs, and isometric between non-leaf organs. Whilst the scaling relationships of P concentration tended to be allometric between roots and non-root organs, and isometric between non-root organs. In arid environments, plant tend to have higher nutrient concentration in leaves at given root or stem nutrient concentration. Evolutionary history affected the scaling relationships of N concentration slightly, but not affected those of P concentration. Despite fairly consistent nutrients allocation strategies existed in independently evolving lineages, evolutionary history and environments still led to variations on these strategies.
Allocation of limiting resources is an important strategy for plants to adapt to changes in their environment1, which has been widely observed in the allocation of biomass2,3,4, functional traits5 and morphological plasticity6,7. Nutrients availability plays an important role in ecosystem function and development, since some essential nutrient elements, such as nitrogen (N) and phosphorus (P), are considered limiting resources in nature8,9. Allocation of nutrients involves in two main ways: strategic allocation of different nutrients within a specific organ and allocation of the same nutrient among organs. It has been widely observed that how different nutrients are coupled within a specific organ, such as leaves10,11,12, stems13, fine roots14,15 and twigs14. However, relatively little is known about how the same nutrient is allocated among organs. Plants need to allocate nutrients they absorb into different organs to meet the requirements of multiple functions such as growth, reproduction, nutrient storage and defense1. Nutrient allocation also reflects a plant's relative investments of essential molecules to different organs, such as N-rich proteins and P-rich RNAs16.
Previous studies have demonstrated that evolutionary history, environmental stresses and plant functional groups are possible factors influencing the relationship of nutrients among different organs16,17,18,19. From an evolutionary perspective, closely related species may share a similar evolutionary history and contain life history strategy5,20,21. Exploring how plants with different evolutionary history allocate N and P among organs can improve our understanding of correlated evolution16. In a meta-analysis, Kerkhoff et al.16 found that the scaling relationships of N and P concentrations between plant organs changed between metabolic and structural organs and proposed a general relationship describing N and P allocation among organs across diverse plant lineages. From a life-history perspective, exploring how plants allocate N and P among organs can improve our understanding of nutrient utilization strategies under climate change scenarios22. Plants need to balance the allocation of limiting resource to maximize their growth and may change allocation strategies as a response to changes in their environments such as climate and soil nutrient availability1,17,18,22,24. For example, Sardans & Peñuelas22 found a higher allocation of nutrients to leaves than to woody biomass in more humid climatic conditions. Plants may contain higher N concentrations in leaves to better adapt to arid environments25,26,27,28,29 through exploiting greater light availability25 while reducing stomatal conductance and increasing water use efficiency28,29. Plant functional groups have also been shown to influence allocation of nutrients through different photosynthetic pathways, fraction of woody tissue and N-fixation ability16,17,18,19. Further studies are needed for a better understanding of the combined impacts of climate, soil and evolutionary history on the allocation of nutrient among plant organs.
Nutrients such as N and P frequently limit plant growth and play important roles in plant functioning14,30,31. As a major element in proteins, N in plant tissues is related to various functions, such as photosynthesis in leaves, tissue respiration, conversion and storage of photosynthates and chemical defense in all plant organs32,33. As a major element of RNAs, P is mainly involved in the production of proteins32,34. Since different plant organs perform different functions in plant growth, the requirements of N and P will differ. For example, leaf N and P concentrations were found to have different scaling relationships with leaf photosynthetic capacity23. These differences may lead to differences in the scaling relationships of N and P concentrations among plant organs. Additionally, N and P concentrations in plants are widely reported to be more similar among closely related species35. Finally, plants are able to coordinate various organs with respect to absorption and allocation of limiting resources and adaptation to environmental constraints18; for example, plants tend to increase leaf N concentration to maintain growth under dry conditions25,26,27,28,29. Given the aforementioned ability of plants to adapt to different allocation strategies, we made the three following hypotheses. First, N and P have different scaling relationships across organs because of their different physiological functions. Second, the scaling relationships of N and P across organs depend on the phylogenetic relatedness of the plants; more similar nutrient allocation strategies exist in more closely related species. Third, we hypothesize that the scaling relationship of nutrients among plant organs changes with environmental factors, such as soil nutrients and precipitation; particularly, we expect higher leaf N concentration at given root or stem N concentration in more arid regions.
We tested the aforementioned hypotheses using an extensive investigation of N and P storage in different organs of shrubs across northern China. Compared to trees, shrubs are generally smaller and relatively more uniform in size among species because they do not have large trunks or roots, which weaken the “dilution effects” of N and P allocation to structural components of woody plants16. Shrubland represent a major vegetation type in Northern China with community types varying from east to west China along an aridity gradient36. In this study, we explored the relationships of N and P among leaves, stems and roots of shrubs in Northern China using scaling approach, which has been applied in exploring generalities of patterns and trade-offs in many plant traits10,16. Nutrient allocation among plant organs involves two facets: concentration and biomass storage16. We compared the scaling relationship for both concentrations and storages of N or P among leaves, stems and roots. The comparison has been conducted at following scales: 1) individual vs. species vs. phylogenetic levels; 2) species from the family Fabaceae vs. those from other families; 3) samples from different soil nutrient concentration levels and 4) samples from different climatic regions.
Results
Scaling of N and P among organs
Both concentrations and storages of N and P showed significant correlation among all organs at the individual, species, and phylogeny levels (Table 1).
Table 1. Summary of reduced major axis (RMA) regression results.
| Individual level | Species level | PICs | P value for likelihood ratio test | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| bind. | 95% CI | r2 | P | N | bsp | 95% CI | r2 | P | Nsp | bPIC | 95% CI | r2 | P | Ntip | bind.vs. bsp | bsp vs. bPIC | |
| Roots vs. leaves: | |||||||||||||||||
| N storage | 1.20 | 1.16–1.25 | 0.88 | <0.001 | 335 | 1.30 | 1.21–1.41 | 0.85 | <0.001 | 98 | 1.20 | 1.10–1.30 | 0.82 | <0.001 | 98 | 0.078 | 0.167 |
| P storage | 1.16 | 1.11–1.2 | 0.87 | <0.001 | 334 | 1.26 | 1.15–1.37 | 0.82 | <0.001 | 98 | 1.20 | 1.09–1.32 | 0.79 | <0.001 | 98 | 0.083 | 0.470 |
| N concentration | 1.79 | 1.68–1.91 | 0.37 | <0.001 | 613 | 2.07 | 1.79–2.38 | 0.51 | <0.001 | 107 | 2.04 | 1.72–2.43 | 0.20 | <0.001 | 107 | 0.073 | 0.721 |
| P concentration | 1.52 | 1.42–1.64 | 0.22 | <0.001 | 608 | 1.56 | 1.34–1.83 | 0.33 | <0.001 | 107 | 1.61 | 1.38–1.89 | 0.32 | <0.001 | 107 | 0.770 | 0.365 |
| Stems vs. leaves: | |||||||||||||||||
| N storage | 1.06 | 1.03–1.09 | 0.91 | <0.001 | 357 | 1.10 | 1.02–1.18 | 0.87 | <0.001 | 104 | 1.02 | 0.97–1.08 | 0.92 | <0.001 | 104 | 0.355 | 0.117 |
| P storage | 1.02 | 0.99–1.05 | 0.91 | <0.001 | 358 | 1.07 | 0.99–1.15 | 0.86 | <0.001 | 105 | 1.01 | 0.95–1.07 | 0.92 | <0.001 | 105 | 0.242 | 0.204 |
| N concentration | 1.55 | 1.45–1.66 | 0.23 | <0.001 | 655 | 1.72 | 1.49–1.99 | 0.37 | <0.001 | 115 | 1.74 | 1.45–2.09 | 0.04 | 0.025 | 115 | 0.201 | 0.962 |
| P concentration | 1.15 | 1.07–1.24 | 0.12 | <0.001 | 650 | 1.07 | 0.91–1.26 | 0.23 | <0.001 | 116 | 1.15 | 0.96–1.37 | 0.08 | <0.001 | 116 | 0.411 | 0.571 |
| Roots vs. stems: | |||||||||||||||||
| N storage | 1.12 | 1.09–1.16 | 0.91 | <0.001 | 343 | 1.14 | 1.07–1.22 | 0.89 | <0.001 | 103 | 1.16 | 1.07–1.25 | 0.84 | <0.001 | 103 | 0.617 | 0.778 |
| P storage | 1.12 | 1.08–1.16 | 0.89 | <0.001 | 344 | 1.15 | 1.06–1.23 | 0.85 | <0.001 | 104 | 1.18 | 1.07–1.26 | 0.8 | <0.001 | 104 | 0.599 | 0.612 |
| N concentration | 1.14 | 1.09–1.19 | 0.67 | <0.001 | 652 | 1.22 | 1.11–1.33 | 0.75 | <0.001 | 113 | 0.94 | 0.80–1.11 | 0.34 | <0.001 | 113 | 0.185 | 0.008 |
| P concentration | 1.34 | 1.26–1.43 | 0.29 | <0.001 | 646 | 1.48 | 1.26–1.74 | 0.22 | <0.001 | 113 | 1.50 | 1.25–1.81 | 0.03 | 0.050 | 113 | 0.286 | 0.894 |
Abbreviations: PIC, phylogenetically independent contrast; CI, confidence interval.
Regression slopes (bind., bsp and bPIC) estimates in bold are significantly different from 1, indicate that the scaling relationship of the two traits are anisometric.
At the individual level, the slopes of N concentration in all organ pairs were significantly different from 1; the slopes for root vs. leaf and stem vs. leaf were significantly larger than that of root vs. stem. At the species level, the slopes were all significantly different from 1; the slope was significantly larger for root vs. leaf and stem vs. leaf than for root vs. stem. At the phylogeny level, the PIC analyses also revealed a similar pattern to individual and species level (Table 1). The slope of the root vs. stem was not significantly different from 1 when PIC is applied. There were no significant differences between the slopes at individual level and species level (P > 0.05). Slopes at the species level were not significantly different (P > 0.05) from the phylogeny level except the root vs. stem (P = 0.012).
Scaling relationship of P concentration among organs was different from that of N (Table 1). At the individual level, the slopes of P concentration in all organ pairs were significantly different from 1; the slope for root vs. leaf or root vs. stem was more deviated than for stem vs. leaf. At the species level, the slope was significantly larger for root vs. leaf and root vs. stem than for stem vs. leaf (Table 1). The PIC analysis depicted that, at the phylogeny level, the slope was significantly larger for root vs. leaf and root vs. stem than for stem vs. leaf. There were no significant differences between the individual, species and phylogeny level (P > 0.05).
The RMA slopes for N and P storages of the same organ pairs were similar at all three levels. For N storage at individual and species levels, the slopes of root vs. leaf, stem vs. leaf and root vs. stem were significantly larger than 1. The PIC analysis depicted that, at the phylogeny level, the slopes of root vs. leaf and root vs. stem were significantly larger than 1, while that of stem vs. leaf was equivalent to 1. For P storage at all levels, the slopes of root vs. leaf and root vs. stem were significantly larger than 1, while the slopes of stem vs. leaf were equivalent to 1.
Influence of functional type on scaling of N and P among organs
All RMA regression slopes were not significantly different between legumes and non-legumes (P > 0.05), except the root N concentration vs. stem N concentration (P = 0.04) (Figure 1).
Figure 1. Scatterplots showing the RMA regressions of N concentration (a–c), P concentration (d–f), N storage (g–i) and P storage (j–l) among organs for legume (black solid dots) and non-legume (gray open cycles) shrubs.
Separate lines indicate the slopes of non-legumes (blue) and legumes (red) are significantly different (likelihood ratio tests, P < 0.05), whereas a single black line indicates otherwise. Lines with the slopes equal to 1 are shown with dotted lines.
All RMA regression slopes of N and P storages among different plant organs were not significantly different between legumes and non-legumes (P > 0.05).
Influence of soil nutrient on scaling of N and P among organs
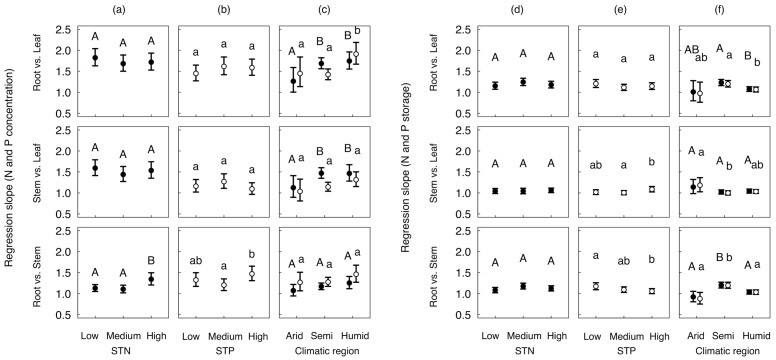
The slopes of N concentration among organs were not significantly different across different soil total N concentration (STN) levels, except that at the highest STN level. The slope of root vs. stem was significantly different than the slopes at the other two STN levels (high STN vs. low STN: P = 0.009 and high STN vs. medium STN: P = 0.005) (Figure 2a). The slopes of N storage among organs were not significantly different across different STN levels (P > 0.05) (Figure 2d).
Figure 2. Comparisons of the RMA regression slopes of N concentration and storage (solid dots), P concentration and storage (open cycles) among different STN levels (a), (d), STP levels (b), (e) and climatic regions (c), (f).
Line bars show 95% confidence intervals (CI). Letters above the line bars show the results of likelihood ratio tests (uppercase for N concentration and storage, lowercase for P concentration and storage). Slopes with the same letters are not significantly different (P ≥ 0.05), while those with different letters are significantly different (P < 0.05).
At different soil total P concentration (STP) levels, the slopes of P concentration among organs were not significantly different across different STP levels, except a slightly larger slope of root vs. stem at the highest STP (high STP vs. medium STP: P = 0.017) (Figure 2b). The slopes of P storage among organs were not significantly different across different STP levels (P > 0.05) (Figure 2e).
Influence of aridity on scaling of N and P among organs
The slopes of N concentration in root vs. leaf were significantly higher in semi-arid/semi-humid (Semi) and humid (Humid) regions than in arid regions (Arid) (Arid vs. Semi: P = 0.020 and Arid vs. Humid: P = 0.014). Similarly, the slopes of stem vs. leaf N concentration were significantly higher in Semi and Humid than in Arid (Arid vs. Semi: P = 0.031 and Arid vs. Humid: P = 0.049). The slope of root vs. stem was not significantly different among three climatic regions (Figure 2c).
The slope of P concentration in root vs. stem was not significantly different between Arid and Semi, and significantly higher in Humid than in Semi (Arid vs. Humid: P = 0.048 and Semi vs. Humid: P < 0.001). The slopes of stem vs. leaf and root vs. stem were not significantly different among three climatic regions (Figure 2c).
The slopes of N and P storages among different organs were quite similar at different climatic regions (Figure 2f). The slopes of N and P storage in root vs. leaf were not significantly different between Arid and Semi, but were significantly lower in Humid than in Semi (P = 0.001 and 0.006 for N and P, respectively). The slopes of P storage in stem vs. leaf were significantly higher in Arid than in Semi (P = 0.028), and were not significantly different between Semi and Humid. Slopes of N storage were not significantly different among three climatic regions. The slopes in root vs. stem were significantly higher in Semi than in Arid and Humid (Arid vs. Semi: P = 0.001 for both N and P; Semi vs. Humid: P < 0.001 for both N and P).
Discussion
Using concentrations and storages of N and P in leaves, stems and roots of 126 shrub species from 172 shrubland sites, we studied the allocation strategies of N and P among organs through a scaling approach. Both concentrations and storages of N and P between all pairs of plant organs are significantly correlated at individual, species and phylogeny levels (Table 1). The coordinate variations of nutrients across different organs demonstrate that plant organs are not independent. The allocation of nutrients among organs might be another important part of life history strategy for plants. For example, plants with high leaf nutrient concentration would also have high nutrient concentration in stem and root to increase nutrient uptaking and phloem loading to meet the requirement of higher photosynthesis rate and photosynthate export. At the community level, this coordination would also help to predict changes in nutrient storage in plants under the climate change scenarios, since increased dominance by species with higher nutrient concentration and storage in one organ will lead to predictable increases in nutrient concentration and storage in other organs16.
Concentrations of nutrients in lower organs increase with those in the neighboring upper organs in a power law form (e.g., roots vs. stems, stems vs. leaves, see Figures 1 and 2, Table 1). This result is consistent with Brouwer's hypothesis that plant organs are competing for nutrients during growth, and that the organ nearest to the nutrient source will be most successful37,38,39, as nutrients are transported to distant organs only after the needs by adjacent organs are met39.
As hypothesized, N and P concentrations show different scaling relationships among organs. Consistent with Kerkhoff et al.16, we find that scaling relationships of N concentration in photosynthetic organ (leaf) vs. N concentration in non-photosynthetic organs (stem and root) tend to be allometric, while that of N concentration between stem vs. root is very close to (although not) isometric (Table 1). Particularly, there is a faster increase of N concentration in non-leaf organs as leaf N concentration increases (b > 1.0), partly because non-leaf organs are closer to the nutrient source (soil)37,38,39. A second possible reason might be related to the activity of vascular tissues. Phloem tissues require more N investments and higher rates of N cycling for the high rate of photosynthate transportation and photosynthetic activity16,40. Among the non- photosynthetic organs, we also observe that the scaling relationships are not strictly isometric, partly because of the difference in distance to the nutrient sources37,38,39. Contrary to N concentration, the scaling slopes of P concentration between non-root organs are close to 1, but higher in root vs. non-root organs, partly because that root is the closest organ to P source. This difference between the scaling relationships of N and P concentrations among organs might because of different requirements of N and P among plant organs to perform various functions. However, it is difficult to fully understand these differences without measuring important physiological processes of plant organs, such as photosynthesis, respiration, nutrient absorption and transportation, or plant functional traits which can represent these physiological processes. Future studies including plant physiological processes will help to further explain the mechanisms behind the scaling relationships of N and P across plant organs.
In contrast to concentrations, the scaling slopes of N and P storages among plant organs are quite similar, which indicates that the allocations of N and P in plants largely depends on the allocation of biomass. This result is reasonable because both N and P are only small portions of total plant tissue, and the pools of N and P in plant tissues mainly reflect the tissue size rather than composition.
Most scaling relationships remain unchanged after phylogeny was controlled, indicating that the scaling relationships of nutrient concentrations across plant organs are independent of phylogenetic relationships between species. We also observed the same scaling relationships for most organ pairs between legume and non-legume shrubs. Although plant N and P concentrations are considered phylogenetically conservative traits16,35,41, that was not the case for the allocation of N and P among organs in this study. The scaling relationships of nutrient concentration across most organs are not simply a result of shared evolutionary history of these species16.
However, we still found significant difference between species and phylogeny level relationships for root N concentration vs. stem N concentration. Similarly, we found significantly different scaling relationships in root N concentration vs. stem N concentration between legumes and non-legumes. These results indicate that the N fixing pathways used by legumes impact the allocation strategy of N in shrubs. The difference in the scaling of N between legumes and non-legumes supports the hypothesis that plant functional group affects plant traits as well as their scaling relationships16,42. Therefore, it is important to take plant functional group into consideration when exploring plant nutrient allocation strategies.
We did not observe a significant influence of STN and STP on the allocation of nutrients among organs, suggesting that plants do not change their nutrients allocation strategies even under soil nutrients deficiency. Available soil N and P can be absorbed directly by plants, and thus might influence N and P concentrations in plants43,44. However, recent studies on leaf traits of shrubs and grasses conducted across Inner Mongolia suggests that total and available soil nutrient concentrations explain similar and a very small amount of variance in leaf N and P concentrations of shrub species45,46. These results indicate that in the region studied, total soil nutrient concentrations do not differ from available soil nutrient concentrations in explaining or predicting plant nutrient concentrations, which is consistent with our results. Other soil properties such as soil age47, soil freezing and disturbance17 may impact nutrient allocation in plants; however, these factors are not included in the present study. Further studies are needed to examine the effect of soil properties on the allocation of nutrient in plants.
Plants in arid conditions tend to have higher leaf N concentration in order to exploit light25 and increase the water use efficiency at the expense of lowering N-use efficiency28,29. As expected, increases of N concentration in non-photosynthetic organs are even faster in humid than in arid regions (Figure 2c). Our result supplements these theories by considering the relationship of nutrient concentrations among plant organs. Although the increase of N concentration in non-leaf organs is always faster than the increase in leaf N concentration in all the three climate regions, leaf N concentration tends to be higher at given root or stem N concentration under arid region than humid region. The scaling relationships of P concentration between non-leaf organs and leaves showed similar variation as N concentration among three climate regions. Another possible reason might be that water is the limiting factor for vegetation growth in shrublands of Northern China and increases in water availability promotes plant growth48. During growth, more photosynthate and nutrients will be transported to non-photosynthetic organs13. This result indicates that shrub species in Northern China display a strategy that favors higher nutrient concentration in leaves than in non-photosynthetic organs in dry environments. The variation tendencies of RMA slopes of N and P storages are almost the same under different aridity conditions, which indicates that aridity can influence plant nutrient allocation through variation in plant biomass allocation. In semi-arid/semi-humid regions, plants tend to allocate more biomass to their root systems than to aboveground organs.
In summary, we investigated the scaling relationship of N and P in leaves, stems and roots based on samples of 126 shrub species from 172 shrubland communities in Northern China. We found that plants have different strategies of allocation for N and P. The scaling relationships of N concentration across different plant organs tended to be allometric between leaves and non-leaf organs, and isometric between non-leaf organs. Whilst the scaling relationships of P concentration tend to be allometric for root vs. stem and for root vs. leaf, it tends to be isometric for stem vs. leaf. The scaling relationships of both concentrations and storages of N and P among organs are affected by aridity, but not by soil nutrient concentration. In arid environments, plant tend to have higher nutrient concentration in leaves at given root or stem nutrient concentration. Most scaling relationships of concentrations and storages of N and P across plant organs don't exhibit phylogenetic signal, except that of N concentration, which could be attribute to the difference of legumes and non-legumes in N utilization. We therefore conclude that consistent nutrient scaling relationships among plant organs exist in independently evolving lineages, but factors such as climate and N-fixation ability lead to variations in these relationships.
Methods
Study site and investigation
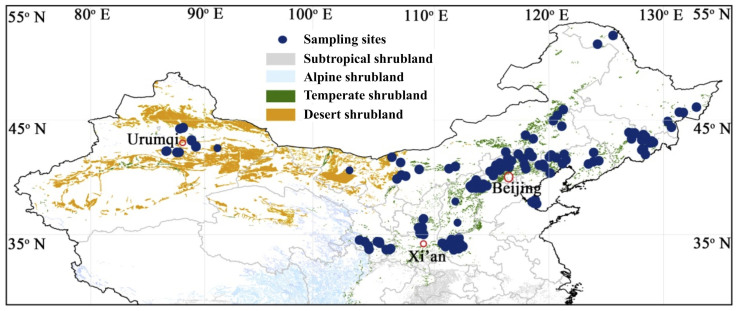
This study was carried out at 172 shrubland sites extending 46.1 degrees in longitude (86.7–132.8°E) and 18.7 degrees in latitude (33.7–52.4°N) in Northern China between July and September (mostly July and August) 2011 (Figure 3). Three 5 m × 5 m plots were selected at each site to represent the natural shrubland communities. We identified all individuals to the species level. In each plot, leaf, stem and root biomass were harvested, and all biomass were harvests separately for each species. For each shrub species encountered in the community, we selected three to five individuals and collected fully expanded leaves, stems and roots (mostly coarse roots in the top 30 cm of soil) at each site. Plant samples were oven-dried and ground after being transported to the laboratory. In total, we sampled 702 individuals of 126 shrub species from 71 genera and 33 families. All of the shrub species sampled were deciduous except the Pinus tabuleaformis in one study site.
Figure 3. Locations of the sampling sites.
The background shows the distribution of shrubland biomes in the northern part of China based on the “Vegetation map of the People's Republic of China (1:1000000)”36. The map was generated using ArcGIS 10.3 (http://www.esri.com/).
We collected soil samples using three one-meter-deep pits along the diagonal of each plot. For each profile, soil at the depths of 0–10, 10–20, 20–30, 30–50, 50–70 and 70–100 cm was sampled and soil samples from the same depth were well mixed. Soil samples were air-dried, had roots removed and were ground to pass through a 100-mesh sieve.
Monthly precipitation and mean monthly temperature was obtained from the WorldClim website49 (available at www.worldclim.org; resolution of 1 km).
Measurements
The plant and soil samples were analyzed at the Measurement Center of the Institute of Botany, Chinese Academy of Sciences. Total nitrogen concentrations of soil (STN) and plant samples (leaf, stem and root N concentrations) were analyzed using an elemental analyzer (2400 II CHNS; Perkin-Elmer, Boston, MA, USA) under 950°C for combustion then reduced to 640°C. Total phosphorus concentrations of soil (STP) and plant samples (leaf, stem and root P concentrations) were analyzed using the molydate/ascorbic acid method after H2SO4-H2O2 digestion50. We only used STN and STP at 0–10 cm depth interval for the analyses because STN and STP at this depth interval were highly correlated with those at the other five depth intervals.
Scaling of N and P among different organs
A scaling approach, Y = aXb, was used to examine the covariation in N and P concentrations because both concentration and storage of N and P tend to be log normally distributed16,51. The power function can be expressed in the form of a linear regression equation after log-transforming, where the exponent b is the regression slope and a is the regression intercept. The N and P storages of organs were calculated by multiplying the concentrations of N and P of the organs by the corresponding biomasses.
We first applied reduced major axis (RMA) to examine the correlation of nutrients among organs. An important advantage of RMA regression compared with ordinary least square regression is that RMA minimizes sums of squares in X and Y simultaneously. We assigned the scaling relationship between Y and X as isometric when the 95% confidence interval (CI) of b contains 1; the scaling relationship is otherwise allometric. A b above 1 indicates that, on average, Y increases faster than linearly with X; whereas a b below 1 indicates Y increases slower than linearly with X10,16. In the RMA analyses, we set the organs closer to the top of the plant as the X variable.
To examine the effects of soil nutrient on the relationship of nutrient concentration and storage across organs, we divided all individuals into three equal subgroups based on soil nutrient (STN and STP) and compared the exponential slopes among different soil and climate levels (STN < 0.9, 0.9 ≤ STN ≤ 2.3 and STN > 2.3 for low, medium and high STN, respectively; STP < 0.47, 0.47 ≤ STP ≤ 0.63 and STP > 0.63 for low, medium and high STP, respectively).
We also identified three climatic areas to examine the effects of aridity on the scaling relationships of nutrient concentrations and storages across plant organs based on the aridity index in growing season (GAI) defined as the ratio of total precipitation to potential evapotranspiration from May to October52. Potential evapotranspiration was calculated using the Thornthwaite equation53. GAI increases from arid to humid climate. We divided the range of GAI of the studied region in to three groups: arid regions, with GAI < 0.59 (the least one third of the GAI, Arid); semi-arid/semi-humid regions, with GAI between 0.59 and 1.17 (the median one third of the GAI, Semi); and humid regions, with GAI > 1.17 (the highest one third of the GAI, Humid).
To test the influence of plant functional groups on the scaling relationship of nutrient concentrations and storage across plant organs, we further grouped all samples into legumes (samples from the family Fabaceae) and non-legumes (samples from other families), as the legume shrubs fix nitrogen whereas the non-legume shrubs do not.
A likelihood ratio test54 was used to test the heterogeneity between RMA regression slopes of different groups, i.e., different soil nutrient level, climatic regions, and functional groups.
Phylogenetic tree and phylogenetic analyses
We also looked for the phylogenetic signals of all RMA regression slopes. To do this, we first constructed a phylogenetic tree for the 126 species using Phylomatic55 based on APG III topology56. The branch lengths were determined using BLADJ algorithm within the Phylocom software (http://www.phylodiversity.net/phylocom/) and the node ages were fossil-estimated57. We then applied a phylogenetically independent contrast (PIC) analysis58. PIC analysis allows us to calculate N-1 standardized contrasts of a trait using a series of trait data for N species. We then conducted RMA regression at both species-level data and PICs, we tested the effects of evolutionary history on covariation of N and P concentrations and storages across organs by comparing the RMA regression slopes at species-level and the PIC analysis using the likelihood ratio test.
All analyses were performed using RStudio with the basic, smatr and picante packages (http://www.R-project.org/).
Author Contributions
X.Y. and Z.T. conceived and designed the study, conducted analyses and wrote the paper. X.Y., Z.T., C.J., H.L., W.M., A.M., Z.S., W.S., T.W., X.P.W., X.W., S.Y., M.Y. and C.Z. contributed data, discussed the draft manuscript and interpreted the results.
Acknowledgments
We are grateful to Yahan Chen from the Institute of Botany, Chinese Academy of Sciences for conducting the measurements. We would also like to thank Alison Beamish at the University of British Columbia for English editing. This work was partly supported by the “National Program on Key Basic Research Project (#2014CB954004 & 2010CB950602)”, “Strategic Priority Research Program” of CAS (#XDA05050301), and the NSFC (#31321061 & 31370620).
Footnotes
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- Bazzaz A. F. & Grace J. Plant resource allocation (Academic Press, San Diego, 1997). [Google Scholar]
- Niklas K. J. & Enquist B. J. Invariant scaling relationships for interspecific plant biomass production rates and body size. Proc. Natl. Acad. Sci. U. S. A. 98, 2922–2927 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niklas K. J. & Enquist B. J. On the vegetative biomass partitioning of seed plant leaves, stems, and roots. Am. Nat. 159, 482–497 (2002). [DOI] [PubMed] [Google Scholar]
- Enquist B. J. & Niklas K. J. Global allocation rules for patterns of biomass partitioning in seed plants. Science 295, 1517–1520 (2002). [DOI] [PubMed] [Google Scholar]
- Reich P. B. et al. The evolution of plant functional variation: traits, spectra, and strategies. Int. J. Plant Sci. 164, S143–S164 (2003). [Google Scholar]
- Fransen B., de Kroon H. & Berendse F. Root morphological plasticity and nutrient acquisition of perennial grass species from habitats of different nutrient availability. Oecologia 115, 351–358 (1998). [DOI] [PubMed] [Google Scholar]
- Grime J. P. & Mackey J. M. L. The role of plasticity in resource capture by plants. Evol. Ecol. 16, 299–307 (2002). [Google Scholar]
- Koerselman W. & Meuleman A. F. M. The vegetation N:P ratio: a new tool to detect the nature of nutrient limitation. J. Appl. Ecol. 33, 1441–1450 (2007). [Google Scholar]
- LeBauer D. S. & Treseder K. K. Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 89, 371–379 (2008). [DOI] [PubMed] [Google Scholar]
- Wright I. J. et al. The worldwide leaf economics spectrum. Nature 428, 821–827 (2004). [DOI] [PubMed] [Google Scholar]
- Han W., Fang J., Guo D. & Zhang Y. Leaf nitrogen and phosphorus stoichiometry across 753 terrestrial plant species in China. New Phytol. 168, 377–385 (2005). [DOI] [PubMed] [Google Scholar]
- He J. et al. Stoichiometry and large-scale patterns of leaf carbon and nitrogen in the grassland biomes of China. Oecologia 149, 115–122 (2006). [DOI] [PubMed] [Google Scholar]
- Helmisaari H.-S. & Siltala T. Variation in nutrient concentrations of Pinus sylvestris stems. Scand. J. For. Res. 4, 443–451 (1989). [Google Scholar]
- Guo D. L., Mitchell R. J. & Hendricks J. J. Fine root branch orders respond differentially to carbon source-sink manipulations in a longleaf pine forest. Oecologia 140, 450–457 (2004). [DOI] [PubMed] [Google Scholar]
- Gordon W. S. & Jackson R. B. Nutrient concentrations in fine roots. Ecology 81, 275–280 (2000). [Google Scholar]
- Kerkhoff A. J., Fagan W. F., Elser J. J. & Enquist B. J. Phylogenetic and growth form variation in the scaling of nitrogen and phosphorus in the seed plants. Am. Nat. 168, E103–E122 (2006). [DOI] [PubMed] [Google Scholar]
- Craine J. M., Lee W. G., Bond W. J., Williams R. J. & Johnson L. C. Environmental constraints on a global relationship among leaf and root traits of grasses. Ecology 86, 12–19 (2005). [Google Scholar]
- Liu G. et al. Coordinated variation in leaf and root traits across multiple spatial scales in Chinese semi-arid and arid ecosystems. New Phytol. 188, 543–553 (2010). [DOI] [PubMed] [Google Scholar]
- Li A., Guo D., Wang Z. & Liu H. Nitrogen and phosphorus allocation in leaves, twigs, and fine roots across 49 temperate, subtropical and tropical tree species: a hierarchical pattern. Funct. Ecol. 24, 224–232 (2010). [Google Scholar]
- Swenson N. G. & Enquist B. J. Ecological and evolutionary determinants of a key plant functional trait: wood density and its community-wide variation across latitude and elevation. Am. J. Bot. 94, 451–459 (2007). [DOI] [PubMed] [Google Scholar]
- Fortunel C., Fine P. V. A. & Baraloto C. Leaf, stem and root tissue strategies across 758 Neotropical tree species. Funct. Ecol. 26, 1153–1161 (2012). [Google Scholar]
- Sardans J. & Peñuelas J. Tree growth changes with climate and forest type are associated with relative allocation of nutrients, especially phosphorus, to leaves and wood. Glob. Ecol. Biogeogr. 22, 494–507 (2013). [Google Scholar]
- Reich P. B. et al. Evidence of a general 2/3-power law of scaling leaf nitrogen to phosphorus among major plant groups and biomes. Proc. R. Soc. B Biol. Sci. 277, 877–883 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han W. X., Fang J. Y., Reich P. B., Ian Woodward F. & Wang Z. H. Biogeography and variability of eleven mineral elements in plant leaves across gradients of climate, soil and plant functional type in China. Ecol. Lett. 14, 788–796 (2011). [DOI] [PubMed] [Google Scholar]
- Cunningham S. A., Summerhayes B. & Westoby M. Evolutionary divergences in leaf structure and chemistry, comparing rainfall and soil nutrient gradients. Ecol. Monogr. 69, 569–588 (1999). [Google Scholar]
- Wright I. J., Reich P. B. & Westoby M. Strategy shifts in leaf physiology, structure and nutrient content between species of high- and low-rainfall and high- and low-nutrient habitats. Funct. Ecol. 15, 423–434 (2001). [Google Scholar]
- Farquhar G. D., Buckley T. N. & Miller J. M. Optimal stomatal control in relation to leaf area and nitrogen content. Silva Fenn. 36, 625–627 (2002). [Google Scholar]
- Wright I. J., Reich P. B. & Westoby M. Least-cost input mixtures of water and nitrogen for photosynthesis. Am. Nat. 161, 98–111 (2003). [DOI] [PubMed] [Google Scholar]
- Palmroth S. et al. On the complementary relationship between marginal nitrogen and water-use efficiencies among Pinus taeda leaves grown under ambient and CO2-enriched environments. Ann. Bot. 111, 467–77 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Güsewell S. N:P ratios in terrestrial plants: variation and functional significance. New Phytol. 164, 243–266 (2004). [DOI] [PubMed] [Google Scholar]
- Vitousek P. M. Nutrient cycling and limitation: hawai'i as a model system (Princeton University Press, Princeton, 2004). [Google Scholar]
- Sterner R. W. & Elser J. J. Ecological stoichiometry: the biology of elements from molecules to the biosphere (Princeton University Press, Princeton, 2002). [Google Scholar]
- Reich P. B. et al. Scaling of respiration to nitrogen in leaves, stems and roots of higher land plants. Ecol. Lett. 11, 793–801 (2008). [DOI] [PubMed] [Google Scholar]
- Vrede T., Dobberfuhl D. R., Kooijman S. A. L. M. & Elser J. J. Fundamental connections among organism C:N:P stoichiometry, macromolecular composition, and growth. Ecology 85, 1217–1229 (2004). [Google Scholar]
- Stock W. D. & Verboom G. A. Phylogenetic ecology of foliar N and P concentrations and N:P ratios across Mediterranean-type ecosystems. Glob. Ecol. Biogeogr. 21, 1147–1156 (2012). [Google Scholar]
- Editorial committee of Vegetation map of China. Vegetation map of the People's Republic of China (1:1000,000) (Geological Publishing House, Beijing, 2007). [Google Scholar]
- Brouwer R. Nutritive influences on the distribution of dry matter in the plant. Neth. J. Agric. Sci. 10, 361–376 (1962). [Google Scholar]
- Brouwer R. Functional equilibrium: sense or nonsense? Neth. J. Agric. Sci. 31, 335–348 (1983). [Google Scholar]
- Yang Z. & Midmore D. J. Modelling plant resource allocation and growth partitioning in response to environmental heterogeneity. Ecol. Modell. 181, 59–77 (2005). [Google Scholar]
- Marschner H., Kirkby E. A. & Engels C. Importance of cycling and recycling of mineral nutrients within plants for growth and development. Bot. Acta. 110, 265–273 (1997). [Google Scholar]
- Broadley M. R. et al. Phylogenetic variation in the shoot mineral concentration of angiosperms. J. Exp. Bot. 55, 321–336 (2004). [DOI] [PubMed] [Google Scholar]
- Reich P. B. et al. Variation in growth rate and ecophysiology among 34 grassland and savanna species under contrasting N supply: a test of functional group differences. New Phytol. 157, 617–631 (2003). [DOI] [PubMed] [Google Scholar]
- McNeill A. & Unkovich M. [The nitrogen cycle in terrestrial ecosystems]. Nutrient cycling in terrestrial ecosystems [Marschner P., & Rengel Z. (eds.)] [37–64] (Springer, Heidelberg, 2007). [Google Scholar]
- Bünemann E. K. & Condron L. M. [Phosphorus and sulphur cycling in terrestrial ecosystems]. Nutrient cycling in terrestrial ecosystems [Marschner P., & Rengel Z. (eds.)] [65–92] (Springer, Heidelberg, 2007). [Google Scholar]
- Liu C. et al. Relative effects of phylogeny, biological characters and environments on leaf traits in shrub biomes across central Inner Mongolia, China. J. Plant Ecol. 6, 220–231 (2013). [Google Scholar]
- Geng Y., Wu Y. & He J.-S. Relationship between leaf phosphorus concentration and soil phosphorus availability across Inner Mongolia grassland. Chinese J. Plant Ecol. 35, 1–8 (2011). [Google Scholar]
- Lambers H., Raven J. A., Shaver G. R. & Smith S. E. Plant nutrient-acquisition strategies change with soil age. Trends Ecol. Evol. 23, 95–103 (2008). [DOI] [PubMed] [Google Scholar]
- Bai Y. et al. Primary production and rain use efficiency across a precipitation gradient on the Mongolia plateau. Ecology 89, 2140–2153 (2008). [DOI] [PubMed] [Google Scholar]
- Hijmans R. J., Cameron S. E., Parra J. L., Jones P. G. & Jarvis A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005). [Google Scholar]
- Jones J. B. Laboratory guide for conducting soil tests and plant analysis. (CRC Press, New York, 2001). [Google Scholar]
- Warton D. I., Wright I. J., Falster D. S. & Westoby M. Bivariate line-fitting methods for allometry. Biol. Rev. Camb. Philos. Soc. 81, 259–291 (2006). [DOI] [PubMed] [Google Scholar]
- UNEP. World atlas of desertification (Arnold Publisher, London, 1992). [Google Scholar]
- Thornthwaite C. W. & Hare F. K. Climatic classification in forest. Unasylva 9, 51–59 (1955). [Google Scholar]
- Warton D. I. & Weber N. C. Common slope tests for bivariate errors-in-variables models. Biometrical J. 44, 161–174 (2002). [Google Scholar]
- Webb C. O. & Donoghue M. J. Phylomatic: tree assembly for applied phylogenetics. Mol. Ecol. Notes 5, 181–183 (2005). [Google Scholar]
- Bremer B. et al. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG III. Bot. J. Linn. Soc. 161, 105–121 (2009). [Google Scholar]
- Wikström N., Savolainen V. & Chase M. W. Evolution of the angiosperms: calibrating the family tree. Proc. Biol. Sci. 268, 2211–2220 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Felsenstein J. Phylogenies and the comparative method. Am. Nat. 125, 1–15 (1985). [DOI] [PubMed] [Google Scholar]