Abstract
Objective
Since dermatomyositis (DM) is associated with an increased risk of malignancy, accurate identification of patients likely to harbor cancers is important. Using immunoprecipitations from radiolabeled cell lysates, several groups recently showed that anti–transcription intermediary factor 1γ (anti–TIF-1γ) antibodies are associated with malignancy in DM. We undertook this study to develop sensitive, specific assays to detect antibodies against TIF-1γ and nuclear matrix protein NXP-2 and to evaluate their association with malignancy in DM.
Methods
To detect anti–TIF-1γ antibodies, immunoprecipitations were performed using lysates made from HeLa cells overexpressing TIF-1γ, with detection by immunoblotting. Anti–NXP-2 antibodies were assayed by immunoprecipitation using 35S-methionine–labeled NXP-2 generated by in vitro transcription/translation. We analyzed patient sera from DM cohorts seen at the Stanford University Dermatology Clinic (n = 111) and the Johns Hopkins Myositis Center (n = 102).
Results
A total of 17% and 38% of patients had antibodies against NXP-2 and TIF-1γ, respectively. Reactivity against either NXP-2 or TIF-1γ identified 83% of patients with cancer-associated DM. In addition to older age and male sex, cancer was associated with antibodies to NXP-2 or TIF-1γ on multivariate analysis (odds ratio 3.78 [95% confidence interval 1.33–10.8]). Stratification by sex revealed that anti–NXP-2 was specifically associated with cancer in males (odds ratio 5.78 [95% confidence interval 1.35–24.7]).
Conclusion
These studies demonstrate that anti–NXP-2 and anti–TIF-1γ antibodies are frequent DM specificities (found in 55% of patients) and are present in most patients with cancer-associated DM.
Dermatomyositis (DM) is a systemic disease characterized by chronic inflammation in the skin and muscle. There is significant clinical heterogeneity with respect to lung or muscle inflammation, patterns of cutaneous inflammation, association with internal malignancy, and response to therapy. Many patients with DM have circulating autoantibodies, which are often associated with distinct clinical phenotypes (1,2). Interestingly, autoantibodies in DM tend to be mutually exclusive, suggesting that specific immune responses might play a role in shaping different phenotypes. It is well established that a subset of DM patients (10–20%) is at increased risk of internal malignancy around the time of DM diagnosis (3). Identification of patients at high risk of cancer remains a high priority for physicians treating these patients.
Recent studies have identified 3 new autoantibody specificities in DM: melanoma differentiation–associated protein 5 (MDA-5), transcription intermediary factor 1γ (TIF-1γ), and nuclear matrix protein NXP-2 (also known as MORC3) (4–9). These antigens all migrate similarly on sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gels (145–155 kd).
Anti–MDA-5 antibodies are found in ~10–20% of DM patients (7,10–12) (and in up to 65% of patients with clinically amyopathic disease [6,10,13,14]) and are associated with interstitial lung disease (often rapidly progressive, especially in Asians), mild or absent muscle disease, cutaneous ulceration, palmar papules, arthritis/arthralgia, and alopecia (6,11,15). Anti–TIF-1γ antibodies are frequently found in cancer-associated DM (4,13,16–18), including in 2 studies with largely Caucasians (19,20). For adult DM patients with antibodies to TIF-1γ, the percentage of patients harboring a malignancy ranges from 42% to 100%, depending on the study (4,13,16–18). In juvenile DM patients, antibodies to TIF-1γ are not associated with malignancy, but rather, with skin ulceration and more extensive cutaneous disease (21). NXP-2 has also been identified as an autoantibody target in a subset (1.6–30%) of adult DM patients (22–25). Originally identified as MJ in the juvenile DM population, this antigen appeared to be targeted by antibodies in patients with a higher risk of cutaneous calcinosis cutis (5,26). In adult DM patients, antibodies to NXP-2 are not associated with any obvious phenotype other than a trend toward increased risk of calcinosis cutis in 1 study (23). One study of adult Japanese DM patients suggested an association with malignancy (3 of 7 anti–NXP-2–positive patients had a primary malignancy); however, this association was difficult to verify given the small numbers of anti–NXP-2–positive patients (24).
There are several major drawbacks of previous studies looking at antibody associations with cancer in DM. The assays used to detect these antibodies lack sensitivity and specificity, given that NXP-2, TIF-1γ proteins, and MDA-5 migrate similarly on gel electrophoresis and may not be optimally expressed using standard cell lines and culture conditions. Additionally, many of the cohorts or antibody-positive patients have been relatively small in number. In this study, we provide definitive evidence that antibodies to NXP-2 and TIF-1γ identify the vast majority of the patients with cancer-associated DM in 2 separate, well-defined DM cohorts.
PATIENTS AND METHODS
Patients
All patients were seen in the outpatient clinics of either the Stanford University Department of Dermatology or Johns Hopkins Myositis Center between January 2003 and March 2012. Both the Stanford University and Johns Hopkins University Institutional Review Boards approved the collection of plasma/serum from the DM patients for use in this analysis. The population from which material was collected represented ~90% of the total number of DM patients seen during this time period. Patients were included only if they were over the age of 18 years and had a diagnosis of probable or definite DM based on the criteria of Bohan and Peter (27) or, for patients with clinically amyopathic disease, based on the characteristic skin findings suggested by Sontheimer (28). All patients with clinically amyopathic DM had skin biopsy findings consistent with DM.
Clinical data were collected as part of routine medical care. Age-appropriate cancer screening and/or computed tomography of the chest, abdomen, and pelvis was performed in all patients at least once, either at clinic presentation or during followup. All malignancies were identified with confirmation by tissue diagnosis. Patients were considered to have cancer-associated DM if they had a diagnosis (or specific signs) of a malignancy (excluding nonmelanoma skin cancer) 3 years before or after the onset of the first DM symptom (29). Patients with clinically amyopathic DM were defined as patients with the characteristic rash of DM for at least 6 months without clinical weakness attributable to inflammatory myopathy and without elevation of muscle enzymes >20% above the upper limit of normal at any time (30).
Immunoprecipitation using 35S-methionine–labeled in vitro transcription/translation (IVTT) proteins
Complementary DNAs (cDNAs) encoding full-length NXP-2 and MDA-5 were purchased from OriGene, and their sequence was verified before use. 35S-methionine–labeled proteins were generated from these cDNAs by IVTT reactions, in accordance with the protocol of the manufacturer (Promega). Immunoprecipitations using these products were performed as described previously (11), and the immunoprecipitates were electrophoresed on 10% SDS-PAGE gels and visualized by fluorography.
Immunoprecipitation/blot assay to detect anti–TIF-1γ antibodies
Complementary DNA encoding full-length FLAG-tagged TIF-1γ (OriGene) was sequence-verified before use, then transiently transfected into HeLa cells using Lipofectamine 2000 (Invitrogen), in accordance with the manufacturer’s protocol. After 24 hours, the cells were lysed with buffer A (1% Nonidet P40 [NP40], 20 mM Tris [pH 7.4], 150 mM NaCl, 1 mM EDTA, and a protease inhibitor cocktail). Robust TIF-1γ expression was confirmed for each lysate batch by immunoblotting gel samples made from equal protein amounts of transfected and nontransfected lysates. The immunoblots were performed with monoclonal antibodies against the FLAG tag (Agilent Technologies) or against TIF-1γ (Novus Biologicals). Levels of TIF-1γ in the transiently transfected lysates were reproducibly ~35–60-fold above those in their nontransfected counterparts.
For use in immunoprecipitations, TIF-1γ –transfected lysates were precleared by incubation with immobilized protein A–agarose (Thermo Scientific) for 20 minutes at 4°C. Immunoprecipitations were then performed as follows. Fifteen-microgram amounts of lysate (~15 μl) were brought to 1 ml by adding buffer A. One microliter of patient serum was added to each, and the tubes were rotated for 1 hour at 4°C. Twenty-five microliters of immobilized protein A–agarose was subsequently added to each tube (20 minutes at 4°C), after which the immunoprecipitates were washed and electrophoresed on 10% SDS-PAGE gels. The gels were transferred to nitrocellulose membrane and immunoblotted with anti–TIF-1γ monoclonal antibody to detect the immunoprecipitates.
Immunoprecipitations from radiolabeled HeLa cell extracts
HeLa cells were radiolabeled for 2 hours with 35S-methionine/cysteine. In some experiments, the cells were treated with 1,000 units/ml interferon-α (IFNα; Sigma) for 22 hours before starting the labeling; in these cases, IFNα was also added to the radiolabeling incubations. The cells were lysed in radioimmunoprecipitation assay buffer (50 mM Tris [pH 7.4], 150 mM NaCl, 5 mM EDTA, 0.5% NP40, 0.5% deoxycholate, 0.1% SDS) and precleared as above. Immunoprecipitations were performed by adding 1 μl of patient serum to the lysate (1 hour at 4°C), followed by protein A–agarose (20 minutes at 4°C), extensive washing, electrophoresis on 10% SDS-PAGE gels, and visualization of radiolabeled immunoprecipitates by fluorography.
Assays for other autoantibodies
Anti–Jo-1 and anti–Ro 52 antibodies were assayed by enzyme-linked immunosorbent assay (Inova Diagnostics). Anti–Mi-2 and anti–SUMO-activating enzyme (anti-SAE1/2) antibodies were determined by immunoprecipitation using 35S-methionine–labeled proteins generated by IVTT from the appropriate cDNAs.
Statistical analysis
Patient characteristics between the Stanford University and Johns Hopkins University cohorts and among the anti–NXP-2–positive, anti–TIF-1γ–positive, and anti–NXP-2–negative/anti–TIF-1γ–negative groups were compared using chi-square tests for categorical variables and Student’s t-test or analysis of variance for continuous variables. Univariate and multivariate logistic regression models were used to assess the odds of cancer associated with anti–NXP-2 and/or anti–TIF-1γ positivity, with the multivariate model correcting for variables that showed a trend toward association with cancer in univariate analysis (P < 0.15). We repeated the analyses stratified by sex and age >60 years. Sensitivity analyses were also performed to assess the odds of cancer associated with anti–NXP-2 positivity alone or anti–TIF-1γ positivity alone compared with patients negative for both antibodies, when excluding patients who were positive for the alternative autoantibody. Odds ratios (ORs) are provided with 95% confidence intervals (95% CIs).
RESULTS
Patient populations
The two DM patient cohorts consisted of 111 patients from Stanford University and 102 patients from Johns Hopkins University (Table 1). Both cohorts were similar in terms of sex breakdown and age at diagnosis. There were significantly more Asian patients in the Stanford University cohort and more African American patients in the Johns Hopkins University cohort. The prevalence of clinically amyopathic DM was 20.7% (23 of 111 patients) in the Stanford University cohort and 14.7% (15 of 102 patients) in the Johns Hopkins University cohort. In all, 71% had a followup time of at least 3 years following the date of diagnosis, with a median duration of followup of 4.1 years.
Table 1.
Characteristics of the patients in the 2 cohorts studied*
| Stanford University (n = 111) | Johns Hopkins University (n = 102) | Total (n = 213) | |
|---|---|---|---|
| Sex | |||
| Female | 78 (70) | 75 (74) | 153 (72) |
| Male | 33 (30) | 27 (27) | 60 (28) |
| Ethnicity | |||
| White (only) | 73 (66) | 82 (80) | 155 (73) |
| White (Latino) | 11 (9.9) | 0 (0) | 11 (5.2) |
| Asian | 20 (18) | 3 (3.0) | 23 (11) |
| African American | 6 (5.4) | 12 (12) | 18 (8.5) |
| Other/missing | 1 (0.9) | 5 (4.9) | 6 (2.8) |
| Age at diagnosis, years | |||
| Median (range) | 47.1 (4.6–86.9) | 46.9 (10.8–80.1) | 47.0 (4.6–86.9) |
| Mean ± SD | 48.1 ± 16 | 47.8 ± 15 | 48.0 ± 16 |
| Followup, years | |||
| Median (range) | 4.3 (0.2–38.4) | 4.0 (0.3–29.4) | 4.1 (0.2–38.4) |
| Mean ± SD | 5.6 ± 5.4 | 4.6 ± 3.4 | 5.2 ± 4.6 |
| Followup ≥3 years | 76 (68) | 74 (73) | 151 (71) |
| Clinically amyopathic disease | 23 (21) | 15 (15) | 38 (18) |
Except where indicated otherwise, values are the number (%) of patients. There were no significant differences between the groups except for ethnicity (P < 0.0001).
Novel, sensitive, and specific methods to assay for antibodies against TIF-1γ and NXP-2
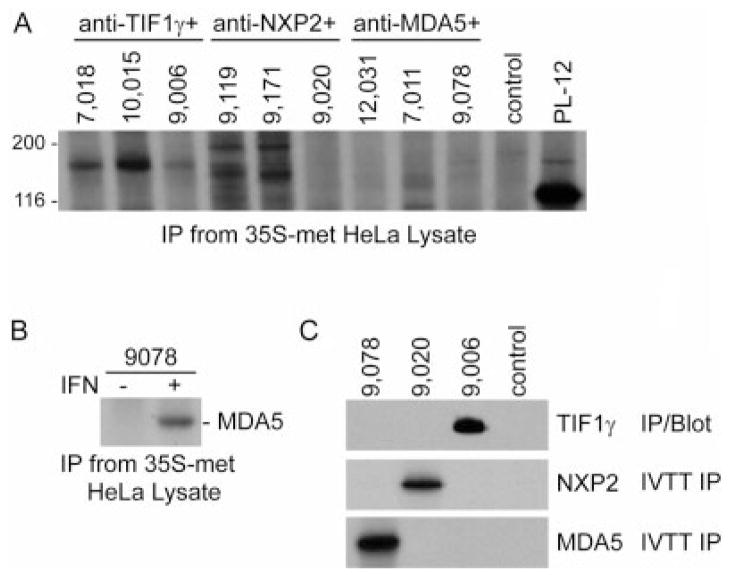
To date, the most commonly used assays to detect anti–TIF-1γ, anti–NXP-2, and anti–MDA-5 antibodies have entailed immunoprecipitation from radiolabeled cell lysates. Detecting and accurately distinguishing these different autoantigens in the resulting immunoprecipitates is challenging because the proteins have similar sizes, and some of these antigens are expressed at low levels. Immunoprecipitation data performed using 3 sera recognizing each of these specificities are demonstrated in Figure 1A. Anti–TIF-1γ–positive sera recognized a 155-kd band and anti–NXP-2–positive sera recognized a 140-kd protein (2 sera also recognized an antigen of ~190 kd), while detection of MDA-5 (an IFN-inducible protein) by this assay was very suboptimal (Figure 1A). Indeed, when patient sera known to be anti–MDA-5 antibody positive by IVTT immunoprecipitation (represented by no. 9,078 in Figure 1) were used to immunoprecipitate radiolabeled lysates made from HeLa cells incubated in the absence or presence of IFNα, MDA-5 was only immunoprecipitated from the IFN-induced cell extracts. This is consistent with the ~20-fold induction of MDA-5 levels in HeLa cells by IFN treatment, as quantitated by immunoblotting (not shown).
Figure 1.
New assays to detect antibodies against transcription intermediary factor 1γ (TIF-1γ), nuclear matrix protein NXP-2, and melanoma differentiation–associated protein 5 (MDA-5). A, Immunoprecipitations (IP) were performed using 35S-methionine (35S-met)–labeled HeLa cell lysate with patient sera known to have antibodies against TIF-1γ, NXP-2, or MDA-5, as indicated (3 sera for each). A control serum and an alanyl–transfer RNA synthetase (PL-12)–positive reference serum were also included. B, Immunoprecipitations were performed with an anti–MDA-5 antibody–positive serum using 35S-methionine–labeled HeLa cell lysate generated from cells incubated in the absence or presence of interferon (IFN) for 24 hours. C, Three sera from patients with dermatomyositis and 1 control serum were tested for antibodies by immunoprecipitation using 35S-methionine–labeled MDA-5 or 35S-methionine–labeled NXP-2 (in vitro transcription/translation immunoprecipitation [IVTT IP]), or by immunoprecipitation from TIF-1γ–transfected lysates followed by immunoblotting with anti–TIF-1γ monoclonal antibody (IP/Blot). These assays show that serum 9,078 is anti–MDA-5 positive, serum 9,020 is anti–NXP-2 positive, and serum 9,006 is anti–TIF-1γ positive.
We next established and carefully optimized and validated assays that specifically detect the 3 similarly sized autoantigens. To detect anti–MDA-5 and anti–NXP-2 antibodies, immunoprecipitations were performed using 35S-methionine–labeled proteins generated by IVTT from the appropriate sequence-verified cDNAs. The products were confirmed by immunoprecipitation using a rabbit polyclonal anti–MDA-5 antibody (American Research Products) and a mouse monoclonal anti–NXP-2 antibody (Novus Biologicals). Neither of these IVTT products was immunoprecipitated using normal control sera (0 of 20 for NXP-2, 0 of 32 for MDA-5).
Since IVTT of TIF-1γ was inconsistent, we established the following assay that does not use IVTT product to detect this specificity. Anti–TIF-1γ antibodies were determined by immunoprecipitation using lysates made from HeLa cells transiently transfected with FLAG-tagged TIF-1γ, followed by detection with immunoblotting using an anti–TIF-1γ monoclonal antibody. Identical data were obtained using an anti-FLAG antibody to detect the immunoprecipitates. TIF-1γ was transiently expressed at levels 35–60-fold above those in nontransfected cells, making this a robust source of antigen for these assays. Using this immunoprecipitation/blot method, anti–TIF-1γ antibodies were detected in sera from 0 of 18 pancreatic cancer patients, 0 of 18 patients with anti–3-hydroxy-3-methylglutaryl-coenzyme A reductase antibody–associated auto-immune necrotizing myopathy, 0 of 36 patients with lupus and cancer, and 0 of 18 normal controls. The utility of these 3 assays is illustrated in Figure 1C, in which a serum with each of these specificities and a normal control is tested with each assay type. The data show that the antibodies are readily distinguishable, in contrast to data obtained from the same sera using immunoprecipitation from 35S-methionine–labeled cell lysates (Figure 1A). These assays were then used to test all sera from the 2 cohorts.
Of note, anti–MDA-5 antibody results from these cohorts have recently been described (11,15). As anti–MDA-5 antibodies have not been found to be associated with cancer and DM in our cohorts, data with the anti–NXP-2 and anti–TIF-1γ antibody specificities will be the exclusive focus for the remainder of this study.
Antibodies and cancer
Antibodies to NXP-2 were seen in 13.5% and 21.6% of patients in the Stanford University and Johns Hopkins University cohorts, respectively (17.4% overall) (Table 2). Antibodies to TIF-1γ were seen in 37.8% and 39.2% of patients in the Stanford University and Johns Hopkins University cohorts, respectively (38.4% overall). Two patients, neither with cancer, had antibodies to both NXP-2 and TIF-1γ. The frequencies of antibodies to either NXP-2 or TIF-1γ did not differ significantly between the 2 cohorts.
Table 2.
Frequency of cancers and autoantibodies*
| Stanford University (n = 111) | Johns Hopkins University (n = 102) | Total (n = 213) | |
|---|---|---|---|
| Cancer | 17 (15.3) | 12 (11.8) | 29 (13.6) |
| Anti–NXP-2 | |||
| With cancer | 5 | 4 | 9 |
| Without cancer | 10 | 18 | 28 |
| Total | 15 (13.5) | 22 (21.6) | 37 (17.4) |
| Anti–TIF-1γ | |||
| With cancer | 8 | 7 | 15 |
| Without cancer | 34 | 33 | 67 |
| Total | 42 (37.8) | 40 (39.2) | 82 (38.4) |
| Other autoantibodies | |||
| With cancer | 4† | 1† | 5‡ |
| Without cancer | 51 | 40 | 91 |
| Total | 55 (49.5) | 41 (40.2) | 96 (45.1) |
Values are the number or number (%) of patients. There were no significant differences between the cohorts. Anti–TIF-1γ = anti–transcription intermediary factor 1γ.
P < 0.05 versus no cancer.
P < 0.005 versus no cancer.
Cancer-associated DM was present in 29 of 213 patients (13.6%). Cancers included breast, colorectal, renal cell carcinoma, thyroid (papillary) carcinoma, lymphoma, myelodysplastic syndrome, chronic lymphocytic leukemia, endometrial, lung, nasopharyngeal carcinoma, neuroendocrine, ovarian, prostate, thymic carcinoma, carcinoma (undifferentiated), urothelial, and Waldenström’s macroglobulinemia. A detailed list of the cancer types and associated autoantibodies is available from the corresponding author. Of these 29 cancer patients, 9 had anti–NXP-2 antibodies and 15 had anti–TIF-1γ antibodies (Table 2). Of the remaining 5 patients with cancer, 1 had antibodies to MDA-5, and the other 4 patients tested negative for all myositis-specific autoantibodies assayed (including anti–NXP-2, anti–TIF-1γ, anti–MDA-5, anti-SAE1/2, anti–Jo-1, anti–Mi-2, and anti–Ro 52). Thus, 24 of 29 patients with cancer-associated DM (83%) had antibodies to either NXP-2 (31%) or TIF-1γ (52%). Interestingly, the frequency of cancer in DM patients with antibodies to TIF-1γ (15 of 82 [18%]) or NXP-2 (9 of 37 [24%]) was increased compared to the frequency of cancer in DM patients without these antibodies (5 of 96 [5%]), and the absence of antibodies to either NXP-2 or TIF-1γ was strongly protective against cancer (P < 0.005 for the entire cohort) (Table 2).
Stratifying cancer risk with antibodies and age
We next examined the relationship between demographic features, autoantibodies, and cancer. On univariate analysis, we found that male sex, older age, and the presence of antibodies to NXP-2 were significantly associated with the presence of cancer (Table 3), while race and having clinically amyopathic DM were not associated with cancer (data not shown). Patients with antibodies to NXP-2 had an increased risk of cancer (OR 2.5 [95% CI 1.0–6.1], P = 0.04). Of note, while reactivity to TIF-1γ had an OR of cancer of 1.9, this was not statistically significant. We realized that any association between anti–TIF-1γ antibodies and cancer was potentially being masked by inclusion of the anti–NXP-2–positive patients (who have a higher risk of cancer) in the anti–TIF-1γ–negative comparator group. When the anti–NXP-2–positive patients were excluded in the univariate analysis, antibodies to TIF-1γ were significantly associated with cancer (OR 4.2 [95% CI 1.5–12.1], P = 0.008). We also performed a test dividing the entire population into 2 groups (those with and those without anti–TIF-1γ or anti–NXP-2 antibodies), and we found that the antibody-positive group was highly associated with cancer (OR 4.7 [95% CI 1.7–12.8], P = 0.003) (Table 3).
Table 3.
Risk factors for cancer (univariate analysis)*
| OR (95% CI) | P | |
|---|---|---|
| Sex | ||
| Female | Referent | |
| Male | 3.3 (1.5–7.4) | 0.003 |
| Age at diagnosis, per year | 1.06 (1.03–1.1) | <0.0001 |
| Anti–NXP-2 | ||
| No | Referent | |
| Yes | 2.5 (1.0–6.1) | 0.042 |
| Anti–TIF-1γ | ||
| No | Referent | |
| Yes | 1.9 (0.9–4.1) | 0.12 |
| Anti–NXP-2 or anti–TIF-1γ | ||
| No | Referent | |
| Yes | 4.7 (1.7–12.8) | 0.0026 |
OR = odds ratio; 95% CI = 95% confidence interval (see Table 2 for other definitions).
We next analyzed the relationship between age, anti–TIF-1γ/anti–NXP-2 status, and cancer. We considered 3 antibody groups–anti–NXP-2, anti–TIF-1γ, and neither antibody. The age range of 40–50 years contained the highest number of patients across the 3 groups (Figure 2A). Antibodies to TIF-1γ and NXP-2 were found across the age spectrum, and the mean age did not differ significantly between antibody groups. However, a higher proportion of patients with these antibodies were over the age of 60 years as compared to the antibody-negative group (35% of patients with either antibody were >60 years old, compared to 19% for the antibody-negative group; P = 0.01). Cancer prevalence increased with age, with an inflection point evident at ~60 years of age, regardless of antibody status (Figure 2B).
Figure 2.
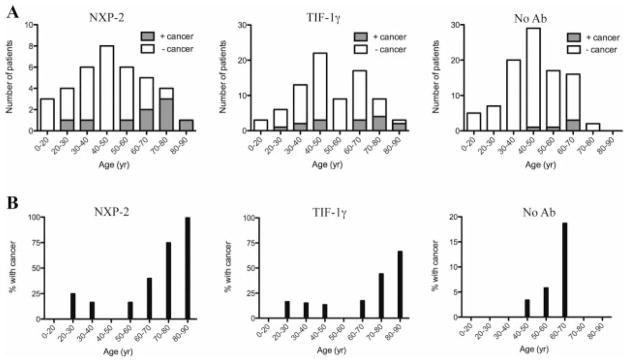
Age and cancer distribution as a function of autoantibody status. A, Total numbers of patients in each age group for each antibody (Ab) class. Shaded portions of the bars represent actual numbers of patients with cancer-associated dermatomyositis (DM). B, Percentage of patients in each age group with cancer-associated DM. See Figure 1 for other definitions.
Since age is the strongest predictor of malignancy, we tested the hypothesis that the association of antibodies with cancer was indirect and a result of their enrichment in older populations. We performed multivariate analysis and found that, when adjusted for age and sex, having antibodies to either TIF-1γ or NXP-2 was still significantly associated with an increased risk of cancer (OR 3.8 [95% CI 1.3–10.8], P = 0.01) (Table 4). When each antibody was considered separately in this multivariate analysis, anti–NXP-2 antibodies showed a trend toward association with cancer, while antibodies to TIF-1γ were not significantly associated with cancer (Table 4). Although the numbers of patients in the older age groups were small, the frequency of cancer in patients over the age of 60 years was 55% for anti–NXP-2–positive patients, 31% for anti–TIF-1γ–positive patients, and 17% for patients without these antibodies. For patients younger than 60 years, the frequency of cancer was 12% for anti–NXP-2–positive patients, 11% for anti–TIF-1γ–positive patients, and 3% for patients without these antibodies.
Table 4.
Age, sex, and antibodies as risk factors for cancer (multivariate analysis)*
| Covariates in model | OR (95% CI) | P |
|---|---|---|
| Anti–NXP-2, age, sex | ||
| Age, per year | 1.06 (1.03–1.09) | 0.0002 |
| Male | 2.7 (1.1–6.4) | 0.025 |
| Anti–NXP-2 | 2.5 (0.9–6.7) | 0.069 |
| Anti–TIF-1γ, age, sex | ||
| Age, per year | 1.06 (1.02–1.09) | 0.0005 |
| Male | 3.0 (1.3–7.0) | 0.012 |
| Anti–TIF-1γ | 1.6 (0.7–3.8) | 0.29 |
| Anti–NXP-2 or anti–TIF-1γ, age, sex | ||
| Age, per year | 1.05 (1.02–1.08) | 0.0013 |
| Male | 2.9 (1.2–6.9) | 0.018 |
| Anti–NXP-2 or anti–TIF-1γ | 3.8 (1.3–10.8) | 0.013 |
| Age, anti–NXP-2 (males only) | ||
| Anti–NXP-2 | 5.8 (1.4–24.7) | 0.018 |
| Age, per year | 1.05 (1.01–1.10) | 0.018 |
| Age, anti–NXP-2 (females only) | ||
| Anti–NXP-2 | 1.1 (0.2–5.4) | 0.95 |
| Age, per year | 1.06 (1.02–1.11) | 0.006 |
OR = odds ratio; 95% CI = 95% confidence interval (see Table 2 for other definitions).
We also evaluated the interaction between sex, antibody positivity, and cancer. Cancer patients in the anti–NXP-2–positive group were mostly male (78%), despite the fact that, overall, anti–NXP-2–positive patients had a sex breakdown similar to that in the rest of the cohort (35% male versus 27% male, respectively; P = 0.32). Conversely, there was no significant difference in sex distribution between cancer-positive patients (33% male) and cancer-negative patients (24% male) among those with antibodies to TIF-1γ. We tested this formally by multivariate analysis stratifying by sex, and we confirmed that antibodies to NXP-2 were associated with an increased risk of cancer only in males (OR 5.8 [95% CI 1.4–24.7], P = 0.02) (Table 4).
DISCUSSION
Several gaps exist in the understanding of the association of autoantibodies and cancer in patients with DM, including whether antibodies to NXP-2 are associated with malignancy, how frequently anti–TIF-1γ antibodies are found in DM with associated cancers, and whether these 2 antibodies might be of clinical use in DM. Our objectives in this study were to fill these gaps by using highly specific assays for these antibodies in a large cohort of DM patients. Maximizing assay specificity was crucial, since at least 4 known autoantigens (NXP-2, TIF-1γ, MDA-5, and TIF-1α) migrate similarly on gel electrophoresis and cannot be unambiguously identified using immunoprecipitation assays from crude lysates.
Our study demonstrated that antibodies to TIF-1γ and NXP-2 are very frequent in DM, occurring in 55% of patients in the combined cohorts (17% for anti–NXP-2 and 38% for anti–TIF-1γ). The frequency of 38% for anti–TIF-1γ antibodies in DM patients was higher than that defined in previous studies (of size >50 patients), which ranged from 7% to 24% (4,13,16,17). This likely reflects differences in the assay, which uses human sera to immunoprecipitate TIF-1γ from transfected cell lysates expressing TIF-1γ at levels 35–60-fold higher than those in nontransfected cells. The assay is highly specific, and none of 90 control sera (disease and healthy controls) recognized TIF-1γ in these transfected lysates (data not shown). For anti–NXP-2 antibodies, we found an overall frequency of 17%, with some variation between our 2 cohorts (14% in the Stanford University cohort and 22% in the Johns Hopkins University cohort). The 3 prior studies evaluating anti–NXP-2 in adult DM patients have noted varying frequencies of 1.6%, 6%, and 30% (22–25).
Interestingly, the anti–TIF-1γ and anti–NXP-2 antibody groups are almost entirely nonoverlapping, with only 2 patients having antibodies to both proteins. Additionally, for a subset of these antibody-positive patients who were analyzed, 84% (47 of 56) did not have antibodies against the other DM-specific or myositis-specific antigens, including Mi-2, SAE1/2, MDA-5, and transfer RNA synthetases (data not shown for this subset analysis). Thus, these assays identify a large subset of DM patients most of whom would have been otherwise “antibody negative.” Interestingly, the proportion of elderly patients with anti–TIF-1γ or anti–NXP-2 antibodies is higher than that seen in the antibody-negative group, although this does not appear to account for their association with cancer. The significance of this differential skewing of age as a function of the different autoantibody groups is unclear, as the numbers are modest in this age range.
A relatively modest percentage (14%) of patients in our cohort had cancer-associated DM (Table 2). These patients were defined using relatively standard criteria of a cancer diagnosis within 3 years of DM diagnosis (29). One limitation of our study is that almost 30% of patients had followup of <3 years (Table 1), and thus, we might not have detected all of the patients with cancer-associated DM. However, in this group of patients with followup of <3 years, we found only 18 (8%) at reasonable risk of developing cancer (as defined by having antibodies to either NXP-2 or TIF-1γ), and those patients had a median followup of 24 months. Thus, we think it is unlikely that we missed a significant number of patients with cancer-associated DM due to insufficient time for followup.
Our studies revealed a robust association of antibodies to NXP-2 with cancer, with an unexpected association with male sex. Although a prior Japanese study identifying 7 patients with anti–NXP-2 antibodies suggested an association with cancer, it was insufficiently powered to definitively demonstrate such an association (24). It is interesting to note that the population in the Japanese study was >50% male, and 6 of 7 of the anti–NXP-2–positive patients were male, which may have allowed preliminary detection of an association between anti–NXP-2 and malignancy. A recent evaluation of an Italian DM cohort did not find an association with cancer (23,31), although the number of anti–NXP-2–positive DM patients was small (8 compared to 37 in one study). That study likely was not powered to detect a cancer association, given the low number of anti–NXP-2–positive male patients. The observation that both age and sex influence association with cancer highlights the importance of studying larger cohorts of patients to accurately delineate these relationships.
The reason for the striking male association of anti–NXP-2 antibodies with cancer in DM is unclear. It cannot be simply explained by an association with male-only tumors (e.g., prostate), as the cancers included hematologic, thyroid, and colon tumors (further information is available from the corresponding author). In addition, our study findings support prior suggestions that males with DM are at a relatively higher risk for cancer (3). Confirming these findings in additional cohorts will be important.
Although our data support a possible association between anti–TIF-1γ antibodies and cancer, this association did not reach statistical significance. This is largely because the prevalence of cancer in our anti–TIF-1γ–positive group was <20%, while in previous studies, it varied between 50% and 100% (4,13,16–18). Although our assay is highly specific, it is also sensitive, identifying more anti–TIF-1γ–positive patients than prior assays (possibly due to the 35–60-fold overexpression of TIF-1γ). It is possible that the assay identifies lower positive titers that are not associated with cancer. However, we did not find any association between cancer risk and anti–TIF-1γ antibody titer, with multiple lower-titer–positive patients in the cancer group (data not shown). It is more likely that the failure to demonstrate a statistically significant relationship between cancer and anti–TIF-1γ antibodies reflects a Type II error (e.g., false-negative association).
Based on the frequency of cancer and anti–TIF-1γ antibodies, we estimate that we would need to study an additional 92 patients to demonstrate this effect with a P value less than 0.05. It is also possible that the differences from previous studies could be explained by a difference in the frequency of anti–TIF-1γ antibodies in different ethnic groups. Regardless of this, our data clearly demonstrate that anti–TIF-1γ antibodies are the most prevalent DM-specific autoantibodies described to date, at least in 2 US cohorts. These tests are thus sensitive, but not specific, markers of cancer-associated DM. Given this, they are by themselves of limited clinical utility if positive, although a patient negative for these antibodies appears to be at lower risk of cancer (Table 2).
When each antibody was analyzed separately in a multivariate analysis adjusting for age and sex, neither antibody was found to be significantly associated with cancer (Table 4). However, in this analysis, anti–NXP-2 antibodies showed a clear trend toward an increased cancer risk (Table 4), and in fact, this was not appreciably different from what was seen in univariate analysis (Table 3). Anti–TIF-1γ antibodies failed to show significant association in the multivariate analysis. In fact, the association with cancer was weakened significantly in the multivariate analysis, and this was largely accounted for by inclusion of age in the multivariate model (date not shown). This is consistent with the notion that much of the risk associated with anti–TIF-1γ antibodies was due to specific enrichment of patients over the age of 70 years in this antibody group, most of whom had cancer (Figure 2). The observations that anti–TIF-1γ and anti–NXP-2 antibodies were expressed in a nonoverlapping way in 98.3% of patients who had these antibodies, that their frequency was higher in older DM patients, and that together, these antibodies identified 83% of patients with DM and cancer (while present in 51% of DM patients without cancer) suggested that they might be grouped together for further analysis. Indeed, when these antibodies were grouped together (i.e., positivity for either anti–TIF-1γ or anti–NXP-2), we found that this was associated with a significant increase in cancer risk, even when adjusting for age and sex (Tables 3 and 4).
The relationship of cancer and age in DM has previously been observed (see ref. 32). Our studies demonstrate that there is a striking inflection point in the curve of the frequency of cancer with age ~60 years and that determining the presence or absence of anti–NXP-2 and anti–TIF-1γ antibodies more precisely identifies the group with the highest cancer prevalence. In patients younger than 60 years without anti–TIF-1γ or anti–NXP-2 antibodies, the frequency of cancer is very low (2.6%). The cancer frequency is higher (11%) in patients younger than 60 years with either anti–TIF-1γ or anti–NXP-2 antibodies. Six of 11 patients over the age of 60 years with anti–NXP-2 antibodies (55%) had cancer. This frequency was 9 of 29 in patients with anti–TIF-1γ antibodies (31%), and only 3 of 18 in patients without either of these antibodies (17%).
One limitation of our study is that it is subject to referral bias, as it is not population based. For example, given that the Stanford University cohort is based in a dermatology center, one might expect a larger percentage of patients with clinically amyopathic disease in that cohort—although this was true, the percentage of patients with clinically amyopathic disease was not vastly different between the Stanford University and Johns Hopkins University cohorts (21% versus 15%). These numbers are consistent with the only population-based study to examine this issue, which estimated that 21% of DM patients in a midwestern county in the US had clinically amyopathic disease (33). Despite this, larger, prospective studies in other populations will be important in the future.
Acknowledgments
Dr. Chung’s work was supported by the Scleroderma Research Foundation. Dr. Christopher-Stine’s work was supported by the Huayi and Siuling Zhang Discovery Fund. Drs. Mammen and Rosen’s work was supported by the NIH (grants K08-AR-054783 and R37-DE-12354, respectively). Dr. Casciola-Rosen’s work was supported by the NIH (grant R01-AR-44684) and the Dorothy and Donald Stabler Foundation. The Johns Hopkins Rheumatic Diseases Research Core Center, where the assays were performed, is supported by the NIH (grant P30-AR-053503).
We thank Bharathi Lingala (Stanford University Biostatistics) for help with the initial statistical analyses and Jessie Werner (Johns Hopkins University) for assistance with the data collection.
Footnotes
Drs. Christopher-Stine and Mammen have patented and licensed a test for anti–3-hydroxy-3-methylglutaryl-coenzyme A reductase antibodies. Dr. Mammen has received consulting fees from aTyr Pharma and Biogen (less than $10,000 each).
AUTHOR CONTRIBUTIONS
All authors were involved in drafting the article or revising it critically for important intellectual content, and all authors approved the final version to be published. Dr. Fiorentino had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study conception and design. Fiorentino, Chung, Rosen, Casciola-Rosen.
Acquisition of data. Fiorentino, Chung, Christopher-Stine, Zaba, Mammen, Casciola-Rosen.
Analysis and interpretation of data. Fiorentino, Chung, Zaba, Li, Rosen, Casciola-Rosen.
References
- 1.Betteridge ZE, Gunawardena H, McHugh NJ. Novel autoantibodies and clinical phenotypes in adult and juvenile myositis. Arthritis Res Ther. 2011;13:209. doi: 10.1186/ar3275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Casciola-Rosen L, Mammen AL. Myositis autoantibodies. Curr Opin Rheumatol. 2012;24:602–8. doi: 10.1097/BOR.0b013e328358bd85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Madan V, Chinoy H, Griffiths CE, Cooper RG. Defining cancer risk in dermatomyositis: part I. Clin Exp Dermatol. 2009;34:451–5. doi: 10.1111/j.1365-2230.2009.03216.x. [DOI] [PubMed] [Google Scholar]
- 4.Fujimoto M, Hamaguchi Y, Kaji K, Matsushita T, Ichimura Y, Kodera M, et al. Myositis-specific anti-155/140 autoantibodies target transcription intermediary factor 1 family proteins. Arthritis Rheum. 2012;64:513–22. doi: 10.1002/art.33403. [DOI] [PubMed] [Google Scholar]
- 5.Gunawardena H, Wedderburn LR, Chinoy H, Betteridge ZE, North J, Ollier WE, et al. for the Juvenile Dermatomyositis Research Group, UK and Ireland. Autoantibodies to a 140-kd protein in juvenile dermatomyositis are associated with calcinosis. Arthritis Rheum. 2009;60:1807–14. doi: 10.1002/art.24547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nakashima R, Imura Y, Kobayashi S, Yukawa N, Yoshifuji H, Nojima T, et al. The RIG-I-like receptor IFIH1/MDA5 is a dermatomyositis-specific autoantigen identified by the anti-CADM-140 antibody. Rheumatology (Oxford) 2010;49:433–40. doi: 10.1093/rheumatology/kep375. [DOI] [PubMed] [Google Scholar]
- 7.Sato S, Hoshino K, Satoh T, Fujita T, Kawakami Y, Fujita T, et al. RNA helicase encoded by melanoma differentiation–associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum. 2009;60:2193–200. doi: 10.1002/art.24621. [DOI] [PubMed] [Google Scholar]
- 8.Targoff IN, Trieu EP, Levy-Neto M, Prasertsuntarasai T, Miller FW. Autoantibodies to transcriptional intermediary factor 1-gamma (TIF1-g) in dermatomyositis [abstract] Arthritis Rheum. 2006;54 (Suppl):S518. [Google Scholar]
- 9.Targoff IN, Trieu EP, Levy-Neto M, Fertig N, Oddis CV. Sera with autoantibodies to the MJ antigen react with NXP2 [abstract] Arthritis Rheum. 2007;56 (Suppl):S787. [Google Scholar]
- 10.Chen F, Wang D, Shu X, Nakashima R, Wang G. Anti-MDA5 antibody is associated with A/SIP and decreased T cells in peripheral blood and predicts poor prognosis of ILD in Chinese patients with dermatomyositis. Rheumatol Int. 2012;32:3909–15. doi: 10.1007/s00296-011-2323-y. [DOI] [PubMed] [Google Scholar]
- 11.Fiorentino D, Chung L, Zwerner J, Rosen A, Casciola-Rosen L. The mucocutaneous and systemic phenotype of dermatomyositis patients with antibodies to MDA5 (CADM-140): a retrospective study. J Am Acad Dermatol. 2011;65:25–34. doi: 10.1016/j.jaad.2010.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hamaguchi Y, Kuwana M, Hoshino K, Hasegawa M, Kaji K, Matsushita T, et al. Clinical correlations with dermatomyositis-specific autoantibodies in adult Japanese patients with dermatomyositis: a multicenter cross-sectional study. Arch Dermatol. 2011;147:391–8. doi: 10.1001/archdermatol.2011.52. [DOI] [PubMed] [Google Scholar]
- 13.Hoshino K, Muro Y, Sugiura K, Tomita Y, Nakashima R, Mimori T. Anti-MDA5 and anti-TIF1-gamma antibodies have clinical significance for patients with dermatomyositis. Rheumatology (Oxford) 2010;49:1726–33. doi: 10.1093/rheumatology/keq153. [DOI] [PubMed] [Google Scholar]
- 14.Sato S, Hirakata M, Kuwana M, Suwa A, Inada S, Mimori T, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum. 2005;52:1571–6. doi: 10.1002/art.21023. [DOI] [PubMed] [Google Scholar]
- 15.Hall JC, Casciola-Rosen L, Samedy LA, Werner J, Owoyemi K, Danoff SK, et al. Anti-MDA5-associated dermatomyositis: expanding the clinical spectrum. Arthritis Care Res (Hoboken) 2013 doi: 10.1002/acr.21992. E-pub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Trallero-Araguas E, Rodrigo-Pendas JA, Selva-O’Callaghan A, Martinez-Gomez X, Bosch X, Labrador-Horrillo M, et al. Usefulness of anti-p155 autoantibody for diagnosing cancer-associated dermatomyositis: a systematic review and meta-analysis. Arthritis Rheum. 2012;64:523–32. doi: 10.1002/art.33379. [DOI] [PubMed] [Google Scholar]
- 17.Ikeda N, Takahashi K, Yamaguchi Y, Inasaka M, Kuwana M, Ikezawa Z. Analysis of dermatomyositis-specific autoantibodies and clinical characteristics in Japanese patients. J Dermatol. 2011;38:973–9. doi: 10.1111/j.1346-8138.2011.01262.x. [DOI] [PubMed] [Google Scholar]
- 18.Kang EH, Nakashima R, Mimori T, Kim J, Lee YJ, Lee EB, et al. Myositis autoantibodies in Korean patients with inflammatory myositis: anti-140-kDa polypeptide antibody is primarily associated with rapidly progressive interstitial lung disease independent of clinically amyopathic dermatomyositis. BMC Musculoskelet Disord. 2010;11:223. doi: 10.1186/1471-2474-11-223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chinoy H, Fertig N, Oddis CV, Ollier WE, Cooper RG. The diagnostic utility of myositis autoantibody testing for predicting the risk of cancer-associated myositis. Ann Rheum Dis. 2007;66:1345–9. doi: 10.1136/ard.2006.068502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Targoff IN, Mamyrova G, Trieu EP, Perurena O, Koneru B, O’Hanlon TP, et al. for the Childhood Myositis Heterogeneity and International Myositis Collaborative Study Groups. A novel autoantibody to a 155-kd protein is associated with dermatomyositis. Arthritis Rheum. 2006;54:3682–9. doi: 10.1002/art.22164. [DOI] [PubMed] [Google Scholar]
- 21.Gunawardena H, Wedderburn LR, North J, Betteridge Z, Dunphy J, Chinoy H, et al. for the Juvenile Dermatomyositis Research Group UK. Clinical associations of autoantibodies to a p155/140 kDa doublet protein in juvenile dermatomyositis. Rheumatology (Oxford) 2008;47:324–8. doi: 10.1093/rheumatology/kem359. [DOI] [PubMed] [Google Scholar]
- 22.Betteridge ZE, Gunawardena H, Chinoy H, Vencovsky J, Allard S, Gordon PA, et al. Autoantibodies to the p140 autoantigen NXP-2 in adult dermatomyositis [abstract] Arthritis Rheum. 2009;60 (Suppl):S304. [Google Scholar]
- 23.Ceribelli A, Fredi M, Taraborelli M, Cavazzana I, Franceschini F, Quinzanini M, et al. Anti-MJ/NXP-2 autoantibody specificity in a cohort of adult Italian patients with polymyositis/dermatomyositis. Arthritis Res Ther. 2012;14:R97. doi: 10.1186/ar3822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ichimura Y, Matsushita T, Hamaguchi Y, Kaji K, Hasegawa M, Tanino Y, et al. Anti-NXP2 autoantibodies in adult patients with idiopathic inflammatory myopathies: possible association with malignancy. Ann Rheum Dis. 2012;71:710–3. doi: 10.1136/annrheumdis-2011-200697. [DOI] [PubMed] [Google Scholar]
- 25.Ishikawa A, Muro Y, Sugiura K, Akiyama M. Development of an ELISA for detection of autoantibodies to nuclear matrix protein 2. Rheumatology (Oxford) 2012;51:1181–7. doi: 10.1093/rheumatology/kes033. [DOI] [PubMed] [Google Scholar]
- 26.Espada G, Maldonado Cocco JA, Fertig N, Oddis CV. Clinical and serologic characterization of an Argentine pediatric myositis cohort: identification of a novel autoantibody (anti-MJ) to a 142-kDa protein. J Rheumatol. 2009;36:2547–51. doi: 10.3899/jrheum.090461. [DOI] [PubMed] [Google Scholar]
- 27.Bohan A, Peter JB. Polymyositis and dermatomyositis (first of two parts) N Engl J Med. 1975;292:344–7. doi: 10.1056/NEJM197502132920706. [DOI] [PubMed] [Google Scholar]
- 28.Sontheimer RD. Dermatomyositis: an overview of recent progress with emphasis on dermatologic aspects. Dermatol Clin. 2002;20:387–408. doi: 10.1016/s0733-8635(02)00021-9. [DOI] [PubMed] [Google Scholar]
- 29.Troyanov Y, Targoff IN, Tremblay JL, Goulet JR, Raymond Y, Senecal JL. Novel classification of idiopathic inflammatory myopathies based on overlap syndrome features and autoantibodies: analysis of 100 French Canadian patients. Medicine (Baltimore) 2005;84:231–49. doi: 10.1097/01.md.0000173991.74008.b0. [DOI] [PubMed] [Google Scholar]
- 30.Gerami P, Schope JM, McDonald L, Walling HW, Sontheimer RD. A systematic review of adult-onset clinically amyopathic dermatomyositis (dermatomyositis sine myositis): a missing link within the spectrum of the idiopathic inflammatory myopathies. J Am Acad Dermatol. 2006;54:597–613. doi: 10.1016/j.jaad.2005.10.041. [DOI] [PubMed] [Google Scholar]
- 31.Ceribelli A, Fredi M, Taraborelli M, Cavazzana I, Franceschini F, Tincani A, et al. Anti-MJ/NXP-2 antibodies are the most common specificity in a cohort of adult Caucasian patients with dermatomyositis [abstract] Arthritis Rheum. 2011;63 (Suppl):S85–6. [Google Scholar]
- 32.Fiorentino D, Callen JP. Dermatomyositis. In: Williams H, Bigby M, Diepgen T, Herxheimer A, Naldi L, Rzany B, editors. Evidence-based dermatology. Oxford: Blackwell Publishing; 2008. pp. 559–72. [Google Scholar]
- 33.Bendewald MJ, Wetter DA, Li X, Davis MD. Incidence of dermatomyositis and clinically amyopathic dermatomyositis: a population-based study in Olmsted County, Minnesota. Arch Dermatol. 2010;146:26–30. doi: 10.1001/archdermatol.2009.328. [DOI] [PMC free article] [PubMed] [Google Scholar]