Abstract
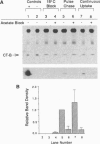
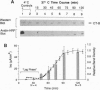
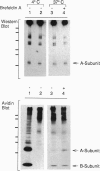
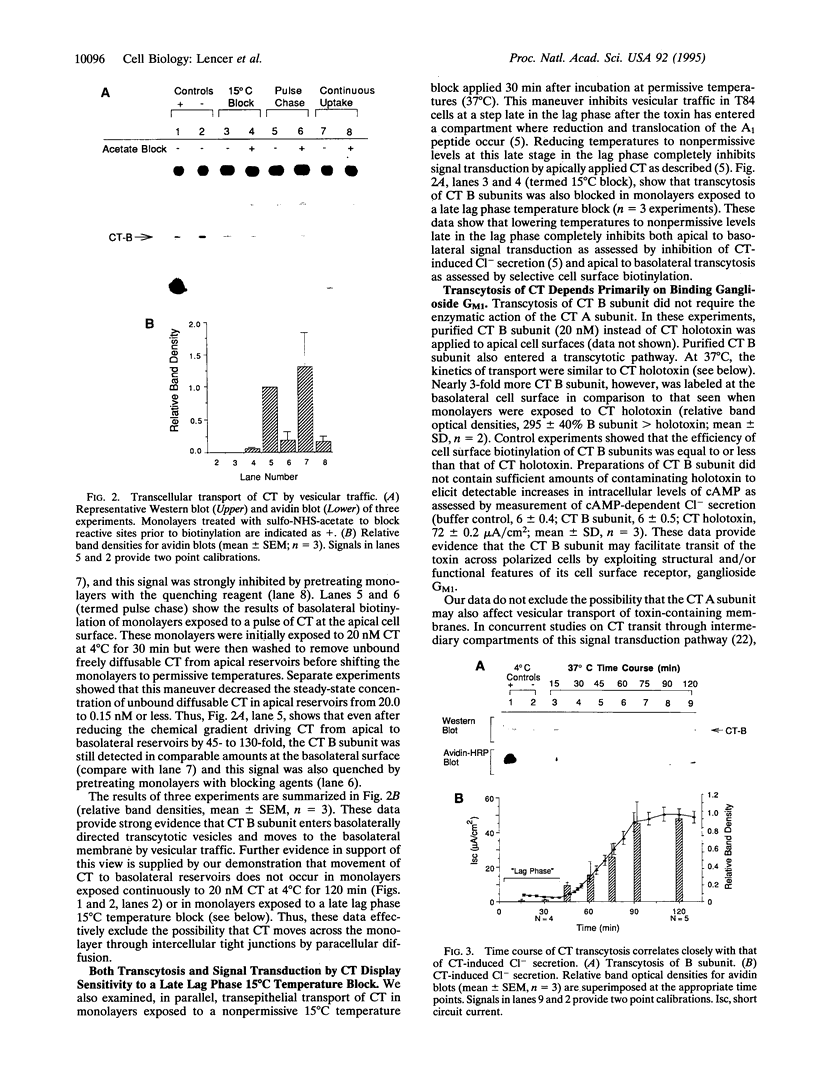
Cholera toxin (CT) elicits a massive secretory response from intestinal epithelia by binding apical receptors (ganglioside GM1) and ultimately activating basolateral effectors (adenylate cyclase). The mechanism of signal transduction from apical to basolateral membrane, however, remains undefined. We have previously shown that CT action on the polarized human intestinal epithelial cell line T84 requires endocytosis and processing in multiple intracellular compartments. Our aim in the present study was to test the hypothesis that CT may actually move to its site of action on the basolateral membrane by vesicular traffic. After binding apical receptors, CT entered basolaterally directed transcytotic vesicles. Both CT B subunits and to a lesser extent CT A subunits were delivered intact to the serosal surface of the basolateral membrane. The toxin did not traverse the monolayer by diffusion through intercellular junctions. Transcytosis of CT B subunits displayed nearly identical time course and temperature dependency with that of CT-induced Cl- secretion--suggesting the two may be related. These data identify a mechanism that may explain the link between the toxin's apical receptor and basolateral effector.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barrett K. E. Positive and negative regulation of chloride secretion in T84 cells. Am J Physiol. 1993 Oct;265(4 Pt 1):C859–C868. doi: 10.1152/ajpcell.1993.265.4.C859. [DOI] [PubMed] [Google Scholar]
- Cassuto J., Jodal M., Tuttle R., Lundgren O. On the role of intramural nerves in the pathogenesis of cholera toxin-induced intestinal secretion. Scand J Gastroenterol. 1981 Apr;16(3):377–384. doi: 10.3109/00365528109181984. [DOI] [PubMed] [Google Scholar]
- Field M., Semrad C. E. Toxigenic diarrheas, congenital diarrheas, and cystic fibrosis: disorders of intestinal ion transport. Annu Rev Physiol. 1993;55:631–655. doi: 10.1146/annurev.ph.55.030193.003215. [DOI] [PubMed] [Google Scholar]
- Goins B., Freire E. Thermal stability and intersubunit interactions of cholera toxin in solution and in association with its cell-surface receptor ganglioside GM1. Biochemistry. 1988 Mar 22;27(6):2046–2052. doi: 10.1021/bi00406a035. [DOI] [PubMed] [Google Scholar]
- Holmgren J., Lycke N., Czerkinsky C. Cholera toxin and cholera B subunit as oral-mucosal adjuvant and antigen vector systems. Vaccine. 1993 Sep;11(12):1179–1184. doi: 10.1016/0264-410x(93)90039-z. [DOI] [PubMed] [Google Scholar]
- Klausner R. D., Donaldson J. G., Lippincott-Schwartz J. Brefeldin A: insights into the control of membrane traffic and organelle structure. J Cell Biol. 1992 Mar;116(5):1071–1080. doi: 10.1083/jcb.116.5.1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lencer W. I., Delp C., Neutra M. R., Madara J. L. Mechanism of cholera toxin action on a polarized human intestinal epithelial cell line: role of vesicular traffic. J Cell Biol. 1992 Jun;117(6):1197–1209. doi: 10.1083/jcb.117.6.1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lencer W. I., de Almeida J. B., Moe S., Stow J. L., Ausiello D. A., Madara J. L. Entry of cholera toxin into polarized human intestinal epithelial cells. Identification of an early brefeldin A sensitive event required for A1-peptide generation. J Clin Invest. 1993 Dec;92(6):2941–2951. doi: 10.1172/JCI116917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matter K., Whitney J. A., Yamamoto E. M., Mellman I. Common signals control low density lipoprotein receptor sorting in endosomes and the Golgi complex of MDCK cells. Cell. 1993 Sep 24;74(6):1053–1064. doi: 10.1016/0092-8674(93)90727-8. [DOI] [PubMed] [Google Scholar]
- Moss J., Vaughan M. Activation of adenylate cyclase by choleragen. Annu Rev Biochem. 1979;48:581–600. doi: 10.1146/annurev.bi.48.070179.003053. [DOI] [PubMed] [Google Scholar]
- Orlandi P. A., Curran P. K., Fishman P. H. Brefeldin A blocks the response of cultured cells to cholera toxin. Implications for intracellular trafficking in toxin action. J Biol Chem. 1993 Jun 5;268(16):12010–12016. [PubMed] [Google Scholar]
- Pelham H. R. The retention signal for soluble proteins of the endoplasmic reticulum. Trends Biochem Sci. 1990 Dec;15(12):483–486. doi: 10.1016/0968-0004(90)90303-s. [DOI] [PubMed] [Google Scholar]
- Peterson J. W., Ochoa L. G. Role of prostaglandins and cAMP in the secretory effects of cholera toxin. Science. 1989 Aug 25;245(4920):857–859. doi: 10.1126/science.2549637. [DOI] [PubMed] [Google Scholar]
- Prydz K., Hansen S. H., Sandvig K., van Deurs B. Effects of brefeldin A on endocytosis, transcytosis and transport to the Golgi complex in polarized MDCK cells. J Cell Biol. 1992 Oct;119(2):259–272. doi: 10.1083/jcb.119.2.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sargiacomo M., Lisanti M., Graeve L., Le Bivic A., Rodriguez-Boulan E. Integral and peripheral protein composition of the apical and basolateral membrane domains in MDCK cells. J Membr Biol. 1989 Mar;107(3):277–286. doi: 10.1007/BF01871942. [DOI] [PubMed] [Google Scholar]
- Sixma T. K., Pronk S. E., Kalk K. H., Wartna E. S., van Zanten B. A., Witholt B., Hol W. G. Crystal structure of a cholera toxin-related heat-labile enterotoxin from E. coli. Nature. 1991 May 30;351(6325):371–377. doi: 10.1038/351371a0. [DOI] [PubMed] [Google Scholar]
- Tomasi M., Montecucco C. Lipid insertion of cholera toxin after binding to GM1-containing liposomes. J Biol Chem. 1981 Nov 10;256(21):11177–11181. [PubMed] [Google Scholar]
- Wan J., Taub M. E., Shah D., Shen W. C. Brefeldin A enhances receptor-mediated transcytosis of transferrin in filter-grown Madin-Darby canine kidney cells. J Biol Chem. 1992 Jul 5;267(19):13446–13450. [PubMed] [Google Scholar]
- Wan X. C., Trojanowski J. Q., Gonatas J. O. Cholera toxin and wheat germ agglutinin conjugates as neuroanatomical probes: their uptake and clearance, transganglionic and retrograde transport and sensitivity. Brain Res. 1982 Jul 15;243(2):215–224. doi: 10.1016/0006-8993(82)90244-x. [DOI] [PubMed] [Google Scholar]
- Watkins P. A., Moss J., Vaughan M. ADP ribosylation of membrane proteins from human fibroblasts. Effect of prior exposure of cells to choleragen. J Biol Chem. 1981 May 25;256(10):4895–4899. [PubMed] [Google Scholar]
- Wisnieski B. J., Bramhall J. S. Photolabelling of cholera toxin subunits during membrane penetration. Nature. 1981 Jan 22;289(5795):319–321. doi: 10.1038/289319a0. [DOI] [PubMed] [Google Scholar]