Abstract
Objective:
To determine the rate of underestimation of malignancy in patients with biopsy-proven stromal fibrosis.
Methods:
Following institutional review board approval, we retrospectively reviewed the charts of patients with biopsy-proven stromal fibrosis who underwent percutaneous breast biopsy in the 5-year period between 1 January 2005 and 31 December 2009. The medical records and the histopathology in patients who underwent repeat biopsy and/or surgical excision at the site of stromal fibrosis within 2 years were reviewed. Interval stability for up to 2 years was documented in patients who did not undergo additional biopsy or surgical excision. An upgrade was defined as any patient with biopsy-proven stromal fibrosis or fibroadenoma with evidence of malignancy at the site of biopsy within 2 years.
Results:
365 cases of stromal fibrosis were identified, of which 25 (7%) were upgraded to in situ or invasive malignancy on repeat biopsy or surgical excision. 7 were upgraded to ductal carcinoma in situ and 18 were upgraded to invasive cancer. Of the upgraded cases, 8 out of 24 (32%) were considered concordant with a benign diagnosis. The false-negative rate, that is, cases of stromal fibrosis concordant with benignity, but with subsequent upgrade, comprised 2% of all cases.
Conclusion:
In biopsy-proven cases of stromal fibrosis, there is a 7% upgrade to malignancy. We recommend that all instances of stromal fibrosis with radiology–pathology discordance undergo repeat biopsy or surgical excision. Cases that demonstrate radiology–pathology concordance can be safely categorized as a Breast Imaging Reporting and Data System 3 (BI-RADS® 3) lesion with a 6-month follow-up, owing to a false-negative rate for missed cancer of 2%.
Advances in knowledge:
We now recommend that concordant cases of stromal fibrosis be categorized as BI-RADS 3 with a short-term follow-up, as this results in a missed cancer rate of 2%.
Stromal fibrosis is a histopathology diagnosis characterized by proliferation of hypocellular fibrous tissue with the obliteration or hypoplasia of mammary acini and ducts.1 Stromal fibrosis is a common finding on percutaneous breast biopsy, with an incidence ranging from 2.1% to 9.0% depending on the series.1 Stromal fibrosis has a wide spectrum of imaging appearances on mammogram, ultrasound and MRI.2,3 Although there is no generally accepted consensus on the management of stromal fibrosis, it has been suggested that the histopathology diagnosis of stromal fibrosis should be considered concordant with a benign diagnosis during radiology–pathology correlation, if accurate targeting is confirmed and in the absence of imaging features that are concerning for malignancy.4
Although the cause of stromal fibrosis has not yet been fully elucidated, it has been observed that stromal fibrosis can occur as a desmoplastic response to malignancy.5 We therefore hypothesized that a proportion of percutaneous breast biopsies that demonstrate stromal fibrosis may be owing to undersampling of an adjacent malignancy (Figure 1).
Figure 1.
Stromal fibrosis can occur as a desmoplastic reaction to an adjacent breast carcinoma.
The false-negative rate of malignancy has been investigated in other studies. Harvey et al6 reported no false-negative cases using a sample size of 38 patients and a follow-up period of 1 year. Rosen et al4 also reported no upgrades using a sample size of 80 patients and a mean follow-up period of 27 months. The series by Sklair-Levy et al1 with a sample size of 74 patients reported an incidence of 2.7% of missed cancer using a spring-loaded device. In our study, we sought to determine the upgrade rate of biopsy-proven stromal fibrosis in a 5-year retrospective review, using both spring-loaded and vacuum-assisted biopsy devices, with a larger sample size of 365 patients and a longer follow-up period of 2 years.
METHODS AND MATERIALS
Institutional ethics approval was obtained with a waiver of informed consent. The records of 9125 percutaneous breast biopsies were reviewed by a senior radiology resident. All consecutive biopsies with histopathology demonstrating stromal fibrosis as the primary histologic diagnosis from 1 January 2005 to 31 December 2009 were identified. Cases of stromal fibrosis with concurrent malignancy or high-risk lesions were excluded. Biopsies were performed under ultrasound, stereotactic or MRI guidance, either by core needle biopsy (14- or 16-G Manan™ Pro-Mag® 2.2; Manan Medical Products, Wheeling, IL) or vacuum-assisted biopsy (10–12 G; SenoRx, Tempe, AZ). A minimum of five cores per lesion with 14-G biopsy needles, seven cores with 16-G biopsy needles and four to six cores with 10–12-G vacuum-assisted biopsy needles were obtained. All ultrasound lesions were initially biopsied with a 14- or 16-G core biopsy needle, while stereotactic and MRI lesions were biopsied with 10- or 12-G vacuum-assisted biopsy needles. For stereotactic biopsies targeting calcifications, specimen radiographs were performed to confirm the presence of calcifications before completing the procedure. Seven radiologists, either with breast imaging fellowship training or a minimum of 5 years' experience, were involved in performing the biopsies and in image interpretation.
For all patients referred for a biopsy, we used the Breast Imaging Reporting and Data System (BI-RADS®) lexicon7 to establish the presence of suspicious features requiring biopsy with ultrasound, stereotactic or MRI guidance. Only BI-RADS 4 and 5 lesions were referred for biopsy.
The electronic medical records for each patient with biopsy-proven stromal fibrosis were accessed and imaging findings, BI-RADS designation, and histopathology findings from repeat biopsy and/or surgical excision within 2 years were recorded. Repeat biopsy was performed with vacuum-assisted biopsy (10–12 G; SenoRx/Bard Biopsy Systems) in all cases except two, in which a suspicious lymph node was targeted and another where repeat 14 G core needle biopsies were taken. In instances where rebiopsy or surgical excision was not performed, interval stability for up to 2 years was recorded.
The pathology was reviewed by one of eight pathologists, each with a minimum of 8 years' work experience. The majority of cases were reviewed by or had a second opinion provided for by one of two fellowship-trained breast pathologists with 10 and 20 years' experience, respectively.
Using the same histopathology records, 365 biopsy-proven cases of non-cellular fibroadenoma between 1 January 2005 and 31 December 2009 were selected. The electronic medical records for these patients were accessed. Histopathology results of patients who underwent repeat biopsy and/or surgical excision were recorded. For all remaining patients, interval stability for up to 2 years was recorded. Patients who were lost to follow-up were cross-referenced with the Champlain Regional Cancer Centre (Ottawa, ON).
RESULTS
The 365 breast biopsies out of the total of 9125 breast biopsies performed had pathology demonstrating stromal fibrosis, yielding an incidence of 4%.
Imaging findings of the 365 biopsy-proven cases of stromal fibrosis included masses (50%), microcalcifications (36%), focal asymmetry (11%), focus of enhancement on MRI (2%), architectural distortion (1%) and non–mass-like enhancement on MRI (0.01%). 263 patients (72%) were assigned BI-RADS 4A, 47 (13%) BI-RADS 4B, 3 (1%) BI-RADS 4 and 10 (3%) BI-RADS 5 and, in 42 biopsies (12%), BI-RADS was not recorded.
As illustrated in Figure 2, of the 365 cases of stromal fibrosis on initial percutaneous breast biopsy, 25 cases (7%) were subsequently upgraded on repeat biopsy (n = 12) or surgical excision (n = 13). Of these 25 cases, 7 were upgraded to ductal carcinoma in situ (DCIS) and 18 to invasive carcinoma. Of the 25 cases that were upgraded to malignancy, 12 were upgraded based on repeat biopsy. Of these, seven were performed by stereotactic-guided vacuum-assisted biopsy, one by ultrasound-guided vacuum-assisted biopsy, one by repeat ultrasound-guided 14-G core needle biopsy within 4 weeks of the initial biopsy and one by ultrasound-guided 18-G biopsy of a suspicious axillary lymph node. In the latter case, initial biopsy of a mass revealed stromal fibrosis, which was felt to be discordant, then a repeat biopsy targeting a suspicious lymph node demonstrated invasive ductal carcinoma (IDC), and both lesion and lymph node were confirmed to be malignant at surgery. The remaining two cases were upgraded by repeat ultrasound-guided 14-G core needle biopsy, at 16 and 24 months, respectively, after initial breast biopsy owing to worsening palpable mass and suspicious MRI features, respectively.
Figure 2.
Outcomes of biopsy-proven cases of stromal fibrosis in a 2-year period. DCIS, ductal carcinoma in situ; LCIS, lobular carcinoma in situ.
Of the 337 patients with stromal fibrosis on percutaneous breast biopsy without upgrade, 190 (56%) patients were followed up with breast imaging, 128 (38%) for at least 2 years (range, 24–91 months; median, 49 months; and mean, 52 months), and 50 (15%) were followed for less than 2 years (range, 4–22 months; median, 12 months; and mean, 11 months). In 11 cases, benignity was proven surgically. 147 (44%) patients were lost to follow-up (Figure 2). Patients who were lost to follow-up were cross-referenced with the Regional Cancer Centre. In total, 13 of these patients who were lost to follow-up had been treated for other malignancies: 9 patients for lung cancer, 1 patient for melanoma, 2 patients for contralateral breast cancer and 1 patient for metastatic cancer of unknown primary malignancy. None of these patients presented with a diagnosis of ipsilateral breast cancer within 2 years of breast biopsy.
There were no upgrades in diagnosis to malignancy in the non-cellular fibroadenoma internal control group (Figure 3).
Figure 3.
Internal control group: outcomes of biopsy-proven cases of non-cellular fibroadenoma in a 2-year period.
As illustrated in Figure 4, of the 25 cases that were upgraded to malignancy, 8 cases (32%) were concordant and 16 cases (64%) were discordant with the histopathology diagnosis of stromal fibrosis. In one case (4%), a radiology–pathology correlation was not provided. Thus, out of the 365 total cases of biopsy-proven fibroadenoma, the upgrade rate in lesions with concordant radiology–pathology correlation was 2% (8 of 365). Table 1 lists the imaging findings in cases of concordance with subsequent upgrade to malignancy.
Figure 4.
Radiology–pathology correlation and outcomes.
Table 1.
Imaging findings in concordant cases of stromal fibrosis
| Patient | Time to upgrade from initial breast biopsy diagnosis of stromal fibrosis | Pathology on repeat biopsy | Pathology at surgical excision | Clinical history |
|---|---|---|---|---|
| 1 | 4 weeks | Not performed | IDC | Surgical excision performed because of suspicious clinical features (nipple inversion and palpable lump) |
| 2 | 3 weeks | Not performed | DCIS | Mastectomy performed for multicentric malignancy |
| 3 | 4 weeks | Not performed | IDC | Patient preference for excision |
| 4 | 3 weeks | Not performed | DCIS | Patient preference for excision |
| 5 | 24 months | IDC | IDC | Suspicious features on MRI follow-up |
| 6 | 8 months | Not performed | IDC | Interval increase in size on follow-up |
| 7 | 4 weeks | Not performed | IDC | Patient preference for excision |
| 8 | 16 months | DCIS | DCIS | Interval increase in size on follow-up |
DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma.
All 16 discordant cases ultimately underwent surgical excision (Table 2). In six cases of discordance, an upgrade was made at the time of surgery. In ten cases, the upgrade occurred at repeat biopsy. Of these ten cases, eight were rebiopsied with vacuum assistance, one was biopsied with 14-G core needle and, in one case, the radiologist felt that the target may not have been biopsied and a biopsy of an axillary lymph node with 18-G core needle was performed resulting in an upgrade.
Table 2.
Imaging findings in discordant cases of stromal fibrosis
| Patient | Repeat biopsy | Imaging features | Final pathology |
|---|---|---|---|
| 1 | 11 G vacuum | Suspicious microcalcifications | DCIS |
| 2 | 11 G vacuum | Spiculated nodule | IDC |
| 3 | 11 G vacuum | Suspicious microcalcifications | IDC |
| 4 | 11 G vacuum | Asymmetry with microcalcifications | IDC |
| 5 | 11 G vacuum | Poorly defined hypoechoic solid mass with possible calcification | IDC |
| 6 | 14 G core | 8-mm solid mass | IDC |
| 7 | 18 G core lymph node biopsy | 8-cm poorly defined mass | IDC |
| 8 | 11 G vacuum | Indeterminate microcalcifications | IDC |
| 9 | Not performed | Solid, irregular 7-mm mass | DCIS |
| 10 | Not performed | Cluster of microcalcifications | IDC |
| 11 | Not performed | Clumped linear enhancement | IDC |
| 12 | 12 G vacuum | Hypoechoic mass | IDC |
| 13 | 11 G vacuum | Suspicious microcalcifications | DCIS |
| 14 | Not performed | Hypoechoic mass | IDC |
| 15 | Not performed | Hypoechoic mass | IDC |
| 16 | Not performed | Hypoechoic mass | IDC |
DCIS, ductal carcinoma in situ.
In the 25 cases of upgrade, stromal fibrosis was the sole histopathology finding in 13 biopsy specimens. The most common finding in addition to the presence of stromal fibrosis was the presence of a fragment of cyst in three cases. Other histopathology findings on biopsy are outlined in Table 3.
Table 3.
Histopathology findings in addition to stromal fibrosis in patients with subsequent upgrade to in situ or invasive malignancy.
| Additional histopathology findings | Number of cases |
|---|---|
| No other histopathology finding | 13 |
| Dilated duct | 2 |
| Typical duct hyperplasia | 1 |
| Epithelial hyperplasia without atypia | 1 |
| Hemosiderin | 2 |
| Periductal elastosis | 1 |
| Cyst | 3 |
| Apocrine metaplasia | 1 |
| Atypical lobular hyperplasia | 1 |
| Atypical duct hyperplasia | 1 |
| Fibroadenoma | 1 |
| Sclerosing adenosis | 1 |
| Columnar cell hyperplasia | 1 |
| Columnar cell change without atypia | 1 |
| Hamartoma | 1 |
Three biopsy specimens had two additional histopathology findings.
DISCUSSION
Desmoplastic proliferation of collagenous and fibrous stroma is a known and accepted host response to the presence of primary breast carcinoma.5 Thus, the histopathology diagnosis of stromal fibrosis on percutaneous breast biopsy might be concerning for the undersampling of an adjacent primary breast cancer. At present, there are no consensus guidelines for the management of the histopathology diagnosis of stromal fibrosis.
In our population, the frequency of stromal fibrosis was found to be 4%, which falls within the range of 2.1–9.0% reported in the literature.1 The upgrade rate to in situ or invasive malignancy in our study was 7%, which is higher than in the series published by Sklair-Levy et al,1 where the reported rate of missed cancer was 2.7%, and Rosen et al4 and Harvey et al,6 where no upgrades were reported. This could be attributed to our broader inclusion criteria, which included cases diagnosed by core needle biopsy and vacuum-assisted biopsy, and biopsies performed with a range of needle gauge. We also included every case where stromal fibrosis was present, not just those where it was the only finding.
In cases of stromal fibrosis that were upgraded to in situ or invasive malignancy, stromal fibrosis was the sole finding in 13 cases. A variety of other pathology was also demonstrated; however, none of these were consistently present and no high-risk lesions were included that confound our findings. Unfortunately, subgroup analysis could not be performed owing to the small sample size of upgraded cases (n = 25). However, of the other pathologies that were present, none of these were present consistently.
There is a wide spectrum of imaging features of stromal fibrosis.2,3 A study characterizing the sonographic imaging features of stromal fibrosis demonstrated that 80% of cases were well-defined hypoechoic masses, 12% were ill-defined masses and 8% demonstrated shadowing without a visible mass. Similarly, stromal fibrosis has variable appearance on mammography and can present as circumscribed masses (14%), lobulated masses (5%), microlobulated masses (2%), obscured masses (25%), architectural distortions (5%) and as asymmetric densities (34%).2 Suspicious microcalcifications have not been described as a feature of stromal fibrosis. Any of these findings may lead to a breast biopsy. Owing to the varied imaging appearance of stromal fibrosis, image findings alone are not sufficient to determine the presence of malignancy in cases of radiology–pathology concordance and discordance (Figure 5).
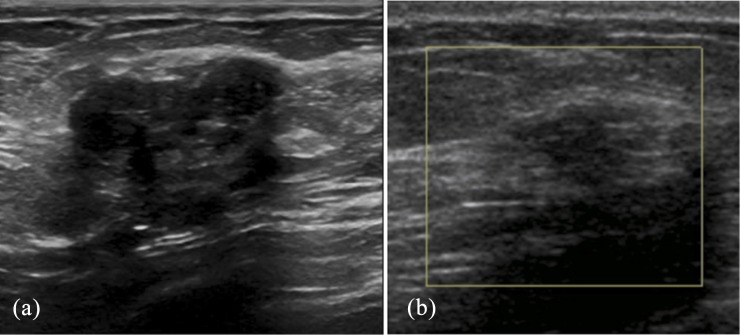
Figure 5.
Discordant and concordant biopsy-proven cases of stromal fibrosis. (a) Solid, hypoechoic mass with irregular margins and posterior acoustic shadowing. Initial 14-G ultrasound-guided core needle biopsy demonstrated stromal fibrosis. The pathology results were discordant, and a repeat 12-G ultrasound-guided vacuum-assisted biopsy was performed 2 weeks later demonstrating ductal carcinoma in situ that was subsequently confirmed at surgery. (b) Small hypoechoic, irregular mass. Initial 14-G ultrasound-guided core needle biopsy demonstrated stromal fibrosis, felt to be concordant with imaging findings. A 16-month follow-up demonstrated interval growth of the lesion. Repeat biopsy was performed and demonstrated invasive ductal carcinoma that was subsequently confirmed at surgery.
From our analysis, we found an upgrade rate of 7% in cases of biopsy-proven stromal fibrosis. It is particularly interesting to note that upon radiology–pathology correlation, 32% of the upgraded cases were felt to be concordant with a benign histopathology diagnosis. However, the percentage of patients with upgrade, who were felt to be concordant, comprises 2% of the entire population of patients with biopsy-proven stromal fibrosis.
Surgical excision or a repeat biopsy for discordant radiologic–pathological correlation is the standard of care in breast imaging.8 Repeat biopsy with a larger needle and with vacuum assistance is generally preferred. In our study, ten cases underwent repeat biopsy, eight of which were performed with vacuum-assisted biopsy. An upgrade to malignancy was demonstrated in all ten cases. In the remaining six cases, upgrade was confirmed upon surgical excision.
A potential limitation of the study is the number of cases that were lost to follow-up. These patients were presumed not to have developed a malignancy on the assumption that any such patient would have returned to our centre for further management. We sought to mitigate this limitation by cross-referencing the Champlain Regional Cancer Centre, ON, Canada, that provides services for 1,276,370 residents in this geographic region (identified as the Champlain Local Health Integration Network). Cancer care services for patients within this region are centrally delivered at designated treatment centres, and any patients diagnosed with cancer who receive treatment between sites within the region are captured in this database.9 In addition, it should be noted that the province of Ontario has a relatively high rate of immigration and a negligible rate of emigration.10 However, patients who may have moved or sought treatment in other regions would still remain unaccounted for, thus artificially lowering our upgrade rate. A second limitation is that variability in interpretation and reporting practices of pathologists was not accounted for. Lastly, an internal departmental database was used as the basis from which cases of fibrosis were identified; however, the reliability and validity of the database was not verified against a central database. As such, it is theoretically possible that not all cases of stromal fibrosis were captured in this analysis.
Despite the limitations of this study, our analysis shows a 7% upgrade rate of biopsy-proven stromal fibrosis. We also show a 2% false-negative rate of cases of stromal fibrosis with features concordant with a benign diagnosis, which were subsequently upgraded to malignancy. For these reasons, we now recommend that all instances of stromal fibrosis on percutaneous breast biopsy with radiology–pathology concordance be categorized as a BI-RADS lesion 3 with a 6-month follow-up. Cases of radiology–pathology discordance should undergo repeat biopsy or surgical excision.
REFERENCES
- 1.Sklair-Levy M, Samuels TH, Catzavelos C, Hamilton P, Shumak R. Stromal fibrosis of the breast. AJR Am J Roentgenol 2001; 177: 573–7. doi: 10.2214/ajr.177.3.1770573 [DOI] [PubMed] [Google Scholar]
- 2.Revelon G, Sherman ME, Gatewood OM, Brem RF. Focal fibrosis of the breast: imaging characteristics and histopathologic correlation. Radiology 2000; 216: 255–9. doi: 10.1148/radiology.216.1.r00jl16255 [DOI] [PubMed] [Google Scholar]
- 3.Lee SJ, Mahoney MC, Khan S. MRI features of stromal fibrosis of the breast with histopathologic correlation. AJR Am J Roentgenol 2011; 197: 755–62. doi: 10.2214/AJR.11.6489 [DOI] [PubMed] [Google Scholar]
- 4.Rosen EL, Soo MS, Bentley RC. Focal fibrosis: a common breast lesion diagnosed at imaging-guided core biopsy. AJR Am J Roentgenol 1999; 173: 1657–62. doi: 10.2214/ajr.173.6.10584816 [DOI] [PubMed] [Google Scholar]
- 5.Walker, RA. The complexities of breast cancer desmoplasia. Breast Cancer Res 2001; 3: 143–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Harvey SC, Denison CM, Lester SC, DiPiro PJ, Smith DN, Meyer JE. Fibrous nodules found at large-core needle biopsy of the breast: imaging features. Radiology 1999; 211: 535–40. doi: 10.1148/radiology.211.2.r99ma42535 [DOI] [PubMed] [Google Scholar]
- 7.American College of Radiology. Breast Imaging Reporting and Data System, 4th edn. Reston, VA: American College of Radiology; 2003. [Google Scholar]
- 8.Youk JH, Kim EK, Kim MJ, Lee JY, Oh KK Missed breast cancers at US-guided core needle biopsy: how to reduce them. Radiographics 2007; 27: 79–94. [DOI] [PubMed] [Google Scholar]
- 9.Champlain Regional Cancer Centre. [Updated 2009, cited 2014]. Available from: http://www.champlain-rcp.on.ca/
- 10.Ministry of Finance. Ontario. Population. Projections. Update, 2011–2036 (Spring 2012). Queen's Printer for Ontario; 2012. [Google Scholar]