Abstract
Aim:
Activation of muscarinic receptors on the detrusor smooth muscle is followed by contraction, which involves both myosin light chain kinase (MLCK) and Rho kinase (ROCK). The aim of this study was to determine the relative contributions of MLCK and ROCK to carbachol-induced contraction of human detrusor smooth muscle in vitro.
Methods:
Detrusor smooth muscle strips were prepared from the macroscopically unaffected bladder wall of patients underwent cystectomy. The strips were fixed in an organ bath, and carbachol or KCl-induced isometric contractions were measured by force transducers.
Results:
Addition of carbachol (0.4-4 μmol/L) into the bath induced concentration-dependent contractions of detrusor specimens, which was completely abolished by atropine (1 μmol/L). Pre-incubation of detrusor specimens with either the MLCK inhibitor ML-9 or the ROCK inhibitors HA1100 and Y-27632 (each at 10 μmol/L) significantly blocked carbachol-induced contractions as compared to the time-control experiments. Moreover, MLCK and ROCK inhibition were equally effective in reducing carbachol-induced contractions. The residual carbachol-induced contractions in the presence of both MLCK and ROCK inhibitors were significantly smaller than the contractions obtained when only one enzyme (either MLCK or ROCK) was inhibited, suggesting an additive effect of the two kinases. Interestingly, ROCK-mediated carbachol-induced contractions were positively correlated to the age of patients (r=o.52, P<0.05).
Conclusion:
Both MLCK and ROCK contribute to carbachol-induced contractions of human detrusor smooth muscle. ROCK inhibitors may be a new pharmacological approach to modulate human bladder hyperactivity.
Keywords: bladder hyperactivity, human detrusor, muscarinic receptor, carbachol, atropine, myosin light chain kinase, Rho kinase, Y-27632, HA1100, ML-9
Introduction
Acetylcholine is a major excitatory transmitter in urogenital organs leading to a pronounced and sustained contraction of smooth muscle cells. Following the activation of post-junctional muscarinic receptors, the prototypical phospholipase C-dependent signaling pathway1 results in an increase in the intracellular Ca2+ concentration that leads to the calmodulin-mediated activation of myosin light chain kinase (MLCK). Alternatively, there is abundant evidence for muscarinic receptor-dependent activation of voltage-dependent Ca2+ channels with subsequent Ca2+ influx from the extracellular space2,3,4. As a consequence, MLCK phosphorylates the regulatory myosin light chain of 20 kDa and thereby initiates contraction5. However, in addition to this MLCK-dependent mechanism, smooth muscle contraction may also be induced by inhibition of the counteracting enzyme – myosin light chain phosphatase – a mechanism, which is often referred to as Ca2+ sensitization. Inhibition of this enzyme is achieved by phosphorylation of a regulatory targeting subunit, which is a major substrate of Rho kinase (ROCK)6,7.
In the human urinary bladder, excitatory cholinergic transmission is particularly present in the detrusor smooth muscle which is responsible for the pronounced bladder contraction during micturition8. Although M2 receptors are more abundantly expressed than M3 receptors, the latter subtype seems to be functionally dominant9,10,11,12. The pharmacological differentiation between the MLCK-dependent and the ROCK-dependent components of detrusor contraction is likely to be translationally significant, since alterations of the ROCK pathway have been implicated in a number of rodent models of bladder hyperactivity13,14,15. In the human detrusor, Ca2+ sensitization following exposure to carbachol has been observed and was found to involve the ROCK pathway3,16. However, the differential contribution of MLCK and ROCK has not been compared directly, and – especially in humans – it is not known which pathway is predominantly involved. Therefore, we recorded isometric contractions from human detrusor muscle strips in an organ bath, and found that both MLCK and ROCK inhibition were equally effective in reducing carbachol-induced contractions, and ROCK-mediated contraction correlates with age.
Materials and methods
Preparation of human detrusor samples
Human detrusor samples were obtained from 26 patients (Table 1). All in vitro experiments with human material performed in this study were approved by the local ethics committee (University of Rostock), and the informed consent to participate in this study was obtained from each patient.
Table 1. Clinical patient data.
| Pat # | Age | Sex | Primary disease | Secondary diseases |
|---|---|---|---|---|
| 1 | 68 | M | Urinary bladder cancer | – |
| 2 | 66 | F | Urinary bladder cancer | Diabetes, hypertension |
| 3 | 61 | M | Urinary bladder cancer | Hypothyreosis, multiple transurethral resections |
| 4 | 80 | M | Urinary bladder cancer | – |
| 5 | 68 | M | Urinary bladder cancer | – |
| 6 | 62 | M | Urinary bladder cancer | Hypertension, coronary heart disease and myocardial infarction, stroke |
| 7 | 73 | M | Urinary bladder cancer | Diabetes, hypertension |
| 8 | 46 | M | Urinary bladder cancer | Renal failure with dialysis, hypertension |
| 9 | 84 | F | Urinary bladder cancer | Renal failure, hypertension |
| 10 | 50 | M | Prostate cancer | Miction dysfunction |
| 11 | 60 | M | Urinary bladder cancer | Diabetes |
| 12 | 80 | F | Urinary bladder cancer | Hypothyreosis |
| 13 | 68 | M | Neurogenic micturition dysfunction, shrinking bladder | Alcoholic polyneuropathy |
| 14 | 74 | M | Urinary bladder cancer | Diabetes, hypertension |
| 15 | 59 | M | Urinary bladder cancer | – |
| 16 | 60 | M | Urinary bladder cancer | Hypertension |
| 17 | 57 | M | Urinary bladder cancer | – |
| 18 | 71 | M | Urinary bladder cancer | Hypertension, coronary heart disease and myocardial infarction |
| 19 | 74 | M | Urinary bladder cancer | Prostate cancer, hypertension |
| 20 | 72 | M | Urinary bladder cancer | Hypertension, aortic aneurysm |
| 21 | 75 | M | Urinary bladder cancer | Prostate cancer, hypertension, renal failure, respiratory insufficiency, aortic aneurysm |
| 22 | 76 | M | Urinary bladder cancer | Prostate cancer, hypertension, cardiac failure, respiratory insufficiency |
| 23 | 82 | M | Urinary bladder cancer | Prostate cancer, cardiac failure, aortic aneurysm |
| 24 | 60 | M | Urinary bladder cancer | Prostate cancer |
| 25 | 62 | M | Urinary bladder cancer | Prostate cancer |
| 26 | 68 | W | Neurogenic micturition dysfunction | Multiple sclerosis |
Immediately following resection of the urinary bladder, a tissue sample of approx 2 cm width was excised from the macroscopically unaffected wall of the detrusor muscle. These samples were submerged into a storage solution containing (in mmol/L) 120 NaCl, 4.5 KCl, 26 NaHCO3, 1.2 NaH2PO4, 1.6 CaCl2, 1.0 MgSO4, 0.025 Na2-EDTA, 5.5 glucose, 5 HEPES (pH=7.4) and kept at 4 °C for the transfer from the operating room to the laboratory (maximum 1 h). Each detrusor sample was then freed from mucosal tissue and cut into 4–12 muscle strips of 1–2 cm length and 2–3 mm width. Thin nylon threads were sutured to either end of these specimens to enable longitudinal fixation in an organ bath (Panlab ML0146/C, ADInstruments, Spechbach, Germany) filled with a buffer that contained (in mmol/L) 120 NaCl, 4.7 KCl, 2.5 CaCl2, 1.2 MgCl2, 30 NaHCO3, 1.2 KH2PO4, 0.5 Na2-EDTA, 5.5 glucose, 2 sodium pyruvate (pH=7.4) and was gassed with carbogen (95% O2 and 5% CO2).
Isometric contractions in vitro
After fixation in the organ bath, the temperature was slowly raised to 37 °C and the detrusor specimens were slightly stretched and allowed to recover for at least 5 h. During this time, the preparations showed a stable baseline tone with rhythmic activity (see insets in Figure 1A). Isometric contractions of the smooth muscle strips induced by carbachol (CCh) or KCl were measured by force transducers (MLT0201), recorded with a bridge amplifier (ML224) connected to an analog-to-digital converter (Powerlab 4/30, LabChart 7, ADInstruments, Australia).
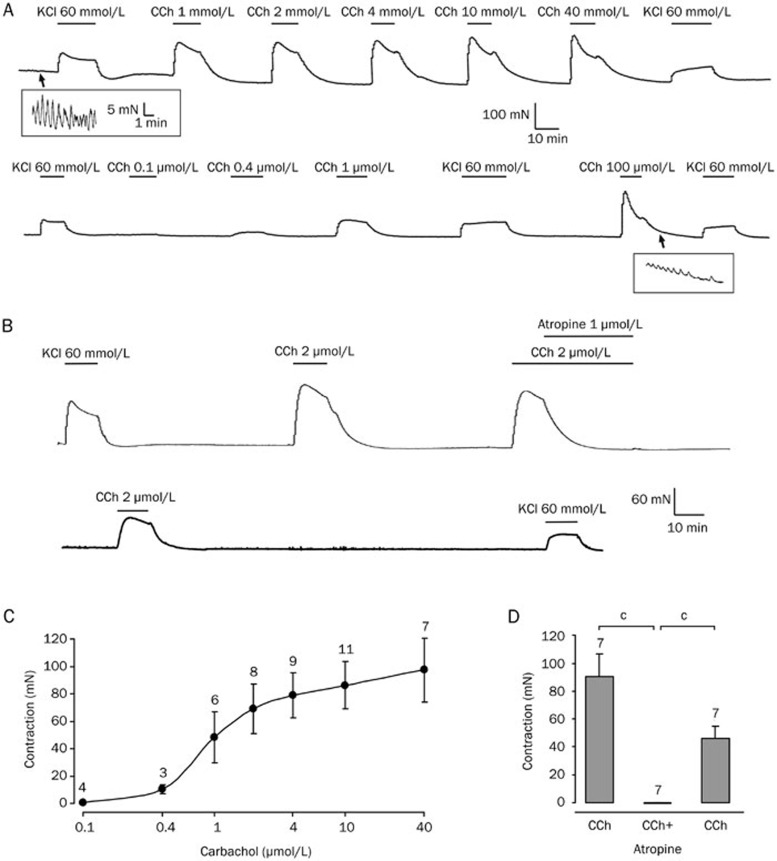
Figure 1.
Carbachol-induced contractions in human detrusor in vitro are atropine-sensitive. (A) Carbachol (CCh, 0.1–100 μmol/L) caused dose-dependent contractions in a human detrusor specimen. The upper trace was started at 6 h following cystectomy. The gap between the upper and the lower trace is 7 h. KCl (60 mmol/L) was used at the beginning and at the end of the experiment to verify stable recording conditions. The insets demonstrate the rhythmic activity of detrusor smooth muscle. Note the different scales within the insets. (B) The upper trace (starting at 5 h following cystectomy) shows contractions induced by carbachol (CCh, 2 μmol/L) in a human detrusor specimen that were abolished by the muscarine receptor antagonist atropine (1 μmol/L). The lower trace shows partial recovery of carbachol-induced contraction following washout of atropine. (C) Dose-response curve of carbachol in human detrusor. The half-maximal contraction was achieved between 1 and 2 μmol/L carbachol (EC50=1.1 μmol/L). Note that a single specimen was tested at multiple concentrations (total number of specimens: n=15 of 4 patients). (D) Carbachol-induced contractions were significantly reduced following atropine (total number of specimens: n=7 of 2 patients). bP<0.05, cP<0.01.
The major aim of this study was to analyze the effect of different kinase blockers on the carbachol-induced contraction. Since both tachyphylaxis and, to some extent, also tissue deterioration cannot be ruled out in the prolonged course of the experiment, we performed time-control experiments in all parts of this study. Time-control experiments contained only the vehicle of the respective compound (occasionally DMSO). Our organ bath is equipped with four chambers, and we always used one chamber for time-control experiments.
Carbachol, the ROCK blockers HA1100 and Y-27632 as well as the MLCK inhibitor ML-9 were purchased from Tocris Bioscience (Bristol, United Kingdom). All other chemicals were obtained from Sigma-Aldrich (Taufkirchen, Germany). The application of drugs was performed by adding 100 μL (carbachol, HA1100, Y-27632, ML-9, verapamil) to the organ bath solution to yield the individual final concentration. In the case of KCl, 1000 μL of stock solution (1.5 mol/L KCl) was added (final KCl concentration of 60 mmol/L). All preparations were challenged with KCl at the beginning as well as at the end of the experiment. Experiments were included into statistics when the KCl response at the end of the experiment was at least 50% of the initial contraction (115 out of 165). On average, exposure of smooth muscle preparations to bath solution containing 60 mmol/L KCl caused a mean contraction of 40±4 mN at the beginning, and of 28±3 mN at the end of the experiment (ie 73%±2%, n=115). The addition of KCl to the organ bath was not corrected for osmolarity, since hyperosmotic control experiments with addition of the same amount of NaCl had no significant effect on smooth muscle tone (contraction of 1.3±0.6 mN compared to 36.7±7.1 mN by KCl application, n=1917). Drugs were applied for 10–15 min, and the interval between individual drug applications was 45–50 min.
Statistical analysis
All data are expressed as mean±SEM. Statistical comparison of drug effects was performed using the two-tailed Student's t test. The level of significance is indicated (bP<0.05; cP<0.01).
Results
Carbachol-induced contractions in human detrusor
Smooth muscle contraction is generally induced by membrane depolarization or by pharmacological activation of G protein-coupled receptors such as muscarinic receptors. The dose-response curve for the cholinergic agonist carbachol (CCh, 0.1–100 μmol/L) obtained with randomly varying concentrations showed dose-dependent contractions of human detrusor smooth muscle preparations with a half-maximal effect between 1–2 μmol/L (EC50=1.1 μmol/L, Figure 1A/1C). The detrusor contraction induced by 2 μmol/L carbachol was entirely due to muscarinic receptor activation, since atropine (1 μmol/L) completely relaxed the pre-contracted detrusor preparation (from 94±17 mN to -1±1 mN, n=7, P<0.05; Figure 1B/1D). Following washout of atropine, CCh-induced contraction partially recovered (46±9 mN). Hence, for the remainder of the study, 2 μmol/L carbachol was used as the routine concentration in order to assess the role of the two main routes of smooth muscle contraction: MLCK and ROCK.
Role of Rho kinase in carbachol-induced contractions
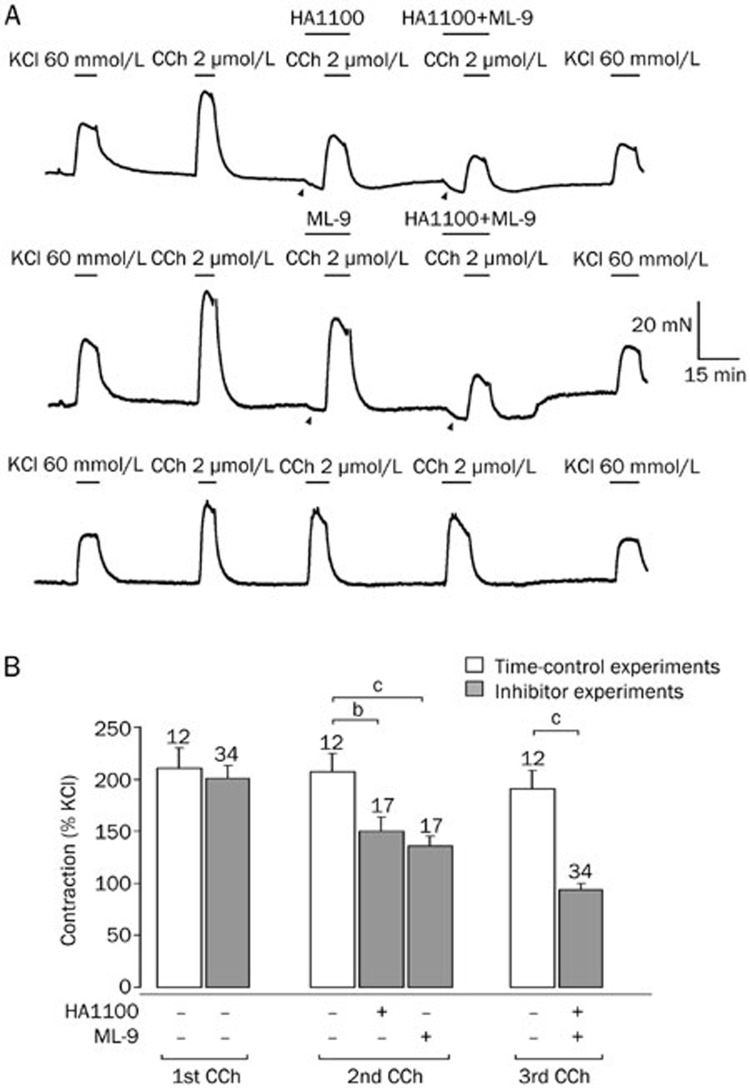
Since we aimed to study the role of these key enzymes in carbachol-induced contractions in the human detrusor muscle, we pre-applied either the MLCK inhibitor ML-9 or the ROCK inhibitor HA1100 (both 10 μmol/L) before the preparations were challenged with 2 μmol/L carbachol. A typical example of such an experiment is depicted in Figure 2A. In different detrusor specimens taken from the same patient, these compounds were tested alone or in combination (upper two traces). Obviously, both HA1100 and ML-9 were able to depress carbachol-induced contractions suggesting a contribution of both MLCK and ROCK. In addition, these blockers caused a marked relaxation of the baseline tone (arrowheads in Figure 2A) indicating that both enzymes were activated under resting conditions. In another subset of specimens taken from the same patient, no enzyme inhibitor was applied. In these time-control experiments, only little run-down of CCh-induced contractions was observed (lower trace, Figure 2A). Hence, we synchronized the inhibitor experiments (Figure 2B, gray bars) with time-control experiments (Figure 2B, white bars) in order to compare the carbachol response in the presence of an enzyme inhibitor to the control carbachol response at the same time point.
Figure 2.
Carbachol-induced contractions are reduced by HA1100 and ML-9. (A) The upper trace (starting at 9 h following cystectomy) shows contractions induced by carbachol (CCh, 2 μmol/L) in a human detrusor specimen that were reduced by the ROCK blocker HA1100 (10 μmol/L) or combined pre-treatment with HA1100 and the MLCK blocker ML-9 (10 μmol/L). Note the reduction of the basal tone (arrow heads) indicating kinase activity at rest. The middle trace shows a similar experiment with another specimen from the same patient demonstrating that both ML-9 alone and combined application of ML-9 and HA1100 reduced the carbachol-induced contraction. The lower trace shows time-control experiments of another specimen from the same patient demonstrating only little tachyphylaxis upon repetitive carbachol applications. Application of KCl (60 mmol/L) was used at the beginning and at the end of the experiment to verify stable recording conditions. (B) Carbachol-induced contractions (in % of initial response to KCl 60 mmol/L) showed little tachyphylaxis (time-control experiments), but were significantly reduced following HA1100 or ML-9 treatment (total number of specimens: n=40 of 4 patients). bP<0.05, cP<0.01.
We observed substantial variation of CCh-induced contractions among different patients, but we found a striking correlation between these contractions and those observed by initial KCl-application (correlation coefficient of 0.92, n=46, P<0.01). Therefore, we used the initial contraction following KCl to normalize the CCh-induced contractions (Figure 2B). Expressed as the percentage of the KCl effect, the initial contraction induced by carbachol was 201%±14% (n=34, P>0.7 versus 211%±19%, n=12, in time-controls; Figure 2B). The ROCK inhibitor HA1100 significantly reduced this carbachol response to 150%±14% (n=17, P<0.05 versus 207%±18%, n=12, in time-controls). When the MLCK blocker ML-9 was used instead, the contraction by carbachol was reduced to 136%±10% (n=17, P<0.01 versus 207%±18%, n=12, in time-controls). Moreover, the combined application of both inhibitors had an additive effect and further reduced the carbachol response to 94%±6% (n=34, P<0.001 versus 191%±17%, n=12, in time-controls; P<0.01 versus HA1100 or ML-9 alone). These results suggest that MLCK and ROCK equally and independently contribute to the CCh-induced contraction of human detrusor smooth muscle.
While ROCK preferably acts via inhibition of myosin light chain phosphatase leading to Ca2+ sensitization in smooth muscle, MLCK-dependent contraction is believed to occur downstream of both Ca2+ influx and Ca2+ release from internal stores. To test the impact of Ca2+ influx via voltage-dependent L-type Ca2+ channels, we compared the effects of HA1100 and the L-type Ca2+ channel blocker verapamil (40 μmol/L). While HA1100 reduced the carbachol-induced contraction to 144%±38% (n=6) of the KCl-induced contraction, verapamil caused a significantly stronger reduction to 104%±33% (n=6, P<0.05 versus HA1100). However, the combination of both compounds strongly and additively suppressed the carbachol-induced response (26%±9%, n=6, P<0.05 versus HA1100 and verapamil). These data may suggest that the remaining CCh-induced contraction following ROCK inhibition is predominantly mediated by Ca2+ influx via L-type Ca2+ channels rather than by Ca2+ release from internal stores.
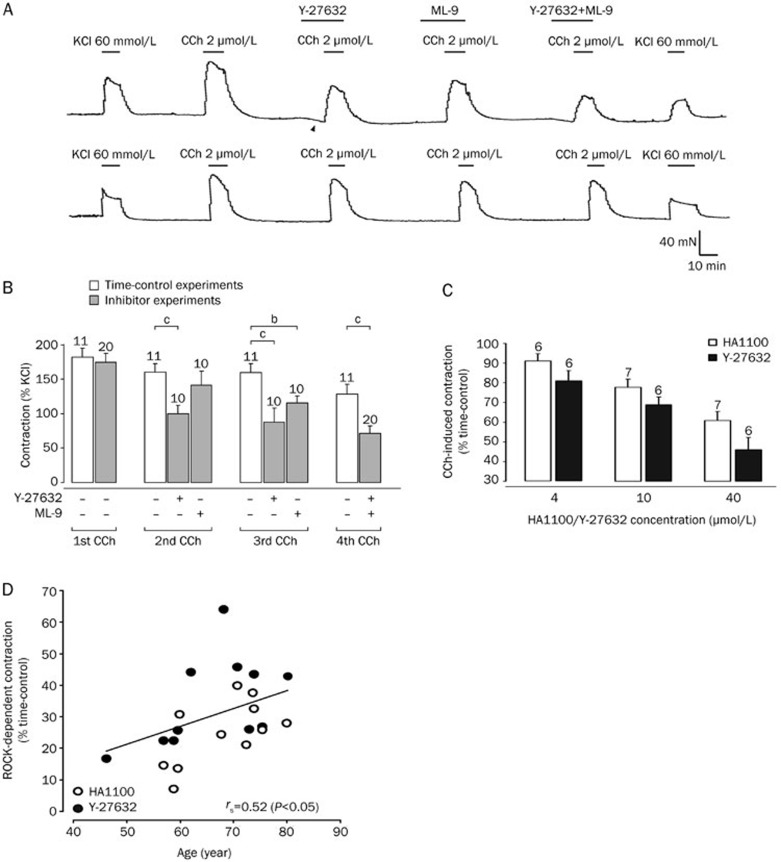
To validate our data showing the involvement of ROCK in carbachol-induced contractions, we chose to also test another ROCK inhibitor, Y-27632 (10 μmol/L), to gauge the effect of this compound on the carbachol response. The effect of carbachol on the detrusor specimens was analyzed in presence of Y-27632 or ML-9 alone (the order of which was randomized), as well as after pre-treatment with both inhibitors in combination (Figure 3A, upper trace). Time-control experiments (Figure 3A, lower trace) indicated a small run-down of the carbachol response. Pooled data of this experiment are illustrated in Figure 3B. As HA1100, the ROCK inhibitor Y-27632 also significantly reduced the contraction by carbachol. The combined application of both inhibitors Y-27632 and ML-9 had again an additive effect and further reduced the carbachol response. These results confirm our previous data and provide further evidence for the finding that ROCK is at least as important as MLCK in carbachol-induced contraction of human detrusor smooth muscle.
Figure 3.
Carbachol-induced contractions are reduced by Y-27632 and ML-9, and Rho kinase-mediated contraction is age-dependent. (A) The upper trace (starting at 7 h following cystectomy) shows contractions induced by carbachol (2 μmol/L) in a human detrusor specimen that were reduced by both the ROCK blocker Y-27632 (10 μmol/L) and the MLCK blocker ML-9 (10 μmol/L). Note the reduction of the basal tone (arrow heads) indicating kinase activity at rest. The lower trace shows time-control experiments of another specimen from the same patient demonstrating only little tachyphylaxis upon repetitive carbachol applications. Application of KCl (60 mmol/L) was used at the beginning and at the end of the experiment to verify stable recording conditions. (B) Carbachol-induced contractions (in % of initial response to KCl 60 mmol/L) showed little tachyphylaxis (time-control experiments), but were significantly reduced following Y-27632 or ML-9 (total number of specimens: n=31 of 5 patients). bP<0.05, cP<0.01. (C) Carbachol-induced contractions following ROCK inhibition (in % of the time-matched control response) was dose-dependent for both Y-27632 and HA1100 (total number of specimens: n=94 of 7 patients). (D) The carbachol-induced contraction following ROCK inhibition with either HA1100 (10 μmol/L) or Y-27632 (10 μmol/L) was significantly age-dependent (Spearman's rank correlation coefficient of 0.52, P<0.05). In this panel, data from Figures 2B and 3B have been included (total number of patients: n=16).
Age-dependence of Rho kinase-mediated carbachol-induced contraction
Our data so far demonstrated that ROCK activation is one of the major pathways for human detrusor smooth muscle contraction. Moreover, both blockers HA1100 and Y-27632, showed a significant concentration-dependent inhibitory effect on the CCh-induced contraction (Figure 3C). Another conspicuous observation in our data was the considerable variability of CCh-induced contractions in inhibitor experiments among patients. Since we hypothesized that the patients' age might in part account for this variation, we re-analyzed our data and performed a correlation analysis between age and CCh-induced contraction in the presence of ROCK inhibitor (Figure 3D). Pooling all data with 10 μmol/L HA1100 and 10 μmol/L Y-27632 from 16 patients, we found a significant positive correlation between age and ROCK-mediated CCh-induced contraction (Spearman's rank correlation coefficient of 0.52; P<0.05). In contrast to the ROCK-dependent contraction, the MLCK-mediated CCh-induced contraction was not significantly correlated to age (Spearman's rank correlation coefficient of 0.38, P=0.31). Hence, ROCK-mediated CCh-induced contractions in human detrusor are strongly age-dependent playing a major role in aging patients.
Discussion
The present study evaluated the differential role of MLCK and ROCK in human urinary bladder smooth muscle. We demonstrated that carbachol is capable to activate the ROCK pathway in the human detrusor and thereby confirmed previous reports3,16. Moreover, we could show that the combined application of both MLCK and ROCK inhibitors was significantly more potent than one of these compounds alone indicating that these pathways had been activated independently. The clinically relevant finding was the significant correlation between age and ROCK contribution to CCh-induced contraction in human detrusor.
Post-junctional muscarinic receptors mediate the CCh-induced detrusor contraction, predominantly involving M3 receptor activation9,10,11,12. The potency of carbachol in the present study is consistent with the EC50 values in previous reports on human tissue (0.7–2 μmol/L)18,19,20,21. This is important to note because it indicates that our results obtained from resected human tissue were not largely influenced by the patient population or the surgical procedures involved.
One major finding of our study was that two different ROCK inhibitors significantly reduced the human detrusor contraction induced by carbachol. Involvement of ROCK activation in muscarinic receptor-dependent detrusor contraction has been observed in mice22, rats23,24,25,26, rabbits27, and humans3,16. Thus, we confirmed that ROCK significantly contributes to the CCh-induced contraction in human tissue. But how does carbachol activate this enzyme? The prototypical signal transduction cascade following muscarinic receptor activation includes phospholipase C (PLC) activation, Ca2+ release from internal stores and subsequent activation of myosin light chain kinase. However, how the cytosolic concentration of Ca2+ ions is raised has already been questioned since L-type Ca2+ channel blockers substantially reduced CCh-induced contractions2,4, and PLC inhibitors failed to block CCh-induced contractions3. We have performed experiments with verapamil and confirmed that Ca2+ entry via L-type Ca2+ channels is a major contributor of the CCh-induced contraction. Moreover, we found that co-application of verapamil and the ROCK inhibitor HA1100 caused a significantly stronger reduction of the CCh-induced contraction than one of these compounds alone, suggesting that ROCK activation appeared independently of Ca2+ influx via L-type Ca2+ channels. A standard experimental procedure to open these channels is depolarization by high concentration of external K+. It is worth to note that throughout the present study, the CCh-induced contraction was about twice the response than 60 mmol/L KCl within the same detrusor preparation. This is in line with Takahashi et al16 who have observed a threefold response to carbachol when compared to 60 mmol/L KCl, and indicates that depolarization per se does not mimic the contraction mediated by muscarinic receptor activation. Hence, Ca2+ entry via depolarization-activated Ca2+-permeable ion channels may only account for a fraction of the CCh-induced contraction in the human detrusor. In general, alternatives to Ca2+ influx might involve Ca2+ release from internal stores and/or Ca2+-independent cascades of contraction. Actually, both Ca2+ release from internal stores and protein kinase C activation are potential downstream effects of M3 receptor-mediated PLC activation which was, however, identified to be only marginally relevant in CCh-induced contractions3. Thus, our results support the idea that the muscarinic receptor-mediated ROCK activation may be PLC-independent.
Regardless of whether Ca2+ influx or Ca2+ release is more relevant for smooth muscle contraction, the rise in intracellular Ca2+ concentration is regarded as a key step by binding to calmodulin and activating MLCK5. We have directly compared the effect of MLCK and ROCK inhibitors, and found that both enzymes are significant and additive contributors to human detrusor contraction. However, this may not imply that both pathways are disjunctive, since Rho-associated kinase may also be activated by L-type Ca2+ channels28, and MLC20 phosphorylation has recently been shown to be a target for Rho kinase, at least in myometrial tissue29. Thus, it is conceivable that both pathways may have cross-links at some certain step within the cascade also in detrusor smooth muscle. With respect to the specificity of the kinase blockers used in this study, the available MLCK inhibitors may also block L-type Ca2+ channels, while HA1100 and Y-27632 do not have this side effect30. Nonetheless, co-application of both inhibitors was significantly more effective than using one of them alone.
Interestingly enough, the MLCK inhibitor ML-9 appeared to be less potent in reducing the CCh-induced contraction as compared to the L-type Ca2+ channel blocker verapamil. This might be due to the metabotropic effect of L-type Ca2+ channels that has recently been discovered in vascular smooth muscle cells and connects depolarization with ROCK activation28,31. In summary, we propose that human detrusor smooth muscle cells have two major mechanisms of muscarinic receptor-mediated contractions: (a) activation of MLCK predominantly following Ca2+-influx via L-type Ca2+ channels and (b) activation of ROCK that is largely independent of an intracellular Ca2+ rise, but may in part occur as a consequence of L-type Ca2+ channel activation.
When analyzing the ROCK contribution with respect to the age of our patients, we discovered a significant and potentially clinically relevant correlation. Thus, ROCK activity appears to be up-regulated with age. Previous reports, mainly in rodent disease models, have indeed suggested an increased ROCK function13,14,15. Hence, our results support the hypothesis that ROCK upregulation may underly both detrusor degeneration during aging and those pathologies are often associated with aging such as overactive bladder. In this context, ROCK is becoming a novel pharmacological target to oppose bladder hyperactivity symptoms32.
Author contribution
Timo KIRSCHSTEIN, Chris PROTZEL, Katrin PORATH, and Tina SELLMANN performed the research. Timo KIRSCHSTEIN, Chris PROTZEL, Rüdiger KÖHLING, and Oliver W HAKENBERG designed the research. Timo KIRSCHSTEIN and Katrin PORATH analyzed the data. Timo KIRSCHSTEIN, Chris PROTZEL, Rüdiger KÖHLING, and Oliver W HAKENBERG wrote the paper.
Acknowledgments
The authors would like to thank both the nursing staff and the patients at the Department of Urology. This study was supported by a grant provided by the Medical Faculty of the University of Rostock (FORUN program).
References
- Caulfield MP, Birdsall NJ. International Union of Pharmacology. XVII. Classification of muscarinic acetylcholine receptors. Pharmacol Rev. 1998;50:279–90. [PubMed] [Google Scholar]
- Masters JG, Neal DE, Gillespie JI. The contribution of intracellular Ca2+ release to contraction in human bladder smooth muscle. Br J Pharmacol. 1999;127:996–1002. doi: 10.1038/sj.bjp.0702640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider T, Fetscher C, Krege S, Michel MC. Signal transduction underlying carbachol-induced contraction of human urinary bladder. J Pharmacol Exp Ther. 2004;309:1148–53. doi: 10.1124/jpet.103.063735. [DOI] [PubMed] [Google Scholar]
- Wuest M, Hiller N, Braeter M, Hakenberg OW, Wirth MP, Ravens U. Contribution of Ca2+ influx to carbachol-induced detrusor contraction is different in human urinary bladder compared to pig and mouse. Eur J Pharmacol. 2007;565:180–9. doi: 10.1016/j.ejphar.2007.02.046. [DOI] [PubMed] [Google Scholar]
- Somlyo AP, Somlyo AV. Signal transduction and regulation in smooth muscle. Nature. 1994;372:231–6. doi: 10.1038/372231a0. [DOI] [PubMed] [Google Scholar]
- Kimura K, Ito M, Amano M, Chihara K, Fukata Y, Nakafuku M, et al. Regulation of myosin phosphatase by Rho and Rho-associated kinase (Rho-kinase) Science. 1996;273:245–8. doi: 10.1126/science.273.5272.245. [DOI] [PubMed] [Google Scholar]
- Schmidt M, Voss M, Weernink PA, Wetzel J, Amano M, Kaibuchi K, et al. A role for rho-kinase in rho-controlled phospholipase D stimulation by the m3 muscarinic acetylcholine receptor. J Biol Chem. 1999;274:14648–54. doi: 10.1074/jbc.274.21.14648. [DOI] [PubMed] [Google Scholar]
- Abrams P, Andersson KE. Muscarinic receptor antagonists for overactive bladder. BJU Int. 2007;100:987–1006. doi: 10.1111/j.1464-410X.2007.07205.x. [DOI] [PubMed] [Google Scholar]
- Abrams P, Andersson KE, Buccafusco JJ, Chapple C, de Groat WC, Fryer AD, et al. Muscarinic receptors: their distribution and function in body systems, and the implications for treating overactive bladder. Br J Pharmacol. 2006;148:565–78. doi: 10.1038/sj.bjp.0706780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chess-Williams R, Chapple CR, Yamanishi T, Yasuda K, Sellers DJ. The minor population of M3-receptors mediate contraction of human detrusor muscle in vitro. J Auton Pharmacol. 2001;21:243–8. doi: 10.1046/j.1365-2680.2001.00231.x. [DOI] [PubMed] [Google Scholar]
- Fetscher C, Fleichman M, Schmidt M, Krege S, Michel MC. M3 muscarinic receptors mediate contraction of human urinary bladder. Br J Pharmacol. 2002;136:641–3. doi: 10.1038/sj.bjp.0704781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamanishi T, Chapple CR, Chess-Williams R. Which muscarinic receptor is important in the bladder. World J Urol. 2001;19:299–306. doi: 10.1007/s003450100226. [DOI] [PubMed] [Google Scholar]
- Bing W, Chang S, Hypolite JA, DiSanto ME, Zderic SA, Rolf L, et al. Obstruction-induced changes in urinary bladder smooth muscle contractility: a role for Rho kinase. Am J Physiol Renal Physiol. 2003;285:F990–7. doi: 10.1152/ajprenal.00378.2002. [DOI] [PubMed] [Google Scholar]
- Chang S, Hypolite JA, DiSanto ME, Changolkar A, Wein AJ, Chacko S. Increased basal phosphorylation of detrusor smooth muscle myosin in alloxan-induced diabetic rabbit is mediated by upregulation of Rho-kinase beta and CPI-17. Am J Physiol Renal Physiol. 2006;290:F650–6. doi: 10.1152/ajprenal.00235.2005. [DOI] [PubMed] [Google Scholar]
- Rajasekaran M, Mehta N, Baquir A, Kuntz S. Rho-kinase inhibition suppresses potassium chloride-induced bladder hyperactivity in a rat model. Urology. 2007;69:791–4. doi: 10.1016/j.urology.2007.01.071. [DOI] [PubMed] [Google Scholar]
- Takahashi R, Nishimura J, Hirano K, Seki N, Naito S, Kanaide H. Ca2+ sensitization in contraction of human bladder smooth muscle. J Urol. 2004;172:748–52. doi: 10.1097/01.ju.0000130419.32165.6b. [DOI] [PubMed] [Google Scholar]
- Kirschstein T, Rehberg M, Bajorat R, Tokay T, Porath K, Köhling R. High K+-induced contraction requires depolarization-induced Ca2+ release from internal stores in rat gut smooth muscle. Acta Pharmacol Sin. 2009;30:1123–31. doi: 10.1038/aps.2009.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kosan M, Tul M, Ozturk B, Hafez G, Inal G, Cetinkaya M. Alteration in contractile responses in human detrusor smooth muscle from obstructed bladders with overactivity. Urol Int. 2008;80:193–200. doi: 10.1159/000112613. [DOI] [PubMed] [Google Scholar]
- Wuest M, Hecht J, Christ T, Braeter M, Schoeberl C, Hakenberg OW, et al. Pharmacodynamics of propiverine and three of its main metabolites on detrusor contraction. Br J Pharmacol. 2005;145:608–19. doi: 10.1038/sj.bjp.0706244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wuest M, Kaden S, Hakenberg OW, Wirth MP, Ravens U. Effect of rilmakalim on detrusor contraction in the presence and absence of urothelium. Naunyn Schmiedebergs Arch Pharmacol. 2005;372:203–12. doi: 10.1007/s00210-005-0015-4. [DOI] [PubMed] [Google Scholar]
- Wuest M, Morgenstern K, Graf EM, Braeter M, Hakenberg OW, Wirth MP, et al. Cholinergic and purinergic responses in isolated human detrusor in relation to age. J Urol. 2005;173:2182–9. doi: 10.1097/01.ju.0000158126.53702.e4. [DOI] [PubMed] [Google Scholar]
- Teixeira CE, Jin L, Priviero FB, Ying Z, Webb RC. Comparative pharmacological analysis of Rho-kinase inhibitors and identification of molecular components of Ca2+ sensitization in the rat lower urinary tract. Biochem Pharmacol. 2007;74:647–58. doi: 10.1016/j.bcp.2007.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braverman AS, Tibb AS, Ruggieri MR., Sr M2 and M3 muscarinic receptor activation of urinary bladder contractile signal transduction. I. Normal rat bladder. J Pharmacol Exp Ther. 2006;316:869–74. doi: 10.1124/jpet.105.097303. [DOI] [PubMed] [Google Scholar]
- Fleichman M, Schneider T, Fetscher C, Michel MC. Signal transduction underlying carbachol-induced contraction of rat urinary bladder. II. Protein kinases. J Pharmacol Exp Ther. 2004;308:54–8. doi: 10.1124/jpet.103.058255. [DOI] [PubMed] [Google Scholar]
- Schneider T, Hein P, Michel MC. Signal transduction underlying carbachol-induced contraction of rat urinary bladder. I. Phospholipases and Ca2+ sources. J Pharmacol Exp Ther. 2004;308:47–53. doi: 10.1124/jpet.103.058248. [DOI] [PubMed] [Google Scholar]
- Wibberley A, Chen Z, Hu E, Hieble JP, Westfall TD. Expression and functional role of Rho–kinase in rat urinary bladder smooth muscle. Br J Pharmacol. 2003;138:757–66. doi: 10.1038/sj.bjp.0705109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speich JE, Borgsmiller L, Call C, Mohr R, Ratz PH. ROK-induced cross-link formation stiffens passive muscle: reversible strain-induced stress softening in rabbit detrusor. Am J Physiol Cell Physiol. 2005;289:C12–21. doi: 10.1152/ajpcell.00418.2004. [DOI] [PubMed] [Google Scholar]
- Fernández-Tenorio M, Porras-González C, Castellano A, Del Valle-Rodríguez A, López-Barneo J, Ureña J. Metabotropic regulation of RhoA/Rho-associated kinase by L-type Ca2+ channels: new mechanism for depolarization-evoked mammalian arterial contraction. Circ Res. 2011;108:1348–57. doi: 10.1161/CIRCRESAHA.111.240127. [DOI] [PubMed] [Google Scholar]
- Wang L, Jia C, Yu Z, Liu X, Kang L, Cong Y, et al. Pennogenin tetraglycoside induces rat myometrial contraction and MLC20 phosphorylation via PLC-IP(3) and RhoA/Rho kinase signaling pathways. PLoS One. 2012;7:e51536. doi: 10.1371/journal.pone.0051536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saponara S, Fusi F, Sgaragli G, Cavalli M, Hopkins B, Bova S. Effects of commonly used protein kinase inhibitors on vascular contraction and L-type Ca2+ current. Biochem Pharmacol. 2012;84:1055–61. doi: 10.1016/j.bcp.2012.07.025. [DOI] [PubMed] [Google Scholar]
- Sakurada S, Takuwa N, Sugimoto N, Wang Y, Seto M, Sasaki Y, et al. Ca2+-dependent activation of Rho and Rho kinase in membrane depolarization-induced and receptor stimulation-induced vascular smooth muscle contraction. Circ Res. 2003;93:548–56. doi: 10.1161/01.RES.0000090998.08629.60. [DOI] [PubMed] [Google Scholar]
- Peters SL, Schmidt M, Michel MC. Rho kinase: a target for treating urinary bladder dysfunction. Trends Pharmacol Sci. 2006;27:492–7. doi: 10.1016/j.tips.2006.07.002. [DOI] [PubMed] [Google Scholar]