Abstract
Aim
The aim of this study was to establish reference intervals in healthy children for two novel urinary biomarkers of acute kidney injury, kidney injury molecule-1 (KIM-1) and neutrophil gelatinase-associated lipocalin (NGAL).
Materials & Methods
Urinary biomarkers were determined in samples from children in the UK (n = 120) and the USA (n = 171) using both Meso Scale Discovery (MSD) and Luminex-based analytical approaches.
Results
95% reference intervals for each biomarker in each cohort are presented and stratified by sex or ethnicity where necessary, and age-related variability is explored using quantile regression. We identified consistently higher NGAL concentrations in females than males (p < 0.0001), and lower KIM-1 concentrations in African–Americans than Caucasians (p = 0.02). KIM-1 demonstrated diurnal variation, with higher concentrations in the morning (p < 0.001).
Conclusion
This is the first report of reference intervals for KIM-1 and NGAL using two analytical methods in a healthy pediatric population in both UK and US-based populations.
Keywords: KIM-1, nephrotoxicity, pediatric nephrology, proximal tubule
Clinical definitions of acute kidney injury (AKI) rely on elevations in serum creatinine (sCr) [1-3], which is delayed in response to renal injury [4]. Therefore, diagnosis of AKI is frequently late and the degree of damage may be underestimated [5]. sCr is a marker of glomerular filtration and therefore not specific to damage in other parts of the nephron. Interpretation of sCr values in children is made more difficult by variation in the production of creatinine which depends on age, sex and weight (in particular, muscle mass). Furthermore, sCr values in the newborn serum initially reflect maternal values. For these reasons, there has been increasing interest in the development of novel urinary biomarkers such as kidney injury molecule-1 (Kim-1 in rodents; KIM-1 in man) and neutrophil gelatinase-associated lipo calin (NGAL). KIM-1 has been qualified by regulatory authorities to support preclinical drug safety evaluation [6]. These biomarkers are not only more sensitive than sCr for the identification of AKI but can also indicate specific damage to the proximal tubular region of the nephron [7,8]. Additionally, the ability to assay the markers noninvasively in urine represents a significant advantage over current methods, especially for the pediatric population.
We recently demonstrated the feasibility of measuring urinary KIM-1 and NGAL, and their quantitative relationship to aminogly-coside-induced nephrotoxicity in neonates [9]. However, a comprehensive understanding of changes in these biomarkers in urine is critical in order to define reference values in healthy populations to allow appropriate interpretation of novel clinical and translational data, and support their ongoing clinical qualification. Historically many pediatric reference intervals have been extrapolated from adult reference data. However, this is not appropriate when one considers the course of renal maturation and other developmental changes during childhood [10]. The aim of this study was to determine the urinary reference intervals for KIM-1 and NGAL in healthy children from both the UK and USA, and to assess the impact of factors including age, sex, ethnicity, time of day and analytical platform.
Patients & methods
Patient recruitment & sample collection
We recruited healthy children aged from birth to 16 years in Liverpool (UK) between April 2012 and February 2013. Eligible children were recruited at a number of sites, including a nursery, primary school, secondary school and elective minor surgery admissions wards at Alder Hey Children’s Hospital (Liverpool, UK). We also recruited children of staff members at Alder Hey Children’s Hospital. Children were excluded if they had a current febrile illness, any history of kidney problems or urinary tract infections, were taking medications known to cause renal problems (especially NSAIDs such as ibuprofen), had a diagnosis of cystic fibrosis (by sweat test or genotype), or had a history of exposure to aminoglycoside antibiotics within the last 3 months. (We specifically excluded children with cystic fibrosis or a history of exposure to aminoglycoside antibiotics as we aim to investigate the utility of urinary biomarkers in these patients in another study, and will use this group as a comparison). The DERIVE study received ethical approval from the National Research Ethics Service (NRES) Committee Northwest – Liverpool East, UK. It was registered on the UK Clinical Research Network portfolio (UKCRN 11810) and received support from the Medicines for Children Research Network (MCRN) and the Primary Care Research Network (PCRN). The study was conducted in accordance with the Declaration of Helsinki.
Informed written consent was obtained from carers or guardians on the behalf of the minors/children involved in our study. Those participants aged 16 years were able to consent for themselves. Consent was obtained from all children to provide one urine sample. In schools each participating child produced a urine sample during the school day, which was collected by the research team. For children attending the nursery and children of staff at Alder Hey Children’s Hospital, parents were asked to collect a morning urine sample at home and bring the sample in to the research team at the site of recruitment on the day of collection. Children recruited on the surgical admissions wards produced a urine sample before their planned surgical procedure.
In addition, some children consented to provide four further urine samples (three morning urine samples at 1-week intervals following their initial urine sample, and one bedtime urine sample collected the evening before one of the morning urine samples). Morning samples were the first micturition of the day, and the evening samples were collected just before the child went to bed and kept in the home refrigerator overnight. Parents were given sample pots to take home to collect these samples. All samples were brought on the morning of collection to the site of recruitment.
Urine samples were collected from each child by an appropriate method dependent on the age of the child. The normally preferred method was a clean catch urine sample into a sterile container. Samples were transferred to (if not already collected in) a sterile container and then centrifuged at 2000 × g for 4 min. Supernatant was aliquoted and then stored at −80°C.
Healthy pediatric subjects were recruited at Children’s Mercy Hospitals and Clinics (MO, USA) to participate in a longitudinal study investigating changes in drug metabolism activity, specifically CYP2D6 and CYP3A4 activities, through puberty using dextromethorphan as a probe. Children of both genders were eligible if they were between the ages of 7 and 16 years; a subset of the study population was diagnosed with attention deficit and hyperactivity disorder (ADHD) or attention deficit disorder (ADD), and the remainder were non-ADHD/ADD controls. Exclusion criteria included: i) current therapy with medications metabolized by or known to inhibit CYP2D6; ii) existing asthma or other respiratory diseases associated with hypercapnia; iii) history of metabolic disease, gastroesophageal reflux disease or gastrointestinal disorders; iv) demonstrated adverse reaction to previous dextromethorphan exposure; v) impaired hepatic activity as determined by routine liver function tests and physical exam; vi) pregnancy; vii) BMI less than 5th percentile.
Subjects were given a complete medical examination, including assessment of Tanner stage, and blood samples were taken for liver function testing and DNA collection at the screening visit. The parent study entitled ‘Exogenous and Endogenous Biomarkers of CYP2D6’ and secondary use of the residual urine was approved by the University of Missouri–Kansas City Health Sciences Pediatric Institutional Review Board. The parent study is also registered as registered as trial NCT01118858 at ClinicalTrials.gov. The study was conducted in accordance with the Declaration of Helsinki.
Subjects were recruited by Children’s Mercy Hospital Clinical Pharmacology staff, and parents and children were given verbal and written information about the study. If subjects agreed to participate in the study, a screening questionnaire was completed with the parents; written informed permission was obtained from the subject’s parents and assent was obtained from children >7 years of age.
Pediatric subjects were instructed to fast overnight and report to the Children’s Mercy Hospitals and Clinics the following morning for dextromethorphan phenotyping. Subjects were given a single oral dose of dextromethorphan (0.5 mg/kg as Robitussin Cough Syrup, Pfizer, Inc., NY, USA). Urine was collected predose and for 4 h following dosing by clean catch into sterile containers. At the end of the 4-h collection period, subjects were instructed to void their bladder. All urine collected during the 4-h collection interval was kept at 4°C until completion of the collection period, pooled, mixed, dispensed into multiple aliquots of various volumes, frozen and stored at −80°C. Unthawed aliquots of 4-h urine samples were shipped on dry ice to the University of Liverpool where they were stored at −80°C until analysis. The 171 samples included in this study represent the first of seven study visits at 6-month intervals for 3 years.
Determination of urinary biomarkers
Collected urine samples were thawed, mixed and centrifuged (3000 rpm, 5 min). Biomarker measurements were performed on the resulting supernatants. Urinary KIM-1 and NGAL were measured using electrochemiluminescent assays (Meso Scale Discovery [MSD], MD, USA). The MSD assays are sandwich immunoassays run on 96-well plates precoated with either a KIM-1- or NGAL-specific capture antibody, and utilize a detection antibody conjugated to an electrochemiluminescent label. The assays were run according to the manufacturer’s protocol, with a turn-around of 6 h. For KIM-1 the upper (ULOQ) and lower limits of quantitation (LLOQ) on a seven-point calibration curve were 5000 and 1.22 pg/ml, respectively. Intra- and inter-assay precision were <6 and <7%, respectively, on three plates on different occasions, recovery of spiked samples was 104–107%, and dilutional linearity was shown at 1 in 10, 1 in 100 and 1 in 1000 dilutions in the assay diluent. For NGAL the ULOQ and LLOQ on a seven-point calibration curve were 10,000 and 40.96 pg/ml, respectively. Intra- and inter-assay precision were <15 and <13%, respectively, on three plates on different occasions, recovery of spiked samples was 94–108%, and dilutional linearity was shown at 1 in 10, 1 in 100 and 1 in 1000 dilutions in the assay diluent. For both assays the lower limit of quantitation is greater than 2 standard deviations above the background value (diluent alone), and therefore the LLOQ is taken as the limit of detection. Urine samples were run in duplicate at a dilution of 1 in 10 in MSD Diluent 37, and repeated at a dilution of 1 in 100 if they remained too concentrated.
Urinary KIM-1 and NGAL measurements were also performed using microsphere-based Luminex technology, as previously validated and described, with a turnaround time of 4–6 h [9,11]. Biomarker values were normalized to urinary creatinine (uCr), which was determined spectrophotometically, as previously described [12]. Laboratory analysis was blinded to participants’ clinical characteristics.
Statistical analysis
Biomarker concentrations from the baseline urine sample produced by each participant were used to calculate the overall reference intervals following approaches recommended for pediatric reference intervals [13], using SAS version 9.3 (SAS Institute, NC, USA) and R [14]. The distributions of KIM-1 and NGAL followed a near Log normal distribution. Therefore, a logarithmic transformation was applied to the biomarker values in order to approximate a Gaussian distribution, allowing a parametric approach to analysis. Biomarker levels in the two reference populations were compared using an independent samples t-test. Reference ranges were then calculated in the two cohorts separately. The US cohort included both patients with ADHD/ADD and healthy controls. Although medications licensed for the management of ADHD/ADD do not commonly have nephrotoxic effects, we cannot exclude the possibility of rare adverse effects. An independent samples t-test was used to compare these groups. There were no differences identified between these two groups for KIM-1 or NGAL, and therefore the two groups were analyzed together.
Outliers were identified and removed according to Tukey’s method [15]. We assessed the need for partitioning of the reference population by the binary factors sex and ethnicity using independent samples t-tests. In order to assess whether reference ranges varied significantly with age, we used the method of quantile regression [16], with age as the independent variable. Quantile regression was applied either on the complete dataset, or on the dataset partitioned by sex and/or ethnicity depending on which factors were found to be statistically significant in the t-tests. Where quantiles were not found to significantly vary with age (p ≥ 0.05 in the quantile regression model), the 2.5th and 97.5th quantiles were referred to in order to calculate the 95% reference intervals for each biomarker. We also report 90% CIs for the upper limit of the reference interval [13,17]. Where quantiles were found to significantly vary with age (p < 0.05), we report the predicted values for the 2.5th and 97.5th quantiles, as obtained from the fitted quantile regression model, for particular age groups, together with 90% CIs for the upper limit.
Differences between morning and evening paired biomarker values were assessed using a paired t-test. Within-subject variation over up to four morning urine samples is measured using mean standard deviation (SD). Reference intervals for urinary creatinine were also calculated using the same statistical approach as for KIM-1 and NGAL. Differences between the MSD and Luminex methods of biomarker analysis were assessed using a regression analysis according to the method of Bland and Altman [18].
Results
A total of 120 healthy children (64 male and 56 female) recruited in the UK provided at least one urine sample as part of the study. The mean age was 9.05 years (standard deviation [SD]: 4.41), and 39 participants provided more than one sample. In parallel, one urine sample was collected from 171 healthy children (108 male and 63 female) recruited in the USA. This cohort had a narrower age range (mean age: 11.20 years; SD: 2.49) than the UK cohort and was more ethnically diverse. While we set out to recruit from birth to 16 years of age in the UK cohort, in practice only one child under 1 year of age was successfully recruited, and we have therefore presented reference intervals for children aged 1 year and above. In the US cohort 65 out of 171 (38%) were patients with ADHD/ADD, and the remainder were controls. There were no differences identified between these two groups for KIM-1 or NGAL, suggesting there is not a significant effect of the diagnosis or of medication exposure on the urinary concentrations of these biomarkers. Table 1 summarizes volunteer demographics for the two cohorts separately.
Table 1. Demographic characteristics of UK and US cohorts.
| Cohort demographics | UK cohort | US cohort |
|---|---|---|
| Number in cohort | 120 | 171 |
| Males:females (n) | 64:56 | 108:63 |
| Age 0–4 years (n) | 27 | |
| Age 5–8 years (n) | 32 | 38 |
| Age 9–12 years (n) | 32 | 85 |
| Age 13–16 years (n) | 29 | 48 |
| Caucasian (n) | 108 | 79 |
| African–American (n) | 77 | |
| Other ethnicity (n) | 12 | 15 |
The US cohort contained children aged 7–16 years only.
Reference intervals for each biomarker were determined in both cohorts. For the UK cohort, participants with non-Caucasian ethnicity (n = 12) were excluded from the calculation of reference ranges, and we were therefore unable to consider the effect of ethnicity in this cohort. For the US cohort, participants with ethnicity other than Caucasian or African–American (n = 15) were excluded. The reference intervals are presented in Table 2, stratified by ethnicity and/or sex as appropriate, and then by age. The predicted quantile values reported reflect the value for the middle age within each age group.
Table 2. Biomarker reference intervals by quantile regression.
| Cohort and biomarker | 2.5th quantile (90% CI), ng/mg Cr | 50th quantile (90% CI), ng/mg Cr | 97.5th quantile (90% CI), ng/mg Cr | CI:RI ratio |
|---|---|---|---|---|
| UK KIM-1 (MSD) Caucasians, male and female (n = 107) | ||||
| Age 1–4 years | 0.08 (0.03–0.12) | 0.46 (0.38–0.57) | 2.39 (1.96–2.91) | 0.41 |
| Age 5–8 years | 0.08 (0.03–0.12) | 0.46 (0.38–0.57) | 1.84 (1.62–2.10) | 0.27 |
| Age 9–12 years | 0.08 (0.03–0.12) | 0.46 (0.38–0.57) | 1.42 (1.22–1.65) | 0.32 |
| Age 13–16 years | 0.08 (0.03–0.12) | 0.46 (0.38–0.57) | 1.10 (0.87–1.39) | 0.51 |
| UK NGAL (MSD) Caucasians, females (n = 48) | ||||
| Age 1–4 years | 8.34 (3.14–22.15) | 10.49 (8.02–13.72) | 128.84 (54.03–307.25) | 2.10 |
| Age 5–8 years | 3.84 (2.64–5.60) | 15.30 (12.78–18.32) | 143.95 (81.17–255.29) | 1.24 |
| Age 9–12 years | 1.77 (0.95–3.31) | 22.31 (16.79–29.66) | 160.83 (104.23–248.19) | 0.91 |
| Age 13–16 years | 0.82 (0.22–3.01) | 32.55 (20.41–51.90) | 179.70 (100.73–320.58) | 1.23 |
| UK NGAL (MSD) Caucasians, males (n = 58) | ||||
| Age 1–16 years | 1.33 (0–1.89) | 5.21 (4.66–6.49) | 64.59 (29.14–Inf)† | Inf |
| UK KIM-1 (Luminex) Caucasians, male and female (n = 108) | ||||
| Age 1–16 years | 0.03 (0.002–0.04) | 0.17 (0.15–0.18) | 0.63 (0.49–Inf)† | Inf |
| UK NGAL (Luminex) Caucasians, females (n = 50) | ||||
| Age 1–4 years | 1.25 (0.11–14.06) | 63.43 (38.09–83.93) | 706.72 (247.84–2015.28) | 2.51 |
| Age 5–8 years | 2.72 (0.92–8.06) | 63.43 (38.09–83.93) | 581.69 (297.53–1137.25) | 1.45 |
| Age 9–12 years | 5.95 (2.41–14.70) | 63.43 (38.09–83.93) | 478.78 (327.30–700.38) | 0.79 |
| Age 13–16 years | 13.00 (1.46–116.09) | 63.43 (38.09–83.93) | 394.08 (256.72–604.94) | 0.91 |
| UK NGAL (Luminex) Caucasians, males (n = 58) | ||||
| Age 1–4 years | 0.36 (0–0.58) | 6.30 (4.53–10.18) | 229.90 (118.14–447.38) | 1.43 |
| Age 5–8 years | 0.36 (0–0.58) | 6.30 (4.53–10.18) | 180.51 (114.13–285.51) | 0.95 |
| Age 9–12 years | 0.36 (0–0.58) | 6.30 (4.53–10.18) | 141.73 (103.94–193.27) | 0.63 |
| Age 13–16 years | 0.36 (0–0.58) | 6.30 (4.53–10.18) | 111.28 (80.96–152.95) | 0.65 |
| US KIM-1 (MSD) Caucasians, male and female (n = 73) | ||||
| Age 7–8 years | 0.014 (0.007–0.032) | 0.23 (0.19–0.29) | 0.79 (0.62–Inf)† | Inf |
| Age 9–12 years | 0.031 (0.022–0.044) | 0.23 (0.19–0.29) | 0.79 (0.62–Inf)† | Inf |
| Age 13–16 years | 0.088 (0.051–0.151) | 0.23 (0.19–0.29) | 0.79 (0.62–Inf)† | Inf |
| US KIM-1 (MSD) African–American, male and female (n = 65) | ||||
| Age 7–8 years | 0.038 (0.018–0.077) | 0.17 (0.13–0.19) | 0.42 (0.34–Inf)† | Inf |
| Age 9–12 years | 0.048 (0.035–0.066) | 0.17 (0.13–0.19) | 0.42 (0.34–Inf)† | Inf |
| Age 13–16 years | 0.067 (0.043–0.106) | 0.17 (0.13–0.19) | 0.42 (0.34–Inf)† | Inf |
| US NGAL (MSD), females (n = 55) | ||||
| Age 7–8 years | 1.30 (0–4.07) | 19.30 (12.36–30.14) | 58.26 (42.06–80.70) | 0.68 |
| Age 9–12 years | 1.30 (0–4.07) | 35.38 (28.40–44.09) | 96.95 (82.72–113.64) | 0.32 |
| Age 13–16 years | 1.30 (0–4.07) | 79.40 (49.91–126.32) | 191.21 (137.58–265.74) | 0.67 |
| US NGAL (MSD), males (n = 90) | ||||
| Age 7–8 years | 1.34 (0–1.50) | 3.10 (2.55–3.77) | 8.28 (6.34–10.81) | 0.64 |
| Age 9–12 years | 1.34 (0–1.50) | 4.23 (3.63–4.95) | 10.38 (8.83–12.20) | 0.37 |
| Age 13–16 years | 1.34 (0–1.50) | 6.42 (4.56–9.03) | 14.02 (10.67–18.42) | 0.61 |
95% RIs (2.5th and 97.5th quantiles) along with median values (50th quantile) and their 90% CIs were estimated using quantile regression methodology. RIs are reported by cohort (UK or USA), biomarker (KIM-1 or NGAL) and analytical method (MSD or Luminex). Where biomarker concentrations were significantly associated with sex or ethnicity (p < 0.05), RIs are presented by these partitions. We report quantile values for the middle age within each age group. A ratio of the 90% CI for the 97.5th quantile to the 95% RI (CI:RI ratio) is given. The US Cohort contained children aged 7–16 years only.
For RIs where there was no significant association with age, it was not possible to calculate upper limits for the CIs due to small sample size and the extreme percentile.
CI: Confidence interval; Cr: Creatinine; Inf: Infinity; MSD: Meso Scale Discovery; RI: Reference interval.
Using MSD, there was a significant difference (p < 0.001) in the mean urinary KIM-1 concentration between the two cohorts (UK [MSD]: 0.43 ng/mg Cr, 95% CI: 0.37–0.50 ng/mg Cr; US [MSD]: 0.18 ng/mg Cr, 95% CI: 0.16–0.20 ng/mg Cr). However, there was no difference between the mean NGAL concentrations (UK [MSD]: 10.71 ng/mg Cr, 95% CI: 8.76–13.11 ng/mg Cr; US (MSD): 8.19 ng/mg Cr, 95% CI: 6.80-9.86 ng/mg Cr).
Within both cohorts, urinary KIM-1 concentrations were not significantly associated with sex. However, we consistently identified a significant difference (p < 0.0001) in mean NGAL concentration between males and females in both cohorts (UK males [MSD]: 6.55 ng/mg Cr, 95% CI: 5.16-8.32 ng/mg Cr; UK females [MSD]: 19.18 ng/mg Cr, 95% CI: 14.63-25.14 ng/mg Cr; US males [MSD]: 3.95 ng/mg Cr, 95% CI: 3.53–4.42 ng/mg Cr; US females [MSD]: 28.56 ng/mg Cr, 95% CI: 22.09–36.93 ng/mg Cr).
The US cohort was more ethnically diverse, with 77 subjects of African-American descent, 79 Caucasians and 15 of dual heritage (although these 15 were excluded from the analysis). Mean NGAL concentrations were not significantly associated with ethnicity. However, for KIM-1 a significant difference was found between Caucasian and African-American ethnic groups (Caucasians [MSD]: 0.21 ng/mg Cr, 95% CI: 0.17–0.26 ng/mg Cr; African–Americans [MSD]: 0.16 ng/mg Cr, 95% CI: 0.13–0.18 ng/mg Cr; p = 0.02).
Using MSD, we assessed intraindividual variability in the UK cohort in 39 individuals who provided one morning sample each week for up to 4 weeks. KIM-1 was less variable within individuals than NGAL (KIM-1 intraindividual SD: 0.16 ng/mg Cr, 95% CI: 0.12–0.22 ng/mg Cr; NGAL intraindividual SD: 3.84 ng/mg Cr; 95% CI: 2.57–5.74 ng/mg Cr). Diurnal effects were assessed using paired urine samples collected in the evening and the following morning in the UK cohort. For KIM-1 (n = 35) there was a significant diurnal variation (p < 0.001) with lower mean concentration in the evening (0.47 ng/mg Cr; 95% CI: 0.36–0.62 ng/mg Cr) compared with the morning (0.77 ng/mg Cr, 95% CI: 0.61–0.97 ng/mg Cr). No diurnal effect was seen for NGAL (n = 36; mean evening concentration: 7.35 ng/mg Cr, 95% CI: 4.96–10.89 ng/mg Cr; mean morning concentration: 6.77 ng/mg Cr, 95% CI: 4.85–9.46 ng/mg Cr; p = 0.36).
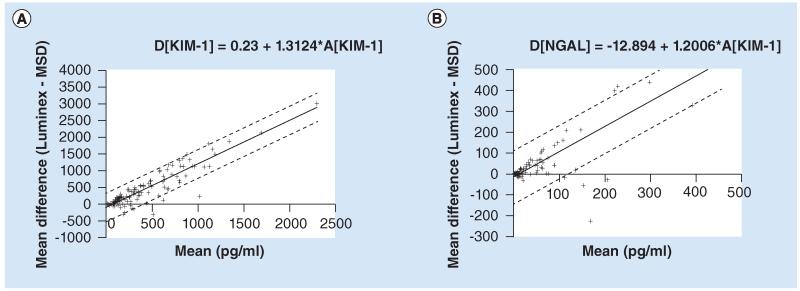
Reference intervals for urinary creatinine were calculated as above utilizing a quantile regression approach, and are given in Table 3. Both the 2.5th and 97.5th quantiles show a significant increase with age. Differences between the MSD and Luminex methods of biomarker analysis were assessed. For both markers there was an increase in the difference in biomarker value as the magnitude of the measurement increased. A regression model was used, first to regress the difference between the methods on the average of the two methods, and then to regress the values of the residuals (Figure 1). Using this approach, the difference (D[KIM-1], pg/ml) between the two methods for KIM-1 (Luminex minus MSD) is described by the equation D[KIM-1] = −105.23 + 1.3124*A[KIM-1], where A[KIM-1] is the mean of the two methods (in pg/ml). 95% limits of agreement are given by the equation D[KIM-1] ± 1.96*216.7. The difference (D[N-GAL], pg/ml) between the two methods for NGAL (Luminex minus MSD) is described by the equation D[NGAL] = −12.894 + 1.2006*A[NGAL], where A[NGAL] is the mean of the two methods (in pg/ml). 95% limits of agreement are given by the equation D[NGAL] ± 1.96*65.44.
Table 3. Reference intervals of urinary creatinine (n = 106).
| Reference interval | 2.5th quantile (90% CI), ng/mg Cr | 50th quantile (90% CI), ng/mg Cr | 97.5th quantile (90% CI), ng/mg Cr | CI:RI ratio |
|---|---|---|---|---|
| Urinary Cr (mg/ml) | ||||
| Age 1–4 years | 0.47 (0.19–0.74) | 0.71 (0.54–0.93) | 1.59 (1.21–2.09) | 0.78 |
| Age 5–8 years | 0.69 (0.53–0.86) | 0.97 (0.81–1.16) | 2.00 (1.70–2.36) | 0.51 |
| Age 9–12 years | 0.92 (0.80–1.04) | 1.32 (1.12–1.56) | 2.52 (2.24–2.84) | 0.38 |
| Age 13–16 years | 1.15 (0.97–1.34) | 1.80 (1.41–2.31) | 3.17 (2.63–3.81) | 0.59 |
95% RIs along with median values and their 90% CIs for urinary creatinine in the UK cohort were derived using quantile regression methodology.
CI: Confidence interval; Cr: Creatinine; RI: Reference interval.
Figure 1. Agreement of Meso Scale Discovery and Luminex methods for analysis of urinary KIM-1 and NGAL.
(A)KIM-1; (B) NGAL. Regression models describing the differences between the Luminex and MSD methods of biomarker analysis. The regression line is given using a solid line, and the upper and lower 95% limits of agreement with dashed lines.
MSD: Meso Scale Discovery.
Discussion
The International Federation of Clinical Chemistry (IFCC) [19] recommends that samples from 120 or more individuals are required for the development of reference intervals for analytes. In this study we were able to meet this figure, although larger numbers would improve the confidence of the reference intervals we have derived (recommended CI:RI ratio is less than 0.2) [13]. We felt it was important to present reference intervals by partitions, where they are significant, in order to give a true picture of our data. However, partitioning the group by sex or ethnicity leads to a loss of confidence, and, in some cases, to very wide confidence intervals. This is a limitation of our study, but was a necessary balance against the difficulty, length of time and investment that would be required for a much larger study. While we set out to recruit from birth to 16 years of age in the UK cohort, in practice only one child under 1 year of age was successfully recruited, and we have therefore presented reference intervals for children aged 1 year and above. Another study will be required to develop useful reference intervals for neonates/infants that take into account the rapid maturation and changes in renal function that occur during this period. We normalized biomarker values to urinary creatinine, but urinary creatinine concentration itself demonstrates age-related changes (Table 3) [20,21], and there is ongoing debate as to the most appropriate standard [22].
In both cohorts, there was a significantly higher mean NGAL concentration in females compared with males. This gender difference has also been observed in adults [23], children [24] and in very low birth weight infants [25,26], although the reasons for the difference remain unclear. One recent study in children, using a different NGAL assay, did not demonstrate this gender difference, although the NGAL values they report are similar to those we found with the MSD assay [27]. We found that for both biomarkers concentration was related to age, as has been previously reported [24]. However, age-related changes were not consistent for each biomarker across both cohorts and analytical platforms, and these results must be interpreted with caution. Significant differences in mean values of KIM-1 were observed between the two cohorts. Factors contributing to this difference may be ethnicity and the time of day of sample collection, as we have shown, for the first time, diurnal variation in KIM-1 concentration with lower values in the evening. The reference intervals for the UK cohort are based on samples collected in the morning, whereas the US samples were collected at varying times of day. Furthermore, urine samples for the UK study were spot collections, whereas in the US cohort they were 4-h collections, although it is unclear whether this would make a difference.
An important aspect for the rolling qualification of novel urinary biomarkers and for their eventual use in man is that independent and robust methodologies are developed for their quantification. KIM-1 and NGAL have primarily been quantified in man via Luminex-based technologies. In the UK cohort we compared this method to a MSD-based method for the quantification of both KIM-1 and NGAL. Although absolute differences in biomarker values were observed, due to differences in the antibodies used and their affinities for the biomarker of interest, there remained strong agreement between both analytical platforms.
Conclusion & future perspective
We have reported reference intervals for two urinary renal injury biomarkers, KIM-1 and NGAL, for the first time in two independent healthy pediatric populations, and have shown that they can be robustly quantified on two independent analytical platforms with a high degree of agreement. This represents a key step in the qualification of these two biomarkers for use in children. A number of groups are currently working on identifying clinical scenarios in which these biomarkers hold diagnostic or prognostic advantages over traditional measures of kidney injury. A key strength over traditional markers lies in their specificity for damage to certain portions of the kidney nephron. This may be particularly advantageous in the early identification of drug-induced kidney injury, which commonly starts with localized injury. While these novel biomarkers may have increased sensitivity and specificity when compared with traditional measures of AKI, in order to provide clinical benefit these diagnostic tools need to be followed by improved interventions, leading to better patient outcomes. Earlier diagnosis may allow earlier interventions, and future studies will need to assess whether interventions made on the basis of changes in novel biomarkers lead to improved outcomes. It will only be when this link is established that novel biomarkers will become routinely used in clinical practice.
Executive summary.
Background
There is increasing interest in a number of novel urinary biomarkers that are not only more sensitive than serum creatinine for the identification of acute kidney injury, but can also indicate the site of damage to different portions of the nephron.
The aim of this study was to describe reference intervals in healthy children for two of these biomarkers, urinary KIM-1 and NGAL.
Methods
Reference intervals were defined in cohorts of healthy children in the UK (n = 120) and the USA (n = 171).
Results
Concentrations of urinary KIM-1 varied with age and with ethnicity, and showed diurnal variation, with higher concentrations in the morning.
In both cohorts, there was a significantly higher mean NGAL concentration in females compared with males. NGAL concentration also varied with age.
We compared the Meso Scale Discovery (MSD) and Luminex methods of biomarker analysis. Although absolute differences in biomarker values were observed, there was strong agreement between both analytical platforms.
Conclusion
The definition of healthy pediatric reference intervals is an important step in the qualification of KIM-1 and NGAL for use in children to identify acute kidney injury.
Acknowledgements
The authors would like to thank the UK Medicines for Children Research Network (MCRN) and Primary Care Research Network (PCRN) for their support of this study. In particular, the authors would like to thank Naomi Wallin for her contribution to recruitment and sample collection in the DERIVE cohort. We acknowledge statistical advice from Steven Lane and Laura Sutton.
Footnotes
Financial & competing interests disclosure
JV Bonventre is co-inventor on KIM-1 patents, which have been licensed by Partners Healthcare to a number of companies. He has received royalty income from Partners Healthcare and grant funding from Novo Nordisk. JV Bonventre or his family have received income for consulting from companies interested in biomarkers: Sekisui, Millenium, Johnson & Johnson and Novartis. M Pirmohamed is a NIHR Senior Investigator. SJ McWilliam is a MRC Clinical Training Fellow supported by the North West England Medical Research Council Fellowship Scheme in Clinical Pharmacology and Therapeutics, which is funded by the Medical Research Council (grant no: G1000417/94909), ICON, GlaxoSmithKline, AstraZeneca and the Medical Evaluation Unit. DJ Antoine would like to acknowledge financial support from a Royal Society International Travelling Research Fellowship and the Wellcome Trust. Urine samples used in this study and collected at Children’s Mercy Hospitals and Clinics were part of a research study supported by NIH grant R01 058556 (JS Leeder and Yvonne Lin, co-Principal Investigators). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
References
Papers of special note have been highlighted as:
• of interest;
•• of considerable interest
- 1.Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL. Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int. 2007;71(10):1028–1035. doi: 10.1038/sj.ki.5002231. [DOI] [PubMed] [Google Scholar]
- 2.Mehta RL, Kellum JA, Shah SV, et al. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit. Care. 2007;11(2):R31. doi: 10.1186/cc5713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Moffett BS, Goldstein SL. Acute kidney injury and increasing nephrotoxic-medication exposure in noncriticallyill children. Clin. J. Am. Soc. Nephrol. 2011;6(4):856–863. doi: 10.2215/CJN.08110910. [• Demonstrates the epidemiology of drug-induced acute kidney injury in a pediatric center, and highlights the importance of improved early diagnostic tools for this population.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Askenazi DJ, Ambalavanan N, Goldstein SL. Acute kidney injury in critically ill newborns: what do we know? What do we need to learn? Pediatr. Nephrol. 2009;24(2):265–274. doi: 10.1007/s00467-008-1060-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Waikar SS, Bonventre JV. Creatinine kinetics and the definition of acute kidney injury. J. Am. Soc. Nephrol. 2009;20(3):672–679. doi: 10.1681/ASN.2008070669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dieterle F, Sistare F, Goodsaid F, et al. Renal biomarker qualification submission: a dialog between the FDA-EMEA and Predictive Safety Testing Consortium. Nat. Biotechnol. 2010;28(5):455–462. doi: 10.1038/nbt.1625. [• Outlines the qualification of seven renal safety biomarkers following the work of the Predictive Safety Testing Consortium’s (PSTC’s) Nephrotoxicity Working Group.] [DOI] [PubMed] [Google Scholar]
- 7.Bonventre JV. Diagnosis of acute kidney injury: from classic parameters to new biomarkers. Contrib. Nephrol. 2007;156:213–219. doi: 10.1159/000102086. [DOI] [PubMed] [Google Scholar]
- 8.Coca SG, Yalavarthy R, Concato J, Parikh CR. Biomarkers for the diagnosis and risk stratification of acute kidney injury: a systematic review. Kidney Int. 2008;73(9):1008–1016. doi: 10.1038/sj.ki.5002729. [DOI] [PubMed] [Google Scholar]
- 9.Mcwilliam SJ, Antoine DJ, Sabbisetti V, et al. Mechanism-based urinary biomarkers to identify the potential for aminoglycoside-induced nephrotoxicity in premature neonates: a proof-of-concept study. PLoS ONE. 2012;7(8):e43809. doi: 10.1371/journal.pone.0043809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Goldman J, Becker ML, Jones B, Clements M, Leeder JS. Development of biomarkers to optimize pediatric patient management: what makes children different? Biomarkers Med. 2011;5(6):781–794. doi: 10.2217/bmm.11.96. [•• Biomarkers for use in the pediatric population should be developed and validated in children, not extrapolated from adult data, in order to take account of child-specific pathogenesis and the effects of ontogeny.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vaidya VS, Waikar SS, Ferguson MA, et al. Urinary biomarkers for sensitive and specific detection of acute kidney injury in humans. Clin. Transl. Sci. 2008;1(3):200–208. doi: 10.1111/j.1752-8062.2008.00053.x. [• Outlines the validation of the Luminex assays.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Waikar SS, Sabbisetti VS, Bonventre JV. Normalization of urinary biomarkers to creatinine during changes in glomerular filtration rate. Kidney Int. 2010;78(5):486–494. doi: 10.1038/ki.2010.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Daly CH, Liu X, Grey VL, Hamid JS. A systematic review of statistical methods used in constructing pediatric reference intervals. Clin. Biochem. 2013;46(13-14):122–1227. doi: 10.1016/j.clinbiochem.2013.05.058. [• Systematic review of statistical methodology in publications of pediatric reference intervals, which addresses both general methodological issues and pediatric-specific issues.] [DOI] [PubMed] [Google Scholar]
- 14.R Development Core Team . R. A language and environment for statistical computing. R Foundation for Statistical Computing; Vienna, Austria: 2011. www.R-project.org ISBN: 3-900051-07-0. [Google Scholar]
- 15.Tukey JW. Exploratory Data Analysis. Vol. 688. Addison-Wesley; MA, USA: 1977. [Google Scholar]
- 16.Koenker R, Hallock KF. Quantile regression. J. Econ. Perspect. 2001;15(4):143–156. [Google Scholar]
- 17.Shaw JLV, Binesh Marvasti T, Colantonio D, Adeli K. Pediatric reference intervals: challenges and recent initiatives. Crit. Rev. Clin. Lab. Sci. 2013;50(2):37–50. doi: 10.3109/10408363.2013.786673. [• Useful review and description of methodology in determination of pediatric reference intervals.] [DOI] [PubMed] [Google Scholar]
- 18.Bland JM, Altman DG. Measuring agreement in method comparison studies. Stat. Methods Med. Res. 1999;8(2):135–160. doi: 10.1177/096228029900800204. [DOI] [PubMed] [Google Scholar]
- 19.Hyltoft P. Prerequisites for establishing common reference intervals. Scand. J. Clin. Lab. Invest. 2004;64:285–292. doi: 10.1080/00365510410006298. [DOI] [PubMed] [Google Scholar]
- 20.Barr DB, Wilder LC, Caudill SP, Gonzalez AJ, Needham LL, Pirkle JL. Urinary creatinine concentrations in the U.S. population: implications for urinary biologic monitoring measurements. Environ. Health Perspect. 2005;113(2):192–200. doi: 10.1289/ehp.7337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martin MD, Woods JS, Leroux BG, et al. Longitudinal urinary creatinine excretion values among preadolescents and adolescents. Transl. Res. 2008;151(1):51–56. doi: 10.1016/j.trsl.2007.10.001. [DOI] [PubMed] [Google Scholar]
- 22.Goldstein SL. Urinary kidney injury biomarkers and urine creatinine normalization: a false premise or not. Kidney Int. 2010;78(5):433–435. doi: 10.1038/ki.2010.200. [DOI] [PubMed] [Google Scholar]
- 23.Cullen MR, Murray PT, Fitzgibbon MC. Establishment of a reference interval for urinary neutrophil gelatinase-associated lipocalin. Ann. Clin. Biochem. 2012;49(2):190–193. doi: 10.1258/acb.2011.011105. [DOI] [PubMed] [Google Scholar]
- 24.Pennemans V, Rigo JM, Faes C, Reynders C, Penders J, Swennen Q. Establishment of reference values for novel urinary biomarkers for renal damage in the healthy population: are age and gender an issue? Clin. Chem. Lab. Med. 2013;51(9):1795–1802. doi: 10.1515/cclm-2013-0157. [DOI] [PubMed] [Google Scholar]
- 25.Huynh TK, Bateman DA, Parravicini E, et al. Reference values of urinary neutrophil gelatinase-associated lipocalin in very low birth weight infants. Pediatr. Res. 2009;66(5):528–532. doi: 10.1203/PDR.0b013e3181baa3dd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Askenazi DJ, Koralkar R, Levitan EB, et al. Baseline values of candidate urine acute kidney injury biomarkers vary by gestational age in premature infants. Pediatr. Res. 2011;70(3):302–306. doi: 10.1203/PDR.0b013e3182275164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cangemi GSS, Cantinotti M, Fortunato A, et al. Reference values for urinary neutrophil gelatinase-associated lipocalin (NGAL) in pediatric age measured with a fully automated chemiluminescent platform. Clin. Chem. Lab. Med. 2013;51(5):1101–1105. doi: 10.1515/cclm-2012-0540. [DOI] [PubMed] [Google Scholar]