Abstract
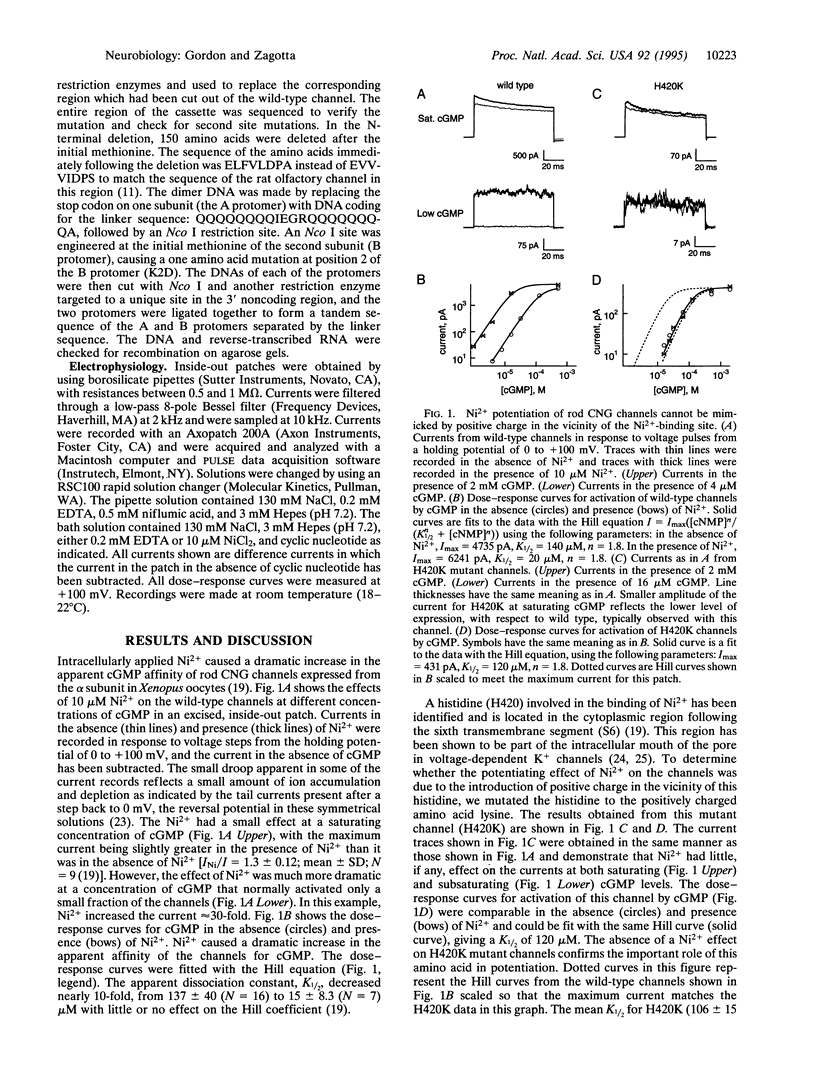
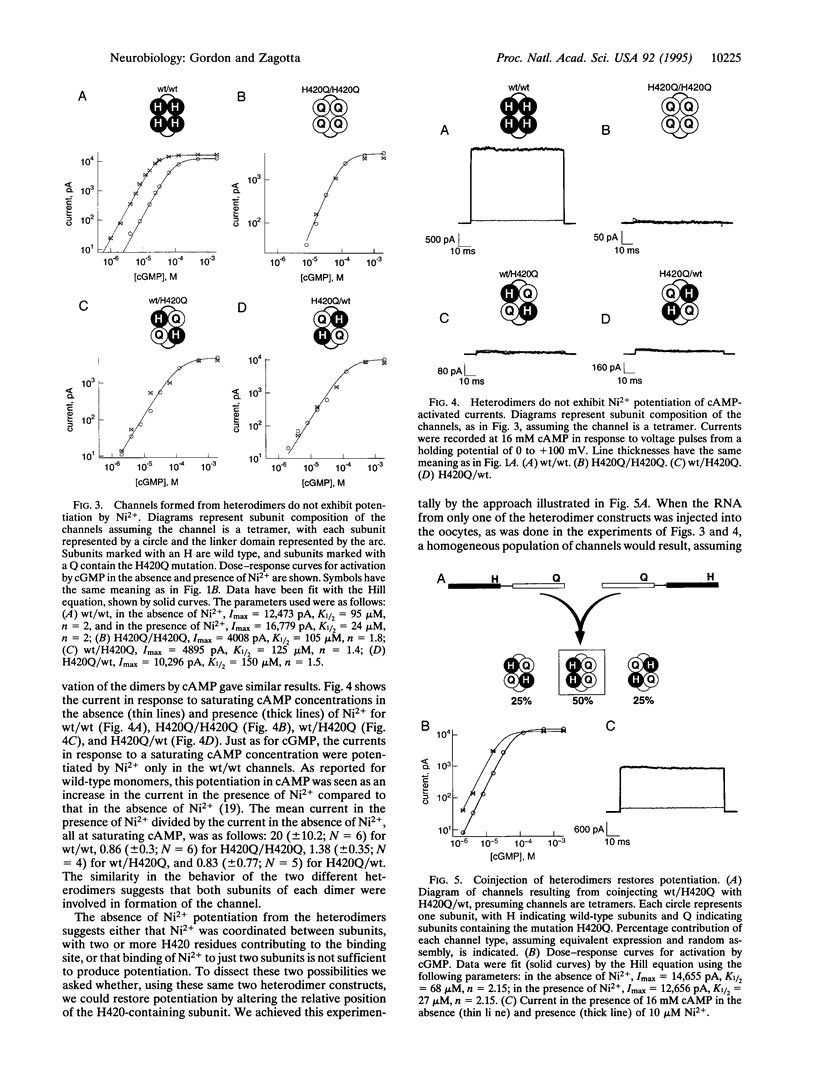
Cyclic nucleotide-gated (CNG) channels present a unique model for studying the molecular mechanisms of channel gating. We have studied the mechanism of potentiation of expressed rod CNG channels by Ni2+ as a first step toward understanding the channel gating process. Here we report that coordination of Ni2+ between histidine residues (H420) on adjacent channel subunits occurs when the channels are open. Mutation of H420 to lysine completely eliminated the potentiation by Ni2+ but did not markedly alter the apparent cGMP affinity of the channel, indicating that the introduction of positive charge at the Ni(2+)-binding site was not sufficient to produce potentiation. Deletion or mutation of most of the other histidines present in the channel did not diminish potentiation by Ni2+. We studied the role of subunit interactions in Ni2+ potentiation by generating heteromultimeric channels using tandem dimers of the rod CNG channel sequence. Injection of single heterodimers in which one subunit contained H420 and the other did not (wt/H420Q or H420Q/wt) resulted in channels that were not potentiated by Ni2+. However, coinjection of both heterodimers into Xenopus oocytes resulted in channels that exhibited potentiation. The H420 residues probably occurred predominantly in nonadjacent subunits when each heterodimer was injected individually, but, when the two heterodimers were coinjected, the H420 residues could occur in adjacent subunits as well. These results suggest that the mechanism of Ni2+ potentiation involves intersubunit coordination of Ni2+ by H420. Based on the preferential binding of Ni2+ to open channels, we suggest that alignment of H420 residues of neighboring subunits into the Ni(2+)-coordinating position may be associated with channel opening.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Biel M., Altenhofen W., Hullin R., Ludwig J., Freichel M., Flockerzi V., Dascal N., Kaupp U. B., Hofmann F. Primary structure and functional expression of a cyclic nucleotide-gated channel from rabbit aorta. FEBS Lett. 1993 Aug 23;329(1-2):134–138. doi: 10.1016/0014-5793(93)80209-d. [DOI] [PubMed] [Google Scholar]
- Biel M., Zong X., Distler M., Bosse E., Klugbauer N., Murakami M., Flockerzi V., Hofmann F. Another member of the cyclic nucleotide-gated channel family, expressed in testis, kidney, and heart. Proc Natl Acad Sci U S A. 1994 Apr 26;91(9):3505–3509. doi: 10.1073/pnas.91.9.3505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley J., Li J., Davidson N., Lester H. A., Zinn K. Heteromeric olfactory cyclic nucleotide-gated channels: a subunit that confers increased sensitivity to cAMP. Proc Natl Acad Sci U S A. 1994 Sep 13;91(19):8890–8894. doi: 10.1073/pnas.91.19.8890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown R. L., Gramling R., Bert R. J., Karpen J. W. Cyclic GMP contact points within the 63-kDa subunit and a 240-kDa associated protein of retinal rod cGMP-activated channels. Biochemistry. 1995 Jul 4;34(26):8365–8370. doi: 10.1021/bi00026a018. [DOI] [PubMed] [Google Scholar]
- Catterall W. A. Structure and function of voltage-gated sodium and calcium channels. Curr Opin Neurobiol. 1991 Jun;1(1):5–13. doi: 10.1016/0959-4388(91)90004-q. [DOI] [PubMed] [Google Scholar]
- Chakrabarti P. Geometry of interaction of metal ions with histidine residues in protein structures. Protein Eng. 1990 Oct;4(1):57–63. doi: 10.1093/protein/4.1.57. [DOI] [PubMed] [Google Scholar]
- Chen T. Y., Peng Y. W., Dhallan R. S., Ahamed B., Reed R. R., Yau K. W. A new subunit of the cyclic nucleotide-gated cation channel in retinal rods. Nature. 1993 Apr 22;362(6422):764–767. doi: 10.1038/362764a0. [DOI] [PubMed] [Google Scholar]
- Dhallan R. S., Yau K. W., Schrader K. A., Reed R. R. Primary structure and functional expression of a cyclic nucleotide-activated channel from olfactory neurons. Nature. 1990 Sep 13;347(6289):184–187. doi: 10.1038/347184a0. [DOI] [PubMed] [Google Scholar]
- Dryer S. E., Henderson D. A cyclic GMP-activated channel in dissociated cells of the chick pineal gland. Nature. 1991 Oct 24;353(6346):756–758. doi: 10.1038/353756a0. [DOI] [PubMed] [Google Scholar]
- Fesenko E. E., Kolesnikov S. S., Lyubarsky A. L. Induction by cyclic GMP of cationic conductance in plasma membrane of retinal rod outer segment. Nature. 1985 Jan 24;313(6000):310–313. doi: 10.1038/313310a0. [DOI] [PubMed] [Google Scholar]
- Gordon S. E., Zagotta W. N. A histidine residue associated with the gate of the cyclic nucleotide-activated channels in rod photoreceptors. Neuron. 1995 Jan;14(1):177–183. doi: 10.1016/0896-6273(95)90252-x. [DOI] [PubMed] [Google Scholar]
- Gordon S. E., Zagotta W. N. Localization of regions affecting an allosteric transition in cyclic nucleotide-activated channels. Neuron. 1995 Apr;14(4):857–864. doi: 10.1016/0896-6273(95)90229-5. [DOI] [PubMed] [Google Scholar]
- Heginbotham L., MacKinnon R. The aromatic binding site for tetraethylammonium ion on potassium channels. Neuron. 1992 Mar;8(3):483–491. doi: 10.1016/0896-6273(92)90276-j. [DOI] [PubMed] [Google Scholar]
- Ildefonse M., Crouzy S., Bennett N. Gating of retinal rod cation channel by different nucleotides: comparative study of unitary currents. J Membr Biol. 1992 Oct;130(1):91–104. doi: 10.1007/BF00233741. [DOI] [PubMed] [Google Scholar]
- Isacoff E. Y., Jan Y. N., Jan L. Y. Evidence for the formation of heteromultimeric potassium channels in Xenopus oocytes. Nature. 1990 Jun 7;345(6275):530–534. doi: 10.1038/345530a0. [DOI] [PubMed] [Google Scholar]
- Jan L. Y., Jan Y. N. A superfamily of ion channels. Nature. 1990 Jun 21;345(6277):672–672. doi: 10.1038/345672a0. [DOI] [PubMed] [Google Scholar]
- Karpen J. W., Brown R. L., Stryer L., Baylor D. A. Interactions between divalent cations and the gating machinery of cyclic GMP-activated channels in salamander retinal rods. J Gen Physiol. 1993 Jan;101(1):1–25. doi: 10.1085/jgp.101.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaupp U. B., Niidome T., Tanabe T., Terada S., Bönigk W., Stühmer W., Cook N. J., Kangawa K., Matsuo H., Hirose T. Primary structure and functional expression from complementary DNA of the rod photoreceptor cyclic GMP-gated channel. Nature. 1989 Dec 14;342(6251):762–766. doi: 10.1038/342762a0. [DOI] [PubMed] [Google Scholar]
- Kavanaugh M. P., Hurst R. S., Yakel J., Varnum M. D., Adelman J. P., North R. A. Multiple subunits of a voltage-dependent potassium channel contribute to the binding site for tetraethylammonium. Neuron. 1992 Mar;8(3):493–497. doi: 10.1016/0896-6273(92)90277-k. [DOI] [PubMed] [Google Scholar]
- Kumar V. D., Weber I. T. Molecular model of the cyclic GMP-binding domain of the cyclic GMP-gated ion channel. Biochemistry. 1992 May 19;31(19):4643–4649. doi: 10.1021/bi00134a015. [DOI] [PubMed] [Google Scholar]
- Liman E. R., Buck L. B. A second subunit of the olfactory cyclic nucleotide-gated channel confers high sensitivity to cAMP. Neuron. 1994 Sep;13(3):611–621. doi: 10.1016/0896-6273(94)90029-9. [DOI] [PubMed] [Google Scholar]
- Liman E. R., Tytgat J., Hess P. Subunit stoichiometry of a mammalian K+ channel determined by construction of multimeric cDNAs. Neuron. 1992 Nov;9(5):861–871. doi: 10.1016/0896-6273(92)90239-a. [DOI] [PubMed] [Google Scholar]
- Lopez G. A., Jan Y. N., Jan L. Y. Evidence that the S6 segment of the Shaker voltage-gated K+ channel comprises part of the pore. Nature. 1994 Jan 13;367(6459):179–182. doi: 10.1038/367179a0. [DOI] [PubMed] [Google Scholar]
- MacKinnon R. Determination of the subunit stoichiometry of a voltage-activated potassium channel. Nature. 1991 Mar 21;350(6315):232–235. doi: 10.1038/350232a0. [DOI] [PubMed] [Google Scholar]
- Nakamura T., Gold G. H. A cyclic nucleotide-gated conductance in olfactory receptor cilia. 1987 Jan 29-Feb 4Nature. 325(6103):442–444. doi: 10.1038/325442a0. [DOI] [PubMed] [Google Scholar]
- Pascual J. M., Shieh C. C., Kirsch G. E., Brown A. M. K+ pore structure revealed by reporter cysteines at inner and outer surfaces. Neuron. 1995 May;14(5):1055–1063. doi: 10.1016/0896-6273(95)90344-5. [DOI] [PubMed] [Google Scholar]
- Root M. J., MacKinnon R. Two identical noninteracting sites in an ion channel revealed by proton transfer. Science. 1994 Sep 23;265(5180):1852–1856. doi: 10.1126/science.7522344. [DOI] [PubMed] [Google Scholar]
- Taglialatela M., Champagne M. S., Drewe J. A., Brown A. M. Comparison of H5, S6, and H5-S6 exchanges on pore properties of voltage-dependent K+ channels. J Biol Chem. 1994 May 13;269(19):13867–13873. [PubMed] [Google Scholar]
- Tsien R. W., Ellinor P. T., Horne W. A. Molecular diversity of voltage-dependent Ca2+ channels. Trends Pharmacol Sci. 1991 Sep;12(9):349–354. doi: 10.1016/0165-6147(91)90595-j. [DOI] [PubMed] [Google Scholar]
- Tytgat J., Hess P. Evidence for cooperative interactions in potassium channel gating. Nature. 1992 Oct 1;359(6394):420–423. doi: 10.1038/359420a0. [DOI] [PubMed] [Google Scholar]
- Weyand I., Godde M., Frings S., Weiner J., Müller F., Altenhofen W., Hatt H., Kaupp U. B. Cloning and functional expression of a cyclic-nucleotide-gated channel from mammalian sperm. Nature. 1994 Apr 28;368(6474):859–863. doi: 10.1038/368859a0. [DOI] [PubMed] [Google Scholar]
- Zagotta W. N., Hoshi T., Aldrich R. W. Gating of single Shaker potassium channels in Drosophila muscle and in Xenopus oocytes injected with Shaker mRNA. Proc Natl Acad Sci U S A. 1989 Sep;86(18):7243–7247. doi: 10.1073/pnas.86.18.7243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmerman A. L., Baylor D. A. Cyclic GMP-sensitive conductance of retinal rods consists of aqueous pores. Nature. 1986 May 1;321(6065):70–72. doi: 10.1038/321070a0. [DOI] [PubMed] [Google Scholar]
- Zimmerman A. L., Karpen J. W., Baylor D. A. Hindered diffusion in excised membrane patches from retinal rod outer segments. Biophys J. 1988 Aug;54(2):351–355. doi: 10.1016/S0006-3495(88)82966-7. [DOI] [PMC free article] [PubMed] [Google Scholar]



