Abstract
We examined superior mesenteric artery (SMA) blood flow velocity (BFV) in response to feeding in babies randomized to trophic feeds (n=16) or nil per os (NPO, n=18) during prior treatment for patent ductus arteriosus. BFV increased earlier in fed babies, but was similar between groups at 30 minutes post-feed.
Keywords: not in title: Premature Infants
The medical literature offers very little insight into whether it is safe to feed infants during treatment of a patent ductus arteriosus (PDA). One reason for not feeding is the possibility that the PDA itself1 and the medications used to treat the PDA2 may reduce superior mesenteric artery (SMA) blood flow and impair the ability of the infant to mount the appropriate postprandial hyperemia3 (the increase in SMA blood flow that follows a bolus feeding and is thought to enable normal digestion). On the other hand, withholding enteral nutrition for even short periods of time potentially may result in gut atrophy4 and impair digestive processes. We recently conducted a randomized controlled trial which addressed whether infants should be fed or kept nil per os (NPO) during medical treatment of a PDA. Infants were randomized to trophic feeds or NPO during indomethacin or ibuprofen treatment5. As part of this parent trial, we tested the hypothesis that feeding infants during PDA treatment will improve the infants’ hemodynamic response to subsequent enteral nutrition. We report the results of this ancillary hemodynamic study.
METHODS
This study was approved by the IRB at each participating institution. Infants in the parent study who were born at Boston Medical Center, Case Western Reserve University Medical Center, Mayo Clinic, University of Pittsburgh Medical Center, Santa Clara Valley Medical Center, and Vanderbilt University Medical Center were eligible to participate if the parents provided written informed consent.
Details of the inclusion/exclusion criteria, randomization and masking process can be found in the parent study manuscript5. Eligible infants were 231/7–306/7 weeks’ gestation, 401–1250 grams at birth, received maximum enteral volumes of 60 mL/kg/d prior to study entry, and were about to be treated for a PDA. Infants were initially randomized to ibuprofen or indomethacin (infused over 30–50 minutes to minimize effects on mesenteric BFV6) and then to “feeding” (15 ml/kg/day bolus feeds) or “NPO” during the “drug treatment period”, which began with the first dose of study drug and ended 24 hours after the last dose. At the end of the drug treatment period (18–24 hours after the last dose of study drug and 3 hours after the last trophic feed for the “feeding” group), babies had a baseline mesenteric flow study. Infants were then given a test feed of 4 ml/kg and the Doppler flow study was repeated 10 and 30 minutes later.
Doppler ultrasound of SMA blood flow velocity (BFV) was obtained using a 7.5 MHz linear transducer to identify the superior mesenteric artery just distal to its origin, and a 5.0–12 MHz transducer for Doppler recording as previously described.7 Angle correction of up to 30° was acceptable. Systolic, mean and end-diastolic SMA BFVs were recorded through two cycles of at least 3 consecutive waves. BFVs were averaged within each cycle, and the higher cycle average was used for statistical calculations.
Data was analyzed using Student t-test, Chi-Square or Fisher Exact, and the Mann-Whitney u-test as appropriate.
RESULTS
Infants were randomized to “feeding” (n=16) or “NPO” (n=18) during the study drug treatment period. Demographic variables were similar in the two groups with the exception of preterm rupture of membranes (Table). Babies in the “feeding” group also tended to have received higher feeding volumes prior to enrollment (Table; p=0.051).
Table.
Baseline Demographic and Clinical Variables
| Fed Group (n=16) | NPO Group (n=18) | p-Value | |
|---|---|---|---|
| Gestational age (weeks) | 26.3 ± 1.8 | 26.4 ± 2.0 | 0.868 |
| Birth weight (grams) | 908 ± 160 | 923 ± 220 | 0.829 |
| Sex (% female) | 10 (59) | 7 (41) | 0.167 |
| Race (% Caucasian) | 8 (44) | 10 (56) | 0.746 |
| Chorioamnionitis | 3 (19) | 6 (33) | 0.332 |
| Preterm rupture of membranes | 16 (100) | 14 (78) | 0.018 |
| Prolonged rupture of membranes | 5 (31) | 6 (33) | 0.897 |
| Preeclampsia | 0 (0) | 1 (6) | 1.000 |
| Betamethasone (any/complete course) | 11 (69)/3 (19) | 14 (78)/7 (39) | 0.552/0.193 |
| Apgar Score at 5 minutes | 6.5 (5, 8) | 6.5 (5, 8) | 0.972 |
| Respiratory Distress Syndrome | 15 (94) | 16 (89) | 0.614 |
| Vasopressors prior to study | 0 (0) | 1 (6) | 1.000 |
| Prophylactic Indomethacin | 7 (44) | 6 (33) | 0.533 |
| Drug for PDA treatment (indomethacin) | 15 (94) | 17 (94) | 1.000 |
| Volume of feed pre-study (ml/kg/day) | 18.6 ± 21.7 | 6.7 ± 11.8 | 0.051 |
| Age at Doppler study (days) | 9 (8, 12) | 10 (7, 13) | 0.756 |
| Postmenstrual age at study(weeks) | 27.8 ± 2.28 | 27.8 ± 1.6 | 0.985 |
| Weight at Doppler study (grams) | 955 ± 222 | 921 ± 242 | 0.676 |
| Volume used for test feed (ml) | 3.2 ± 1.2 | 3.1 ± 1.1 | 0.616 |
| Time: last PDA drug to Doppler study (hr) | 20.5 ± 7.0 | 30.5 ± 25.6 | 0.128 |
| Time: test feed to first post-feed study (min) | 10.1 ± 0.5 | 12.1 ± 5.0 | 0.133 |
| Time: test feed to 2nd post-feed study (min) | 30.3 ± 1.3 | 32.3 ± 6.4 | 0.246 |
| Heart Rate pre-study (beats/min) | 157 ± 11 | 159 ± 13 | 0.686 |
| Mean BP pre-study (mmHg) | 41 ± 9 | 43 ± 11 | 0.437 |
| O2 Sat pre-study (%) | 93 ± 6 | 93 ± 5 | 0.995 |
| Baseline Max Systolic BFV (cm/sec) | 77.4 ± 26.1 | 71.5 ± 20.5 | 0.467 |
| Baseline Mean BFV (cm/sec) | 33.8 ± 15.5 | 31.8 ± 10.5 | 0.648 |
| Baseline End-Diastolic BFV (cm/sec) | 11.3 ± 5.6 | 15.3 ± 9.8 | 0.157 |
| Baseline Resistance Index* | 0.84 ± 0.10 | 0.79 ± 0.11 | 0.116 |
| Baseline Pulsitility Index† | 2.14 ± 0.78 | 1.87 ± 0.57 | 0.258 |
t-test for continuous variables, Data: mean±SD; Chi-Square or Fisher Exact Test for counts, as appropriate, Data: n(%); Mann-Whitney for ordinal variables, Data: median (Q1–Q3);
Resistance index [(systolic-diastolic BFV)/systolic BFV];
Pulsatility index [(systolic-diastolic BFV)/mean BFV]
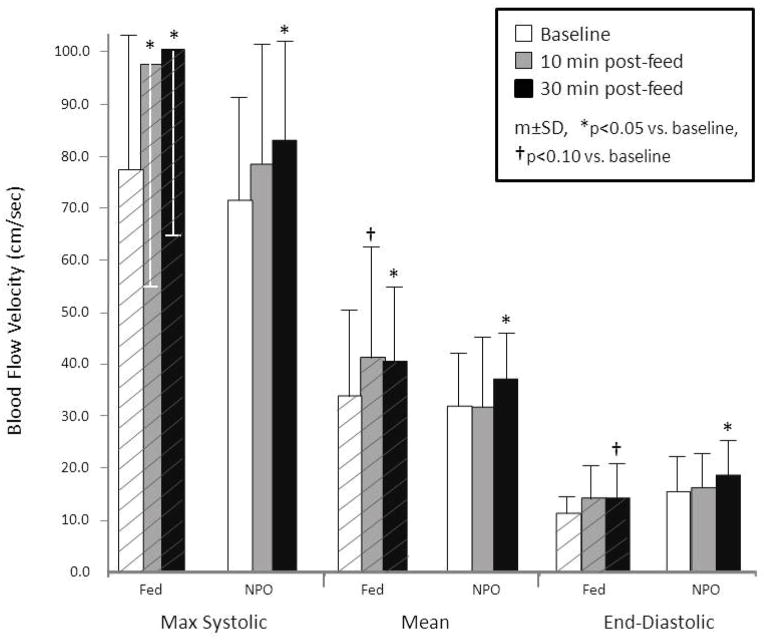
There were no significant differences between the “feeding” and “NPO” groups in either the baseline or post-feed SMA flow velocities (Figure). Both groups had similar and significant increases in systolic, mean, and diastolic SMA BFVs by 30 minutes after the test feed. However, infants in the “feeding” group started to increase their systolic and mean BFVs earlier after the test feed than infants in the “NPO” group (Figure). The response to feeding was not significantly different for the one baby in each group who recieved ibuprofen compared with babies in the same group who received indomethacin.
Figure.
Max systolic, mean, and end-diastolic BFV for babies fed (hatched bars) and NPO (open bars) during medical treatment of the PDA. By 30 minutes after the test feed, both groups had a significant increase in systolic, mean and end-diastolic mesenteric BFV. Babies who fed during treatment of the PDA had an earlier rise in systolic and mean mesenteric BFV.
DISCUSSION
The “parent” trial found that infants who receive “trophic” enteral feedings, while they receive drug treatment for a PDA, require less time to reach near-full enteral nutrition. Prior studies have shown that the SMA’s postprandial BFV response can predict feeding tolerance in preterm infants8. Therefore, we designed an ancillary study to determine if “trophic” feedings during PDA treatment also improve postprandial hemodynamic responses during the post-treatment period.
In our study, more than 90% of the infants were treated with indomethacin. We had planned to examine an equal number of infants treated with indomethacin and ibuprofen. However, due to manufacturing problems, ibuprofen was unavailable for the majority of the study. Bolus indomethacin reduces SMA BFV for 2–4 hours after administration.2 Slow infusion of indomethacin has minimal effect on mesenteric BFVs6. We eliminated any interfering effects of drug treatment on our study groups’ postprandial hyperemic responses by both administering the drug slowly over 30–50 minutes and also waiting 18–24 hours after the last dose before testing the infants.
Infants who were fed during the drug administration period appeared to have an earlier rise in postprandial SMA BFVs compared with those kept NPO. However, the magnitude of the postprandial hemodynamic response was similar between the groups. Additionally, despite recent PDA and PDA treatment, the baseline BFV and the BFV responses to feeding were similar in magnitude to those we reported previously in anemic preterm infants7. The findings support the results of the parent clinical trial5 in which there was no significant difference between the groups when feeding intolerance was examined in a multivariate model that included all of the relevant demographic risk factors (P=0.1)5. Importantly, there were also no differences in the incidence of necrotizing enterocolitis or spontaneous ileal perforation between groups.
In conclusion, very low birth weight infants treated medically for a PDA can mount an appropriate SMA BFV response to feeding whether they are fed trophic feeds or kept NPO during PDA treatment.
Acknowledgments
Supported by the Gerber Foundation and a gift from the Jamie and Bobby Gates Foundation.
Abbreviations
- SMA
superior mesenteric artery
- BFV
blood flow velocity
- PDA
patent ductus arteriosus
Footnotes
The authors declare no conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Shimada S, Kasai T, Hoshi A, Murata A, Chida S. Cardiocirculatory effects of patent ductus arteriosus in extremely low-birth-weight infants with respiratory distress syndrome. Pediatr Int. 2003;45:255–62. doi: 10.1046/j.1442-200x.2003.01713.x. [DOI] [PubMed] [Google Scholar]
- 2.Van Bel F, Van Zoeren D, Schipper J, Guit GL, Baan J. Effect of indomethacin on superior mesenteric artery blood flow velocity in preterm infants. J Pediatr. 1990;116:965–70. doi: 10.1016/s0022-3476(05)80662-6. [DOI] [PubMed] [Google Scholar]
- 3.McCurnin D, Clyman RI. Effects of a patent ductus arteriosus on postprandial mesenteric perfusion in premature baboons. Pediatrics. 2008;122:e1262–7. doi: 10.1542/peds.2008-2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hernandez G, Velasco N, Wainstein C, Castillo L, Bugedo G, Maiz A, et al. Gut mucosal atrophy after a short enteral fasting period in critically ill patients. J Crit Care. 1999;14:73–7. doi: 10.1016/s0883-9441(99)90017-5. [DOI] [PubMed] [Google Scholar]
- 5.Clyman R, Wickremasinghe A, Jhaveri N, Hassinger DC, Attriridge JT, Sanocka U, et al. Enteral Feeding during Indomethacin and Ibuprofen Treatment of a Patent Ductus Arteriosus. J Pediatr. 2013 doi: 10.1016/j.jpeds.2013.01.057. S0022-3476(13)00132-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coombs RC, Morgan MEI, Durbin GM, Booth IW, McNeish AS. Gut blood flow velocities in the newborn: effects of patent ductus arteriosus and parenteral indomethacin. Archives of Disease in Childhood. 1990;65:1067–71. doi: 10.1136/adc.65.10_spec_no.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Krimmel G, Baker R, Yanowitz TD. Blood transfusion alters superior mesenteric artery blood flow response to feedings in premature infants. Am J Perinatol. 2009;26:99–106. doi: 10.1055/s-0028-1090595. [DOI] [PubMed] [Google Scholar]
- 8.Fang S, Kempley ST, Gamsu HR. Prediction of early tolerance to enteral feeding in preterm infants by measurement of superior mesenteric artery blood flow velocity. Archives of Disease in Childhood Fetal & Neonatal Edition. 2001;85:F42–5. doi: 10.1136/fn.85.1.F42. [DOI] [PMC free article] [PubMed] [Google Scholar]