Abstract
Background and objectives
Alge et al. recently reported that urinary renin may be a prognostic biomarker for AKI after cardiac surgery. However, their urinary renin levels far exceeded published plasma renin levels, whereas normally, urinary renin is <10% of plasma renin. This result raises questions about the specificity of the new Quantikine Renin ELISA Kit used in the work by Alge et al., which is claimed to detect total renin (i.e., renin and prorenin). Therefore, this study tested this assay.
Design, setting, participants, & measurements
Plasma and urine from 30 patients with hypertension, diabetes, or preeclampsia and 10 healthy pregnant women (randomly selected from sample sets obtained earlier to investigate urinary renin-angiotensin system components) were used to compare the ELISA with a validated renin immunoradiometric assay and an in-house enzyme kinetic assay. Measurements were performed before and after in vitro prorenin activation, representing renin and total renin, respectively.
Results
Total renin measurements by ELISA, immunoradiometric assay, and enzyme kinetic assay were highly correlated. However, ELISA results were consistently ≥10-fold higher. The ELISA standard yielded low to undetectable levels in the immunoradiometric assay and enzyme kinetic assay, except after prorenin activation, when the results were ≥10-fold lower than the ELISA results. In plasma, prorenin activation increased ELISA results by 10%–15%. Urine contained no detectable prorenin.
Conclusions
The ELISA renin kit standard is prorenin, and its immunoreactivity and enzymatic activity after conversion to renin do not match the International Reference Preparation of human renin that has been used to validate previous immunoradiometric assays and enzyme kinetic assays; in fact, they are at least 10-fold lower, and thus, any measurements obtained with this ELISA kit yield levels that are at least 10-fold too high. The ELISA antibodies detect both renin and prorenin, with a preference for the former. Given these inconsistencies, urinary renin levels should be measured by established renin assays.
Keywords: ACE inhibitors, renin-angiotensin system, albuminuria, chronic heart, failure, CKD
Introduction
Renin-angiotensin system (RAS) components in urine are currently being evaluated as renal disease markers, the underlying concept being that they reflect the activity of the renal RAS independent from the circulating RAS and that the renal RAS contributes to renal disease (1,2). For instance, Alge et al. (3) recently suggested that urinary angiotensinogen is a prognostic biomarker for the progression of AKI after cardiac surgery. In a subsequent study, Alge et al. (4) additionally studied urinary renin to improve prognostic power. Remarkably, the urinary renin-to-creatinine ratios in that study, involving patients between 58 and 79 years old, ranged from 114 to 2894 pg/mg. Given the urinary creatinine concentration in such patients (approximately 1 mg/ml), these results would translate to urinary renin levels of 114–2894 pg/ml (5,6). Such levels are several orders of magnitude higher than the urinary renin levels reported by others (approximately 1–2 pg/ml; range=0.03–157 pg/ml) (7–9) and even substantially higher than the plasma renin levels in cardiac patients (10). This finding raises questions about the specificity of the ELISA used in the works by Alge et al. (4) to measure renin (Quantikine ELISA; R&D Systems, Abingdon, UK). One reason for this discrepancy might be that the ELISA simultaneously detects prorenin, the inactive precursor of renin (i.e., it actually measures total renin [renin and prorenin]), and not renin. However, although prorenin levels in plasma are generally 10-fold higher than plasma renin levels, urine contains no detectable prorenin (7), thereby ruling out this possibility.
Classically, renin is measured immunoreactively, making use of antibodies that recognize the active site of renin, or enzyme kinetically (i.e., based on its capacity to generate angiotensin I [Ang I]) (11). The latter is more sensitive but can obviously not be used in samples from patients treated with a renin inhibitor. In the present study, we compared the new ELISA kit with both an established renin immunoradiometric assay (IRMA) and an in-house enzyme kinetic assay (EKA). We focused on urine samples obtained from various patient groups but also tested plasma samples.
Materials and Methods
Samples
Urine samples were obtained from 10 hypertensive patients (six men and four women; age=58±10 years), 10 patients with diabetes mellitus (eight men and two women; age=59±10 years), 10 healthy pregnant women (age=33±4 years), and 10 women with preeclampsia (age=31±5 years). From the latter two groups, plasma samples were also available. The former two groups have been described previously (7,8), and the latter two groups are part of an ongoing study investigating the RAS in pregnancy and preeclampsia. The four patients groups were chosen on the basis of availability and their wide variety in renin and prorenin levels; diabetic patients and preeclamptic women have relatively low renin and high prorenin levels versus hypertensive patients, and pregnant women have relatively high renin and prorenin levels (12,13). The 10 representatives from each group were chosen in a random manner. All studies were approved by the Medical Ethical Review Board and performed in accordance with the Declaration of Helsinki and Good Clinical Practice. Informed consent was obtained from each subject. Blood was collected in EDTA tubes and centrifuged, and plasma was stored at −20°C until analysis. Urine was frozen without inhibitors and also stored at −20°C until analysis. The samples from the healthy pregnant women and the women with preeclampsia had not been thawed before, whereas the samples from the diabetic and hypertensive patients had been thawed one time. This information is important, because repetitive thawing and freezing of plasma, but not urine, is known to cause prorenin activation (i.e., prorenin-to-renin conversion) without affecting the total amount of renin and prorenin.
Biochemical Measurements
Total Renin Measurement in Plasma and Urine by Quantikine ELISA.
Total renin in urine and plasma was measured by Quantikine ELISA, which makes use of a 96-well plate precoated with a (pro)renin antibody (i.e., an antibody that recognizes both renin and prorenin). After binding renin and prorenin, a secondary (pro)renin-specific enzyme-linked monoclonal antibody is added, which after washing, is incubated with a substrate solution that allows a color to develop in proportion to the amount of renin bound in the initial step. The color is subsequently detected making use of a microplate reader (VersaMax ELISA Microplate Reader; Molecular Devices, Sunnyvale, CA). The detection limit is 4.4 pg/ml, and the standard curve ranges from 31.3 to 2000 pg/ml.
Renin and Total Renin Measurement in Plasma by Cisbio IRMA.
Renin in plasma was measured with an IRMA (Renin III; Cisbio, Gif-sur-Yvette, France), making use of an active site-directed radiolabeled antibody (11). Total renin can also be measured with this assay either after converting prorenin to renin with trypsin (proteolytic activation; i.e., the prosegment is cleaved off) or by incubating the sample for 48 hours with 10 μmol/L aliskiren (14). The latter procedure induces a conformational change in the prorenin molecule (nonproteolytic activation; involving the removal of the prosegment from the enzymatic cleft), allowing its recognition by the active site-directed antibody. The detection limit of the IRMA is 1 pg/ml, and the standard curve ranges from 2.5 to 320 pg/ml.
Renin Measurement in Urine by Cisbio IRMA or EKA.
Renin in urine was measured by either IRMA or EKA (11). The latter involves the incubation with excess sheep angiotensinogen and angiotensinase inhibitors and the subsequent detection of the generated Ang I by radioimmunoassay. The detection limit of the EKA is 0.05 ng Ang I/ml per hour. Ang I–generating activities were converted to renin concentrations based on the fact that 1 ng Ang I/ml per hour corresponds with 2.6 pg human renin/ml (15). Before their application in the IRMA, the urine samples from the patients with hypertension and diabetes were concentrated 5-fold with Amicon Ultra-10 centrifugal filter devices (EMD Millipore, Cork, Ireland). This concentration step was not applied to the samples from pregnant women because of their higher renin content. Prorenin activation did not increase the renin content of the urine samples (7), suggesting that urine does not contain prorenin.
Statistical Analyses
Results are expressed as mean and SD. Levels that were below the detection limit were considered to be equal to the detection limit to allow for statistical evaluation. Differences between levels before and after prorenin activation were assessed by paired t tests. Data were logarithmically transformed before analysis in case of non-normal distribution. Correlations between assay results were assessed by linear regression. P<0.05 was considered statistically significant. Statistical analyses were performed with SPSS (version 21; IBM).
Results
Urinary renin in 40 samples, when measured by IRMA or EKA, amounted to 1.9±2.2 pg/ml. Results were identical after prorenin activation (data not shown), suggesting that urine does not contain prorenin. When measuring these same urine samples in the ELISA, which measures renin and prorenin (i.e., total renin), total renin levels were ≥10-fold higher than levels measured by IRMA or EKA (Figure 1A). An identical 10-fold–higher value was obtained when detecting the IRMA renin standard in the ELISA (Figure 1B). Importantly, urinary total renin levels, when measured by ELISA, were below the detection limit in 26 samples compared with 1 sample when applying the IRMA or EKA. Assuming that the levels in the samples that were below the detection limit equaled the detection limit, the urinary total renin levels on the basis of the ELISA were 20±31 pg/ml.
Figure 1.
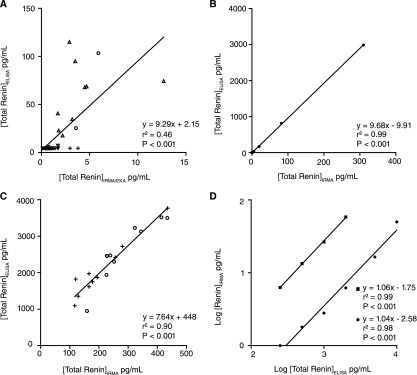
Comparison of immunoradiometric assay (IRMA) and ELISA total renin measurements. (A) urine samples; (B) IRMA renin standard; and (C) plasma samples. Samples were obtained from patients with hypertension (▿), diabetes (Δ), and preeclampsia (○) and healthy pregnant women (+). D compares the ELISA standard before (●) and after (■) prorenin activation; not enough standard was available to have the same number of observations after prorenin activation as before prorenin activation. In C, the relationship between the two measurements was not different when limiting it to samples that were above the detection limit.
Plasma renin in 20 samples, when measured by IRMA, was 30±18 pg/ml and increased to 244±100 pg/ml after prorenin activation (P<0.001). The latter values represent renin and prorenin (i.e., total renin). The same samples, when measured by ELISA, yielded approximately 10-fold higher total renin levels (2315±810 pg/ml) (Figure 1C). Taken together, these data suggest that the ELISA is less sensitive than the other two approaches and yields total renin levels that are ≥10-fold higher.
The most logical explanation of the above findings is that the ELISA standard differs from the IRMA standard. We therefore measured the ELISA standard in both the EKA and IRMA before and after prorenin activation with trypsin. Figure 1D shows the comparison between IRMA and ELISA. Before prorenin activation, the ELISA standard yielded levels in the IRMA that were approximately 300-fold lower than expected, whereas after activation, the levels were approximately 30-fold lower than expected. Results obtained by EKA were identical to results by IRMA (data not shown). Given the approximately 10-fold increase in both renin immunoreactivity and enzymatic activity after prorenin activation, it is clear that the ELISA standard is actually prorenin. The standard curve of the ELISA is claimed to run from 31.3 to 2000 pg/ml. In reality, it now seems to be from 1 to 67 pg/ml.
Finally, because the ELISA antibodies detect renin and prorenin equally well, it should not matter whether samples are measured before or after nonproteolytic prorenin activation by aliskiren. However, when measuring 8 individual plasma samples (4 samples from healthy pregnant women and 4 samples from women with preeclampsia randomly chosen from 10 samples of each group that were used in this study) before and after aliskiren treatment pairwise in the ELISA, we consistently observed that the levels detected after activation (2626±756 pg/ml) were 10%–15% higher than before activation (2361±719 pg/ml; P<0.01). This finding suggests that at least one of two antibodies applied in this assay has a preference for renin.
Discussion
Urinary renin is increasingly believed to serve as a prognostic biomarker (1,4,7,8). It may be filtered from plasma or could be kidney-derived (1). Normally, urinary renin levels are 5%–10% of plasma renin levels (7–9). Particularly, under conditions where plasma renin levels are high, a substantial percentage of urinary renin may be plasma-derived (8). Thus, to what degree urinary renin yields information independent from the circulating RAS remains to be determined. Alge et al. (4) recently reported that urinary renin, together with urinary angiotensinogen, associates with AKI after cardiac surgery. Their urinary renin levels were measured with the Quantikine Renin ELISA Kit and exceeded published plasma renin levels (11,12).
However, the present study reveals that this ELISA does not provide reliable renin levels. In fact, the kit simultaneously detects both renin and prorenin, and thus, given that in most body fluids prorenin greatly exceeds renin (up to 100-fold), it provides information on prorenin (11,12,16). Remarkably for a renin kit, the ELISA uses recombinant prorenin as a standard, and even more concerning, this prorenin has not been calibrated against the International Reference Preparation of human renin. As a consequence, its total renin (renin and prorenin) results are more than one order of magnitude from published total renin levels over the past 3 decades. Indeed, the normal range claimed by the manufacturer is 201±1851 pg/ml in plasma and nondetectable to 96.7 pg/ml in urine (i.e., >10-fold higher than the levels reported earlier by others in normal subjects) (7,8,11,12).
The Quantikine prorenin standard, when converted to renin, yielded ≥10-fold lower levels than expected in the well established Cisbio renin IRMA and our in-house renin EKA. According to the manufacturer’s manual, the ELISA kit relies on antibodies that detect both renin and prorenin (i.e., antibodies that recognize epitopes on renin’s main body and not its active site or the prosegment). Nevertheless, our data consistently showed that the ELISA kit results after prorenin activation were 10%–15% higher than before activation, which is suggestive of a renin preference of at least one of two antibodies. Unfortunately, the manufacturer did not provide information on the precise epitopes recognized by these antibodies, and thus, we cannot explain this renin preference.
Given the noncalibrated standard of this ELISA, it is not surprising that Alge et al. (4), when using this kit, obtained urinary renin levels that are far above the normal range in plasma (11) and, most likely, even above the plasma levels in patients who developed AKI after cardiac surgery. It should be noted that plasma renin values were not reported in their study but that their highest urinary total renin levels (2894 pg/ml) exceed the plasma levels of total renin measured in heart failure patients treated with RAS blockers (10).
Clearly, the urinary renin levels detected with this kit do not reflect reality, and realistic values would only be obtained when taking into consideration that the ELISA standard contains ≥10 times less prorenin than claimed. Without such correction, the ELISA results do not allow comparison with published urinary renin levels. However, even when corrected for the lower prorenin content of the ELISA standard, the kit does not offer a greater sensitivity than the Cisbio IRMA or the EKA. Nevertheless, in view of the correlation between the levels measured with this kit and the levels measured with validated assays (Figure 1), it is still possible that urinary renin is a prognostic biomarker for AKI after cardiac surgery. We suggest that, in future studies, urinary (and plasma) renin levels should be measured by established renin assays that have been rigorously validated by making use of the International Reference Preparation of human renin (11). EKAs are most sensitive and can be applied directly to nonconcentrated urine samples. However, they depend on the availability of angiotensinogen and require the measurement of Ang I by radioimmunoassay. IRMA kits are easier to handle but display a lower sensitivity. Ideally, urine samples obtained from healthy individuals should, therefore, be concentrated approximately 5-fold before the application of such assays. The concentration step is not required when studying samples from patients with an activated RAS.
Disclosures
None.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Roksnoer LCW, Verdonk K, van den Meiracker AH, Hoorn EJ, Zietse R, Danser AHJ: Urinary markers of intrarenal renin-angiotensin system activity in vivo. Curr Hypertens Rep 15: 81–88, 2013 [DOI] [PubMed] [Google Scholar]
- 2.Kobori H, Nangaku M, Navar LG, Nishiyama A: The intrarenal renin-angiotensin system: From physiology to the pathobiology of hypertension and kidney disease. Pharmacol Rev 59: 251–287, 2007 [DOI] [PubMed] [Google Scholar]
- 3.Alge JL, Karakala N, Neely BA, Janech MG, Tumlin JA, Chawla LS, Shaw AD, Arthur JM, SAKInet Investigators : Urinary angiotensinogen and risk of severe AKI. Clin J Am Soc Nephrol 8: 184–193, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alge JL, Karakala N, Neely BA, Janech MG, Tumlin JA, Chawla LS, Shaw AD, Arthur JM, SAKInet Investigators : Association of elevated urinary concentration of renin-angiotensin system components and severe AKI. Clin J Am Soc Nephrol 8: 2043–2052, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barr DB, Wilder LC, Caudill SP, Gonzalez AJ, Needham LL, Pirkle JL: Urinary creatinine concentrations in the U.S. population: Implications for urinary biologic monitoring measurements. Environ Health Perspect 113: 192–200, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Waikar SS, Sabbisetti VS, Bonventre JV: Normalization of urinary biomarkers to creatinine during changes in glomerular filtration rate. Kidney Int 78: 486–494, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.van den Heuvel M, Batenburg WW, Jainandunsing S, Garrelds IM, van Gool JMG, Feelders RA, van den Meiracker AH, Danser AHJ: Urinary renin, but not angiotensinogen or aldosterone, reflects the renal renin-angiotensin-aldosterone system activity and the efficacy of renin-angiotensin-aldosterone system blockade in the kidney. J Hypertens 29: 2147–2155, 2011 [DOI] [PubMed] [Google Scholar]
- 8.Persson F, Lu X, Rossing P, Garrelds IM, Danser AHJ, Parving HH: Urinary renin and angiotensinogen in type 2 diabetes: Added value beyond urinary albumin? J Hypertens 31: 1646–1652, 2013 [DOI] [PubMed] [Google Scholar]
- 9.Lumbers ER, Skinner SL: Observations on the origin of renin in human urine. Circ Res 24: 689–697, 1969 [DOI] [PubMed] [Google Scholar]
- 10.Klotz S, Burkhoff D, Garrelds IM, Boomsma F, Danser AHJ: The impact of left ventricular assist device-induced left ventricular unloading on the myocardial renin-angiotensin-aldosterone system: Therapeutic consequences? Eur Heart J 30: 805–812, 2009 [DOI] [PubMed] [Google Scholar]
- 11.Campbell DJ, Nussberger J, Stowasser M, Danser AHJ, Morganti A, Frandsen E, Ménard J: Activity assays and immunoassays for plasma Renin and prorenin: Information provided and precautions necessary for accurate measurement. Clin Chem 55: 867–877, 2009 [DOI] [PubMed] [Google Scholar]
- 12.Danser AHJ, Derkx FHM, Schalekamp MADH, Hense HW, Riegger GA, Schunkert H: Determinants of interindividual variation of renin and prorenin concentrations: Evidence for a sexual dimorphism of (pro)renin levels in humans. J Hypertens 16: 853–862, 1998 [DOI] [PubMed] [Google Scholar]
- 13.Verdonk K, Visser W, van den Meiracker AH, Danser AHJ: The renin-angiotensin-aldosterone system in pre-eclampsia: The delicate balance between good and bad. Clin Sci (Lond) 126: 537–544, 2014 [DOI] [PubMed] [Google Scholar]
- 14.Krop M, van Gool JMG, Day D, Hollenberg NK, Danser AHJ: Evaluation of a direct prorenin assay making use of a monoclonal antibody directed against residues 32-39 of the prosegment. J Hypertens 29: 2138–2146, 2011 [DOI] [PubMed] [Google Scholar]
- 15.Krop M, Garrelds IM, de Bruin RJA, van Gool JMG, Fisher ND, Hollenberg NK, Danser AHJ: Aliskiren accumulates in renin secretory granules and binds plasma prorenin. Hypertension 52: 1076–1083, 2008 [DOI] [PubMed] [Google Scholar]
- 16.Schalekamp MADH, Derkx FHM, Deinum J, Danser AHJ: Newly developed renin and prorenin assays and the clinical evaluation of renin inhibitors. J Hypertens 26: 928–937, 2008 [DOI] [PubMed] [Google Scholar]


