Abstract
Objectives:
Obesity may alter the pharmacokinetics of β-lactams. The goal of this study was to evaluate if and why serum concentrations are inadequate when standard β-lactam regimens are administered to obese, non-critically ill patients.
Subjects and methods:
During first year, we consecutively included infected, obese patients (body mass index (BMI) ⩾30 kg m−2) who received meropenem (MEM), piperacillin-tazobactam (TZP) or cefepime/ceftazidime (CEF). Patients with severe sepsis or septic shock, or those hospitalized in the intensive care unit were excluded. Serum drug concentrations were measured twice during the elimination phase by high-performance liquid chromatography. We evaluated whether free or total drug concentrations were >1 time (fT>minimal inhibition concentration (MIC)) or >4 times (T>4MIC) the clinical breakpoints for Pseudomonas aeruginosa during optimal periods of time: ⩾40% for MEM, ⩾50% for TZP and ⩾70% for CEF.
Results:
We included 56 patients (median BMI: 36 kg m−2): 14 received MEM, 31 TZP and 11 CEF. The percentage of patients who attained target fT>MIC and T>4MIC were 93% and 21% for MEM, 68% and 19% for TZP, and 73% and 18% for CEF, respectively. High creatinine clearance (107 (range: 6–398) ml min−1) was the only risk factor in univariate and multivariate analyses to predict insufficient serum concentrations.
Conclusions:
In obese, non-critically ill patients, standard drug regimens of TZP and CEF resulted in insufficient drug concentrations to treat infections due to less susceptible bacteria. Augmented renal clearance was responsible for these low serum concentrations. New dosage regimens need to be explored in this patient population (EUDRA-CT: 2011-004239-29).
Introduction
Obesity, defined by the World Health Organization as a body mass index (BMI) greater or equal to 30 kg m−2, is a worldwide uncontrolled epidemic: in 2005 it was estimated that 400 million adults were obese, whereas it is projected that more than 700 million will be obese in 2015.1 Obesity is a risk factor for more frequent and longer hospital stays, and for more post-operative and hospital-related infections. Optimal management of these infections requires early administration of appropriate antibiotics. Both the choice of the antibiotic and the administration of an appropriate dose (according to pharmacokinetic/pharmacodynamic (PK/PD) principles) are important factors to improve the outcome of severe infections.2
Recommended daily doses of antibiotics are based on PK studies performed essentially in non-obese patients. However, the PK of hydrophilic antibiotics that are essentially eliminated by renal clearance, like β-lactams, may be altered in obese patients. Indeed, the volume of distribution (VD) may be increased because of greater lean body mass and more adipose tissue, and renal clearance may be either increased because of greater kidney mass and global filtration or decreased because of chronic hypertensive or diabetic nephropathy.2
Lower β-lactam serum concentrations in obese than in non-obese patients have been reported in sparse case reports and limited case series.3, 4, 5, 6 However, we previously showed that in critically ill patients, inadequate serum concentrations of broad-spectrum β-lactams were mainly due to sepsis alone, rather than to obesity.7 In a recent study, Sturm et al. also showed that obese, critically ill patients do not need higher dosage regimens of piperacillin-tazobactam (TZP) than non-obese individuals.8 The question remains: is obesity responsible for significant PK changes of β-lactams in infected, but not critically ill patients? The aim of this study was to evaluate the adequacy of standard β-lactam dosage regimens in infected, obese but not critically ill patients and to identify risk factors associated with inadequate drug concentrations.
Subjects and Methods
Study design and data sources
This prospective study was conducted at Erasme hospital, an 858-bed university hospital, between 1 October 2011 and 30 September 2012. Adult patients (⩾18 years of age) were included if they had a BMI ⩾30 kg m−2, and were receiving ceftazidime or cefepime (CEF), TZP or meropenem (MEM) for a suspected or confirmed infection. Exclusion criteria were severe sepsis or septic shock and those admitted to the intensive care unit. Patients could be included more than once if they received another study antibiotic.
Antibiotic treatment and serum sampling
The clinician's choice of antibiotic therapy was based on local guidelines. All patients received standard dosage regimens: 2 g three times a day for CEF, 4 g four times a day for TZP or 1 g three times a day for MEM. In case of renal failure, the first dose remained unchanged, but the subsequent doses were adapted to the creatinine clearance (CrCL) calculated using the Cockroft-Gault formula (CrCLCG)9 as shown in Supplementary Table 1.
Two blood samples (3 ml each) were taken to assess drug concentrations: one just before (T0) and the second, 2 h (T2) after the onset of the 30-min infusion of the antibiotic. Exact sampling times were recorded. Samples were kept on ice and sent directly to the clinical chemistry laboratory, where they were centrifuged at 3000 r.p.m. at 4 °C for 10 min before the supernatant was removed and analyzed. High-performance liquid chromatography connected to ultraviolet spectrophotometry was used to measure the serum concentrations of β-lactams. For TZP concentrations, only piperacillin levels were measured because the PK of piperacillin and tazobactam are highly correlated. The lower and upper limits of quantification for each β-lactam analyzed were 2 and 200 mg l−1, respectively. The coefficient of variation for each β-lactam never exceeded 11%.10
Data collection
Demographic data, co-morbidities, site of infection, causative pathogens, antibiotic dosage regimen administered, number of days of antibiotic therapy, the day of serum sampling and biological data were recorded.
Estimation of serum concentrations at any given time and PK analyses
A one-compartment model was used to perform PK analyses: assuming that the steady state was reached, considering the exponential elimination of drugs in one-compartment models, and that sampling was performed during the elimination phase, the following equation [ln Ct=−ket+lnC0] was used to estimate the serum concentrations of the drug at one given time, where Ct is the measured serum concentration at the specified time, C0 is the virtual serum concentration at the beginning of the elimination phase and ke is the elimination constant. On the basis of the dose interval, the time in % spent above the clinical breakpoint was estimated. Other PK variables, such as VD, total body clearance (CL) and elimination half-life (t½), could then be calculated by using conventional PK equations: t½=0.693/ke, and VD=(dose of antibiotic administered)/C0 and CL=VD × ke. PK parameters were evaluated for the entire cohort, and in the subgroup of patients with CrCL >80 ml min−1.10
Clinical breakpoints, PK and PD criteria
The European Committee on Antimicrobial Susceptibility Testing (EUCAST) has defined clinical breakpoints of microorganisms for different antimicrobial therapies by taking into account in vitro and in vivo PK/PD data. The clinical breakpoints for susceptibility have been fixed to ensure a good probability of therapeutic success, translating into a probability of PD target attainment greater than 90%. The PD target for β-lactams used by EUCAST is the free fraction (fT)>1 × minimal inhibition concentration (fT>MIC) during 40–50%, 50–60% and 60–70% of the dosage interval for MEM, TZP and CEF, respectively.11 However, another PD target also frequently used is the total fraction (T)>4 × MIC (T>4MIC) for 40%, 50% and 70% of the dosage interval for MEM, TZP and CEF, respectively.7, 10, 12
Each patient was classified as having a serum concentration less than or greater than the 2 PD targets for pathogens with different MIC values. To calculate the fT, we considered that 2%, 30% and 15% of MEM, TZP and CEF, respectively, were bound to protein.13, 14, 15, 16 The percentage of patients who reached each PD target was calculated for different MICs, and more specifically for infections caused by Enterobacteriaceae spp. and P. aeruginosa, based on EUCAST's clinical breakpoints for these pathogens: 2 mg l−1 for MEM, 8 mg l−1 and 16 mg l−1 for TZP and 1 mg l−1 and 8 mg l−1 for CEF, respectively.17 In accord with EUCAST, a percentage of PD target attainment less than 90% was considered insufficient.
Urinary CrCL
Twenty-four-hour urine collections were performed. Urinary CrCL was calculated using the following equation: CrCL24h (ml min−1)=(Urine creatinine (mg dl−1) × volume (ml))/(Plasma creatinine concentration (mg dl−1) × 1440). CrCL24h values were compared with CrCLCG values;9 if significantly different, then the impact on the delivered drug regimen was assessed. Correlations between CrCL24h and the percentage of patients who attained the PD target were also examined.
Body size descriptors
The patient's total body weight (TBW; kg), height (HT; cm) and abdominal circumference were measured on day of serum sampling. This information was then used to estimate different body size descriptors as described by Green et al.18 The body surface area (BSA; m2), the ideal body weight (IBW; kg), the fat-free mass (FFM; kg), the lean body weight (LBW; kg), the adjusted body weight (ABW; kg) and the percent ideal body weight (PIBW; %) were calculated using the following equations: BSA=TBW0.425 × HT0.725 × 0.007184; IBW=45.4+0.89 × HT (cm)−152.4)+4.5 for males and 45.4+0.89 × HT (cm)−152.4) for females; FFM=0.285 × TBW+12.1 × HT (m)2 for males and 0.287 × TBW+9.74 × HT (m)2 for females; LBW=1.1 × TBW–0.0128 × BMI × TBW for males and 1.07 × TBW–0.0148 × BMI × TBW for females; ABW=IBW+0.4(TBW–IBW); and finally PIBW=(TBW−IBW)/IBW × 100.
Risk factors for failure to reach PD targets
Risk factors for failure to reach PD targets were evaluated. The explored variables were: age, sex, early sampling (defined as sampling performed during the first 48 h of antibiotic treatment), CrCL24h and the different body size descriptors.
Ethics
The research was conducted in accordance with the Declaration of Helsinki, national and institutional standards. The study was approved by the Erasme Hospital's Ethics Committee (registration number: P2011/258), and approved by the Belgian Regulatory Agency (2011-004239-29). Written informed consent was obtained from all patients.
Statistical analysis
Descriptive statistics were performed for all study variables. Discrete variables are expressed as counts (percentage) and continuous variables as median (range). Categorical data were compared using the χ2-test or Fisher's exact test, as appropriate. Continuous variables were compared using the Mann–Whitney U-test. Multivariate logistic regression analysis with insufficient drug concentrations as the dependent variable was performed in all patients: only variables associated with a higher risk of inadequate concentrations (P<0.2) on a univariate basis were introduced in the multivariate model. Co-linearity between variables was excluded before modeling. Odds ratio with 95% confidence intervals were computed. All tests were two-sided and P-values less than 0.05 were considered significant.
Post-hoc analysis
As patients with neoplasia represented 30% of our patients, we re-analyzed our data after excluding this subgroup of patients.
Furthermore, because high CrCL was identified as the only factor associated with insufficient serum concentrations to treat bacteria with high MICs in our cohort, we performed a post-hoc analysis concerning CrCL. We also compared age, sex and TBW between patients with CrCL24h greater than 150 ml min−1 to those with a CrCL24h lower than 150 ml min−1.
Results
We included 56 patients: 14 received MEM, 31 received TZP and 11 received CEF. Serum sampling were performed after 3 (1–10) days of treatment. Demographic, biological and clinical characteristics and size descriptors are presented in Table 1.
Table 1. Demographic, biological and clinical characteristics of all patients.
| Variables | Patients (n=56) |
|---|---|
| Age (years) | 61 (18–84) |
| Male/female | 28/28 |
| Weight (kg) | 103 (81–153) |
| Abdominal circumference (cm) | 127 (99–177) |
| Body size descriptors | |
| Body mass index (kg m−2) | 36 (30–54) |
| Ideal body weight (kg) | 64 (41–80) |
| Percent of ideal body weight (%) | 163 (128–272) |
| Fat-free mass (kg) | 58 (47–74) |
| Lean body weight (kg) | 60 (35–80) |
| Adjusted body weight (kg) | 80 (61–105) |
| Body surface area (m2) | 2.1 (1.8–2.6) |
| Co-morbidities | |
| COPD | 12 (21) |
| Hypertension | 18 (32) |
| Diabetes | 24 (43) |
| Chronic renal insufficiency (serum creatinine> 2.5 mg dl−1) | 3 (5) |
| Malignancy | 18 (32) |
| Neutropenia or immunosuppressive treatment | 9 (16) |
| Site of infection | |
| Abdomen | 19 (34) |
| Skin | 12 (21) |
| Lungs | 11 (20) |
| Urinary tract | 6 (11) |
| Other | 8 (14) |
| Pathogens | |
| Enterobacteriaceae | 24 (43) |
| Streptococcus spp. | 10 (18) |
| Staphylococcus spp. | 7 (13) |
| Pseudomonas aeruginosa | 6 (11) |
| Stenotrophomonas | 1 (2) |
| Unidentified | 11 (20) |
| Positive blood cultures | 16 (29) |
Abbreviation: COPD, chronic obstructive pulmonary disease.
Data are expressed as counts (percentage) or median (range).
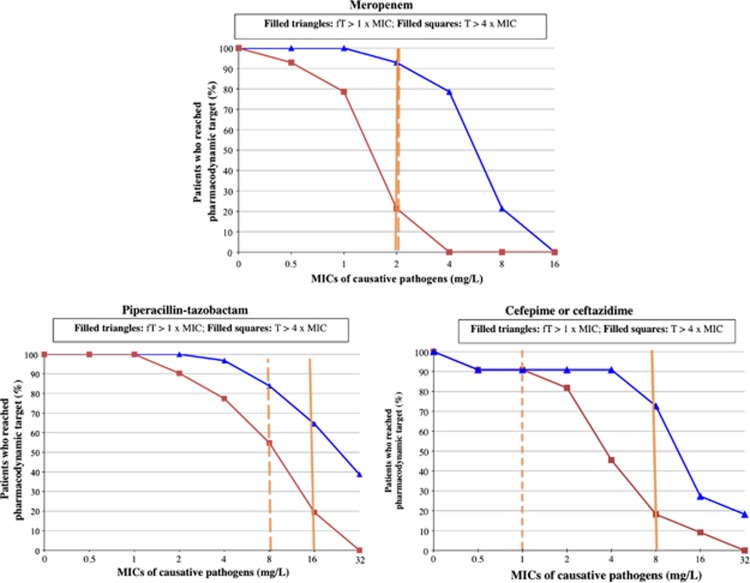
The PK variables of β-lactams obtained in our cohort of obese patients were compared with those found in the literature in non-obese patients,4, 13, 14, 16, 19, 20, 21, 22, 23, 24 which are presented in Table 2. Total serum concentrations and fT at 40%, 50% and 70% of the dosage intervals of MEM, TZP and CEF, respectively, were as follows: 6.2 (2.0–13.4) mg l−1 and 6.1 (2.0–13.1) mg l−1 for MEM, 36.7 (5.4–108.8) mg l−1 and 25.7 (3.8–76.2) mg l−1 for TZP and 16.1 (2–64.4) mg l−1 and 12.5 (2.0–54.7) mg l−1 for CEF. Figure 1 shows the percentage of patients that attained the 2 PD targets for the different MICs studied. When evaluating the fT>MIC for Enterobacteriacae spp., adequate serum concentrations were obtained in 93% of patients receiving MEM, 84% of patients receiving TZP and 91% of patients receiving CEF. For infections due to P. aeruginosa, adequate fT>MIC were obtained in 93% of patients receiving MEM, 68% of patients receiving TZP and 73% of patients receiving CEF. When we evaluated the T>4MIC for Enterobacteriacae spp., lower percentages of patients reached PD targets for all antibiotics, except for CEF: 21% for MEM, 55% for TZP and 91% for CEF. For infections due to P. aeruginosa, even fewer patients reached optimal T>4MIC for all antibiotics: 21% for MEM, 19% for TZP and 18% for CEF. In the post-hoc analysis, overall results were no different than those obtained when the sub-group of patients with neoplasia were excluded from the data analysis (data not shown).
Table 2. PK variables of MEM, TZP and CEF in the obese cohort, in those with a measured or estimated creatinine clearance greater than 80 ml min−1 and data in non-obese patients found in the literature.
| PK variables for each antibiotic | Obese cohort (N=56) | Obese cohort with CrCL24h or CrCLCG >80 ml min−1 (N=36) | Non-obese patients [4,13–14,16,19–24] |
|---|---|---|---|
| MEM | |||
| VD (L) | 48.0±25.1 | 54.6±23.6 | 12.5–17.8 |
| VD (L)/kg | 0.5±0.2 | 0.5±0.2 | 0.2 |
| CL (L/H) | 18.0±9.0 | 19.9±8.8 | 9.4–13.2 |
| t½ (H) | 2.0±0.9 | 2.0±0.9 | 1.0 |
| TZP | |||
| VD (L) | 31.4±26.0 | 26.9±15.9 | 8.2–15.8 |
| VD (L)/kg | 0.3±0.3 | 0.2±0.1 | 0.2 |
| CL (L/H) | 8.9±4.7 | 13.1±10.4 | 8.0–14.5 |
| t½ (H) | 2.2±1.8 | 1.5±1.4 | 0.6–1.1 |
| CEF | |||
| VD (L) | 33.9±27.7 | 42.9±31.6 | 14.3–19.3 |
| VD (L)/kg | 0.3±0.2 | 0.4±0.3 | 0.2 |
| CL (L/H) | 13.1±20.1 | 18.7±23.9 | 5.6–9.1 |
| t½ (H) | 3.1±2.2 | 2.2±0.8 | 1.3–2.0 |
Abbreviations: CEF, cefepime/ceftazidime; CL, total body clearance; CrCL24h, measured urinary creatinine clearance using 24-h urine collect; CrCLCG, calculated creatinine clearance using the Cockcroft-Gault equation; MEM, meropenem; PK, pharmacokinetic; t½: half-life; TZP, piperacillin-tazobactam; VD, volume of distribution.
Data for the obese cohort are expressed as mean values with standard deviations. Data for the non-obese patients are expressed in ranges of mean values found in the literature.
Figure 1.
Percent of patients who attained different pharmacodynamic targets for different minimal inhibition concentrations (MICs). fT, serum concentration of the free fraction of the β-lactam; T, serum concentration of the total fraction of the β-lactam. Solid vertical line, clinical susceptibility breakpoint for Pseudomonas aeruginosa, according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST); Dashed vertical line: clinical susceptibility breakpoint for Enterobacteriacae spp, according to EUCAST.
The CrCL24h was 107.0 (6.0–389.0) ml min−1: more than 50% of the patients had a CrCL24h >107 ml min−1 and over 25% of patients had a CrCL24h >150 ml min−1. CrCLCG and CrCL24h were compared. Four patients did not have a 24-h urine collection, and were thus excluded from this part of the analysis. CrCL obtained by CrCLCG (134 (26–469) ml min−1) was no different from CrCL24h (107 (6–389) ml min−1, P=0.86). However, 5 patients (10%) would have received even lower total daily doses if based on CrCL24h, and not on CrCLCG. When patients with a CrCL24h greater than 150 ml min−1 were compared with those with a CrCL24h less than 150 ml min−1, patients were not more frequently male ((8/13 (62%) versus 17/39 (44%), P=NS) and not younger (37 (18–77) versus 64 (26–87), P=NS). Plasma creatinine values were also similar (0.6 (0.3–1.0) mg dl−1 versus 1.0 (0.4–4.4) mg dl−1, P=NS).
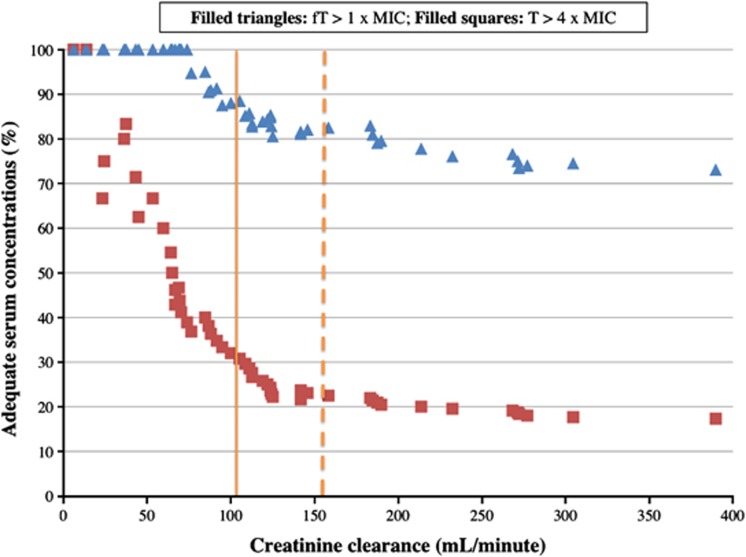
Risk factors for failure to reach PD targets for infections due to P. aeruginosa when standard doses were administered are shown in Table 3. Size descriptors were not identified as risk factors. High CrCL24h was the only factor detected by both univariate and multivariate analysis. The relationship between CrCL24h and the percentage of patients with adequate serum concentrations is shown in Figure 2: as CrCL24h increased, the percentage of patients with adequate serum concentrations decreased.
Table 3. Risk factors for lower β-lactam serum concentrations than pharmacodynamic targets of the free fraction greater than 1 times the MIC (fT >MIC) and the total serum concentration greater than 4 times the MIC (T >4MIC) of P. aeruginosa for 40%, 50% and 70% of dosing interval for meropenem, piperacillin-tazobactam and cefepime/ceftazidime, respectively.
| Variable | fT <MIC for specified time period (n=14) | fT >MIC for specified time period (n=42) |
Univariate analysis |
Multivariate analysis |
T <4MIC for specified time period (n=45) | T >4MIC for specified time period (n=11) |
Uni-variate analysis |
Multivariate analysis |
||
|---|---|---|---|---|---|---|---|---|---|---|
| P-value | P-value | OR (95% CI) | P-value | P-value | OR (95% CI) | |||||
| Male | 7 (50.0) | 21 (50.0) | 1.0 | 21(46.7) | 7 (63.6) | 0.32 | ||||
| Weight | 106 (91–141) | 103 (80–153) | 0.43 | 102.5 (85–152.5) | 110 (80–152.5) | 0.58 | ||||
| Body mass index (kg m−2) | 37 (30–46.1) | 35 (30–54) | 0.93 | 36 (30–48) | 38 (30–54) | 0.55 | ||||
| CrCL24h (ml min−1; n=52) | 155 (76–390) | 86 (6–305) | 0.01 | 0.01 | 0.980–0.996 | 113 (23–305) | 37 (6–142) | 0.003 | 0.006 | 0.929–0.987 |
| Early serum sampling | 6 (42.9) | 17 (40.5) | 0.88 | 17 (37.8) | 6 (54.6) | 0.32 | ||||
| Abdominal circumference (cm; n=47) | 127 (118–158) | 127 (99–177) | 0.83 | 125 (99–177) | 132 (124–161) | 0.03 | 0.067 | 0.997–1.104 | ||
| Body surface area (m2) | 2.2 (2–2.5) | 2.1 (1.8–2.6) | 0.25 | 2.1 (1.8–2.6) | 2.1 (1.9–2.6) | 0.68 | ||||
| Ideal body weight (kg) | 68 (54–78) | 60 (41–80) | 0.23 | 65 (41–78) | 60 (48–80) | 0.75 | ||||
| Fat-free mass (kg) | 58 (53–72) | 58 (47–74) | 0.25 | 58 (47–74) | 57 (52–74) | 0.68 | ||||
| Lean body weight (kg) | 64 (50–77) | 58 (35–80) | 0.46 | 58 (38–77) | 61 (35–80) | 0.72 | ||||
| Adjusted body weight (kg) | 82 (71–101) | 79 (61–105) | 0.26 | 80 (61–101) | 79 (70–105) | 0.61 | ||||
| Percent ideal body weight (%) | 176 (129–214) | 160 (131–272) | 0.75 | 162 (128–272) | 165 (129–225) | 0.77 | ||||
| Meropenem | 1 (7.1) | 13 (31.0) | 0.20 | 12 (26.7) | 2 (18.2) | 0.84 | ||||
| Piperacillin-tazobactam | 10 (71.4) | 21 (50.0) | 0.10 | 24 (53.3) | 7 (63.6) | 0.87 | ||||
| Cefepime or ceftazidime | 3 (21.4) | 8 (19.1) | 0.23 | 9 (20.0) | 2 (18.2) | 0.98 | ||||
Abbreviations: CI, confidence interval; CrCL24h, measured creatinine clearance with 24-h urine collect; MIC, minimal inhibition concentration; OR, odds ratio.
Data are expressed as counts (percentage) or median (range). Boldface indicates significant P-value.
Figure 2.
Percent of patients with greater serum concentrations than the pharmacodynamic target of the total serum concentration (T) greater than four times the minimal inhibition concentration (MIC; T>4MIC) and the free fraction serum concentration (fT) greater than 1 time the MIC (fT>MIC) of P. aeruginosa for 40%, 50% and 70% of the dosing interval for meropenem, piperacillin-tazobactam and cefepime/ceftazidime, respectively, in function of the measured creatinine clearance obtained from 24-h urine collects. Vertical solid line: 50% of patients have a clearance greater than 107.0 ml min−1; Vertical dashed line: 25% of patients have a clearance greater than 158.9 ml min−1.
Discussion
In our study, we showed that when standard doses were administered to infected, obese, non-critically ill patients, an insufficient number of patients treated with TZP or CEF reached therapeutic serum concentrations according to established criteria to treat an infection due to P. aeruginosa. However, when aiming to treat infections due to Enterobacteriaceae spp., only patients treated with TZP did not attain these therapeutic concentrations. When considering more severe PD targets (T>4MIC), only treatment of infections due to Enterobacteriaceae spp. with CEF permitted more than 90% of patients to attain therapeutic serum concentrations and none of the antibiotics attained the PD target when treating infections due to P. aeruginosa. The low antibiotic serum concentrations observed in our obese cohort was attributed to an increase in VD and CL for all of the studied drugs, compared with non-obese patients. In agreement with the observed increased drug CL, an augmented renal clearance (ARC), defined by a CrCL of more than 150 ml min−1, was found in 25% of our obese cohort. ARC was the only risk factor identified for failure to reach therapeutic concentrations for infections due to P. aeruginosa.
There are sparse data in the literature on the PK of broad-spectrum β-lactams in obese, non-critically ill patients: one PK study in 10 obese patients who received cefepime for prophylaxis during bariatric surgery,3 two case reports on the PK of TZP in 2 morbidly obese individuals4, 5 and one PK study on the administration of TZP as a prolonged infusion in 14 obese, infected individuals.6 Our data are concordant with these data, suggesting that PK parameters are altered in obese, non-critically ill patients. The VD and CL of the antibiotics in our cohort were increased compared with values reported in PK studies in non-obese patients.4, 13, 14, 16, 19, 20, 21, 22, 23, 24 Together, these data suggest that standard dosage regimens, particularly for TZP, are insufficient in obese, non-critically ill patients. Insufficient serum concentrations could be the cause for the lower cure rates observed by Zakrison et al. in obese patients when compared with non-obese patients treated with TZP for complicated intra-abdominal infections.25 The greater VD and CL may justify both higher loading and total daily doses in obese patients.
The t1/2 of the antibiotics in our cohort was also increased compared with values reported in PK studies in non-obese patients.4, 13, 14, 16, 19, 20, 21, 22, 23, 24 The t1/2 depends upon both the VD and CL, and changes in t1/2 may reflect changes in VD, CL or both. In our study, we believe that the increase in t1/2 was due to the increase in the VD, as suggested by Barbour et al. in a PK study on cefuroxime in obese patients.26 The VD in our study appears to have a greater impact on the t1/2 of the β-lactams studied than the increase in CL, because otherwise we should have observed no change or a decrease in t½.
We showed that when using EUCAST criteria for clinical efficacy, less than 90% of patients attained PD targets for TZP and CEF for the treatment of infections due to P. aeruginosa or less susceptible pathogens. Until now, the literature concerning the impact of inadequate antibiotic serum concentrations on the clinical cure of infections is sparse outside of the intensive care unit setting. However, the worldwide epidemiology is changing and the MICs of different pathogens are increasing over time. In the future, patients with lower antibiotic serum concentrations will be at greater risk for therapeutic failure, or selection of even more resistant pathogens.
The less frequently used PD target of T>4MIC was explored concomitantly to EUCAST's PD targets because certain clinical situations may justify the use of this more severe PD target to ensure therapeutic success and limit the emergence of resistance to antibiotics. In vitro studies have shown that maximum killing of P. aeruginosa occurs at four to six times the MIC.27, 28 A patient with septic shock from P. aeruginosa septicaemia was clinically cured only when serum concentrations of MEM reached T>4MIC of the P. aeruginosa for 40% of the dosing interval.12 Furthermore, 30% of our patients had malignancies and were undergoing oncological treatment; in vitro studies have confirmed that neutropenic individuals need higher PK/PD targets than non-neutropenic individuals for effective antibiotic treatment.29
The only risk factor for failure to reach PD targets, identified by univariate and multivariate analysis, was increased CrCL, but surprisingly not body size descriptors. A high CrCL24h was observed in our cohort despite a median age of 61 years and many co-morbidities (43% were diabetic and 32% hypertensive). The high CrCL was not more frequent in case of neoplasia or in function of demographic data (age, sex and so on), however, our cohort was small. More than half of the patients had a CrCL24h greater than 100 ml min−1, and 25% had a CrCL24h greater than 150 ml min−1, concordant with the observed increased antibiotic CL in our cohort compared with non-obese individuals. As CrCL increased, the percentage of patients with adequate serum concentrations decreased. Only patients with a CrCL of less than 80 ml min−1 had a high probability (>90%) of attaining therapeutic serum concentrations when treating infections due to P. aeruginosa.
ARC refers to enhanced renal elimination of circulating hydrophilic solutes and has been mostly described in critically ill patients. A strong association between increased CrCL and insufficient β-lactam concentrations has already been reported, suggesting the need for higher than recommended dosage regimens in this setting.30 Furthermore, ARC is associated with worse clinical outcome in critically ill patients receiving standard doses of antimicrobial therapy.31
Increased CrCL has also been described in the obese individual,32 with reported CrCL similar to those observed in our cohort. Salazar and Corcoran33 and Chew-Harris et al.34 reported significantly higher CrCL24h in obese than non-obese, non-infected patients (169.0±34.5 ml min−1 and 159.2 (112.4–211.9) ml min−1 in obese patients compared with 123.0±14.4 ml min−1 and 135.9 (104.6–195.6) ml min−1 in non-obese individuals, respectively). A median CrCL greater than 150 ml min−1 with an upper range up to 443 ml min−1 was also reported in small case series of obese individuals (healthy volunteers, infected and non-infected patients).6, 35, 36, 37 However, high CrCL has never been associated with insufficient β-lactam serum concentrations or therapeutic failure in this patient population. The only studies showing a correlation between CrCL and CL of a hydrophilic molecule in obese patients are PK studies on vancomycin, resulting in lower than predicted serum concentrations compared with normal-weight patients.37, 38
It has already been shown that none of the current existing equations to estimate glomerular filtration are adequate to properly identify ARC in the intensive care unit patient; ARC is best identified in the clinical setting by urine collects lasting several hours.30 In our study, we found no difference in measured and estimated CrCL, but our study was probably underpowered for this analysis. In other studies, CrCLCG has been shown to over-estimate CrCL24h in obese patients,32 thus providing an explanation why five patients in our cohort received greater doses of antibiotics than if their dosage regimen had been adapted to their CrCL24h. ARC has probably been overlooked in the obese patient population because very few studies actually measure or estimate CrCL in infected, obese, non-critically ill patients, particularly when patients appear to have a ‘normal' renal function such as a serum creatinine value less than 1.0 mg dl−1. Furthermore, PK studies in obese patients are very limited, and cohorts are small. More studies are needed to identify obese patients with ARC. However, even if a patient with ARC is identified, there is currently no recommended upward daily dose adjustment for patients with a CrCL >80 ml min−1.
Our study has some limitations. First, this is a preliminary, simplified PK study on a heterogeneous cohort in terms of type of infection, causative pathogens and time of blood sampling, not designed to evaluate the impact of adequate dosage on outcome. Furthermore, we calculated the percentage of patients who reached PD targets, instead of using techniques such as Monte–Carlo simulation to calculate the probability of PD target attainment.
Second, as in all other published series on obese, infected patients, the fT of β-lactams was not measured, but estimated, despite that the fT may be different in obese than in non-obese patients. Third, we did not perform a case–control study but compared our results to data from the literature. We did use PK/PD criteria for efficacy as proposed by EUCAST for the general population. If obese patients were like the general population in terms of PK parameters, more than 90% of our patients receiving MEM, TZP and CEF should have attained the PD target of fT>MIC. Fourth, only ARC was identified as a factor for insufficient serum concentrations by univariate and multivariate analysis. The sample size of our cohort may have been too small to identify other factors. Finally, the median BMI of our patients was 36 kg m−2, with only a very few patients with a BMI >50 kg m−2. Serum concentrations may be even lower in a more obese patient cohort.
This study was performed to determine whether or not a more detailed PK study on β-lactams in obese, infected not critically ill patients is warranted. Because our study does suggest that PKs of β-lactams are truly altered in obese patients, a PK study on obese and non-obese individuals, with detailed serum sampling, measurement of fT and utilization of techniques such as Monte–Carlo simulation is currently being carried out.
Conclusions
Standard doses of MEM, TZP and CEF resulted in serum concentrations too low to treat infections due to bacteria with high MICs in obese, non-critically ill patients. We identified ARC as the major risk factor for these low serum concentrations. Based on this study, we have initiated a more detailed PK study on broad-spectrum β-lactams in obese patients. PK variables and the impact of factors like ARC on this patient population will be better characterized to recommend appropriate daily doses in a near future.
Acknowledgments
We thank Mr H Njimi for his help with the statistics.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on the Nutrition & Diabetes website (http://www.nature.com/nutd)
Supplementary Material
References
- World Health Organization. Obesity . http://www.who.int/topics/obesity/en/ . Accessed on 13 June 2013.
- Janson B, Thursky K. Dosing of antibiotics in obesity. Curr Opin Infect Dis. 2012;25:634–649. doi: 10.1097/QCO.0b013e328359a4c1. [DOI] [PubMed] [Google Scholar]
- Rich BS, Keel R, Ho VP, Turbendian H, Afaneh CI, Dakin GF, et al. Cefepime dosing in the morbidly obese patient population. Obes Surg. 2012;22:465–471. doi: 10.1007/s11695-011-0586-8. [DOI] [PubMed] [Google Scholar]
- Newman D, Scheetz MH, Adeyemi OA, Montevecchi M, Nicolau DP, Noskin GA, et al. Serum piperacillin/tazobactam pharmacokinetics in a morbidly obese individual. Ann Pharmacother. 2007;41:1734–1739. doi: 10.1345/aph.1K256. [DOI] [PubMed] [Google Scholar]
- Deman H, Verhaegen J, Willems L, Spriet I. Dosing of piperacillin/tazobactam in a morbidly obese patient. J Antimicrob Chemother. 2012;67:782–783. doi: 10.1093/jac/dkr503. [DOI] [PubMed] [Google Scholar]
- Cheatham SC, Fleming MR, Healy DP, Chung CE, Shea KM, Humphrey ML, et al. Steady-state pharmacokinetics and pharmacodynamics of piperacillin and tazobactam administered by prolonged infusion in obese patients. Int J Antimicrob Agents. 2013;41:52–56. doi: 10.1016/j.ijantimicag.2012.09.004. [DOI] [PubMed] [Google Scholar]
- Hites M, Taccone FS, Wolff F, Cotton F, Beumier M, De Backer D, et al. Case-control study of drug monitoring of β-lactams in obese critically ill patients. Antimicrob Agents Chemother. 2013;57:708–715. doi: 10.1128/AAC.01083-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sturm AW, Allen N, Rafferty KD, Fish DN, Toschlog E, Newell M, et al. Pharmacokinetic analysis of Piperacillin administered with Tazobactam in Critically Ill, Morbidly Obese Surgical Patients. Pharmacotherapy. 2013;34:28–35. doi: 10.1002/phar.1324. [DOI] [PubMed] [Google Scholar]
- Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16:31–41. doi: 10.1159/000180580. [DOI] [PubMed] [Google Scholar]
- Wolff F, Deprez G, Seyler L, Taccone FS, Hites M, Gulbis B, et al. Rapid quantification of six beta-lactams to optimize dosage regimens in severely septic patients. Talanta. 2013;103:153–160. doi: 10.1016/j.talanta.2012.10.024. [DOI] [PubMed] [Google Scholar]
- Mouton JW, Brown DF, Apfalter P, Canton R, Giske CG, Ivanova M, et al. The role of pharmacokinetics/pharmacodynamics in setting clinical MIC breakpoints: the EUCAST approach. Clin Microbiol Infect. 2012;18:E37–E45. doi: 10.1111/j.1469-0691.2011.03752.x. [DOI] [PubMed] [Google Scholar]
- Taccone FS, Cotton F, Roisin S, Vincent JL, Jacobs F. Optimal meropenem concentrations to treat multidrug-resistant Pseudomonas aeruginosa septic shock. Antimicrob Agents Chemother. 2012;56:2129–2131. doi: 10.1128/AAC.06389-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drusano GL, Hutchison M. The pharmacokinetics of meropenem. Scand J Infect Dis. 1995;96 (Suppl 1:11–16. [PubMed] [Google Scholar]
- Sorgel F, Kinzig M. The chemistry, pharmacokinetics and tissue distribution of piperacillin/tazobactam. J Antimicrob Chemother. 1993;31 (Suppl A:39–60. doi: 10.1093/jac/31.suppl_a.39. [DOI] [PubMed] [Google Scholar]
- Crandon JL, Bulik CC, Kuti JL, Nicolau DP. Clinical pharmacodynamics of cefepime in patients infected with Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2010;54:1111–1116. doi: 10.1128/AAC.01183-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulfeuerborn W, Müller HJ, Borner K, Koeppe P, Lode H. Comparative pharmacokinetics and serum bactericidal activities of SCE-2787 and ceftazidime. Antimicrob Agents Chemother. 1993;37:1835–1841. doi: 10.1128/aac.37.9.1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- European Committee on Antimicrobial Susceptibility Testing Clinical breakpoints-bacteria http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/Breakpoint_table_v_2.0_120221.pdf . Accessed on 14 July 2013.
- Green B, Duffull SB. What is the best size descriptor to use for pharmacokinetic studies in the obese. Br J Clin Pharmacol. 2004;58:119–133. doi: 10.1111/j.1365-2125.2004.02157.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi Y, Roberts JA, Paterson DL, Lipman J. Pharmacokinetic evaluation of piperacillin-tazobactam. Expert Opin Drug Metab Toxicol. 2010;6:1017–1031. doi: 10.1517/17425255.2010.506187. [DOI] [PubMed] [Google Scholar]
- Bulitta JB, Duffull SB, Kinzig-Schippers M, Holzgrabe U, Stephan U, Drusano GL, et al. Systematic comparison of the population pharmacokinetics and pharmacodynamics of piperacillin in cystic fibrosis patients and healthy volunteers. Antimicrob Agents Chemother. 2007;51:2497–2507. doi: 10.1128/AAC.01477-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mouton JW, Horrevorts AM, Mulder PG, Prens EP, Michel MF. Pharmacokinetics of ceftazidime in serum and suction blister fluid during continuous and intermittent infusions in healthy volunteers. Antimicrob Agents Chemother. 1990;34:2307–2311. doi: 10.1128/aac.34.12.2307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbhaiya RH, Forgue ST, Gleason CR, Knupp CA, Pittman KA, Weilder DJ, et al. Pharmacokinetics of cefepime after single and multiple intravenous administrations in healthy subjects. Antimicrob Agents Chemother. 1992;36:552–557. doi: 10.1128/aac.36.3.552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsson-Ehle I, Hutchison M, Hawarth SJ, Norrby SR. Pharmacokinetics of meropenem compared to imipenem-cilastatin in young, healthy males. Eur J Clin Microbiol Infect Dis. 1991;10:85–88. doi: 10.1007/BF01964413. [DOI] [PubMed] [Google Scholar]
- Bax RP, Bastain W, Featherstone A, Wilkinson DM, Hutchinson M, Haworth SJ. The pharmacokinetics of meropenem in volunteers. J Antimicrob Chemother. 1989;24 (Suppl A:311–320. doi: 10.1093/jac/24.suppl_a.311. [DOI] [PubMed] [Google Scholar]
- Zakrison TL, Hille DA, Namias N. Effect of body mass index on treatment of complicated intra-abdominal infections in hospitalized adults: comparison of ertapenem with piperacillin-tazobactam. Surg Infect (Larchmt) 2012;13:38–42. doi: 10.1089/sur.2010.095. [DOI] [PubMed] [Google Scholar]
- Barbour A, Schmidt S, Rout WR, Ben-David K, Burkhardt O, Derendorf H. Soft tissue penetration of cefuroxime determined by clinical microdialysis in morbidly obese patients undergoing abdominal surgery. Int J Antimicrob Agents. 2009;34:231–235. doi: 10.1016/j.ijantimicag.2009.03.019. [DOI] [PubMed] [Google Scholar]
- Craig WA, Redington J, Ebert SC. Pharmacodynamics of amikacin in vitro and in mouse thigh and lung infections. J Antimicrob Chemother. 1991;27 (Suppl C:29–40. doi: 10.1093/jac/27.suppl_c.29. [DOI] [PubMed] [Google Scholar]
- Mouton JW, Punt N, Vinks AA. A retrospective analysis using Monte Carlo simulation to evaluate recommended ceftazidime dosing regimens in healthy volunteers, patients with cystic fibrosis, and patients in the intensive care unit. Clin Ther. 2005;27:762–772. doi: 10.1016/j.clinthera.2005.06.013. [DOI] [PubMed] [Google Scholar]
- Drusano GL, Liu W, Kulawy R, Louie A. Impact of granulocytes on the antimicrobial effect of tedizolid in a mouse thigh infection model. Antimicrob Agents Chemother. 2011;55:5300–5305. doi: 10.1128/AAC.00502-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Udy AA, Roberts JA, Boots RJ, Paterson DL, Lipman J. Augmented renal clearance: implications for antibacterial dosing in the critically ill. Clin Pharmacokinet. 2010;49:1–16. doi: 10.2165/11318140-000000000-00000. [DOI] [PubMed] [Google Scholar]
- Claus BO, Hoste EA, Colpaert K, Robays H, Decruyenaere J, De Waele JJ. Augmented renal clearance is a common finding with worse clinical outcome in critically ill patients receiving antimicrobial therapy. J Crit Care. 2013;28:695–700. doi: 10.1016/j.jcrc.2013.03.003. [DOI] [PubMed] [Google Scholar]
- Bosma RJ, Krikken JA, Homan van der Heide JJ, de Jong PE, Navis GJ. Obesity and renal hemodynamics. Contrib Nephrol. 2006;151:184–202. doi: 10.1159/000095329. [DOI] [PubMed] [Google Scholar]
- Salazar DE, Corcoran GB. Predicting creatinine clearance and renal drug clearance in obese patients from estimated fat-free body mass. Am J Med. 1988;84:1053–1060. doi: 10.1016/0002-9343(88)90310-5. [DOI] [PubMed] [Google Scholar]
- Chew-Harris JSC, Florkowski CM, George PM, Elmslie JL, Endre ZH. The relative effects of fat muscle mass on cystatin C and estimates of renal function in healthy young men. Ann Clin Biochem. 2013;50:39–46. doi: 10.1258/acb.2012.011241. [DOI] [PubMed] [Google Scholar]
- Dionne RE, Bauer LA, Gibson GA, Griffin WO, Blouin RA. Estimating creatinine clearance in morbidly obese patients. Am J Hosp Pharm. 38:841–844. [PubMed] [Google Scholar]
- Demirovic JA, Pai AB, Pai MP. Estimation of creatinine clearance in morbidly obese patients. Am J Health-Syst Pharm. 2009;66:642–648. doi: 10.2146/ajhp080200. [DOI] [PubMed] [Google Scholar]
- Blouin R, Bauer L, Miller D, Record K, Griffen W. Vancomycin pharmacokinetics in normal and morbidly obese subjects. Antimicrob Agents Chemother. 1982;21:575–580. doi: 10.1128/aac.21.4.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grace E. Altered vancomycin pharmacokinetics in obese and morbidly obese patients: what we have learned over the past 30 years. J Antimicrob Chemother. 2012;67:1305–1310. doi: 10.1093/jac/dks066. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.