Abstract
In addition to the classical motor symptoms, motivational and affective deficits are core impairments of Parkinson's disease (PD). We recently demonstrated, by lesional approaches in rats, that degeneration of the substantia nigra pars compacta (SNc) dopaminergic (DA) neurons is likely to have a crucial role in the development of these neuropsychiatry symptoms. We have also shown that, as in clinical investigations, chronic treatment with levodopa or the DA D2/D3 receptor (D2/D3R) agonist ropinirole specifically reverses these PD-related motivational deficits. The roles of specific DA receptor subtypes in such reversal effects remain, however, unknown. We therefore investigated here the precise involvement of D1, D2 and D3R in the reversal of the motivational and affective deficits related to SNc DA neuronal loss. Three weeks after bilateral and partial 6-hydroxydopamine (6-OHDA) SNc lesions, rats received 14 daily intraperitoneal administrations of the selective D1R agonist SKF-38393 (2.5 or 3.5 mg kg−1), the selective D2R agonist sumanirole (0.1 or 0.15 mg kg−1), or the preferring D3R gonist PD-128907 (0.1 or 0.15 mg kg−1). Anxiety-, depressive-like and motivated behaviors were assessed in an elevated-plus maze, a forced-swim test, and an operant sucrose self-administration procedure, respectively. All DA agonists attenuated anxiety- and depressive-like behaviors. However, only PD-128907 reversed the motivational deficits induced by 6-OHDA SNc lesions. This effect was blocked by a selective D3R (SB-277011A, 10 mg kg−1), but not D2R (L-741,626, 1.5 mg kg−1), antagonist. These data provide strong evidence for the role of D3R in motivational processes and identify this receptor as a potentially valuable target for the treatment of PD-related neuropsychiatric symptoms.
Introduction
Parkinson's disease (PD) is a motor disorder resulting from the progressive degeneration of midbrain dopaminergic (DA) neurons in the substantia nigra pars compacta (SNc). However, in addition to the classical motor symptoms of the disease, a plethora of neuropsychiatric and cognitive impairments, including apathy, contribute significantly to patient's high morbidity, impairing critically their quality of life.1 Apathy is observed in between 16.5 and 70% of PD patients, depending on the evaluation scale used and the population studied2,3 and as such, is considered to be a major neuropsychiatric feature of the disease.1 Apathy is classically defined as a lack of motivation, or a reduction in the ‘cognitive and emotional concomitants of goal-directed behaviors'.4 Apathetic patients indeed exhibit a global deficit in self-initiation and maintenance of voluntary and purposeful behavior, frequently accompanied by affective disorders, such as anxiety and depression,1,2,5 resulting in low levels of activity and a loss of interest in sources of reinforcement.6,7 A recent study revealed that apathy in PD and non PD patients is associated with an inability to translate expected reward into effort and action, with no change in the perception of reward value,7 indicating that the core element of apathy resides within the motivational preparatory processes responsible for initiating voluntary actions. In addition, PD-related apathetic, anxiety and depressive symptoms have been shown to fluctuate with DA replacement therapy, occurring particularly in conditions in which DA medications are highly reduced, whereas these symptoms can be reversed by treatments with DA agonists,1,2,5,6,8 suggesting a common underlying pathophysiological mechanism linked to DA neurotransmission in the basal ganglia.5,8,9
We have recently demonstrated that a part of these neuropsychiatric symptoms may stem directly from the loss of nigral DA neurons.10 Specifically, we found that partial bilateral 6-hydroxydopamine (6-OHDA) lesions of SNc DA neurons in rats induced depressive- and anxiety-like behaviors and impairments of motivated behaviors, reminiscent of the apathetic symptoms observed in PD patients. Indeed, evaluation of behavior in several operant and nonoperant tasks revealed a profound deficit in the preparatory aspect of instrumental responses, with changes neither in locomotor activity nor in reward and Pavlovian processes. Moreover, we have shown, as in clinical investigations,1,5,6 that this adverse phenotype was reversed by levodopa or the D2/D3R agonist ropinirole,10 confirming the critical implication of DA.
DA acts. however, through multiple receptor subtypes with differential roles in motor, limbic and cognitive functions, depending on their neuronal and brain localizations in corticostriatal and limbic circuits.11, 12, 13, 14 As such, D1R, D2R and D3R have been shown to have important roles in regulating affective,15, 16, 17, 18 reward-related and motivational processes.19, 20, 21 In the present study, we therefore set out to determine the respective contributions of these DA receptors in the reversal of the motivational and affective impairments induced by SNc DA neuronal loss. Using DA agonists selective for D1R (SKF-38393), D2R (sumanirole) or preferential for D3R (PD-128907),22, 23, 24, 25, 26 we showed that all DA agonists corrected the affective impairments, but only the preferential D3R agonist reversed the motivational deficits induced by the SNc DA lesions, providing a potential specific target for the treatment of PD-related neuropsychiatric symptoms.
Materials and methods
See Supplementary Material for details regarding, animals, drugs and reagents, surgery, immunohistochemistry, DA denervation analyses and behavioral procedures, as well as data and statistical analysis.
Bilateral 6-OHDA lesions
A total of 6 μg of 6-OHDA dissolved in 2.3 μl of sterile 0.9% NaCl (6-OHDA conditions) or 2.3 μl of sterile 0.9% NaCl (sham conditions) were bilaterally and stereotaxically infused into the SNc as described in the Supplementary Material, at the following coordinates relative to bregma:27 anterior-posterior, −5.4 mm; medial-lateral, ±1.8 mm and dorsal-ventral, −8.1 mm. The posterior placement of the injectors is a determinant factor for sparing a part of DA ascending fibers, and therefore to lead to a partial (<80%) DA denervation of the dorsal striatum that circumvent severe alterations of the motor function. Indeed, we previously shown that the SNc DA lesions with these parameters did not affect ambulatory behaviors, neither induced significant deficits in sensorimotor coordination or in stepping adjustement of the forepaws.10
Immunohistochemistry and DA denervation analyses
Immunohistochemical processing and analyses are described in the Supplementary Material. Coronal mesencephalic and striatal sections were processed for tyrosine hydroxylase (TH)- immunohistochemistry. Only TH-immunoreactivity (TH-IR) in the nucleus accumbens (NAc) and the dorsal striatum of sections localized around +1.2 mm anterior to bregma was quantified, as we previously observed that the dorsostriatal DA denervation at this level, is highly representative of the lesional extent and predictive of the motivational deficits.10 We have ensured that pharmacological treatment did not modify TH-IR in sham-operated animals (no effect of treatment and no treatment × structure interaction were found: Fs<1.26).
Experimental design
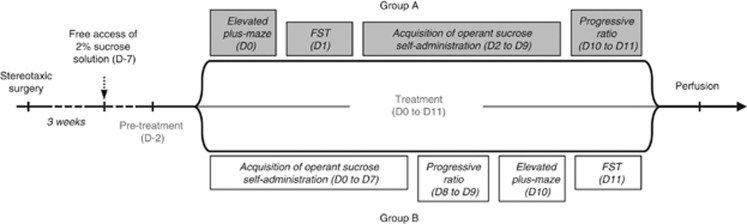
Three weeks after surgery and habituation to the 2% sucrose solution, rats were subjected to a sequence of behavioral tests, as summarized in Figure 1. Intraperitoneal administration of SKF-38393 (2.5 or 3.5 mg kg−1), sumanirole (0.1 or 0.15 mg kg−1), PD-128907 (0.1 or 0.15 mg kg−1) or vehicle (0.9% NaCl, 1 ml kg−1) began two days before the beginning of the behavioral sequence and were repeated daily for 14 days (Figure 1). Injections were carried out 30 min before the beginning of the behavioral sessions. Each of the selected doses has been shown to have electrophysiological or neurochemical and behavioral effects through a specific action on the preferred DR subtype.22,24, 25, 26,28,29,30,31
Figure 1.
Behavioral experiment schedules. Evaluation of the effects of different pharmacological treatments on the behavioral deficits induced by the 6-OHDA lesion. The behavioral studies began 3 weeks after surgery and tests were carried out in opposite orders for groups A and B. FST, forced-swim test; 6-OHDA, 6-hydroxydopamine.
Two subgroups of animals were constituted, the order of the tests being reversed in the second subgroup (see group A and B in Figure 1), to ensure that the effects of the lesion or of a pharmacological treatment did not change over the course of the study: half the animals were subjected first to the elevated-plus maze (EPM; day 0) and then the forced-swim test (FST; day 1), and operant sucrose self-administration (from day 2 to day 9) followed by a progressive ratio (PR) test (from day 10 to day 11), whereas the other half was subjected first to the self-administration procedure (from day 0 to day 7), followed by the PR test (from day 8 to day 9), the EPM (day 10) and the FST (day 11; Figure 1). No effect of order was found in any of the conditions tested (data not shown). In each experiment, all conditions (lesions and/or pharmacological treatments) were distributed among the different test chambers according to a Latin-square design.
This study was conducted in two separated experiments: a first one with the low dose of each agonists and a second with the higher doses. As the results obtained from sham-vehicle and 6-OHDA-vehicle rats were similar among the two experiments (data not shown), the data were pooled. The final number of subjects in each group for the sham and 6-OHDA condition, respectively, was as followed: n=21 and 13 for vehicle, n=12 and 8 for 2.5 mg kg−1 SKF-38393, n=9 and 10 for 3.5 mg kg−1 SKF-38393, n=9 and 8 for 0.10 mg kg−1 sumanirole, n=11 and 9 for 0.15 mg kg−1 sumanirole, n=13 and 12 for 0.10 mg kg−1 PD-128907 and n=12 and 10 for 0.15 mg kg−1 PD-128907.
At the end of the second experiment, the self-administration procedure was continued or resumed (if after evaluation of affective-related behaviors) for 10 days, for the groups sham and 6-OHDA vehicle, 3.5 mg kg−1 SKF-38393 and 0.15 mg kg−1 PD-128907. During this period, SKF-38393 administration were discontinued and replaced after 2 days of withdrawal by the administration of the high-efficacy D1R agonist SKF-8295820,22 for 8 days. PD-128907 continued to be administered 30 min before each self-administration session. On days 3, 5 and 8, PD-128907 administration in sham and lesioned animals was preceded by a subcutaneous injection of the respective D3R SB-277011A (10 mg kg−1) or D2R L-741626 (1.5 mg kg−1) antagonist,32 or vehicle (5% ethanol and 20% dimethyl sulfoxide in 0.9% NaCl, 1 ml kg−1), in a within-subject design. These doses were chosen on the basis of previous reports,25,32,33 and we had verified that they did not disrupt sucrose self-administration in sham-vehicle animals (no effect of treatment: F(2,14)=0.39, data not shown). A second PR test was performed at the end of this 10-day period and then, locomotor activity of 6-OHDA 0.15 mg kg−1 PD-128907 animals was evaluated in an open area for two more days, in combination with SB-277011A or its vehicle (within-subject design).
Results
6-OHDA-induced nigrostriatal DA lesions
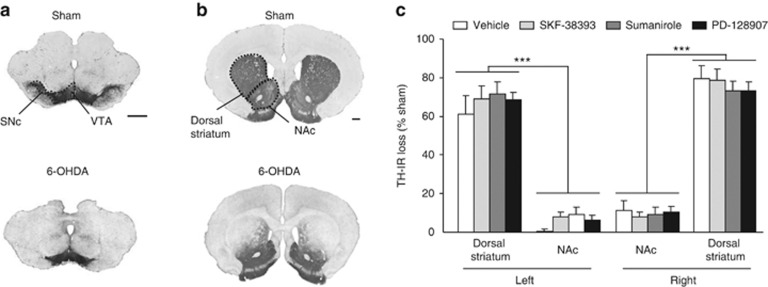
Bilateral 6-OHDA infusion into the SNc led to a loss of DA neurons within this area, as revealed by TH immunolabeling (Figure 2a), and a less than 80% concomitant decrease in TH-IR in the dorsal striatum (Supplementary Table S1 and Figures 2b and c, significant effect of lesion: F(1,702)=1066.01, P<0.001), predominantly in the lateral portion (Figure 2b). By contrast, the mesolimbic pathway was relatively well-preserved (Figures 2a and b), with a less than 15% TH-IR loss in NAc (Supplementary Table S1 and Figure 2c, significant effect of structure and significant lesion × structure interaction: Fs>240.86, Ps<0.001). We previously showed that such mild degeneration of this pathway did not alter accumbal extracellular DA levels,34 and did not provoke nor contribute to the behavioral phenotype induced by the 6-OHDA SNc lesions.10 Moreover, analogous patterns of DA striatal denervation were observed for each treatment condition (Supplementary Table S1 and Figure 2c, no effect of treatment, structure × treatment interaction, lesion × treatment interaction, or lesion × treatment × structure interaction: Fs<0.65).
Figure 2.
Bilateral partial lesions of the SNc result in a partial DA denervation of the dorsal striatum. (a, b) Photographs of coronal sections stained for TH at mesencephalic (a, anteriority −5.2 mm from bregma) and striatal (b, anteriority +1.2 mm from bregma) levels, according to Paxinos and Watson (2005). (c) Quantifications of the loss of TH staining in the NAc and the dorsal striatum for each treatment, independently of the dose used, expressed as the percentage of the mean value obtained for sham-operated animals. Scale bar, 1 mm. DA, dopamine; NAc, nucleus accumbens; 6-OHDA, 6-hydroxydopamine; SNc, substantia nigra pars compacta; TH, tyrosine hydroxylase; VTA, ventral tegmental area. ***P<0.001 (n=13–22 per group).
DA receptors stimulation reverses the affect-related impairments induced by 6-OHDA SNc lesions
We first tested whether systemic administration of SKF-38393 (selective D1R agonist), sumanirole (selective D2R agonist) or PD-128907 (preferring D3R agonist), would correct the anxiety- and depression-like behaviors induced by the SNc DA lesions.10
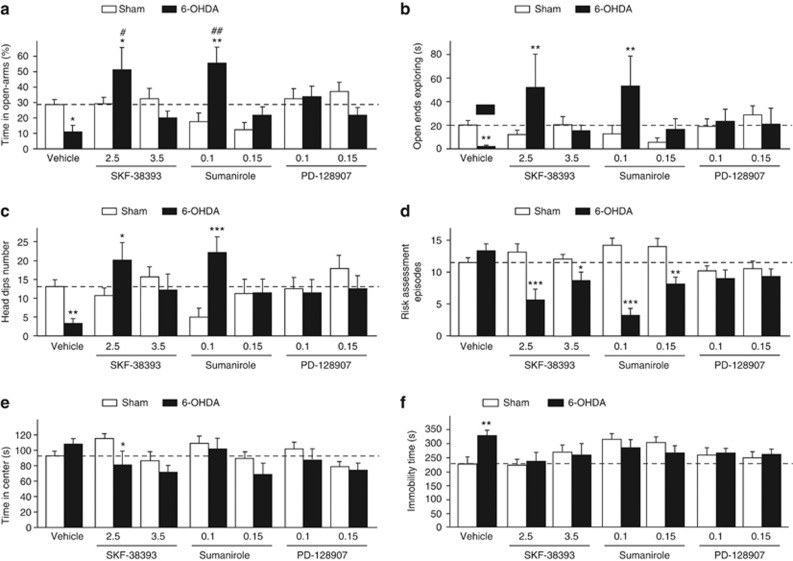
All DA receptor agonists fully reversed the decrease in the time spent in the open arms induced by the SNc lesion in an EPM (Figure 3a, significant lesion × treatment interaction: F(6,143)=5.49, P<0.001), as well as the decrease in the time spent exploring the end of the open arms and in the number of nonprotected head dips (Figures 3b and c, significant lesion × treatment interaction: Fs=2.77, Ps<0.02), indicating a decrease in anxiety-like behaviors in lesioned-treated animals. Depending on the dose, SKF-38393- and sumanirole even increased the time spent in the open arms of the EPM in lesioned rats compared with sham animals (Figure 3a), as well as nonprotected exploratory behaviors (Figures 3b and c). They also reduced the number of stretch attend postures toward the open arms (Figure 3d, significant lesion x treatment interaction: F(6,143)=8.98, P<0.001), a robust marker of risk assessment,35 and tended to decrease the time spent in the central area (Figure 3e, significant effect of treatment: F(6,143)=3.13, P<0.01 and no lesion × treatment interaction: F(6,143)=1.50), whereas PD-128907 normalized anxiety-related behaviors in lesioned-treated animals to control levels (Figures 3a–d). Conversely, sumanirole tends to reduce in sham animals exploration of the open arms (Figures 3a and b) and nonprotected head dipping (Figure 3c).
Figure 3.
Effects of dopaminergic agonists on affective-related deficits induced by 6-OHDA SNc lesions. Evaluation of the effects of subchronic administration of SKF-38193 (2.5 and 3.5 mg kg−1), sumanirole (0.1 and 0.15 mg kg−1) and PD-128907 (0.1 and 0.15 mg kg−1) in the elevated-plus maze (a–e) and the forced-swim test (f), in sham-operated (white histograms) and 6-OHDA SNc-lesioned (black histograms) animals. Indexes of anxiety-related behaviors evaluated here were the percentage of time spent in the open arms (a), the time spent specifically at the end of the open arms (b), the number of head dips when animals are exploring the open arms (c), the number of stretched attend episodes toward the open arms (d) and the time spent in the central area (e). Overall, DA agonists reversed the anxiety-like behaviors induced by 6-OHDA SNc lesions, as well as the increase in the time spent immobile in the forced-swim test. Dot lines represented the mean of the behavioral performances of vehicle-treated sham animals. *P<0.05; **P<0.01, ***P<0.01 sham-operated versus lesioned within the same treatment and #P<0.05, ##P<0.01 between the treatments for sham-operated and lesioned conditions, respectively (n=8–21 per group). 6-OHDA, 6-hydroxydopamine; PD, Parkinson's disease; SNc, substantia nigra pars compacta.
In addition, SKF-38393 and PD-128907 also reversed the depression-like behavior displayed in the FST (Figure 3f). Indeed, the method of contrasts (marginal lesion × treatment interaction: F(6,143)=2.07, P=0.06), found a significant increase in time spent immobile, a classical behavioral index of depression-like state,36 in vehicle-treated, but not in SKF-38393- or PD-128907-treated SNc-lesioned animals, when compared with their respective sham conditions. This effect was also observed in SNc-lesioned rats treated with sumanirole, but to a lesser extent as chronic administration of sumanirole tended to increase immobility time in sham animals (Figure 3f).
Only D3R agonist corrects the motivational deficits induced by 6-OHDA SNc lesions
We investigated whether sumanirole or PD-128907, as well as SKF-38393, could reverse the deficit in instrumental responses for sucrose self-administration induced by the SNc DA lesions, an impairment that was clearly linked to a motivational deficit and was notably reversed by the D2/D3R agonist ropinirole.10
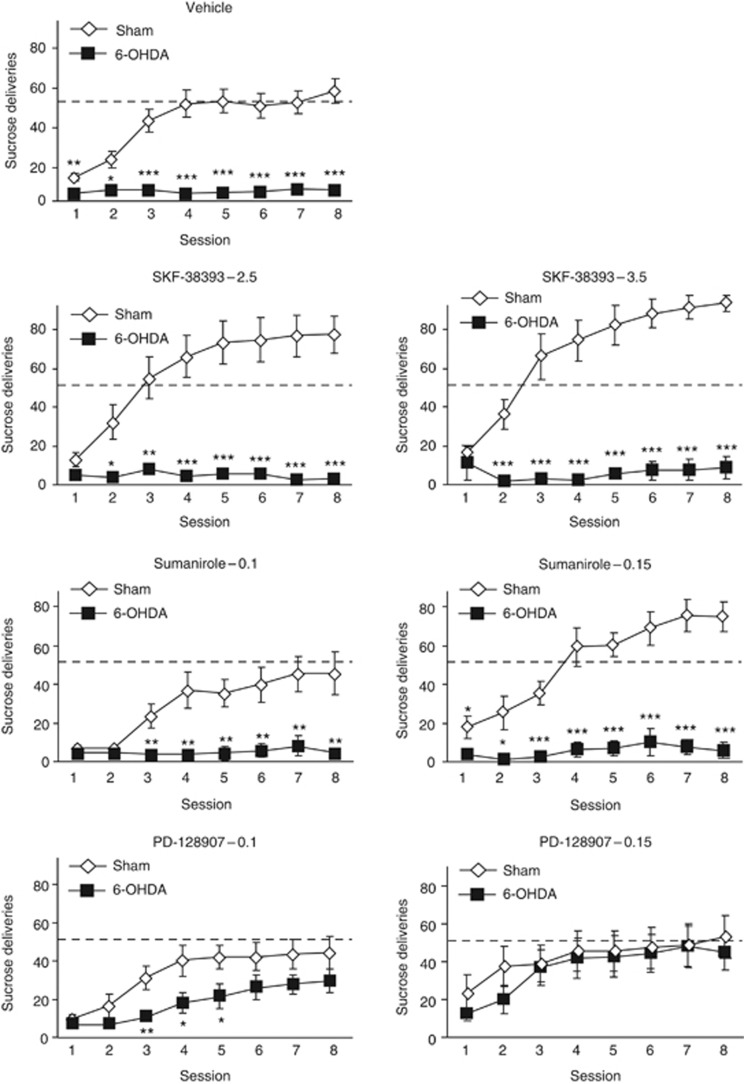
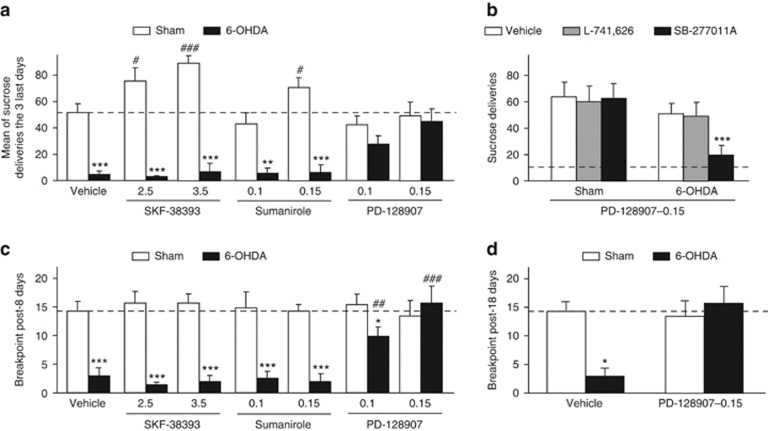
Analysis of operant performances session by session revealed that only PD-128907 reversed the deleterious impact of the SNc lesion on sucrose self-administration under a continuous schedule of reinforcement (Figure 4, vehicle, SKF-38393 and sumanirole treatments: significant effect of lesion and session × lesion interaction: Fs>5.26, Ps<0.001; 0.1 mg kg−1 PD-128907 treatment: significant effect of lesion: F(1,168)=7.68, P=0.01, but no session × lesion interaction: F(7,168)=0.81; 0.15 mg kg−1 PD-128907 treatment: no significant effect of lesion and interaction; Fs<0.22), progressively increasing the number of sucrose deliveries in lesioned animals, to eventually reach the level of reinforcers earned by sham animals (see also Figure 5a, significant lesion × treatment interaction: F(6,143)=8.08, P<0.001). The absence of positive effects of the other treatments in lesioned animals was particularly striking, as the D1R and D2R agonist dose-dependently improved the instrumental performances of the sham animals. Similar results were observed with the potent full D1R agonist SKF-8295822 (Supplementary Figure S1). Importantly, PD-128907 administration in 6-OHDA rats selectively increased the number of presses on the active lever over the number of presses on the inactive lever (effect of lever: Fs>14.95, P<0.004 and significant session × lever interaction: Fs>5.64, P<0.001; data not shown), indicating that PD-128907 did not induce a general increase in operant activity, but selectively toward the reinforced lever. This reversal effect of PD-128907 (0.15 mg kg−1) in SNc-lesioned animals was selectively blocked by the D3R antagonist SB-277011A, but not the D2R antagonist L-741626 (Figure 5b, significant lesion × treatment interaction: F(2,38)=9.15, P<0.001). This effect was not due to a disruptive action of SB-277011A in lesioned animals, as locomotor behavior was not affected by the antagonist (Supplementary Figure S1).
Figure 4.
Only the preferential D3R agonist, PD-128907, reverses the instrumental deficit induced by 6-OHDA SNc lesions. Evolution of operant sucrose (2% in water) self-administration performances, under a fixed ratio 1 (FR1) schedule of reinforcement, in sham-operated (white chips) and lesioned (black chips) animals, during the eight instrumental sessions. Subchronic administration of PD-128907 (0.1 and 0.15 mg kg−1), but not of SKF-38393 (2.5 and 3.5 mg kg−1) or sumanirole (0.1 and 0.15 mg kg−1), improved the instrumental performances of 6-OHDA-lesioned rats. Dot lines represented the mean of the instrumental performances of the three last FR1 sessions of vehicle-treated sham animals. *P<0.05; **P<0.01; ***P<0.001, sham-operated versus lesioned within the same treatment (n=8–21 per group). 6-OHDA, 6-hydroxydopamine; PD, Parkinson's disease; SNc, substantia nigra pars compacta.
Figure 5.
Reversion of the motivational deficits induced by 6-OHDA SNc lesions by the D3R agonist PD-128907 was blocked by the D3R antagonist SB-277011A, but not by the D2R antagonist L-741626. (a) Representation of the mean number of sucrose deliveries of the three last fixed ratio 1 (FR1) sessions in sham-operated (white histograms) and lesioned (black histograms) animals. Subchronic administration of PD-128907 (0.1 and 0.15 mg kg−1), but not of SKF-38393 (2.5 and 3.5 mg kg−1) or sumanirole (0.1 and 0.15 mg kg−1), improved instrumental performances of the 6-OHDA-lesioned rats. (b) SB-277011A (10 mg kg−1) but not L-741626 (1.5 mg kg−1) blocked specifically this reversal effect of PD-128907 (0.15 mg kg−1) in lesioned rats. (c) PD-128907, but not SKF-38393 or sumanirole enhanced the breakpoint (that is, last ratio completed) of 6-OHDA-lesioned rats after 8 days of sucrose self-administration. (d) A similar effect of PD-128907 (0.15 mg kg−1) was observed after 18 days of sucrose self-administration. Dot lines represented the mean of the instrumental performances of the three last FR1 sessions of vehicle-treated sham animals (a), the mean of the number of sucrose deliveries of vehicle-treated 6-OHDA-lesioned animals during antagonists tests (b) or the mean of the breakpoint of vehicle-treated 6-OHDA-lesioned animals (c–d). *P<0.05; **P<0.01; ***P<0.001, sham-operated versus lesioned within the same treatment and #P<0.05, ##P<0.01, ###P<0.001 between the treatments for sham-operated and lesioned conditions, respectively (n=8–21 per group). 6-OHDA, 6-hydroxydopamine; PD, Parkinson's disease; SNc, substantia nigra pars compacta.
Moreover, when the workload required to obtain the reward increased exponentially under a PR reinforcement schedule, an index of motivation,37 only PD-128907 significantly increased the performance of lesioned animals, leading to a partial or a full reversion of the deficit, depending of the dose administered (Figure 5c, significant lesion × treatment interaction: F(6,143)=5.08, P<0.001). Ten more days of PD-128907 (0.15 mg kg−1) administration, when responding for sucrose is stable, asymptotic and instrumental strategies become presumably exploitative,38 led to a similar reversal of PR performance deficits (Figure 5d, marginal lesion × treatment interaction: F(1,30)=3.67, P=0.06).
Discussion
By using DA agonists with different affinities for D1R, D2R and D3R, we show here a differential implication of these receptors in the reversal of the affective and motivational impairments induced by a DA denervation of the nigrostriatal system in rats. D1R, D2R and D3R agonists reversed the anxiety- and depressive-like behaviors induced by partial and bilateral 6-OHDA SNc lesions. The most important finding was that only the pharmacological stimulation of D3R reversed the instrumental and motivational deficits of lesioned animals, thereby suggesting a critical role for this receptor in the reversal of processes that are directly related to the pathophysiology of PD.
SKF-38393 is a well-characterized agonist of D1R, with a much higher selectivity for this receptor than for D2R and D3R,22 contrasting with sumanirole and PD-128907 that exhibit a very low affinity for D1R.24,26 Sumanirole also has a 250-fold selectivity for D2 over D3 receptors,26 but the affinity of PD-128907 for D3R is only 15 to 30 times higher than that for D2R in rats.23 Thus, sumanirole is unlikely to interact efficiently with D3R at relevant pharmacological concentrations, but PD-128907, depending on the dose used, may act as a D3R or a D2/D3R agonist.25,26,30 Pharmacological and genetic studies have provided strong evidence that two very characteristic effects of DA agonists, hypothermia and yawning, are mediated by D2R and D3R respectively.39, 40, 41 On the basis of these findings, Collins and colleagues39, 40, 41 elegantly demonstrated that systemic administration of PD-128907 in Sprague–Dawley rats, the strain used here, induced robust yawning with no effects on body temperature for doses ranging from 0.01 to 0.1 mg kg−1, thereby indicating a functional in vivo selective activation of D3R over D2R within this range of doses. In line with these results, it has been shown in D3R knockout mice, that PD-128907 must be administered at doses higher than 0.1 mg kg−1 to induce a significant D2 autoreceptors-mediated reduction of basal striatal DA extracellular levels.30 Consistently, Zapata et al.31 have shown in D3R knockout mice that low doses of PD-128907, ranging from 0.03 to 0.3 mg kg−1, reduce basal striatal DA extracellular levels through a selective activation of D3R. By using these selective agonists, together with a D3R preferring over D2R agonist at low doses, that selectively affect D3R but not D2R-mediated behaviors, we were able to accurately dissociate the effects of D1R, D2R and D3R activation on the behavioral deficits induced by a lesion of the DA nigrostriatal system.
We previously showed that anxiety- and depression-like behaviors induced by a DA SNc lesion can be reversed by levodopa and ropinirole.10 SKF-38393, sumanirole and PD- 128907 fully abolished the lesion-induced increase in anxiety observed in the EPM and exhibited beneficial effects in the FST, indicating a broad implication of DA receptors in the reversal of such affective impairments. It notably suggests that activation of both D2R and D3R may account for the reversal effects of ropinirole, a D2/D3R agonist, but that activation of D1R may, in addition, contribute to the effects of levodopa, which has, by its presynaptic mechanisms, no selective action on DA receptors. It has long been suggested that the DA function is involved in the physiopathology of depression and anxiety and as such, is considered as a potential target for the treatment of these disorders.42, 43, 44 However, its precise role has just begun to be clarified (for example, as seen in Tye et al.45). Notably, the specific role of the different DA receptor subtypes remains to be determined. Consistent with our data, loss of D1R in transgenic mice has been shown to promote anxiety-related behaviors,18 and the antidepressant-like effects of DA agents in the FST are at least partly mediated by D1R,16 identifying this receptor as a potentially interesting target for the treatment of affective disorders. The relationships between D2R and D3R and mood are less clear. Indeed, although D2/D3R agonists, such as pramipexole and ropinirole, have been shown to exhibit antidepressant and anxiolytic activities in preclinical models and clinical studies,1,10,46,47 conflicting results have been yielded concerning the relative contribution of D2R and D3R,15,17,47,48 probably due to the pre- and postsynaptic localization of these receptors15,16,49 that frequently leads to dual, opposite functions. Our data suggest that, at least under pathophysiological conditions, associated with a loss of or a decrease in presynaptic function and a complex postsynaptic redistribution of D2R and D3R following DA denervation and treatments,13,50,51 these receptors may be of particular interest for the treatment of affective-related disorders.
Such receptors modifications may also account for the strong anxiolytic properties that SKF-38393 and sumanirole acquired following DA denervation. Indeed, depending on the dose, the D1R and D2R agonist not only induced a dramatic increase in the time spent in the open arms of the EPM, but also strongly influences ethological factors related to disinhibition and risk-taking behaviors,34,52 by increasing head dippings and exploration in nonprotected areas and reducing risk assessments. Although it deserves further investigation, these data may provide useful information for understanding the iatrogenic mechanisms of DA medications that lead to the development of impulse control behaviors in PD.53
In addition to reversing affective impairments, PD-128907 was the only agonist which improved the instrumental deficits in lesioned rats, a beneficial effect blocked by a D3R, but not a D2R antagonist, thereby suggesting a specific implication of the D3R in the reversion of motivational impairments related to nigrostriatal DA denervation. It is also unlikely that the lack of effect of the D1R and D2R agonists resulted from the administration of too low doses. Indeed, the doses of SKF-38393, SKF-82958 and sumanirole used here have been shown to be behaviorally effective in previous studies,26,28,29,54 as well as in the present work. It is particularly striking for SKF-38393 that, despite its lack of effect on 6-OHDA-lesioned animals, significantly increased operant performance under a continuous ratio in controls, consistent with the function of D1R in conditioned reinforcement.20,55 Interestingly, this effect does not appear to be strictly motivational, as SKF-38393 did not lead to a higher breakpoint under a progressive ratio procedure. Taken with the results above, this differential implication of DA receptors suggests that the development of affective and motivational deficits following DA SNc lesioning may be underlain by dysfunctions of different neurobiological substrates. For instance, our previous study clearly associated the motivational impairments with striatal DA denervation, but mood alterations may result from a loss of DA signals in extrastriatal regions, such as the amygdala which also receives DA inputs from the SNc.10
Our data further suggest that D3R is crucial for mediating the beneficial effects of DA agonists on the behavioral alterations induced by nigrostriatal DA denervation. D3R is thought to significantly contribute to the control of affective and motivated behaviors and as such, has been identified as a potential target for the treatment of neuropsychiatric symptoms in several disorders,19 including PD.56,57 However, the relationship between D3R and motivation has been investigated mostly in the framework of drug addiction21 and behavioral evidences of a wider involvement of this receptor in affective and motivational functions are still lacking. Our data strongly suggest a pivotal role of D3R in the control of mood and motivation and in behavioral dysfunctions related to the pathophysiology of PD. Overall, these data identify D3R as an interesting pharmacological target for the treatment of apathy and affective disorders, such as anxiety and depression in PD.
In conclusion, this study provides new insights into the specific roles of DA receptors in motivational and affective functions, highlighting the critical role of D3R in the beneficial effects of DA agonists on the nonmotor behavioral alterations associated with nigral DA neuronal loss. Our data therefore support the use of D2/D3R agonists as effective treatments for PD-related neuropsychiatric symptoms and put forward D3R as a promising therapeutic target.
Acknowledgments
This work was supported by the Institut National de la Sante et de la Recherche Medicate, Fondation NeuroDis, Association France Parkinson, Ministere de la Recherche et de la Technologie (MRT), Region Rhone-Alpes (ARC 2), and Grenoble University. We would like to thank Pfizer Global Research and Development (request no. WS2025483) and Mr Donnie W Owens for providing sumanirole and we also thank Gaelle Frenois-Veyrat for technical assistance.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on the Translational Psychiatry website (http://www.nature.com/tp)
Supplementary Material
References
- Chaudhuri KR, Schapira AH. Non-motor symptoms of Parkinson's disease: dopaminergic pathophysiology and treatment. Lancet Neurol. 2009;8:464–474. doi: 10.1016/S1474-4422(09)70068-7. [DOI] [PubMed] [Google Scholar]
- Aarsland D, Marsh L, Schrag A. Neuropsychiatric symptoms in Parkinson's disease. Mov Disord. 2009;24:2175–2186. doi: 10.1002/mds.22589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leentjens AF, Dujardin K, Marsh L, Martinez-Martin P, Richard IH, Starkstein SE, et al. Apathy and anhedonia rating scales in Parkinson's disease: critique and recommendations. Mov Disord. 2008;23:2004–2014. doi: 10.1002/mds.22229. [DOI] [PubMed] [Google Scholar]
- Marin RS, Biedrzycki RC, Firinciogullari S. Reliability and validity of the Apathy Evaluation Scale. Psychiatry Res. 1991;38:143–162. doi: 10.1016/0165-1781(91)90040-v. [DOI] [PubMed] [Google Scholar]
- Thobois S, Ardouin C, Lhommee E, Klinger H, Lagrange C, Xie J, et al. Non-motor dopamine withdrawal syndrome after surgery for Parkinson's disease: predictors and underlying mesolimbic denervation. Brain. 2010;133:1111–1127. doi: 10.1093/brain/awq032. [DOI] [PubMed] [Google Scholar]
- Czernecki V, Schupbach M, Yaici S, Levy R, Bardinet E, Yelnik J, et al. Apathy following subthalamic stimulation in Parkinson disease: a dopamine responsive symptom. Mov Disord. 2008;23:964–969. doi: 10.1002/mds.21949. [DOI] [PubMed] [Google Scholar]
- Schmidt L, d'Arc BF, Lafargue G, Galanaud D, Czernecki V, Grabli D, et al. Disconnecting force from money: effects of basal ganglia damage on incentive motivation. Brain. 2008;131:1303–1310. doi: 10.1093/brain/awn045. [DOI] [PubMed] [Google Scholar]
- Lhommee E, Klinger H, Thobois S, Schmitt E, Ardouin C, Bichon A, et al. Subthalamic stimulation in Parkinson's disease: restoring the balance of motivated behaviours. Brain. 2012;135:1463–1477. doi: 10.1093/brain/aws078. [DOI] [PubMed] [Google Scholar]
- Volkmann J, Daniels C, Witt K. Neuropsychiatric effects of subthalamic neurostimulation in Parkinson disease. Nat Rev Neurol. 2010;6:487–498. doi: 10.1038/nrneurol.2010.111. [DOI] [PubMed] [Google Scholar]
- Drui G, Carnicella S, Carcenac C, Favier M, Bertrand A, Boulet S, et al. Loss of dopaminergic nigrostriatal neurons accounts for the motivational and affective deficits in Parkinson's disease. Mol Psychiatry. 2014;19:358–367. doi: 10.1038/mp.2013.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beaulieu JM, Gainetdinov RR. The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol Rev. 2011;63:182–217. doi: 10.1124/pr.110.002642. [DOI] [PubMed] [Google Scholar]
- Weiner DM, Levey AI, Sunahara RK, Niznik HB, O'Dowd BF, Seeman P, et al. D1 and D2 dopamine receptor mRNA in rat brain. Proc Natl Acad Sci USA. 1991;88:1859–1863. doi: 10.1073/pnas.88.5.1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guillin O, Diaz J, Carroll P, Griffon N, Schwartz JC, Sokoloff P. BDNF controls dopamine D3 receptor expression and triggers behavioural sensitization. Nature. 2001;411:86–89. doi: 10.1038/35075076. [DOI] [PubMed] [Google Scholar]
- Bentivoglio AR, Morelli M.The organization and circuits of mesencephalic dopaminergic neurons and the distribution of dopamine receptors in the brain. In: Dunnett SB, Bentivoglio AR, Bjorklund A, Hokfelt B (eds). Handbook of Chemical Neuroanatomy vol. 21Elsevier; 2005 [Google Scholar]
- de la Mora MP, Gallegos-Cari A, Arizmendi-Garcia Y, Marcellino D, Fuxe K. Role of dopamine receptor mechanisms in the amygdaloid modulation of fear and anxiety: structural and functional analysis. Prog Neurobiol. 2010;90:198–216. doi: 10.1016/j.pneurobio.2009.10.010. [DOI] [PubMed] [Google Scholar]
- D'Aquila PS, Galistu A. Possible role of dopamine D1-like and D2-like receptors in behavioural activation and evaluation of response efficacy in the forced swimming test. Neuropharmacology. 2012;62:1717–1729. doi: 10.1016/j.neuropharm.2011.11.018. [DOI] [PubMed] [Google Scholar]
- Basso AM, Gallagher KB, Bratcher NA, Brioni JD, Moreland RB, Hsieh GC, et al. Antidepressant-like effect of D(2/3) receptor-, but not D(4) receptor-activation in the rat forced swim test. Neuropsychopharmacology. 2005;30:1257–1268. doi: 10.1038/sj.npp.1300677. [DOI] [PubMed] [Google Scholar]
- Short JL, Ledent C, Drago J, Lawrence AJ. Receptor crosstalk: characterization of mice deficient in dopamine D1 and adenosine A2A receptors. Neuropsychopharmacology. 2006;31:525–534. doi: 10.1038/sj.npp.1300852. [DOI] [PubMed] [Google Scholar]
- Sokoloff P, Diaz J, Le Foll B, Guillin O, Leriche L, Bezard E, et al. The dopamine D3 receptor: a therapeutic target for the treatment of neuropsychiatric disorders. CNS Neurol Disord Drug Targets. 2006;5:25–43. doi: 10.2174/187152706784111551. [DOI] [PubMed] [Google Scholar]
- Katz JL, Kopajtic TA, Terry P. Effects of dopamine D1-like receptor agonists on food-maintained operant behavior in rats. Behav Pharmacol. 2006;17:303–309. doi: 10.1097/01.fbp.0000205015.67079.f7. [DOI] [PubMed] [Google Scholar]
- Newman AH, Blaylock BL, Nader MA, Bergman J, Sibley DR, Skolnick P. Medication discovery for addiction: translating the dopamine D3 receptor hypothesis. Biochem Pharmacol. 2012;84:882–890. doi: 10.1016/j.bcp.2012.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumeyer JL, Kula NS, Bergman J, Baldessarini RJ. Receptor affinities of dopamine D1 receptor-selective novel phenylbenzazepines. Eur J Pharmacol. 2003;474:137–140. doi: 10.1016/s0014-2999(03)02008-9. [DOI] [PubMed] [Google Scholar]
- van Vliet LA, Rodenhuis N, Dijkstra D, Wikstrom H, Pugsley TA, Serpa KA, et al. Synthesis and pharmacological evaluation of thiopyran analogues of the dopamine D3 receptor-selective agonist (4aR,10bR)-(+)-trans-3,4,4a,10b-tetrahydro-4-n-propyl- 2H,5H [1]b enzopyrano[4,3-b]-1,4-oxazin-9-ol (PD 128907) J Med Chem. 2000;43:2871–2882. doi: 10.1021/jm0000113. [DOI] [PubMed] [Google Scholar]
- Pugsley TA, Davis MD, Akunne HC, MacKenzie RG, Shih YH, Damsma G, et al. Neurochemical and functional characterization of the preferentially selective dopamine D3 agonist PD 128907. J Pharmacol Exp Ther. 1995;275:1355–1366. [PubMed] [Google Scholar]
- Collins GT, Newman AH, Grundt P, Rice KC, Husbands SM, Chauvignac C, et al. Yawning and hypothermia in rats: effects of dopamine D3 and D2 agonists and antagonists. Psychopharmacology (Berl) 2007;193:159–170. doi: 10.1007/s00213-007-0766-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCall RB, Lookingland KJ, Bedard PJ, Huff RM. Sumanirole, a highly dopamine D2- selective receptor agonist: in vitro and in vivo pharmacological characterization and efficacy in animal models of Parkinson's disease. J Pharmacol Exp Ther. 2005;314:1248–1256. doi: 10.1124/jpet.105.084202. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C.The Rat Brain in Stereotaxic Coordinates5th edn. Elsevier Academic Press: San Diego, CA, USA; 2005 [Google Scholar]
- Weber M, Chang WL, Breier MR, Yang A, Millan MJ, Swerdlow NR. The effects of the dopamine D2 agonist sumanirole on prepulse inhibition in rats. Eur Neuropsychopharmacol. 2010;20:421–425. doi: 10.1016/j.euroneuro.2010.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernando AB, Economidou D, Theobald DE, Zou MF, Newman AH, Spoelder M, et al. Modulation of high impulsivity and attentional performance in rats by selective direct and indirect dopaminergic and noradrenergic receptor agonists. Psychopharmacology (Berl) 2012;219:341–352. doi: 10.1007/s00213-011-2408-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koeltzow TE, Xu M, Cooper DC, Hu XT, Tonegawa S, Wolf ME, et al. Alterations in dopamine release but not dopamine autoreceptor function in dopamine D3 receptor mutant mice. J Neurosci. 1998;18:2231–2238. doi: 10.1523/JNEUROSCI.18-06-02231.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zapata A, Witkin JM, Shippenberg TS. Selective D3 receptor agonist effects of (+)-PD 128907 on dialysate dopamine at low doses. Neuropharmacology. 2001;41:351–359. doi: 10.1016/s0028-3908(01)00069-7. [DOI] [PubMed] [Google Scholar]
- van Gaalen MM, Unger L, Jongen-Relo AL, Schoemaker H, Gross G. Amphetamine decreases behavioral inhibition by stimulation of dopamine D2, but not D3, receptors. Behav Pharmacol. 2009;20:484–491. doi: 10.1097/FBP.0b013e3283305e3b. [DOI] [PubMed] [Google Scholar]
- Song R, Yang RF, Wu N, Su RB, Li J, Peng XQ, et al. YQA14: a novel dopamine D3 receptor antagonist that inhibits cocaine self-administration in rats and mice, but not in D3 receptor-knockout mice. Addict Biol. 2012;17:259–273. doi: 10.1111/j.1369-1600.2011.00317.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Favier M, Duran T, Carcenac C, Drui G, Savasta M, Carnicella S. Pramipexole reverses Parkinson's disease-related motivational deficits in rats. Mov Disord. advance online publication, 11 February 2014; doi:10.1002/mds.25837 (e-pub ahead of print). [DOI] [PubMed]
- Rodgers RJ, Johnsono NJT. Factor analysis of spatiotemporal and ethological measures in the murine elevated plus-maze test of anxiety. Pharmacol Biochem Behav. 1995;52:297–303. doi: 10.1016/0091-3057(95)00138-m. [DOI] [PubMed] [Google Scholar]
- Castagne V, Moser P, Roux S, Porsolt RD.Rodent models of depression: forced swim and tail suspension behavioral despair tests in rats and mice Curr Protoc Neurosci 2011Chapter 8, Unit 8.10A. [DOI] [PubMed] [Google Scholar]
- Hodos W. Progressive ratio as a measure of reward strength. Science. 1961;134:943–944. doi: 10.1126/science.134.3483.943. [DOI] [PubMed] [Google Scholar]
- Cohen JD, McClure SM, Yu AJ. Should I stay or should I go? How the human brain manages the trade-off between exploitation and exploration. Philos Trans R Soc Lond B Biol Sci. 2007;362:933–942. doi: 10.1098/rstb.2007.2098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulay D, Depoortere R, Perrault G, Borrelli E, Sanger DJ. Dopamine D2 receptor knock-out mice are insensitive to the hypolocomotor and hypothermic effects of dopamine D2/D3 receptor agonists. Neuropharmacology. 1999;38:1389–1396. doi: 10.1016/s0028-3908(99)00064-7. [DOI] [PubMed] [Google Scholar]
- Boulay D, Depoortere R, Rostene W, Perrault G, Sanger DJ. Dopamine D3 receptor agonists produce similar decreases in body temperature and locomotor activity in D3 knock-out and wild-type mice. Neuropharmacology. 1999;38:555–565. doi: 10.1016/s0028-3908(98)00213-5. [DOI] [PubMed] [Google Scholar]
- Collins GT, Witkin JM, Newman AH, Svensson KA, Grundt P, Cao J, et al. Dopamine agonist-induced yawning in rats: a dopamine D3 receptor-mediated behavior. J Pharmacol Exp Ther. 2005;314:310–319. doi: 10.1124/jpet.105.085472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willner P. The mesolimbic dopamine system as a target for rapid antidepressant action. Int Clin Psychopharmacol. 1997;12:S7–14. doi: 10.1097/00004850-199707003-00002. [DOI] [PubMed] [Google Scholar]
- Nestler EJ, Carlezon WA., Jr The mesolimbic dopamine reward circuit in depression. Biol Psychiatry. 2006;59:1151–1159. doi: 10.1016/j.biopsych.2005.09.018. [DOI] [PubMed] [Google Scholar]
- Durant C, Christmas D, Nutt D. The pharmacology of anxiety. Curr Top Behav Neurosci. 2010;2:303–330. doi: 10.1007/7854_2009_8. [DOI] [PubMed] [Google Scholar]
- Tye KM, Mirzabekov JJ, Warden MR, Ferenczi EA, Tsai HC, Finkelstein J, et al. Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature. 2013;493:537–541. doi: 10.1038/nature11740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers DC, Costall B, Domeney AM, Gerrard PA, Greener M, Kelly ME, et al. Anxiolytic profile of ropinirole in the rat, mouse and common marmoset. Psychopharmacology (Berl) 2000;151:91–97. doi: 10.1007/s002130000485. [DOI] [PubMed] [Google Scholar]
- Siuciak JA, Fujiwara RA. The activity of pramipexole in the mouse forced swim test is mediated by D2 rather than D3 receptors. Psychopharmacology (Berl) 2004;175:163–169. doi: 10.1007/s00213-004-1809-7. [DOI] [PubMed] [Google Scholar]
- Chourbaji S, Brandwein C, Vogt MA, Dormann C, Mueller R, Drescher KU, et al. Dopamine receptor 3 (D3) knockout mice show regular emotional behaviour. Pharmacol Res. 2008;58:302–307. doi: 10.1016/j.phrs.2008.09.002. [DOI] [PubMed] [Google Scholar]
- Bartoszyk GD. Anxiolytic effects of dopamine receptor ligands: I. Involvement of dopamine autoreceptors. Life Sci. 1998;62:649–663. doi: 10.1016/s0024-3205(97)01160-0. [DOI] [PubMed] [Google Scholar]
- Bezard E, Ferry S, Mach U, Stark H, Leriche L, Boraud T, et al. Attenuation of levodopa-induced dyskinesia by normalizing dopamine D3 receptor function. Nat Med. 2003;9:762–767. doi: 10.1038/nm875. [DOI] [PubMed] [Google Scholar]
- Savasta M, Dubois A, Benavides J, Scatton B. Different plasticity changes in D1 and D2 receptors in rat striatal subregions following impairment of dopaminergic transmission. Neurosci Lett. 1988;85:119–124. doi: 10.1016/0304-3940(88)90440-5. [DOI] [PubMed] [Google Scholar]
- Reymann JM, Naudet F, Pihan M, Saïkali S, Laviolle B, Bentué-Ferrer D. Subthalamic nucleus modulates social and anxiogenic-like behaviors. Behav Brain Res. 2013;252:356–362. doi: 10.1016/j.bbr.2013.05.059. [DOI] [PubMed] [Google Scholar]
- Voon V, Potenza MN, Thomsen T. Medication-related impulse control and repetitive behaviors in Parkinson's disease. Curr Opin Neurol. 2007;20:484–492. doi: 10.1097/WCO.0b013e32826fbc8f. [DOI] [PubMed] [Google Scholar]
- Cooper SJ, Al-Naser HA. Dopaminergic control of food choice: contrasting effects of SKF 38393 and quinpirole on high-palatability food preference in the rat. Neuropharmacology. 2006;50:953–963. doi: 10.1016/j.neuropharm.2006.01.006. [DOI] [PubMed] [Google Scholar]
- El-Ghundi M, O'Dowd BF, Erclik M, George SR. Attenuation of sucrose reinforcement in dopamine D1 receptor deficient mice. Eur J Neurosci. 2003;17 (4:851–862. doi: 10.1046/j.1460-9568.2003.02496.x. [DOI] [PubMed] [Google Scholar]
- Leentjens AF, Koester J, Fruh B, Shephard DT, Barone P, Houben JJ. The effect of pramipexole on mood and motivational symptoms in Parkinson's disease: a meta- analysis of placebo-controlled studies. Clin Ther. 2009;31:89–98. doi: 10.1016/j.clinthera.2009.01.012. [DOI] [PubMed] [Google Scholar]
- Joyce JN. Dopamine D3 receptor as a therapeutic target for antipsychotic and antiparkinsonian drugs. Pharmacol Ther. 2001;90:231–259. doi: 10.1016/s0163-7258(01)00139-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.