Abstract
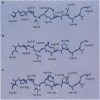
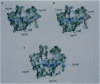
Molecular mimicry, normally defined by the level of primary-sequence similarities between self and foreign antigens, has been considered a key element in the pathogenesis of autoimmunity. Here we describe an example of molecular mimicry between two overlapping peptides within a single self-antigen, both of which are recognized by the same human self-reactive T-cell clone. Two intervening peptides did not stimulate the T-cell clone, even though they share nine amino acids with the stimulatory peptides. Molecular modeling of major histocompatibility complex class II-peptide complexes suggests that both of the recognized peptides generate similar antigenic surfaces, although these are composed of different sets of amino acids. The molecular modeling of a peptide shifted one residue from the stimulatory peptide, which was recognized in the context of the same HLA molecule by another T-cell clone, generated a completely different antigenic surface. Functional studies using truncated peptides confirmed that the anchor residues of the two "mimicking" epitopes in the HLA groove differ. Our results show, for two natural epitopes, how molecular mimicry can occur and suggest that studies of potential antigenic surfaces, rather than sequence similarity, are necessary for analyzing suspected peptide mimicry.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander J., Snoke K., Ruppert J., Sidney J., Wall M., Southwood S., Oseroff C., Arrhenius T., Gaeta F. C., Colón S. M. Functional consequences of engagement of the T cell receptor by low affinity ligands. J Immunol. 1993 Jan 1;150(1):1–7. [PubMed] [Google Scholar]
- Altmann D. M., Sansom D., Marsh S. G. What is the basis for HLA-DQ associations with autoimmune disease? Immunol Today. 1991 Aug;12(8):267–270. doi: 10.1016/0167-5699(91)90124-C. [DOI] [PubMed] [Google Scholar]
- Bertoletti A., Sette A., Chisari F. V., Penna A., Levrero M., De Carli M., Fiaccadori F., Ferrari C. Natural variants of cytotoxic epitopes are T-cell receptor antagonists for antiviral cytotoxic T cells. Nature. 1994 Jun 2;369(6479):407–410. doi: 10.1038/369407a0. [DOI] [PubMed] [Google Scholar]
- Bhardwaj V., Kumar V., Geysen H. M., Sercarz E. E. Degenerate recognition of a dissimilar antigenic peptide by myelin basic protein-reactive T cells. Implications for thymic education and autoimmunity. J Immunol. 1993 Nov 1;151(9):5000–5010. [PubMed] [Google Scholar]
- Brown J. H., Jardetzky T. S., Gorga J. C., Stern L. J., Urban R. G., Strominger J. L., Wiley D. C. Three-dimensional structure of the human class II histocompatibility antigen HLA-DR1. Nature. 1993 Jul 1;364(6432):33–39. doi: 10.1038/364033a0. [DOI] [PubMed] [Google Scholar]
- Davis M. M., Bjorkman P. J. T-cell antigen receptor genes and T-cell recognition. Nature. 1988 Aug 4;334(6181):395–402. doi: 10.1038/334395a0. [DOI] [PubMed] [Google Scholar]
- Dayan C. M., Londei M., Corcoran A. E., Grubeck-Loebenstein B., James R. F., Rapoport B., Feldmann M. Autoantigen recognition by thyroid-infiltrating T cells in Graves disease. Proc Natl Acad Sci U S A. 1991 Aug 15;88(16):7415–7419. doi: 10.1073/pnas.88.16.7415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Magistris M. T., Alexander J., Coggeshall M., Altman A., Gaeta F. C., Grey H. M., Sette A. Antigen analog-major histocompatibility complexes act as antagonists of the T cell receptor. Cell. 1992 Feb 21;68(4):625–634. doi: 10.1016/0092-8674(92)90139-4. [DOI] [PubMed] [Google Scholar]
- Dyrberg T., Petersen J. S., Oldstone M. B. Immunological cross-reactivity between mimicking epitopes on a virus protein and a human autoantigen depends on a single amino acid residue. Clin Immunol Immunopathol. 1990 Feb;54(2):290–297. doi: 10.1016/0090-1229(90)90090-d. [DOI] [PubMed] [Google Scholar]
- Fairchild P. J., Wildgoose R., Atherton E., Webb S., Wraith D. C. An autoantigenic T cell epitope forms unstable complexes with class II MHC: a novel route for escape from tolerance induction. Int Immunol. 1993 Sep;5(9):1151–1158. doi: 10.1093/intimm/5.9.1151. [DOI] [PubMed] [Google Scholar]
- Fremont D. H., Matsumura M., Stura E. A., Peterson P. A., Wilson I. A. Crystal structures of two viral peptides in complex with murine MHC class I H-2Kb. Science. 1992 Aug 14;257(5072):919–927. doi: 10.1126/science.1323877. [DOI] [PubMed] [Google Scholar]
- Fujinami R. S., Oldstone M. B. Molecular mimicry as a mechanism for virus-induced autoimmunity. Immunol Res. 1989;8(1):3–15. doi: 10.1007/BF02918552. [DOI] [PubMed] [Google Scholar]
- Garrett T. P., Saper M. A., Bjorkman P. J., Strominger J. L., Wiley D. C. Specificity pockets for the side chains of peptide antigens in HLA-Aw68. Nature. 1989 Dec 7;342(6250):692–696. doi: 10.1038/342692a0. [DOI] [PubMed] [Google Scholar]
- Gautam A. M., Pearson C. I., Smilek D. E., Steinman L., McDevitt H. O. A polyalanine peptide with only five native myelin basic protein residues induces autoimmune encephalomyelitis. J Exp Med. 1992 Aug 1;176(2):605–609. doi: 10.1084/jem.176.2.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hogquist K. A., Jameson S. C., Heath W. R., Howard J. L., Bevan M. J., Carbone F. R. T cell receptor antagonist peptides induce positive selection. Cell. 1994 Jan 14;76(1):17–27. doi: 10.1016/0092-8674(94)90169-4. [DOI] [PubMed] [Google Scholar]
- Jorgensen J. L., Esser U., Fazekas de St Groth B., Reay P. A., Davis M. M. Mapping T-cell receptor-peptide contacts by variant peptide immunization of single-chain transgenics. Nature. 1992 Jan 16;355(6357):224–230. doi: 10.1038/355224a0. [DOI] [PubMed] [Google Scholar]
- Kagnoff M. F., Austin R. K., Hubert J. J., Bernardin J. E., Kasarda D. D. Possible role for a human adenovirus in the pathogenesis of celiac disease. J Exp Med. 1984 Nov 1;160(5):1544–1557. doi: 10.1084/jem.160.5.1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klenerman P., Rowland-Jones S., McAdam S., Edwards J., Daenke S., Lalloo D., Köppe B., Rosenberg W., Boyd D., Edwards A. Cytotoxic T-cell activity antagonized by naturally occurring HIV-1 Gag variants. Nature. 1994 Jun 2;369(6479):403–407. doi: 10.1038/369403a0. [DOI] [PubMed] [Google Scholar]
- Kwok W. W., Mickelson E., Masewicz S., Milner E. C., Hansen J., Nepom G. T. Polymorphic DQ alpha and DQ beta interactions dictate HLA class II determinants of allo-recognition. J Exp Med. 1990 Jan 1;171(1):85–95. doi: 10.1084/jem.171.1.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Londei M., Bottazzo G. F., Feldmann M. Human T-cell clones from autoimmune thyroid glands: specific recognition of autologous thyroid cells. Science. 1985 Apr 5;228(4695):85–89. doi: 10.1126/science.3871967. [DOI] [PubMed] [Google Scholar]
- Matzinger P. Tolerance, danger, and the extended family. Annu Rev Immunol. 1994;12:991–1045. doi: 10.1146/annurev.iy.12.040194.005015. [DOI] [PubMed] [Google Scholar]
- Nepom G. T., Erlich H. MHC class-II molecules and autoimmunity. Annu Rev Immunol. 1991;9:493–525. doi: 10.1146/annurev.iy.09.040191.002425. [DOI] [PubMed] [Google Scholar]
- Sette A., Alexander J., Ruppert J., Snoke K., Franco A., Ishioka G., Grey H. M. Antigen analogs/MHC complexes as specific T cell receptor antagonists. Annu Rev Immunol. 1994;12:413–431. doi: 10.1146/annurev.iy.12.040194.002213. [DOI] [PubMed] [Google Scholar]
- Shimoda S., Nakamura M., Ishibashi H., Hayashida K., Niho Y. HLA DRB4 0101-restricted immunodominant T cell autoepitope of pyruvate dehydrogenase complex in primary biliary cirrhosis: evidence of molecular mimicry in human autoimmune diseases. J Exp Med. 1995 May 1;181(5):1835–1845. doi: 10.1084/jem.181.5.1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern L. J., Brown J. H., Jardetzky T. S., Gorga J. C., Urban R. G., Strominger J. L., Wiley D. C. Crystal structure of the human class II MHC protein HLA-DR1 complexed with an influenza virus peptide. Nature. 1994 Mar 17;368(6468):215–221. doi: 10.1038/368215a0. [DOI] [PubMed] [Google Scholar]
- Sutcliffe M. J., Haneef I., Carney D., Blundell T. L. Knowledge based modelling of homologous proteins, Part I: Three-dimensional frameworks derived from the simultaneous superposition of multiple structures. Protein Eng. 1987 Oct-Nov;1(5):377–384. doi: 10.1093/protein/1.5.377. [DOI] [PubMed] [Google Scholar]
- Sutcliffe M. J., Hayes F. R., Blundell T. L. Knowledge based modelling of homologous proteins, Part II: Rules for the conformations of substituted sidechains. Protein Eng. 1987 Oct-Nov;1(5):385–392. doi: 10.1093/protein/1.5.385. [DOI] [PubMed] [Google Scholar]
- Theofilopoulos A. N. The basis of autoimmunity: Part I. Mechanisms of aberrant self-recognition. Immunol Today. 1995 Feb;16(2):90–98. doi: 10.1016/0167-5699(95)80095-6. [DOI] [PubMed] [Google Scholar]
- Tian J., Lehmann P. V., Kaufman D. L. T cell cross-reactivity between coxsackievirus and glutamate decarboxylase is associated with a murine diabetes susceptibility allele. J Exp Med. 1994 Nov 1;180(5):1979–1984. doi: 10.1084/jem.180.5.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wraith D. C., Bruun B., Fairchild P. J. Cross-reactive antigen recognition by an encephalitogenic T cell receptor. Implications for T cell biology and autoimmunity. J Immunol. 1992 Dec 1;149(11):3765–3770. [PubMed] [Google Scholar]
- Wucherpfennig K. W., Strominger J. L. Molecular mimicry in T cell-mediated autoimmunity: viral peptides activate human T cell clones specific for myelin basic protein. Cell. 1995 Mar 10;80(5):695–705. doi: 10.1016/0092-8674(95)90348-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamvil S. S., Mitchell D. J., Moore A. C., Kitamura K., Steinman L., Rothbard J. B. T-cell epitope of the autoantigen myelin basic protein that induces encephalomyelitis. Nature. 1986 Nov 20;324(6094):258–260. doi: 10.1038/324258a0. [DOI] [PubMed] [Google Scholar]