Abstract
Tumor cells employ various methods of immune suppression to overcome antitumor immunity. One such method is that of programmed death ligand-1 (PD-L1 or B7-H1), which upon binding its receptor programmed death-1 (PD-1) on T cells triggers apoptotic death of the activated T cells. Over-expression of the costimulatory molecule CD80 on PD-L1+ tumor cells, or inclusion of a soluble form of CD80 (CD80-Fc) maintains the activation of PD-1+ activated T cells. Using T cells from CD28-deficient mice and antibodies to block CD28 on human T cells, we now report that a soluble form of CD80 mediates this effect by simultaneously neutralizing the PD-1/PD-L1-mediated immune suppression and by providing CD80-CD28 costimulation. In summary, PD-L1 is a potent mediator of immune suppression that inhibits antitumor immunity in many cancer patients. In this study, we show that a soluble form of the costimulatory molecule CD80 increases the production of IFNγ by PD-1+ activated T cell more effectively than antibodies to PD-1 or PD-L1. Therefore, soluble CD80 may be a more effective therapeutic than these checkpoint antibodies for facilitating the development and maintenance of antitumor immunity because it has the dual functions of preventing PD-L1-mediated immune suppression and simultaneously delivering the second signal for T-cell activation.
Keywords: Tumor immunity, T cell activation, T cells, immune suppression
Introduction
Programmed death ligand-1 (PD-L1), also known as B7 homolog 1 (B7-H1) or CD274, is expressed by many human and mouse tumor cells either constitutively or in response to exposure to IFNγ (1, 2). Expression of PD-L1 results in the suppression of antitumor immunity through multiple mechanisms. PD-L1 renders tumor cells resistant to cytotoxic T lymphocyte (CTL) and FasL-mediated lysis (3). It also induces apoptosis of activated T cells by signaling through its receptor PD-1. PD-L1 also reverse signals through T cell-expressed CD80 to anergize T cells, and its expression promotes the induction and expansion of regulatory T cells (Tregs) (1, 4-7). Some T and B cells, dendritic cells, Tregs, macrophages, and myeloid-derived suppressor cells (MDSC) may also express PD-L1 (8-10), and thus contribute to the inhibition of antitumor immunity.
Human and mouse tumor cells modified to express CD80 as an integral membrane protein prevent PD-L1 from binding its receptor PD-1 (11, 12). As a result, PD-1+ T cells remain activated. Treatment with a fusion protein consisting of the extracellular domains of CD80 fused to the Fc region of human IgG1 (CD80-Fc) similarly maintains the viability of activated PD-1+ T cells (12).
In addition to overcoming suppression by PD-L1, membrane-bound CD80 or CD80-Fc has the potential to costimulate T-cell activation via T cell-expressed CD28 (13). We now report that CD80-Fc maintains the activation of PD-1+ T cells by simultaneously preventing PD-1/PD-L1 suppression and by providing costimulation through CD28, and is more effective than antibodies to PD-L1 or PD-1 for maintaining IFNγ production by activated T cells. These findings suggest the potential of CD80-Fc as a therapeutic agent to overcome immune suppression and sustain antitumor immunity.
Materials and Methods
Cell lines and transfections
Human cutaneous melanoma cell line C8161 was kindly provided by Dr. Elisabeth Seftor (Children's Memorial Research Center, Northwestern University) in 2011 and was cultured as described (11). Since C8161 cells were not obtained from a cell bank, they cannot be authenticated; however, the line has maintained a unique profile by STR analysis and is routinely tested for mycoplasma infection. C8161/CD80 transfectants were generated and maintained as described (11). Cell lines and procedures with human materials were approved by the UMBC Institutional Review Board.
Mice
Breeding stock for C57BL/6 and CD28-deficient C57BL/6 (CD28−/−) mice were from The Jackson Laboratory. Mice were bred and maintained in the UMBC animal facility. All animal procedures were approved by the UMBC Institutional Animal Care and Use Committee.
Antibodies, reagents, and flow cytometry
Mouse CD3-Pacific Blue (clone 17A2), mouse CD28-PE-Cy7 (clone E18), mouse PD-1-APC (clone RMP1-30), mouse PD-L1-PE (clone 10F.9G2), mouse CD152-APC (clone UC10-4B9), and low endotoxin, azide-free human CD80 (clone 2D10) monoclonal antibodies (mAb) were from BioLegend (San Diego, CA). Mouse CD69-FITC (clone H1.2F3) and functional grade mouse IgG1 (clone P3.6.2.8.1) were from BD Biosciences (San Jose, CA) and eBioscience (San Diego, CA), respectively. Anti-PD-L1 (clone 5H1) was provided by Dr. Eugene Kwon (Mayo Clinic). Anti-human IgG1-Alexa Fluor 488 and anti-mouse IgG-Alexa Fluor 647 polyclonal antibodies were from Invitrogen (Grand Island, NY). Cells were stained for cell-surface expression and subjected to flow cytometry as described (14, 15), and analyzed using a Beckman Coulter Cyan ADP flow cytometer and Summit V4.3. For internal staining, cells were fixed with 2% paraformaldehyde and permeabilized prior to staining.
Human PBMC activation
Cryopreserved PBMC were obtained from healthy human donors as described (16). PBMC (6×104) and tumor cells (50 Gy-irradiated, 3×104) were co-cultured in 96-well plates as described (11). Human CD80-Fc, human TROY-Fc (TNF receptor superfamily member) recombinant fusion proteins (R&D Systems, Minneapolis, MN), and antibodies to human PD-L1 (clone 29E.2A3, BioLegend) and human PD-1 (clone PD1.3.1.3, Miltenyi Biotec, Auburn, CA) were added to some wells at 10 μg/ml. Mouse IgG1 (clone P3.6.2.8.1) and antibodies to human CD80 (clone 2D10) were from eBioscience and BioLegend, respectively. Human IFNγ production was measured by ELISA on day 3 as described (16).
CD28-Fc and PD-1-Fc binding
C8161/CD80 cells were incubated in the presence of 10 or 100 μg/ml anti-human CD80 (BioLegend) or mouse IgG1 (eBioscience) mAbs, followed by incubation with a fusion protein consisting of the extracellular region of CD28 fused to the Fc region of human IgG1 (CD28-Fc, R&D Systems), and detected by anti-human IgG-Alexa Fluor 488 (Invitrogen). C8161 and C8161/CD80 cells were incubated in the presence of human PD-1-Fc, followed by anti-human IgG-Alexa Fluor 488 (Invitrogen) as described (11).
Mouse splenocyte activation
Splenocytes from C57BL/6 and CD28−/− mice were depleted of red blood cells and cultured at 37°C, 5% CO2 in 96-well plates at 1×105 cells/well with 20 ng/ml phorbol-12-myristate-13-acetate (PMA) (Sigma-Aldrich, St. Louis, MO) plus 1μg/well ionomycin (EMD Millipore, Billerica, MA) in the presence or absence of mouse TROY- Fc or CD80-Fc (R&D Systems) at 1, 5, and 10 μg/ml. IFNγ production was measured on day 2 by ELISA according to the manufacturer's directions (BioLegend).
Statistical Analysis
Standard deviation, one-way analysis of variance (ANOVA), and Tukey's honestly significant difference (HSD) test were performed using Excel version 2010. Mann-Whitney test was performed using http://faculty.vassar.edu/lowry/VassarStats.html.
Results
CD80-mediated costimulation is not exclusively responsible for maintaining activated T cells in the presence of PD-L1+ human tumor cells
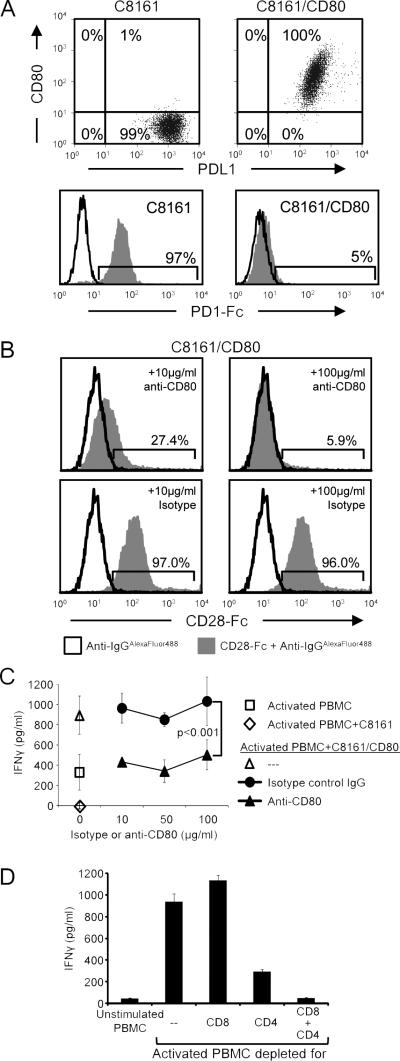
Co-expression of membrane-bound CD80 by PD-L1+ tumor cells prevents the binding of PD-1 to tumor cell-expressed PD-L1 (Fig. 1A), suggesting that CD80 may sustain T-cell activation by preventing PD-1/PD-L1-mediated immune suppression. In addition to binding PDL1, CD80 also functions as a costimulatory molecule by binding to T cell-expressed CD28 (13). We have shown previously that PD-L1+ human and mouse tumor cells modified to express CD80 or treated with a soluble recombinant CD80 (CD80-Fc) facilitate the activation of T cells (11), an effect that could be due to either blocking of PD-1/PD-L1 interactions and/or CD80-CD28 costimulation. To test the latter possibility, PD-L1+ human melanoma C8161 cells transfected with CD80 (C8161/CD80) were incubated with 10 or 100 μg/ml anti-CD80 blocking mAb or IgG1 isotype control followed by human CD28-Fc. Binding of CD28-Fc was detected by staining with fluorescently-labeled goat anti-human IgG and analysis by flow cytometry (Fig. 1B). Pretreatment with 10 or 100 μg/ml anti-CD80 mAb decreased CD28-Fc binding by 73% and 94%, respectively, whereas, pretreatment with 10 or 100 μg/ml of control IgG mAb decreased the binding by no more than 4%. Since treatment with100 μg/ml of anti-CD80 antibodies inhibited the binding of CD28, it is reasonable to expect that CD80 antibody-treated PD-L1+ cells cannot bind to CD28 and costimulate.
Figure 1. CD80-mediated costimulation is not exclusively responsible for maintaining activated T cells in the presence of PD-L1+ human tumor cells.
A, CD80-transfected tumor cells co-express CD80 and PD-L1, and do not bind PD-1. Human cutaneous melanoma C8161 and C8161/CD80 cells were stained for cell surface CD80 and PD-L1 (mAb 5H1) (top panels), or incubated with or without recombinant human PD-1-Fc followed by staining with anti-human IgG-Alexa Fluor 488 (bottom panels) and analyzed by flow cytometry. B, Anti-CD80 mAbs block CD28-Fc binding to CD80+ tumor cells. C8161/CD80 human tumor cells were incubated with 10 or 100 μg/ml of CD80 blocking mAbs or IgG1 control followed by incubation with recombinant human CD28-Fc, staining with anti-human IgG-Alexa Fluor 488, and analysis by flow cytometry. C, CD80 blockade diminishes, but does not eliminate the ability of CD80 to maintain T-cell activation. PHA-activated PBMC from healthy human donors were co-cultured with human C8161 or C8161/CD80 cells. Ten, 50, or 100 μg/ml of CD80 blocking mAb or IgG1 control were included in some wells. IFNγ production was measured by ELISA. Statistical evaluation was performed by Mann-Whitney test. D, Human PBMC from healthy donors were undepleted, CD8-depleted, CD4-depleted, or CD8- and CD4-depleted, and subsequently incubated with PHA for 72hours. IFNγ production was measured by ELISA. Data are from three, two, two, and two independent experiments for A, B, C, and D, respectively. Slightly modified versions of this experiment using 5 different tumor cells and different quantities of CD80-Fc have been done with PBMC from six different healthy donors.
To determine if CD80-CD28 costimulation was exclusively responsible for maintaining T-cell activation in the presence of PD-L1+ tumor cells, PHA-activated human PBMC were co-cultured with PD-L1+ C8161 or with C8161/CD80 cells in the presence or absence of increasing concentrations of either anti-CD80 blocking mAb or IgG1 isotype control antibody (Fig. 1C). T-cell activation was measured by the production of IFNγ. If CD80 only costimulates, then cultures of C8161/CD80 cells with anti-CD80 mAbs will display suppression similar to cultures containing CD80− C8161 cells. C8161 cells suppressed the activation, whereas C8161/CD80 cells increased IFNγ production. Inclusion of anti-CD80 mAb, but not IgG1 isotype control, at 10, 50, or 100 μg/ml, decreased the activation to levels similar to that of activated PBMC alone, but more than that of activated PBMC plus C8161 cells. IFNγ was produced by CD4+ and CD8+ T cells since depletion of these cells eliminated IFNγ production (Fig. 1D). Therefore, anti-CD80 mAbs partially decreased T cell activation by C8161/CD80 cells, suggesting that in the absence of costimulation, CD80 maintains T-cell activation by preventing PD-1/PD-L1-mediated suppression. This latter conclusion is supported by our finding that CD80+ PD-L1+ tumor cells do not bind PD-1 (Fig. 1A).
CD80-Fc overcomes suppression by PD-L1+ PBMC, including PD-L1-expressing activated CD4+ and CD8+ T cells
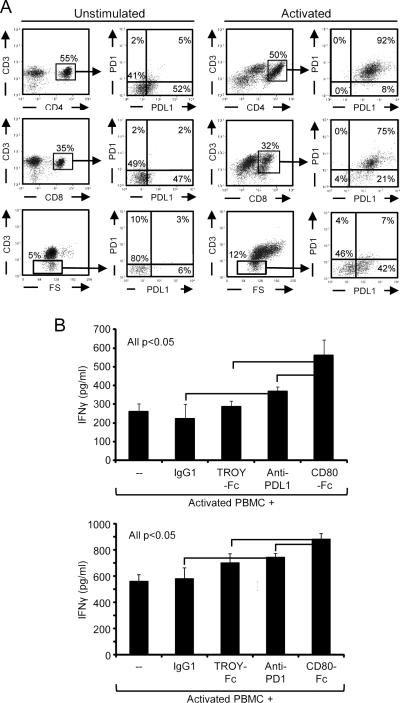
In addition to tumor cells, activated CD4+ and CD8+ T cells as well as non-CD3+ PBMC express PD-1 and PD-L1 (Fig. 2A), and may contribute to suppression. To test this possibility, human PBMC were activated for 72 hrs with PHA in the presence of anti-PD-L1 mAb (clone 29E.2A3), anti-PD-1 mAb (PD1.3.1.3), or an isotype control (Fig. 2B). IFNγ production was measured by ELISA. Antibodies to both PD-L1 and PD-1 increased the activation relative to the untreated or IgG controls, confirming that T cell-expressed PD-L1 suppresses activation. We have shown previously that a soluble form of CD80 (CD80-Fc) is equally effective as membrane-bound CD80 in maintaining the activation of both mouse and human T cells (12), and therefore included CD80-Fc and a negative control recombinant protein (TROY-Fc) in this experiment. CD80-Fc enhanced activation more efficiently than treatment with either antibody (Fig. 2B). Experiments with four other anti-PD-L1 mAbs (MIH1, 5H1, MIH3, and 130021) and four other anti-PD-1 mAbs (EH12.1, EH12.2H7, J116, and MIH4) gave similar results. Therefore, expression of PD-L1 by activated T cells and other PBMC also reduces T-cell activation, and T-cell production of IFNγ is more effectively enhanced by CD80-Fc than by antibodies to PD-L1 or PD-1.
Figure 2. T cell-expressed PD-L1 suppresses T-cell activation and is overcome by CD80-Fc.
A, Activated CD4+ and CD8+ T cells up-regulate PD-1 and PD-L1. Human PBMC from healthy donors were cultured for 72 hours with or without PHA, subsequently stained for CD3, CD4, CD8, and either PD-1 or PD-L1 mAbs, and analyzed by flow cytometry. B, T cell-expressed PD-1 or PD-L1 suppresses T-cell activation. Human PBMC were activated as in A in the presence or absence of 10 μg/ml IgG isotype control, TROY-Fc, PD-L1 blocking mAb, PD-1 blocking mAb, or CD80-Fc. IFNγ production was measured by ELISA. Data are from three and two-three independent experiments for A and B, respectively. Statistical evaluation was performed by one-way ANOVA followed by Tukey's HSD test. Similar results were obtained in additional experiments using 4 other mAbs to PD-L1 (MIH1, 5H1, MIH3, and 130021) and 4 other mAbs to PD-1 (EH12.1, EH12.2H7, J116, and MIH4).
CD80 increases T-cell activation independent of costimulation
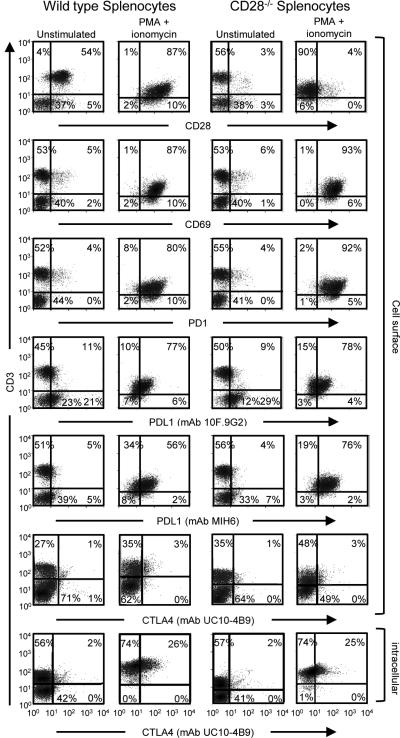
The experiments of Figures 1 and 2 demonstrated that CD80, either as a membrane-bound protein or as a soluble recombinant protein, contributes to maintaining T-cell activation, and that its effect is partially due to the costimulation via CD28. To test the function of CD80 in the absence of costimulation, wild type and CD28−/− C57BL/6 mouse splenocytes were treated with PMA plus ionomycin for 48 hours and the resulting T cells were analyzed by flow cytometry for the activation marker CD69 and for the expression of PD-1 and PD-L1. Activated splenocytes were also tested for CTLA-4 expression because CD80 can cause anergy/apoptosis by binding to T cell-expressed CTLA-4 (17). Since cell surface CTLA-4 has a short half-life, both intracellular and cell surface CTLA-4 were measured (Fig. 3; Supplementary Fig. S1). Both wild-type and CD28−/− T cells displayed increased CD69 and PD-1 expression following treatment with PMA plus ionomycin, demonstrating that both cell populations were activated. Mouse T cells also up-regulated PD-L1 after activation, indicating that activated mouse T cells, like activated human T cells, may also contribute to PD-L1-mediated immune suppression. CTLA-4 was also expressed by activated mouse T cells.
Figure 3. Murine T cells activated with PMA plus ionomycin express CD69, PD-1, PD-L1, and CTLA-4.
Splenocytes from wild-type and CD28−/− C57BL/6 mice were activated for 48-72 hours with PMA plus ionomycin, subsequently surface-stained with CD3 and either CD28, CD69, PD-1, PD-L1, or CTLA-4 mAbs, and analyzed by flow cytometry. Splenocytes were also fixed and permeabilized and stained for intracellular CTLA-4. Data are from one of three (CD69, PD-1, PD-L1) or two (CTLA-4) independent experiments.
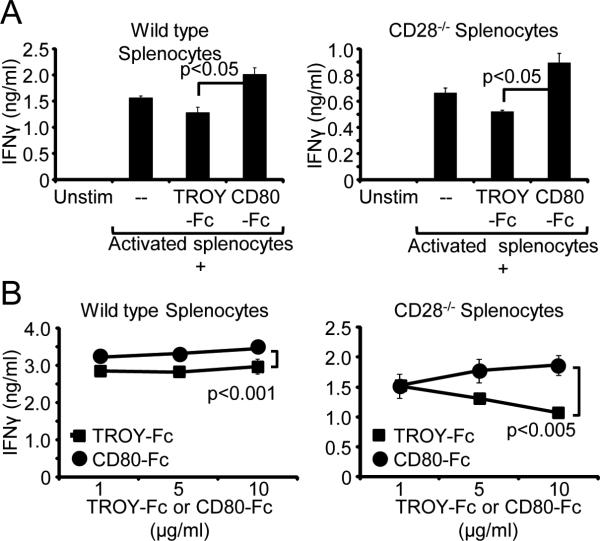
To test if murine T cell-expressed PD-L1 decreased T-cell activation, splenocytes from C57BL/6 wild-type and CD28−/− mice were activated with PMA plus ionomycin and cultured for 48 hours with either CD80-Fc or control TROY-Fc. T-cell activation was assessed by measuring IFNγ production (Fig. 4A). If CD80-Fc prevents suppression by blocking PD-1/PD-L1 interactions, then CD80-Fc will increase IFNγ production by CD28−/− T cells. However, if CD80-Fc only increases T-cell activation by costimulation, then there will be no effect when CD80-Fc is added to the CD28−/− T cells. Inclusion of CD80-Fc, but not TROY-Fc, increased IFNγ production by both wild-type and CD28−/− T cells, and the dose-dependent increase was most dramatic for the CD28−/− T cells (Fig. 4A, 4B). IFNγ production was increased by CD80-Fc despite the expression of CTLA-4 on activated T cells. This finding suggests that CD80-Fc is not inducing T cell anergy/apoptosis via CTLA-4 or that such interactions are minimal. Collectively, these results demonstrate that similar to the effects of CD80 on human T cells, CD80 also sustains the activation of murine T cells through a costimulation-independent mechanism.
Figure 4. CD80-Fc increases T-cell activation independent of costimulation.
A, PMA plus ionomycin-activated splenocytes from wild-type and CD28−/− C57BL/6 mice were cultured for 48 hours in the presence or absence of 10 μg/ml of mouse CD80-Fc or TROY-Fc. IFNγ production was measured by ELISA. Statistical evaluation was performed by one-way ANOVA followed by Tukey's HSD test. B, Murine wild-type and CD28−/− splenocytes were activated as in panel A in the presence or absence of increasing quantities of CD80-Fc or TROY-Fc. IFNγ production was measured by ELISA. Data are from one of two independent experiments. Statistical evaluation was performed by Mann-Whitney test.
Discussion
Previously we have demonstrated that treatment of PD-L1+ tumor cells with a soluble form of the extracellular domains of CD80 or co-expression of the membrane-bound CD80 maintains the activation of both human and mouse T cells (11, 12). In these studies it was unclear if and how much of the effect of CD80 was due to its ability to costimulate through CD28 vs. its ability to prevent PD-1/PD-L1-mediated immune evasion. The present report demonstrates that both mechanisms contribute to the ability of CD80-Fc to maintain IFNγ production by mouse and human PD-1+ T cells in the presence of PD-L1+ cells. As recent clinical trials have demonstrated, PD-L1-mediated suppression is a potent mechanism employed by many tumor cells to evade antitumor immunity, and antibody blocking of PD-L1 or PD-1 significantly increases patient responses and survival times (18, 19). In the present studies, CD80-Fc was more effective than antibodies to PD-L1 or PD-1 at maintaining T-cell production of IFNγ. This enhanced activity may be because of the dual action of CD80-Fc in simultaneously blocking PD-1/PD-L1 suppression and providing costimulation, and therefore, CD80-Fc may be a more effective therapeutic than mAbs to PD-L1 and/or PD-1 for restoring and maintaining antitumor immunity in cancer patients.
Supplementary Material
Acknowledgments
Financial support: NIH RO1 CA84232 and CA115880 (SOR)
LH was partially supported by GAANN P200A12-17 and STH by NIH R25GM55036
Footnotes
The authors have no conflicts of interest to declare.
References
- 1.Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med. 2002;8:793–800. doi: 10.1038/nm730. [DOI] [PubMed] [Google Scholar]
- 2.Lee SJ, Jang BC, Lee SW, Yang YI, Suh SI, Park YM, et al. Interferon regulatory factor-1 is prerequisite to the constitutive expression and IFN-gamma-induced upregulation of B7-H1 (CD274). FEBS Lett. 2006;580:755–62. doi: 10.1016/j.febslet.2005.12.093. [DOI] [PubMed] [Google Scholar]
- 3.Azuma T, Yao S, Zhu G, Flies AS, Flies SJ, Chen L. B7-H1 is a ubiquitous antiapoptotic receptor on cancer cells. Blood. 2008;111:3635–43. doi: 10.1182/blood-2007-11-123141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Latchman YE, Liang SC, Wu Y, Chernova T, Sobel RA, Klemm M, et al. PD-L1-deficient mice show that PD-L1 on T cells, antigen-presenting cells, and host tissues negatively regulates T cells. Proc Natl Acad Sci USA. 2004;101:10691–6. doi: 10.1073/pnas.0307252101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ. Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T cell responses. Immunity. 2007;27:111–22. doi: 10.1016/j.immuni.2007.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Park JJ, Omiya R, Matsumura Y, Sakoda Y, Kuramasu A, Augustine MM, et al. B7-H1/CD80 interaction is required for the induction and maintenance of peripheral T-cell tolerance. Blood. 2010;116:1291–8. doi: 10.1182/blood-2010-01-265975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Francisco LM, Salinas VH, Brown KE, Vanguri VK, Freeman GJ, Kuchroo VK, et al. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. The J Exp Med. 2009;206:3015–29. doi: 10.1084/jem.20090847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yamazaki T, Akiba H, Iwai H, Matsuda H, Aoki M, Tanno Y, et al. Expression of programmed death 1 ligands by murine T cells and APC. J Immunol. 2002;169:5538–45. doi: 10.4049/jimmunol.169.10.5538. [DOI] [PubMed] [Google Scholar]
- 9.Liu Y, Zeng B, Zhang Z, Zhang Y, Yang R. B7-H1 on myeloid-derived suppressor cells in immune suppression by a mouse model of ovarian cancer. Clin Immunol. 2008;129:471–81. doi: 10.1016/j.clim.2008.07.030. [DOI] [PubMed] [Google Scholar]
- 10.Lin PY, Sun L, Thibodeaux SR, Ludwig SM, Vadlamudi RK, Hurez VJ, et al. B7-H1-dependent sex-related differences in tumor immunity and immunotherapy responses. J Immunol. 2010;185:2747–53. doi: 10.4049/jimmunol.1000496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Haile ST, Bosch JJ, Agu NI, Zeender AM, Somasundaram P, Srivastava MK, et al. Tumor cell programmed death ligand 1-mediated T cell suppression is overcome by coexpression of CD80. J Immunol. 2011;186:6822–9. doi: 10.4049/jimmunol.1003682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Haile ST, Dalal SP, Clements V, Tamada K, Ostrand-Rosenberg S. Soluble CD80 Restores T Cell Activation and Overcomes Tumor Cell Programmed Death Ligand 1-Mediated Immune Suppression. J Immunol. 2013;191:2829–36. doi: 10.4049/jimmunol.1202777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jenkins M, Taylor P, Norton S, Urdahl K. CD28 delivers a costimulatory signal involved in antigen-specific IL-2 production by human T cells. J Immunol. 1991;147:2461–7. [PubMed] [Google Scholar]
- 14.Dissanayake SK, Thompson JA, Bosch JJ, Clements VK, Chen PW, Ksander BR, et al. Activation of tumor-specific CD4(+) T lymphocytes by major histocompatibility complex class II tumor cell vaccines: a novel cell-based immunotherapy. Cancer Res. 2004;64:1867–74. doi: 10.1158/0008-5472.can-03-2634. [DOI] [PubMed] [Google Scholar]
- 15.Thompson JA, Srivastava MK, Bosch JJ, Clements VK, Ksander BR, Ostrand-Rosenberg S. The absence of invariant chain in MHC II cancer vaccines enhances the activation of tumor-reactive type 1 CD4+ T lymphocytes. Cancer Immunol Immunother. 2008;57:389–98. doi: 10.1007/s00262-007-0381-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bosch JJ, Thompson JA, Srivastava MK, Iheagwara UK, Murray TG, Lotem M, et al. MHC class II-transduced tumor cells originating in the immune-privileged eye prime and boost CD4(+) T lymphocytes that cross-react with primary and metastatic uveal melanoma cells. Cancer Res. 2007;67:4499–506. doi: 10.1158/0008-5472.CAN-06-3770. [DOI] [PubMed] [Google Scholar]
- 17.Walunas TL, Lenschow DJ, Bakker CY, Linsley PS, Freeman GJ, Green JM, et al. CTLA-4 can function as a negative regulator of T cell activation. Immunity. 1994;1:405–13. doi: 10.1016/1074-7613(94)90071-x. [DOI] [PubMed] [Google Scholar]
- 18.Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ, Topalian SL, Hwu P, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366:2455–65. doi: 10.1056/NEJMoa1200694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443–54. doi: 10.1056/NEJMoa1200690. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.