Abstract
Objectives
To determine whether hearing impairment, defined using objective audiometry, is associated with multiple categories of self-reported physical functioning in a cross-sectional, nationally representative sample of older adults.
Design
Multivariate secondary analysis of cross-sectional data.
Setting
The 2005–06 and 2009–10 cycles of the National Health and Nutrition Examination Survey.
Participants
Adults aged 70 and older who completed audiometric testing (N=1,669).
Measurements
Hearing was measured using pure-tone audiometry. Physical functioning was assessed using a structured interview.
Results
In a model adjusted for age and demographic and cardiovascular risk factors, greater hearing impairment (per 25 dB hearing level (HL)) was associated with greater odds of physical disability in activities of daily living (odds ratio (OR)=1.4, 95% confidence interval (CI)=1.1–1.9), instrumental activities of daily living (OR=1.6, 95% CI=1.2–2.2), leisure and social activities (OR=1.5, 95% CI=1.1–2.0), lower extremity mobility (OR=1.4, 95% CI=1.1–1.7), general physical activities (OR=1.3, 95% CI=1.1–1.6), work limitation (OR=1.4, 95% CI=1.0–1.9), walking limitation (OR=1.6, 95% CI=1.3–2.0), and limitation due to memory or confusion (OR=1.4, 95% CI=1.1–1.8). Hearing impairment was not associated with limitations in amount or type of work done (OR=1.2, 95% CI=1.0–1.6).
Conclusion
Hearing impairment in older adults is independently associated with greater disability and limitations in multiple self-reported categories of physical functioning.
Keywords: hearing impairment, physical functioning, older adults
Hearing impairment affects approximately 70% of older adults in the United States, and its prevalence nearly doubles with every decade of age.1 Recent epidemiological studies have demonstrated that hearing impairment is independently associated with accelerated cognitive decline2 and the risk of incident dementia in older adults.3 Effects of hearing impairment on cognitive load4,5 and social isolation6 may mediate these associations.
Similar mediating pathways may also underlie contributions of hearing impairment to poorer physical functioning in older adults. Social isolation7 and lack of attentional resources from cognitive load8 have been associated with poorer functioning in elderly adults, although prior epidemiological studies of the association of hearing impairment with physical functioning have demonstrated inconsistent results, with some studies demonstrating a positive association9–18 and others demonstrating no significant association.19–21 It is likely that differences between studies in how hearing and physical functioning were measured explain this heterogeneity in study results. (Hearing has been measured using subjective self-report10,12,19,21 and objective clinical audiometry9,13–18,20, whereas physical functioning has been measured using activities of daily living (ADLs),11,12,14,17,19,21 instrumental activities of daily living (IADLs),10–12,19 the Sickness Impact Profile,9 Rosow-Breslau indices,18 and the Medical Outcomes Study 36-item Short-Form Survey.13) It is likely that biased or imprecise assessments of hearing thresholds would decrease the sensitivity to detect associations due to greater variance.
In the present study, cross-sectional data from a nationally representative sample of older adults in whom hearing was objectively measured using audiometry were used to perform exploratory analyses of whether hearing impairment is associated with a broad range of self-reported metrics of physical functioning and activities. These comprehensive self-reported measures of physical functioning included ADLs, IADL, and other indicators of physical functioning such as leisure and social activities, lower extremity mobility, and general physical activities.
Methods
Study Participants
The National Health and Nutrition Examination Survey (NHANES) is an ongoing series of studies that the National Center for Health Statistics conducts to assess the health, functional, and nutritional status of the civilian noninstitutionalized U.S. population. Each 2-year cross-sectional study cycle uses a complex multistage probability sampling design to survey a representative sample of the population from multiple counties across the United States, with selective oversampling of low-income individuals, racial minorities, and older adults. The sample does not include persons residing in nursing homes, members of the armed forces, institutionalized persons, or U.S. nationals living abroad. Sampling weights allow for analyses that account for the complex survey design and yield results that are generalizable to the U.S. population. NHANES data from 2005–06 and 2009–10 were combined because these were the only 2-year cycles when hearing was measured in adults aged 70 and older. One thousand eight hundred eighty-five individuals aged 70 and older participated in the examination component of NHANES in these cycles, 1,669 (89%) of whom completed audiometric testing.
Audiometric Assessment
A trained examiner performed audiometry according to established NHANES protocols.22 Briefly, air conduction hearing thresholds were obtained from both ears in a sound-isolating room in the mobile examination center. Testing was conducted according to a modified Hughson-Westlake procedure using the automated testing mode of the audiometer (Interacoustics Model AD226, Interacoustics, Eden Prairie, MN) or manually, according to the testing protocol. Quality assurance and control were established through daily calibration of equipment and monitoring of ambient noise levels using a sound-level meter. As an additional quality measure, thresholds were measured twice at 1 kHz in both ears, and audiometry was repeated if there was greater than 10 dB discrepancy between the threshold measurements. Hearing ability was defined according to a speech-frequency pure tone average of thresholds at 0.5, 1, 2, and 4 kHz in the better-hearing ear, according to the definition of hearing impairment that the World Health Organization uses.23 Thresholds of hearing impairment severity were based on American Speech-Language Hearing Association guidelines24 (normal hearing <25.0 dB, mild impairment 25.0–39.9 dB, moderate impairment 40.0–69.9 dB, severe to profound impairment ≥70.0 dB).
Physical Functioning Assessment
Study personnel interviewed participants regarding their physical functioning using the Computer-Assisted Personal Interviewing system. Participants were asked a series of questions regarding any difficulties they had performing physical tasks. These physical tasks were categorized into five domains (ADLs, IADLs, leisure and social activities, lower-extremity mobility, general physical activities), as previously described.25 Difficulty (yes/no) in a physical domain was defined as a response of some or more difficulty in at least one of the physical task questions within a domain.25 Participants were also asked about general functional limitations in ability to work, amount and type of work performed, walking, and limitations due to confusion and memory. Limitations in these categories were defined as a response of “yes” on the survey. Survey items pertaining to these physical domains and functional limitations are summarized in Table 1.
Table 1. National Health and Nutrition Examination Survey Questionnaire Items Comprising Physical Domains and Functional Limitations As Described Previously25.
| Measure of Disability | Survey Items | Possible Responses |
|---|---|---|
| Physical domain | ||
| Activities of daily living | By yourself and without using any special equipment, how much difficulty do you have… Getting in and out of bed Eating Dressing yourself |
No difficulty, some difficulty, much difficulty, unable to do, do not do this activity |
| Instrumental activities of daily living | By yourself and without using any special equipment, how much difficulty do you have… Managing money Performing house chores Preparing meals |
No difficulty, some difficulty, much difficulty, unable to do, do not do this activity |
| Leisure and social activities | By yourself and without using any special equipment, how much difficulty do you have… Going to the movies Attending social events Performing leisure activity at home |
No difficulty, some difficulty, much difficulty, unable to do, do not do this activity |
| Lower extremity mobility | By yourself and without using any special equipment, how much difficulty do you have… Walking one-quarter of a mile Walking up 10 steps Stopping, kneeling, crouching Walking between rooms on the same floor Standing up from an armless chair |
No difficulty, some difficulty, much difficulty, unable to do, do not do this activity |
| General physical activities | By yourself and without using any special equipment, how much difficulty do you have… Lifting or carrying heavy objects Reaching up overhead Grasping/holding small objects Standing for long periods Sitting for long periods |
No difficulty, some difficulty, much difficulty, unable to do, do not do this activity |
| Functional limitations | ||
| Ability to work | Does a physical, mental, or emotional problem now keep you from working at a job or business? | Yes, no |
| Amount and type of work | Are you limited in the kind or amount of work you can do because of a physical, mental, or emotional problem? | Yes, no |
| Walking | Because of a health problem, do you have difficulty walking without using any special equipment? | Yes, no |
| Memory and confusion | Are you limited in any way because of difficulty remembering or because you experience periods of confusion? | Yes, no |
Difficulty (yes/no) in a physical domain was defined as a response of some difficulty or more in at least one of the physical task questions within a domain.
Other Study Variables
Other covariates that could potentially confound the association between hearing impairment and physical functioning were included in this analysis. Data on demographic variables and medical history were obtained from interviews. For confidentiality purposes, the NHANES database truncates the age variable at 80. Therefore, age was treated as a categorical rather than a continuous variable in the primary analysis, with the categories 70 to 74, 75 to 79, and 80 and older. A sensitivity analysis was also conducted limiting the analytical cohort to individuals aged 70 to 79 to allow for adjustment of age as a continuous variable. Self-reported race and ethnicity was grouped as Mexican American or other Hispanic (Hispanic), non-Hispanic white (white), non-Hispanic black (black), or other race. Education was collapsed into a three-level variable (<12th grade, high school graduate, some college or more), and household income was collapsed into a five-level variable (<$20,000, $20,000–44,000, $45,000–74,000, ≥$75,000, refused/do not know). Variables related to medical history included hypertension, cardiovascular disease, diabetes mellitus, stroke, and smoking status (current, former, never). Hypertension was defined as having been diagnosed by a physician on at least two visits with high blood pressure or taking antihypertensive medication. Cardiovascular disease was defined as any self-reported history of congestive heart failure, coronary artery disease, angina pectoris, or myocardial infarction. Hearing aid use was defined as whether an individual reported having worn a hearing aid at least 5 hours per week in the 12 months before the interview.
Statistical Methodology
The complex sampling design was accounted for in all analyses using sample weights according to National Center for Health Statistics guidelines,26 except for Table 2. The purpose of Table 2 was to provide descriptive statistics for the characteristics of the study cohort rather than nationally generalizable estimates, so weights were not used. Baseline characteristics of the study participants were compared using the Pearson chi-square test. Multivariate logistic regression was used to examine the relationship between hearing impairment as a continuous variable and physical functioning while adjusting for age and other covariates. Sensitivity analyses included restricting the analytical cohort to individuals younger than 80 (to allow for adjustment of age as a continuous variable) and excluding participants with severe hearing impairment (better-ear pure-tone average (BPTA) ≥70dB HL). To assess effect modification according to sex and age, analyses were also conducted using a hearing impairment–sex interaction term and a hearing impairment–age interaction term, with age coded as a binary variable (<80 or ≥80). In all models, individuals with missing data accounted for less than 10% of the study population, with the exception of analyses involving IADLs, in which IADL data were missing in 267 individuals (16.0%). This high proportion of missing IADL data was due to a number of participants responding, “Do not do this activity” to questions comprising the IADL domain. All significant tests were conducted using two-sided tests with a type I error of 0.05. All analyses were conducted using Stata 11.1 (StataCorp, College Station, TX).
Table 2. Demographic Characteristics of Participants Aged 70 and Older with Audiometric Testing, National Health and Nutrition Examination Survey, 2005–06 and 2009–10.
| Characteristic | Normal Hearing, n=529 (32%) | Hearing Impairment, n=1,140 (68%) | P-Value |
|---|---|---|---|
|
|
|||
| n (%) | |||
| Age | <.001 | ||
| 70–74 | 289 (55) | 319 (30) | |
| 75–79 | 138 (26) | 283 (25) | |
| ≥80 | 102 (19) | 538 (47) | |
| Male | 217 (41) | 631 (55) | <.001 |
| Race | <.001 | ||
| White | 327 (62) | 859 (75) | |
| Black | 117 (22) | 117 (10) | |
| Hispanic | 68 (13) | 131 (12) | |
| Other | 17 (3) | 33 (3) | |
| Education | .001 | ||
| <12th grade | 161 (30) | 428 (38) | |
| High school graduate | 128 (24) | 300 (26) | |
| Some college or more | 239 (45) | 410 (36) | |
| Income, $ | .004 | ||
| <20,000 | 129 (24) | 353 (31) | |
| 20,000–44,999 | 176 (33) | 406 (36) | |
| 45,000–74,999 | 114 (22) | 182 (16) | |
| ≥75,000 | 71 (13) | 119 (10) | |
| Refused or do not know | 39 (7) | 75 (7) | |
| Hypertension | 353 (67) | 703 (62) | .050 |
| Cardiovascular diseasea | 103 (17) | 307 (27) | .001 |
| Stroke | 38 (7) | 130 (11) | .008 |
| Diabetes mellitus | 113 (21) | 247 (22) | .890 |
| Smoking | .240 | ||
| Never | 258 (49) | 534 (47) | |
| Former | 226 (43) | 529 (46) | |
| Current | 45 (9) | 77 (7) | |
| Degree of hearing impairment | |||
| Mild | - | 590 (52) | |
| Moderate | - | 518 (45) | |
| Severe to profound | - | 32 (3) | |
| Hearing aid use | 1 (0) | 198 (17) | <.001 |
| Physical function difficultyb | |||
| Activities of daily living | 93 (18) | 272 (24) | .004 |
| Instrumental activities of daily living | 103 (19) | 261 (23) | .020 |
| Leisure and social activities | 95 (18) | 249 (22) | .030 |
| Lower extremity mobility | 261 (49) | 718 (63) | <.001 |
| General physical activities | 260 (49) | 630 (55) | .004 |
| Work limitation | 82 (15.5) | 229 (20.1) | .020 |
| Limitation in amount or type of work | 172 (32.5) | 451 (39.6) | .006 |
| Walking limitation | 85 (16.1) | 285 (25.0) | <.001 |
| Memory and confusion limitation | 60 (11.3) | 178 (15.6) | .020 |
Hearing impairment is defined as a speech-frequency pure-tone average of hearing thresholds at 0.5, 1, 2, and 4 kilohertz in the better hearing ear of 25 dB of hearing loss or greater.
Self-reported history of congestive heart failure, coronary artery disease, angina pectoris, or myocardial infarction
Some difficulty or more in at least one of the physical task questions within a domain. Presence of a functional limitation (work, amount or type of work, walking, memory, and confusion) was defined as a response of “yes” in a category.
Results
One thousand six hundred sixty-nine participants aged 70 and older were included in this study, 529 (31.7%) of whom had normal hearing (Table 2). Of those with hearing impairment (n=1,140), 590 (51.8%) had mild hearing impairment, 518 (45.4%) had moderate hearing impairment, and 32 (2.8%) had severe to profound hearing impairment. Hearing impairment was associated with age, sex, race, education, income, hypertension, cardiovascular disease, and stroke (Table 2).
In a model adjusted for age and demographic and cardiovascular risk factors, a 25-dB difference in hearing threshold was associated with greater odds of poorer ADL functioning (OR=1.4, 95% CI=1.1–1.9; p=.01), IADL functioning (OR=1.6, 95% CI=1.2–2.2; p=.003, leisure and social activities (OR=1.5, 95% CI=1.1–2.0; p=.01), lower extremity mobility (OR=1.4, 95% CI=1.1–1.7; p=.005, and general physical activities (OR=1.3, 95% CI=1.1–1.6; p=.02). For functional limitations, a 25-dB difference in hearing threshold was associated with greater odds of work limitation (OR=1.4, 95% CI=1.0–1.9; p=.03), walking limitation (OR=1.6, 95% CI=1.3–2.0; p<.001), and limitation due to memory or confusion (OR=1.4, 95% CI=1.1–1.8; p=.01) (Figure 1). Hearing impairment was not associated with limitations in amount or type of work done (OR=1.2, 95% CI=1.0–1.6; p=.11).
Figure 1.
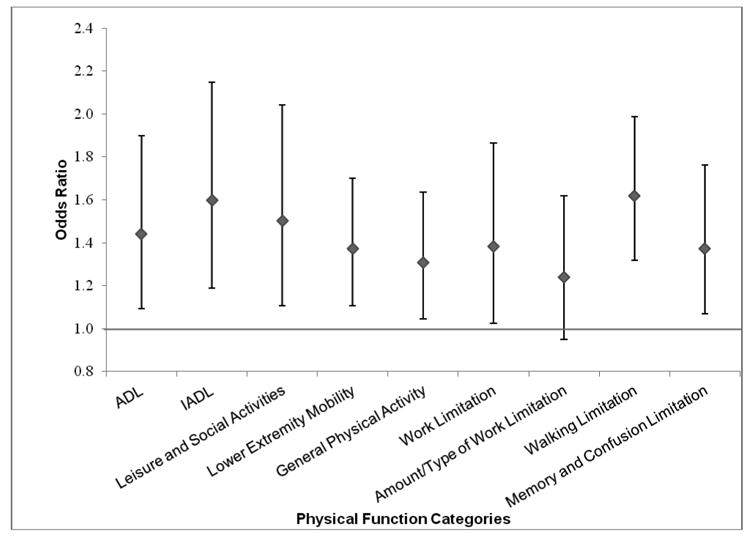
Adjusted odds ratios (adjusted for age, sex, race, education, income, hypertension, cardiovascular disease, stroke, diabetes, and smoking status) of disabilities and functional limitations per 25-dB difference in hearing level (hearing defined according to the speech-frequency pure tone average of hearing thresholds at 0.5, 1, 2, and 4 kHz in the better ear). Error bars indicate 95% confidence intervals.
A sensitivity analysis restricting the analytical cohort to individuals younger than 80 (to allow for adjustment of age as a continuous covariate) did not substantially change the results (data not shown) from those shown in Figure 1, with the exception of wider CIs being observed for work limitations (OR=1.4, 95% CI=0.9–2.1; p=.17) and memory and confusion limitations (OR=1.2, 95% CI=0.9–1.7; p=.20). An additional sensitivity analysis excluding participants with severe hearing impairment (BPTA ≥70dB HL) also yielded results similar to those shown in Figure 1 (data not shown), with the exception of wider CIs being observed for general physical activities (OR=1.3, 95% CI=1.0–1.6; p=.10) and work limitations (OR=1.3, 95% CI=0.9–1.8; p=.17).
Effect modification was also investigated according to sex and age. Sex did not significantly moderate the relationship between hearing and any of the outcome variables except amount or type of work done and limitations due to confusion and memory. Men had greater odds of limitation in the former category (p=.02), and women had greater limitation in the latter category (p=.04). Age did not significantly moderate the association between hearing impairment and physical functioning in any of the categories. Finally, the association between hearing aid use and physical functioning was also investigated. In individuals with hearing impairment (BPTA ≥25dB HL), hearing aid use was not significantly associated with any of the domains of physical functioning (data not shown) except for greater odds of having ADL impairments (OR=2.2, 95% CI 1.2–3.9).
Discussion
In this nationally representative study of adults aged 70 and older, greater hearing impairment was independently associated with poorer functioning in multiple self-reported measures including ADLs, IADLs, leisure and social activities, lower extremity mobility, general physical activities, work limitation, walking limitation, and limitations due to confusion and memory. These results were robust to adjustment for multiple confounders and in sensitivity analyses excluding individuals with severe hearing impairment or aged 80 and older. A 25 dB difference in hearing thresholds (equivalent to the magnitude of difference between normal hearing and mild hearing impairment) was associated with 60% greater odds of having self-reported IADL impairments, 50% greater odds of impairments in leisure and social activities, and 62% greater odds of walking limitations.
These findings are consistent with prior research demonstrating significant associations between hearing impairment and ADL, IADL10–12,17 and other measures of physical functioning.9,12,13,15,18 In contrast, several studies have found no association between hearing impairment and functioning or functional decline.19–21 One study that used longitudinal data from NHANES I and the NHANES Epidemiologic Follow-up Study found that hearing impairment at baseline was predictive of poorer Rosow-Breslau functional indices but not ADLs or IADLs after 10 years, although the cohort in this study was younger, and hearing impairment was defined using a cutoff of 40 rather than 25 dB. The measurement and definition of hearing impairment varied considerably in prior studies. Several studies relied on self-report or other subjective tests to measure hearing,10–12,19,21 whereas those that used pure-tone audiometry used varying audiometric definitions of hearing impairment and testing environments.9,13–18,20 With the exception of two studies,18,21 prior reports were conducted in study populations in which results may not be generalizable. The strengths of the current study are that the results are based on a nationally representative sample, and a standardized audiometric testing protocol using a definition of hearing impairment that the World Health Organization23 provided was applied to all individuals. Physical functioning was also assessed using a broad range of functional metrics not limited to ADLs and IADLs. Several explanations could account for the observed association between hearing impairment and physical functioning. First, poor verbal reception resulting from hearing impairment may have interfered with participants' understanding of the questionnaire items, but it is likely that this would lead to a nondifferential bias rather than a specific bias toward a positive association. The results were also not sensitive to exclusion of individuals with severe hearing impairment from the analytical cohort. Second, a shared impairment of the cochlear and vestibular sense organs, given their common location in the inner ear,27 could contribute to impaired balance and poorer physical functioning, although a previous analysis demonstrated associations between hearing impairment and falls that were robust to adjustment for vestibular function.28 Third, pure-tone audiometry has been found to be weakly associated with cardiovascular risk factors,29,30 and underlying microvascular disease or another common pathologic process may contribute to hearing impairment and poorer physical functioning. Nevertheless, the current results demonstrate an association between hearing impairment and physical functioning even after adjustment for multiple cardiovascular risk factors.
Hearing impairment could also be associated with physical functioning through mechanistic pathways involving cognitive load and social isolation. Age-related hearing impairment leads to impaired neural encoding of sounds by the cochlea, and the cognitive resources required for processing of degraded sound signals may place a load on the listener's cognitive and attentional resources,4,5 both of which are important determinants of physical mobility and functioning.32,33 Consistent with this hypothesis, hearing impairment has been associated with poorer performance on tests of executive function requiring attentional resources,31 which are necessary for maintaining postural control and balance32 and predict gait speed declines in older adults.33 Communication impairments due to hearing impairment may also contribute to social isolation and loneliness in older adults,12,34 which in turn could mediate the association with poorer physical functioning.35,36
It was found that hearing aid use was not significantly associated with any domains of physical functioning except for greater odds of ADL impairments. The basis of this association is unclear but may be due to a greater likelihood of Medicaid eligibility in individuals with ADL impairments. Many state Medicaid programs provide benefits for hearing aids, in contrast to the federal Medicare program.37 Because the data are cross-sectional, the direction of this observed association cannot be established (whether hearing aid use is a marker of incipient ADL difficulty or vice versa). Ultimately, investigating whether rehabilitative hearing treatments could affect physical functioning will require a randomized clinical trial.
This study has several limitations. First, the pathways underlying the observed associations between hearing impairment and physical functioning cannot be determined. Although there is unlikely to be reverse causation (poor physical functioning leading to hearing impairment), it is likely that there is some degree of residual confounding by other medical or environmental factors (e.g., inflammation, mitochondrial aging). Established confounders for hearing impairment and physical functioning were adjusted for in the models, and multiple pathways (e.g., shared pathology, cognitive load, social isolation) could contribute synergistically to poorer physical functioning. Second, although data from NHANES is understood to be representative of the U.S. population, individuals who are institutionalized (e.g., older adults in nursing homes) are specifically excluded from the NHANES cohort. The association between hearing impairment and physical functioning could be expected to be even stronger in these institutionalized individuals who are excluded. Third, results from the two functional limitation categories regarding ability to work and amount or type of work done may not be relevant to some individuals in the study population who may no longer be working. Fourth, the analyses are also based on cross-sectional data and may thus be subject to bias according to cohort effects or interindividual heterogeneity in participant characteristics. Further prospective research will be needed to determine whether hearing impairment is associated with accelerated declines in physical functioning over. Specifically, longitudinal studies using objective physical performance measurements may help clarify the temporal relationship between hearing impairment and functional parameters such as gait speed and balance.
Physical functioning is a critical determinant of overall health and vitality in older adults. Given the high prevalence of hearing impairment in elderly adults and the increasing proportion of older adults in the general population, clarifying the role of hearing impairment as a potentially modifiable risk factor for poorer physical functioning is a public health priority. Further research is needed to investigate the basis of the observed association between hearing impairment and physical disability, as well as to determine whether hearing rehabilitative therapies could help mitigate declines in physical functioning.
Acknowledgments
This manuscript was supported in part by National Institutes of Health Grant K23DC011279, the Eleanor Schwartz Charitable Foundation, and a Triological Society/American College of Surgeons Clinician Scientist Award.
The National Institute on Deafness and Other Communication Disorders (including an interagency agreement with the National Institute for Occupational Safety and Health for training health technicians to perform audiometry), National Institute on Aging, and National Center for Health Statistics generously provided funding for the NHANES hearing and disability data.
Footnotes
Conflict of Interest: Dr. Lin is a consultant to Cochlear, Autifony, and Pfizer.
References
- 1.Lin FR, Niparko JK, Ferrucci L. Hearing impairment prevalence in the United States. Arch Intern Med. 2011;171:1851–1852. doi: 10.1001/archinternmed.2011.506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lin FR, Yaffe K, Xia J, et al. Hearing impairment and cognitive decline among older adults. JAMA Intern Med. 2013;73:299. doi: 10.1001/jamainternmed.2013.1868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lin FR, Metter EJ, O'Brien RJ, et al. Hearing impairment and incident dementia. Arch Neurol. 2011;68:214–220. doi: 10.1001/archneurol.2010.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tun PA, McCoy S, Wingfield A. Aging, hearing acuity, and the attentional costs of effortful listening. Psychol Aging. 2009;24:761–766. doi: 10.1037/a0014802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wingfield A, Tun PA, McCoy SL. Hearing impairment in older adulthood—what it is and how it interacts with cognitive performance. Curr Dir Psychol Sci. 2005;14:144–148. [Google Scholar]
- 6.Barnes LL, Mendes de Leon CF, Wilson RS, et al. Social resources and cognitive decline in a population of older African Americans and whites. Neurology. 2004;63:2322–2326. doi: 10.1212/01.wnl.0000147473.04043.b3. [DOI] [PubMed] [Google Scholar]
- 7.Perissinotto CM, Stijacic Cenzer I, Covinsky KE. Loneliness in older persons: A predictor of functional decline and death. Arch Intern Med. 2012;172:1078–1083. doi: 10.1001/archinternmed.2012.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Coppin AK, Shumway-Cook A, Saczynski JS, et al. Association of executive function and performance of dual-task physical tests among older adults: Analyses from the InChianti study. Age Ageing. 2006;35:619–624. doi: 10.1093/ageing/afl107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bess FH, Lichtenstein MJ, Logan SA, et al. Hearing impairment as a determinant of function in the elderly. J Am Geriatr Soc. 1989;37:123–128. doi: 10.1111/j.1532-5415.1989.tb05870.x. [DOI] [PubMed] [Google Scholar]
- 10.Dargent-Molina P, Hays M, Breart G. Sensory impairments and physical disability in aged women living at home. Int J Epidemiol. 1996;25:621–629. doi: 10.1093/ije/25.3.621. [DOI] [PubMed] [Google Scholar]
- 11.Keller BK, Morton JL, Thomas VS, et al. The effect of visual and hearing impairments on functional status. J Am Geriatr Soc. 1999;47:1319–1325. doi: 10.1111/j.1532-5415.1999.tb07432.x. [DOI] [PubMed] [Google Scholar]
- 12.Strawbridge WJ, Wallhagen MI, Shema SJ, et al. Negative consequences of hearing impairment in old age: A longitudinal analysis. Gerontologist. 2000;40:320–326. doi: 10.1093/geront/40.3.320. [DOI] [PubMed] [Google Scholar]
- 13.Chia EM, Wang JJ, Rochtchina E, et al. Hearing impairment and health-related quality of life: The Blue Mountains Hearing Study. Ear Hear. 2007;28:187–195. doi: 10.1097/AUD.0b013e31803126b6. [DOI] [PubMed] [Google Scholar]
- 14.Lopez-Torres Hidalgo J, Boix Gras C, Tellez Lapeira J, et al. Functional status of elderly people with hearing impairment. Arch Gerontol Geriatr. 2009;49:88–92. doi: 10.1016/j.archger.2008.05.006. [DOI] [PubMed] [Google Scholar]
- 15.Viljanen A, Kaprio J, Pyykko I, et al. Hearing acuity as a predictor of walking difficulties in older women. J Am Geriatr Soc. 2009;57:2282–2286. doi: 10.1111/j.1532-5415.2009.02553.x. [DOI] [PubMed] [Google Scholar]
- 16.Viljanen A, Kaprio J, Pyykko I, et al. Hearing as a predictor of falls and postural balance in older female twins. J Gerontol A Biol Sci Med Sci. 2009;64A:312–317. doi: 10.1093/gerona/gln015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gopinath B, Schneider J, McMahon CM, et al. Severity of age-related hearing impairment is associated with impaired activities of daily living. Age Ageing. 2012;41:195–200. doi: 10.1093/ageing/afr155. [DOI] [PubMed] [Google Scholar]
- 18.Reuben DB, Mui S, Damesyn M, et al. The prognostic value of sensory impairment in older persons. J Am Geriatr Soc. 1999;47:930–935. doi: 10.1111/j.1532-5415.1999.tb01286.x. [DOI] [PubMed] [Google Scholar]
- 19.Laforge RG, S W, Sternberg J. The relationship of vision and hearing impairment to one-year mortality and functional decline. J Aging Health. 1992;4:126–148. [Google Scholar]
- 20.Lin MY, Gutierrez PR, Stone KL, et al. Vision impairment and combined vision and hearing impairment predict cognitive and functional decline in older women. J Am Geriatr Soc. 2004;52:1996–2002. doi: 10.1111/j.1532-5415.2004.52554.x. [DOI] [PubMed] [Google Scholar]
- 21.Rudberg MA, Furner SE, Dunn JE, et al. The relationship of visual and hearing impairments to disability: An analysis using the longitudinal study of aging. J Gerontol. 1993;48:M261–M265. doi: 10.1093/geronj/48.6.m261. [DOI] [PubMed] [Google Scholar]
- 22.Centers for Disease Control and Prevention. National Centers for Health Statistics. The National Health and Nutrition Examination Survey. [Accessed 9 Dec 2012]; Audiometry Manual 2005–2006 [on-line]. Available at http://www.cdc.gov/nchs/data/nhanes/nhanes_05_06/AU.pdf.
- 23.World Health Organization Prevention of Blindness and Deafness (PBD) Program. Prevention of Deafness and Hearing Impaired Grades of Hearing Impairment [on-line] [Accessed 9 Dec 2012]; Available at http://www.who.int/pbd/deafness/hearing_impairment_grades/en/index.html.
- 24.Clark JG. Uses and abuses of hearing impairment classification. ASHA. 1981;23:493–500. [PubMed] [Google Scholar]
- 25.Plantinga LC, Johansen K, Crews DC, et al. Association of CKD with disability in the United States. Am J Kidney Dis. 2011;57:212–227. doi: 10.1053/j.ajkd.2010.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Control CfD. NHANES Analytic and Reporting Guidelines [on-line] [Accessed February 1, 2011]; Available at www.cdc.gov/nchs/data/nhanes/nhanes_03_04/nhanes_analytic_guidelines_dec_2005.pdf.
- 27.Agrawal Y, Carey JP, Della Santina CC, et al. Disorders of balance and vestibular function in US adults: Data from the National Health and Nutrition Examination Survey, 2001–2004. Arch Intern Med. 2009;169:938–944. doi: 10.1001/archinternmed.2009.66. [DOI] [PubMed] [Google Scholar]
- 28.Lin FR, Ferrucci L. Hearing impairment and falls among older adults in the United States. Arch Intern Med. 2012;172:369–371. doi: 10.1001/archinternmed.2011.728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cruickshanks KJ, Klein R, Klein BE, et al. Cigarette smoking and hearing impairment: The Epidemiology of Hearing Impairment Study. JAMA. 1998;279:1715–1719. doi: 10.1001/jama.279.21.1715. [DOI] [PubMed] [Google Scholar]
- 30.Dalton DS, Cruickshanks KJ, Klein R, et al. Association of NIDDM and hearing impairment. Diabetes Care. 1998;21:1540–1544. doi: 10.2337/diacare.21.9.1540. [DOI] [PubMed] [Google Scholar]
- 31.Lin FR, Ferrucci L, Metter EJ, et al. Hearing impairment and cognition in the Baltimore Longitudinal Study of Aging. Neuropsychology. 2011;25:763–770. doi: 10.1037/a0024238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Woollacott M, Shumway-Cook A. Attention and the control of posture and gait: A review of an emerging area of research. Gait Posture. 2002;16:1–14. doi: 10.1016/s0966-6362(01)00156-4. [DOI] [PubMed] [Google Scholar]
- 33.Watson NL, Rosano C, Boudreau RM, et al. Executive function, memory, and gait speed decline in well-functioning older adults. J Gerontol A Biol Sci Med Sci. 2010;65A:1093–1100. doi: 10.1093/gerona/glq111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Weinstein BE, Ventry IM. Hearing impairment and social isolation in the elderly. J Speech Hear Res. 1982;25:593–599. doi: 10.1044/jshr.2504.593. [DOI] [PubMed] [Google Scholar]
- 35.Unger JB, McAvay G, Bruce ML, et al. Variation in the impact of social network characteristics on physical functioning in elderly persons: MacArthur Studies of Successful Aging. J Gerontol B Psychol Sci Soc Sci. 1999;54B:S245–S251. doi: 10.1093/geronb/54b.5.s245. [DOI] [PubMed] [Google Scholar]
- 36.Mendes de Leon CF, Glass TA, Beckett LA, et al. Social networks and disability transitions across eight intervals of yearly data in the New Haven EPESE. J Gerontol B Psychol Sci Soc Sci. 1999;54B:S162–S172. doi: 10.1093/geronb/54b.3.s162. [DOI] [PubMed] [Google Scholar]
- 37.Cohen-Mansfield J, Infeld DL. Hearing aids for nursing home residents: current policy and future needs. Health Pol. 2006;79:49–56. doi: 10.1016/j.healthpol.2005.11.016. [DOI] [PubMed] [Google Scholar]


