Abstract
Background: Maternal obesity is a growing public health concern in Belgium as well as in other European countries and is now becoming the most common risk factor associated with pregnancy complications with impact on the health of the women and her offspring. At this moment, there is no specific management strategy for obese pregnant women and mothers, focusing on physical health and psychological well-being.
Objectives: We aimed (1) to study the influence of socio-demographic and obstetrical correlates on pre-pregnancy body mass index (BMI) and gestational weight gain (GWG) in different regions of Flanders, Belgium, (2) to review the literature on the onset and progression of labour in normal weight and obese pregnant women, (3) to compare levels and evolution of anxiety and depressed mood during pregnancy between obese women and normal-weight women, (4) to examine whether a prenatal lifestyle intervention programme, based on principles of motivational interviewing, in obese pregnant women reduces GWG and lowers levels of anxiety and depressed mood during pregnancy, (5) to examine associations between inter-pregnancy weight change from the first to the second pregnancy and the risk for adverse perinatal outcomes during the second pregnancy and finally (6) to study predictors of postpartum weight retention (PPWR) in obese mothers at six months after delivery in order to provide clues for the design of interventions aimed at preventing weight retention related to childbearing.
Methods: We performed an epidemiological study, an intervention study during pregnancy with postpartum follow up and a literature review.
Results: One in three Flemish women start pregnancy being overweight or obese and this prevalence has slowly been rising since 2009 in the Flanders. We identified women at risk for a high pre-pregnancy BMI and excessive GWG, both being important predictors for increased pregnancy and birth related complications. In a literature review, we showed that the combination of a higher incidence of post-term deliveries and increased inadequate contraction pattern during the first stage of labour in obese women suggests an influence of obesity on myometrial activity. Given the low compliance for adequate GWG in obese women in the general Flemish population and their increased psycho-social vulnerability compared to the normal weight pregnant women, counselling obese pregnant women can lead to a reduced GWG and increased psychological comfort. Stabilizing inter-pregnancy maternal weight for all women is an important target for reducing adverse perinatal outcomes in the subsequent pregnancy. Psychological discomfort during pregnancy does impact on PPWR in obese mothers six months after delivery.
Discussion and conclusion: Focusing on weight management in obese women before, during and after a pregnancy has advantages for both the mother and her infant. Theoretical and practice based training modules should be developed and focus on: (1) awareness of techniques for identifying the clearly identified risk groups with a high pre-pregnancy BMI and excessive GWG, (2) the increased perinatal risks, (3) an adapted perinatal management and (4) counselling techniques for an adequate weight management and psychological wellbeing in obese pregnant women. To achieve better care for the future, we must focus on tackling maternal obesity. This means that obese women should be reached before they get pregnant for the first time. Targeting primary and community based care, promotion and education are challenging, but the psychosocial context should be acknowledged.
Keywords: Pregnancy, obesity, epidemiological study, intervention study, interpregnancy, perinatal outcome, psychological outcome
Introduction
Obesity is a medical condition which is characterized by an abnormal or excessive fat accumulation that may impair health. The body mass index (BMI) is defined as weight (kg), divided by the square of the height (m) and is commonly used to diagnose obesity in adult men and women. When the BMI (kg/m²) exceeds a predetermined threshold as shown in table I, a classification that forms the basis of a diagnosis of the degree of fat mass can be made. Although the BMI is highly correlated with the degree of fat mass, it does not directly measure the percentage of body fat, fat distribution or composition. Nevertheless, the BMI offers a more accurate assessment of excess adiposity than weight alone, and is a well reproducible indicator of metabolic risk. Due to its simplicity, most epidemiological and clinical studies consider the BMI as a useful and easy method to measure surrogate marker for increased health risks related to excess body fat (Fattah et al., 2009; WHO, 2012).
Obesity is rising worldwide, including in women of reproductive age. In Belgium, 47% of the general population is either overweight (BMI ≥ 25 kg/m², 33%) or obese (14%) (Van der Heyden J. et al., 2010). Depending on the cohorts studied, the BMI cut-offs used and the period of evaluation, the prevalence of obesity in pregnant women (hereafter referred to as maternal obesity) ranges from 1.8% to 25.3% (Guelinckx, Devlieger, Beckers, & Vansant, 2008). Because of the absence of systematically recorded regional data on maternal pre-pregnancy weight, height, and weight at delivery, no comprehensive representative picture of the epidemic burden of maternal obesity is available in most countries (Institute of Medicine, 2009). Most published figures are derived from cross-sectional and regional cohorts. In Belgium, the Flemish Study Centre for Perinatal Epidemiology (SPE) has routinely collected data on maternal pre-pregnancy weight (kg) and height (m) as well as maternal weight at delivery (kg) since 2009. This enables us to make a representative regional picture of maternal obesity and gestational weight gain (GWG) in the region of Flanders, the Northern part of Belgium.
It is well documented that a high pre-pregnancy BMI (Guelinckx et al., 2008; Heslehurst et al., 2008) and excessive GWG (Nohr et al., 2008; Institute of Medicine, 2009) are both associated with obstetric complications including hypertensive disorders, gestational diabetes mellitus (GDM), caesarean section (CS) and higher postpartum weight retention (PPWR) (Heslehurst et al., 2008; Poobalan et al., 2009; Nelson et al., 2010). Foetal risks include miscarriage, neural-tube defects, heart defects, macrosomia and stillbirth (Callaway et al., 2006; Sarwer et al., 2006). Complications are even higher when pre-pregnancy obesity is combined with excessive GWG, especially the increased risk of caesarean delivery, macrosomia and postpartum weight retention (Cedergren, 2006; Nohr et al., 2008; Institute of Medicine, 2009). In 2009, the IOM released new guidelines with specified lower and upper limits for GWG in obese pregnant women (Table I). Concurrently, they highlighted the importance of interventions in pregnancy to achieve weight gain within these recommended ranges, in order to prevent adverse gestational outcomes, maternal post-partum obesity and childhood obesity (Institute of Medicine, 2009).
Table I. Recommended ranges of gestational weight gain (GWG) by pre-pregnancy BMI according to the Institute of Medicine (IOM) guidelines, 2009.
| Pre-pregnancy BMI category | Recommended weight gain ranges (kg) | Rates of weight gain in the second and third trimesters, mean (range) kg/week |
|---|---|---|
| Underweight (< 18.5 kg/m²) | 12.5-18 | 0.51 (0.44-0.58) |
| Normal weight (18.5-24.9 kg/m²) | 11.5-16 | 0.42 (0.35-0.50) |
| Overweight (25-29.9 kg/m²) | 7-11.5 | 0.28 (0.23-0.33) |
| Obese (≥ 30 kg/m²) | 5-9 | 0.22 (0.17-0.27) |
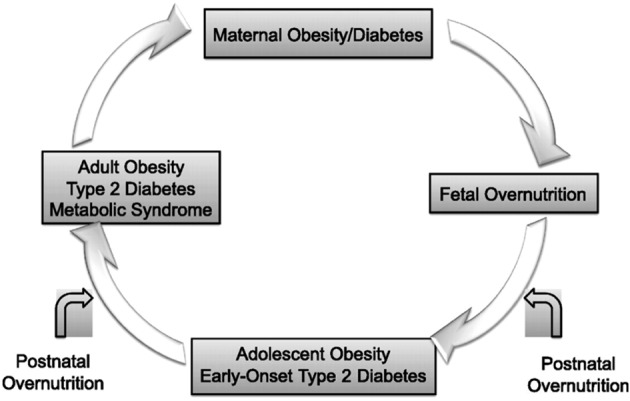
Maternal obesity not only impacts on the metabolism of the mother, but may also confer risk of obesity to her offspring (Nelson et al., 2010; O’Reilly & Reynolds, 2013). Epidemiological studies and animal models confirm the developmental origins of obesity. Offspring born to obese mothers are often larger at birth and show increased adipose tissue mass and obesity and diabetes risk in later life (O’Reilly & Reynolds, 2013). As the prevalence of maternal obesity continues to rise, increasing numbers of children are exposed to an ‘obese intrauterine environment’. Obesity among pregnant women has therefore the potential to result in an ‘intergenerational cycle’ through in utero programming (Fig. 1). Concurrently, a positive correlation between the pre-pregnancy BMI, GWG and anxiety and depressive symptoms was suggested in earlier studies, indicating a possible correlation between (pre)-pregnancy weight status and psychological vulnerability (Bodnar et al., 2009; Laraia et al., 2009). Obesity and stress, both are stimulating factors for in utero programming effects. In the light of the in utero programming theory, the mental health of obese pregnant women should be explored thoroughly and findings should be taken into account when developing perinatal care programmes for pregnancy high-risk groups.
Fig. 1. Intergenerational cycle of obesity: in utero programming of obesity and metabolic dysfunction.
The first six months after delivery are an important time frame to explore clinically important predictors of PPWR which can have an important long term impact on maternal weight and health (Linne et al., 2004; Rooney et al., 2005). Inter-pregnancy weight gain is often a result of PPWR and plays an important role in the development of obesity among women of childbearing age. The influence of inter-pregnancy weight changes on perinatal outcomes in the next pregnancy are understudied.
To develop a tailored management strategy for obese pregnant women and mothers that results in improved physical and psychological outcomes during the perinatal period, six research questions regarding the pregnancy, delivery and inter-pregnancy period were formulated (Table II).
Table II. Research questions and study designs according to the time of investigation.
| PREGNANCY | DELIVERY | INTER-PREGNANCY | |
|---|---|---|---|
| Postpartum period 1-6 months | Inter-pregnancy period > 6 months | ||
| 1. What is the association between socio-demographic and obstetrical correlates with pre-pregnancy BMI and GWG in Flanders? Epidemiological study. | |||
| 2. How does obesity affects the onset and progression of labour? Review. | |||
| 3. Are levels and evolution of anxiety and depressed mood in obese pregnant women different from pregnant women with a normal pre-pregnancy BMI? Prospective controlled cohort study. | |||
| 4. Can a targeted antenatal lifestyle intervention programme for obese pregnant women influence gestational weight gain and mental health? Randomized controlled trial. | |||
| 5. What is the association between inter-pregnancy weight change and the risk for adverse perinatal outcomes in the subsequent pregnancy? Epidemiological study. | |||
| 6. What are predictors of postpartum weight retention in obese mothers six months after delivery? Longitudinal cohort study. | |||
1. Socio-demographic and obstetrical correlates of pre-pregnancy body mass index and gestational weight gain
In a first part of this thesis, the influence of socio-demographic (i.e., maternal age, education, profession, employment, ethnicity and marital state) and obstetrical (i.e., duration of pregnancy, parity, hypertension and GDM) correlates on pre-pregnancy BMI and GWG in different regions of the Belgian Flanders were studied.
1.1. Subjects and Methods
The Flemish Study Centre for Perinatal Epidemiology (SPE) routinely registers perinatal data from all deliveries in Flanders. Perinatal data from all maternity units are collated centrally, subjected to an error detection program, and checked for accuracy and completeness through feedback with the individual units and reassessment of patient records when needed (Cammu et al., 2011). For this study, we initially considered all 66312 live-born singleton deliveries in 2009. We excluded multiple pregnancies as these pregnancies have specific guidelines regarding GWG (Institute of Medicine, 2009). We excluded women with missing data on maternal height (n = 7295), maternal pre-pregnancy weight (n = 5702) and maternal weight at delivery (n = 6247). Cumulative missing data for individual women meant that a total of 9153 cases were excluded. We also excluded women who delivered before 37 completed weeks of gestation (n = 3137). Ranges of maternal height (1.40-1.92 m), pre-pregnancy weight (35-154 kg) and GWG, (-26 - +50 kg) were found to be acceptable. A total of 54022 pregnancies were included in the analysis.
BMI was categorized in accordance to the WHO (Table I). The obese pregnant women group was further divided into class I (BMI 30 – 34.9kg/m² - moderate obesity), class II (BMI 35 – 39.9kg/m²- extreme obesity) and class III (BMI ≥ 40 kg/m²- morbid obesity). GWG was calculated by subtracting the maternal pre-pregnancy weight from the weight at delivery and was categorized as insufficient, adequate or excessive in accordance to the Institute of Medicine (IOM) guidelines for each category of pre-pregnancy BMI (Institute of Medicine, 2009) (Table I).
1.2. Results
Nearly one-third of women are overweight (21.6%) or obese (10.1%) at the start of pregnancy and GWG as recommended by the IOM (5-9 kg) occurs in only 28% of obese women. Figure 2 shows the distribution of maternal pre-pregnancy BMI (a) and GWG (b) in the total study population.
Fig. 2. Distribution of maternal BMI (a) and GWG (b) in total population (n = 54 022).
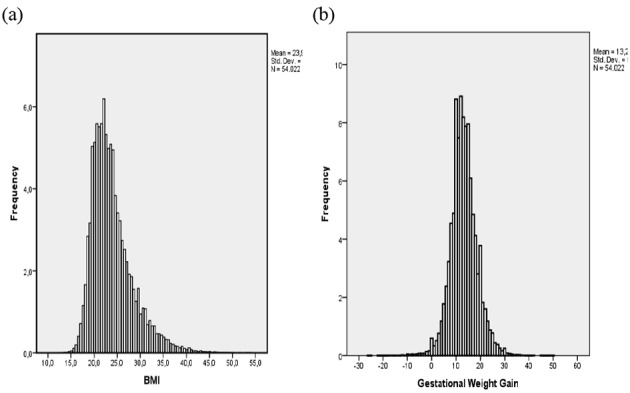
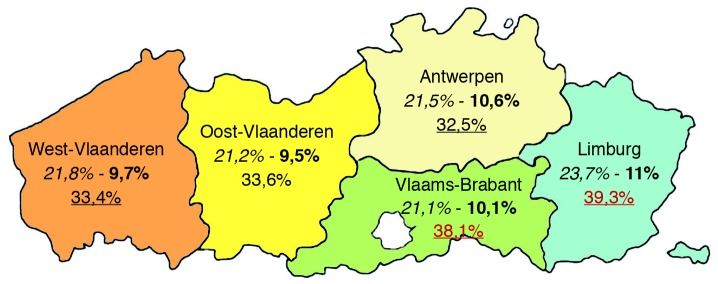
A high pre-pregnancy BMI is significantly more prevalent in older multiparous women with lower levels of education and in those with a lower professional state and belonging to ethnic minority groups. Compared to adequate GWG (as recommended by the IOM by BMI category), excessive GWG (more than recommended by the IOM) is more common in younger women (<2 0 years), with a higher pre-pregnancy BMI and pregnancy-induced hypertension. Moreover, younger (20-24 years), single women, or women from ethnic minority groups show higher odds for excessive as well as for insufficient GWG (less than recommended by the IOM); while those with a high/highest educational level have lower odds for excessive and insufficient GWG. Regional differences became apparent with the highest mean pre-pregnancy BMI (24.2 kg/m², SD 4.6) and prevalence of maternal obesity (11%) in Limburg, the most eastern part of the Flanders (Fig. 3).
Fig. 3. Percentage of maternal overweight, obesity and excessive GWG, respectively in 2009 by region in Flanders (N = 54022).
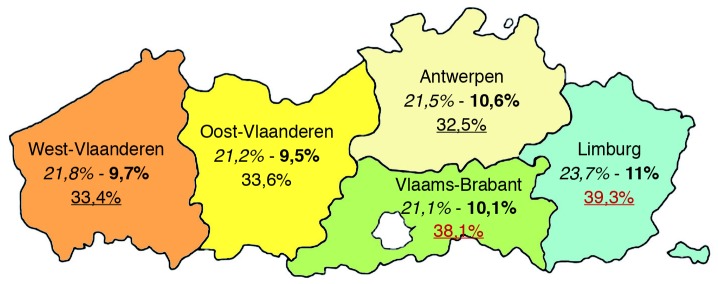
The results of this study highlight the scale of the problem of maternal obesity and excessive GWG for this region and offers opportunities to target educational campaigns and intervention programmes in a clinical setting.
2. Obesity in pregnancy: altered onset and progression of labour
Labour is often complicated in obese pregnant women, and this in an apparently contradictory way. On one hand, a high BMI is associated with an increased risk for induced and PPROM (preterm prelabour rupture of membranes) related preterm childbirth (Nohr et al., 2007). In contrast to that, there is a decreased risk for spontaneous preterm childbirth (Hendler et al., 2005; Beyer et al., 2011). Also, obese women have a higher risk than overweight and lean women to progress beyond term, with a higher incidence of post-term childbirths (Heslehurst et al., 2008). Finally, obese women tend to have higher odds for induction of labour and failure to progress with labour (Heslehurst et al., 2008; Schrauwers & Dekker, 2009). Therefore the aim of this literature review was to investigate the current knowledge on the pathophysiology of the onset and progression of labour in normal weight and obese pregnant women in order to understand the specificities of labour initiation and progression in these two groups of women and to provide the reader with practical recommendations regarding the management of labour in this obstetrically high risk population.
2.1. Methods
Articles for this review were identified by consulting the electronic databases Medline, Cochrane Library, Web of Science and Science Direct, using the keywords ’pregnancy’ and ‘obesity’combined with respectively ‘labour obstetric’ ‘labour onset’ ‘labour first stage’, ‘labour second stage’and ‘uterine contraction’. The search was limited to English papers, published between 2000 and 2010. We only included original research articles concerning labour related complications in obese pregnant women. We excluded opinion articles, case reports, anecdotes and clinical descriptions. Key themes were ‘postdate’, ‘induction of labour’, ‘failed induction of labour’, ‘onset of labour’, ‘progression of labour’ and ‘prolonged first stage of labour’. We chose to undertake a critical summary of research, in order to determine associations, gaps or inconsistencies in this specific but limited body of research attuned to practical recommendations regarding adequate labour management in the obese population (Polit & Beck, 2008).
2.2. Results
The literature of the pathophysiology of onset and progression of labour in obese women is reviewed to understand the specificities of labour onset and progression and to provide practical suggestions for an adapted labour approach in this high-risk group. In human pregnancy, the onset of labour is a not yet fully understood for reasons of multifactorial physiological and psychological process involving mechanical and hormonal interactions between the mother and her foetus. The physiological pathway appears to involve activation of the foetal hypothalamic-pituitary-adrenal (HPA) axis and placental corticotrophin-releasing hormone (CRH). However, the precise biological role of CRH in foeto-maternal tissue remains subject to debate (Norwitz et al., 1999; Grammatopoulos, 2008). Figure 4 summarises the current knowledge in a hypothetical model developed by the authors on maternal, foetal and placental factors playing a role in the initiation of labour. This model was based on most relevant and available sources at the moment of construction (Norwitz et al., 1999; Mesiano, 2004; Martin W.L. & Hutchon, 2004; Li & Challis, 2005; Bisits et al., 2005; Mesiano & Welsh, 2007; Smith, 2007; Grammatopoulos, 2008; Anubhuti & Arora, 2008; Madan et al., 2009).
Fig. 4. Hypothetical model, on maternal, foetal and placental factors playing a role in the initiation of labour.
The combination of a higher incidence of post maturity and increased inadequate contraction pattern during the first stage of labour in obese women, suggests an influence of obesity on myometrial activity. Standard definitions of poor labour progress and an adapted interpretation of labour curves for obese women should be re-evaluated in the light of this information.
3. Anxiety and depressed mood in obese pregnant women: a prospective controlled cohort study
Although several studies have looked at maternal anxiety and depression during pregnancy (Andersson et al., 2006; Faisal-Cury & Rossi, 2007; Breitkopf et al., 2006; Austin et al., 2007; Heron et al., 2004), little research has focused on these emotions in obese pregnant women. It is generally well known that obese women are more likely to suffer from higher levels of anxiety and depression, compared to normal-weight women (Zhao et al., 2009; Zender & Olshansky, 2009; Gariepy et al., 2010). In a recent Danish cohort study, a positive association was observed between psychosocial factors such as feelings of depression and levels of anxiety during pregnancy, and postpartum weight retention at 6 and 18 months (Pedersen et al., 2011), indicating that pregnant women burdened by psychological stressors are particularly vulnerable to excessive weight retention related to childbearing. The aim of our study was to compare the levels and evolution of anxiety and depressed moods during pregnancy between obese women (study group) and normal-weight women (controls), as a first step towards the development of a global lifestyle-coaching health programme for obese pregnant women.
3.1. Materials and methods
This study evaluates levels of anxiety and depressed mood in obese (n = 63) and normal weight pregnant woman (n = 156), whilst taking into account socio-demographic and medical pregnancy variables. Women were included prospectively during routine antenatal visits and they all received the normal standard antenatal care. None of those hospitals offered specific antenatal care programmes for obese pregnant women at the time of the study. Obese and normal-weight women were recruited for participation in the study between 7 and 14 weeks of pregnancy. Women who had pre-existing type 1 diabetes, a multiple pregnancy, who had a primary need for nutritional advice or insufficient knowledge of the Dutch language were excluded. Socio-demographic and psychological data were obtained at the time of study entry, before 15 weeks of pregnancy (base line measurement, trimester 1). Psychological evaluation was furthermore performed between 18 and 22 weeks (trimester 2) and between 30 and 34 weeks of pregnancy (trimester 3) in both groups. None of the study women were on antidepressants. Levels of anxiety were measured with the use of a Dutch standardized version of the State-Trait Anxiety Inventory (STAI)(Spielberger et al., 2000). The STAI comprises two self-report scales for measuring two distinct anxiety aspects: state-anxiety and trait-anxiety. Both scales contain 20 statements asking the pregnant women to describe how they feel. The state-anxiety scale includes statements about the intensity of feelings at a particular moment in time, whereas the trait-anxiety scale includes statements about the frequency of general feelings. A composite score is generated for each subscale after a reversal of the negative items, ranging for each scale from a minimum of 20 to a maximum of 80. High scores mean more state or trait anxiety. Feelings of depression, i.e. depressed moods, during pregnancy were assessed by using the 10-item Edinburgh Postnatal Depression Scale (EPDS). This scale was originally designed as a screening instrument for postnatal depression, but was also validated later for usage during pregnancy (Rich-Edwards et al., 2006; Cox, Holden, & Sagovsky, 1987; Pop et al., 1992) and named Edinburgh Depression Scale (EDS).
Linear mixed-effect models with repeated measures were used to analyse group differences for levels of anxiety and depressed mood, checking for the effects of those covariates that correlated significantly with state or trait anxiety, or depressed mood. The covariates considered were: maternal age and education, marital state, employment, profession, ethnicity, parity, history of miscarriages, gravidity, cigarette smoking and alcohol consumption, spontaneous or assisted conception, psychological history and diabetes and hypertensive disorders in the current pregnancy. All analyses were performed using the statistical software SAS Enterprise Guide 4.2, by Cary, NC: SAS Institute Inc.
3.2. Results
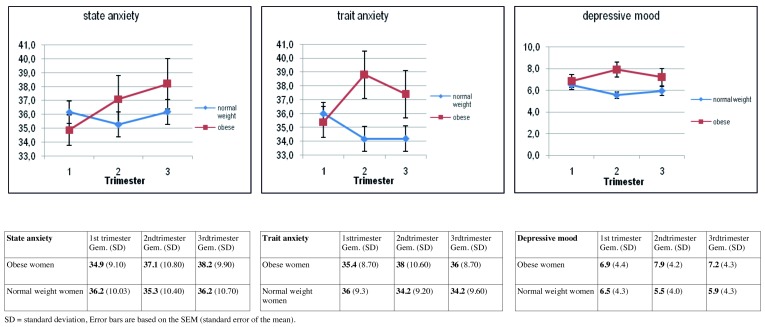
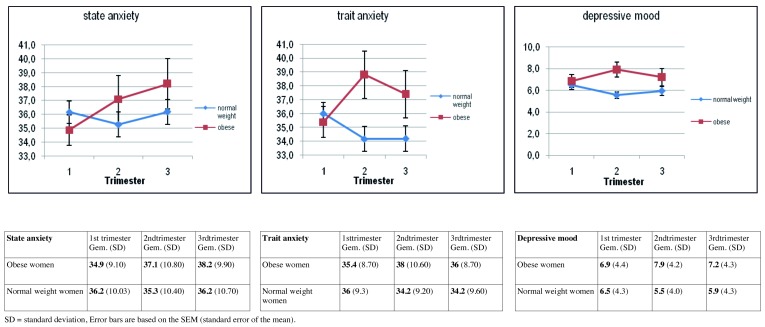
Compared to normal weight pregnant women, obese pregnant women demonstrate higher levels of anxiety from trimester 1 to 3 (β = 3.70, p = 0.007), while no difference is shown during the pregnancy in normal weight women, or after controlling for relevant confounding variables. The significant decrease in levels of trait anxiety and depressed mood during pregnancy in normal weight women in our sample is not found in obese pregnant women (Fig. 5). The decreasing level of psychological discomfort throughout pregnancy which has been described in other studies and also in normal weight pregnant women in this cohort, is not present in obese pregnant women. Maternal characteristics like lower level of education, belonging to an ethnic minority group, living without a partner, psychological history of depression and miscarriages and higher parity, which are more prevalent in obese pregnant women compared to their normal weight counterparts, significantly influence levels of anxiety and depressed mood during pregnancy. The increased psychosocial vulnerability of obese pregnant women has to be taken into account when developing interventional programmes and tailored counselling.
Fig. 5. Mean levels and error bars of State and Trait Anxiety and Depressed mood in obese and normal-weight pregnant women by trimester.
4. Effects of lifestyle intervention in obese pregnant women on gestational weight gain and mental health: a randomized controlled trial
Lifestyle intervention could help obese pregnant women to limit their weight gain during pregnancy and improve their psychological comfort, but has not yet been evaluated in randomized controlled trials. We evaluated whether a targeted antenatal lifestyle intervention programme for obese pregnant women influences gestational weight gain (GWG) and levels of anxiety/depressed mood.
4.1. Materials and methods
This study used a longitudinal interventional design. Of the 235 eligible obese pregnant women, 205 [mean age (y): 29 ± 4.5; body mass index (BMI, kg/m²): 34.7 ± 4.6] were randomized to a control group, a brochure group receiving written information on healthy lifestyle and an experimental group receiving an additional four antenatal lifestyle intervention sessions by a midwife trained in motivational lifestyle intervention. Exclusion criteria were: gestational age > 15 weeks, pre-existing type 1 diabetes, multiple pregnancy, primary need for nutritional advice and insufficient knowledge of the Dutch language. Measurement of feelings of anxiety (State and Trait Anxiety Inventory, STAI) and depressed mood (Edinburgh Depression Scale, EDS) were obtained at entry into the study, before 15 weeks of gestation (baseline measurement, trimester 1) in all the 3 groups. Psychological assessment was furthermore repeated in the second (between 18 and 22 weeks) and third trimester (between 30 and 34 weeks) of gestation. All women received a weight gain chart to fill in each time they went for a prenatal visit. Socio-demographical, behavioural, psychological and medical variables were used for controlling and correcting outcome variables.
A maximum of three obese pregnant women were brought together in a 1,5 to 2h session always supported by the same midwife (AB). Principles of this lifestyle intervention programme for obese pregnant women were based on the stages of the behavioural change model of Prochaska & Diclemente (Johnson et al., 2008), as well as on the concept of motivational interviewing (DiMarco et al., 2009). Personal barriers to behavioural change were explored and as much as possible positive verbal reinforcement given to increase each pregnant woman’s self-confidence and self-efficacy. Although the main focus was on nutritional advice and physical activity, worries and personal questions concerning their pregnancy were addressed.
For levels of anxiety and depressed mood multivariate linear mixed effects model with repeated measures were used, since every woman could have three different outcomes (one for every trimester). Method of delivery was analysed by a logistic regression. The use of a direct likelihood model, which was the case in our analysis, seems to be the best to deal with missing data in a longitudinal data collection within controlled trials (Polit & Beck, 2008). Attrition bias was analysed in this longitudinal data collection by comparing socio-demographic differences as well as differences in main outcome variables between the groups with complete and incomplete lifestyle intervention sessions, and between those with complete and missing data concerning psychological measurements throughout pregnancy. All analyses were performed with the SAS statistical software Enterprice Guide 4.2, by Cary, NC: SAS Institute Inc.
4.2. Results
We found a significant reduction of GWG in the brochure (9.5kg) and lifestyle intervention (10.6kg) group compared to the normal care group (13.5kg) (p = 0.007). Furthermore, levels of anxiety significantly decreased in the lifestyle intervention group and increased in the normal care group during pregnancy (p = 0.02) (Fig. 6); no differences were demonstrated in the brochure group. Pre-pregnancy BMI was positively related to levels of anxiety. Obese pregnant women who stopped smoking recently showed a significant higher GWG (β = 3.04; p = 0.01); those with concurrent Gestational Diabetes Mellitus (GDM) (β = 3.54; p = 0.03) and those who consumed alcohol on a regular base (β = 3.69; p = 0.04) showed significant higher levels of state anxiety. No differences in depressed mood or obstetrical/neonatal outcomes were observed between the three groups. The number needed to treat with lifestyle interventions as organised in this trial to observe one more obese women with a GWG less than 9 kg and a low (< 40) state anxiety level in the third trimester of pregnancy is 17. The cost-benefit analysis of counselling obese pregnant women on long-term health outcomes for the mother and her child should be further investigated.
Fig. 6. Levels of state anxiety by trimester in the 3 study groups.
5. Interpregnancy weight retention and risk for adverse perinatal outcome: an epidemiological study
Perinatal complications in obese women and in those with excessive gestational weight gain (GWG) are well documented (Nohr et al., 2008; Guelinckx et al., 2008; Heslehurst et al., 2008; Madan et al., 2010; Mamun et al., 2011; Weiss et al., 2004; Yogev & Catalano, 2009). The cross-sectional design of most studies on the subject calls for caution when interpreting the results. Longitudinal data from large cohorts of pregnant women are needed to get stronger and valid estimates on the temporal and causal relation between maternal obesity and perinatal outcomes. This means that perinatal risks should also be analysed against changes in maternal weight between two consecutive pregnancies. We therefore examined associations between maternal weight changes between the start of the first and the second pregnancy and the risk for adverse perinatal outcomes in a regional representative cohort of women who had their first two consecutive singleton births in Belgium between 2009 and 2011.
5.1. Methods
All live born singleton births delivered at 20-42 weeks of gestation in women who had their first two consecutive births between 2009 and 2011 (N = 7897). By merging the variables for the first and second delivery in the same mother, interpregnancy weight difference was calculated. Multiple pregnancies were excluded. 200,706 singleton births occurred between January 2009 and December 2011. After exclusion of those with missing first date of birth in their second pregnancy data file (n = 2 821) and missing maternal height or pre-pregnancy weight or weight at delivery (n = 17 216), the data for 180,669 births remained. After merging women who had their first and second birth between 2009-2011 (n = 8792) and excluding those with differences in maternal height more than or equal to 5 cm between the two consecutive pregnancies (n = 895), the final cohort for analysis consisted of 7897 women.
Ranges of maternal height (1.42-1.90 m), pre-pregnancy weight (35-144 kg) and weight at delivery (44-160 kg) were found to be acceptable. Interpregnancy weight change (ΔIPW) was calculated as the difference between the pre-pregnancy BMI of the first pregnancy and the pre-pregnancy BMI of the second pregnancy. Besides analysing it as a continuous variable, ΔIPW was categorized as less than -1 (BMI loss more than 1 unit (kg/m²)), -1 to less than 1 (reference group), 1 to less than 2, 2 to less than 3 and 3 or more BMI units.
The main outcomes of interest for maternal pregnancy related complications were: pregnancy induced hypertension (PIH), gestational diabetes mellitus (GDM) and caesarean section (CS); for neonatal and birth related complications, macrosomia (≥ 4000 g), low birth weight (< 2500 g) and congenital malformations were examined. All these outcomes were retrieved from the SPE database.
As we found statistically significantly interactions between the pre-pregnancy BMI at first pregnancy and categorical BMI changes between pregnancies, we decided also to build logistic regression models for each outcome stratified by whether the woman’s pre-pregnancy BMI at first pregnancy was below or above 25 kg/m². To visualize the impact of ΔIPW on the five outcome variables, we constructed logistic regression models as described above, but this time keeping all categories of the ΔIPW in the model. All p values were 2-sided and values less than .05 were considered statistically significant. SAS version 9.3 (SAS Institute, Cary, North Carolina) was used for all analyses.
5.2. Results
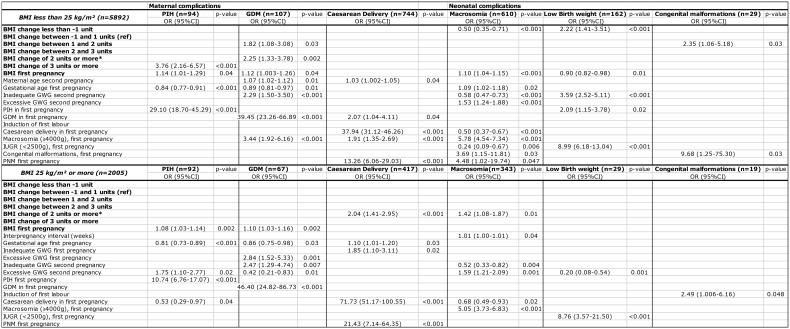
Mean pre-pregnancy BMI increased from 23.3 kg/m² (SD 4.0) at the start of the first pregnancy to 23.9 kg/m² (SD 4.4) at the start of the second pregnancy (p = 0.0001). Between two consecutive pregnancies, we noticed an increase in the prevalence of overweight and obese women, from 25.4% to 31.4%. In the total cohort, 17.2% of women gained ≥ 2 BMI units (± 5 kg) at the start of the second pregnancy. In the group of obese women at the onset of the first pregnancy, 28.7% gained ≥ 2 BMI units at the start of the second pregnancy compared to 15% in the normal weight women (p < 0.001). Overall, 19% of women shifted between BMI categories between their first two consecutive pregnancies; 13.7% shifted to a higher BMI category, while 5.3% shifted to a lower BMI category. The largest shifts occurred in the group of normal weight women, of whom 8.6% shifted from normal weight to overweight; followed by 3.0% of overweight women who shifted to the obese category.
The adjusted odds ratio (aOR) for PIH was 3.76 (95% CI 2.16-6.57; p < 0.001) with an increase of ≥ 3 BMI units between pregnancies; the aOR for GDM was 1.82 (95% CI 1.08-3.08; p = 0.03) for IPW retention between one and two BMI units, and 2.25 (95% CI 1.33-3.78; p = 0.002) for IPW retention of ≥ 2 BMI units; but these associations were only present in underweight and normal weight women. In overweight and obese women, the aOR was 2.04 (95% CI 1.41-2.95; p < 0.001) for caesarean section and 1.42 (95% CI 1.08-1.87; p = 0.01) for macrosomia for an IPW retention of ≥ 2 BMI units. In underweight and normal weight women, the risk for macrosomia was halved if women lost more than one BMI unit between pregnancies, but at the same time the risk for low birth weight doubled (table III).
Table III. Adjusted odds ratios for adverse perinatal outcomes during second pregnancy in relation to change in categories of BMI between the first and second pregnancy, stratified by prepregnancy BMI at first pregnancy.
6. Anxiety during early pregnancy predicts postpartum weight retention in obese mothers
Postpartum weight retention (PPWR) plays an important role in the development of obesity among women of childbearing age. Given the scarce literature on the topic, we aimed to study predictors of PPWR in obese mothers at six months after delivery, in a longitudinal study in order to provide clues for the design of interventions aimed at preventing interpregnancy weight gain in this high risk obstetric population. Our hypothesis is that psychosocial (i.e. levels of anxiety and depression) and behavioural (smoking, sleeping and breastfeeding behaviour) variables during the perinatal period influence six-month post-partum weight retention in obese mothers.
6.1. Methods
We performed a prospective longitudinal cohort study to identify and understand factors that could be used to predict PPWR in obese mothers. Postpartum data from an interventional trial in obese pregnant women (n = 197), conducted in three regional hospitals in the Belgian Flanders, between March 2008 and June 2012, were available from 150 mothers. Predictors were examined from the pregnancy and post-partum period. Descriptive statistics were performed and linear regression models constructed. PPWR was calculated as maternal weight six months after delivery minus pre-pregnancy weight. Pre-pregnancy weight was self-reported at the time of study inclusion as described before (Bogaerts et al., 2012) and maternal weight after delivery was self-reported by questionnaires obtained at six months after delivery. To perform multivariate analysis, PPWR was used as a continuous variable. We chose to explore PPWR at six months after delivery because failure to lose pregnancy related weight in an appreciable time of six months after delivery is an important indicator of obesity in midlife (Rooney et al., 2005). Demographic (maternal age, education and parity), socio-economic (ethnicity, household income), behavioural (smoking behaviour, duration of maternal sleep and breastfeeding) and psychological data (levels of anxiety and depressed mood) were obtained through interviews and self-administered questionnaires at entry into the study, before 15 weeks of gestation, and from self-administered questionnaires, six months after delivery. Anxiety and levels of depression were measured as mentioned earlier in this thesis.
6.2. Results
The mean pre-pregnancy BMI in this study group was 34.4 kg/m² (SD 4.5), mean maternal age was 28.9 year (SD 4.1). The mean GWG was 10.9 kg (SD 6.8, range 9, +30 kg), maternal height 1.66 m (SD 0.06) and maternal weight at six months after delivery 93.6 kg (SD 13.6, range 69, 135 kg). Most obese mothers (59.3%) in this cohort were of class I (BMI 29-34.99 kg/m²), 27.3% of class II (BMI 35-39.99 kg/m²) and 13.3% of class III (BMI ≥ 40 kg/m²) before pregnancy.
PPWR at six months after delivery ranges between – 17 and + 19 kg with a mean of -1.28 kg (SD 6.05; median -1 kg). Overall, 39% showed PPWR (> 0 kg) and 13% of obese mothers reported a high PPWR (≥ 5 kg). Mean GWG and psychological discomfort (i.e. levels of anxiety and depressed mood) were significantly higher in obese mothers with high and any PPWR compared to those with low and no PPWR. Mean duration of breastfeeding in this cohort of obese mothers was 9.5 weeks (SD 8.7); no significant difference was demonstrated for any duration of breastfeeding between those with PPWR compared to those with low or no PPWR. Those with high PPWR were more likely to sleep less (15.8% versus 3.8%; p = 0.09) and smoked less (5.3% versus 17.7%; p = 0.31) compared to those with low PPWR, although these differences were not statistically significant.
The final multivariate analysis showed that GWG (β = 0.337; p = 0.001) and maternal trait anxiety in the first trimester of pregnancy (β = 0.255; p = 0.001) were significant positive predictors for PPWR at six months after delivery. Pre-pregnancy BMI (β = -0.283; p = 0.001) was a significant negative predictor for PPWR (Table III). For each kg gained during pregnancy, there was risk of approximately 0.300 kg increase of PPWR at six months after delivery. If trait anxiety in obese pregnant women increased by one unit at the start of pregnancy, there was a 0.190 kg increase of PPWR six months after delivery. In the multivariate model, all the p-values were smaller than 0.005 (Table IV), indicating a chance of Type 1 error of less than 0.5%.
Table IV. Multivariate linear model of PPWR at 6 months after delivery in obese mothers.
| >PPWR | Unstandardized β | Standardized β | Std error | 95% CI for β | ||
|---|---|---|---|---|---|---|
| Model 1 | Intercept | -5.44 | 0.85 | -7.11 | -3.76 | |
| Total GWG | 0.38 | .42 | 0.07 | 0.24 | 0.51 | |
| Model 2 | Intercept | 5.07 | 3.81 | -2.46 | 12.60 | |
| Total GWG | 0.32 | .36 | 0.07 | 0.18 | 0.45 | |
| BMI | -0.29 | -0.22 | 0.10 | -0.49 | -0.09 | |
| Model 3 | Intercept | 1,59 | 3.80 | -5.93 | 9.10 | |
| Total GWG | 0.30 | .34 | 0.06 | 0.17 | 0.43 | |
| BMI | -0.37 | -.28 | 0.10 | -0.57 | -0.17 | |
| Trait anxiety, T1 | 0.19 | .25 | 0.05 | 0.08 | 0.30 | |
p ≤ 0.001
R² = 28.4%, adjusted R² = 26.9%
Discussion
Maternal obesity is now becoming one of the most common risk factors in pregnancy, leading to complications that impact on the health of both the woman and her offspring and are therefore also public health issues. Together with the related perinatal medical and psychological complications as mentioned in this thesis, this represents an added important economic burden. Indeed, it is estimated that the annual extra medical cost for obese adults represents about 3000 Euro (De Jonghe & Annemans, 2009). We therefore believe that the problem of maternal overweight has to be brought to the attention of policy makers as a matter of urgency. Older pregnant women, women with lower levels of education and a lower professional status, multiparous women and those ethnic minorities are at higher risk for being obese at the onset of pregnancy. These socio-demographic characteristics, which often occur simultaneously in obese women, are also correlated with higher levels of intrapartal interventions and poorer perinatal outcomes (Stein et al., 2009; Cammu et al., 2011; Schempf et al., 2007). The predictive value of the multivariate models for the variables related with pre-pregnancy BMI and GWG ranged between 3.4 and 10.5%, which was quite low. This means that other lifestyle factors such as food intake, physical activity, alcohol consumption and smoking behaviour will certainly be associated with unhealthy weight and weight gain. Although smoking is a much stronger risk factor for mortality than most other risk factors, obesity, as a mediator of the effect of a low socio-economic status on mortality, fills part of the gap in terms of health inequalities (i.e. mortality rates) between lower and higher socioeconomic positions if smoking would be eliminated (Mackenbach, 2011).
Although the onset of labour in humans is not yet fully understood, stimulatory triggers from the foetal and maternal HPA-axis, myometrial stretch, progesterone withdrawal, inflammation and local prostaglandines, as well as psychological factors, are involved (Mesiano & Welsh, 2007; Smith, 2007). From few observational cohort studies, controlling for induction of labour, we can conclude that obese pregnant women need more time to reach the active labour phase compared to non-obese women (Kominiarek et al., 2011; Verdiales et al., 2009). After 6 cm of cervical dilatation, progression is comparable in both obese and non-obese women.
Pregnancy is an important period in a woman’s lifecycle and is associated with specific physiological and psychosocial changes. Obese pregnant women appear to be rather insensitive to the ‘physiological’ increase in psychological comfort that we found in normal-weight pregnant women. Levels of anxiety are comparable in pregnant and non-pregnant women; but compared to normal weight pregnant women, obese pregnant women behave psychologically differently during the three trimesters of pregnancy. We must take into account the fact that psychological well being during pregnancy, traditionally often characterized by levels of anxiety and depressed mood, cannot be considered unidimensional. A comprehensive assessment of personal coping strategies to facilitate a healthy psychology should be involved (Jomeen, 2004).
Pregnancy is considered to be an ideal ‘teachable moment’ for changes towards a healthy lifestyle behaviour (Phelan, 2010). Lifestyle interventions in obese pregnant women have the potential to improve maternal and foetal outcomes by limiting GWG (Institute of Medicine, 2009). We learned from our prospective controlled cohort study on anxiety and depressed mood that obese pregnant women are more vulnerable compared to their normal weight counterparts. Because psychological factors were often neglected in earlier intervention studies, we performed a RCT aimed at reducing GWG as well as increasing psychological well-being (operationalized by levels of anxiety and depressed mood) in obese pregnant women. To produce significant behavioural changes, a combination of health education with psychological interventions are more effective than information alone (World Health Organisation, 2003). We showed that an intensive specialised follow-up during pregnancy, including motivational behaviour coaching, resulted in an improvement of the psychological well being in obese pregnant women. At the same time, a significant decrease in GWG (3-4 kg) was observed. A possible inter-dependence of behavioural change (weight reduction) and psychological factors (levels of anxiety) can be questioned. It seems that an increase of psychological comfort, or at least a stable degree, is important to achieve weight-related behavioural changes in obese pregnant women.
Maternal weight change during the postpartum period is a combination of retention of GWG and weight change caused by other lifestyle related factors. To study the effects of pregnancy related weight retention on perinatal outcomes, we chose to analyse maternal weight changes within a short inter-pregnancy mean time interval of one year. These data can give support to defining ‘excessive’ PPWR by pre-pregnancy BMI group, related to adverse perinatal outcomes in the next pregnancy. A more intensive follow up of the postpartum period is essential, and initial underweight and normal weight women also need to be monitored for weight management after delivery. The postpartum period in general, which can be the pre-conceptional period for the next pregnancy, is an understudied period. A longitudinal follow up in the postnatal period with adequate weight management could be useful. However, well-designed postpartum lifestyle intervention trials should be developed, because the literature is scarce on this topic and study results are inconsistent.
The increased psychosocial vulnerability of obese pregnant women and the weight related problems during pregnancy and the postpartum period provide arguments for an integration of psychological counselling during the perinatal follow up. This means that healthcare providers, involved in the care of obese pregnant women, should be trained to provide a correct and effective approach.
Conclusion
From this thesis, we can conclude that maternal obesity and excessive GWG are both important health care issues contributing to increased perinatal complications in the short, medium and long term. The epidemiological and psychological identification of the problem of maternal obesity and related socio-demographic and obstetrical correlates, taking into account various important confounding variables, provides evidence for a tailored weight management strategy for obese women before, during and after a pregnancy. In this doctoral thesis, we can identify socio-demographic, obstetrical and psychological characteristics of maternal obesity, we find evidence for beneficial outcomes of a lifestyle intervention programme in obese pregnant women, and we can find support for longer term perinatal complications with PPWR between the first and second pregnancy. Further development and implementation of preconception programmes based on a bio-psycho-social model that explicitly recognizes the individual needs and interacting lifestyle factors in obese women of reproductive age in order to prevent pre-pregnancy obesity, excessive GWG and PPWR is a challenge for the near future.
Promotor: Devlieger R., Department of Obstetrics & Gynaecology, Division Mother & Child, University Hospitals Leuven, Belgium.
Promotor: Van den Bergh B.R.H., Department of Psychology, Tilburg University, The Netherlands; Department of Welfare, Public Health and Family, Flemish Government, Brussels, Belgium.
Co-promotor: Witters I., Department of Obstetrics, East Limburg Hospital, Genk, Belgium; Center of Human Genetics, University Hospitals Leuven, Belgium.
References
- Andersson L, Sundstrom-Poromaa I, Wulff M, et al. Depression and anxiety during pregnancy and six months postpartum: a follow-up study. Acta Obstet Gynecol Scand. 2006;85:937–944. doi: 10.1080/00016340600697652. [DOI] [PubMed] [Google Scholar]
- Anubhuti and Arora S. Leptin and its metabolic interactions: an update. Diabetes Obes Metab. 2008;10:973–993. doi: 10.1111/j.1463-1326.2008.00852.x. [DOI] [PubMed] [Google Scholar]
- Austin MP, Tully L, Parker G. Examining the relationship between antenatal anxiety and postnatal depression. J Affect Disord. 2007;101:169–174. doi: 10.1016/j.jad.2006.11.015. [DOI] [PubMed] [Google Scholar]
- Beyer DA, Amari F, Ludders DW, et al. Obesity decreases the chance to deliver spontaneously. Arch Gynecol Obstet. 2011;28:981–988. doi: 10.1007/s00404-010-1502-5. [DOI] [PubMed] [Google Scholar]
- Bisits AM, Smith R, Mesiano S, et al. Inflammatory aetiology of human myometrial activation tested using directed graphs. PLoS Comput Biol. 2005;1:132–136. doi: 10.1371/journal.pcbi.0010019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bodnar LM, Wisner KL, Moses-Kolko E, et al. Prepregnancy body mass index, gestational weight gain, and the likelihood of major depressive disorder during pregnancy. J Clin Psychiatry. 2009;70:1290–1296. doi: 10.4088/JCP.08m04651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bogaerts AF, Devlieger R, Nuyts E et. Effects of lifestyle intervention in obese pregnant women on gestational weight gain and mental health: a randomized controlled trial. Int J Obes (Lond) 2012;37:814–821. doi: 10.1038/ijo.2012.162. [DOI] [PubMed] [Google Scholar]
- Breitkopf CR, Primeau LA, Levine RE. Anxiety symptoms during pregnancy and postpartum. J Psychosom Obstet Gynaecol. 2006;27:157–162. doi: 10.1080/01674820500523521. [DOI] [PubMed] [Google Scholar]
- Callaway LK, Prins JB, Chang AM, et al. The prevalence and impact of overweight and obesity in an Australian obstetric population. Med J Aust. 2006;184:56–59. doi: 10.5694/j.1326-5377.2006.tb00115.x. [DOI] [PubMed] [Google Scholar]
- Cammu H, Martens G, Keirse MJ. Mothers’ Level of Education and Childbirth Interventions: A Population-based Study in Flanders, Northern Belgium. Birth. 2011;38:191–199. doi: 10.1111/j.1523-536X.2011.00476.x. [DOI] [PubMed] [Google Scholar]
- Cedergren M. Effects of gestational weight gain and body mass index on obstetric outcome in Sweden. Int J Gynaecol Obstet. 2006;93:269. doi: 10.1016/j.ijgo.2006.03.002. [DOI] [PubMed] [Google Scholar]
- Cox JL, Holden JM, Sagovsky R. Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br J Psychiatry. 1987;150:782–786. doi: 10.1192/bjp.150.6.782. [DOI] [PubMed] [Google Scholar]
- De Jonghe E, Annemans L. De maatschappelijke kosten van obesitas in Vlaanderen. Scriptie 2de master arts. Faculteit Geneeskunde en Gezondheidswetenschappen; 2009. [Google Scholar]
- DiMarco ID, Klein DA, Clark VL. The use of motivational interviewing techniques to enhance the efficacy of guided self-help behavioral weight loss treatment. Eat Behav. 2009;10:134–136. doi: 10.1016/j.eatbeh.2009.02.001. [DOI] [PubMed] [Google Scholar]
- Cury A, Rossi MP. Prevalence of anxiety and depression during pregnancy in a private setting sample. Arch Womens Ment Health. 2007;10:25–32. doi: 10.1007/s00737-006-0164-6. [DOI] [PubMed] [Google Scholar]
- Fattah C, Farah N, Barry S, et al. The measurement of maternal adiposity. J Obstet Gynaecol. 2009;29:686–689. doi: 10.3109/01443610903184025. [DOI] [PubMed] [Google Scholar]
- Gariepy G, Nitka D, Schmitz N. The association between obesity and anxiety disorders in the population: a systematic review and meta-analysis. Int J Obes (Lond) 2010;34:407–419. doi: 10.1038/ijo.2009.252. [DOI] [PubMed] [Google Scholar]
- Grammatopoulos DK. Placental corticotrophin-releasing hormone and its receptors in human pregnancy and labour: still a scientific enigma. J Neuroendocrinol. 2008;20:432–438. doi: 10.1111/j.1365-2826.2008.01660.x. [DOI] [PubMed] [Google Scholar]
- Guelinckx I, Devlieger R, Beckers K, et al. Maternal obesity: pregnancy complications, gestational weight gain and nutrition. Obes Rev. 2008;9:140–150. doi: 10.1111/j.1467-789X.2007.00464.x. [DOI] [PubMed] [Google Scholar]
- Hendler I, Goldenberg RL, Mercer BM, et al. The Preterm Prediction Study: association between maternal body mass index and spontaneous and indicated preterm birth. Am J Obstet Gynecol. 2005;192:882–886. doi: 10.1016/j.ajog.2004.09.021. [DOI] [PubMed] [Google Scholar]
- Heron J, O'Connor TG, Evans J, et al. The course of anxiety and depression through pregnancy and the postpartum in a community sample. J Affect Disord. 2004;80:65–73. doi: 10.1016/j.jad.2003.08.004. [DOI] [PubMed] [Google Scholar]
- Heslehurst N, Simpson H, Ells LJ, et al. The impact of maternal BMI status on pregnancy outcomes with immediate short-term obstetric resource implications: a meta-analysis. Obes Rev. 2008;9:635–683. doi: 10.1111/j.1467-789X.2008.00511.x. [DOI] [PubMed] [Google Scholar]
- Institute of Medicine. Weight Gain during Pregnancy: Re-examining the Guidelines. Washington (DC): National Academy Press; 2009. [Google Scholar]
- Johnson SS, Paiva AL, Cummins CO, et al. Transtheoretical model-based multiple behavior intervention for weight management: effectiveness on a population basis. Prev Med. 2008;46:238–246. doi: 10.1016/j.ypmed.2007.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jomeen J. The importance of assessing psychological status during pregnancy, childbirth and the postnatal period as a multidimensional construct: A literature review. Clinical Effectiveness in Nursing. 2004;8:143–155. [Google Scholar]
- Kominiarek MA, Zhang J, Vanveldhuisen P, et al. Contemporary labor patterns: the impact of maternal body mass index. Am J Obstet Gynecol. 2011;205:244–248. doi: 10.1016/j.ajog.2011.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laraia BA, Siega-Riz AM, Dole N, et al. Pregravid weight is associated with prior dietary restraint and psychosocial factors during pregnancy. Obesity (Silver.Spring) 2009;17:550–558. doi: 10.1038/oby.2008.585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W, Challis JR. Corticotropin-releasing hormone and urocortin induce secretion of matrix metalloproteinase-9 (MMP-9) without change in tissue inhibitors of MMP-1 by cultured cells from human placenta and fetal membranes. J Clin Endocrinol Metab. 2005;90:6569–6574. doi: 10.1210/jc.2005-1445. [DOI] [PubMed] [Google Scholar]
- Linne Y, Dye L, Barkeling B, et al. Long-term weight development in women: a 15-year follow-up of the effects of pregnancy. Obes Res. 12:1166–1178. doi: 10.1038/oby.2004.146. [DOI] [PubMed] [Google Scholar]
- Mackenbach JP. What would happen to health inequalities if smoking were eliminated? BMJ. 2011;342:d3460. doi: 10.1136/bmj.d3460. [DOI] [PubMed] [Google Scholar]
- Madan J, Chen M, Goodman E, et al. Maternal obesity, gestational hypertension, and preterm delivery. J Matern Fetal Neonatal Med. 2010;23:82–88. doi: 10.3109/14767050903258738. [DOI] [PubMed] [Google Scholar]
- Madan JC, Davis JM, Craig WY WY, et al. Maternal obesity and markers of inflammation in pregnancy. Cytokine. 2009;47:61–64. doi: 10.1016/j.cyto.2009.05.004. [DOI] [PubMed] [Google Scholar]
- Mamun AA, Callaway LK, Callaghan MJ, et al. Associations of maternal pre-pregnancy obesity and excess pregnancy weight gains with adverse pregnancy outcomes and length of hospital stay. BMC Pregnancy Childbirth. 2011;11:62. doi: 10.1186/1471-2393-11-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin WL, Hutchon SP. Mechanism and management of normal labour. Curr Obstet Gynaecol. 2004;14:301–308. [Google Scholar]
- Mesiano S. Myometrial progesterone responsiveness and the control of human parturition. J Soc Gynecol Investig. 2004;11:193–202. doi: 10.1016/j.jsgi.2003.12.004. [DOI] [PubMed] [Google Scholar]
- Mesiano S, Welsh TN. Steroid hormone control of myometrial contractility and parturition. Semin Cell Dev Biol. 2007;18:321–331. doi: 10.1016/j.semcdb.2007.05.003. [DOI] [PubMed] [Google Scholar]
- Nelson SM, Matthews P, Poston L. Maternal metabolism and obesity: modifiable determinants of pregnancy outcome. Hum Reprod Update. 2010;16:255–275. doi: 10.1093/humupd/dmp050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nohr EA, Bech BH, Vaeth M, et al. Obesity, gestational weight gain and preterm birth: a study within the Danish National Birth Cohort. Paediatr Perinat Epidemiol. 2007;21:5–14. doi: 10.1111/j.1365-3016.2007.00762.x. [DOI] [PubMed] [Google Scholar]
- Nohr EA, Vaeth M, Baker JL, et al. Combined associations of prepregnancy body mass index and gestational weight gain with the outcome of pregnancy. Am J Clin Nutr. 2008;87:1750–1759. doi: 10.1093/ajcn/87.6.1750. [DOI] [PubMed] [Google Scholar]
- Norwitz ER, Robinson JN, Challis JR. The control of labor. N Engl J Med. 1999;341:660–666. doi: 10.1056/NEJM199908263410906. [DOI] [PubMed] [Google Scholar]
- Reilly JR, Reynolds RM. The risk of maternal obesity to the long-term health of the offspring. Clin Endocrinol (Oxf) 2013;78:9–16. doi: 10.1111/cen.12055. [DOI] [PubMed] [Google Scholar]
- Pedersen P, Baker JL, Henriksen TB, et al. Influence of Psychosocial Factors on Postpartum Weight Retention. Obesity. 2011;19:639–646. doi: 10.1038/oby.2010.175. [DOI] [PubMed] [Google Scholar]
- Phelan S. Pregnancy: a “teachable moment” for weight control and obesity prevention. Am J Obstet Gynecol. 2010;202:135–138. doi: 10.1016/j.ajog.2009.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polit DE, Beck CT. Nursing Research, Generating and Assessing Evidence for Nursing Practice. Philadelphia: 2008. [Google Scholar]
- Poobalan AS, Aucott LS, Gurung T, et al. Obesity as an independent risk factor for elective and emergency caesarean delivery in nulliparous women – systematic review and meta-analysis of cohort studies. Obes Rev. 2009;10:28–35. doi: 10.1111/j.1467-789X.2008.00537.x. [DOI] [PubMed] [Google Scholar]
- Pop VJ, Komproe IH, van Son MJ. Characteristics of the Edinburgh Post Natal Depression Scale in The Netherlands. J Affect Disord. 1992;26:105–110. doi: 10.1016/0165-0327(92)90041-4. [DOI] [PubMed] [Google Scholar]
- Edwards JW, Kleinman K, Abrams A, et al. Sociodemographic predictors of antenatal and postpartum depressive symptoms among women in a medical group practice. J Epidemiol Community Health. 2006;60:221–227. doi: 10.1136/jech.2005.039370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rooney BL, Schauberger CW, Mathiason MA. Impact of perinatal weight change on long-term obesity and obesity-related illnesses. Obstet Gynecol. 2005;106:1349–1356. doi: 10.1097/01.AOG.0000185480.09068.4a. [DOI] [PubMed] [Google Scholar]
- Sarwer DB, Allison KC, Gibbons LM, et al. Pregnancy and obesity: a review and agenda for future research. J Womens Health (Larchmt.) 2006;15:720–733. doi: 10.1089/jwh.2006.15.720. [DOI] [PubMed] [Google Scholar]
- Schempf AH, Branum AM, Lukacs SL, et al. Maternal age and parity-associated risks of preterm birth: differences by race/ethnicity. Paediatr Perinat Epidemiol. 2007;21:34–43. doi: 10.1111/j.1365-3016.2007.00785.x. [DOI] [PubMed] [Google Scholar]
- Schrauwers C, Dekker G. Maternal and perinatal outcome in obese pregnant patients. J Matern Fetal Neonatal Med. 2009;22:218–226. doi: 10.1080/14767050902801652. [DOI] [PubMed] [Google Scholar]
- Smith R. Parturition. N Engl J Med. 2007;356:271–283. doi: 10.1056/NEJMra061360. [DOI] [PubMed] [Google Scholar]
- Spielberger CD, Gorsuch RL, Lushene RE. STAI manual for the State-Trait Anxiety Inventory. California: Consulting Psychologists Press, Palo Alto; 1970. [Google Scholar]
- Stein CR, Savitz DA, Janevic T, et al. Maternal ethnic ancestry and adverse perinatal outcomes in New York City. Am J Obstet Gynecol. 2009;201:584–589. doi: 10.1016/j.ajog.2009.06.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Heyden J, Gisle L, Demarest S, et al. Gezondheidsenquête België, 2008. Rapport I - Gezondheidstoestand. Operationele Directie Volksgezondheid en surveillance. Brussel: Wetenschappelijk Instituut Volksgezondheid; 2010. [Google Scholar]
- Verdiales M, Pacheco C, Cohen WR. The effect of maternal obesity on the course of labor. J Perinat Med. 2009;37:651–655. doi: 10.1515/JPM.2009.110. [DOI] [PubMed] [Google Scholar]
- Weiss JL, Malone FD, Emig D, et al. Obesity, obstetric complications and cesarean delivery rate – a population-based screening study. Am J Obstet Gynecol. 2004;190:1091–1097. doi: 10.1016/j.ajog.2003.09.058. [DOI] [PubMed] [Google Scholar]
- WHO. Obesity and Overweight. 2012. [Google Scholar]
- World Health Organisation. Adherence to Long-Term Therapies: Evidence for Action. Geneva: WHO; 2003. [Google Scholar]
- Yogev Y, Catalano PM. Pregnancy and obesity. Obstet Gynecol Clin North Am. 2009;36:285–300. doi: 10.1016/j.ogc.2009.03.003. [DOI] [PubMed] [Google Scholar]
- Zender R, Olshansky E. Women’s mental health: depression and anxiety. Nurs Clin North Am. 2009;44:355–364. doi: 10.1016/j.cnur.2009.06.002. [DOI] [PubMed] [Google Scholar]
- Zhao G, Ford ES, Dhingra S, et al. Depression and anxiety among US adults: associations with body mass index. Int J Obes (Lond) 2009;33:257–266. doi: 10.1038/ijo.2008.268. [DOI] [PubMed] [Google Scholar]