Abstract
Calcifying epithelial odontogenic tumors (CEOTs) and ghost cell odontogenic tumors (GCOTs) are characteristic odontogenic origin epithelial tumors which produce calcifying materials from transformed epithelial tumor cells. CEOT is a benign odontogenic tumor composed of polygonal epithelial tumor cells that show retrogressive calcific changes, amyloid-like deposition, and clear cytoplasm. Differentially, GCOTs are a group of transient tumors characterized by ghost cell presence, which comprise calcifying cystic odontogenic tumor (CCOT), dentinogenic ghost cell tumor (DGCT), and ghost cell odontogenic carcinoma (GCOC), all derived from calcifying odontogenic cysts (COCs). There is considerable confusion about COCs and GCOTs terminology, but these lesions can be classified as COCs or GCOTs, based on their cystic or tumorous natures, respectively. GCOTs include ameloblastomatous tumors derived from dominant odontogenic cysts classified as CCOTs, ghost cell-rich tumors producing dentinoid materials as DGCTs, and the GCOT malignant counterpart, GCOCs. Many authors have reported CEOTs and GCOTs variably express keratins, β-catenin, BCL-2, BSP, RANKL, OPG, Notch1, Jagged1, TGF-β, SMADs, and other proteins. However, these heterogeneous lesions should be differentially diagnosed to allow for accurate tumor progression and prognosis prediction.
Keywords: Calcifying, Dentinoid, Ghost cell odontogenic tumors
Odontogenic tissue is programmed to produce dentin and enamel due to active interactions between odontogenic mesenchyme and epithelium. Tooth formation is achieved via odontogenic mesenchyme and epithelium stage- and spatial-specific differentiation from early tooth development to late maturation.1,2 Therefore, when odontogenic tissue becomes undifferentiated and undergoes tumorous change, it has the potential to produce abnormal calcifications with enameloid, dentinoid, and cementum-like material histologic features. For this reason, these odontogenic calcifications are important odontogenic tumor characteristics and occasionally are accompanied by odontogenic epithelium ghost cell change and amorphous odontogenic mesenchyme hyalinization.3
Ghost cells are anucleate epithelial cells with swollen homogeneous pale eosinophilic cytoplasm and pale to clear central areas occupied by the previous nucleus. In addition, they are highly keratinized with degenerated nuclei shadow images. Ghost cells are frequently found in calcifying odontogenic cyst (COC) derivatives, including craniopharyngioma, pilomatricoma, etc., in association with calcified foci.4
Calcifying epithelial odontogenic tumors (CEOTs) and COCs produce calcifying materials by transforming odontogenic epithelial cells. In particular, COC tumorous growth can lead to ghost cell odontogenic tumor (GCOT) development, comprising the calcifying cystic odontogenic tumor (CCOT), dentinogenic ghost cell tumor (DGCT), and ghost cell odontogenic carcinoma (GCOC), in which many ghost cells emerge. However, for prognostic reasons, epithelial odontogenic tumors producing calcifications and/or ghost cells should be differentially diagnosed based on tumor cellular differentiation.
CALCIFYING EPITHELIAL ODONTOGENIC TUMOR
General features
CEOT is an uncommon, benign odontogenic lesion that accounts for less than 1% of all odontogenic tumors. Dr. Pindborg (1958)5 first described this tumor, and thereafter, CEOT was termed Pindborg tumor by Shafer et al.6,7 CEOT mainly presents discrete polyhedral eosinophilic epithelial cell islands, strands, or sheets in a fibrous stroma (Fig. 1A). Its tumor cells produce eosinophilic deposits resembling amyloid, which react positively to Congo red and calcify gradually. The epithelial cellular outlines are distinct, and intercellular bridges may be seen. Nuclei show considerable variation, and giant nuclei may be seen. Some tumors show considerable nuclear pleomorphism, but this feature is not considered to indicate malignancy (Fig. 1C).8,9
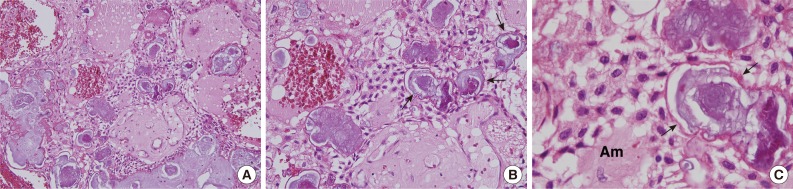
Fig. 1.
Calcifying epithelial odontogenic tumor photomicrographs. (A-C) Polyhedral epithelial cell sheets, resembling enamel epithelium stratum intermedium, are proliferative and produce concentric calcifications, called Liesegang rings (arrows), and amyloid-like materials (Am).
CEOT is usually encountered in the posterior mandible, and is most common in patients between 30 and 50 years of age, with no gender predominance. It usually presents as a slow-growing, painless swelling, and is asymptomatic, but locally invasive.10 CEOT manifests as an intraosseous lesion (central type) in the majority of cases (95%). Extraosseous or peripheral lesions account for fewer than 5% of cases.8,11,12 It can be associated with an impacted tooth and radiographically simulate a dentigerous cyst. Most CEOTs are solid masses, and rarely show cyst spaces. They appear either as monocystic or polycystic radiolucent lesions with radiopaque spots or masses, and also exhibit peripheral destruction upon imaging. Calcifications are usually scattered within tumors,13 and are a distinctive feature. They develop within amyloid-like materials and form concentric rings (Liesegang ring calcifications) (Fig. 1B, C), which tend to fuse to form large, complex masses. These amyloid-like materials stain positively with Congo red or thioflavine T. Furthermore, after Congo red staining, amyloid exhibits an apple-green birefringence in polarized light.8,11
Types and variants
Several CEOT variants may appear with clear cell focal areas, cementum-like components, abundant Langerhans cells, combined epithelial odontogenic tumor (adenomatoid odontogenic tumor), and abundant myoepithelial cells.12,14
The extraosseous CEOT clear cell variant is extremely rare, and shows polyhedral cells, hyaline materials, and calcified areas. The hyaline materials are usually positive for Congo red, crystal violet, and Lugol's iodine, but negative for Coomassie blue. The clear polyhedral cells often exhibit stained granules after periodic acid-Schiff staining. Therefore, these hyaline materials are considered amyloid-like. Furthermore, clear cell presence in extraosseous CEOT imply aggressive tumor growth in vascular connective tissue.15
The intraosseous CEOT noncalcifying variant is also very rare, and commonly appears in association with Langerhans cells. The noncalcifying, Langerhans cell-rich variant has only been reported in four cases to date,16 and most were located in the anterior and premolar maxillary regions, in contrast to classical CEOTs, which are usually located in the mandible molar and ascending ramus areas. Furthermore, the intraosseous CEOT noncalcifying variant is believed to be aggressive.17
A few peripheral (extraosseous) CEOT cases have been reported, and manifest as nonspecific, sessile gingival masses, usually on the anterior gingiva. In some cases, this lesion is associated with underlying bone cupped-out erosion.18
The adenomatoid odontogenic tumor and CEOT combination is rare; 22 cases have been published.19 Because many adenomatoid odontogenic tumors contain variable sized CEOT-like areas, CEOT-like change may be considered a normal feature in the adenomatoid odontogenic tumor histomorphological spectrum. The combination variant is benign, and represents odontogenic epithelium dedifferentiation.19
The multifocal CEOT variant shows multiple CEOT lesions in intraosseous and extraosseous jaw regions, including mandible, maxilla, and gingiva.20 Each multifocal tumor mass must be carefully evaluated for clinical and histologic evidence of neoplasia, because they may exhibit different tumorigenic propensities.
Pathogenesis
CEOT epithelial tumor cells bear close morphologic resemblance to enamel organ stratum intermedium cells (Fig. 1C). However, based on its anatomic distribution in jaw, some investigators have recently suggested that the tumor arises from dental lamina remnants.8
The CEOT polyhedral epithelial cells commonly express laminins 1 and 5, fibronectin, cytokeratins, and vimentin, whereas its amyloid-like materials do not.21 Tumor epithelial cells are slightly positive or negative for PKK1 (specific for the 44, 46, 52, and 53 kD keratins) detectable keratins, but slightly to strongly positive for KL1 (specific for the 55-57 kD keratins) and TK (41-65 kD keratins). Tumor epithelium is slightly positive for vimentin but negative for desmin.22 Notably, CEOT epithelial tumor cells exhibit alkaline phosphatase and ATPase localization in their membranes, indicating active membrane transport.23 These high alkaline phosphatase and ATPase activities may be relevant to abnormal eosinophilic materials (amyloid-like) and irregular calcification production.24
At the electron microscope level, CEOT epithelial tumor cells consist of polyhedral epithelial cells and myoepithelial-like cells, containing large numbers of electron-dense tonofilament bundles, electron-dense bodies, and fine lamina densa filaments.25 Amyloid-like materials compose fine filament sheets measuring 10-12 nm in diameter and lamina densa fragment aggregates, which are probably secreted by polyhedral epithelial and myoepithelial-like cells. The fine filamentous materials are a form of amyloid, and their formation results from lamina densa material degradation.26 Recently, the odontogenic ameloblast-associated protein (ODAM) fibril-forming region was found in CEOT amyloid-like materials, thus suggesting ODAM might have amyloidogenic potential.27
A number of dendritic cells, which are frequently found among CEOT epithelial sheets, are strongly positive for S-100 and CD-1a antisera. Ultrastructurally, these dendritic cells show indented nuclei and Birbeck's granules similar to Langerhans cells. Thus, they are likely to be Langerhans cells, and play a role in antigen presentation from epithelial tumor cell abortive products.21,28
The fact that an ameloblastin mutation was found exclusively in CEOT suggests that alterations to this gene may be relevant to CEOT pathogenesis.29 However, both ameloblastin transgenic and ameloblastin-null mice showed amelogenesis imperfecta features. In particular, ameloblastin-null mice developed unidentified type odontogenic tumors.30 More mutational analyses are required to elucidate the mutation nature in CEOT odontogenic genes.
Prognosis
CEOTs are generally considered benign, but can be locally aggressive, and exhibit 10% to 15% recurrence rates.31 Maxillary CEOTs tend to be more aggressive and spread more rapidly to possibly involve surrounding vital structures than mandibular CEOTs.32 Therefore, maxillary CEOT should be treated more aggressively, and five years is considered the absolute minimum follow-up period.12
Although it was originally believed that CEOT is similar to ameloblastoma biologic behavior, growing evidence indicates it tends to be less aggressive.8,12 Thus, due to CEOT's relatively indolent biological behavior, mutilating procedures, such as wide resection or mandible hemisection, seem unwarranted. Accordingly, enucleation within macroscopically normal tissue is recommended for CEOT involving the mandible.8 However, repeated local recurrence may imply malignant transformation and tumor metastasis with vascular invasion and spread to a cervical lymph node.33 Contrary to primary CEOT benign histological features, malignant CEOT shows nuclear pleomorphism with frequent mitotic figures and increased proliferative activity, as assessed by immunostaining for Ki-67.34 Therefore, recurrent CEOT carries the threat of malignant transformation, and may justify radical surgery, and adjuvant radiotherapy.35
CALCIFYING ODONTOGENIC CYST
COC was first described by Gorlin et al. in 1962,36 and since then several hundred cases have been reported.37 COC exhibits considerable histopathologic diversity and variable clinical behavior. Cyst epithelium is usually thin and tends to be detached easily, but is focally thickened by keratinized epithelial cells and ghost cells. Hyperplastic basal cells often grow into fibrous cyst walls, resulting in daughter cysts. Unlike other odontogenic cysts, COCs may contain highly-differentiated lining epithelium composed of columnar cells or stellate reticulum-like cells associated with ghost cells, and undifferentiated lining epithelium resembling reduced enamel epithelium (Fig. 2A).38,39
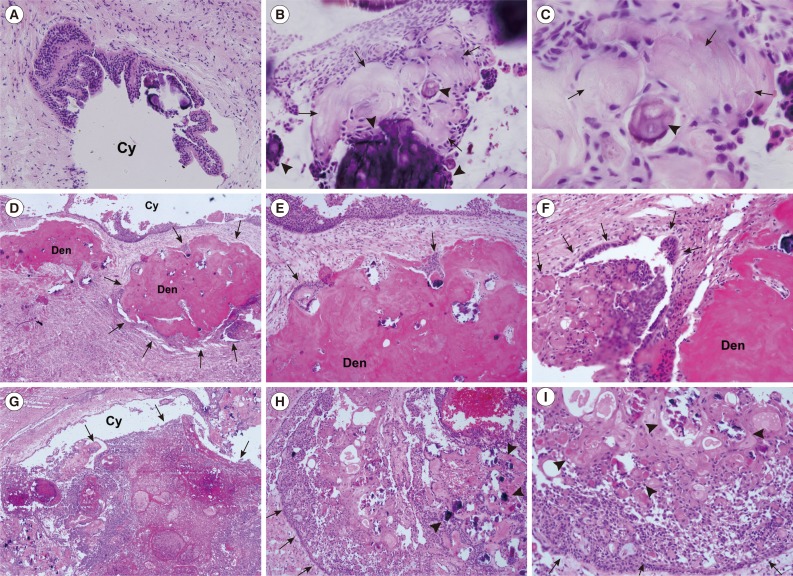
Fig. 2.
Calcifying odontogenic cyst (COC) and calcifying cystic odontogenic tumor (CCOT) photomicrographs. (A-C) COC, cystic epithelium is keratinized and produces irregular calcifications (arrowheads) and aberrantly keratinized ghost cells (arrows). (D-I) CCOT. (D-F) Odontoma-associated CCOT, proliferating tumor mass (arrows) containing dysplastic dentinoid materials (Den). (G-I) Ameloblastomatous proliferating CCOT, infiltratively proliferating tumor cells (arrows), accompanying multiple ghost cell calcifications (arrowheads). Cy, cyst space (Fig. 2D-I; Courtesy of Professor Kyung-Ja Cho, Department of Pathology, University of Ulsan College of Medicine, Korea).
Ghost cell masses frequently fuse to form large amorphous, acellular sheets. Furthermore, ghost cells are commonly associated with dystrophic calcification (Fig. 2B, C), and rarely with aberrant ossification.40 Confocal laser scanning microscopy observations have disclosed that COC ghost cells autofluoresce. However, autofluorescence intensities are variable, possibly due to hard keratin presence.4
Immunohistochemically, amelogenin protein is expressed chiefly in ghost cells,41 whereas cytokeratin 19 and B-cell leukemia/lymphoma-2 (BCL-2) are expressed chiefly in odontogenic cyst lining epithelial cells. Lining epithelial cells are sporadically positive for Ki-67 antigen, especially in proliferative and ameloblastomatous epithelium. These findings suggest that ghost cells or lining epithelial cells show ameloblastic cytodifferentiation or odontogenic epithelial characteristics and consistently proliferate and increasingly survive under cystic conditions.42
COC is predominantly an intraosseous lesion, although in 13% to 30% of reported cases, COC has manifested as a peripheral lesion. About 65% of cases are found in the incisor or canine areas. Mean age is 33 years, and most cases are diagnosed in the second and third decades of life. However, COCs that are associated with odontomas tend to occur in younger patients (mean age, 17 years).43,44
Central COC is usually a unilocular, well-defined radiolucency, although the lesion may occasionally appear multilocular. Radiopaque structures within lesions, either irregular calcifications or tooth-like densities, are present in about one third to one half of cases. The radiolucent lesion is associated with an unerupted tooth, usually a canine, in approximately one third of cases.45,46
Extraosseous COCs are usually localized sessile or pedunculated gingival masses with no distinctive clinical features, and can resemble common gingival fibromas, gingival cysts, or peripheral giant cell granulomas. They appear as variably sized odontogenic epithelial islands in a fibrous stroma composed of peripheral palisading columnar cells and central stellate reticulum, reminiscent of ameloblastoma. However, ghost cell nests are present within epithelium, and juxtaepithelial dentinoid is commonly present. These features differentiate this lesion from peripheral ameloblastoma.46
COC prognosis is relatively good, and few recurrences have been reported after simple enucleation. Peripheral neoplastic COC appears to have the same prognosis as a peripheral ameloblastoma with minimal recurrence chance after simple surgical excision. Accordingly, when a COC is associated with some other recognized odontogenic tumor, such as, ameloblastoma, treatment and prognosis are likely to be the same as that of the associated tumor.47
The term COC is consistent with a unicystic lesion with or without associated odontoma, while other related lesions identified as benign or malignant tumors must be classified separately.44 Although the COC term represents a cyst, some investigators prefer to classify it as a neoplasm.8 The cyst lining unifocal or multifocal epithelial proliferation into the lumen may resemble ameloblastoma, but the COC is intermixed with varying ghost cell numbers. About 20% of COCs are associated with odontoma, but the neoplastic solid COCs are relatively uncommon, and account for 2% to 16% of all COCs in reported series.48 The 2005 World Health Organization (WHO) Classification of Odontogenic Tumors divided COC neoplastic lesions into GCOTs comprised of CCOT, DGCT, and GCOC.49
COC may have more heterogeneous components than envisaged, and thus, careful pathological observation and long-time follow-up are required. Given the uneventful outcomes for 35 cases filed in our Gangneung-Wonju National University Dental Hospital over 18 years, we consider the diagnostic term, COC, is still useful. However, some authors have described COC as CCOT, or calcifying ghost cell odontogenic cyst. Therefore, these heterogeneous lesions can be classified as COCs when benign cystic lesions, and as GCOTs when tumorous.
GHOST CELL ODONTOGENIC TUMORS
According to the WHO International Histological Classification of Odontogenic Tumors guideline (2005) GCOTs comprise CCOT, DGCT, and GCOC.50 In this guideline, CCOT is defined as a benign cystic neoplasm of odontogenic origin, characterized by ameloblastoma-like epithelium with ghost cells that may be calcified,51 and DGCT as a locally invasive neoplasm characterized by epithelial cell ameloblastoma-like islands in mature connective tissue stroma. Aberrant ghost cell keratinization may be found in association with varying dysplastic dentin (so called dentinoid materials) amounts.52 Furthermore, previously reported odontogenic ghost cell carcinoma and malignant epithelial odontogenic ghost cell tumor cases were re-named by WHO as GCOC (Table 1).53,54
Table 1.
Pathological findings and COC diagnoses and its derivative tumors (GCOTs)
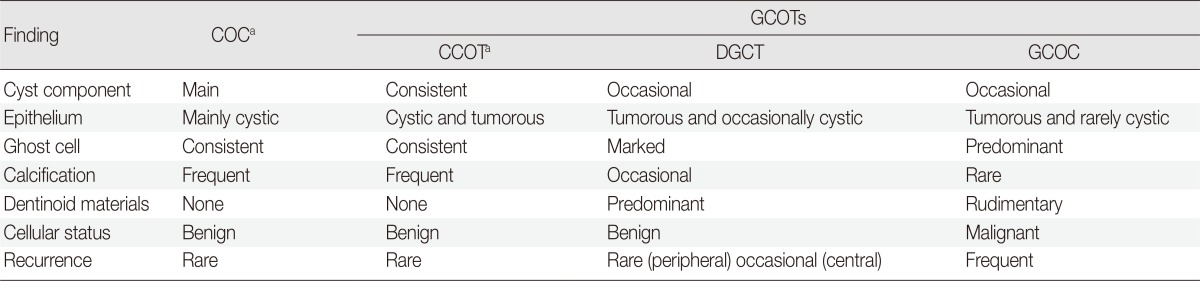
COC, calcifying odontogenic cyst; GCOT, ghost cell odontogenic tumor; CCOT, calcifying cystic odontogenic tumor; DGCT, dentinogenic ghost cell tumor; GCOC, ghost cell odontogenic carcinoma.
aWorld Health Organization (WHO) classification suggests COC and CCOT are similar lesions.50
GCOTs usually occur within maxillary and mandibular bones (central GCOTs), and in gingival soft tissues (peripheral GCOTs). Central GCOTs can be associated with odontoma arising from an impacted tooth, but are gradually replaced by solid epithelial odontogenic tumor tissue. On the other hand, peripheral GCOTs may present on gingiva as a painless nodule that clinically appears as a reactive hyperplastic mass.55 Peripheral GCOTs are usually treated by simple excision and show good prognosis with few recurrences, but central GCOTs should be treated carefully depending on neoplastic epithelium infiltrative growth.56
A clear cell GCOT variant was reported without further attempt at classification.57 It showed glycogen sheets and islands containing clear epithelial cells separated by a thin fibrous connective tissue stroma. Furthermore, both ameloblastic and clear cells were immunopositive for cytokeratin 19 and AE1/3.58 Although it is unclear whether this tumor represented a pre-existing GCOT clear cell change or a separate and distinct neoplasm derived de novo from odontogenic epithelium, the clear cell components were the most prominent clear cell GCOT distinguishing feature.44
Previous reports indicate GCOTs have wide neoplastic potential.59 CCOT is a cystic, painless, slowly growing tumor that commonly presents as a well-defined radiolucent or combined lesion that rarely recurs, whereas DGCT is aggressive and recurrences may be expected, and GCOC is in actuality a malignant neoplasm.50 Furthermore, the term GCOT is useful for describing a solid neoplastic COC variant characterized by ameloblastomatous epithelial components accompanied by abundant ghost cell clusters and dentinoid materials (Table 1).
CALCIFYING CYSTIC ODONTOGENIC TUMOR
General features
CCOT is a benign odontogenic origin cystic neoplasm characterized by an ameloblastoma-like epithelium and ghost cells. Its cystic epithelial lining shows a well-defined columnar cell basal layer and an overlying layer often composed of many epithelial cells, which may resemble the enamel organ stellate reticulum. CCOT usually contains ghost cell masses that may be located within the epithelial lining or in the fibrous capsule. The CCOT neoplastic epithelium is closely associated with COC, and exhibits ghost cells that may undergo calcification (Fig. 2F).60,61
CCOT usually presents as a painless, slow-growing mass involving both maxilla and mandible, primarily in the anterior area (incisors and canines). It generally affects young adults in the third to fourth decades and has no gender predominance.62 CCOT may occur in an intraosseous or extraosseous area. Peripheral CCOT accounts for about 26% of all reported cases.63
CCOT typical microscopic features include ameloblastomatous epithelium containing ghost cells clusters and the simple COC lesion (Fig. 2G-I). The cystic lesion may sometimes dominate and be associated with a hard dental tissue area resembling odontoma. However, a limited dysplastic dentin amount can usually be found (Fig. 2D-F).61
CCOT calcification appears as a thin radiopaque line, and discrete radiopaque foci, which contrast with those in adenomatoid odontogenic tumors, which exhibit numerous dispersed or clustered radiopaque foci.64 These characteristic radiopacities may facilitate differential diagnosis by revealing internal calcifications in cone-beam computed tomography images.65 CCOT is also associated with benign odontogenic tumors other than odontoma, such as, ameloblastic fibroma, ameloblastic fibro-odontoma, adenomatoid odontogenic tumor, or odontoameloblastoma.66
Types and variants
Although CCOT is designated histologically as a compound lesion composed of COC and tumorous odontogenic epithelium, it can be classified into four sub-types using the following predominant findings, 1) simple cystic (Fig. 2A-C), 2) odontoma associated (Fig. 2D-F), 3) ameloblastomatous proliferating (Fig. 2G-I), or 4) CCOT associated with a benign odontogenic tumor other than odontoma.54,66
The simple CCOT cystic type resembles ordinary COC, but with polygonal odontogenic epithelium and ghost cell tumorous growth. Furthermore, this cystic lesion may have ghost cells without odontoma features.54
Odontoma-associated CCOT contains discrete, abnormal tooth structures originating from odontogenic mesenchyme. Dentin, enamel, cementum, and pulpal tissues are present as a complex or compound odontoma. Furthermore, CCOT enamel epithelium is relatively well differentiated, which implies this CCOT type has a good prognosis.54
Ameloblastomatous proliferating CCOT shows predominant ameloblastoma-like epithelial proliferation features, producing anastomosing epithelial strands with palisading basal cells (Fig. 2G-I). Its epithelium is less infiltrative than ameloblastoma, and is usually localized at subepithelial connective tissue with sparse odontogenic myxoid mesenchyme. Ameloblastomatous proliferating CCOT is characterized by intraluminal or capsular plexiform growths resembling those seen in the cystic ameloblastoma plexiform variant. Sometimes follicles simulating solid ameloblastoma are observed in the connective tissue capsule.54
The existence of benign odontogenic tumor-associated CCOT implies CCOT's heterogeneous nature. Hybrid odontogenic tumors can occur. To date, five CCOT adenomatoid odontogenic tumor cases,67 and one CCOT plexiform ameloblastoma case have been reported.68
Pathogenesis
CCOT tumor epithelium shows positive reactions for keratin-14 and keratin-10/13 in its basal and upper cell layers, respectively, which indicate its epithelium differentiates towards the squamous type.69 Of its epithelial basement membrane components (laminins 1 and 5, collagen type IV, and fibronectin) laminin 5 is found faintly in suprabasal cells, but expressed strongly in ghost cells. Therefore, it is presumed that laminin 5 is involved in ghost cell formation and in calcification initiation.70
CCOT calcified masses demonstrate positive immunoreactivity to human bone sialoprotein (BSP) antibodies, and hybridization signals for BSP are located in cells near calcified particles. In COC, BSP signals are also seen in cells surrounding the characteristic ghost cell nests, which often subsequently calcify. BSP may play an important role in ghost tumor formation and differentiation involving pathological calcification.71
Ghost cells demonstrate Notch1 and Jagged1 overexpression, suggesting Notch1-Jagged1 signaling subserves the main transduction mechanism responsible for ghost cell fate decision in CCOT. Protein localizations are largely membranous and/or cytoplasmic. In particular, mineralized ghost cells are strongly positive, which suggests ghost cell calcification processes might be associated with Notch1 and Jagged1 upregulation.72 However, both polygonal epithelial and stromal cells show positive immunoreactivity for nuclear factor kappa-B (NF-κB; RANK), RANK ligand (RANKL), and osteoprotegerin receptor activation.73
The transforming growth factor-β/SMAD signaling pathway is less activated in CCOT than in ameloblastoma, indicating CCOT may show less cellular proliferation and less advanced cellular differentiation than ameloblastoma.74 Furthermore, CCOTs express fewer matrix metalloproteases (MMPs), tissue inhibitors of metalloproteinases (TIMP), growth factors, epidermal growth factor receptor, and extracellular signal-regulated kinase than odontogenic keratocysts (keratocystic odontogenic tumors). Therefore, it is believed that CCOT is less aggressive than odontogenic keratocysts.75 In addition, CCOT has been reported to show lower MMPs, TIMPs, and growth factor expression than ameloblastoma.76
NF-κB, Ki-67, and MMP-9 are weakly stained in CCOT, but nevertheless, may consistently affect its progression and local invasiveness.77 Podoplanin, a representative immunohistochemical marker for lymphatic endothelial cells, is localized in both CCOT basal and polyhedral cells, which are coincidentally positive for proliferating cell nuclear antigen and integrin β1. Thus, podoplanin-positive cells may be closely associated with extracellular matrix signaling and cell proliferation in CCOT.78
Prognosis
CCOT is a noninvasive odontogenic neoplasm with ameloblastomatous epithelium with fewer aggressive features than ameloblastoma. CCOT is usually uneventful after simple enucleation and curettage, whereas extensive radical surgery is recommended for ameloblastoma. CCOTs associated with odontoma, a supernumerary tooth, or a dentigerous cyst of embedded canine may show good prognosis after simple enucleation.65
DENTINOGENIC GHOST CELL TUMOR
General features
DGCT is a rare benign odontogenic solid tissue tumor derived from a neoplastic COC variant, but neoplastic epithelium is more predominant than the cystic component, which is contrary to that observed in CCOT.54 The peripheral DGCT variant appears as a well-circumscribed mass mimicking a nonspecific gingival enlargement, whereas intraosseous DGCTs are more aggressive than extraosseous DGCTs and have a high propensity for local recurrence.79,80
DGCT is histologically characterized by odontogenic epithelium islands showing ameloblastoma-like features in fibrous tissue, where dysplastic dentin and ghost cells are frequently observed. The neoplastic DGCT epithelia are considered to be locally invasive. A microscopic dentinoid formation with ghost cells may provide a definitive DGCT diagnosis. Van Giesson histochemical stain further confirms the dentinoid-like materials (Fig. 3).81 DGCT is sometimes accompanied by COC, and the DGCT dentinoid materials are eosinophilic, amorphous, and sometimes undergo transformation into osteodentin.82 Furthermore, dendritic cells representing Langerhans cells and melanocytes often infiltrate into tumoral epithelium.83
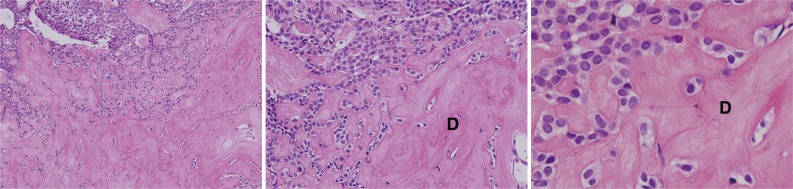
Fig. 3.
Dentinogenic ghost cell tumor photomicrographs. The polygonal epithelial strands are proliferative in the vicinity of eosinophilic dentinoid materials (D).
Types and variants
DGCT can be separated into central aggressive and peripheral non-aggressive types.54 As peripheral (extraosseous) lesions commonly show gingival swelling, they can be easily confused with other gingival lesions, such as, reactive or inflammatory lesions, or with other peripheral odontogenic tumors. Central lesions have the potential to invade locally, thus may have variable recurrence rates.79
DGCT is defined as a solid neoplastic growth formed by epithelial cell groups and islands visualized as an ameloblastoma-like basal cell layer admixed with variable quantities of dentin-like materials and ghost cell groups.54 These DGCT ameloblastomatous features are sometimes difficult to distinguish from ameloblastoma. In one case, ameloblastoma was associated with DGCT with a predominant proliferating neoplastic epithelium feature.84
Pathogenesis
The DGCT odontogenic tumor epithelium shows variable reactions for keratins, and demonstrates PKK1 staining in peripheral tumor cells, staining for KL1, and involucrin in centrally located cells. Lectin binding in amorphous dentinoid materials was reported to be comparatively strong for PNA and SBA, moderate for WGA, RCA-1, and UEA-1, and slight for DBA and ConA. In the same study, amorphous dentinoid material elemental analysis by electron probe X-ray microanalysis showed a pattern similar to that found in normal dentin.82
β-Catenin and lymphoid enhancer-binding factor 1 are frequently positive in odontogenic epithelial cell cytoplasm and nuclei adjacent to ghost cells, which suggests involvement of the Wnt signaling pathway during ghost cell formation.85 Nucleated tumor cells adjacent to ghost cells are occasionally terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay positive. Furthermore, β-catenin mutations have been described in DGCT and in COC, suggesting β-catenin plays an important role in DGCT tumorigenesis.86
Prognosis
DGCT is a locally invasive neoplasm characterized by ameloblastoma-like epithelial islands, ghost cells, and dentinoid.87 Peripheral DGCT is absolutely benign with no features of invasive growth and is invariably uneventful after surgical removal. On the other hand, intraosseous DGCT is potentially invasive and tends to recur, and hence requires radical surgery.54,79,80
In one report on seven intraosseous DGCTs, five treated by conservative surgery recurred, but the two treated by aggressive local resection did not.88 Thus, intraosseous DGCT should be treated by resection with an adequate safety margin of at least 0.5 cm (as is recommended for ameloblastoma), and adequately followed for signs of recurrence.80,88,89
GHOST CELL ODONTOGENIC CARCINOMA
General features
GCOC is an uncommon malignant neoplasm exhibiting prominent mitotic activity, nuclear atypia, cellular pleomorphism, ghost epithelial cell groups, necrosis, and sometimes scarce mineralized or dentin-like materials. GCOC has an infiltrative growth pattern with regional metastasis. Furthermore, as tumor cells undergo malignant processes, ghost cell may be difficult to find.90
This neoplasm has a locally aggressive, destructive behavior, and at times metastatic deposits can be found. It may be diagnosed as CCOT, DGCT, or GCOTs before its malignant transformation, although it often co-occurs with benign variant odontogenic tumors derived from COC.91
In one 30 case GCOC study, mean age at diagnosis was 40.3 years, 22 (73%) involved the maxilla, and 12 (40%) were secondary benign CCOT malignant manifestations, or DGCTs. Five patients died of recurrence or distant metastasis.92 It has also been reported that GCOC is more prevalent in Asians (12/18) than in other racial groups.93 A radiological study found a mixed radiolucent and radiopaque lesion pattern was most frequent (14/19) compared to a radiolucent lesion pattern (5/19). In addition, 89% (17/19) showed poorly defined borders and 11% (2/19) showed well defined borders. Root resorption was reported in 31% (6/19) of patients, and tooth displacements in 21% (4/19).93,94,95
Types and variants
Three GCOC variants are identified according to their pathogenic mechanisms, as follows: 1) GCOC arising de novo is a malignant neoplasm not associated with previous DGCT or CCOT, although sometimes DGCT-like areas may be present;96 2) GCOC ex-CCOT is considered when a GCOC is found to be derived from previously diagnosed CCOT, or when a GCOC is associated with benign CCOT;54,92 and 3) GCOC ex-DGCT is a recurrent malignant neoplasm previously diagnosed as benign DGCT, in which occasional dentin-like material areas are demonstrated.90
Pathogenesis
Although GCOC has different histological presentations and unpredictable indolent or locally aggressive growth behaviors, the tumor usually arises due to preexisting benign COC (CCOT) malignant transformation, and sometimes from other odontogenic tumors.93 However, immunohistochemical reactivities for Ki-67 and MMP-9 are significantly stronger in GCOC than in CCOT, or in DGCT.77,97 In particular, nucleated cells adjacent to ghost cells in GCOC express cytokeratins, involucrin, and BCL-2 associated X (BAX), and they are frequently positive for Ki-67 and p53, and occasionally TUNEL positive.98 These findings support the observation that GCOC exhibits significantly greater proliferative activity and invasive growth than CCOT or DGCT.
Despite their retrogressive and apoptotic status, GCOC ghost cells consistently express BAX and vitronectin receptor proteins, and are strongly positive for the tartrate-resistant acid phosphatase reaction.99 These facts indicate anucleated ghost cells still play important roles in the extensive bone resorption observed in GCOC.91,98
Prognosis
GCOCs may grow slowly and be locally invasive or grow rapidly and be infiltrative. Therefore, wide local excision with histologically clean margins is recommended for GCOCs. Furthermore, close long-term surveillance for recurrence is required for GCOCs arising from long-standing benign COCs or GCOTs.100
To date 31 GCOC cases have been described in English publications, and three cases involved Koreans.91,98 As was suggested by another author,50 we recommend more GCOC cases be reported (using the WHO tumor classification) to allow their variable malignant potential to be better defined.
SUMMARY
This review of the currently available CEOT and GCOTs literature shows that CEOT and GCOTs are uncommon tumors exhibiting variable calcification features and ghost cells produced during odontogenic epithelium tumorigenic differentiation. We advise CEOT and GCOTs differential diagnosis should be undertaken carefully, despite the confusion and controversy associated with histological diagnoses of these heterogeneous and transient odontogenic tumors. Furthermore, in order to more precisely define their biological and pathological behaviors, further advanced studies are required, and publication should be encouraged to provide comprehensive data.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Main JH, Waheed MA. Epitheliomesenchymal interactions in the proliferative response evoked by polyoma virus in odontogenic epithelium in vitro. J Natl Cancer Inst. 1971;47:711–726. [PubMed] [Google Scholar]
- 2.Slavkin HC, Cummings E, Bringas P, Honig LS. Epithelial-derived basal lamina regulation of mesenchymal cell differentiation. Prog Clin Biol Res. 1982;85(Pt B):249–259. [PubMed] [Google Scholar]
- 3.Badger KV, Gardner DG. The relationship of adamantinomatous craniopharyngioma to ghost cell ameloblastoma of the jaws: a histopathologic and immunohistochemical study. J Oral Pathol Med. 1997;26:349–355. doi: 10.1111/j.1600-0714.1997.tb00229.x. [DOI] [PubMed] [Google Scholar]
- 4.Lucchese A, Scivetti M, Pilolli GP, Favia G. Analysis of ghost cells in calcifying cystic odontogenic tumors by confocal laser scanning microscopy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:391–394. doi: 10.1016/j.tripleo.2006.09.005. [DOI] [PubMed] [Google Scholar]
- 5.Pindborg JJ. A calcifying epithelial odontogenic tumor. Cancer. 1958;11:838–843. doi: 10.1002/1097-0142(195807/08)11:4<838::aid-cncr2820110423>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 6.Shafer WG. Cysts, neoplasms, and allied conditions of odontogenic origin. Semin Roentgenol. 1971;6:403–413. doi: 10.1016/0037-198x(71)90052-6. [DOI] [PubMed] [Google Scholar]
- 7.Singh N, Sahai S, Singh S, Singh S. Calcifying epithelial odontogenic tumor (Pindborg tumor) Natl J Maxillofac Surg. 2011;2:225–227. doi: 10.4103/0975-5950.94489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Neville B, Damm DD, Allen CM, Bouquot J. Oral and maxillofacial pathology. Philadelphia: WB Saunders; 2009. [Google Scholar]
- 9.Krolls SO, Pindborg JJ. Calcifying epithelial odontogenic tumor: a survey of 23 cases and discussion of histomorphologic variations. Arch Pathol. 1974;98:206–210. [PubMed] [Google Scholar]
- 10.Sharma U, Gulati A, Batra H, Singh D. Calcifying epithelial odontogenic tumor in anterior maxilla associated with a supernumerary tooth: a case report. J Dent Res Dent Clin Dent Prospects. 2013;7:51–54. doi: 10.5681/joddd.2013.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ai-Ru L, Zhen L, Jian S. Calcifying epithelial odontogenic tumors: a clinicopathologic study of nine cases. J Oral Pathol. 1982;11:399–406. doi: 10.1111/j.1600-0714.1982.tb00181.x. [DOI] [PubMed] [Google Scholar]
- 12.Philipsen HP, Reichart PA. Calcifying epithelial odontogenic tumour: biological profile based on 181 cases from the literature. Oral Oncol. 2000;36:17–26. doi: 10.1016/s1368-8375(99)00061-5. [DOI] [PubMed] [Google Scholar]
- 13.Channappa NK, Krishnapillai R, Rao JB. Cystic variant of calcifying epithelial odontogenic tumor. J Investig Clin Dent. 2012;3:152–156. doi: 10.1111/j.2041-1626.2011.00092.x. [DOI] [PubMed] [Google Scholar]
- 14.Ng KH, Siar CH. A clinicopathological and immunohistochemical study of the calcifying epithelial odontogenic tumour (Pindborg tumour) in Malaysians. J Laryngol Otol. 1996;110:757–762. doi: 10.1017/s0022215100134887. [DOI] [PubMed] [Google Scholar]
- 15.Habibi A, Saghravanian N, Zare R, Jafarzadeh H. Clear cell variant of extraosseous calcifying epithelial odontogenic tumor: a case report. J Oral Sci. 2009;51:485–488. doi: 10.2334/josnusd.51.485. [DOI] [PubMed] [Google Scholar]
- 16.Kaushal S, Mathur SR, Vijay M, Rustagi A. Calcifying epithelial odontogenic tumor (Pindborg tumor) without calcification: a rare entity. J Oral Maxillofac Pathol. 2012;16:110–112. doi: 10.4103/0973-029X.92985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang YP, Lee JJ, Wang JT, et al. Non-calcifying variant of calcifying epithelial odontogenic tumor with Langerhans cells. J Oral Pathol Med. 2007;36:436–439. doi: 10.1111/j.1600-0714.2007.00533.x. [DOI] [PubMed] [Google Scholar]
- 18.Ide F, Obara K, Mishima K, et al. Peripheral odontogenic tumor: a clinicopathologic study of 30 cases. General features and hamartomatous lesions. J Oral Pathol Med. 2005;34:552–557. doi: 10.1111/j.1600-0714.2005.00355.x. [DOI] [PubMed] [Google Scholar]
- 19.Montes Ledesma C, Mosqueda Taylor A, Romero de Leon E, de la Piedra Garza M, Goldberg Jaukin P, Portilla Robertson J. Adenomatoid odontogenic tumour with features of calcifying epithelial odontogenic tumour (The so-called combined epithelial odontogenic tumour): clinico-pathological report of 12 cases. Eur J Cancer B Oral Oncol. 1993;29B:221–224. doi: 10.1016/0964-1955(93)90026-b. [DOI] [PubMed] [Google Scholar]
- 20.Abrahão AC, Camisasca DR, Bonelli BR, et al. Recurrent bilateral gingival peripheral calcifying epithelial odontogenic tumor (Pindborg tumor): a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108:e66–e71. doi: 10.1016/j.tripleo.2009.04.037. [DOI] [PubMed] [Google Scholar]
- 21.Poomsawat S, Punyasingh J. Calcifying epithelial odontogenic tumor: an immunohistochemical case study. J Mol Histol. 2007;38:103–109. doi: 10.1007/s10735-007-9082-9. [DOI] [PubMed] [Google Scholar]
- 22.Mori M, Tatemoto Y, Yamamoto N, Okada Y. Immunohistochemical localization of intermediate filament proteins in calcifying epithelial odontogenic tumors. J Oral Pathol. 1988;17:236–240. doi: 10.1111/j.1600-0714.1988.tb01531.x. [DOI] [PubMed] [Google Scholar]
- 23.Morimoto C, Tsujimoto M, Shimaoka S, Shirasu R, Takasu J. Ultrastructural localization of alkaline phosphatase in the calcifying epithelial odontogenic tumor. Oral Surg Oral Med Oral Pathol. 1983;56:409–414. doi: 10.1016/0030-4220(83)90352-3. [DOI] [PubMed] [Google Scholar]
- 24.Chomette G, Auriol M, Guilbert F. Histoenzymological and ultrastructural study of a bifocal calcifying epithelial odontogenic tumor: characteristics of epithelial cells and histogenesis of amyloid-like material. Virchows Arch A Pathol Anat Histopathol. 1984;403:67–76. doi: 10.1007/BF00689339. [DOI] [PubMed] [Google Scholar]
- 25.El-Labban NG, Lee KW, Kramer IR. The duality of the cell population in a calcifying epithelial odontogenic tumour (CEOT) Histopathology. 1984;8:679–691. doi: 10.1111/j.1365-2559.1984.tb02380.x. [DOI] [PubMed] [Google Scholar]
- 26.El-Labban NG, Lee KW, Kramer IR, Harris M. The nature of the amyloid-like material in a calcifying epithelial odontogenic tumour: an ultrastructural study. J Oral Pathol. 1983;12:366–374. doi: 10.1111/j.1600-0714.1983.tb00349.x. [DOI] [PubMed] [Google Scholar]
- 27.Murphy CL, Kestler DP, Foster JS, et al. Odontogenic ameloblast-associated protein nature of the amyloid found in calcifying epithelial odontogenic tumors and unerupted tooth follicles. Amyloid. 2008;15:89–95. doi: 10.1080/13506120802005965. [DOI] [PubMed] [Google Scholar]
- 28.Takata T, Ogawa I, Miyauchi M, Ijuhin N, Nikai H, Fujita M. Non-calcifying Pindborg tumor with Langerhans cells. J Oral Pathol Med. 1993;22:378–383. doi: 10.1111/j.1600-0714.1993.tb01093.x. [DOI] [PubMed] [Google Scholar]
- 29.Perdigão PF, Carvalho VM, De Marco L, Gomez RS. Mutation of ameloblastin gene in calcifying epithelial odontogenic tumor. Anticancer Res. 2009;29:3065–3067. [PubMed] [Google Scholar]
- 30.Paine ML, Wang HJ, Luo W, Krebsbach PH, Snead ML. A transgenic animal model resembling amelogenesis imperfecta related to ameloblastin overexpression. J Biol Chem. 2003;278:19447–19452. doi: 10.1074/jbc.M300445200. [DOI] [PubMed] [Google Scholar]
- 31.Franklin CD, Pindborg JJ. The calcifying epithelial odontogenic tumor: a review and analysis of 113 cases. Oral Surg Oral Med Oral Pathol. 1976;42:753–765. doi: 10.1016/0030-4220(76)90098-0. [DOI] [PubMed] [Google Scholar]
- 32.Kamath G, Abraham R. Recurrent CEOT of the maxilla. Dent Res J (Isfahan) 2012;9:233–236. doi: 10.4103/1735-3327.95242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Veness MJ, Morgan G, Collins AP, Walker DM. Calcifying epithelial odontogenic (Pindborg) tumor with malignant transformation and metastatic spread. Head Neck. 2001;23:692–696. doi: 10.1002/hed.1097. [DOI] [PubMed] [Google Scholar]
- 34.Kawano K, Ono K, Yada N, et al. Malignant calcifying epithelial odontogenic tumor of the mandible: report of a case with pulmonary metastasis showing remarkable response to platinum derivatives. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:76–81. doi: 10.1016/j.tripleo.2006.04.014. [DOI] [PubMed] [Google Scholar]
- 35.Somayaji G, Rajeshwary A, Ramesh S, Dinesh S. Recurrent Pindborg tumor of the maxilla: a case report and review of the literature. Ear Nose Throat J. 2013;92:84–87. [PubMed] [Google Scholar]
- 36.Gorlin RJ, Pindborg JJ, Odont, Clausen FP, Vickers RA. The calcifying odontogenic cyst: a possible analogue of the cutaneous calcifying epithelioma of Malherbe: an analysis of fifteen cases. Oral Surg Oral Med Oral Pathol. 1962;15:1235–1243. doi: 10.1016/0030-4220(62)90159-7. [DOI] [PubMed] [Google Scholar]
- 37.Freedman PD, Lumerman H, Gee JK. Calcifying odontogenic cyst: a review and analysis of seventy cases. Oral Surg Oral Med Oral Pathol. 1975;40:93–106. doi: 10.1016/0030-4220(75)90351-5. [DOI] [PubMed] [Google Scholar]
- 38.Siar CH, Ng KH. Histological variation of the lining epithelium of the calcifying odontogenic cyst: a light microscopic study. Dent J Malays. 1988;10:42–47. [PubMed] [Google Scholar]
- 39.Kwon HI, Lim WB, Kim JS, et al. Odontogenic keratocyst associated with an ectopic tooth in the maxillary sinus: a report of two cases and a review of the literature. Korean J Pathol. 2011;45(Suppl 1):S5–S10. [Google Scholar]
- 40.Kim YS, Lee SK. Aberrant ossification in basophilic ghost cells of calcifying odontogenic cyst resembling to pilomatricoma. Korean J Oral Maxillofac Pathol. 2012;36:287–292. [Google Scholar]
- 41.Mori M, Yamada K, Kasai T, Yamada T, Shimokawa H, Sasaki S. Immunohistochemical expression of amelogenins in odontogenic epithelial tumours and cysts. Virchows Arch A Pathol Anat Histopathol. 1991;418:319–325. doi: 10.1007/BF01600161. [DOI] [PubMed] [Google Scholar]
- 42.Yoshida M, Kumamoto H, Ooya K, Mayanagi H. Histopathological and immunohistochemical analysis of calcifying odontogenic cysts. J Oral Pathol Med. 2001;30:582–588. doi: 10.1034/j.1600-0714.2001.301002.x. [DOI] [PubMed] [Google Scholar]
- 43.Johnson A, 3rd, Fletcher M, Gold L, Chen SY. Calcifying odontogenic cyst: a clinicopathologic study of 57 cases with immunohistochemical evaluation for cytokeratin. J Oral Maxillofac Surg. 1997;55:679–683. doi: 10.1016/s0278-2391(97)90574-7. [DOI] [PubMed] [Google Scholar]
- 44.Li TJ, Yu SF. Clinicopathologic spectrum of the so-called calcifying odontogenic cysts: a study of 21 intraosseous cases with reconsideration of the terminology and classification. Am J Surg Pathol. 2003;27:372–384. doi: 10.1097/00000478-200303000-00011. [DOI] [PubMed] [Google Scholar]
- 45.Lucchese A, Petruzzi M, Scivetti M, et al. Calcifying odontogenic cysts associated with odontomas: confocal laser scanning microscopy analysis of 13 cases. Ultrastruct Pathol. 2011;35:146–150. doi: 10.3109/01913123.2010.548111. [DOI] [PubMed] [Google Scholar]
- 46.Hong SP, Ellis GL, Hartman KS. Calcifying odontogenic cyst: a review of ninety-two cases with reevaluation of their nature as cysts or neoplasms, the nature of ghost cells, and subclassification. Oral Surg Oral Med Oral Pathol. 1991;72:56–64. doi: 10.1016/0030-4220(91)90190-n. [DOI] [PubMed] [Google Scholar]
- 47.Gallana-Alvarez S, Mayorga-Jimenez F, Torres-Gómez FJ, Avellá-Vecino FJ, Salazar-Fernandez C. Calcifying odontogenic cyst associated with complex odontoma: case report and review of the literature. Med Oral Patol Oral Cir Bucal. 2005;10:243–247. [PubMed] [Google Scholar]
- 48.Basile JR, Klene C, Lin YL. Calcifying odontogenic cyst with odontogenic keratocyst: a case report and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;109:e40–e45. doi: 10.1016/j.tripleo.2009.12.026. [DOI] [PubMed] [Google Scholar]
- 49.Philipsen HP, Reichart PA. Classification of odontogenic tumours: a historical review. J Oral Pathol Med. 2006;35:525–529. doi: 10.1111/j.1600-0714.2006.00470.x. [DOI] [PubMed] [Google Scholar]
- 50.Barnes L, Eveson JW, Reichart P, Sidransky D. World Health Organization classification of tumours: pathology and genetics of head and neck tumours. Lyon: IARC Press; 2005. pp. 1–41. [Google Scholar]
- 51.Prætorius F, Ledesma-Montes C. Calcifying cystic odontogenic tumour. In: Barnes L, Eveson JW, Reichart P, Sidransky D, editors. World Health Organization classification of tumours: pathology and genetics of head and neck tumours. Lyon: IARC Press; 2005. p. 313. [Google Scholar]
- 52.Prætorius F, Ledesma-Montes C. Dentinogenic ghost cell tumour. In: Barnes L, Eveson JW, Reichart P, Sidransky D, editors. World Health Organization classification of tumours: pathology and genetics of head and neck tumours. Lyon: IARC Press; 2005. p. 314. [Google Scholar]
- 53.Takata T, Lu Y. Ghost cell odontogenic carcinoma. In: Barnes L, Eveson JW, Reichart P, Sidransky D, editors. World Health Organization classification of tumours: pathology and genetics of head and neck tumours. Lyon: IARC Press; 2005. p. 293. [Google Scholar]
- 54.Ledesma-Montes C, Gorlin RJ, Shear M, et al. International collaborative study on ghost cell odontogenic tumours: calcifying cystic odontogenic tumour, dentinogenic ghost cell tumour and ghost cell odontogenic carcinoma. J Oral Pathol Med. 2008;37:302–308. doi: 10.1111/j.1600-0714.2007.00623.x. [DOI] [PubMed] [Google Scholar]
- 55.Lombardi T, Kuffer R, Di Felice R, Samson J. Epithelial odontogenic ghost cell tumour of the mandibular gingiva. Oral Oncol. 1999;35:439–442. doi: 10.1016/s1368-8375(98)00128-6. [DOI] [PubMed] [Google Scholar]
- 56.Ellis GL. Odontogenic ghost cell tumor. Semin Diagn Pathol. 1999;16:288–292. [PubMed] [Google Scholar]
- 57.Hogge M, Velez I, Kaltman S, Movahed R, Yeh F. Ghost cell odontogenic tumor associated with odontoma: report of two rare cases. J Clin Pediatr Dent. 2012;36:373–376. doi: 10.17796/jcpd.36.4.h0x50662vj624912. [DOI] [PubMed] [Google Scholar]
- 58.Yoon JH, Ahn SG, Kim SG, Kim J. Odontogenic ghost cell tumour with clear cell components: clear cell odontogenic ghost cell tumour? J Oral Pathol Med. 2004;33:376–379. doi: 10.1111/j.1600-0714.2004.00200.x. [DOI] [PubMed] [Google Scholar]
- 59.Fejerskov O, Krogh J. The calcifying ghost cell odontogenic tumor - or the calcifying odontogenic cyst. J Oral Pathol. 1972;1:273–287. doi: 10.1111/j.1600-0714.1972.tb01666.x. [DOI] [PubMed] [Google Scholar]
- 60.de Lima AP, Kitakawa D, Almeida JD, Brandão AA, Anbinder AL. Peripheral calcifying cystic odontogenic tumour of the maxillary gingiva. BMC Res Notes. 2012;5:455. doi: 10.1186/1756-0500-5-455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kler S, Palaskar S, Shetty VP, Bhushan A. Intraosseous calcifying cystic odontogenic tumor. J Oral Maxillofac Pathol. 2009;13:27–29. doi: 10.4103/0973-029X.48753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Habibi A, Saghravanian N, Salehinejad J, Jafarzadeh H. Thirty years clinicopathological study of 60 calcifying cystic odontogenic tumors in Iranian population. J Contemp Dent Pract. 2011;12:171–173. doi: 10.5005/jp-journals-10024-1029. [DOI] [PubMed] [Google Scholar]
- 63.Buchner A, Merrell PW, Carpenter WM. Relative frequency of peripheral odontogenic tumors: a study of 45 new cases and comparison with studies from the literature. J Oral Pathol Med. 2006;35:385–391. doi: 10.1111/j.1600-0714.2006.00437.x. [DOI] [PubMed] [Google Scholar]
- 64.Chindasombatjaroen J, Poomsawat S, Kakimoto N, Shimamoto H. Calcifying cystic odontogenic tumor and adenomatoid odontogenic tumor: radiographic evaluation. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114:796–803. doi: 10.1016/j.oooo.2012.08.452. [DOI] [PubMed] [Google Scholar]
- 65.Chindasombatjaroen J, Poomsawat S, Klongnoi B. Calcifying cystic odontogenic tumor associated with other lesions: case report with cone-beam computed tomography findings. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113:414–420. doi: 10.1016/j.oooo.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 66.Poleti ML, Duarte BG, Lara VS, Freitas-Faria P, Rubira-Bullen IR, Goncales ES. Odontoma associated with calcifying cystic odontogenic tumor in deciduous dentition: case report. Oral Maxillofac Surg. 2013;17:77–80. doi: 10.1007/s10006-012-0333-x. [DOI] [PubMed] [Google Scholar]
- 67.Soares EC, Costa FW, Neto IC, Bezerra TP, do Socorro Vidal Patrocínio RM, Alves AP. Rare hybrid odontogenic tumor in a 2-year-old child. J Craniofac Surg. 2011;22:554–558. doi: 10.1097/SCS.0b013e3182074616. [DOI] [PubMed] [Google Scholar]
- 68.Chaubey SS, Mishra SS, Degwekar SS, Chaubey S. A rare presentation of hybrid odontogenic tumor involving calcifying cystic odontogenic tumor and plexiform ameloblastoma. Contemp Clin Dent. 2013;4:406–408. doi: 10.4103/0976-237X.118369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Crivelini MM, Felipini RC, Coclete GA, Soubhia AM. Immunoexpression of keratins in the calcifying cystic odontogenic tumor epithelium. J Oral Pathol Med. 2009;38:393–396. doi: 10.1111/j.1600-0714.2008.00745.x. [DOI] [PubMed] [Google Scholar]
- 70.Poomsawat S, Punyasingh J, Vejchapipat P. Expression of basement membrane components in odontogenic tumors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:666–675. doi: 10.1016/j.tripleo.2006.08.025. [DOI] [PubMed] [Google Scholar]
- 71.Chen J, Aufdemorte TB, Jiang H, Liu AR, Zhang W, Thomas HF. Neoplastic odontogenic epithelial cells express bone sialoprotein. Histochem J. 1998;30:1–6. doi: 10.1023/a:1003228910529. [DOI] [PubMed] [Google Scholar]
- 72.Siar CH, Kawakami T, Buery RR, et al. Notch signaling and ghost cell fate in the calcifying cystic odontogenic tumor. Eur J Med Res. 2011;16:501–506. doi: 10.1186/2047-783X-16-11-501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Andrade FR, Sousa DP, Mendonça EF, Silva TA, Lara VS, Batista AC. Expression of bone resorption regulators (RANK, RANKL, and OPG) in odontogenic tumors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106:548–555. doi: 10.1016/j.tripleo.2008.05.042. [DOI] [PubMed] [Google Scholar]
- 74.Karathanasi V, Tosios KI, Nikitakis NG, et al. TGF-beta1, Smad-2/-3, Smad-1/-5/-8, and Smad-4 signaling factors are expressed in ameloblastomas, adenomatoid odontogenic tumors, and calcifying cystic odontogenic tumors: an immunohistochemical study. J Oral Pathol Med. 2013;42:415–423. doi: 10.1111/jop.12016. [DOI] [PubMed] [Google Scholar]
- 75.Ribeiro AL, Nobre RM, Alves-Junior SM, et al. Matrix metalloproteinases, tissue inhibitors of metalloproteinases, and growth factors regulate the aggressiveness and proliferative activity of keratocystic odontogenic tumors. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114:487–496. doi: 10.1016/j.oooo.2012.06.011. [DOI] [PubMed] [Google Scholar]
- 76.Siqueira AS, Carvalho MR, Monteiro AC, Freitas VM, Jaeger RG, Pinheiro JJ. Matrix metalloproteinases, TIMPs and growth factors regulating ameloblastoma behaviour. Histopathology. 2010;57:128–137. doi: 10.1111/j.1365-2559.2010.03596.x. [DOI] [PubMed] [Google Scholar]
- 77.Gong Y, Wang L, Wang H, Li T, Chen X. The expression of NF-kappaB, Ki-67 and MMP-9 in CCOT, DGCT and GCOC. Oral Oncol. 2009;45:515–520. doi: 10.1016/j.oraloncology.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 78.Tsuneki M, Maruyama S, Yamazaki M, Cheng J, Saku T. Podoplanin expression profiles characteristic of odontogenic tumor-specific tissue architectures. Pathol Res Pract. 2012;208:140–146. doi: 10.1016/j.prp.2011.12.016. [DOI] [PubMed] [Google Scholar]
- 79.Wong YK, Chiu SC, Pang SW, Cheng JC. Peripheral dentinogenic ghost cell tumour presenting as a gingival mass. Br J Oral Maxillofac Surg. 2004;42:173–175. doi: 10.1016/S0266-4356(03)00239-0. [DOI] [PubMed] [Google Scholar]
- 80.Ramaglia L, Esposito D, Bruno MP, Siano M. Dentinogenic ghost cell tumor: histopathology and clinical aspects in pediatric age. Minerva Stomatol. 2012;61:509–517. [PubMed] [Google Scholar]
- 81.Kumar U, Vij H, Vij R, Kharbanda J, Aparna I, Radhakrishnan R. Dentinogenic ghost cell tumor of the peripheral variant mimicking epulis. Int J Dent. 2010;2010:519494. doi: 10.1155/2010/519494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Mori M, Kasai T, Nakai M, et al. Dentinogenic ghost cell tumor: histologic aspects, immunohistochemistry, lectin binding profiles, and biophysical studies. Oral Oncol. 2000;36:134–143. doi: 10.1016/s1368-8375(99)00056-1. [DOI] [PubMed] [Google Scholar]
- 83.Candido GA, Viana KA, Watanabe S, Vencio EF. Peripheral dentinogenic ghost cell tumor: a case report and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108:e86–e90. doi: 10.1016/j.tripleo.2009.04.033. [DOI] [PubMed] [Google Scholar]
- 84.Kim TJ, Lee YS, Kim BK, Lee KY. Ameloblastoma associated with dentinogenic ghost cell tumor: a case report. Korean J Pathol. 2006;40:297–302. [Google Scholar]
- 85.Tanaka A, Okamoto M, Yoshizawa D, et al. Presence of ghost cells and the Wnt signaling pathway in odontomas. J Oral Pathol Med. 2007;36:400–404. doi: 10.1111/j.1600-0714.2007.00550.x. [DOI] [PubMed] [Google Scholar]
- 86.Kim SA, Ahn SG, Kim SG, et al. Investigation of the beta-catenin gene in a case of dentinogenic ghost cell tumor. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:97–101. doi: 10.1016/j.tripleo.2005.10.037. [DOI] [PubMed] [Google Scholar]
- 87.Singhaniya SB, Barpande SR, Bhavthankar JD. Dentinogenic ghost cell tumor. J Oral Maxillofac Pathol. 2009;13:97–100. doi: 10.4103/0973-029X.57679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sun G, Huang X, Hu Q, Yang X, Tang E. The diagnosis and treatment of dentinogenic ghost cell tumor. Int J Oral Maxillofac Surg. 2009;38:1179–1183. doi: 10.1016/j.ijom.2009.06.016. [DOI] [PubMed] [Google Scholar]
- 89.Park HR, Min JH, Huh KH, et al. Distant metastasis of intraosseous dentinogenic ghost cell tumour to the donor site of a bone graft. Dentomaxillofac Radiol. 2013;42:20120172. doi: 10.1259/dmfr.20120172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Li BB, Gao Y. Ghost cell odontogenic carcinoma transformed from a dentinogenic ghost cell tumor of maxilla after multiple recurrences. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107:691–695. doi: 10.1016/j.tripleo.2009.01.008. [DOI] [PubMed] [Google Scholar]
- 91.Li BH, Cho YA, Kim SM, Kim MJ, Hong SP, Lee JH. Recurrent odontogenic ghost cell carcinoma (OGCC) at a reconstructed fibular flap: a case report with immunohistochemical findings. Med Oral Patol Oral Cir Bucal. 2011;16:e651–e656. doi: 10.4317/medoral.17207. [DOI] [PubMed] [Google Scholar]
- 92.Arashiyama T, Kodama Y, Kobayashi T, et al. Ghost cell odontogenic carcinoma arising in the background of a benign calcifying cystic odontogenic tumor of the mandible. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114:e35–e40. doi: 10.1016/j.oooo.2012.01.018. [DOI] [PubMed] [Google Scholar]
- 93.Lu Y, Mock D, Takata T, Jordan RC. Odontogenic ghost cell carcinoma: report of four new cases and review of the literature. J Oral Pathol Med. 1999;28:323–329. doi: 10.1111/j.1600-0714.1999.tb02048.x. [DOI] [PubMed] [Google Scholar]
- 94.Cheng Y, Long X, Li X, Bian Z, Chen X, Yang X. Clinical and radiological features of odontogenic ghost cell carcinoma: review of the literature and report of four new cases. Dentomaxillofac Radiol. 2004;33:152–157. doi: 10.1259/dmfr/67909783. [DOI] [PubMed] [Google Scholar]
- 95.Kim HJ, Choi SK, Lee CJ, Suh CH. Aggressive epithelial odontogenic ghost cell tumor in the mandible: CT and MR imaging findings. AJNR Am J Neuroradiol. 2001;22:175–179. [PMC free article] [PubMed] [Google Scholar]
- 96.Nazaretian SP, Schenberg ME, Simpson I, Slootweg PJ. Ghost cell odontogenic carcinoma. Int J Oral Maxillofac Surg. 2007;36:455–458. doi: 10.1016/j.ijom.2006.10.007. [DOI] [PubMed] [Google Scholar]
- 97.Gong YL, Wang L, Chen XM, Wang HK, Wang XH. Expression of nuclear factor-kappaB, Ki-67 and matrix metalloproteinase-9 in calcifying odontogenic cyst. Zhonghua Kou Qiang Yi Xue Za Zhi. 2006;41:627–630. [PubMed] [Google Scholar]
- 98.Kim J, Lee EH, Yook JI, Han JY, Yoon JH, Ellis GL. Odontogenic ghost cell carcinoma: a case report with reference to the relation between apoptosis and ghost cells. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90:630–635. doi: 10.1067/moe.2000.109016. [DOI] [PubMed] [Google Scholar]
- 99.Roh GS, Jeon BT, Park BW, et al. Ghost cell odontogenic carcinoma of the mandible: a case report demonstrating expression of tartrate-resistant acid phosphatase (TRAP) and vitronectin receptor. J Craniomaxillofac Surg. 2008;36:419–423. doi: 10.1016/j.jcms.2008.06.001. [DOI] [PubMed] [Google Scholar]
- 100.Goldenberg D, Sciubba J, Tufano RP. Odontogenic ghost cell carcinoma. Head Neck. 2004;26:378–381. doi: 10.1002/hed.10376. [DOI] [PubMed] [Google Scholar]