Abstract
AIM: To elucidate the impact of hemodynamic para-meters on ultrasonography and serum fibrosis markers for the assessment of liver fibrosis in the children with infant hepatitis syndrome (IHS).
METHODS: Forty-one children with IHS and 46 healthy infants were examined by ultrasonography, and several hemodynamic indices such as peak systolic velocity (PSV) and resistant index (RI) of proper hepatic artery (PHA) were measured. Serum fibrosis markers including hyaluronic acid (HA), pre-collagen type-III (PC-III), collagen type IV (C-IV), and laminin (LN) were assayed by radioimmunoassays. In children with IHS, liver tissues were obtained either by ultrasound-guided liver biopsy (n = 35) or in the course of operation (n = 6). The stages of hepatic fibrosis were scored as mild (S1 and S2), moderate (S3), or severe (S4) according to liver histological diagnosis. Multiple groups comparative and Spearman correlative analyses were carried out.
RESULTS: Histopathologically, 39 children (95.1%) were found to have hepatic fibrosis, 12 of them stage S1 or S2, 12 stage S3, and 15 stage S4. PSV, RI of the PHA, and serum HA showed a consecutive increase from mild to severe hepatic fibrosis and a close positive correlation with hepatic fibrosis in IHS group (r = 0.717, 0.745 and 0.712, respectively, P = 0.001). The Doppler waveform of HV was also positively correlated with the degree of hepatic fibrosis in IHS group (n = 0.783, P < 0.001).
CONCLUSION: Combination of ultrasonic studies on the hepatic hemodynamics with the evaluation of serum HA may provide an indicator for hepatic fibrosis in patients with IHS. This may be a useful noninvasive method for the diagnosis and evaluation of the prognosis of IHS.
Keywords: Infant hepatitis syndrome, Hepatic fibrosis, Doppler ultrasonography, Hemodynamics, Hyaluronic acid
INTRODUCTION
Infant hepatitis syndrome (IHS)[1] is defined as elevation of serum levels of conjugated bilirubin in the first 12 mo of life, may be due to infectious, genetic, metabolic, or undefined abnormalities, giving rise either to mechanical obstruction of bile flow or to functional impairment of hepatic excretory function and bile secretion. Main symptoms are prolonged jaundice, hepatomegaly or hepatosplenomegaly, and impairment of the liver focused. Although IHS has multiple etiologies, hepatic fibrosis is one of the main pathologic features. Histology is the gold standard for assessing the degree of hepatic fibrosis and for estimating prognosis[2-6]; however, there are several limitations in the usage of liver biopsy in general clinical practice due to the invasive nature of the examination, especially in infant age group. So there is a need for a non-invasive examination, which is clinically useful and accurately reproducible.
In adults, two-dimensional ultrasonography (2DUS)[2-5,7,8], Doppler blood stream[7-10,11-14,26], and determi-nation of serum fibrosis markers[15-19,21-23,25,26] have been performed widely for the noninvasive diagnosis of hepatic fibrosis and therapeutic surveillance[20,24], and they were also useful in the assessment of cirrhosis in the pediatric age group[27-30]. But until recently, only a few studies[31,32] focussed on infants, especially on infants with IHS.
This prospective study investigated the association between the hepatic fibrosis in infants with IHS and hepatic hemodynamic parameters and serum fibrosis markers, correlated the results with the histological findings, and evaluated the clinical relevance of the noninvasive methods.
MATERIALS AND METHODS
Patients
Forty-one infants (27 boys and 14 girls; mean age 5.2 ± 3.6 mo) who were admitted in the Department of pediatrics in our hospital from January 2000 to December 2003 were included. The patients were diagnosed as infant hepatitis syndrome (IHS) according to the diagnosis criteria of the national infant virus hepatitis prevention and cure symposium[1]. Infants presented with jaundice, hepatomegaly or hepatosplenomegaly, and impairment of the liver function. They didn’t have any contraindication for liver biopsy. Those with clinically overt heart failure were excluded. Etiologies included viral infection, metabolic disorders, familial cholestasis, or idiopathic neonatal hepatitis. Etiologies were categorized on the basis of their clinic and pathologic diagnosis (Table 1). Forty-six infants (34 boys and 12 girls; mean age 4.6 ± 3.6 mo) who presented with normal liver size and function, negative HBV status, and without any history of clinical signs of liver disease served as healthy controls.
Table 1.
Etiology of IHS
| Etiology | n | % |
| Cytomegalovirus hepatitis (CMVH) | 18 | 43.90 |
| CMVH + Glucose-6-phosphate dehydrogenase | 3 | 7.32 |
| CMVH + congenital choledochocele | 2 | 4.88 |
| CMVH + intrahepatic biliary atresia | 1 | 2.44 |
| CMVH + hemangioma | 1 | 2.44 |
| Glycogen storage disease | 5 | 12.20 |
| Infant Hepatitis Syndrome (undefined) | 8 | 19.51 |
| Congenital deformity of biliary tract (Extrahepatic biliary atresia) | 1 | 2.44 |
| Congenital choledochocele | 1 | 2.44 |
| Drug-induced hepatitis | 1 | 2.44 |
| Total | 41 | 100.00 |
Ultrasonography
Ultrasonography was performed on all children (healthy and those with IHS) by two physicians, who were unaware of the results of the liver biopsy and any laboratory finding at the time of the examinations. All children were held in the arms of their mothers and studied after a fasting period of 2-4 h. Some of them were examined during normal sleep, some were helped by use of a sugar nipple, if necessary, and some were given a sedative drug before the examination. GE-LOGIQ9 ultrasound scanner (GE, Milwaukee, and American) equipped with an 8-MHz electric curved scanning transducer or HP 8500 ultrasound scanner (HP, Andover, American) with a 7.5-MHz electric curved scanning transducer were used for the examination. Evaluation of liver parenchyma includes the assessment of the size, configuration, homogeneity, contour, and the vascular structure. Spleen and visibility of the gallbladder were also evaluated and compared with normal data. Color Flow Mapping(CFM) was used to determine the direction of flow of the hepatic blood vessels, and Pulse Wave Doppler (PW) was used to measure the Doppler waveforms of the PHA, portal vein (PV), and hepatic vein (HV). During the PW examination, the angle between the Doppler beam and longitudinal axis of the blood vessels was kept at 30°, and Doppler sample volume was smaller than the inner diameter of vessels. In each individual, the Doppler waveforms of the PHA, PV, and HV were measured. Doppler waveform analysis was calculated from 3-5 consecutive waveforms on the Doppler tracing in each subject. Flow in the middle hepatic vein (MHV) was evaluated at subcostal or intercostals, a distance of at least 2 cm from the inferior vena cava (IVC). This distance was important to rule out the possibility that the MHV flow pattern was influenced more by the flow pattern of the IVC than by the surrounding liver parenchyma.The waveforms of the MHV were classified into 3 types[11,14]: Type 1 (HV0), regular triphasic waveforms (with a short phase of reversed flow) which were considered to be normal flow, whereas type 2 (HV1), biphasic waveforms (without reversed flow) and type 3 (HV2), flat waveforms (with continuous blood flow) were considered to be abnormal.
Laboratory tests
Serum total bilirubin (T-Bil), indirect bilirubin (I-Bil), direct bilirubin (D-Bil), aspartate aminotransferase (AST), alanine aminotransferase (ALT), γ-glutamyl transpeptidase (γ-GTP), alkaline phosphatase (AKP), total protein, albumin, globulin, the ratio of albumin to globulin (A/G), and prothrombin time were measured by routine methods. The etiologic differential diagnosis was made according to the presence of serological markers for viral hepatitis type B, toxoplasma, rubella, cytomegalovirus, herpes simplex, and syphilis (TORCHS) (positive IgM or more than a fourfold rise in IgG titer), and urine cytomegalovirus (CMV) culture. The serum specimens for fibrosis indices, including HA, PC-III, C-IV and LN, were determined by commercially available radioimmunoassays (RIA kits; Shanghai Navy Medical Institution, Shanghai, China). Samples of peripheral venous blood of the subjects were collected, immediately separated, and preserved at -40°C until they could be assayed.
Liver biopsy and histological analysis
Liver biopsy was performed in 41 children with IHS, 35 cases underwent US-guidance liver biopsies and 6 cases underwent operation biopsies. Intercostals or subcostal ultrasound-guided liver biopsies were performed during the scanning with an 18G biopsy-cut (CR Bard, Inc., Covington, GA, USA) needle driven by the spring-loaded automatic biopsy gun (CR Bard, Inc., MAGNER, USA). The Bard biopsy gun consists of a hand-held device that triggers rapid firing of an 18G cutting needle. When the gun is fired, an inner trocar with its 1.9 cm sample notch thrusts forward, followed by its outer cannula which shears a core of tissue with minimum crushing of the specimen. For a satisfactory sampling, an average of two specimens was obtained in each ultrasound-guided core-needle biopsy case. All biopsy specimens were 0.8-1.9 cm in length and were fixed in 10% buffered formalin. Sections were stained with hematoxylin-eosin; Masson’s trichrome stain was used specially for staining of fibrous tissue components.
The stage of hepatic fibrosis was scored according to the diagnostic criteria of viral hepatitis in Xian, China, 2000[33]. S0 corresponds to the absence of fibrosis, S1 to fibrosis within portal tract areas, S2 to fibrosis around the portal tract areas with formation of fibrosis segregation while maintain lobular structure, S3 to the formation of fibrosis segregation and disorder of lobular structure without hepatic cirrhosis, and S4 to early or late stage cirrhosis. Then these patients were divided into three groups: mild (S1-S2 grade), moderate (S3 grade) and severe (S4 grade).
Statistical analysis
The one-way ANOVA was used to compare numerical variables between multiple groups. The chi-square test and Fisher exact test were used to compare categorical variables. Association between the ranked values was evaluated using Spearman rank correlation. P < 0.05 was considered statistically significant. All statistical analyses were done with SPSS for windows, version 10.0 (SPSS Inc. American).
RESULTS
In the IHS group, there was an increase in T-Bil which reached up to 810 μmol/L. D-Bil increased in almost all except for 5 children (I-Bil was higher than D-Bil). There was an increase in serum ALT activity in all cases, the highest value was 598 U/L, mean 126.9 U/L. Serum AST activity increased in all except for 3 cases, and the highest value was 2816 U/L, mean 282.4 U/L. The AST/ALT ratio was 3.5 (normal 1.15), the ratio of albumin to globulin (A/G) decreased in 29 cases, and inversed in 5 cases. Pathology showed liver fibrosis in 39 cases (95.1%), 12 cases with S1 or S2, 12 cases with S3, and 15 cases with S4. Further findings were ballooning change (n = 35, 85.4%), hepatocyte necrosis (n = 30, 73.2%), cholestasis in liver cells (n = 26, 63.4%), cholestasis in the hepatic lobules (n = 21, 51.2%), cholestasis in portal area (n = 7, 17.1%), and cytomorphosis of giant corpuscle (n = 20, 48.8%).
2DUS findings
In the 39 cases of liver fibrosis, 15 cases with S4 stage liver fibrosis (confirmed by histological diagnosis) showed that the liver parenchyma was more echogenic and coarse than normal, among which 6 severe cases with obvious echogenic changes showed nodularity of the liver edge and ascites by two-dimensional ultrasonography (2DUS). The other 24 cases with S1-S3 stage liver fibrosis (confirmed by histological diagnosis) were all hepatomegaly but the liver parenchyma was normal, except for 5 cases of Glycogen Storage Disease (1 case in S0 stage, 2 in S2 stage and 2 in S3 stage) presented with significant hepatomegaly, increased echogenicity, and slightly increased attenuation. Additionally, we found that proper hepatic arteries (PHA) in the IHS group were enlarged and even were easy to display on the 2DUS.
CDUS findings
We found that PHA in the IHS group can be displayed easily on the B-Mode, as well as on the Color Flow Mappings and even better (Figure 1A). PHA can be seen in 100% of patients in IHS group but only in 58.7% of the control subjects, there was significant difference between IHS group and control group (P < 0.001). And then, PW parameters showed an increase in PSV, EDV, and RI (Figure 1A). PSV and RI in IHS group were significantly higher than in the control group (P < 0.05), as well as, with development of hepatic fibrosis from mild to severe, PSV and RI of the PHA increase gradually (Figure 2A and 2C), and EDV of the PHA decreased gradually (Figure 2B), there was a significant difference between groups (P < 0.05). Furthermore, PW waveform of the MHV developed from HV1 (damped biphasic waveform without reversed flow) to HV2 (flat waveform) with the severity of the hepatic fibrosis (Table 2).
Figure 1.
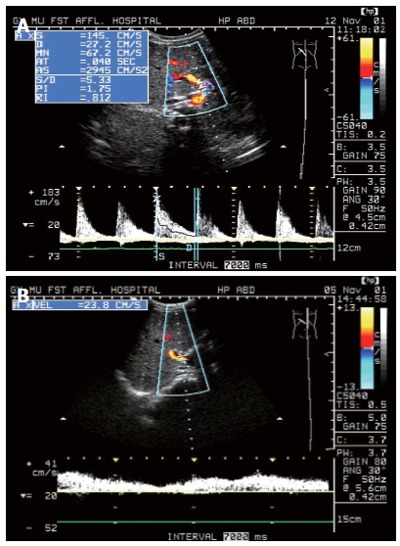
A: Duplex Doppler Sonogram of case with IHS showed the PHA was easy to see as well as PSV and RI of the PHA increased significantly; B: Duplex Doppler Sonogram of the normal subject showed normal waveform of PV, and PHA cannot display clearly.
Figure 2.
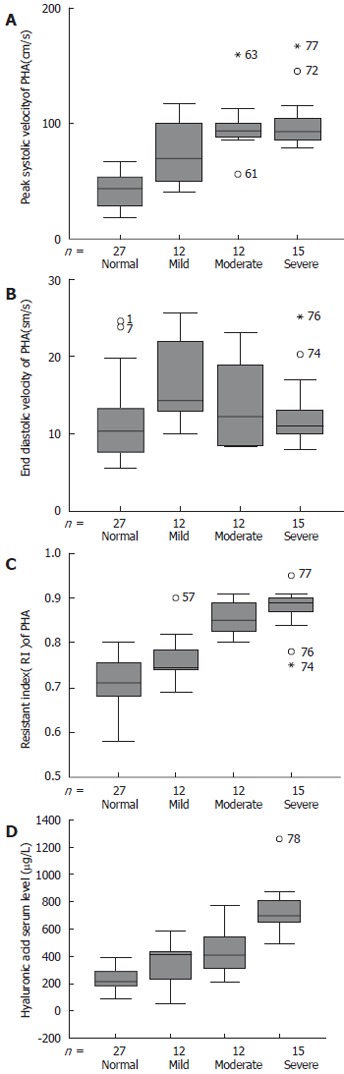
Boxplots showing A: Significantly higher PSV of PHA in patients with advanced liver fibrosis; B: Decreased gradually EDV of PHA in patients with advanced liver fibrosis; C: Significantly higher RI of PHA in patients with advanced liver fibrosis; D: Significantly higher serum HA level in patients with advanced liver fibrosis.
Table 2.
Doppler sonograms in hepatic veins in patients with different fibrosis stage of IHS n (%)
| Group | Patients (n) | HV0 | HV1 | HV2 | Total |
| Control group | 46 | 43 (93.50) | 3 (6.50) | 0 (0.00) | 46 (100) |
| Mild group | 12 | 2 (16.67) | 10 (83.33) | 0 (0.00) | 12 (100) |
| Moderate group | 12 | 1 (8.33) | 10 (83.4) | 1 (8.33) | 12 (100) |
| Severe group | 15 | 1 (6.67) | 10 (66.67) | 4 (26.67) | 15 (100) |
Chi-square test, χ2 = 37.67, P < 0.001.
Serum fibrosis index
Serum concentration of fibrosis markers was higher in the IHS than in the control group (P < 0.05). Especially serum HA increased gradually from the mild to the severe group along with the severity of the hepatic fibrosis (Figure 2D), and it was significantly higher in IHS group than in the control group (P < 0.05). For PC-III, C-IV and LN, there were also significant differences between infants with severe hepatic fibrosis and the control group (P < 0.05). When infants of the IHS group with mild or moderate fibrosis were compared to the healthy group, differences did not reach statistical significance (Table 3).
Table 3.
Serum fibrosis indexes in patients with different fibrosis stage of IHS (mean ± SD, μg/L)
| Group | Patients (n) | HA | PC- III | C IV | LN |
| Control group | 46 | 225.18 ± 80.58 | 432.32 ± 166.68 | 129.69 ± 33.08 | 141.63 ± 34.63 |
| Mild group | 12 | 352.11 ± 157.33a | 445.73 ± 191.11 | 149.85 ± 49.27 | 151.22 ± 36.57 |
| Moderate group | 12 | 447.48 ± 175.57b | 480.77 ± 217.15 | 141.91 ± 47.51 | 132.56 ± 41.29 |
| Severe group | 15 | 745.32 ± 231.12bd | 580.36 ± 147.79 a | 185.00 ± 45.82b | 167.90 ± 29.41a |
HA, Serum hyaluronic acid; PC- III: Pre-Collagen type III; C-IV: Collagen type IV; LN: Laminin.
P < 0.05, and
P < 0.01 vs control group;
P < 0.01, vs mild and moderate group.
Correlative analyses
We found close positive correlations between the serum HA and hepatic fibrosis (r = 0.712, P < 0.001), between PSV of PHA and hepatic fibrosis (r = 0.717, P = 0.001), and between RI of PHA and hepatic fibrosis (r = 0.745, P = 0.001). The changes of hepatic vein waveforms were also positively related to liver fibrosis (r = 0.783, P < 0.001). In contrast, liver size, the size of the spleen and hepatic capsule, the echogenicity of the liver parenchyma, the inner diameter of the PV, nor the velocity of the PV were found to be useful for the diagnosis of hepatic fibrosis in IHS patients.
DISCUSSION
The basic pathological changes of chronic liver disease in the adults have been recognized more accurately and profoundly[33-36]. According to a new pathological classification method which was carried out in the adults in our country[33], staging of hepatic fibrosis can help us to recognize the development of the hepatic fibrosis in infants with IHS in our study. In the IHS group, 39 cases (95.1%) showed to have hepatic fibrosis by pathology. Further findings were ballooning change (n = 35, 85.4%), hepatocyte necrosis (n = 30, 73.2%), cholestasis in liver cells (n = 26, 63.4%), cholestasis in the hepatic lobules (n = 21, 51.2%), cholestasis in portal area (n = 7, 17.1%), and cytomorphosis of giant corpuscle (n = 20, 48.8%) of. Our findings showed that hepatic fibrosis was also one of the main pathologic features in the patients of this age group. We found no significant changes between the mild and the moderate hepatic fibrosis in 2DUS examination, it is compatible with other reports in the literature about adults[2,3,5-8].
PSV and RI of the PHA were significantly higher in infants with IHS than in the control group (P < 0.05). This phenomenon might be due to shrunken intrahepatic blood vessel space and the correspondingly increased peripheral resistance of the artery microvasscular bed, which increased the PSV and RI. This might also be the result of the severity of diffuse liver parenchyma impairment, hepatocellular degeneration and necrosis, inflammatory cell infiltration and fibrous connective tissue development[9,10]. Our findings also showed that PSV, RI of the PHA increased gradually and EDV decreased gradually with advanced liver fibrosis, and there were significant differences between their groups (P < 0.05). PSV and RI of the PHA were positively correlated with the severity of liver fibrosis (r = 0.717 and 0.745, respectively P = 0.001). Hemodynamic changes of chronic liver disease in adults[7,9,10] and in the pediatric age group[29] have been described by other researchers. Some results of our study were inconsistent with the findings of Xue et al[10] whose work was focused on chronic liver disease and confirmed cirrhosis in adults. One point was EDV of PHA elevating in IHS group instead of declining, another was velocity of PHA increasing without deficiency of PV. These may be concerned with a relatively short course of disease,abundant hepatic blood flow,active liver fibrosis and inflammation,or vasodilatation induced by inflammatory substances in patients with IHS.
In our findings, the changes in the waveforms of the hepatic vein were positively correlated with the severity of liver fibrosis (r = 0.783, P < 0.001). Other groups described a damped waveform in the hepatic veins of patients with liver cirrhosis, and our results were consistent with the studies of Bolondi et al[14]. This may be due to increased parenchyma stiffness, which impaired the compliance of the hepatic veins and caused the waveforms to change from HV0 to HV1. With the development of the hepatic fibrosis, diffuse fibrosis can cause disorganization of lobular architecture. Therefore, the hepatic veins were stenosis by pressure of the fibrosis tissues, pseudo lobule and the hepatic capsule. Furthermore, a damped waveform (HV2) in the hepatic veins was associated with focal hepatic vein stenosis[11-14].
Many groups addressed serum indexes such as HA, PC-III, LN and C-IV, which reflected the stage of hepatic fibrosis. There is agreement that the four serum fibrosis indexes are relevant for the serodiagnosis of hepatic fibrosis, either alone[2,5,12,15,17-19,21,24,31,32] or in combination[4,16,20,22,23,25-28]. In our study, although we found some cases were inconsistent with pathological diagnosis, even with a high stage of hepatic fibrosis, the four serum fibrosis indexes were still around normal range, but statistical results indicated that the serum fibrosis index HA increased gradually from the mild to severe group as severity of liver fibrosis increased, and was significantly higher in each of fibrosis groups than in the control group. And then, the serum fibrosis index HA has a high positive correlation with the severity of hepatic fibrosis in IHS group. For the other serum fibrosis indices, PC-III, C-IV and LN, there was only a significant difference between the severe hepatic fibrosis group and control group. And PC-III, C-IV had a low positive correlation with the severity of hepatic fibrosis in IHS group. There was no correlation between LN and the severity of hepatic fibrosis in IHS group. One researcher’s result showed the rate of inconsistency between the four serum fibrosis indexes and stage of hepatic fibrosis: HA was 29.2%, C-IV 31.9%, PC-III 34.5% and LN 38.1%, the rate of consistency between serum HA and stage of hepatic fibrosis was the highest one, which was followed by C-IV. This suggested that among the four serum fibrosis indexes, HA was the most ideal index for the diagnosis of hepatic fibrosis[15]. Our results were also consistent with other reports in the literature[5,15-17,22]. Furthermore, infants less than one year old belong to a special group in the mostly active developing phase, the concentration of serum fibrosis indices was affected by many factors. In particular, serum PC-III is very high in neonates, but quickly decreases by one year of age showing a marked negative correlation with age[30].
Liver biopsy guided by ultrasound in infants in 35 cases were performed during the scanning with an 18 G biopsy-cut needle driven by spring-loaded automatic biopsy gun, satisfactory sampling were get in all cases without any complication. A level of serum total bilirubin ≤ 200 μmol/L( former 136 μmol/ L) was indicated for liver biopsy in IHS group. According to our results, we consider that in clinical application, ultrasound-guided liver biopsy is also a valuable technology, secure, rapid, with high achievement ratio and no complication in the infant age group.
Our findings showed that young age, short course of disease, incompatibility with age and degree of liver fibrosis were the features of IHS group, parameters such as serum HA, PSV, and RI of PHA as well as the changes in the waveforms of the hepatic veins were more sensitive and reliable indicators in evaluating hepatic fibrosis in patients with IHS. Combination of ultrasonic studies on the hepatic hemodynamics with the evaluation of serum HA may provide an indicator for revealing hepatic fibrosis in children with IHS. This may be a useful noninvasive method for the diagnosis and estimation of the prognosis of IHS.
ACKNOWLEDGMENTS
Qi-Ming Feng, Xiang-Hong Li and Yan-Ning Li for their statistical analysis.
Footnotes
Supported by the Bureau of Education, Guangxi Zhuang Autonomous Region, China, No. A9909
S- Editor Liu Y L- Editor Mihm S E- Editor Liu WF
References
- 1.Symposium of the national infant virus hepatitis prevention and cure. Zhonghua Erke Zazhi. 1982;20:120–126. [Google Scholar]
- 2.Chen Y, Wang BE, Jia JD, Qian LX, Wang TL, Chen MH, Chen GY, He W, Ding HG, Yin SS, et al. [Noninvasive evaluation of liver fibrosis in chronic hepatitis B patients] Zhonghua Ganzangbing Zazhi. 2003;11:354–357. [PubMed] [Google Scholar]
- 3.Lu LG, Zeng MD, Wan MB, Li CZ, Mao YM, Li JQ, Qiu DK, Cao AP, Ye J, Cai X, et al. Grading and staging of hepatic fibrosis, and its relationship with noninvasive diagnostic parameters. World J Gastroenterol. 2003;9:2574–2578. doi: 10.3748/wjg.v9.i11.2574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zheng RQ, Lv MD, Xie SB, Ren J, Su ZZ, Yao JL. Comparison of Ultrasonography with serology in evaluating fibrosis degrees and diagnosing liver cirrhosis. Zhonghua Chaosheng Yingxiangxue Zazhi. 2002;11:29–32. [Google Scholar]
- 5.Li C, Wan M, Zeng M, Su B, He Q, Lu L, Mao Y. [A preliminary study of the combination of noninvasive parameters in the diagnosis of liver fibrosis] Zhonghua Ganzangbing Zazhi. 2001;9:261–263. [PubMed] [Google Scholar]
- 6.Colli A, Fraquelli M, Andreoletti M, Marino B, Zuccoli E, Conte D. Severe liver fibrosis or cirrhosis: accuracy of US for detection--analysis of 300 cases. Radiology. 2003;227:89–94. doi: 10.1148/radiol.2272020193. [DOI] [PubMed] [Google Scholar]
- 7.Khan KN, Yamasaki M, Yamasaki K, Inoue O, Yatsuhashi H, Koga M, Yano M. Proposed abdominal sonographic staging to predict severity of liver diseases: analysis with peritoneoscopy and histology. Dig Dis Sci. 2000;45:554–564. doi: 10.1023/a:1005491018415. [DOI] [PubMed] [Google Scholar]
- 8.Aubé C, Oberti F, Korali N, Namour MA, Loisel D, Tanguy JY, Valsesia E, Pilette C, Rousselet MC, Bedossa P, et al. Ultrasonographic diagnosis of hepatic fibrosis or cirrhosis. J Hepatol. 1999;30:472–478. doi: 10.1016/s0168-8278(99)80107-x. [DOI] [PubMed] [Google Scholar]
- 9.Hirata M, Akbar SM, Horiike N, Onji M. Noninvasive diagnosis of the degree of hepatic fibrosis using ultrasonography in patients with chronic liver disease due to hepatitis C virus. Eur J Clin Invest. 2001;31:528–535. doi: 10.1046/j.1365-2362.2001.00840.x. [DOI] [PubMed] [Google Scholar]
- 10.Xue ES, Lin LW, Ye Z. Gao SD, He YW. Study on Intrahepatic Arterial Hemodynamics by Doppler Ultrasound. Zhongguo Chaosheng Yixue Zazhi. 1993;9:233–235. [Google Scholar]
- 11.von Herbay A, Frieling T, Häussinger D. Association between duplex Doppler sonographic flow pattern in right hepatic vein and various liver diseases. J Clin Ultrasound. 2001;29:25–30. doi: 10.1002/1097-0096(200101)29:1<25::aid-jcu4>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 12.Yin J, Yin L, Zhou XX, Lu B, Sheng MX, Wu P, Chen JL, He ZR. Diagnosis of for liver fibrosis by Doppler ultrasound assessment of hepatic veins compared with serological investigation of fibrosis parameter. Zhongguo Chaosheng Yixue Zazhi. 2000;16:291–294. [Google Scholar]
- 13.Colli A, Cocciolo M, Riva C, Martinez E, Prisco A, Pirola M, Bratina G. Abnormalities of Doppler waveform of the hepatic veins in patients with chronic liver disease: correlation with histologic findings. AJR Am J Roentgenol. 1994;162:833–837. doi: 10.2214/ajr.162.4.8141001. [DOI] [PubMed] [Google Scholar]
- 14.Bolondi L, Li Bassi S, Gaiani S, Zironi G, Benzi G, Santi V, Barbara L. Liver cirrhosis: changes of Doppler waveform of hepatic veins. Radiology. 1991;178:513–516. doi: 10.1148/radiology.178.2.1987617. [DOI] [PubMed] [Google Scholar]
- 15.Tao J, Peng HQ, Cai WM, Dong FQ, Weng HL, Liu RH. Influence factors of serum fibrosis markers in liver fibrosis. World J Gastroenterol. 2003;9:2497–2500. doi: 10.3748/wjg.v9.i11.2497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cai W, Zheng M, Weng H, Liu R. [Determination and significance of serum markers for fibrosis in patients with chronic hepatitis] Zhonghua Neike Zazhi. 2001;40:448–451. [PubMed] [Google Scholar]
- 17.Luo R, Yang S, Xie J, Zhao Z, He Y, Yao J. [Diagnostic value of five serum markers for liver fibrosis] Zhonghua Ganzangbing Zazhi. 2001;9:148–150. [PubMed] [Google Scholar]
- 18.McHutchison JG, Blatt LM, de Medina M, Craig JR, Conrad A, Schiff ER, Tong MJ. Measurement of serum hyaluronic acid in patients with chronic hepatitis C and its relationship to liver histology. Consensus Interferon Study Group. J Gastroenterol Hepatol. 2000;15:945–951. doi: 10.1046/j.1440-1746.2000.02233.x. [DOI] [PubMed] [Google Scholar]
- 19.Plevris JN, Haydon GH, Simpson KJ, Dawkes R, Ludlum CA, Harrison DJ, Hayes PC. Serum hyaluronan--a non-invasive test for diagnosing liver cirrhosis. Eur J Gastroenterol Hepatol. 2000;12:1121–1127. doi: 10.1097/00042737-200012100-00009. [DOI] [PubMed] [Google Scholar]
- 20.Fabris P, Marranconi F, Bozzola L, Biasin MR, De Lazzari F, Plebani M, Benedetti P, Tositti G, Pellizzer G, Stecca C, et al. Fibrogenesis serum markers in patients with chronic hepatitis C treated with alpha-IFN. J Gastroenterol. 1999;34:345–350. doi: 10.1007/s005350050272. [DOI] [PubMed] [Google Scholar]
- 21.Pontinha N, Pessegueiro H, Barros H. Serum hyaluronan as a marker of liver fibrosis in asymptomatic chronic viral hepatitis B. Scand J Clin Lab Invest. 1999;59:343–347. doi: 10.1080/00365519950185535. [DOI] [PubMed] [Google Scholar]
- 22.Gu S, Zhang H, Zhang L. [Relationship between serum fibrosis markers and fibrosis quantitative analysis of liver tissue] Zhonghua Ganzangbing Zazhi. 1999;7:199–200. [PubMed] [Google Scholar]
- 23.Zheng M, Cai WM, Weng HL, Liu RH. ROC curves in evaluation of serum fibrosis indices for hepatic fibrosis. World J Gastroenterol. 2002;8:1073–1076. doi: 10.3748/wjg.v8.i6.1073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ninomiya T, Yoon S, Hayashi Y, Sugano M, Kumon Y, Seo Y, Shimizu K, Kasuga M. Clinical significance of serum hyaluronic acid as a fibrosis marker in chronic hepatitis C patients treated with interferon-alpha: histological evaluation by a modified histological activity index scoring system. J Gastroenterol Hepatol. 1998;13:68–74. doi: 10.1111/j.1440-1746.1998.tb00548.x. [DOI] [PubMed] [Google Scholar]
- 25.Pilette C, Rousselet MC, Bedossa P, Chappard D, Oberti F, Rifflet H, Maïga MY, Gallois Y, Calès P. Histopathological evaluation of liver fibrosis: quantitative image analysis vs semi-quantitative scores. Comparison with serum markers. J Hepatol. 1998;28:439–446. doi: 10.1016/s0168-8278(98)80318-8. [DOI] [PubMed] [Google Scholar]
- 26.Oberti F, Valsesia E, Pilette C, Rousselet MC, Bedossa P, Aubé C, Gallois Y, Rifflet H, Maïga MY, Penneau-Fontbonne D, et al. Noninvasive diagnosis of hepatic fibrosis or cirrhosis. Gastroenterology. 1997;113:1609–1616. doi: 10.1053/gast.1997.v113.pm9352863. [DOI] [PubMed] [Google Scholar]
- 27.Xu HG, Fang JP, Huang SL, Li HG, Zhong FY, Guo HX, Su H. [Diagnostic values of serum levels of HA, PC III, C IV and LN to the liver fibrosis in children with beta-thalassemia major] Zhonghua Erke Zazhi. 2003;41:603–606. [PubMed] [Google Scholar]
- 28.Jiang DM, Zhou JQ, Zhang XL, Sun M. Determination and clinical significance of four serum markers of liver fibrosis in children with chronic hepatitis. Zhonghua Erke Zazhi. 2001;39:465–468. [Google Scholar]
- 29.Tüney D, Aribal ME, Ertem D, Kotiloğlu E, Pehlivanoğlu E. Diagnosis of liver cirrhosis in children based on colour Doppler ultrasonography with histopathological correlation. Pediatr Radiol. 1998;28:859–864. doi: 10.1007/s002470050483. [DOI] [PubMed] [Google Scholar]
- 30.Vocke AK, Kardorff R, Ehrich JH. Sonographic measurements of the portal vein and its intrahepatic branches in children. Eur J Ultrasound. 1998;7:121–127. doi: 10.1016/s0929-8266(98)00016-0. [DOI] [PubMed] [Google Scholar]
- 31.Kobayashi H, Horikoshi K, Yamataka A, Yamataka T, Okazaki T, Lane GJ, Miyano T. Hyaluronic acid: a specific prognostic indicator of hepatic damage in biliary atresia. J Pediatr Surg. 1999;34:1791–1794. doi: 10.1016/s0022-3468(99)90314-7. [DOI] [PubMed] [Google Scholar]
- 32.Xiong Y, Wang SY. The characteristic of serum procollagen type III in healthy children. Zhonghua Erke Zazhi. 1998;36:682–683. [Google Scholar]
- 33.Chinese Society of Infectious disease and Parasitology and Chinese Society of Hepatology of Chinese medical association. The programme of prevention and cure for viral hepatitis. Zhonghua Ganzangbing Zazhi. 2000;8:324–329. [Google Scholar]
- 34.Wang TL, Liu X, Zhou YP, He JW, Zhang J, Li NZ, Duan ZP, Wang BE. A semiquantitative scoring system for evaluation of hepatic inflammation and fibrosis in chronic viral hepatitis. Zhonghua Ganzangbing Zazhi. 1998;6:195–197. [Google Scholar]
- 35.Chevallier M, Guerret S, Chossegros P, Gerard F, Grimaud JA. A histological semiquantitative scoring system for evaluation of hepatic fibrosis in needle liver biopsy specimens: comparison with morphometric studies. Hepatology. 1994;20:349–355. [PubMed] [Google Scholar]
- 36.Scheuer PJ. Classification of chronic viral hepatitis: a need for reassessment. J Hepatol. 1991;13:372–374. doi: 10.1016/0168-8278(91)90084-o. [DOI] [PubMed] [Google Scholar]


