Abstract
AIM: To evaluate the prognostic significance of p27kip1 in colorectal cancer patients.
METHODS: Cytoplasmic and nuclear p27kip1 expression was evaluated in 418 colorectal cancers using tissue microarrays. Data were associated with known patient and tumor variables and long-term patient outcomes, providing further insight into the mechanisms by which p27kip1 may influence tumor development.
RESULTS: Nuclear and cytoplasmic p27Kip1 expressions were detected in 59% and 19% of tumors respectively. Cytoplasmic p27Kip1 was almost invariably associated with positive nuclear p27Kip1 expression. Neither case correlated with known clinical or pathological variables, including tumor stage, grade or extramural vascular invasion. Furthermore, nuclear p27kip1 expression had no impact on survival. However, we identified a significant correlation between expression of cytoplasmic p27kip1 and longer disease-specific survival times. On multivariate analysis, TNM stage and extramural vascular invasion were highly significant independent prognostic factors, with positive cytoplasmic p27 expression showing a trend towards improved patient survival (P = 0.059).
CONCLUSION: These findings support the recent evidence that cytoplasmic p27kip1 has a distinct and important biological role that can influence tumor outcome.
Keywords: Colorectal cancer, Tissue microarray, p27Kip1, Prognostic factor
INTRODUCTION
p27, also known as Kip1, is expressed in most cells and its role is to bind and inhibit cyclin/cyclin-dependent kinase (cyclin-CDK) complexes, thereby inhibiting cell cycle progression[1]. This role in the cell cycle makes p27kip1 a key player in multiple cell fate decisions including proliferation, motility, differentiation and apoptosis. Numerous studies have shown that p27kip1 is a tumor suppressor gene whose loss co-operates with mutations in several oncogenes and tumor suppressor genes in order to facilitate tumor growth. p27kip1 is rarely mutated, and is predominantly regulated at the post-transcriptional level by degradation in the ubiquitin-proteasome pathway[2]. However, in contrast to most tumor suppressor genes, which are recessive, tumor suppression by p27kip1 appears critically dependent upon on the absolute level of p27kip1[3]. Several clinical studies have correlated absent or low p27kip1 expression with a poor prognosis in a range of malignancies, including breast, colorectal, gastric, ovary, prostate, bladder and oesophageal tumors[4-11]. Other investigators have failed to reproduce, or have only partially confirmed, the previously published results[12-14]. Some controversy also exists over the importance of cytoplasmic expression of p27kip1, which was originally thought to represent a mechanism for inactivating p27kip1 by sequestering it away from its site of action within the nucleus[15]. Cytoplasmic expression of p27kip1 has been identified in colorectal cancer, ovarian cancer and Barrett’s oesophagus[6,16-18], and recent evidence suggests that it may have an active role, as it has been shown to bind to RhoA and inhibit its activity[19]. Members of the Rho family include Cdc42, Rac and RhoA. These act as molecular switches in signalling pathways affecting gene transcription and cytoskeletal rearrangements. In particular, the Rho proteins regulate and co-ordinate the cytoskeletal remodelling that underlies changes in cell adhesion and migration[20]. Consistent with these studies, p27’s ability to regulate motility is distinct from its ability to inhibit cellular proliferation. Indeed, the region of p27kip1 that binds to RhoA and is required for migration is different from the region that binds cyclin-CDKs[21].
Using high-throughput Tissue Microarray (TMA) technology[22] we have investigated the expression of both cytoplasmic and nuclear p27kip1 in a series of over 400 paraffin wax-embedded colorectal tumor specimens. Data derived from this analysis was then associated with known patient and tumor variables, and with long-term patient outcome data, in order to gain further insight into the mechanisms by which p27 may influence tumor development.
MATERIALS AND METHODS
Patients
Four hundred and sixty-two patients were included in this study. A detailed description of these cases has been provided previously[23,24]. Briefly, all patients included in the TMA underwent elective resection of a histologically proven primary colorectal cancer between 1st January 1993 and 31st December 2000 at the University Hospital Nottingham, with prospective collection of associated clinical and pathological data including tumor site, TNM stage, histological tumor type and grade, and the presence of extramural vascular invasion. Histological factors more recently identified as having potential prognostic value in colorectal cancer, such as tumor budding and the tumor border configuration, were not recorded routinely at our institution and hence were not considered in the data analysis. Patients with lymph node positive disease were characteristically treated with adjuvant chemotherapy, consisting of 5-flurouracil and folinic acid. The UK Office for National Statistics has provided comprehensive follow-up regarding the date and cause of death for this cohort of patients. The length of follow-up was determined from the date of primary tumor resection, with surviving cases censored for analysis on the 31st December 2003. Disease specific survival was used as the primary end-point of the study.
Immunohistochemical procedures
Arrayed tumors were analysed for the expression of p27kip1 using a murine monoclonal anti-human p27kip1 antibody (clone SX53G8; Dako Ltd, Ely, UK) and standard immunohistochemistry with an avidin-biotin/horseradish peroxidase development system. Five microns formalin-fixed, wax-embedded tissue array sections were dewaxed in an incubator for 20 min at 60°C, deparaffinised with xylene, rehydrated through graded alcohol and immersed in 0.3% hydrogen peroxide-methanol solution to block endogenous peroxidase activity. Antigen retrieval was achieved by microwaving slides for 20 min in pH 9.0 EDTA buffer. Endogenous avidin/biotin activity was blocked using an avidin/biotin blocking kit (Vector Labs, USA). One hundred microliters of normal swine serum (NSS) was added to the sections for 10 min to block non-specific binding of the primary antibody. 100 μL of anti-p27kip1 antibody diluted 1:25 (v/v) in NSS/TBS was then applied to the test sections for 1hr at room temperature. Positive controls consisted of multi-tissue sections containing human kidney and tonsil, with omission of the primary antibody from negative control sections. After washing with TBS, sections were incubated with 100 μL of biotinylated goat anti-mouse/rabbit immunoglobulin (Dako Ltd, Ely, UK) diluted 1:100 in NSS for 30 min. One hundred microliters of pre-formed streptavidin-biotin/horseradish peroxidase (HRP) complex (Dako Ltd, Ely, UK) was then applied for 60 min at room temperature. Finally, bound antibody visualisation was accomplished using 3, 3’-Diaminobenzidine tetrahydrochloride (Dab, Dako Ltd, Ely, UK).
Evaluation of immunohistochemical staining
Immunohistochemical staining patterns were interpreted by two observers (DSGS and NFSW) blinded to the associated clinicopathological data. Nuclear p27kip1 expression was scored as follows: cases with < 10% tumor cell nuclei stained = 0, 10%-20% tumor cell nuclei stained = 1 and > 20% tumor cell nuclei stained = 2, irrespective of the staining intensity. Cytoplasmic staining was scored: complete absence of cytoplasmic staining or cytoplasmic staining of any intensity in < 10% tumor cells = 0, weak/moderate intensity cytoplasmic staining in > 10% tumor cells = 1, and intense cytoplasmic staining in > 10% tumor cells = 2. These cutoffs were based on previously published reports investigating the prognostic significance of p27Kip1 expression in colorectal tumors[16,25]. For the purposes of statistical analysis, these scores were re-categorized, with tumors considered either positive (score 1 or 2) or negative (score 0) for both nuclear and cytoplasmic p27kip1 expression. It has previously been established that in colorectal cancers, no significant differences in p27 immunoreactivity are usually seen between the centre and the invasive edge of the tumors[26].
Statistical analysis
All calculations were performed using SPSS software (version 11.5 for Windows, SPSS Inc., Chicago, IL). Associations between categorical variables were examined using crosstabulation and the Pearson chi-square test. Kaplan-Meier curves were plotted in order to assess correlations with disease-specific survival and the significance of differences in disease-specific survival between groups was calculated using the log-rank test. Patients whose deaths related to their colorectal cancer, including early deaths from post-operative complications, were considered in the disease-specific survival calculations. Deaths resulting from non-colorectal cancer related causes were censored at the time of death. Multivariate analysis was conducted using the Cox proportional-hazards model to determine hazard ratios, and to identify variables with independent prognostic significance in this cohort. In all cases P values < 0.05 were considered statistically significant.
RESULTS
Clinicopathological data
The characteristics of the 462 patients included in this study are summarized in Table 1. Male patients comprised 58% of the cohort, and the median patient age at the time of surgery was 72 years. At the time of censoring for data analysis 49% of patients had died from their colorectal cancer, with a further 14% deceased from non-colorectal cancer causes. The remaining 37% were still alive. The majority of tumors were of a moderately differentiated histological grade (77%). Similarly, 85% of tumors were adenocarcinomas, with a further 11% showing mucinous differentiation. Surviving patients had a median length follow-up of 75 (range 36-116) mo. Of the conventional clinicopathological variables, strong correlations were observed between tumor stage and disease specific survival (DSS) (Log rank = 207.33, P < 0.001) and between the presence of extramural vascular invasion and DSS (Log rank = 44.30, P < 0.001).
Table 1.
Patient and tumor characteristics (n = 462)
| Variable | Category | n (%) |
| Age (yr) | Median Range | 72 57-89 |
| Gender | Male Female | 266 (58) 196 (42) |
| Status | Alive Dead (cancer related) Dead (unrelated causes) Unknown | 169 (37) 228 (49) 64 (14) 1 |
| Histological type | Adenocarcinoma Adenocarcinoma with Mucinous differentiation Adenocarcinoma with Signet ring differentiation Other Unknown | 392 (85) 51 (11) 7 (1) 4 (1) 8 (2) |
| Histological grade | Well differentiated Moderately differentiated Poorly differentiated Unknown | 29 (6) 353 (77) 71 (15) 9 (2) |
| Tumor site | Colon Rectal Unknown | 238 (52) 181 (39) 43 (9) |
| TNM stage | 0 (Tis) 1 2 3 4 Unknown | 3 (1) 69 (15) 174 (38) 155 (33) 54 (12) 7 (2) |
| Extramural vascular invasion | Negative Positive Unknown | 224 (48) 128 (28) 110 (24) |
Of the 462 tumor specimens analyzed, 44 (9.5%) were subsequently uninterpretable due to loss of tissue from the TMA during the Immunohistochemical procedure. Among the remaining 418 tumors, three distinct patterns of p27kip1 expression were observed (Figure 1A-1C). These comprised staining of the tumor cell nuclei alone, staining of both tumor cell nuclei and cytoplasm, and absent staining of tumor elements. In contrast, no tumor cell membrane or stromal expression of p27kip1 was detected in any specimen.
Figure 1.
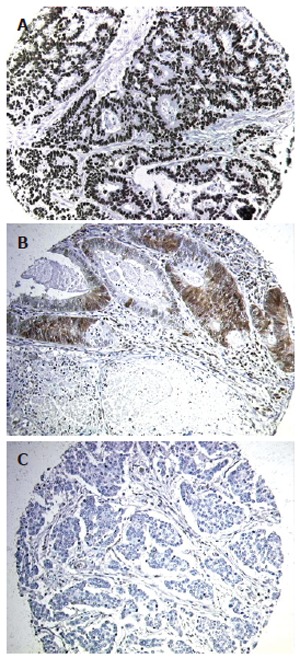
p27Kip1 expression in representative tumor samples. Tumors displayed either nuclear p27Kip1 alone (A), both nuclear and cytoplasmic p27Kip1 (B), or absent p27Kip1 expression (C).
Nuclear p27kip1 expression
Variable expression of nuclear p27kip1 was observed in 217/418 (51.9%) tumors (Table 2). In 29 of these cases nuclear p27kip1 was present in 10%-20% of tumor cells within the core, and in the remainder, nuclear p27kip1 was present in > 20% of tumor cells. There were no significant associations detected between any level of nuclear p27kip1 expression and known clinicopathological variables, including tumor grade, stage and the presence of extramural vascular invasion. Furthermore, on Kaplan-Meier analysis, no association was found between nuclear p27kip1 expression and DSS (Figure 2A, Log rank = 1.701, P = 0.1815).
Table 2.
p27kip1 localisation within 418 colorectal tumor specimens
| p27 localisation | Positive n (%) | Negative n (%) | Missing n (%) |
| Nuclear | 217 (51.9) | 201 (48.1) | 44 (9.5) |
| Cytoplasmic | 79 (18.9) | 339 (81.1) | 44 (9.5) |
Figure 2.
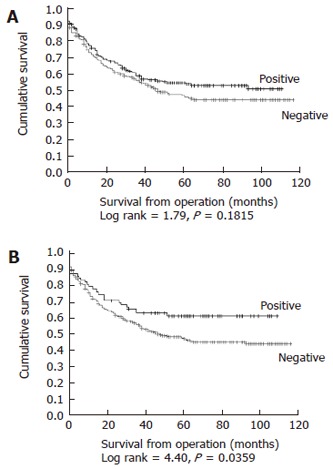
Kaplan-Meier plots for disease specific survival in relation to expression of nuclear (A) and cytoplasmic (B) p27Kip1.
Cytoplasmic p27kip1 expression
Expression of cytoplasmic p27kip1 was detected in 79/418 (18.9%) tumors (Table 2). These tumors appeared to form a distinct subset of the nuclear p27kip1 positive tumors, as co-expression of nuclear and cytoplasmic p27kip1 was noted in 74/79 cases (93.6%). In 55 of the cytoplasmic p27kip1 positive tumors, expression was of weak or moderate intensity and in the remaining 24 cases strong expression was noted. As with nuclear p27kip1, no statistically significant associations were found between the presence of cytoplasmic p27kip1 within the tumor and clinicopathological variables for the patient cohort. However, a significant relationship was identified between the presence of cytoplasmic p27kip1 and DSS. Kaplan-Meier analysis revealed that patients with cytoplasmic p27(+) tumors, had a significantly longer mean DSS of 72 (95% CI 62-83) mo, compared with a mean DSS of 62 (95% CI 57-68) mo in patients with cytoplasmic p27kip1(-) tumors (Figure 2B, Log rank = 4.40, P = 0.0359).
Multivariate analysis
A multivariate analysis of factors influencing survival in 418 available cases was performed using the Cox proportional hazards model (Table 3). Of the conventional clinicopathological variables analysed, tumor stage (P < 0.001) and extramural vascular invasion status (P = 0.001) were demonstrated to confer independently significant prognostic information. In this model expression of cytoplasmic p27kip1 within the tumor samples was associated with a strong trend towards improved DSS (HR for death in cytoplasmic p27kip1 positive tumors = 0.681, 95% CI 0.458-1.015), which approached statistical significance (P = 0.059).
Table 3.
Multivariate analysis of Cytoplasmic p27kip expression in relation to known clinical and pathological variables (n = 418)
| Variable | Category | Hazard ratio (HR) | 95% CI for HR | P |
| Gender | Female | 1 | ||
| Male | 1.055 | 0.787-1.415 | 0.719 | |
| Patient age (yr) | < 65 | 1 | ||
| 65-79 | 1.262 | 0.843-1.889 | ||
| 80+ | 1.609 | 0.961-2.694 | 0.194 | |
| Tumor site | Colon | 1 | ||
| Rectal | 1.117 | 0.813-1.535 | ||
| Unknown | 1.452 | 0.870-2.423 | 0.353 | |
| Tumor histological type | Adenocarcinoma | 1 | ||
| Non-adenocarcinoma | 0.956 | 0.621-1.472 | 0.838 | |
| Tumor grade | Well differentiated | 1 | ||
| Moderately differentiated | 1.167 | 0.582-2.337 | ||
| Poorly differentiated | 1.139 | 0.528-2.457 | ||
| Unknown | 0.869 | 0.245-3.079 | 0.926 | |
| Vascular invasion | Negative | 1 | ||
| Positive | 1.884 | 1.344-2.641 | ||
| Unknown | 1.308 | 0.876-1.954 | 0.001 | |
| Tumor (TNM) stage | 0/I | 1 | ||
| II | 2.021 | 1.102-3.708 | ||
| III | 3.741 | 2.066-6.774 | ||
| IV | 16.977 | 8.829-32.645 | ||
| Unknown | 4.211 | 1.049-16.902 | < 0.001 | |
| Cytoplasmic p27Kip1 expression | Negative | 1 | ||
| Positive | 0.681 | 0.458-1.015 | 0.059 |
DISCUSSION
p27kip1 is a universal CDK inhibitor that acts in G0 and early G1 to inhibit cyclin E/CDK2 and thereby prevents entry into the S phase of the cell cycle. It can also bind other cyclin-CDK complexes, including those involving the D-cyclins, and complexes of cyclin A/cdk2. Mitogenic growth factor signalling causes a decrease of p27kip1 levels and/or activity and, conversely, p27kip1 levels and/or activity increase in response to differentiation signals, loss of adhesion to extracellular matrix, or signalling by growth-regulatory factors such as TGF-β, c-AMP and IFN-γ. Studies in animals have shown that loss of p27kip1 increases the formation of tumors and also increases tumor associated deaths rates. Several studies have shown that loss of nuclear p27kip1 is an independent predictor of poor prognosis in colorectal cancer[6,27,28], although other studies have failed to confirm this observation[29,30]. Loss of nuclear p27kip1 was observed in 48% of tumors in our study. However, this showed no significant associations with known clinical or pathological variables. Additionally, on Kaplan-Meier analysis, no association was found between nuclear p27kip1 expression and survival.
Several clinical studies have detected cytoplasmic expression of p27kip1, and found an inverse correlation with disease free survival[17,28,31]. In contrast, in this large study of 418 colorectal tumors, cytoplasmic expression of p27kip1 was significantly associated with a good prognosis. This may be related to the findings from recent studies showing that p27kip1 can bind and inactivate Rho proteins. For tumors to metastasise, cells must alter their connections to both their neighbours and their substrate, and then migrate. Efficient migration requires a tightly balanced activation and deactivation of Cdc42, Rac and RhoA in both time and space. Indeed, two modes of tumor cell motility have been described in 3D matrices. Rho signalling through ROCK promotes a rounded bleb-associated mode of motility that does not require pericellular proteolysis. In contrast, elongated cell motility is associated with Rac-dependent F-actin-rich protrusions and does not require Rho or ROCK[32]. Consistent with this observation are other studies that have shown inhibition of ROCK reduces the invasive behaviour of tumor cells in vivo[33,34]. Sequestrations of RhoA by cytoplamsic p27kip1 may inhibit RhoA, resulting in inhibition of ROCK and less aggressive tumors. In the current study, cytoplasmic expression of p27kip1 was almost invariably associated with nuclear expression. These tumors may therefore have relatively controlled cell proliferation together with a reduced capacity for migration, resulting in a less aggressive tumor and a good prognosis. In contrast, loss of both cytoplasmic and nuclear p27kip1 may result in uncontrolled proliferation and increased invasion, leading to an aggressive tumor and poor prognosis. Tumor cells expressing only nuclear p27kip1 would have reduced proliferation but may still be invasive, resulting in an intermediate prognosis. This may explain the inconsistency of previous studies linking p27kip1 expression with prognosis, as studies considering nuclear p27kip1 expression alone have not identified the subgroup of tumors with additional cytoplasmic p27kip1 expression. It is anticipated that the role of cytoplasmic p27kip1 in promoting tumor progression may also depend upon the site and mechanism of invasion, as in a study of 61 patients with pancreatic cancer in which those with exclusively nuclear p27kip1 expression were shown to have a better prognosis than those with both nuclear and cytoplasmic p27kip1 expression[31].
Recently, it has been suggested that the presence of high numbers of detached clusters of tumor cells in adjacent stroma (termed tumor budding) may serve as an adverse histopathological prognostic feature in colorectal cancer[35]. As this tumor budding is the morphological counterpart of epithelial-mesenchymal transition, micro-invasion, and acquisition of individual cell motility, it would be of interest to correlate p27 immuno-expression patterns with colorectal tumor budding activity. However, this would be best performed using conventional whole tissue section analysis rather than TMA’s. Similarly, as the size of this array does not provide us with the statistical power to co-analyze two rare events, we have not performed analysis of DNA mismatch repair gene status and therefore cannot comment on whether cytoplasmic p27 expression may have a diverse role in colorectal tumors of differing microsatelite instability status. Finally, although out of the scope of this manuscript, it would be of interest to compare disease specific survival between p27Kip1 -/- and wild-type mouse in a colorectal cancer mouse model.
The identification of novel molecular and genetic markers of prognosis will eventually allow us to provide a cancer patient with individually tailored therapy based upon on the specific molecular fingerprint of his or her tumor. Our findings in a large cohort of unselected patients with colorectal cancer suggest that cytoplasmic p27Kip1 expression deserves further consideration as a marker of prognosis, as patients with colorectal tumors showing cytoplasmic expression of p27kip1 appear to have a more favourable disease specific survival.
ACKNOWLEDGMENTS
We are grateful to Mr Rob Moss for his technical assistance and expert advice.
Footnotes
Supported by the Special Trustees of Nottingham Hospitals (grant No. STR 62/03/M)
S- Editor Liu Y L- Editor Lutze M E- Editor Liu WF
References
- 1.Lacy ER, Filippov I, Lewis WS, Otieno S, Xiao L, Weiss S, Hengst L, Kriwacki RW. p27 binds cyclin-CDK complexes through a sequential mechanism involving binding-induced protein folding. Nat Struct Mol Biol. 2004;11:358–364. doi: 10.1038/nsmb746. [DOI] [PubMed] [Google Scholar]
- 2.Pagano M, Tam SW, Theodoras AM, Beer-Romero P, Del Sal G, Chau V, Yew PR, Draetta GF, Rolfe M. Role of the ubiquitin-proteasome pathway in regulating abundance of the cyclin-dependent kinase inhibitor p27. Science. 1995;269:682–685. doi: 10.1126/science.7624798. [DOI] [PubMed] [Google Scholar]
- 3.Philipp-Staheli J, Payne SR, Kemp CJ. p27(Kip1): regulation and function of a haploinsufficient tumor suppressor and its misregulation in cancer. Exp Cell Res. 2001;264:148–168. doi: 10.1006/excr.2000.5143. [DOI] [PubMed] [Google Scholar]
- 4.Slingerland J, Pagano M. Regulation of the cdk inhibitor p27 and its deregulation in cancer. J Cell Physiol. 2000;183:10–17. doi: 10.1002/(SICI)1097-4652(200004)183:1<10::AID-JCP2>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 5.Lloyd RV, Erickson LA, Jin L, Kulig E, Qian X, Cheville JC, Scheithauer BW. p27kip1: a multifunctional cyclin-dependent kinase inhibitor with prognostic significance in human cancers. Am J Pathol. 1999;154:313–323. doi: 10.1016/S0002-9440(10)65277-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Loda M, Cukor B, Tam SW, Lavin P, Fiorentino M, Draetta GF, Jessup JM, Pagano M. Increased proteasome-dependent degradation of the cyclin-dependent kinase inhibitor p27 in aggressive colorectal carcinomas. Nat Med. 1997;3:231–234. doi: 10.1038/nm0297-231. [DOI] [PubMed] [Google Scholar]
- 7.Tan P, Cady B, Wanner M, Worland P, Cukor B, Magi-Galluzzi C, Lavin P, Draetta G, Pagano M, Loda M. The cell cycle inhibitor p27 is an independent prognostic marker in small (T1a,b) invasive breast carcinomas. Cancer Res. 1997;57:1259–1263. [PubMed] [Google Scholar]
- 8.Catzavelos C, Bhattacharya N, Ung YC, Wilson JA, Roncari L, Sandhu C, Shaw P, Yeger H, Morava-Protzner I, Kapusta L, et al. Decreased levels of the cell-cycle inhibitor p27Kip1 protein: prognostic implications in primary breast cancer. Nat Med. 1997;3:227–230. doi: 10.1038/nm0297-227. [DOI] [PubMed] [Google Scholar]
- 9.Masciullo V, Sgambato A, Pacilio C, Pucci B, Ferrandina G, Palazzo J, Carbone A, Cittadini A, Mancuso S, Scambia G, et al. Frequent loss of expression of the cyclin-dependent kinase inhibitor p27 in epithelial ovarian cancer. Cancer Res. 1999;59:3790–3794. [PubMed] [Google Scholar]
- 10.Wu J, Shen ZZ, Lu JS, Jiang M, Han QX, Fontana JA, Barsky SH, Shao ZM. Prognostic role of p27Kip1 and apoptosis in human breast cancer. Br J Cancer. 1999;79:1572–1578. doi: 10.1038/sj.bjc.6690250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tsuchiya A, Zhang GJ, Kanno M. Prognostic impact of cyclin-dependent kinase inhibitor p27kip1 in node-positive breast cancer. J Surg Oncol. 1999;70:230–234. doi: 10.1002/(sici)1096-9098(199904)70:4<230::aid-jso6>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 12.Gillett CE, Smith P, Peters G, Lu X, Barnes DM. Cyclin-dependent kinase inhibitor p27Kip1 expression and interaction with other cell cycle-associated proteins in mammary carcinoma. J Pathol. 1999;187:200–206. doi: 10.1002/(SICI)1096-9896(199901)187:2<200::AID-PATH228>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 13.Barbareschi M, van Tinteren H, Mauri FA, Veronese S, Peterse H, Maisonneuve P, Caffo O, Scaioli M, Doglioni C, Galligioni E, et al. p27(kip1) expression in breast carcinomas: an immunohistochemical study on 512 patients with long-term follow-up. Int J Cancer. 2000;89:236–241. [PubMed] [Google Scholar]
- 14.Leong AC, Hanby AM, Potts HW, Tan DS, Skilton D, Ryder K, Harris WH, Liebmann RD, Barnes DM, Gillett CE. Cell cycle proteins do not predict outcome in grade I infiltrating ductal carcinoma of the breast. Int J Cancer. 2000;89:26–31. doi: 10.1002/(sici)1097-0215(20000120)89:1<26::aid-ijc5>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- 15.Sgambato A, Ratto C, Faraglia B, Merico M, Ardito R, Schinzari G, Romano G, Cittadini AR. Reduced expression and altered subcellular localization of the cyclin-dependent kinase inhibitor p27(Kip1) in human colon cancer. Mol Carcinog. 1999;26:172–179. doi: 10.1002/(sici)1098-2744(199911)26:3<172::aid-mc6>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- 16.Zhang H, Sun XF. Loss of p27 expression predicts poor prognosis in patients with Dukes' B stage or proximal colorectal cancer. Int J Oncol. 2001;19:49–52. [PubMed] [Google Scholar]
- 17.Rosen DG, Yang G, Cai KQ, Bast RC Jr, Gershenson DM, Silva EG, Liu J. Subcellular localization of p27kip1 expression predicts poor prognosis in human ovarian cancer. Clin Cancer Res. 2005;11:632–637. [PubMed] [Google Scholar]
- 18.Singh SP, Lipman J, Goldman H, Ellis FH Jr, Aizenman L, Cangi MG, Signoretti S, Chiaur DS, Pagano M, Loda M. Loss or altered subcellular localization of p27 in Barrett's associated adenocarcinoma. Cancer Res. 1998;58:1730–1735. [PubMed] [Google Scholar]
- 19.Besson A, Gurian-West M, Schmidt A, Hall A, Roberts JM. p27Kip1 modulates cell migration through the regulation of RhoA activation. Genes Dev. 2004;18:862–876. doi: 10.1101/gad.1185504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ridley AJ, Schwartz MA, Burridge K, Firtel RA, Ginsberg MH, Borisy G, Parsons JT, Horwitz AR. Cell migration: integrating signals from front to back. Science. 2003;302:1704–1709. doi: 10.1126/science.1092053. [DOI] [PubMed] [Google Scholar]
- 21.McAllister SS, Becker-Hapak M, Pintucci G, Pagano M, Dowdy SF. Novel p27(kip1) C-terminal scatter domain mediates Rac-dependent cell migration independent of cell cycle arrest functions. Mol Cell Biol. 2003;23:216–228. doi: 10.1128/MCB.23.1.216-228.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kononen J, Bubendorf L, Kallioniemi A, Bärlund M, Schraml P, Leighton S, Torhorst J, Mihatsch MJ, Sauter G, Kallioniemi OP. Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med. 1998;4:844–847. doi: 10.1038/nm0798-844. [DOI] [PubMed] [Google Scholar]
- 23.Watson NF, Ramage JM, Madjd Z, Spendlove I, Ellis IO, Scholefield JH, Durrant LG. Immunosurveillance is active in colorectal cancer as downregulation but not complete loss of MHC class I expression correlates with a poor prognosis. Int J Cancer. 2006;118:6–10. doi: 10.1002/ijc.21303. [DOI] [PubMed] [Google Scholar]
- 24.Watson NF, Durrant LG, Madjd Z, Ellis IO, Scholefield JH, Spendlove I. Expression of the membrane complement regulatory protein CD59 (protectin) is associated with reduced survival in colorectal cancer patients. Cancer Immunol Immunother. 2006;55:973–980. doi: 10.1007/s00262-005-0055-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rosati G, Chiacchio R, Reggiardo G, De Sanctis D, Manzione L. Thymidylate synthase expression, p53, bcl-2, Ki-67 and p27 in colorectal cancer: relationships with tumor recurrence and survival. Tumour Biol. 2004;25:258–263. doi: 10.1159/000081389. [DOI] [PubMed] [Google Scholar]
- 26.Galizia G, Lieto E, Ferraraccio F, Orditura M, De Vita F, Castellano P, Imperatore V, Romano C, Ciardiello F, Agostini B, et al. Determination of molecular marker expression can predict clinical outcome in colon carcinomas. Clin Cancer Res. 2004;10:3490–3499. doi: 10.1158/1078-0432.CCR-0960-03. [DOI] [PubMed] [Google Scholar]
- 27.Palmqvist R, Stenling R, Oberg A, Landberg G. Prognostic significance of p27(Kip1) expression in colorectal cancer: a clinico-pathological characterization. J Pathol. 1999;188:18–23. doi: 10.1002/(SICI)1096-9896(199905)188:1<18::AID-PATH311>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- 28.Günther K, Jung A, Völker U, Meyer M, Brabletz T, Matzel KE, Reymond MA, Kirchner T, Hohenberger W. p27(kip1) expression in rectal cancer correlates with disease-free survival. J Surg Res. 2000;92:78–84. doi: 10.1006/jsre.2000.5871. [DOI] [PubMed] [Google Scholar]
- 29.Hoos A, Nissan A, Stojadinovic A, Shia J, Hedvat CV, Leung DH, Paty PB, Klimstra D, Cordon-Cardo C, Wong WD. Tissue microarray molecular profiling of early, node-negative adenocarcinoma of the rectum: a comprehensive analysis. Clin Cancer Res. 2002;8:3841–3849. [PubMed] [Google Scholar]
- 30.Li GQ, Zhang HF. Mad2 and p27 expression profiles in colorectal cancer and its clinical significance. World J Gastroenterol. 2004;10:3218–3220. doi: 10.3748/wjg.v10.i21.3218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fukumoto A, Ikeda N, Sho M, Tomoda K, Kanehiro H, Hisanaga M, Tsurui Y, Tsutsumi M, Kato JY, Nakajima Y. Prognostic significance of localized p27Kip1 and potential role of Jab1/CSN5 in pancreatic cancer. Oncol Rep. 2004;11:277–284. [PubMed] [Google Scholar]
- 32.Sahai E, Marshall CJ. Differing modes of tumour cell invasion have distinct requirements for Rho/ROCK signalling and extracellular proteolysis. Nat Cell Biol. 2003;5:711–719. doi: 10.1038/ncb1019. [DOI] [PubMed] [Google Scholar]
- 33.Itoh K, Yoshioka K, Akedo H, Uehata M, Ishizaki T, Narumiya S. An essential part for Rho-associated kinase in the transcellular invasion of tumor cells. Nat Med. 1999;5:221–225. doi: 10.1038/5587. [DOI] [PubMed] [Google Scholar]
- 34.Takamura M, Sakamoto M, Genda T, Ichida T, Asakura H, Hirohashi S. Inhibition of intrahepatic metastasis of human hepatocellular carcinoma by Rho-associated protein kinase inhibitor Y-27632. Hepatology. 2001;33:577–581. doi: 10.1053/jhep.2001.22652. [DOI] [PubMed] [Google Scholar]
- 35.Prall F, Nizze H, Barten M. Tumour budding as prognostic factor in stage I/II colorectal carcinoma. Histopathology. 2005;47:17–24. doi: 10.1111/j.1365-2559.2005.02161.x. [DOI] [PubMed] [Google Scholar]


