Summary
Background
Prostate cancer is one of the leading causes of death from malignant disease among men in the Western world. One strategy to decrease the risk of dying from this disease is screening with Prostate-Specific Antigen (PSA); however, the extent of benefit and harm with such screening is under continuous debate.
Methods
In December 1994, 20 000 men born 1930 to 1944, randomly sampled from the Population Register, were computer randomised in a 1:1 ratio to a screening group invited for biennial PSA testing or to a control group not invited. In each arm, 48 men were excluded from analysis due to either death or emigration before randomization date or prevalent prostate cancer. The primary endpoint was prostate cancer specific mortality analyzed according to the intention-to-screen principle. Men in the screening group were invited up to the upper age limit (median 69, range 67–71 years) and only men with elevated PSA were offered additional tests such as digital rectal examination and prostate biopsies. The study is still ongoing inviting men who have not yet reached the upper age limit. This is the first planned report on cumulative prostate cancer incidence and mortality calculated up to Dec 31 2008. This study is registered [as an International Standard Randomised Controlled Trial], number [ISRCTN49127736].
Findings
Among men randomised to screening 7578/9952 (76%) attended at least once (attendees). During a median follow-up of 14 years, 1138 men in the screening group and 718 in the control group were diagnosed with prostate cancer resulting in a cumulative incidence of prostate cancer of 12.7% in the screening arm and 8.2% in the control arm (hazard ratio 1.64; 95% confidence interval [CI] 1.50–1.80; p<0.0001). The absolute cumulative risk reduction of death from prostate cancer at 14 years was 0.40% (95% CI 0.17–0.64%), from 0.90% in the control group to 0.50% in the screening group. The incidence rate ratio for death from prostate cancer was 0.56 (95% CI 0.39–0.82; p=0.002) in the screening compared to the control group. The incidence rate ratio of attendees compared to the control group was 0.44 (95% CI 0.28–0.68; p=0.0002). Overall, 293 men needed to be invited for screening and 12 to be diagnosed to prevent one prostate cancer death.
Interpretation
The benefit of prostate cancer screening compares favourably to other cancer screening programs and in this study prostate cancer mortality was reduced almost by half over 14 years. However, the risk of over diagnosis is substantial and the number needed to treat is at least as high as in breast cancer screening.
Funding
The Swedish Cancer Society, the Swedish Research Council and the National Cancer Institute.
Introduction
The European Randomized Study of Screening for Prostate Cancer (ERSPC) compares a group of men invited for prostate cancer screening based on prostate-specific antigen (PSA) to a control group without any active intervention. In interim analyses based on a median follow-up of 9 years,1–2 men randomised to active screening had a statistically significant reduction in prostate cancer mortality, risk ratio (RR) 0.80 (95% CI 0.65 to 0.98, adjusted p = 0.04).1 The number of men needed to be screened (NNS) to prevent one death from prostate cancer was 1410 (or 1068 in men who actually underwent screening1), which is similar to that reported for breast and colorectal cancer screening.3–6 However, the number needed to treat (NNT) to prevent one death was notably high, 48. This might be explained by only 9 years’ follow-up or by screening resulting in the detection of a large proportion of indolent cancers.
These reports are important as they contribute the first “level 1” evidence that PSA-based prostate cancer screening can reduce prostate cancer mortality. An open question, however, is whether the modest benefit in reduced cancer mortality documented thus far outweighs the harms of over-detection. This issue is emphasized by the report from another large screening trial, the US-based Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial (PLCO), which found no difference in prostate cancer mortality between men randomised to screening and those in the control group at 11.5 years’ follow-up.7 Other randomised studies have either been too small8–9 or criticized for methodological problems.10–11
The Göteborg Randomised Prostate Cancer Screening Trial is a prospective, randomised trial planned and started in 1995 evaluating the effects of biennial PSA-based screening.
The trial is truly population-based, as individuals from the Population Register were randomised to screening or controls without prior information, which results in a more representative study than does randomization after informed consent. The study design allows for an analysis of both how a screening program will be accepted by the population and also its effectiveness in terms of prostate cancer mortality reduction on a population level. The trial was designed and initiated independently of the ERSPC, although it was subsequently agreed to include a subset of subjects in the ERSPC. According to the Ethical Committee approval from 1994 this study was planned to be analyzed after 15 years and the present report is the first publication from the Göteborg trial evaluating prostate cancer mortality.
Patients and Methods
The Ethical Review Committee at the University of Göteborg approved this study in 1994.
Randomisation and masking
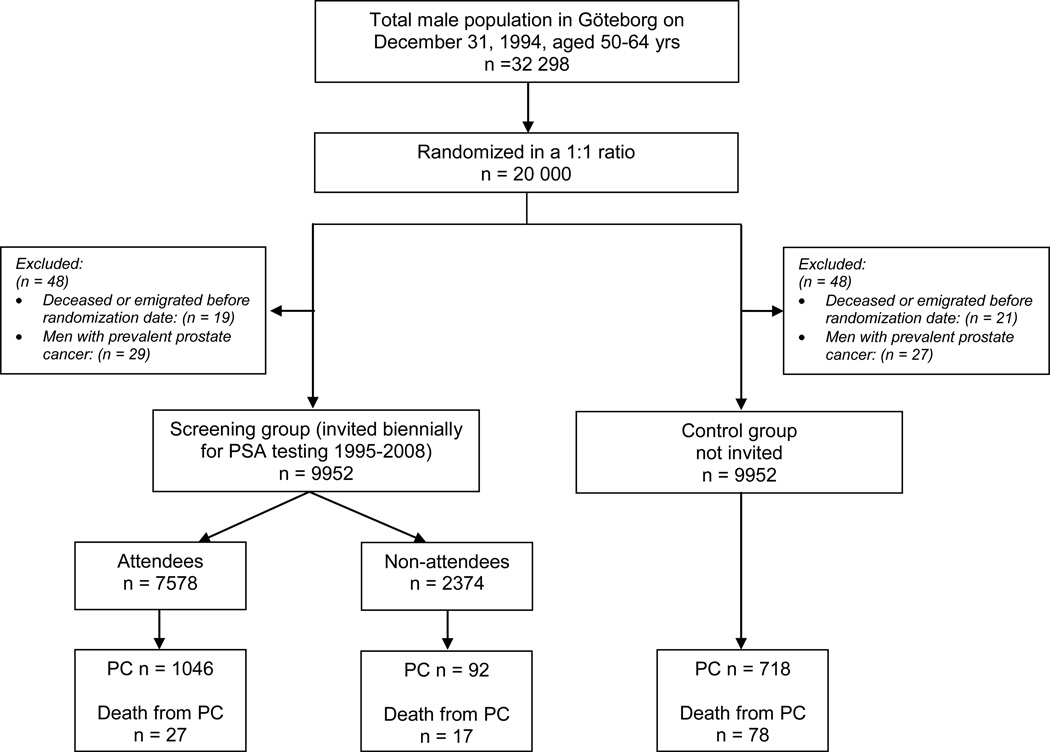
As of December 31, 1994 the Population Register documented 32,298 men born during 1930 through 1944 (ages 50–64, median 56 years) living in the city of Göteborg, Sweden. By computer randomisation 20,000 of these men were identified and allocated to either the intervention arm (screening group) or to a control group. The number of men in each birth cohort (1930–34, 1935–39, 1940–44) was calculated to be proportional to the distribution in the original cohort. This resulted in that birth cohorts from the 40thies were larger than those from the early 30thies. The randomization procedure was done at the Department of Statistics at University of Göteborg. The 10-digit personal identifiers were the only available personal data for those conducting the computer randomization. No informed consent was required from those in the control group according to the approval from the Ethical Review Committee. Masking of group assignment was only done for the Cause of Death committee, see below. However, possible discrepancies due to group assignments were analysed for differences in treatments given, see below. Subsequent to randomization, we excluded from analysis 56 men with a prior diagnosis of prostate cancer, 34 who had died, and 6 who had emigrated but had not been removed from the Population Register at time of randomization.
Screening algorithm
Figure 1 depicts the study design. Invitations to screening began in January 1995, and in 1996 the study became associated with the ERSPC without any changes in the protocol. Results from the men born during 1930–1939 have been published within the previous ERSPC study.1
Figure 1.
CONSORT diagram of the study
Men allocated to the screening arm were invited every second year for PSA-testing, until they reached the upper age limit.12 The mean age at last invitation to screening was 69 years (range 67–71). The written invitation informed men about the study design, the complexity of PSA screening, and the voluntariness of participation. Blood was processed within 3 hours of venipuncture, frozen, and shipped frozen on dry ice for analyses within two weeks from the blood draw. Total PSA was measured using the dual-label DELFIA Prostatus® total/free PSA-Assay (Perkin-Elmer, Turku, Finland) under the supervision of Dr. Hans Lilja at Lund University. Calibration of this assay changed in 2004 to reflect the World Health Organization 96/670 calibrator; a correction factor was applied to the earlier measurements, and all figures given in this paper are in accordance with this calibration.
The PSA threshold to invite men to further urological work-up was 3.4 ng/ml (WHO-corrected value; the nominal value was 3.0 ng/ml) during 1995–1998; in 1999 the threshold was changed to 2.9 ng/ml (nominal value 2.5 ng/ml) for consistency with other ERSPC sites. Due to the change of assay-calibrator, the threshold further changed to 2.5 ng/ml at the start of 2005. Men with PSA below the threshold were not further evaluated, but were invited again after two years. Only men with PSA at or above the threshold were invited for further urological work-up including digital rectal examination (DRE), trans-rectal ultrasound (TRUS) examination, and at least laterally directed sextant biopsies. For men diagnosed with prostate cancer, the protocol did not specify any particular treatment; further evaluation and treatment was at the discretion of their physicians. Men with a benign finding at the biopsy were re-invited for screening after two years. Men with persistently elevated PSA were recommended a new prostate biopsy at each visit PSA was elevated. Seven screening rounds were completed by the end of 2008. Minor changes in the screening algorithm have been made during the study period.13
In both arms of the study, the incidence of prostate cancer was checked by linking with the West- Swedish Regional Cancer Registry every third month since the study start. In 2009, we linked to all six Regional Cancer Registries in Sweden and obtained data for prostate cancers diagnosed through December 31, 2008. For every man with prostate cancer, all available medical documentation was retrieved for establishing tumour stage, treatment, and disease course. In addition, for all deceased men we obtained a copy of the cause of death (COD) certificate. Two cases of prostate cancer not registered in the Regional Cancers Registries were detected from COD certificates. Linkage with the Population Register was carried out every third month to identify all men who died or emigrated. The last date of follow-up was date of death, date of emigration, or December 31, 2008.
Causes of death
Causes of death for men diagnosed with prostate cancer were determined by an independent COD committee. The committee performed a blinded review of all cases diagnosed with prostate cancer (including all medical records, pathology reports, autopsy protocols) according to a standard algorithm used in the ERSPC.14 The COD certificates were not available to the COD committee. Deaths classified as definitive prostate cancer deaths, intervention-related deaths (i.e. deaths from diagnostic procedures or treatment), or probable prostate cancer deaths were regarded as deaths caused by prostate cancer, while other classifications were regarded as non–prostate cancer deaths.
Statistical analysis
The main outcome measures were absolute and relative risk reduction in cumulative prostate cancer mortality between study arms. Secondary measures were the cumulative prostate cancer incidence and the proportion of screening attendees. A pre-study power calculation (two-sided test, p<0.05 and 80% power) was performed with the assumption of a 70% participation rate. A 40% mortality difference between the study arms was calculated to become significant 15 years after study start (at the end of 2009). A new power calculation in 2009 incorporated the observed 76% participation rate in the Swedish branch in the published ERSPC results.1 The new calculation implied significant power to analyze the data through 2008.
Cumulative incidences of prostate cancer in screening and control groups were plotted as 1 – the Kaplan-Meier estimator. The corresponding hazard ratio for the incidence of prostate cancer between the groups was estimated by Cox regression. The proportional hazard assumption was tested with Schoenfeld residuals15. A time-dependent covariate approach was used to estimate the hazard ratio at different time periods after the start of screening to avoid violence to the proportional hazard assumption. The Nelson-Aalen method was used to calculate the cumulative hazard for prostate cancer mortality.16 Poisson regression analysis was used to estimate the rate ratio of mortality in the screening group to the mortality in the control group. All p-values are two-sided. The number needed to screen (NNS) was calculated as (1/absolute reduction in prostate cancer mortality). As this study is an intention-to-screen analysis, we refer to NNS as number needed to invite for screening. The number needed to treat (NNT) was calculated as ((1/absolute reduction in prostate cancer mortality)*excess prostate cancer incidence). This measure was also rephrased to number needed to diagnose, because many patients actually were not treated. The analyses were done by use of Stata, release 11 (StataCorp, College Station, TX). This study is registered [as an International Standard Randomised Controlled Trial], controlled-trials.com identifier number [ISRCTN49127736].
Role of the funding source
The funding sources had no roles in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript. The funding sources had no access to the database which is kept at the Sahlgrenska University Hospital. All authors of this manuscript have had full access to the database. The corresponding author (JH) has had the final responsibility to submit the manuscript for publication.
Results
Screening and control groups each consisted of 9952 evaluable men. In the screening group, 7578/9952 men (76%) participated in at least one screening round (attendees) (Table 1). These men received 29,315 PSA tests during the study period. In 2469/7578 attendees (33%), PSA was elevated above the threshold at least once and a total of 4693 elevated PSA tests were recorded during the study (Table 1). Among these men with elevated PSA, 2298/2469 (93%) underwent prostate biopsy at least once, and 4153 biopsy procedures were performed in the study. The maximum follow-up time of 14 years was reached by 15 501 (77%) of randomised men.
Table 1.
Number and outcome of participants related to screening visit
| Screening visit | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Invitation round (years) |
Numbers | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | Total |
| 1st (1995–1996) | No. invited | 9890 | |||||||
| No. participating | 5855 | 5855 | |||||||
| No. with elevated PSA level | 661 | 661 | |||||||
| No. with PC | 144 | 144 | |||||||
| 2nd (1997–1999) | No. invited | 9525 | |||||||
| No. participating | 580 | 4680 | 5260 | ||||||
| No. with elevated PSA level | 66 | 543 | 609 | ||||||
| No. with PC | 15 | 98 | 113 | ||||||
| 3rd (1999–2000)* | No. invited | 6920 | |||||||
| No. participating | 460 | 632 | 2283 | 3375 | |||||
| No. with elevated PSA level | 79 | 130 | 621 | 830 | |||||
| No. with PC | 29 | 23 | 108 | 160 | |||||
| 4th (2001–2002) | No. invited | 7873 | |||||||
| No. participating | 291 | 549 | 2251 | 1531 | 4622 | ||||
| No. with elevated PSA level | 49 | 63 | 125 | 497 | 734 | ||||
| No. with PC | 13 | 13 | 19 | 87 | 132 | ||||
| 5th (2003–2004) | No. invited | 6598 | |||||||
| No. participating | 207 | 342 | 547 | 1880 | 1138 | 4114 | |||
| No. with elevated PSA level | 38 | 62 | 54 | 110 | 351 | 615 | |||
| No. with PC | 9 | 11 | 6 | 20 | 65 | 111 | |||
| 6th (2005–2006) | No. invited | 5733 | |||||||
| No. participating | 117 | 188 | 296 | 468 | 1556 | 850 | 3475 | ||
| No. with elevated PSA level | 34 | 34 | 51 | 61 | 104 | 418 | 702 | ||
| No. with PC | 13 | 6 | 14 | 11 | 20 | 81 | 145 | ||
| 7th (2007–2008) | No. invited | 4148 | |||||||
| No. participating | 68 | 94 | 145 | 241 | 374 | 1157 | 535 | 2614 | |
| No. with elevated PSA level | 20 | 11 | 24 | 42 | 64 | 87 | 294 | 542 | |
| No. with PC | 8 | 3 | 3 | 11 | 10 | 11 | 45 | 91 | |
| Total (1995–2008) | Total number of invitations in the study | 50687 | |||||||
| No. participating | 7578 | 6334 | 3794 | 4393 | 3325 | 2452 | 1439 | 29315** | |
| No. with elevated PSA level | 947 | 843 | 875 | 710 | 519 | 505 | 294 | 4693*** | |
| No. with PC | 231 | 154 | 150 | 129 | 95 | 92 | 45 | 896 | |
PSA = Prostate-Specific Antigen
PC = prostate cancer
The low attendance rate in the 3rd invitation round was due to that men with total PSA < 1 ng/ml in the 2nd invitation round were not invited (except those born 1930–1931).
Relates to the total number of PSA tests performed in the study
Relates to the total number of PSA tests exceeding the PSA cut off during the study
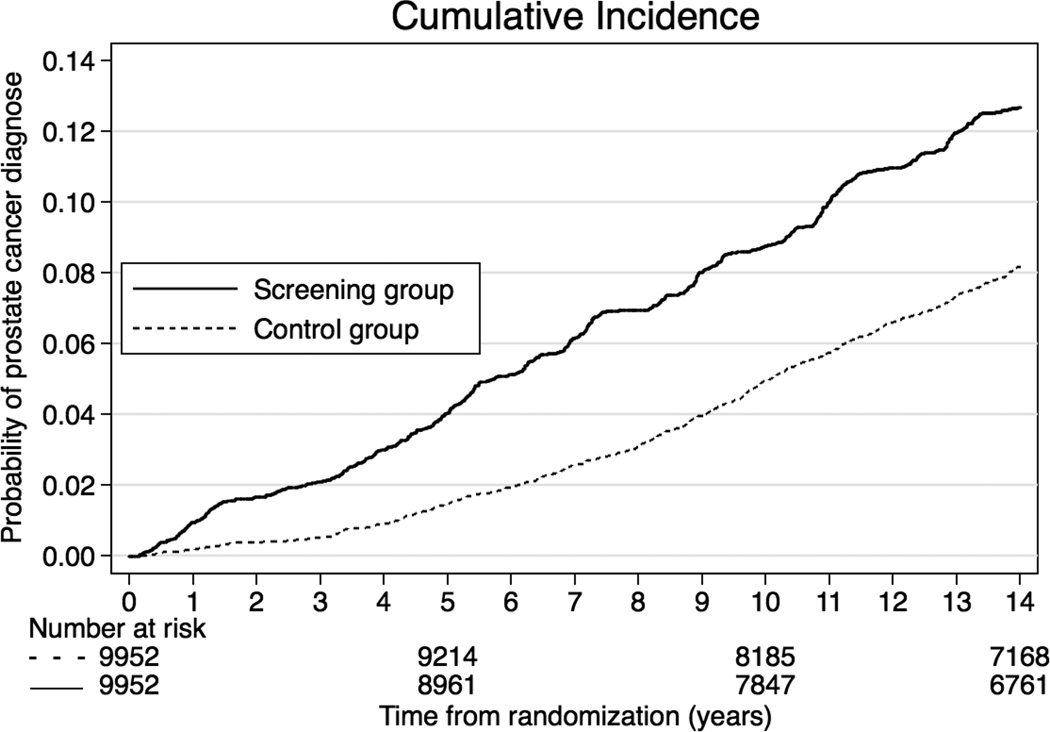
Prostate cancer was diagnosed in 1138 men in the screening group and 718 in the control group. Of those detected in the screening group 896/1138 cases (78.7%) were diagnosed as a result of invitation to the study (Table 1). Of these 896 cases 231 were detected at their first screening visit and 665 during subsequent screening rounds. At the first screening visit (7578 participants) 3671/7578 (48.4%) had PSA below 1 ng/ml, 2960/7578 (39.1%) had a PSA between 1and 2.99 ng/ml and 947/7578 (12.5%) a PSA >= 3 ng/ml. Their risk of being diagnosed with prostate cancer during follow-up was 2.6%, 17.6% and 45.5% respectively. The cumulative incidence of prostate cancer at 14 years was 12.7% in the screening group versus 8.2% in the control group, corresponding to a hazard ratio of 1.64 (95% confidence interval [CI] 1.50–1.80, p<0.0001) (Figure 2)
Figure 2.
Cumulative incidence of prostate cancer in the screening group and in the control group
During the first year after the start of screening the hazard ratio was 5.2 (95% confidence interval [CI] 3.1–8.6), which subsequently decreased to 3.7 (2.2–6.2), 2.6 (1.9–3.6), 2.1 (1.7–2.7), 1.7 (1.3–2.1), and 1.2 (1.0–1.3) (Fig. 2) at year 1–2, 2–4, 4–6, 6–8, and >8 years, respectively.
The majority of prostate cancers diagnosed in the screening group were early-stage disease (Table 2). The number of men with advanced prostate cancer (metastases or PSA >100 ng/ml at diagnosis) was lower in the screening arm: 46 men compared to 87 in the control arm (p=0.0003). Notably, among the non-attendees in the screening group, a high proportion of cancer cases were advanced at diagnosis.
Table 2.
Prostate cancers diagnosed in the study groups
| Screening Group |
||||
|---|---|---|---|---|
| Characteristics | Control group (n=9952) |
All (n=9952) |
Attendees (n=7578) |
Non- attendees (n=2374) |
| Number of men with Prostate cancers diagnosed (%) | 718 (7.2) | 1138 (11.4) | 1046 (13.8) | 92 (3.9) |
| Tumour Grouping (%) | ||||
| Low riska | 199 (2.00) | 604 (6.07) | 590 (7.79) | 14 (0.59) |
| Moderate riskb | 249 (2.50) | 363 (3.65) | 339 (4.47) | 24 (1.01) |
| High riskc | 126 (1.27) | 96 (0.96) | 76 (1.00) | 20 (0.84) |
| Advanced diseased | 87 (0.87) | 46 (0.46) | 25 (0.33) | 21 (0.88) |
| Unknowne | 57 (0.57) | 29 (0.29) | 16 (2.11) | 13 (0.55) |
Defined as T1 and Gleason score ≤ 6 and PSA <10 and not N1 or M1
Defined as T1–2 and/or Gleason score ≤ 7 and/or PSA <20, but not N1 or M1, and not meeting the criteria for low risk
Defined as T1–4 and/or Gleason score ≥ 8 and/or PSA <100, but not N1 or M1, and not meeting the criteria for low or moderate risk
Defined as N1 or M1 or PSA ≥100
Includes 7 cases detected at autopsy
Table 3 presents the primary treatment in the two groups. The difference in stage distribution was mirrored by treatments, with more hormonal therapy in the control arm and more surveillance or treatment with curative intent in the screening arm. However, among men with low- and moderate-risk tumours (as defined in Table 2), the proportion receiving curative treatment was similar between trial arms: 476/967 (49%) in the screening arm and 228/448 (51%) in the control arm. Among the men diagnosed with prostate cancer, the median follow-up after diagnosis was 6.7 years in the screening group and 4.3 years in the control group. In evaluating whether deaths were attributable to prostate cancer, the COD committee and COD certificates were highly concordant. According to the COD committee review, 78 men in the control group died from prostate cancer (77 according to death certificates) compared to 44 (45 according to death certificates) in the screening group. Within the screening group, 27 prostate-cancer–specific deaths were registered among 7578 attendees versus 17 among 2374 non-attendees (Fig. 1). Among attendees who died from prostate cancer, 13 were diagnosed with prostate cancer at their first screening visit (prevalence screen), and the youngest of these men was 59 years at diagnosis. Attendees who were older than 60 at study entry appeared to be at higher risk related to the control group (19/35) of dying from prostate cancer compared to men of younger age (8/43) at study entry (Table 4).
Table 3.
Treatments for prostate cancer
| Treatment, No. (%) | Control group (n=718) |
Screening Group |
||
|---|---|---|---|---|
| All (n=1138) |
Attendees (n=1046) |
Non- attendees (n=92) |
||
| Primary radical prostatectomya | 241 (33.6) | 468 (41.1) | 439 (42.0) | 29 (31.5) |
| Primary radiation | 75 (10.4) | 93 (8.2) | 81 (7.7) | 12 (13.0) |
| Primary endocrine treatment | 162 (22.6) | 80 (7.0) | 47 (4.5) | 33 (35.9) |
| Primary surveillance followed by curative treatmentb | 36 (5.0) | 142 (12.5) | 141 (13.5) | 1 (1.1) |
| Primary surveillance followed by endocrine treatment | 20 (2.8) | 23 (2.0) | 21 (2.0) | 2 (2.2) |
| Surveillance at last follow-up | 152 (21.2) | 314 (27.6) | 301 (28.8) | 13 (14.1) |
| Not treatedc | 32 (4.5) | 18 (1.6) | 16 (1.5) | 2 (2.2) |
Includes 9 cryosurgeries and 6 cystoprostatectomies
Includes 2 cystoprostatectomies
Includes 7 cases detected at autopsy
Table 4.
Outcome related to age at entry in the study
| Screening group |
|||||
|---|---|---|---|---|---|
| Birth year | Total | Control group | All | Attendees | Non-attendees |
|
1930–34 Total No. |
5563 | 2789 | 2774 | 2064 | 710 |
| No. with PC | 615 | 259 | 356 | 318 | 38 |
| No. of deaths | 1689 | 853 | 836 | 488 | 348 |
| No. of PC deaths | 62 | 35 | 27 | 19 | 8 |
|
1935–39 Total No. |
6284 | 3161 | 3123 | 2420 | 703 |
| No. with PC | 654 | 252 | 402 | 372 | 30 |
| No. of deaths | 1284 | 650 | 634 | 360 | 274 |
| No. of PC deaths | 47 | 35 | 12 | 6 | 6 |
|
1940–44 Total No. |
8057 | 4002 | 4055 | 3094 | 961 |
| No. with PC | 587 | 207 | 380 | 356 | 24 |
| No. of deaths | 990 | 479 | 511 | 267 | 244 |
| No. of PC deaths | 13 | 8 | 5 | 2 | 3 |
|
Total Total No. |
19904 | 9952 | 9952 | 7578 | 2374 |
| No. with PC | 1856 | 718 | 1138 | 1046 | 92 |
| No. of deaths | 3963 | 1982 | 1981 | 1115 | 866 |
| No. of PC deaths | 122 | 78 | 44 | 27 | 17 |
PC = prostate cancer
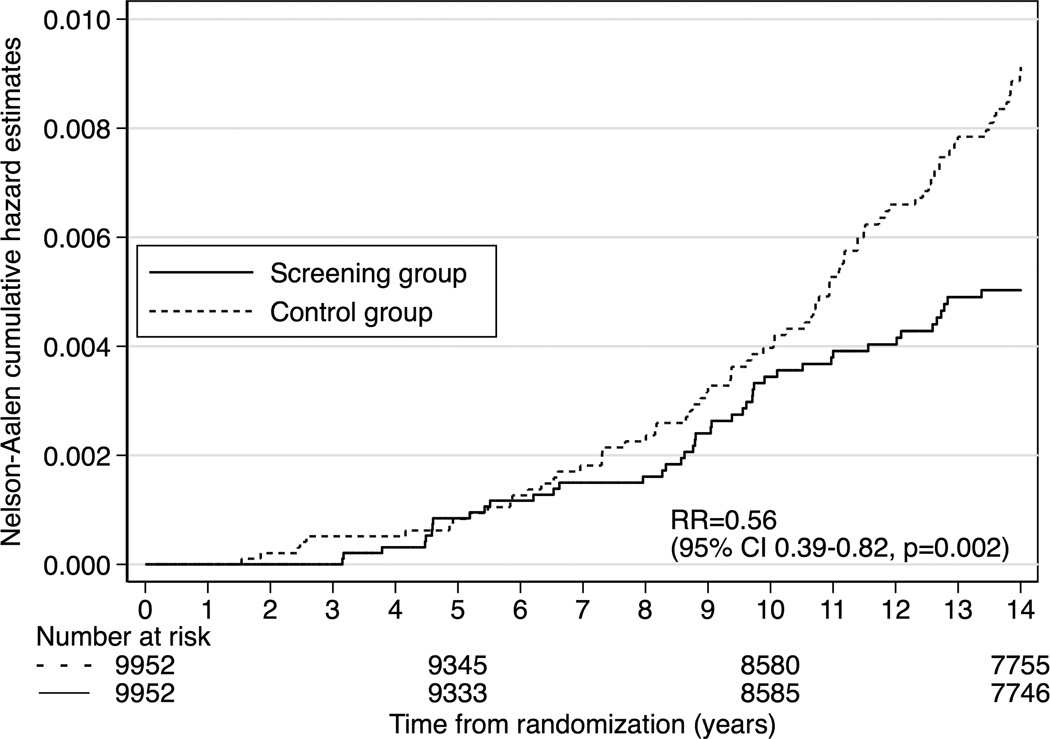
The main outcome analysis performed on intention-to-screen showed that the RR of dying from prostate cancer was 0.56 (95% CI 0.39–0.82, p=0.002) in the screening compared to the control group. The absolute cumulative risk reduction (Kaplan-Meier estimates) of death from prostate cancer at 14 years was 0.40% (95% CI 0.17–0.64%), from 0.90% in the control group to 0.50% in the screening group (Fig. 3). A secondary analysis showed that the RR of attendees compared to the control group was 0.44 (95% CI 0.28–0.68, p=0.0002) and the RR between non-attendees and the control group was 1.05 (95% CI 0.62–1.78 p=0.84).
Figure 3.
Cumulative risk of death from prostate cancer using Nelson-Aalen cumulative hazard estimates
The number of men with prostate cancer dying from unrelated causes was 109/1138 (9.6%) in the screening group and 54/718 (7.5%) in the control group. However, the follow-up time was significantly longer for the men diagnosed with prostate cancer in the screening compared to the control group (6.7 versus 4.3 years, see above). Therefore, the cumulative risk (KM estimates) of non-prostate cancer deaths measured from the date of prostate cancer diagnosis was similar at 10 years, 13.1% in the screening group and 15% in the control group (N.S.).
The number of men needed to invite for screening (NNS) to prevent one prostate cancer death was calculated as 293 while the number needed to diagnose (NNT) was calculated as 12. If the calculations were restricted to attendees, the respective numbers were 234 and 15.
Discussion
This prospective, population-based randomised screening study aimed at evaluating the effectiveness of a screening program in which men were first randomised and then asked to participate. This design results in a more representative study than does randomization after informed consent, and it mirrors the situation when screening is introduced in the population. The study yielded two major findings. First, a PSA-based screening program is acceptable to the male population of ages 50 and older, with 76% attending at least once. Second, with such a participation rate, a screening program will decrease prostate cancer mortality by as much as 44% and by 56% for screening attendees, over 14 years’ follow-up.
Half of the attendees who died from prostate cancer were diagnosed at their first screening visit and many of these men were at age 60 or higher at diagnosis. In a program according to which all men were to start screening at age 50, some of these men might instead have been diagnosed at a possibly curable stage; therefore, there is potential for larger mortality reduction (Table 4).12
This study showed a much higher mortality reduction than in previous studies: a RR of 0.56 in this study compared to 0.80 (0.84 if the Swedish cohort was withdrawn) in the ERSPC publication1 and no reduction in the PLCO study.7 Several factors might account for this. First, the men in our study were younger (median 56 years at baseline) than in both previous publications (median >60 years), and younger men are less likely to have incurable prostate cancer at the first screening and are therefore more likely to realize the full benefit of screening. Second, the PSA threshold for biopsy was lower in our study than in most other ERSPC branches and in the PLCO. However, DRE was never used as a screening tool in our study, which is contrary to the incorporation of DRE by most ERSPC-centres during their first screening round and the design of PLCO. It is possible that addition of DRE in our study would have resulted in an even larger mortality reduction, albeit only few incurable cancers were found in participants who attended the program, and some of these incurable cancers were still non-palpable at diagnosis.12
Third, the interval of screening in this study (every 2 years) was shorter than in the other ERSPC branches (every 4 years), although longer than in the PLCO (every year). Fourth, compared to PLCO, this study had a much higher rate of biopsy among men with a positive screening result (93%, vs 30–40% in PLCO17) and a much lower rate of PSA testing before the start of the study (estimated as 3% vs 44% in PLCO) and probably a lower rate of contamination in the control group. Fifth, the much longer follow-up in the present study (median 14 years from randomization, versus 9 years for the previous ERSPC publication and 11.5 years for PLCO). Up to 10 years of follow-up, the Nelson-Aalen plot in our current study resembles the one previously published from ERSPC, suggesting that most of the benefit from screening occurs after 10 years (Figure 3). This is to be expected from a disease with long lead-time and long natural course18–19.
Although the median follow-up from randomization is long, the follow-up time measured from prostate cancer diagnosis is rather short, 6.7 years for screened men versus 4.3 years for controls in this study compared to 6.3 versus 5.2 years in the PLCO study.7
The reasons as to why our current study showed important mortality reduction and PLCO did not despite a similar follow-up after diagnosis in the two studies may in parts be explained by that men in our study were not pre-screened and thus many aggressive cancers were still “detectable” as well as the fact that contamination in the control group was low - at least during the first five year of our current study. One indication of these important differences is that despite that PLCO randomised 76,693 men compared to 19,904 in the present study “only” 174 prostate cancer deaths were recorded in PLCO7 compared to 122 in our study. The men in the PLCO were also older.
The RR of 0.56 within 14 years corresponds to an absolute risk reduction of 0.40% and obviously no effect on overall mortality (see number of men at risk in Figure 3). These low mortality figures are related to the young age at study start and the comparatively short follow-up after prostate cancer diagnosis. As approximately 5% of deaths among Swedish men are caused by prostate cancer,20 it is obvious that we so far have studied only the early effects of screening. If the relative risk reduction is sustained over time, the mortality reduction, even measured in terms of absolute risk reduction, may become important. One indication of this is the large difference in endocrine treatment between the arms, 79 more men in the control arm were treated with endocrine treatment. Reducing the number of men needing hormonal treatment from 182 (1.8%) in the control group to 103 (1.0%) in the screening group may also be regarded as an important advantage. The increased ratio of unrelated deaths observed in the screening- compared to the control-group (9.6 versus 7.5%) is explained by the longer follow-up of prostate cancer patients in the former group as there is no difference in non-prostate cancer mortality if Kaplan-Meier estimates are calculated from diagnosis.
The high rate of attendance to this PSA-based screening program is corroborated by findings from several uncontrolled trials. For example, Bartsch and co-workers reported that 86.6% accepted an offer of a free PSA test and that 85% of those with elevated levels consented to additional urological evaluation with prostate biopsies.21 Also, all centres within the ERSPC reported a high acceptance rate for screening.1 The screening procedures with PSA testing and prostate biopsy are seldom associated with severe psychological distress, even for men with repeatedly elevated PSA levels.22–23 We therefore conclude that acceptance is not an obstacle for a population-based prostate cancer screening program.
The differences between screening and control groups in stage and grade illustrate the stage migration introduced by a PSA-based screening program. Although 1.6 times as many prostate cancers were diagnosed in the screening arm, the absolute number of advanced cases was lower in the screening arm than in the control arm. Therefore, screening caused a true stage migration. This resulted in a different distribution of treatments between the two groups, However, in men with early cancer (low- and moderate-risk cancer, table 2), treatment with curative intent was equally frequent in the control and screening groups (51% versus 49%), implying that the mortality difference resulted from screening and not from different treatments.
At 14 years of follow-up, the number who need to be invited to screening (corresponding to NNS) to prevent one prostate cancer death was 293, and the number who needed to be diagnosed (corresponding to NNT) was 12. These figures, and the RR of 0.56 in our study, can be compared with those of the commonly recommended practices of screening for breast and colon cancer. Because these figures are time-dependent, we focus this comparison on studies with fairly similar follow-up. For mammography, a 2009 meta-analysis of randomised trials yielded a number needed to invite to screening of 377 (credible interval 230–1050) for women aged 60–69 years and 1339 (credible interval 322–7455) for women aged 50–59 years, and RR’s of 0.68 and 0.86 respectively, at 11–20 years of follow-up.5 Separate or other mammography studies have yielded similar numbers.3, 24–31 In a separate 2009 Cochrane review, the NNT for mammography was 10 throughout 10 years.3 For colorectal cancer screening by fecal occult blood test, the RR’s have varied between 0.67 and 0.87 among four randomised trials32–35 and was 16% overall in both a 2008 Cochrane review4 (after 11.7–18.4 years) and a meta-analysis by Towler et al (after 7.8–13 years).6 In the latter, the NNS after 10 years was estimated as 1173 (95% CI 741–2807). A recent multicentre study presents a RR of 0.69 for colorectal cancer mortality with flexible sigmoidoscopy screening for colorectal cancer and a lower NNS of 489 at a median follow-up of 11.2 years36. As screening for colorectal cancer is associated with a reduced colorectal cancer incidence, NNT is not possible to calculate for comparison.
The NNT in our study is substantially lower compared to previous ERSPC publication indicating two things. First, the NNT is very dependent on the length of follow-up and it is not easy to predict when and at which level NNT will stabilize. Second, as NNT in prostate cancer screening mainly reflects the risk of over-diagnosis it is not easy at this point to make estimates of this risk but it is probably not that high as some has feared37, at least if screening is restricted to the age groups included in this study. NNT in prostate cancer screening is better rephrased to “Number needed to diagnose” as many men were not treated. As many as 314 (30%) of screen attendees were in active surveillance at last follow-up in this study. This strategy will at least lower the risk of over-treatment and the risk associated seems low38.
However, as it takes a long time to achieve the benefit from prostate cancer screening, only marginal benefit the first 10 years from starting prostate cancer screening, one should be cautious to recommend elderly men screening. As the risk of over-diagnosis and over-treatment still are the major concerns it seems questionable to invite men over the age of 70 for PSA screening.
Conclusion
To conclude, prostate cancer screening is well accepted by the general population and may result in a relevant reduction in cancer mortality greater than what has been seen in screening for breast or colorectal cancer. A disadvantage of PSA screening is the long and varying lead time, resulting in a risk of over-diagnosis that is substantial but still of largely unknown magnitude.
Acknowledgements
This study was supported by the Swedish Cancer Society (Contract numbers 09 0107, 080315 and 083455), the Swedish Research Council (Medicine) [20095] and the National Cancer Institute [grant number R21-CA127768-01A1]. Grants were also received from the Stichting af Jochnick Foundation, Catarina and Sven Hagstroms family foundation, Gunvor and Ivan Svensson’s foundation, Johanniterorden, King Gustav V Jubilée Clinic Cancer Research Foundation, Sahlgrenska University Hospital, Abbott Pharmaceuticals, Sweden, and Schering Plough, Sweden. Sidney Kimmel Center for Prostate and Urologic Cancers, David H. Koch through the Prostate Cancer Foundation, and Fundación Federico SA are also acknowledged for their funding support.
We thank: The COD committee, Bo Johan Norlén, Professor Emeritus in Urology; Silas Pettersson, Professor Emeritus in Urology; and Eberhard Varenhorst, Professor Emeritus in Urology. Helén Ahlgren, data manager and responsible for the study secretary. Maria Nyberg, study nurse. Charlotte Becker, MD, PhD, Clinical chemist. Gun-Britt Eriksson, Kerstin Håkansson, and Mona Hassan Al-Battat for expert assistance with immunoassays. Björn Zackrisson, Urologist, former PhD-student Roland Frösing, Urologist, Erik Pileblad, Urologist, Rebecka Godtman, MD and Anna Grenabo Bergdahl, MD, PhD-student all at the Urology department at Sahlgrenska Hospital for various aspects in this screening project and especially for helping with all biopsies performed. Janet Novak, PhD, at Helix Editing for her assistance with editing of the manuscript, which was paid for by Memorial Sloan-Kettering Cancer Center. Andrew Vickers, PhD at Memorial Sloan-Kettering Cancer Center for valuable criticism and suggestions.
Footnotes
To view the protocol of this trial see http://media.erspc-media.org/sweden/
Contributions
The corresponding author (JH) had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. JH is the principal investigator of the study and was responsible for planning of the study. HL participated in conception and design of the study and contributed with supervision and administrative support. JH and HL were responsible for funding. Data collections were made by JH, AK, PL, JS, SB and C–G P. The biopsy procedures were carried out by JH, GA, SC, AK, PL, JS and SB. The pathological examination of all specimens in the study was performed by C–G P. The data analysis (extraction of results and the statistical analysis) was made by EH, JH, SC, HL and GA. The interpretation of data was made by JH, JS, SC, HL and GA. SC performed the literature search. All authors were involved in the writing (JH, SC, GA, HL, JS), revision (JH, SC, GA, HL, PL, AK, SB) or review (EH, C–G P) of the manuscript. Other persons involved and their role in this work is presented under Acknowledgements.
Disclosure
Dr. Hans Lilja holds patents for free PSA and hK2 assays. Dr. Lilja reports having received honoraria from GlaxoSmithKline. Dr. Hugosson reports receiving lecture fees from GlaxoSmithKline and Abbott Pharmaceuticals. All other authors declare that they have no conflicts of interest.
References
- 1.Schroder FH, Hugosson J, Roobol MJ, Tammela TL, Ciatto S, Nelen V, et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med. 2009 Mar 26;360(13):1320–1328. doi: 10.1056/NEJMoa0810084. [DOI] [PubMed] [Google Scholar]
- 2.Roobol MJ, Kerkhof M, Schroder FH, Cuzick J, Sasieni P, Hakama M, et al. Prostate cancer mortality reduction by prostate-specific antigen-based screening adjusted for nonattendance and contamination in the European Randomised Study of Screening for Prostate Cancer (ERSPC) Eur Urol. 2009 Oct;56(4):584–591. doi: 10.1016/j.eururo.2009.07.018. [DOI] [PubMed] [Google Scholar]
- 3.Gotzsche PC, Nielsen M. Screening for breast cancer with mammography. Cochrane Database Syst Rev. 2009(4):CD001877. doi: 10.1002/14651858.CD001877.pub3. [DOI] [PubMed] [Google Scholar]
- 4.Hewitson P, Glasziou P, Watson E, Towler B, Irwig L. Cochrane systematic review of colorectal cancer screening using the fecal occult blood test (hemoccult): an update. Am J Gastroenterol. 2008 Jun;103(6):1541–1549. doi: 10.1111/j.1572-0241.2008.01875.x. [DOI] [PubMed] [Google Scholar]
- 5.Nelson HD, Tyne K, Naik A, Bougatsos C, Chan BK, Humphrey L, et al. Screening for breast cancer: an update for the U.S. Preventive Services Task Force. Ann Intern Med. 2009 Nov 17;151(10):727–737. W237–W242. doi: 10.1059/0003-4819-151-10-200911170-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Towler B, Irwig L, Glasziou P, Kewenter J, Weller D, Silagy C. A systematic review of the effects of screening for colorectal cancer using the faecal occult blood test, hemoccult. BMJ. 1998 Aug 29;317(7158):559–565. doi: 10.1136/bmj.317.7158.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Andriole GL, Grubb RL, 3rd, Buys SS, Chia D, Church TR, Fouad MN, et al. Mortality results from a randomized prostate-cancer screening trial. N Engl J Med. 2009 Mar 26;360(13):1310–1319. doi: 10.1056/NEJMoa0810696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sandblom G, Varenhorst E, Lofman O, Rosell J, Carlsson P. Clinical consequences of screening for prostate cancer: 15 years follow-up of a randomised controlled trial in Sweden. Eur Urol. 2004 Dec;46(6):717–723. doi: 10.1016/j.eururo.2004.08.011. discussion 24. [DOI] [PubMed] [Google Scholar]
- 9.Kjellman A, Akre O, Norming U, Tornblom M, Gustafsson O. 15-year followup of a population based prostate cancer screening study. J Urol. 2009 Apr;181(4):1615–1621. doi: 10.1016/j.juro.2008.11.115. discussion 21. [DOI] [PubMed] [Google Scholar]
- 10.Labrie F, Candas B, Cusan L, Gomez JL, Belanger A, Brousseau G, et al. Screening decreases prostate cancer mortality: 11-year follow-up of the 1988 Quebec prospective randomized controlled trial. Prostate. 2004 May 15;59(3):311–318. doi: 10.1002/pros.20017. [DOI] [PubMed] [Google Scholar]
- 11.Ilic D, O'Connor D, Green S, Wilt TA. Screening for prostate cancer: A Cochrane systematic review. Cancer Causes Control. 2007 Apr;18(3):279–285. doi: 10.1007/s10552-006-0087-6. [DOI] [PubMed] [Google Scholar]
- 12.Bergdahl AG, Aus G, Lilja H, Hugosson J. Risk of dying from prostate cancer in men randomized to screening: differences between attendees and nonattendees. Cancer. 2009 Dec 15;115(24):5672–5679. doi: 10.1002/cncr.24680. [DOI] [PubMed] [Google Scholar]
- 13. [cited 2010-05-21];ERSPC-website. Available from: http://media.erspc-media.org/sweden/
- 14.De Koning HJ, Blom J, Merkelbach JW, Raaijmakers R, Verhaegen H, Van Vliet P, et al. Determining the cause of death in randomized screening trial(s) for prostate cancer. BJU Int. 2003 Dec;92(Suppl 2):71–78. doi: 10.1111/j.1465-5101.2003.04402.x. [DOI] [PubMed] [Google Scholar]
- 15.Grambsch P, Therneau T. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika. 1994;81(3):515–526. [Google Scholar]
- 16.Aalen O. Nonparametric inference for a family of counting processes. Ann Stat. 1978;6:701–726. [Google Scholar]
- 17.Grubb RL, 3rd, Pinsky PF, Greenlee RT, Izmirlian G, Miller AB, Hickey TP, et al. Prostate cancer screening in the Prostate, Lung, Colorectal and Ovarian cancer screening trial: update on findings from the initial four rounds of screening in a randomized trial. BJU Int. 2008 Dec;102(11):1524–1530. doi: 10.1111/j.1464-410X.2008.08214.x. [DOI] [PubMed] [Google Scholar]
- 18.Tornblom M, Eriksson H, Franzen S, Gustafsson O, Lilja H, Norming U, et al. Lead time associated with screening for prostate cancer. Int J Cancer. 2004 Jan 1;108(1):122–129. doi: 10.1002/ijc.11554. [DOI] [PubMed] [Google Scholar]
- 19.Albertsen PC, Hanley JA, Fine J. 20-year outcomes following conservative management of clinically localized prostate cancer. JAMA. 2005 May 4;293(17):2095–2101. doi: 10.1001/jama.293.17.2095. [DOI] [PubMed] [Google Scholar]
- 20. [cited 2009-11-15];Causes of death 2007. The National Board of Health and Welfare, 2009. Official Statistics of Sweden. Available from: http://www.socialstyrelsen.se/Statistik/statistik_amne/dodsorsaker/index.htm.
- 21.Bartsch G, Horninger W, Klocker H, Pelzer A, Bektic J, Oberaigner W, et al. Tyrol Prostate Cancer Demonstration Project: early detection, treatment, outcome, incidence and mortality. BJU Int. 2008 Apr;101(7):809–816. doi: 10.1111/j.1464-410X.2008.07502.x. [DOI] [PubMed] [Google Scholar]
- 22.Brindle LA, Oliver SE, Dedman D, Donovan JL, Neal DE, Hamdy FC, et al. Measuring the psychosocial impact of population-based prostate-specific antigen testing for prostate cancer in the UK. BJU Int. 2006 Oct;98(4):777–782. doi: 10.1111/j.1464-410X.2006.06401.x. [DOI] [PubMed] [Google Scholar]
- 23.Carlsson S, Aus G, Wessman C, Hugosson J. Anxiety associated with prostate cancer screening with special reference to men with a positive screening test (elevated PSA) - Results from a prospective, population-based, randomised study. Eur J Cancer. 2007 Sep;43(14):2109–2116. doi: 10.1016/j.ejca.2007.06.002. [DOI] [PubMed] [Google Scholar]
- 24.Miller AB, To T, Baines CJ, Wall C. The Canadian National Breast Screening Study-1: breast cancer mortality after 11 to 16 years of follow-up. A randomized screening trial of mammography in women age 40 to 49 years. Ann Intern Med. 2002 Sep 3;137(5 Part 1):305–312. doi: 10.7326/0003-4819-137-5_part_1-200209030-00005. [DOI] [PubMed] [Google Scholar]
- 25.Miller AB, To T, Baines CJ, Wall C. Canadian National Breast Screening Study-2: 13- year results of a randomized trial in women aged 50–59 years. J Natl Cancer Inst. 2000 Sep 20;92(18):1490–1499. doi: 10.1093/jnci/92.18.1490. [DOI] [PubMed] [Google Scholar]
- 26.Nystrom L, Andersson I, Bjurstam N, Frisell J, Nordenskjold B, Rutqvist LE. Long-term effects of mammography screening: updated overview of the Swedish randomised trials. Lancet. 2002 Mar 16;359(9310):909–919. doi: 10.1016/S0140-6736(02)08020-0. [DOI] [PubMed] [Google Scholar]
- 27.Moss SM, Cuckle H, Evans A, Johns L, Waller M, Bobrow L, et al. Effect of mammographic screening from age 40 years on breast cancer mortality at 10 years' follow-up: a randomised controlled trial. Lancet. 2006 Dec 9;368(9552):2053–2060. doi: 10.1016/S0140-6736(06)69834-6. [DOI] [PubMed] [Google Scholar]
- 28.Tabar L, Fagerberg G, Chen HH, Duffy SW, Smart CR, Gad A, et al. Efficacy of breast cancer screening by age. New results from the Swedish Two-County Trial. Cancer. 1995 May 15;75(10):2507–2517. doi: 10.1002/1097-0142(19950515)75:10<2507::aid-cncr2820751017>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 29.Shapiro S, Venet W, Strax P, Venet L, Roeser R. Ten- to fourteen-year effect of screening on breast cancer mortality. J Natl Cancer Inst. 1982 Aug;69(2):349–355. [PubMed] [Google Scholar]
- 30.Frisell J, Lidbrink E, Hellstrom L, Rutqvist LE. Followup after 11 years--update of mortality results in the Stockholm mammographic screening trial. Breast Cancer Res Treat. 1997 Sep;45(3):263–270. doi: 10.1023/a:1005872617944. [DOI] [PubMed] [Google Scholar]
- 31.Bjurstam N, Bjorneld L, Warwick J, Sala E, Duffy SW, Nystrom L, et al. The Gothenburg Breast Screening Trial. Cancer. 2003 May 15;97(10):2387–2396. doi: 10.1002/cncr.11361. [DOI] [PubMed] [Google Scholar]
- 32.Kronborg O, Jorgensen OD, Fenger C, Rasmussen M. Randomized study of biennial screening with a faecal occult blood test: results after nine screening rounds. Scand J Gastroenterol. 2004 Sep;39(9):846–851. doi: 10.1080/00365520410003182. [DOI] [PubMed] [Google Scholar]
- 33.Lindholm E, Brevinge H, Haglind E. Survival benefit in a randomized clinical trial of faecal occult blood screening for colorectal cancer. Br J Surg. 2008 Aug;95(8):1029–1036. doi: 10.1002/bjs.6136. [DOI] [PubMed] [Google Scholar]
- 34.Mandel JS, Church TR, Ederer F, Bond JH. Colorectal cancer mortality: effectiveness of biennial screening for fecal occult blood. J Natl Cancer Inst. 1999 Mar 3;91(5):434–437. doi: 10.1093/jnci/91.5.434. [DOI] [PubMed] [Google Scholar]
- 35.Scholefield JH, Moss S, Sufi F, Mangham CM, Hardcastle JD. Effect of faecal occult blood screening on mortality from colorectal cancer: results from a randomised controlled trial. Gut. 2002 Jun;50(6):840–844. doi: 10.1136/gut.50.6.840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Atkin WS, Edwards R, Kralj-Hans I, Wooldrage K, Hart AR, Northover JM, et al. Once-only flexible sigmoidoscopy screening in prevention of colorectal cancer: a multicentre randomised controlled trial. Lancet. 2010 May 8;375(9726):1624–1633. doi: 10.1016/S0140-6736(10)60551-X. [DOI] [PubMed] [Google Scholar]
- 37.Albertsen PC. The Unintended Burden of Increased Prostate Cancer Detection Associated With Prostate Cancer Screening and Diagnosis. Urology. 2009 Nov 19; doi: 10.1016/j.urology.2009.08.078. [DOI] [PubMed] [Google Scholar]
- 38.Khatami A, Aus G, Damber JE, Lilja H, Lodding P, Hugosson J. PSA doubling time predicts the outcome after active surveillance in screening-detected prostate cancer: results from the European randomized study of screening for prostate cancer, Sweden section. Int J Cancer. 2007 Jan 1;120(1):170–174. doi: 10.1002/ijc.22161. [DOI] [PubMed] [Google Scholar]