Abstract
Autoimmune responses targeting synaptic proteins are associated with a wide range of neurologic symptoms. Among these disorders are those associated with antibodies to ionotropic glutamate receptors, including the NMDAR (N-methyl-D-aspartate receptor) and AMPAR (α-amino-3-hydrozy-5-methyl-4-isoxazolepropionic acid receptor). Patients with anti-NMDAR encephalitis present with psychiatric symptoms, seizures, movement disorders, impaired consciousness, and autonomic derangements; half of patients have an associated ovarian teratoma, and most patients respond to immunosuppressive therapies. Patients’ antibodies bind to the amino terminal domain of the NMDAR, and result in loss of NMDARs from synapses with subsequent NMDAR hypofunction. Anti-NMDAR antibodies have now been reported in other neuropsychiatric conditions, including psychosis, dementia, and HSV encephalitis. The pathophysiologic relevance of anti-NMDAR antibodies in these disorders is not yet clear, but their presence may indicate a role for immunotherapy in some patients. Although considerable work remains to be done, our understanding of disorders associated with anti-glutamate receptor antibodies has grown exponentially since they were first described just over 7 years ago, revolutionizing neurology. These antibodies, by interfering with synaptic function, readily link basic science and clinical medicine, and have revealed the impact of sudden but sustained loss of specific neurotransmitter receptors in humans. Improved understanding of their pathophysiology will lead to better treatments for these diseases while providing novel insights regarding the roles of glutamate receptors in learning, memory, and neuropsychiatric disease.
In recent years it has been found that autoimmune responses can target a number of synaptic proteins, generating specific autoantibodies associated with a wide range of neurologic symptoms. These autoimmune responses frequently result in encephalitis, leading to the broad designation of “synaptic autoencephalitides” (Kayser and Dalmau 2011) Among the synaptic proteins that have been identified as autoantigens are proteins associated with voltage-gated potassium channels, such as Caspr2 (contactin-associated protein-like 2) (Lancaster et al. 2011) and LGI-1 (leucine-rich glioma-inactivated protein 1) (Lai et al., 2010); metabotropic receptors, such as GABA-B (γ-aminobutyric acid receptor B)(Lancaster et al. 2010) and mGluR5 (metabotropic glutamate receptor 5) (Lancaster et al. 2011); and ionotropic receptors, such as the glycine receptor (Hutchinson et al. 2008). Perhaps the best known are the encephalidities associated with antibodies to ionotropic glutamate receptors, including the NMDAR (N-methyl-D-aspartate receptor) (Dalmau et al., 2007) and AMPAR (α-amino-3-hydrozy-5-methyl-4-isoxazolepropionic acid receptor) (Lai et al., 2009). In these disorders, autoantibodies are directed against surface proteins and appear to be directly pathogenic, as compared to autoantibodies to cytosolic antigens, which are believed to simply be epiphenomena or non-specific markers of disease progression (Wiley et al., 1998). In this regard, the newly discovered synaptic autoencephalidites resemble the more well known autoimmune diseases of the peripheral nervous system, such as myasthenia gravis (MG) and Lambert-Eaton myasthenic syndrome (LEMS), in which surface nicotinic acetylcholine receptors or voltage-gated calcium channels, respectively, are targeted, resulting in functional consequences (Lindstrom, 2002; Titulaer et al., 2011).
This difference in antibody target has direct consequences for patient treatment: paraneoplastic autoimmune CNS disorders with cytosolic antibodies frequently show high levels of CD8-positive cytotoxic T cells, which mediate the majority of damage (Bien et al. 2012; Koike et al. 2011) and tend to be poorly responsive to immunosuppression (Keime-Guibert et al., 2000) In these patients, aggressive tumor treatment is the most common course of action and outcome is worse in cases in which the tumor is not found (Graus et al., 2001) . In patients with synaptic autoencephalitides, however, there is no evidence of T cell cytotoxicity, minimal parenchymal inflammation, and no evidence of neuro-axonal injury (Bien et al., 2012). Accordingly, immunosuppression is much more effective, and, even in disorders such as anti NMDAR encephalitis, in which the acute phase is severe and potentially life threatening, overall long-term outcomes are good (Titulaer et al., 2013b).
Anti-NMDAR encephalitis
Clinical presentation of anti-NMDAR encephalitis
Anti-NMDAR encephalitis is the most well studied disorder within this newly discovered category of synaptic autoencephalidites. First described in 2007 in 12 patients (Dalmau et al., 2007), followed by a more extensive report of 100 patients in 2008 (Dalmau et al., 2008), the hallmark of this form of encephalitis is the presence of antibodies against the GluN1 subunit of the NMDA receptor. Since its recent discovery, awareness of anti-NMDAR encephalitis has grown, and currently this disorder is the most commonly identified cause of encephalitis (Granerod et al., 2010; Gable et al., 2012).
Initially, most patients present with psychiatric symptoms, including behavioral changes, paranoia, memory loss and hallucinations. Over weeks to months, the disorder progresses to include seizures, movement disorders, impaired consciousness, and autonomic derangements such as central hypoventilation, hypertension, and bradycardia. This pattern of progressive neurologic involvement is characteristic of the disease, and, by one month after disease onset, only 1% of patients are monosymptomatic (Titulaer et al., 2013b).
The precipitating autoimmune event that triggers antibody formation is unclear. In the initial cohort of 12 patients, all had teratomas (11/12 were ovarian), and all tested teratomas expressed NMDARs (Dalmau et al., 2007). However, the disorder has since expanded in scope to include male patients and pediatric patients in whom tumors are rare (Florance et al., 2009; Irani et al., 2010; Kashyape et al., 2012) In the large cohort of patients reported by Titulaer et al. (2013), only 38% of patients had ovarian teratomas. NMDAR-expressing teratomas presumably trigger the development of autoantibodies with spread of the immune response to the CNS . These antibodies are not ubiquitous in teratoma patients in general; a recent prospective study of patients with ovarian teratomas without encephalitis failed to detect any anti-NMDAR antibodies (Mangler et al., 2013). The instigating factor in patients without teratomas remains unknown, although a post-viral etiology has been proposed (Dalmau et al., 2008). Other than the very characteristic symptoms of anti-NMDAR encephalitis, additional neurological studies are generally non-specific and not useful in diagnosis of the disease. Brain MRI scans are normal in approximately half of patients, and when changes are seen they are typically non-specific (Dalmau et al., 2008). In most patients, non-specific EEG abnormalities (slowing, epileptiform discharges) are seen and cerebrospinal fluid studies are mildly abnormal, including mild lymphocytic pleiocytosis, elevated protein, and less often, the presence of oligoclonal bands (Dalmau et al., 2008).
The immunology and treatment of anti-NMDAR encephalitis
The immunologic basis of anti-NMDAR receptor encephalitis is largely unexplored. T helper 17 cell (Th17) responses may play a role (Ulusoy et al., 2012) . In contrast to what is seen in T-cell mediated neuro-immune diseases associated with cytosolic autoantigens, on autopsy in anti-NMDAR encephalitis, there is minimal parenchymal inflammation without prominent T-cell infiltration, absent complement deposition, and no evidence of neuro-axonal injury (Dalmau et al., 2007; Martinez-Hernandez et al., 2011; Bien et al., 2012). Instead, antibody–secreting plasma cells infiltrate the CNS, and the disease seems to be mediated directly by antibodies (Martinez-Hernandez et al., 2011). Plasma exchange and/or intravenous immunoglobulin (IVIG) treatment, both of which remove of antibodies from the blood stream, are effective in reducing serum anti-NMDAR antibody titers. However, intrathecal antibody synthesis, likely by memory B-cells within the CNS, often results in persistently high CSF antibody titers and persistent disease (Dalmau et al., 2007; Florance et al., 2009; Hughes et al., 2010). In those cases in which a tumor is found, its removal improves outcome (Dalmau et al. 2008). Approximately half of patients respond well to these first line therapies, defined by significant improvement within one month. These patients are almost all at their neurologic baseline by two years follow up. The remaining half of patients may require more aggressive second-line immunotherapies such as the rituximab and/or cyclophosphamide to target antibody-producing lymphocytes. . In these refractory patients, the use of second-line immunotherapy predicts better outcomes, with just over 75% of aggressively treated patients at or near their baseline after two years, as compared to just over 50% for those refractory patients in whom treatment was not escalated. In addition, patients treated with agents such as rituximab and cyclophosphamide were less likely to go on to have relapses of the disease (Titulaer et al., 2013b).
Effects of patients’ antibodies on NMDARs
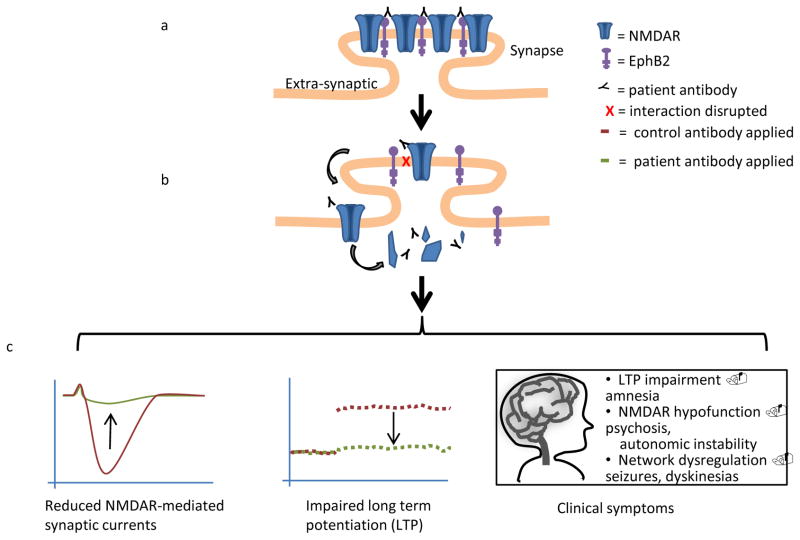
The identity of the autoantigen in this disorder was first hinted at by intense hippocampal immunostaining following application of patients’ antibodies to rat brain sections (Dalmau et al., 2007). Subsequent work using transfected HEK cells demonstrated that the predominantly recognized antigen is the GluN1 subunit of the NMDAR receptor (Dalmau et al., 2008). Acutely, binding of patients’ antibodies to the NMDAR increases channel open time (Figure 2E) (Gleichman et al., 2012). However, over the longer term, application of patients’ antibodies to live, cultured, hippocampal neurons results in loss of surface NMDARs in a specific and reversible manner via antibody-mediated capping and internalization of receptors (Figure 1A-B) (Hughes et al., 2010). Fluorescence recovery after photobleaching (FRAP) and high-resolution nanoparticle imaging has shown that autoantibody application results in lateral displacement of surface GluN2A-containing NMDARs out of synapses and into the extrasynaptic space. Meanwhile, the mobile fraction of extrasynaptic GluN2B-containing NMDARs is reduced. Taken together, these results suggest that antibodies either actively displace NMDARs from synapses or disrupt a critical synaptic-retention mechanism; extrasynaptic NMDARs are then cross-linked and internalized (Figure 1A-B) (Mikasova et al., 2012). The impacted mechanism of synaptic retention may involve Ephrin-B2 receptors (Eph-B2Rs), as patients’ antibodies weaken the interaction between the extracellular domains of NMDARs and Eph-B2Rs (Figure 1B); furthermore, antibody-induced loss of NMDARs can be prevented by exogenous activation of EPHB2 receptors (Mikasova et al., 2012).
Figure 2.
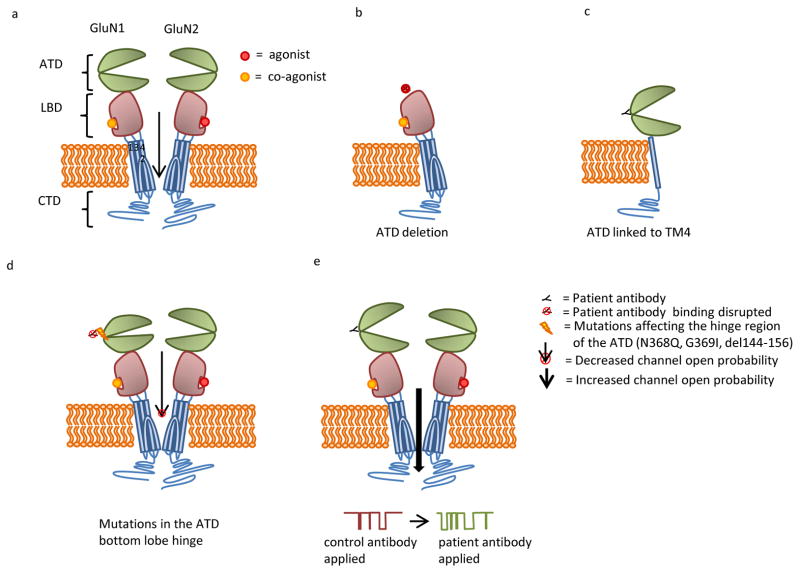
(A) NMDAR subunits contain two extracellular domains: amino terminal domain (ATD) and a ligand binding domain (LBD) comprised of an S1 and S2 domain, three transmembrane domains (TMs 1, 3, 4), a transmembrane loop (TM 2), and a cytosolic c-terminal domain (CTD). GluN1 and GluN2 subunits typically combine to form a heterotetrameric receptor. (B) Deletion of the ATD eliminates patients’ antibody binding. (C) Patients’ antibodies bind to a construct that contains the ATD linked to TM4. This construct lacks the LBD and TM domains 1–3. (D) Patients’ antibody binding is eliminated by mutations to the ATD hinge region, including N368Q, G369I, and deletion of amino acids 144–156. These mutations appear to decrease channel open time. (E) Patients’ antibody binding results in increased channel open time.
Figure 1.
(A) Synaptic NMDARs interact with EphB2; patients’ antibodies bind to NMDARs. (B) After patients’ antibody binding, the NMDAR – EphB2 interaction is disrupted. NMDARs leave synapses, are cross-linked, and internalized. (C) Patients’ antibodies reduce synaptic NMDAR currents and impair LTP induction. These electrophysiologic effects are linked to the clinical symptoms of the disease, including amnesia, psychosis, seizures, and dyskinetic movements.
Recordings of miniature EPSCs in cultured hippocampal neurons treated with patients’ antibodies demonstrate a specific reduction in synaptic NMDAR-mediated currents (Figure 1C) (Hughes et al., 2010). In contrast, AMPAR-mediated currents are not affected, and patients’ antibodies do not affect other synaptic proteins such as AMPARs, PSD-95, or Bassoon, nor do they alter synaptic morphology . In vivo studies have shown that infusion of patients’ antibodies results in loss of NMDARs within rat hippocampi (Hughes et al., 2010). Patients’ CSF is able to suppress the induction of long-term potentiation (LTP) at Schaffer collateral- CA1 synapses in mouse hippocampal slices (Figure 1C). This effect is blocked by preabsorption of patients’ antibodies by NMDAR-expressing cultured HEK cells, thereby confirming that it is the anti-NMDAR antibodies present in patients’ CSF that are responsible for suppressing LTP (Zhang et al., 2012). Similarly, exposure of cultured hippocampal neurons to patient CSF blocks the induction of chemical LTP, as measured by a failure to increase synaptic content of surface AMPARs (Mikasova et al., 2012).
Epitope recognition by anti-NMDAR antibodies
Even in the original reports, the epitope recognized by patients’ antibodies appeared highly conformation-dependent, as antibodies were unable to recognize NMDARs on Western blots, and their staining was dependent on fixation technique (Dalmau et al., 2007; Gleichman et al., 2012). Recent work based on epitope mapping using transfected HEK cells has confirmed this original impression, demonstrating that a small and conformation dependent region of the amino terminal domain (ATD) of GluN1 is recognized by patients’ antibodies. NMDAR subunits contain two extracellular domains: a 400 amino acid ATD and a ligand binding domain (LBD) comprised of an S1 and S2 domain, three transmembrane domains (TMs I, III, IV), a transmembrane loop (TM II), and a cytosolic domain that mediates scaffolding, localization, and coupling to intracellular signaling domains (Figure 2A) (Traynelis et al., 2010) . Patients’ antibody staining to HEK cells transfected with various GluN1 constructs has shown that deletion of the ATD eliminates antibody binding (Figure 2B), and that binding is unaffected by a GluN1 construct that links the end of the ATD directly to TM4 (therefore lacking the S1, S2, and several trans-membrane domains) (Figure 2C). Taken together, these results show the GluN1 ATD is both necessary and sufficient for patients’ antibody binding (Gleichman et al., 2012).
Within the ATD of GluN1, posttranslational modifications may play an important role in epitope recognition. Blockade of N-linked glycosylation by tunicamycin abolishes patients’ antibody staining. Mutation of only one of the seven N-linked glycosylation consensus sites in the ATD, N368Q, has an impact similar to tunicamycin’s abolition of patients’ antibody staining (Figure 2D). However, a different mutation of that same glycosylation consensus site (T370A) has a less dramatic effect, indicating that glycosylation state is likely not the only factor important for antibody recognition of this region (Gleichman et al., 2012). GluN1-N368 also may undergo deamidation, a nonenzymatic post-translational modification. A series of mutations to G369 that slowed deamidation of N368 to different degrees resulted in staining that correlated with deamidation rates of model peptides (Robinson et al., 2004). Taken together, these results suggest that the structural requirements for glycosylation and deamidation of N368 are also crucial for creating the exact receptor conformation recognized by patients’ antibodies (Figure 2D).
The ATD of NMDARs is not directly involved in ligand binding or channel opening, but does serve multiple discrete functions (Paoletti, 2011; Furukawa, 2012). In addition to being needed for receptor assembly (Meddows et al., 2001; Hansen et al., 2010), it is also bound to by multiple allosteric modulators of receptor function (Paoletti, 2011). The ATD is composed of a top and bottom lobe, arranged in a clamshell-like structure with the two lobes separated by a middle cleft (Figure 2A) (Jin et al., 2009; Karakas et al., 2009; Sobolevsky et al., 2009). The cleft’s exact conformation may be altered by interactions with other subunits and also reflect the binding of various modulators (Karakas et al., 2011; Furukawa, 2012). Changes in the cleft conformation then alter the probability of channel opening and closing through as yet unclear mechanisms (Gielen et al., 2009; Paoletti, 2011). Residues N368 and G369, crucial for epitope formation, are located in the bottom lobe of the ATD, near the “clamshell” hinge of the two lobes. Residues 144–156 form an alpha-helix that is in close proximity to N368/G369 based on predicted structure, although far removed in primary sequence (Farina et al., 2011; Karakas et al., 2011). Deletion of this alpha helix also abolishes patients’ antibody staining, further confirming that this region of the bottom lobe near the hinge is critically important determinant of the epitope (Figure 2D).
Since this hinge region is important for control of receptor physiology (Gielen et al., 2009; Yuan et al., 2009; Hansen et al., 2010), mutations in this area could affect the single channel properties of the receptor. Interestingly, it may be receptor physiology itself that is the crucial determinant of antibody binding. In agreement with this hypothesis, single channel recordings of these GluN1 variants show that receptor closed time is inversely correlated with patients’ antibody staining. Furthermore, if antibodies specifically recognize an ATD conformation that is closed for shorter durations, their binding in this crucial hinge region may impact receptor physiology. This concept is confirmed by the observation that acute application of patient CSF prolongs receptor open time in response to agonist, suggesting that patients’ antibodies bind to a receptor that is more prone to opening and then act to stabilize the open state (Figure 2E) (Gleichman et al., 2012).
NMDA receptor activity in physiologic and pathologic circumstances
The distinct constellation of symptoms observed in patients with anti-NMDAR encephalitis is reflected in the known roles of NMDA receptors in physiology, pathology, and pharmacology. NMDARs play a crucial role in learning and memory; their activation has repeatedly been shown both pharmacologically and genetically (Miyamoto, 2006) to be necessary for the induction of long-term potentiation (LTP) and long-term depression (LTD), thought to be the molecular correlates of memory formation (Tsien, 2000). NMDAR activity is required for both memory acquisition and consolidation (Robbins and Murphy, 2006; Wang et al., 2006) , and an absence of NMDAR-mediated current has been shown to play a role in the decreased LTP observed in aged mice (Robillard et al., 2011). Therefore, autoantibody-mediated decreases in NMDAR-mediated LTP may explain the fact that patients with anti-NMDAR encephalitis are generally amnestic for all or most of their experience (Figure 1C) (Dalmau et al., 2008).
Similarly, NMDAR hypofunction has long been hypothesized to underlie schizophrenia (Olney JW, 1995). NMDAR function appears to be decreased in affected regions of postmortem schizophrenic brain (Hahn et al., 2006), and a number of polymorphisms that increase the risk of schizophrenia can act to decrease NMDAergic tone (Bennett, 2009). NMDAR protein levels themselves may also be affected, although many of these results are disputed (Kristiansen et al., 2007). Drugs that decrease NMDAR function, including the antagonists ketamine, MK-801 and phencyclidine (PCP), can induce psychosis in unaffected individuals and trigger relapses in schizophrenic patients (Belsham, 2001). Decreasing NMDAR function genetically with a 90–95% decrease of the obligatory NMDAR subunit GluN1 induces endophenotypes of schizophrenia in mice, including increased activity levels and altered social interactions (Mohn et al., 1999). Two main characteristics of schizophrenia are paranoia and hallucinations, both so-called “positive” symptoms of the disorder. These symptoms are also conspicuous features of anti-NMDAR encephalitis (Figure 1C). Later features of anti-NMDAR encephalitis include catatonia, with reduced purposeful movement and lack of responsiveness to the environment, as may be seen in patients with schizophrenia or other psychiatric disorders (Dalmau et al., 2008).
Over-activation of NMDARs has been implicated in a number of pathologic situations: excess calcium entry through NMDARs can cause excitotoxicity, leading to neuronal damage and death in a variety of neurodegenerative disorders including stroke (Arundine and Tymianski, 2004), Huntington’s disease (Fan and Raymond, 2007), Alzheimer’s disease (Hynd et al., 2004), Parkinson’s disease (Koutsilieri and Riederer, 2007), HIV-associated neurocognitive disorders (Cook et al., 2011), and others. Because of this, a significant amount of time and effort has gone into developing clinically useful NMDAR antagonists. However, while many of these antagonists successfully reduced or eliminated damage in animal models, all failed in clinical trials, in large part due to their unacceptable side effects, including psychosis (Wood PL, 1997; Lai et al., 2011). This double-edged sword demonstrates the importance of carefully calibrated NMDAergic activity in normal neurologic function.
Although the autonomic roles of NMDARs are less well studied, there is considerable evidence that NMDARs have a significant role in autonomic stability and ventilation (Waters and Machaalani, 2005). GluN1 knockout mice die within one day due to a failure of respiration (Forrest et al., 1994), and NMDA receptor modulation in respiratory neurons has bidirectional effects on respiration: NMDAR activation through NMDA injection speeds respiration, while NMDAR inhibition decreases phrenic nerve activity (Bonham, 1995). Similarly, pharmacologic NMDAR inhibition via PCP in humans can result in coma (Baldridge EB, 1990), which is particularly reminiscent of the autonomic instability seen in anti-NMDAR encephalitis patients (Figure 1C) (Dalmau et al., 2011) . These data provide yet more evidence that antibody-mediated NMDAR hypofunction may explain many of the symptoms found in anti-NMDAR encephalitis.
Other syndromes associated with anti-NMDAR antibodies
Anti-NMDAR antibodies have now been reported in a variety of other neurological disorders, including isolated case reports describing these antibodies in opsoclonus-myoclonus syndrome, narcolepsy with psychosis, and bipolar disorder with hemidystonia (Smith et al., 2011; Tsutsui et al., 2012; Choe et al., 2013). Antibodies to NMDARs are now also being reported in CNS disorders not previously believed to have an autoimmune component, such as schizophrenia, Creutzfeldt-Jakob disease and HSV encephalitis (Fujita et al., 2012; Prüss et al., 2012a; Hacohen et al., 2013; Steiner et al., 2013).It is not clear that this recent plethora of studies, reporting wide-ranging associations with anti-NMDAR antibodies, adhere to the same strict criteria that defined the original syndrome, which include: (1) immunohistochemistry of brain tissue optimized for cell-surface antigens resulting in a highly sensitive test with a highly specific immunostaining pattern; and (2) a cell-based assay using HEK cells expressing the GluN1 subunit of the NMDAR to detect IgG antibodies . With these criteria, Dalmau and collaborators have identified no IgG GluN1 antibodies in approximately 8000 patients with varied neurological and other disorders, including 80 patients with schizophrenia, or in 200 normal control subjects(Titulaer et al., 2013a). Thus, GluN1 IgG antibodies, defined strictly, appear to be specific for the syndrome of anti-NMDAR encephalitis. GluN1 IgM or IgA antibodies may not be as specific as these IgG antibodies, and have been reported in a few other syndromes, including schizophrenia, dementia, and HSV encephalitis (Prüss et al., 2012a, 2012b; Höftberger et al., 2013; Steiner et al., 2013).
Anti-NMDAR antibodies associated with schizophrenia or isolated psychosis
As discussed above, NMDAR hypofunction is hypothesized to underlie schizophrenia, and psychosis is a core feature of anti-NDMAR encephalitis. Thus, it seems reasonable to speculate that anti-NDMAR antibodies may result in isolated psychosis or a schizophrenia-like illness in some individuals. Recently, a subgroup analysis of Dalmau’s large anti-NMDAR cohort was published (Kayser et al., 2013). Of the 571 patients in the original cohort, 23 (4%) developed isolated psychiatric episodes; five of these were at disease onset and 18 were during relapse. Greater than 80 % had full or substantial recovery after immunotherapy and tumor resection when appropriate. These results suggest that there is a rare subtype of anti-NMDAR encephalitis consisting of isolated psychosis. However, it is not yet known how often patients with isolated psychosis or schizophrenia have anti-NMDAR antibodies. Multiple studies have attempted to address this question, but the results have been mixed. In 2011, Zandi et al. reported that three out of 46 patients with new onset isolated psychosis had detectable anti-NDMAR antibodies in their serum; one of these patients was treated with immunotherapy and had a positive response (Zandi et al., 2011). A subsequent study reported that four out of 51 patients with schizophrenia or schizophreniform disorders had anti-NDMAR antibodies(Tsutsui et al., 2012). However, three of these antibody-positive patients had other features, such as seizures and/or ovarian teratoma, that made a diagnosis of typical anti-NMDAR encephalitis more likely. In 2013, a study of 121 patients with schizophrenia found diverse anti-NMDAR antibodies in 15 subjects (Steiner et al., 2013). However, only two had IgG antibodies specific to GluN1a, and these were subsequently re-classified as having typical anti-NMDAR encephalitis. The other patients had IgA or IgM antibodies, or antibodies that were not specific to GluN1a alone. In addition to these potentially positive studies, there have been several negative studies. In one study of seven patients with chronic schizophrenia, no patients were found to have antibodies to GluN1(Rhoads et al., 2011). In a larger study, 80 patients with newly diagnosed schizophrenia, as defined by one year of symptoms, were also negative for IgG antibodies to GluN1 in serum collected at the onset of their symptoms (Masdeu et al., 2012). These conflicting results likely are due to variation how the psychotic syndromes were classified, differences in the timing of when samples were collected relative to onset of psychiatric symptoms, variations laboratory methodology, the inclusion of non-IgG antibodies and binding to NMDAR subunits other than GluN1, and the study of serum rather than CSF. A conclusive answer to this question may require specifically designed prospective studies examining CSF in patients with strictly defined new onset psychosis.
Anti-NMDAR antibodies associated with dementia
Of particular interest is a recent report by Prusse et al. (2012) describing IgA, but not IgG, antibodies against the NMDAR in seven patients with a rapidly progressive dementia resembling a primary degenerative disorder. Patients developed progressive, disabling, cognitive impairment over 1–2 years. Most had cerebral atrophy on MRI, but normal EEGs and CSF studies. Tumors were found in a minority of patients. In the index patient, there was a transient, but dramatic improvement following plasmapheresis, associated with a substantial decrease of the IgA-NMDAR antibody titer and improved of cerebral metabolism as measured via PET.
Patient serum was shown to have IgA, but not IgG antibodies that recognized the GluN1 subunit of the NMDAR. Application of patient serum to cultured hippocampal neurons resulted in a reversible loss of surface NMDARs reminiscent to that seen in the IgG-mediated encephalitis. However, in contrast to IgG-mediated encephalitis, application of patient serum also affected the expression of other synaptic proteins such synaptophysin, synapsin , VGLUT1 and VGAT. Network-driven, spontaneous activity was also impaired, with significantly reduced incidence and mean amplitude of sEPSCs.
The more protracted clinical course and global effect of NMDAR-IgA on synapses is markedly different from the rapidly progressive and clinically distinct syndrome associated with NMDAR-IgG antibodies, as well as from the more specific action of the IgG antibodies on NMDARs. These differences suggest that NMDAR-IgA has a distinct impact upon NMDARs and synapses, possibly mediated by recognition of a different epitope(s). Alternatively, these differences could be due to non-specific destructive properties of these antibodies, or due to the presence in patient serum of a more extensive repertoire of antibodies against other as yet unknown synaptic proteins. In addition, IgA-NMDAR antibodies may not be directly pathogenic, but may rather represent a secondary immune response against synaptic proteins that were exposed to the immune system as a result of another neurodegenerative process. Clearly, further work investigating these possibilities is needed, but the possibility remains that this novel syndrome may prove to be a newly identified and treatable form of dementia.
Anti-NMDAR antibodies associated with herpes simplex encephalitis
In addition, NMDAR antibodies of the IgA, IgG, or IgM subtypes were recently reported in the serum or CSF of 30% of patients with herpes simplex virus (HSV) encephalitis.(Prüss et al., 2012a) The application of antibody-positive sera to cultured hippocampal neurons resulted in reduction of membrane GluN1, as well as reduction in the number of presynaptic puncta, an effect similar to that reported for the autoantibodies in NMDAR-IgA associated dementia. There were not obvious differences in the acute symptoms of antibody positive and antibody negative HSV encephalitis patients. The most likely explanation for the presence of these antibodies is that virus-induced neuronal injury may expose antigens, such as the NMDAR, normally shielded from systemic immunity. A second possibility is molecular mimicry, where the immune response to a HSV protein is misdirected, attacking a structurally similar epitope in the NMDAR (although no such epitope is known to exist) (Höftberger et al., 2013) .
HSV encephalitis typically is a severe, monophasic limbic encephalitis, often with necrosis of limbic structures, resulting in seizures, personality change, memory dysfunction and other focal neurological deficits(Höftberger et al., 2013). The disorder is diagnosed via PCR of spinal fluid and is treated with antiviral agents. In approximately one quarter of patients, often children, there is a recurrent episode after treatment of the initial infection (Kimura H, Aso K, Kuzushima K, Hanada N, Shibata M, 1992; Ito et al., 2000; De Tiege et al., 2003). In a subset of relapsing patients, HSV is no longer detected via PCR and symptoms do not respond to antivirals. This subacute syndrome typically occurs 1–2 months after the initial episode of HSV encephalitis. In children, dyskinesias and choreoathetosis, similar to the movement disorder seen in anti-NMDAR encephalitis, are prominent symptoms, although in adults cognitive and psychiatric symptoms are more common (Joos et al., 2003; Sköldenberg et al., 2006). Although anti-NMDAR antibodies do not appear to be related to clear clinical differences during the initial acute phase of HSV encephalitis, it may be that relapse or choreoathetosis post-herpes simplex encephalitis is, in fact, a form of anti-NMDAR encephalitis (Armangue et al., 2012; Prüss et al., 2012a; Höftberger et al., 2013).
Anti-AMPA receptor encephalitis
Clinical presentation and treatment of anti-AMPAR encephalitis
Anti-AMPA receptor encephalitis, in which patients develop antibodies against the AMPA-type glutamate receptor, was first described in 2009 in a cohort of 10 patients with limbic encephalitis (LE)(Lai et al., 2009). Nine patients presented with short-term memory loss and confusion, while four patients had seizures. Seven patients had tumors, indicating that anti-AMPAR encephalitis may be a paraneoplastic disorder, although there was little consistency in tumor type (thymic carcinoma, malignant thymoma, non-small cell lung cancer, thymoma, small cell lung cancer, and two patients with breast cancer). Nine of the ten patients were females over the age of 35. Patient CSF contained antibodies to the AMPA receptor subunits GluA1, GluA2, or both. Since the initial report, there has been one additional report of four AMPAR encephalitis cases(Graus et al., 2010), as well as a case study of one additional patient(Bataller et al., 2010). All five of these additional patients were women over 50 years old; three of these patients had LE and tumors (two patients with small cell lung cancer, one with breast cancer), but interestingly, two other patients had no history of LE and symptomatology more closely resembling acute-onset psychosis rather than memory loss and seizures. All patients responded well to corticosteroids or IVIG and chemotherapy, indicating an immune basis to symptoms.
Based on the number of patients identified thus far, it appears that AMPAR encephalitis is far less common than NMDAR encephalitis; however, given the current state of uncertainty as to which symptoms should act as diagnostic indicators of AMPAR encephalitis, it is possible that the disease is currently underdiagnosed. The syndrome can be treated in many individuals with tumor removal or plasmapheresis, suggesting that it is mediated directly by antibodies(Lai et al., 2009). However, when compared to anti-NMDAR encephalitis, the underlying neoplasms are more diverse, response is less complete, and relapses are more common.
Effects of patients’ antibodies on AMPARs
Antibodies and CSF from patients cause internalization of AMPARs in model systems, resulting in loss of receptors from the neuronal surface, analogous to the pathogenesis of myasthenia gravis and anti-NMDAR encephalitis. Internalization of AMPARs can be demonstrated both by immunostaining of unpermeabilized neurons to label surface receptors followed by fluorescent microscopy as well as by traditional biochemical approaches such as surface biotinylation (Lai et al., 2009). Although the antibodies react with GluR1, GluR2 or both, the exact site of immunogenicity is not known, although our preliminary data have identified a region of GluR1/GluR2 in the amino terminal domain (ATD) that may contribute to immunoreactivity to patient-generated AMPAR antibodies (unpublished observations).
AMPA receptor activity in physiologic and pathologic circumstances
To date, anti-AMPAR encephalitis has been associated primarily with memory loss, seizures, and psychosis (Lai et al., 2009; Bataller et al., 2010; Graus et al., 2010); all of these symptoms, to varying degrees, reflect known functions of AMPAR activity. AMPARs traffic rapidly in and out of the membrane in an activity-dependent manner, tightly controlling the excitability of synapses. This trafficking appears to play a major role in learning and memory. Membrane insertion of GluA1-containing, GluA2-lacking receptors is elevated in a number of learning paradigms and is frequently necessary for the development of LTP (Plant et al., 2006; Whitlock et al., 2006; Makino and Malinow, 2009), although the exact GluA subunit requirements differ somewhat across different learning tasks (Kessels and Malinow, 2009; Keifer and Zheng, 2010). Blocking AMPAR receptor insertion blocks LTP(Kopec et al., 2007) and learning (Li et al., 2005; Slipczuk et al., 2009), while enhancing AMPAR synaptic delivery enhances LTP and improves learning (Lynch, 2004; Knafo et al., 2012). Thus, this syndrome mimics the loss of surface AMPAR seen in LTD in model systems and if understood provides a manner to link clinical and basic understanding of glutamatergic transmission and its role in synaptic remodeling.
Antibodies to the AMPAR have also been associated with Rasmussen’s encephalitis, a childhood onset severe progressive epilepsy; in a subset of cases, these individuals make antibodies to the S2 region of GluA3 that may act to activate the receptor (Twyman et al., 1995), although subsequent work has shown that these antibodies are not present in the majority of cases(Watson et al., 2004). It is not yet clear whether anti-AMPAR encephalitis overlaps at all with the subjects with Rasmussen’s encephalitis who have antibodies to GluA3 (Twyman et al., 1995; Ganor et al., 2005; Takahashi et al., 2005). In the modest size group of individuals presently identified, anti-AMPAR encephalitis does not have any association with GluA3 antibodies
Conclusions
Although considerable work remains to fully understand these complex disorders, our understanding of the biology of anti-NMDAR and anti-AMPAR encephalitis has grown exponentially since they were first described just over 7 years ago.
The discovery of various forms of encephalitis mediated by antibodies targeted to synaptic proteins is revolutionizing neurology. As such antibodies typically interfere with critical events in synaptic transmission or modulation, they readily link basic science and clinical medicine, and understanding these disorders provides important insight into not only their clinical features but also basic neuroscience. With these diseases, for the first time in humans we are able to appreciate the impact of sudden but sustained loss of specific neurotransmitter receptors. For the encephalitides directed to glutamatergic receptors, improved understanding of their pathophysiology might not only lead to better treatments, but lead to novel insights into human glutamate receptor function and understanding of their roles in learning, memory, neurological and psychiatric disease.
References
- Armangue T, Titulaer MJ, Málaga I, Bataller L, Gabilondo I, Graus F, Dalmau J. Pediatric Anti-N-methyl-D-Aspartate Receptor Encephalitis-Clinical Analysis and Novel Findings in a Series of 20 Patients. J Pediatr. 2012 doi: 10.1016/j.jpeds.2012.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arundine M, Tymianski M. Molecular mechanisms of glutamate-dependent neurodegeneration in ischemia and traumatic brain injury. Cell Mol Life Sci. 2004;61:657–668. doi: 10.1007/s00018-003-3319-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baldridge EB, Bensen HA. Phencyclidine. Emerg Med Clin North Am. 1990;8:541–550. [PubMed] [Google Scholar]
- Bataller L, Galiano R, Garcia-Escrig M, Martinez M, Sevilla T, Vilchez J, Dalmau J. Reversible paraneoplastic limbic encephalitis associated with antibodies to the AMPA receptor. Neurology. 2010;74:265–267. doi: 10.1212/WNL.0b013e3181cb3e52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belsham B. Glutamate and its role in psychiatric illness. Hum Psychopharmacol. 2001;16:139–146. doi: 10.1002/hup.279. [DOI] [PubMed] [Google Scholar]
- Bennett M. Positive and negative symptoms in schizophrenia: the NMDA receptor hypofunction hypothesis, neuregulin/ErbB4 and synapse regression. Aust N Z J Psychiatry. 2009;43:711–721. doi: 10.1080/00048670903001943. [DOI] [PubMed] [Google Scholar]
- Bien CG, Vincent A, Barnett MH, Becker AJ, Blümcke I, Graus F, Jellinger KA, Reuss DE, Ribalta T, Schlegel J, Sutton I, Lassmann H, Bauer J. Immunopathology of autoantibody-associated encephalitides: clues for pathogenesis. Brain. 2012;135:1622–1638. doi: 10.1093/brain/aws082. [DOI] [PubMed] [Google Scholar]
- Bonham AC. Neurotransmitters in the CNS control of breathing. Respir Physiol. 1995;101:219–230. doi: 10.1016/0034-5687(95)00045-f. [DOI] [PubMed] [Google Scholar]
- Choe C-U, Karamatskos E, Schattling B, Leypoldt F, Liuzzi G, Gerloff C, Friese Ma, Mulert C. A clinical and neurobiological case of IgM NMDA receptor antibody associated encephalitis mimicking bipolar disorder. Psychiatry Res. 2013;208:194–196. doi: 10.1016/j.psychres.2012.09.035. [DOI] [PubMed] [Google Scholar]
- Cook DR, Gleichman AJ, Cross SA, Doshi S, Ho W, Jordan-Sciutto KL, Lynch DR, Kolson DL. NMDA receptor modulation by the neuropeptide apelin: implications for excitotoxic injury. J Neurochem. 2011;118:1113–1123. doi: 10.1111/j.1471-4159.2011.07383.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalmau J, Gleichman AJ, Hughes EG, Rossi JE, Peng X, Lai M, Dessain SK, Rosenfeld MR, Balice-Gordon R, Lynch DR. Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol. 2008;7:1091–1098. doi: 10.1016/S1474-4422(08)70224-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalmau J, Lancaster E, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R. Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. Lancet Neurol. 2011;10:63–74. doi: 10.1016/S1474-4422(10)70253-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalmau J, Tüzün E, Wu H, Masjuan J, Rossi JE, Voloschin A, Baehring JM, Shimazaki H, Koide R, King D, Mason W, Sansing LH, Dichter MA, Rosenfeld MR, Lynch DR. Paraneoplastic anti-N-methyl-D-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol. 2007;61:25–36. doi: 10.1002/ana.21050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Tiege X, Rozenberg F, Portes V, Des Lobut JB, Lebon P, Ponsot GHB. Herpes simplex encephalitis relapses in children: Differentiation of two neurologic entities. Neurology. 2003;61:241–243. doi: 10.1212/01.wnl.0000073985.71759.7c. [DOI] [PubMed] [Google Scholar]
- Fan MMY, Raymond LA. N-methyl-D-aspartate (NMDA) receptor function and excitotoxicity in Huntington’s disease. Prog Neurobiol. 2007;81:272–293. doi: 10.1016/j.pneurobio.2006.11.003. [DOI] [PubMed] [Google Scholar]
- Farina AN, Blain KY, Maruo T, Kwiatkowski W, Choe S, Nakagawa T. Separation of domain contacts is required for heterotetrameric assembly of functional NMDA receptors. J Neurosci. 2011;31:3565–3579. doi: 10.1523/JNEUROSCI.6041-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Florance NR, Davis RL, Lam C, Szperka C, Zhou L, Ahmad S, Campen CJ, Moss H, Peter N, Gleichman AJ, Glaser CA, Lynch DR, Rosenfeld MR, Dalmau J. Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis in children and adolescents. Ann Neurol. 2009;66:11–18. doi: 10.1002/ana.21756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forrest D, Yuzaki M, Soares HD, Ng L, Luk DC, Sheng M, Stewart CL, Morgan JI, Connor JA, Curran T. Targeted disruption of NMDA receptor 1 gene abolishes NMDA response and results in neonatal death. Neuron. 1994;13:325–338. doi: 10.1016/0896-6273(94)90350-6. [DOI] [PubMed] [Google Scholar]
- Fujita K, Yuasa T, Takahashi Y, Tanaka K, Sako W, Koizumi H, Iwasaki Y, Yoshida M, Izumi Y, Kaji R. Antibodies to N-methyl-D-aspartate glutamate receptors in Creutzfeldt-Jakob disease patients. J Neuroimmunol. 2012;251:90–93. doi: 10.1016/j.jneuroim.2012.06.010. [DOI] [PubMed] [Google Scholar]
- Furukawa H. Structure and function of glutamate receptor amino terminal domains. J Physiol. 2012;590:63–72. doi: 10.1113/jphysiol.2011.213850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gable MS, Sheriff H, Dalmau J, Tilley DH, Glaser CA. The frequency of autoimmune N-methyl-D-aspartate receptor encephalitis surpasses that of individual viral etiologies in young individuals enrolled in the California Encephalitis Project. Clin Infect Dis. 2012;54:899–904. doi: 10.1093/cid/cir1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganor Y, Goldberg-Stern H, Lerman-Sagie T, Teichberg VI, Levite M. Autoimmune epilepsy: distinct subpopulations of epilepsy patients harbor serum autoantibodies to either glutamate/AMPA receptor GluR3, glutamate/NMDA receptor subunit NR2A or double-stranded DNA. Epilepsy Res. 2005;65:11–22. doi: 10.1016/j.eplepsyres.2005.03.011. [DOI] [PubMed] [Google Scholar]
- Gielen M, Siegler Retchless B, Mony L, Johnson JW, Paoletti P. Mechanism of differential control of NMDA receptor activity by NR2 subunits. Nature. 2009;459:703–707. doi: 10.1038/nature07993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gleichman AJ, Spruce LA, Dalmau J, Seeholzer SH, Lynch DR. Anti-NMDA receptor encephalitis antibody binding is dependent on amino acid identity of a small region within the GluN1 amino terminal domain. J Neurosci. 2012;32:11082–11094. doi: 10.1523/JNEUROSCI.0064-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Granerod J, Ambrose HE, Davies NW, Clewley JP, Walsh AL, Morgan D, Cunningham R, Zuckerman M, Mutton KJ, Solomon T, Ward KN, Lunn MP, Irani SR, Vincent A, Brown DW, Crowcroft NS. Causes of encephalitis and differences in their clinical presentations in England: a multicentre, population-based prospective study. Lancet Infect Dis. 2010;10:835–844. doi: 10.1016/S1473-3099(10)70222-X. [DOI] [PubMed] [Google Scholar]
- Graus F, Boronat A, Xifró X, Boix M, Svigelji V, Garcia A, Palomino A, Sabater L, Alberch J, Saiz A. The expanding clinical profile of anti-AMPA receptor encephalitis. Neurology. 2010;74:857–859. doi: 10.1212/WNL.0b013e3181d3e404. [DOI] [PubMed] [Google Scholar]
- Graus F, Keime-Guibert F, Reñe R, Benyahia B, Ribalta T, Ascaso C, Escaramis G, Delattre JY. Anti-Hu-associated paraneoplastic encephalomyelitis: analysis of 200 patients. Brain. 2001;124:1138–1148. doi: 10.1093/brain/124.6.1138. [DOI] [PubMed] [Google Scholar]
- Hacohen Y, Deiva K, Pettingill P, Waters P, Siddiqui A, Chretien P, Menson E, Lin J-P, Tardieu M, Vincent A, Lim MJ. N-methyl-D-aspartate receptor antibodies in post-herpes simplex virus encephalitis neurological relapse. Mov Disord. 2013;00:1–7. doi: 10.1002/mds.25626. [DOI] [PubMed] [Google Scholar]
- Hahn C-G, Wang H-Y, Cho D-S, Talbot K, Gur RE, Berrettini WH, Bakshi K, Kamins J, Borgmann-Winter KE, Siegel SJ, Gallop RJ, Arnold SE. Altered neuregulin 1-erbB4 signaling contributes to NMDA receptor hypofunction in schizophrenia. Nat Med. 2006;12:824–828. doi: 10.1038/nm1418. [DOI] [PubMed] [Google Scholar]
- Hansen KB, Furukawa H, Traynelis SF. Control of Assembly and Function of Glutamate Receptors by the Amino-Terminal Domain. Mol Pharmacol. 2010;78:535–549. doi: 10.1124/mol.110.067157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Höftberger R, Armangue T, Leypoldt F, Graus F, Dalmau J. Clinical Neuropathology practice guide 4-2013: post-herpes simplex encephalitis: N-methyl-Daspartate receptor antibodies are part of the problem. Clin Neuropathol. 2013;32:251–254. doi: 10.5414/NP300666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes EG, Peng X, Gleichman AJ, Lai M, Zhou L, Tsou R, Parsons TD, Lynch DR, Dalmau J, Balice-Gordon RJ. Cellular and synaptic mechanisms of anti-NMDA receptor encephalitis. J Neurosci. 2010;30:5866–5875. doi: 10.1523/JNEUROSCI.0167-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutchinson M, Waters P, McHugh J. Progressive encephalomyelitis, rigidity, and myoclonus: a novel glycine receptor antibody. Neurology. 2008:1289–1292. doi: 10.1212/01.wnl.0000327606.50322.f0. [DOI] [PubMed] [Google Scholar]
- Hynd MR, Scott HL, Dodd PR. Glutamate-mediated excitotoxicity and neurodegeneration in Alzheimer’s disease. Neurochem Int. 2004;45:583–595. doi: 10.1016/j.neuint.2004.03.007. [DOI] [PubMed] [Google Scholar]
- Irani SR, Bera K, Waters P, Zuliani L, Maxwell S, Zandi MS, Friese MA, Galea I, Kullmann DM, Beeson D, Lang B, Bien CG, Vincent A. N-methyl-D-aspartate antibody encephalitis: temporal progression of clinical and paraclinical observations in a predominantly non-paraneoplastic disorder of both sexes. Brain. 2010;133:1655–1667. doi: 10.1093/brain/awq113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito Y, Kimura H, Yabuta Y, Ando Y, Murakami T, Shiomi M, Morishima T. Exacerbation of herpes simplex encephalitis after successful treatment with acyclovir. Clin Infect Dis. 2000;30:185–187. doi: 10.1086/313618. [DOI] [PubMed] [Google Scholar]
- Jin R, Singh SK, Gu S, Furukawa H, Sobolevsky AI, Zhou J, Jin Y, Gouaux E. Crystal structure and association behaviour of the GluR2 amino-terminal domain. EMBO J. 2009;28:1812–1823. doi: 10.1038/emboj.2009.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joos AAB, Ziyeh S, Rauer S, Keller E, Huzly D, Lücking CH. Postinfectious Autoimmune-Mediated Encephalitis Eight Months after Herpes simplex Encephalitis. Eur Neurol. 2003;50:54–56. doi: 10.1159/000070860. [DOI] [PubMed] [Google Scholar]
- Karakas E, Simorowski N, Furukawa H. Structure of the zinc-bound amino-terminal domain of the NMDA receptor NR2B subunit. EMBO J. 2009;28:3910–3920. doi: 10.1038/emboj.2009.338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karakas E, Simorowski N, Furukawa H. Subunit arrangement and phenylethanolamine binding in GluN1/GluN2B NMDA receptors. Nature. 2011;475:249–253. doi: 10.1038/nature10180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashyape P, Taylor E, Ng J, Krishnakumar D, Kirkham F, Whitney A. Successful treatment of two paediatric cases of anti-NMDA receptor encephalitis with cyclophosphamide: the need for early aggressive immunotherapy in tumour negative paediatric patients. Eur J Paediatr Neurol. 2012;16:74–78. doi: 10.1016/j.ejpn.2011.07.005. [DOI] [PubMed] [Google Scholar]
- Kayser M, Dalmau J. The emerging link between autoimmune disorders and neuropsychiatric disease. J neuropsychiatry Clin. 2011:1–12. doi: 10.1176/appi.neuropsych.23.1.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kayser MS, Titulaer MJ, Gresa-Arribas N, Dalmau J. Frequency and Characteristics of Isolated Psychiatric Episodes in Anti-N-Methyl-d-Aspartate Receptor Encephalitis. JAMA Neurol. 2013;70:1133–1139. doi: 10.1001/jamaneurol.2013.3216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keifer J, Zheng Z. AMPA receptor trafficking and learning. Eur J Neurosci. 2010;32:269–277. doi: 10.1111/j.1460-9568.2010.07339.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keime-Guibert F, Graus F, Fleury A, René R, Honnorat J, Broet P, Delattre JY. Treatment of paraneoplastic neurological syndromes with antineuronal antibodies (Anti-Hu, anti-Yo) with a combination of immunoglobulins, cyclophosphamide, and methylprednisolone. J Neurol Neurosurg Psychiatry. 2000;68:479–482. doi: 10.1136/jnnp.68.4.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kessels HW, Malinow R. Synaptic AMPA receptor plasticity and behavior. Neuron. 2009;61:340–350. doi: 10.1016/j.neuron.2009.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura H, Aso K, Kuzushima K, Hanada N, Shibata M, Morishima T. Relapse of herpes simplex encephalitis in children. Pediatrics. 1992;89:891–894. [PubMed] [Google Scholar]
- Knafo S, Venero C, Sánchez-Puelles C, Pereda-Peréz I, Franco A, Sandi C, Suárez LM, Solís JM, Alonso-Nanclares L, Martín ED, Merino-Serrais P, Borcel E, Li S, Chen Y, Gonzalez-Soriano J, Berezin V, Bock E, Defelipe J, Esteban JA. Facilitation of AMPA receptor synaptic delivery as a molecular mechanism for cognitive enhancement. PLoS Biol. 2012;10:e1001262. doi: 10.1371/journal.pbio.1001262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koike H, Tanaka F, Sobue G. Paraneoplastic neuropathy: wide-ranging clinicopathological manifestations. Curr Opin Neurol. 2011;24:504–510. doi: 10.1097/WCO.0b013e32834a87b7. [DOI] [PubMed] [Google Scholar]
- Kopec CD, Real E, Kessels HW, Malinow R. GluR1 links structural and functional plasticity at excitatory synapses. J Neurosci. 2007;27:13706–13718. doi: 10.1523/JNEUROSCI.3503-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koutsilieri E, Riederer P. Excitotoxicity and new antiglutamatergic strategies in Parkinson’s disease and Alzheimer's disease. Parkinsonism Relat Disord. 2007;13(Suppl 3):S329–31. doi: 10.1016/S1353-8020(08)70025-7. [DOI] [PubMed] [Google Scholar]
- Kristiansen LV, Huerta I, Beneyto M, Meador-Woodruff JH. NMDA receptors and schizophrenia. Curr Opin Pharmacol. 2007;7:48–55. doi: 10.1016/j.coph.2006.08.013. [DOI] [PubMed] [Google Scholar]
- Lai M, Hughes EG, Peng X, Zhou L, Gleichman AJ, Shu H, Matà S, Kremens D, Vitaliani R, Geschwind MD, Bataller L, Kalb RG, Davis R, Graus F, Lynch DR, Balice-Gordon R, Dalmau J. AMPA receptor antibodies in limbic encephalitis alter synaptic receptor location. Ann Neurol. 2009;65:424–434. doi: 10.1002/ana.21589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai M, Huijbers MGM, Lancaster E, Graus F, Bataller L, Balice-Gordon R, Cowell JK, Dalmau J. Investigation of LGI1 as the antigen in limbic encephalitis previously attributed to potassium channels: a case series. Lancet Neurol. 2010;9:776–785. doi: 10.1016/S1474-4422(10)70137-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai TW, Shyu W-C, Wang YT. Stroke intervention pathways: NMDA receptors and beyond. Trends Mol Med. 2011;17:266–275. doi: 10.1016/j.molmed.2010.12.008. [DOI] [PubMed] [Google Scholar]
- Lancaster E, Huijbers MGM, Bar V, Boronat A, Wong A, Martinez-Hernandez E, Wilson C, Jacobs D, Lai M, Walker RW, Graus F, Bataller L, Illa I, Markx S, Strauss KA, Peles E, Scherer SS, Dalmau J. Investigations of caspr2, an autoantigen of encephalitis and neuromyotonia. Ann Neurol. 2011a;69:303–311. doi: 10.1002/ana.22297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lancaster E, Lai M, Peng X, Hughes E, Constantinescu R, Raizer J, Friedman D, Skeen MB, Grisold W, Kimura A, Ohta K, Iizuka T, Guzman M, Graus F, Moss SJ, Balice-Gordon R, Dalmau J. Antibodies to the GABA(B) receptor in limbic encephalitis with seizures: case series and characterisation of the antigen. Lancet Neurol. 2010;9:67–76. doi: 10.1016/S1474-4422(09)70324-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lancaster E, Titulaer MJ, Boulos M, Weaver S, Liebers E, Kornblum C, Bien CG, Honnorat J, Wong S, Xu J, Contractor A, Dalmau J. Antibodies to metabotropic glutamate receptor 5 in the Ophelia syndrome. Neurolo. 2011b:77. doi: 10.1212/WNL.0b013e3182364a44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q, Roberts AC, Glanzman DL. Synaptic facilitation and behavioral dishabituation in Aplysia: dependence on release of Ca2+ from postsynaptic intracellular stores, postsynaptic exocytosis, and modulation of postsynaptic AMPA receptor efficacy. J Neurosci. 2005;25:5623–5637. doi: 10.1523/JNEUROSCI.5305-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindstrom J. Autoimmune diseases involving nicotinic receptors. J Neurobiol. 2002;53:656–665. doi: 10.1002/neu.10106. [DOI] [PubMed] [Google Scholar]
- Lynch MA. Long-Term Potentiation and Memory. Phsyiol Rev. 2004;84:87–136. doi: 10.1152/physrev.00014.2003. [DOI] [PubMed] [Google Scholar]
- Makino H, Malinow R. AMPA receptor incorporation into synapses during LTP: the role of lateral movement and exocytosis. Neuron. 2009;64:381–390. doi: 10.1016/j.neuron.2009.08.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mangler M, Trebesch de Perez I, Teegen B, Stöcker W, Prüss H, Meisel A, Schneider A, Vasiljeva J, Speiser D. Seroprevalence of anti-N-methyl-D-aspartate receptor antibodies in women with ovarian teratoma. J Neurol. 2013 doi: 10.1007/s00415-013-7074-0. [DOI] [PubMed] [Google Scholar]
- Martinez-Hernandez E, Horvath J, Shiloh-Malawsky Y, Sangha N, Martinez-Lage M, Dalmau J. Analysis of complement and plasma cells in the brain of patients with anti-NMDAR encephalitis. Neurology. 2011;77:589–593. doi: 10.1212/WNL.0b013e318228c136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masdeu J, Gonzalez-Pinto A, Matute C, Ruiz de Azua S, Palomino A. Serum IgG Antibodies Against the NR1 Subunit of the NMDA Receptor Not Detected in Schizophrenia. Am J Psychiatry. 2012;169:1120–12. doi: 10.1176/appi.ajp.2012.12050646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meddows E, Le Bourdelles B, Grimwood S, Wafford K, Sandhu S, Whiting P, McIlhinney RA. Identification of molecular determinants that are important in the assembly of N-methyl-D-aspartate receptors. J Biol Chem. 2001;276:18795–18803. doi: 10.1074/jbc.M101382200. [DOI] [PubMed] [Google Scholar]
- Mikasova L, De Rossi P, Bouchet D, Georges F, Rogemond V, Didelot A, Meissirel C, Honnorat J, Groc L. Disrupted surface cross-talk between NMDA and Ephrin-B2 receptors in anti-NMDA encephalitis. Brain. 2012;135:1606–1621. doi: 10.1093/brain/aws092. [DOI] [PubMed] [Google Scholar]
- Miyamoto E. Review Molecular Mechanism of Neuronal Plasticity : Induction and Maintenance of Long-Term Potentiation in the Hippocampus. 2006;442:433–442. doi: 10.1254/jphs.cpj06007x. [DOI] [PubMed] [Google Scholar]
- Mohn AR, Gainetdinov RR, Caron MG, Koller BH, Hill C, Carolina N, Hughes H. Mice with Reduced NMDA Receptor Expression Display Behaviors Related to Schizophrenia. Vol. 98. University of North Carolina; Chapel Hill: 1999. pp. 427–436. [DOI] [PubMed] [Google Scholar]
- Olney JWFN. Glutamate receptor dysfunction and schizophrenia. Arch Gen Psychiatry. 1995;52:998–1007. doi: 10.1001/archpsyc.1995.03950240016004. [DOI] [PubMed] [Google Scholar]
- Paoletti P. Molecular basis of NMDA receptor functional diversity. Eur J Neurosci. 2011;33:1351–1365. doi: 10.1111/j.1460-9568.2011.07628.x. [DOI] [PubMed] [Google Scholar]
- Plant K, Pelkey KA, Bortolotto ZA, Morita D, Terashima A, McBain CJ, Collingridge GL, Isaac JTR. Transient incorporation of native GluR2-lacking AMPA receptors during hippocampal long-term potentiation. Nat Neurosci. 2006;9:602–604. doi: 10.1038/nn1678. [DOI] [PubMed] [Google Scholar]
- Prüss H, Finke C, Höltje M, Hofmann J, Klingbeil C, Probst C, Borowski K, Ahnert-Hilger G, Harms L, Schwab JM, Ploner CJ, Komorowski L, Stoecker W, Dalmau J, Wandinger KP. N-methyl-D-aspartate receptor antibodies in herpes simplex encephalitis. Ann Neurol. 2012a;72:902–911. doi: 10.1002/ana.23689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prüss H, Höltje M, Maier N, Gomez A, Buchert R, Harms L, Ahnert-Hilger G, Schmitz D, Terborg C, Kopp U, Klingbeil C, Probst C, Kohler S, Schwab JM, Stoecker W, Dalmau J, Wandinger KP. IgA NMDA receptor antibodies are markers of synaptic immunity in slow cognitive impairment. Neurology. 2012b;78:1743–1753. doi: 10.1212/WNL.0b013e318258300d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhoads J, Guirgis H, McKnight C, Duchemin AM. Lack of anti-NMDA receptor autoantibodies in the serum of subjects with schizophrenia. Schizophr Res. 2011;129:213–214. doi: 10.1016/j.schres.2010.12.018. [DOI] [PubMed] [Google Scholar]
- Robbins TW, Murphy ER. Behavioural pharmacology: 40+ years of progress, with a focus on glutamate receptors and cognition. Trends Pharmacol Sci. 2006;27:141–148. doi: 10.1016/j.tips.2006.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robillard JM, Gordon GR, Choi HB, Christie BR, MacVicar BA. Glutathione restores the mechanism of synaptic plasticity in aged mice to that of the adult. PLoS One. 2011;6:e20676. doi: 10.1371/journal.pone.0020676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson NE, Robinson ZW, Robinson BR, Robinson AL, Robinson JA, Robinson ML, Robinson AB. Structure-dependent nonenzymatic deamidation of glutaminyl and asparaginyl pentapeptides. J Pept Res. 2004;63:426–436. doi: 10.1111/j.1399-3011.2004.00151.x. [DOI] [PubMed] [Google Scholar]
- Sköldenberg B, Aurelius E, Hjalmarsson A, Sabri F, Forsgren M, Andersson B, Linde A, Strannegård O, Studahl M, Hagberg L, Rosengren L. Incidence and pathogenesis of clinical relapse after herpes simplex encephalitis in adults. J Neurol. 2006;253:163–170. doi: 10.1007/s00415-005-0941-6. [DOI] [PubMed] [Google Scholar]
- Slipczuk L, Bekinschtein P, Katche C, Cammarota M, Izquierdo I, Medina JH. BDNF activates mTOR to regulate GluR1 expression required for memory formation. PLoS One. 2009;4:e6007. doi: 10.1371/journal.pone.0006007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith JH, Dhamija R, Moseley BD, Sandroni P, Lucchinetti CF, Lennon VA, Kantarci OH. N-methyl-D-aspartate Receptor Autoimmune Encephalitis Presenting With Opsoclonus-Myoclonus. Arch Neurol. 2011;68:1069–1072. doi: 10.1001/archneurol.2011.166. [DOI] [PubMed] [Google Scholar]
- Sobolevsky AI, Rosconi MP, Gouaux E. X-ray structure, symmetry and mechanism of an AMPA-subtype glutamate receptor. Nature. 2009;462:745–756. doi: 10.1038/nature08624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steiner J, Walter M, Glanz W, Sarnyai Z, Bernstein H-G, Vielhaber S, Kästner A, Skalej M, Jordan W, Schiltz K, Klingbeil C, Wandinger KP, Bogerts B, Stoecker W. Increased prevalence of diverse N-methyl-D-aspartate glutamate receptor antibodies in patients with an initial diagnosis of schizophrenia: specific relevance of IgG NR1a antibodies for distinction from N-methyl-D-aspartate glutamate receptor encephalitis. JAMA Psychiatry. 2013;70:271–278. doi: 10.1001/2013.jamapsychiatry.86. [DOI] [PubMed] [Google Scholar]
- Takahashi Y, Mori H, Mishina M, Watanabe M, Kondo N, Shimomura J, Kubota Y, Matsuda K, Fukushima K, Shiroma N, Akasaka N, Nishida H, Imamura A, Watanabe H, Ikezawa M, Fujiwara T. Autoantibodies and Cell-mediated Autoimmunity to NMDA-type GluR ε 2 in Patients with Rasmussen ’ s Encephalitis and Chronic Progressive Epilepsia Partialis Continua. Epilepsia. 2005;46:152–158. doi: 10.1111/j.1528-1167.2005.01024.x. [DOI] [PubMed] [Google Scholar]
- Titulaer MJ, Kayser MS, Dalmau J. Authors’ reply. Lancet Neurol. 2013a;12:425–426. doi: 10.1016/S1474-4422(13)70072-3. [DOI] [PubMed] [Google Scholar]
- Titulaer MJ, Lang B, Verschuuren JJ. Lambert-Eaton myasthenic syndrome: from clinical characteristics to therapeutic strategies. Lancet Neurol. 2011;10:1098–1107. doi: 10.1016/S1474-4422(11)70245-9. [DOI] [PubMed] [Google Scholar]
- Titulaer MJ, McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, Honig LS, Benseler SM, Kawachi I, Martinez-Hernandez E, Aguilar E, Gresa-Arribas N, Ryan-Florance N, Torrents A, Saiz A, Rosenfeld MR, Balice-Gordon R, Graus F, Dalmau J. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. 2013b;12:157–165. doi: 10.1016/S1474-4422(12)70310-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traynelis SF, Wollmuth LP, Mcbain CJ, Menniti FS, Vance KM, Ogden KK, Hansen KB, Yuan H, Myers SJ, Dingledine R. Glutamate Receptor Ion Channels : Structure , Regulation , and Function. Pharmacol Rev. 2010;62:405–496. doi: 10.1124/pr.109.002451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsien J. Linking Hebb’s coincidence-detection to memory formation. Curr Opin Neurobiol. 2000;10:266–273. doi: 10.1016/s0959-4388(00)00070-2. [DOI] [PubMed] [Google Scholar]
- Tsutsui K, Kanbayashi T, Tanaka K, Boku S, Ito W, Tokunaga J, Mori A, Hishikawa Y, Shimizu T, Nishino S. Anti-NMDA-receptor antibody detected in encephalitis, schizophrenia, and narcolepsy with psychotic features. BMC Psychiatry. 2012;12:37. doi: 10.1186/1471-244X-12-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Twyman RE, Gahring LC, Spiess J, Rogers SW. Glutamate receptor antibodies activate a subset of receptors and reveal an agonist binding site. Neuron. 1995;14:755–762. doi: 10.1016/0896-6273(95)90219-8. [DOI] [PubMed] [Google Scholar]
- Ulusoy C, Tüzün E, Kürtüncü M, Türkoğlu R, Akman-Demir G, Eraksoy M. Comparison of the Cytokine Profiles of Patients With Neuronal-Antibody-Associated Central Nervous System Disorders. Int J Neurosci. 2012;122:284–289. doi: 10.3109/00207454.2011.648762. [DOI] [PubMed] [Google Scholar]
- Wang H, Hu Y, Tsien JZ. Molecular and systems mechanisms of memory consolidation and storage. Prog Neurobiol. 2006;79:123–135. doi: 10.1016/j.pneurobio.2006.06.004. [DOI] [PubMed] [Google Scholar]
- Waters KA, Machaalani R. Role of NMDA receptors in development of respiratory control. Respir Physiol Neurobiol. 2005;149:123–130. doi: 10.1016/j.resp.2005.03.009. [DOI] [PubMed] [Google Scholar]
- Watson R, Jiang Y, Bermudez I, Houlihan L, Clover L, McKnight K, Cross JH, Hart IK, Roubertie A, Valmier J, Hart Y, Palace J, Beeson D, Vincent A, Lang B. Absence of antibodies to glutamate receptor type 3 (GluR3) in Rasmussen encephalitis. Neurology. 2004;63:43–50. doi: 10.1212/01.wnl.0000132651.66689.0f. [DOI] [PubMed] [Google Scholar]
- Whitlock JR, Heynen AJ, Shuler MG, Bear MF. Learning induces long-term potentiation in the hippocampus. Science. 2006;313:1093–1097. doi: 10.1126/science.1128134. [DOI] [PubMed] [Google Scholar]
- Wiley J, Molinuevo JL, Graus F, Guerrero A. Utility of Anti-Hu Antibodies in the Diagnosis of Paraneoplastic Sensory Neuropathy. 1998:976–980. doi: 10.1002/ana.410440620. [DOI] [PubMed] [Google Scholar]
- Wood PL, Hawkinson JE. N-methyl-D-aspartate antagonists for stroke and head trauma. Expert Opin Investig Drugs. 1997;6:389–397. doi: 10.1517/13543784.6.4.389. [DOI] [PubMed] [Google Scholar]
- Yuan H, Hansen KB, Vance KM, Ogden KK, Traynelis SF. Control of NMDA receptor function by the NR2 subunit amino-terminal domain. J Neurosci. 2009;29:12045–12058. doi: 10.1523/JNEUROSCI.1365-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zandi MS, Irani SR, Lang B, Waters P, Jones PB, McKenna P, Coles AJ, Vincent A, Lennox BR. Disease-relevant autoantibodies in first episode schizophrenia. J Neurol. 2011;258:686–688. doi: 10.1007/s00415-010-5788-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Q, Tanaka K, Sun P, Nakata M, Yamamoto R, Sakimura K, Matsui M, Kato N. Suppression of synaptic plasticity by cerebrospinal fluid from anti-NMDA receptor encephalitis patients. Neurobiol Dis. 2012;45:610–615. doi: 10.1016/j.nbd.2011.09.019. [DOI] [PubMed] [Google Scholar]