Abstract
We for the first time investigated the effect and mechanism of the total flavones of Rhododendron simsii Planch (TFR), a widely-used Chinese herb for a thousand years, on vasodilatation and hyperpolarization in middle cerebral artery (MCA) of rats subject to global cerebral ischemia-reperfusion (CIR). TFR (11~2700 mg/L) evoked dose-dependent vasodilation and hyperpolarization in MCA of both sham and CIR that were partially inhibited by 30 μM N-nitro-L-arginine-methyl-ester and 10 μM indomethacin and further attenuated by endogenous H2S synthese-CSE inhibitor PPG (100 μM) or Ca2+-activated potassium channel (Kca) inhibitor TEA (1 mM). In whole-cell patch clamp recording, TFR remarkably enhanced the outward current that was inhibited by TEA. CIR increased CSE mRNA expression and the contents of H2S that were further increased by TFR. We conclude that, in MCA of CIR rats, TFR induces non-NO and non-PGI2-mediated effects of vasodilatation and hyperpolarization involving Kca and increases CSE mRNA expression level in endothelial cells and H2S content in the cerebrum. These findings suggest that the response induced by TFR is potentially related to endothelium-derived hyperpolarizing factor mediated by the endogenous H2S and promote the use of TFR in protection of brain from ischemia-reperfusion injury.
1. Introduction
Ischemic cerebrovascular disease is one of the common entities threatening human health with high morbidity, disability rate, and mortality. Hunting for more effective drugs against cerebral ischemia-reperfusion injury (CIR) is one of the particular concerns in the pharmaceutical research.
Vascular tone plays an important role in the occurrence, development, and outcome of ischemic cerebrovascular disease. Endothelium-derived hyperpolarizing factor (EDHF) is the third kind of relaxing factor and autacoids [1, 2], described as the nonnitric oxide (NO) and nonprostaglandin I2 (PGI2) factor [3–7]. It induces the hyperpolarizing response and consecutive dilatation of the vascular smooth muscle cells by activating vascular calcium-activated potassium channels (Kca) [8]. Studies have indicated that EDHF is a modulator in regulating blood flow and vascular resistance during normal physiological states [9–12] and plays an even greater role following pathological conditions like organ ischemia, acidosis, and hypoxia [13, 14]. The role of EDHF has been demonstrated in several blood vessels, including mesenteric arteries [15, 16], coronary arteries [17, 18], carotid artery [19], femoral artery [20], and human arteries [12, 21]. However, the chemical nature of EDHF in cerebral arteries is still unclear. Others [22] and we [23] have found that the endogenous gas hydrogen sulphide (H2S) is related to the action of EDHF in cerebral arteries.
H2S is a gas signaling molecule and released from vascular endothelium. It has been shown that H2S could protect the heart from CIR damage in rats, leading to vasodilatation in rat aorta and mesenteric arteries [24, 25]. Furthermore, the effect of H2S-induced endothelium-derived dilatations was partly inhibited by antagonist of potassium ion channels [26, 27]. This indicates that H2S is potentially an EDHF candidate. Recent studies in our laboratory have found that H2S is possibly the EDHF in middle cerebral arteries (MCAs) from healthy and CIR rats [23, 28, 29].
Medicinal plants in nature may represent a desirable source to develop valid and safe drugs for treatment of diseases including ischemic brain injury. Flavonoids are effective ingredients in many Chinese herbs and exist extensively in natural plants. Flavonoids have various biological activities and pharmacological functions including vasorelaxing and anti-inflammatory effect as well as protective effects against CIR and free radical.
Rhododendron simsii Planch is a traditional Chinese medicine that has been used for a thousand years in China. The leaves of Rhododendron simsii Planch are used for the antitussive and expectorant effects as well as for the antiallergic and anti-inflammatory effects used as topical medicine. The root of Rhododendron simsii Planch, however, has been used for treatment of abnormal menstruation, diarrhea, cough, and exec [30]. These effects often lead the clinical application in treatment of chronic bronchitis, asthma, and rheumatic arthritis [30–32].
Total flavones of Rhododendron simsii Planch (TFR) are the active extract from the flowers of Rhododendron simsii Planch consisting of essential components of hyperion, quercetin, matteucinol, and rutin. The pharmacological effect of these components has been reported before. It has been shown that hyperin generates protective effects to attenuate the effect of free radicals, to reduce cerebral edema as well as cerebral vascular resistance, to increase cerebral blood flow, and to relax cerebral basilar artery in rats against cerebral ischemic injury [28, 33, 34]. In contrast, quercetin is reported to be able to inhibit pig platelet aggregation induced by thrombin through restraining Ca2+ influx in platelets [21] and protect from neuronal damage after transient global cerebral ischemia [35]. In addition, Qing et al. demonstrated that matteucinol could produce concentration-dependent vasodilatation in the isolated rat aorta, which was dependent on endothelium [36]. As to the role of rutin, it was reported to exert endothelium-dependent vasodilation by nitric oxide/guanylate cyclase (NO/GC) pathway and refrains platelet activating factor [37, 38]. Finally, as total flavones of Rhododendron simsii Planch, TFR has been found to have antispasmodic, analgesic, and anti-inflammatory roles [39].
Previous studies by others and us have shown that TFR has protective effects against cerebral ischemia-reperfusion injury [40] and that TFR may induce vascular dilatation in CBA and MCA from healthy rats [28, 29]. The present study was undertaken, for the first time, to investigate the underlying mechanisms of TFR against CIR in rats subject to global cerebral ischemia-reperfusion injury.
2. Methods
2.1. Drugs and Reagents
Total flavones of Rhododendron simsii Planch (TFR), content of flavones greater than 85%, were furnished by Hefei Heyuan Medicine Technology Limited Company (Hefei, China). N-Nitro-L-arginine-methyl-ester (L-NAME), indomethacin (Indo), DL-propargylglycine (PPG), tetraethylammonium (TEA), and collagenase were obtained from Sigma (St. Louis, MO, USA). Evans blue (EB) were purchased from Tiangen Biotechnology (Beijing, China). Mouse anti-rat CD31 FITC, anti-FITC microBeads, and Mini/Micli MACS Starting kit were purchased from Miltenyi Biotechnology (Auburn, CA, USA). PCR-Marker, RT-PCR test kit, RNAiso Plus, CSE, and β-actin primer were purchased from Baoshengwu Biological Company (Dalian, China). Diethyl pyrocarbonate (DEPC) was purchased from Solarbio. Fetal bovine serum was purchased from Hyclone (Logan, UT, USA). Phosphate saline solution (PSS) [41] including (in mM) 118 NaCl, 3.4 KCl, 2.5 CaCl2, 1.2 KH2PO4, 1.2 MgSO4, 25 NaHCO3, and 11.1 glucose was effervesced with 95% O2 and 5% CO2, pH adjusted to 7.4 with NaOH. The PSS solution was always oxygenated during the latency period.
2.2. Animals and Experimental Protocol
Male Sprague-Dawley rats weighing 250–350 g were provided by the Experimental Animal Center of Anhui Medical University (Hefei, China) (Certificate number SCXK 2005-001). All animal studies and surgical procedures were conformed to the regulations defined by the Animal Care Committee of Anhui Medical University, which is in line with the Guide for the Care and Use of laboratory Animals published by the US National Institute of Health (NIH publication number 85-23, revised 1996).
All animals were fed with free access to food and tap water in clean plexiglass cages with stainless steel wire lids and filter tops at a controlled temperature 22 ± 3°C in room and kept with a 12 hours light/dark cycles. Rats were used in the experiment during the photostage of the light/dark cycles.
For RT-PCR test, rats were divided into the 6 groups (n = 8 for each), that is, sham, NS (control with treatment of normal saline), TFR 25 mg/kg, TFR 50 mg/kg, TFR 100 mg/kg, TFR 100 mg/kg, and PPG 37.5 mg/kg, and the animals were administered via tail vein injection at 20 min before ischemia.
In ELIASA experiment, rats were allocated to 5 groups (n = 8 for each), that is, sham, NS, TFR 25 mg/kg, TFR 50 mg/kg, and TFR 100 mg/kg and were treated via tail vein injection at 20 min before ischemia.
2.3. Extraction of Total Flavones from Rhododendron simsii Planch
A large number of components may be identified from Rhododendron simsii Planch [42]. In brief, the dried flowers of Rhododendron simsii Planch (500 g × 3) were dunked into 75% alcohol for 8 hours and then boiled under reflux for 2 hours. After being filtered, the boiled liquid was concentrated. The concentration was chromatographed on polyamide thread columns (25 × 18 cm; Wako Pure Chemical Industry Co., Osaka, Japan) to procure crude total flavones, which were further depurated by ethyl acetate and ethanol (V : V 76 : 38). The final freeze-dried yellow powder of total flavones was acquired and utilized for this study. UV-spectrophotometry was used to determine the content of TFR.
2.4. Global Cerebral Ischemia-Reperfusion Rat Models
Rat models of CIR were established by the modified method of Pulsinelli-Brierley (four-vessel occlusion, 4-VO) [43, 44]. In brief, rats were anesthetized with 10% chloral hydrate (0.3–0.35 g/kg body wt, ip) (Shanghai, China). The bilateral vertebral arteries were electrocoagulated and the incision was managed with suturing, followed by exposing and isolating the bilateral common carotids. All animals were fed in rearing cages for 24 h with adlibitum feeding. In the experiment, bilateral carotid arteries were simultaneously clamped by using two nontraumatic arteriole clips with keeping the core body temperature at 37°C. After 0.5 h of ischemia, the microvascular clamps were gently removed to recover cerebral blood flow through bilateral carotid arteries. The eligible experimental animals presented stiffness of the forepaws and loss of righting reflex during ischemia and survived after 0.5 h of ischemia and 2 h of reperfusion. In the sham-operation group, vertebral arteries were not electrocoagulated and carotid arties were not occluded.
Electroencephalogram (EEG; EB Neuro Corp., S.p.A, Firenze, Italy) was recorded at 5 min before and 0.5 h after ischemia and reperfusion by 5 min, 15 min, 30 min, 45 min, 60 min, and 120 min, respectively.
2.5. Vascular Experiments
Rats were decapitated after anesthetizing with 10% chloral hydrate via peritoneal injection. The brain was fleetly removed from the pericranium and immersed in ice-cold physiological salt solution (PSS). Then the MCA was carefully dissected, made into segments of 0.6–0.8 cm in length, and placed in a vessel container. The end of each MCA segment was inserted into two glass micropipettes and kept within a vessel chamber pressurized to a mean of 85 mmHg to establish a flow discharge of 150 μL/min through the lumen. The vessel was bathed with PSS aerated with 95% O2-5% CO2 (PH 7.3–7.5) and kept at 37°C using a fixed heat-exchanger device. After MCA segment was mounted, the container was placed on the stage of a stereoscopic microscope (Bengbu, China) equipped with a digital camera (Nikon, Tokyo) and a computer screen. The reproducible constriction was acquired by adding 30 mM KCl to the superfusate. L-NAME (30 μM) and Indo (10 μM) were utilized to inhibit the products of NO synthase and cyclooxygenase in experiments, respectively. Outer diameter of each vessel was measured directly from the video screen (magnification of ×100). The average values of maximum and minimum diameters were recorded on MCA during vasomotion. MCA dilatations were represented as the percentage of the maximum diameter (% D max) using the following equation:
| (1) |
where D represents the vessel diameter after luminal administration of the reagents, either TFR or L-NAME, and so forth, D min expresses the diameter after the addition of 30 mM KCl, and D max stands for the maximum diameter obtained at 85 mmHg luminal pressure after 1 h equilibrium.
Mechanical method is used to remove vascular endothelial cell of MCA [45]. Briefly, a slimsy hairline of head is utilized to rub back and forth into lumens of MCA for removal vascular endothelial cell. Ach (10−5 mol/L) was used to validate the integrity of endothelium and usually it was less than 10% [46]. As suggested, it is considered that endothelium is completely removed when ACh-induced relaxation is less than 30% [47].
2.6. Microelectrode Experiments
Isolated MCA segments for sham operation and CIR vessels were longitudinally incised, fixed within 10 mL silica gel slot (a mean pressure of 85 mmHg), superfused with PSS, maintained at 37°C, and oxygenated with 95% O2-5% CO2. In some experiments, superfusate was filled with 30 μM L-NAME plus 10 μM Indo. Vascular smooth muscle cells (VSMCs) of the MCA were impaled by microelectrodes surveyed intracellular membrane potential (E m) as previously reported. In this experiment, KCl was deleted from those special protocols as it could lead to vasoconstriction with the consequent tissue motion being disadvantageous to keeping intracellular membrane potential recordings. Each VSMC was pierced by using glass microelectrodes perfused with 3 M KCl (electrode resistances ranged from 40 to 80 MΩ). The typical pierce was successfully emerged a sudden drop in E m kept stable for at least 2 min before initiating the experiment. A single E m value for each condition in a specified MCA was acquired by averaging four to six different VSMC impalements. The conventional high impedance amplifier (Intra 767; World Precision Instruments, Sarasota, FL, USA) was utilized to record the potential difference and interference (50 Hz) at the amplifier output that was selectively moved aside. The MacLab system connected with Chart 5 software (AD Instruments, Castle Hill, NSW, Australia) was used to monitor and analyze E m. The n value of Figure 1 refers to the number of experimental rats, which is not the number of impalements.
Figure 1.
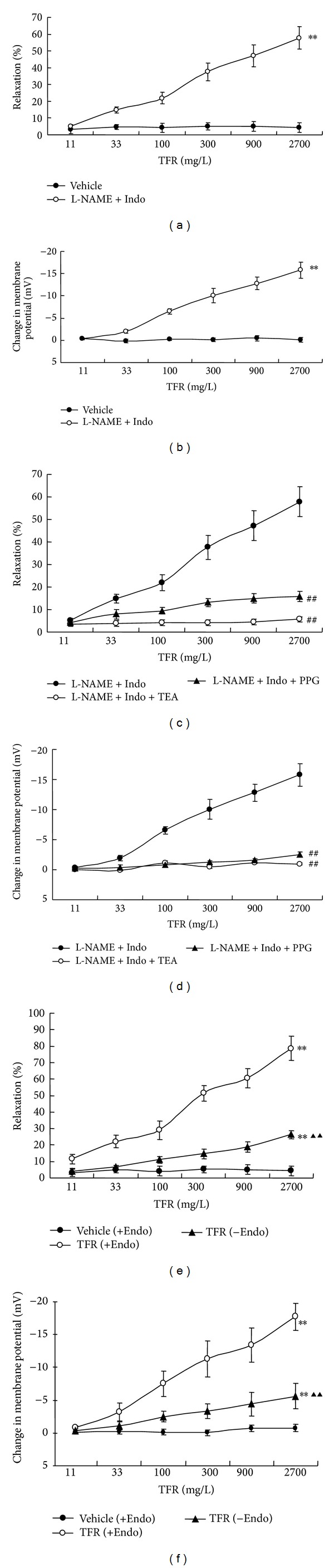
The dilatation (a) and hyperpolarization (b) of middle cerebral arteries of rats subjected to cerebral ischemia-reperfusion to TFR in the presence of 30 μM L-NAME (an inhibitor of nitric oxide synthase) and 10 μM Indo (an inhibitor of cyclooxygenase); effect of TEA (1 mM, an inhibitor of Ca2+-activated potassium channel) or PPG (100 μM, an inhibitor of cystathionine-γ-lyase) on non-NO and non-PGI2-mediated vasodilation (c) and hyperpolarization (d) elicited by TFR in VSMC from middle cerebral artery of rats subjected to cerebral ischemia-reperfusion; effect of endothelial denudation on the dilatation (e) and hyperpolarization (f) to TFR in middle cerebral arteries of rats subjected to cerebral ischemia reperfusion. Values are presented as means ± SE. +Endo: endothelium-intact and −Endo: endothelium-denudated. **P < 0.01 compared with vehicle group; ## P < 0.01 compared with L-NAME+Indo group; ▲▲ P < 0.01 compared with TFR (+Endo) group. Comparisons were performed by unpaired Student's t-test.
2.7. Whole Cell Patch Clamp Recording Experiments
The acute separation of MCA smooth muscle cells in rats has been described elsewhere. Briefly, Rats were killed by detruncation and MCA was cleanly isolated and placed in PSS containing 1.0 mg/mL Type II collagenase and 0.5 mg/mL papain to incubate at 37°C for 35 min. Mechanical separation of the digested tissue into a single vascular smooth muscle cell and the formation of cell suspension were performed by using polished Pasteur pipettes. The cell was plated onto glass coverslips and incubated for 45 min prior to electrophysiological study and left to stick to the glass coverslip before an experiment was started.
Electrophysiological measurements from MCA smooth muscle cells were made using the whole cell patch clamp recording technique. Whole cell currents were made using an EPC-10 amplifier and pulse software. The membrane currents were filtered at 1 kHz and stored. When starting each experiment, the junction potential between the bath solution and pipette solution was correctly adjusted to zero. Test pulses were performed with a 10 mV increase from −60 to 100 mV with a holding potential of −60 mV for 500 ms. The patch pipettes were drawn out of borosilicate glass (resistance 3 to 5 MΩ, P-97-type microelectrode puller instrument from Sutter, USA). The bath solution for recording Kca current was composed of (in mM) 140 NaCl, 1 MgCl2, 5 H-HEPES, 1 CaCl2, 5 KCl, and 10 glucose (pH adjusted to 7.4 with NaOH). The recording pipette solution contained (in mM) 105 K-gluconate, 1 MgCl2, l30 KCl, 10 H-HEPES, 2.1 CaCl2, and 5 Na2ATP; pH was set to 7.2 with NaOH. Cells were continuously superfused with the bath solution containing testing chemicals. The recording values of the current were shown by the current density (pA/pF) and the experimental results analyzed and mapped using Igor 5 software.
2.8. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
The endothelial cells from MCA of rats were isolated and purified using magnetic activated cell sorting (MACS) by the method described by DeLeve et al. and Kader et al. All reagents for MACS separation were purchased from Miltenyi Biotec (Bergisch Gladbach, Germany). In brief, 10 μL FITC was incubated in 100 μL buffer (35~40 min at 4°C) and added in 2 mL phosphate saline solution (PSS) and centrifugated for 10 min (1000 rpm). The endothelial cell of middle cerebral artery cells in rats was marked by using mouse anti-rat CD31 FITC. Anti-FITC microBeads (10 μL) were incubated with 107 cells per mL for 30 min (4~8°C). Endotheliocytes of MCA (positive cells) in rats were separated and purified by utilizing a magnetic separation column, which was washed 3× with PSS. A total of 105 cells/mL were placed in cryogenic vial and maintained in nitrogen canister for reserve. The level of endotheliocyte CSE mRNA was determined by semiquantitative reverse transcription (RT)-PCR [48]. Total RNA was isolated using RNA isolation kit (Baoshengwu, Dalian, China). Extraction of RNA was successful, and the RNA integrity and quantity were estimated ordinarily by absorbability (A260/A280: 1.8~2.0). Ethidium bromide fluorescence of RNA was separated by electrophoretic technique on 1% tetrabromoethane (TBE) containing agarose gels. A standardized semiquantitative RT-PCR method was utilized on amplification of the target genes and β-actin, a constitutively expressed gene, as reference gene. The sequence specific primers for the RT-PCR were used as follows (Baoshengwu, Dalian, China): (forward) 5′-CCACCACAACGATTACCCA-3′, (reverse) 5′-TCAGCACCCAGAGCCAAAG-3′, and 334 bp for MCA CSE and (forward) 5′-CTGTCCCTGTATGCCTCTG-3′, (reverse) 5′-ATGTCACGCACGATTTCC-3′, and 236 bp for β-actin. Sample RNA 1 μL was reversely transcribed in 10 μL of a solution including 1 μL of dNTP mixture (10 mM each), 1 μL of oligo dT primer (2.5 μM), and RNase free dH2O. The mixture was heated to 65°C for 5 min for RNA denaturation and cooled quickly to 4°C. Next, 4 μL of 5× primescript buffer, 0.5 μL of RNase inhibitor (40 U/μL), 0.5 μL of primescript RTase (for 2 step), and 5 μL of RNase free dH2O mix were added, and the reaction was performed in following conditions: 30°C for 10 min, 42°C for 30 min, 95°C for 5 min, and 4°C for 5 min. Then the RNA obtained was amplified by PCR in total of 35 cycles (2 min at 94°C, 30 s at 50°C/53°C, and 45 s at 72°C). β-Actin was utilized as an internal control. After amplification, each PCR product (5 μL) was analyzed by electrophoresis on a 1% agarose gel in 1×TBE buffer, stained with ethidium bromide, and photographed under ultraviolet light with a multi-image light cabinet.
2.9. Measurement of Endogenous H2S Production
Tissue H2S production rate was determined as described previously [49] with modifications. In brief, the right cerebral cortex of rats killed by detruncation was stored in liquid nitrogen and homogenized 12% (w/v) tissue homogenate in 50 mM ice-cold potassium phosphate buffer solution (pH 8.0), and then the homogenate was centrifugated for 10 min (47000 ×g, 4°C). The pipette was used to move homogenate (75 μL) into another a centrifugal tube in which 1% zinc acetate (0.25 mL) and distilled water (0.45 mL) were added, respectively. After incubating for 10 min, 0.25 mL of 10% three chloroacetic acid was added into the reaction mixture to stop the reaction, followed by second 10-min centrifugation (14000 ×g, 4°C). The supernate in the centrifugal tub was then transferred to test tubes. Subsequently, 133 μL of 20 mM N,N-dimethyl-p-phenylenediamine sulphate in 7.2 M HCl was added immediately, followed by adding 133 μL of 30 mM FeCl3 in 1.2 M HCl. The absorbance of the resulting solution at 670 nm was measured 20 min later with an automatic ELISA (BioTEK, USA). The content of H2S was calculated by the calibration curve of the standard H2S solutions.
2.10. Statistical Analysis
All data are expressed as the mean ± standard deviation of the mean. Quantitative analysis was estimated by using one-way analysis of variance (ANOVA) with Bonferroni's posttest. The differences between sham and CIR vessels were tested using the unpaired Student's t-test. A probability (P) value <0.05 was considered as statistically significant. All analyses were performed using the Statistical Package for Social Sciences (SPSS) 11.5 software.
3. Results
3.1. Effect of L-NAME+Indo on TFR-Induced Vasodilatation in CIR Vessels
Figure 1 shows that in vessels precontracted by 30 mM KCl, after treatment with 30 μM L-NAME and 10 μM Indo, TFR (11~2700 mg/L) evoked dose-dependent vasodilation (the percentage of maximal dilatation, E max: 57.8 ± 6.6%, Figure 1(a)) and hyperpolarization (the maximal change in E m: −15.8 ±1.9 mV, Figure 1(b)) in MCA of CIR rats (P < 0.01, versus Vehicle).
3.2. Combined Effect of L-NAME+Indo+TEA on TFR-Induced Dilation in CIR Vessels
TEA (1 mM), an inhibitor of Ca2+-activated potassium channel, markedly attenuated TFR-elicited non-NO and non-PGI2 dilatation (57.8 ± 6.6% versus 5.7 ± 1.2%, P < 0.01, Figure 1(c)) and hyperpolarization (−15.8 ± 1.9 mV versus −0.9 ± 0.2 mV, P < 0.01, Figure 1(d)) in the presence of L-NAME (30 μM) plus Indo (10 μM). These data revealed that the TFR-mediated non-NO, non-PGI2 effects were potentially associated with calcium-activated potassium channels.
3.3. Effect of L-NAME+Indo+PPG on TFR-Induced Dilatation in CIR Vessels
After inhibition of NO and PGI2 synthase with 30 μM L-NAME and 10 μM Indo, TFR-induced vasodilatation and hyperpolarization were notably inhibited by 100 μM PPG, a blocker of endogenous H2S synthese-cystathionine-γ-lyase (CSE), resulting in reduced dilatation (57.8 ± 6.6% versus 15.8 ± 2.4%, Figure 1(c)) and hyperpolarization (−15.8 ± 1.9 mV versus −2.5 ± 0.4 mV, P < 0.01, Figure 1(d)). These findings indicated that TFR-induced non-NO and non-PGI2 mediated effects were potentially related to the endogenic release of H2S.
3.4. Effect of Endothelial Denudation on TFR-Induced Vasodilatation in CIR Vessels
The TFR (11~2700 mg/L) induced vasodilatation and hyperpolarization were significantly attenuated by removal of endothelium in MCA of CIR rat. In fact, in vessels precontracted by 30 mM KCl, the maximal dilatation reduced from 78.6 ± 7.5% to 26.4 ± 2.5% (Figure 1(e)). Similarly, the maximal hyperpolarization reduced from −17.7 ± 2.1 mV to −5.6 ± 1.9 mV (Figure 1(f)).
3.5. Effect of TFR on Calcium-Dependent Potassium Current in VSMC from MCA of CIR Rat
The outward current shown in Figure 2 was induced in VSMC from MCA of rats subject to CIR. At 300–2700 mg/L, TFR remarkably enhanced the outward current and the enhancement was inhibited by 1 mM TEA, an inhibitor of Kca channel. The results suggested that the enhanced effect of TFR on calcium-dependent potassium current in VSMC from MCA of CIR rats involves the opening of Kca channels.
Figure 2.
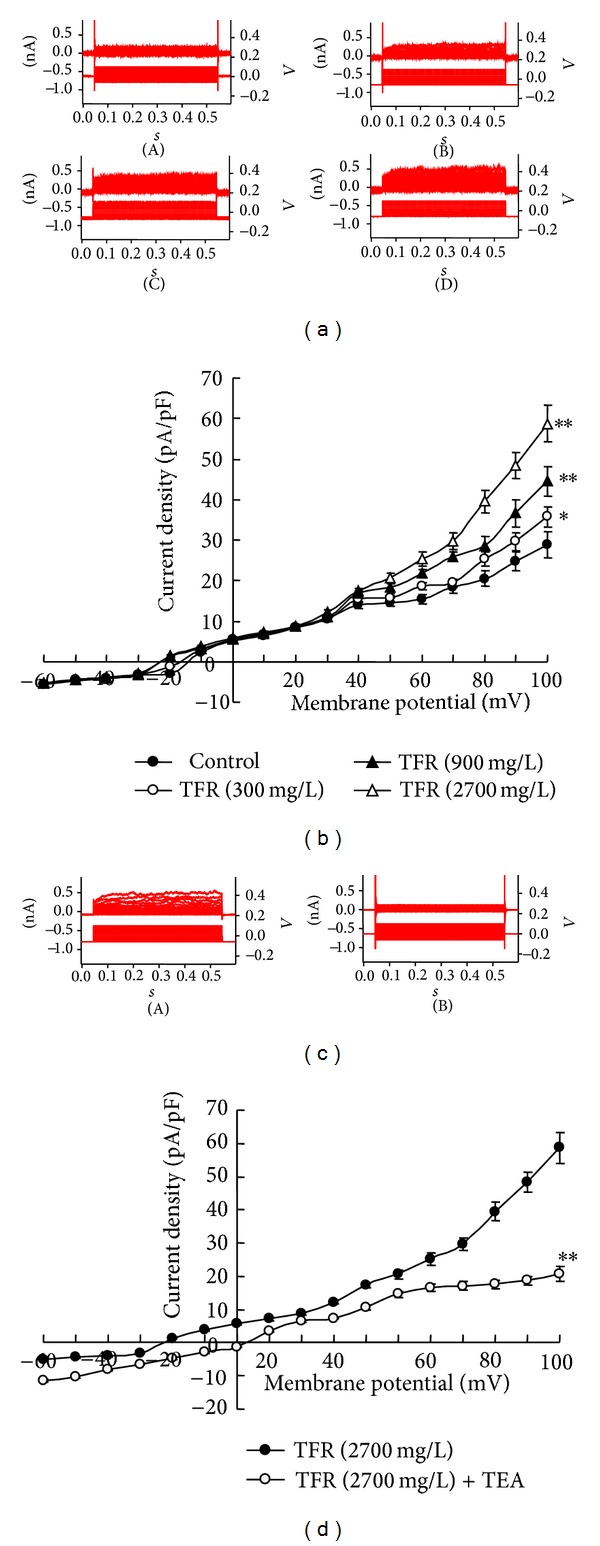
The enhancement of TFR on the outward current in VSMC from middle cerebral arteries of rats subjected to global cerebral ischemia-reperfusion. (a) The trace of outward current in one VSMC, A: control, B ~ D: 300,900 and 2700 mg/L TFR and (b) the curve of current-voltage relationship. (n = 6); effect of TEA on the TFR-induced enhancement of outward current in VSMC from middle cerebral artery of rats subjected to global cerebral ischemia-reperfusion. (c) The trace of outward potassium current in one VSMC. A: TFR 2700 mg/L, B: TEA 1 mM + TFR 2700 mg/L and (d) the curve of current-voltage relationship. n = 6. Values are presented as means ± SE. *P < 0.05, **P < 0.01, compared with control group. **P < 0.01 compared with TFR (2700 mg/L) group. Comparisons were performed by unpaired Student's t-test.
3.6. Effect of TFR on the Level of CSE mRNA Expression in MCA Endothelial Cells of CIR Rats
The level of CSE mRNA expression in the purified and isolated endothelial cells from MCA of CIR rats was increased (P < 0.01, versus control, Figure 3). TFR (50, 100 mg/kg) markedly potentiated the CSE mRNA expression, which was upregulated in the endothelial cells from MCA of CIR rats (P < 0.01, versus NS, Figure 3).
Figure 3.
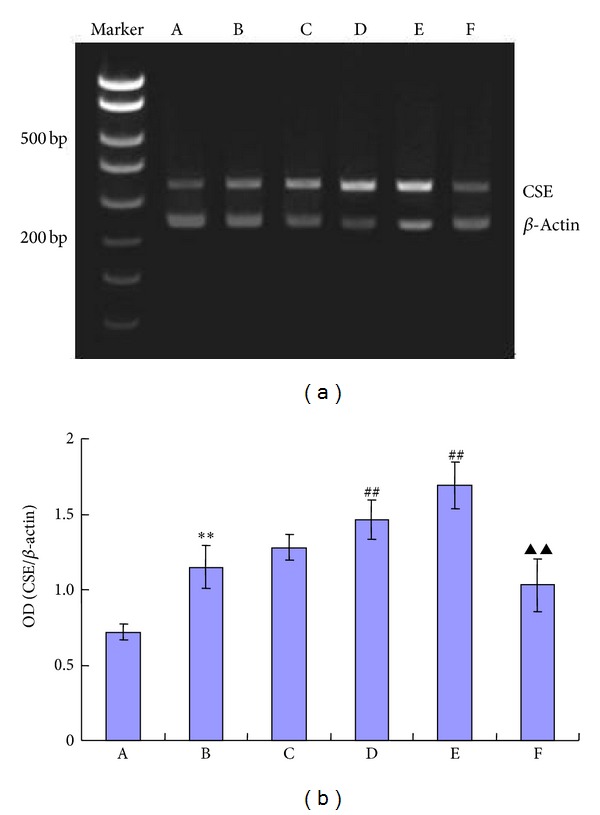
Effects of TFR on the levels of endothelium cells of middle cerebral artery CSE mRNA expression in rats subjected to cerebral ischemia-reperfusion. A: sham, B: NS, C~E: 25, 50, and 100 mg/kg TFR, and F: 37.5 mg/kg PPG + 100 mg/kg TFR. All values are presented as means ± SE. All comparisons were assessed by unpaired Student's t-test. **P < 0.01 compared with sham group, ## P < 0.01 compared with NS group, and ▲▲ P < 0.01 compared with TFR (100 mg/kg) group.
3.7. Effect of TFR on H2S Content in the Cerebrum of CIR Rats
The contents of H2S were significantly increased in the cerebrum of rats subject to CIR and pretreatment with TFR in the range of 25 to 100 mg/kg further increased the H2S contents (P < 0.05).
4. Discussion
In this study, we have found that (1) TFR induces endothelium-dependent vasodilatation and hyperpolarization in MCA of CIR rat; (2) the non-NO and non-PGI2-mediated effects of vasodilatation and hyperpolarization of VSMCs induced by TFR involves Kca channels and therefore the response is related to EDHF; and (3) TFR increases the level of CSE mRNA expression in MCA endothelial cells and H2S content in the cerebrum of CIR rats.
4.1. TFR Induced Endothelium-Dependent and Endothelium-Independent Responses
The present study showed that 11~2700 mg/L TFR-induced vasodilatation and hyperpolarization have endothelium-dependent and endothelium-independent components in MCA of CIR rats. The endothelium-dependent component is reflected by the significant attenuation of the response by removal of the endothelium (Figures 1(e) and 1(f)). The endothelium-independent component is reflected by the remaining response after removal of the endothelium (Figures 1(e) and 1(f)).
4.2. TFR Induced EDHF-Mediated Dilatation and Hyperpolarization of the VSMCs
In the present study, we found that in MCA of CIR rats with blockage of NO and PGI2 formation with combination of L-NAME and Indo, TFR (11~2700 mg/L) induced concentration-dependent vasodilatation and hyperpolarization of smooth muscle cells, indicating that TFR may induce non-NO, non-PGI2-mediated vasodilatation and hyperpolarization. It is likely that TFR-mediated cerebrovascular dilatation may be related to the endothelium-dependent vasodilatation of hyperin, matteucinol, and rutin in TFR that were demonstrated previously [28, 36, 37]. The present study further demonstrated that these responses involve the activation of Kca channels. As is well known now, EDHF opens Kca channels on the smooth muscle cell and induces hyperpolarization of smooth muscle cell membrane potential, resulting in reduced Ca2+ influx and vascular dilatation [9, 50–52]. Therefore, an important characteristic of EDHF response is that it may be inhibited by Kca channel blockers.
In acutely isolated smooth muscle cell of MCA in rats, we found that the calcium-dependent potassium current of the whole cell in both control and TFR groups exhibited a voltage-dependent manner, significantly and outwardly rectifying characteristics in high voltage and that the outward current density induced by TFR (300~2700 mg/L) was significantly enhanced. This was abolished by 1 mM TEA, indicating that the outward current was calcium-activated potassium current and TFR directly enhanced this current in smooth muscle cells from MCA in CIR rats.
Previous studies have shown that 1 mM TEA, nonselective potassium channel blocker, specifically blocks the opening of Kca channels [9, 27, 53]. Indeed, we found that 1 mM TEA almost abolished the non-NO non-PGI2-mediated vasodilation and hyperpolarization induced by TFR in the presence of L-NAME and Indo in MCA of CIR rats. These results indicate that the effects of TFR involve the opening of Kca channels and this finding clearly shows that TFR-induced non-NO non-PGI2 effects are related to EDHF.
4.3. EDHF-Mediated Responses in Rat Cerebral Vessels Is Related to H2S
Studies from others and ourselves have suggested that H2S induces hyperpolarization of smooth muscle cells and vasodilatation [23, 24, 53] and H2S modulates the functions of the central nervous system by regulating vascular smooth muscle tone and cerebral blood supply [25]. In the cardiovascular system, CSE is considered to be the only enzyme to produce H2S and is expressed in vascular smooth muscle cells and endothelial cells, with endothelial cells being more prominent [54]. We have proposed that H2S produced in vascular endothelium of CBA of rats may be EDHF [23].
In the present study, 100 μM PPG, an inhibitor of endogenous H2S synthese-cystathionine-γ-lyase (CSE), combined with L-NAME and Indo, eliminated EDHF-mediated vasodilatation and hyperpolarization of smooth muscle cell induced by TFR in MCA of CIR rats, suggesting that TFR- induced EDHF effects are associated with the production of endogenous H2S. Further, our RT-PCR experiments by using endothelial cells from MCA in CIR rats separated and purified by the magnetic activated cell sorting technique (MACS), currently the most advanced technology on cell separation [55], demonstrated the expression of CSE mRNA, indicating that the synthesis of endogenous H2S enzymes is expressed in endothelial cells from MCA. In addition, the endothelial cells CSE mRNA expression was significantly higher than that of sham group, indicating that CIR promotes endothelial cells of MCA to synthesize CSE and increases the generation of H2S. This is in accordance with our previous experimental results [23]. We speculate that the increase of CSE mRNA expression and H2S production of the endothelial cells from MCA might serve as a compensative protection to reserve endothelium-derived vasodilatation in CIR rats. Such protective and vasodilator effect is beneficial to the vessel subject to ischemia-reperfusion injury.
The present study also demonstrated that the content of H2S in the brain tissue was significantly higher after ischemia-reperfusion (compare NS and sham, P < 0.01) and this increase was further spurred by pretreatment of TFR (25–100 mg/kg) (Table 1).
Table 1.
Effect of TFR on cerebral H2S level in rats subjected to cerebral ischemia-reperfusion.
| Group | Dose (mg/kg) | n | H2S (nmol/g) |
|---|---|---|---|
| Sham | — | 8 | 11.5 ± 1.1 |
| NS | — | 8 | 17.6 ± 1.6∗∗ |
| TFR | 25 | 8 | 19.8 ± 1.7∗∗# |
| 50 | 8 | 22.0 ± 2.3∗∗## | |
| 100 | 8 | 25.6 ± 2.5∗∗## |
**P < 0.01 compared to the sham control, # P < 0.05, ## P < 0.01 compared to the NS control. Each group consisted of 8 rats. TFR: total flavones of Rhododendron simsii Planch, Sham: sham operation control group, and NS: normal saline control group.
Interestingly, the present study demonstrated for the first time that TFR may increase the CSE expression levels of endothelial cells of MCA in CIR rats and therefore may increase the synthesis of endogenous H2S that results in vasodilatation effect in cerebrovascular system against brain ischemia-reperfusion injury. This result is in accordance with previous reports that showed the protective effects against cerebral ischemia by hyperin and quercetin, which are primarily active constituents in TFR [28, 35]. The findings from our study, together with previous reports, may promote the development and utilization of TFR as the new strategy in management of ischemia-reperfusion injury.
In summary, we for the first time found that in MCA of CIR rats, TFR induces the non-NO and non-PGI2-mediated effects of vasodilatation and hyperpolarization of VSMCs, involving Kca channels, and increases the level of CSE mRNA expression in MCA endothelial cells and H2S content in the cerebrum of CIR rat. This finding suggests that the non-NO and non-PGI2 response induced by TFR is potentially related to EDHF, mediated by the endogenous H2S. Therefore, our study promotes the use of Chinese medicine in protection of brain and may form a new strategy in prevention and treatment of ischemia-reperfusion injury by using TFR.
Acknowledgments
The authors appreciated the contributions from the other members of their laboratory: YF Fan, B Song, and YF Chen. This study was supported by the National Natural Science Foundation of China under Grant nos. 81173596 and 81170148, the Doctor Subject Fund of Education of Ministry of China under Grant no. 20070366005, the Natural Science Foundation of the Department of Education of Anhui Province under Grant no. kj2011B200, and the Natural Science Foundation of Wannan Medical College under Grant no. WK2012Z03.
Abbreviations
- TFR:
Total flavones of Rhododendron simsii Planch
- CIR:
Cerebral ischemia-reperfusion
- EDHF:
Endothelium-derived hyperpolarizing factor
- H2S:
Hydrogen sulfide
- Kca:
Calcium-activated potassium channel
- CSE:
Cystathionine-γ-lyase.
Conflict of Interests
This study was supported by an unrestricted grant from Recordati SpA, which only supported the project financially and was not involved in the design, production, or handling of the intellectual content of the paper. Authors declare no competing financial interests.
References
- 1.Jin X, Satoh-Otonashi Y, Zamami Y, et al. New molecular mechanisms for cardiovascular disease: contribution of endothelium-derived hyperpolarizing factor in the regulation of vasoconstriction in peripheral resistance arteries. Journal of Pharmacological Sciences. 2011;116(4):332–336. doi: 10.1254/jphs.10r30fm. [DOI] [PubMed] [Google Scholar]
- 2.del Valle-Rodríguez A, López-Barneo J, Ureña J. Ca2+ channel-sarcoplasmic reticulum coupling: a mechanism of arterial myocyte contraction without Ca2+ influx. EMBO Journal. 2003;22(17):4337–4345. doi: 10.1093/emboj/cdg432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bolton TB, Gordienko DV, Pucovský V, Parsons S, Povstyan O. Calcium release events in excitation-contraction coupling in smooth muscle. Novartis Foundation Symposium. 2002;246:154–173. doi: 10.1002/0470853050.ch12. [DOI] [PubMed] [Google Scholar]
- 4.Fleming I. Cytochrome P450 epoxygenases as EDHF synthase(s) Pharmacological Research. 2004;49(6):525–533. doi: 10.1016/j.phrs.2003.11.016. [DOI] [PubMed] [Google Scholar]
- 5.Chataigneau T, Félétou M, Duhault J, Vanhoutte PM. Epoxyeicosatrienoic acids, potassium channel blockers and endothelium-dependent hyperpolarization in the guinea-pig carotid artery. British Journal of Pharmacology. 1998;123(3):574–580. doi: 10.1038/sj.bjp.0701629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fleming I. Vascular cytochrome p450 enzymes: physiology and pathophysiology. Trends in Cardiovascular Medicine. 2008;18(1):20–25. doi: 10.1016/j.tcm.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 7.Hercule HC, Schunck WH, Gross V, et al. Interaction between P450 eicosanoids and nitric oxide in the control of arterial tone in mice. Arteriosclerosis, Thrombosis, and Vascular Biology. 2009;29(1):54–60. doi: 10.1161/ATVBAHA.108.171298. [DOI] [PubMed] [Google Scholar]
- 8.Fissithaler B, Popp R, Kiss L, et al. Cytochrome P450 2C is an EDHF synthase in coronary arteries. Nature. 1999;401(6752):493–497. doi: 10.1038/46816. [DOI] [PubMed] [Google Scholar]
- 9.Ge ZD, Zhang XH, Fung PC, He GW. Endothelium-dependent hyperpolarization and relaxation resistance to N(G)-nitro-L-arginine and indomethacin in coronary circulation. Cardiovascular Research. 2000;46(3):547–556. doi: 10.1016/s0008-6363(00)00040-7. [DOI] [PubMed] [Google Scholar]
- 10.Yang Q, Yim AP, He GW. The significance of endothelium-derived hyperpolarizing factor in the human circulation. Current Vascular Pharmacology. 2007;5(1):85–92. doi: 10.2174/157016107779317206. [DOI] [PubMed] [Google Scholar]
- 11.Zhang RZ, Yang Q, Yim AP, Huang Y, He GW. Role of NO and EDHF-mediated endothelial function in the porcine pulmonary circulation: comparison between pulmonary artery and vein. Vascular Pharmacology. 2006;44(3):183–191. doi: 10.1016/j.vph.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 12.He GW, Liu ZG. Comparison of nitric oxide release and endothelium-derived hyperpolarizing factor-mediated hyperpolarization between human radial and internal mammary arteries. Circulation. 2001;104(1):I344–I349. doi: 10.1161/hc37t1.094930. [DOI] [PubMed] [Google Scholar]
- 13.Dong YY, Wu M, Yim AP, He GW. Hypoxia-reoxygenation, St. Thomas cardioplegic solution, and nicorandil on endothelium-derived hyperpolarizing factor in coronary microarteries. Annals of Thoracic Surgery. 2005;80(5):1803–1811. doi: 10.1016/j.athoracsur.2005.04.047. [DOI] [PubMed] [Google Scholar]
- 14.Powers RW, Gandley RE, Lykins DL, Roberts JM. Moderate hyperhomocysteinemia decreases endothelial-dependent vasorelaxation in pregnant but not nonpregnant mice. Hypertension. 2004;44(3):327–333. doi: 10.1161/01.HYP.0000137414.12119.f6. [DOI] [PubMed] [Google Scholar]
- 15.Brancaleone V, Roviezzo F, Vellecco V, de Gruttola L, Bucci M, Cirino G. Biosynthesis of H2S is impaired in non-obese diabetic (NOD) mice. British Journal of Pharmacology. 2008;155(5):673–680. doi: 10.1038/bjp.2008.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang R. Hydrogen sulfide: a new EDRF. Kidney International. 2009;76(7):700–704. doi: 10.1038/ki.2009.221. [DOI] [PubMed] [Google Scholar]
- 17.Ahluwalia A, Hobbs AJ. Endothelium-derived C-type natriuretic peptide: more than just a hyperpolarizing factor. Trends in Pharmacological Sciences. 2005;26(3):162–167. doi: 10.1016/j.tips.2005.01.005. [DOI] [PubMed] [Google Scholar]
- 18.Villar IC, Panayiotou CM, Sheraz A, et al. Definitive role for natriuretic peptide receptor-C in mediating the vasorelaxant activity of C-type natriuretic peptide and endothelium-derived hyperpolarising factor. Cardiovascular Research. 2007;74(3):515–525. doi: 10.1016/j.cardiores.2007.02.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chauhan SD, Nilsson H, Ahluwalia A, Hobbs AJ. Release of C-type natriuretic peptide accounts for the biological activity of endothelium-derived hyperpolarizing factor. Proceedings of the National Academy of Sciences of the United States of America. 2003;100(3):1426–1431. doi: 10.1073/pnas.0336365100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Leuranguer V, Vanhoutte PM, Verbeuren T, Félétou M. C-type natriuretic peptide and endothelium-dependent hyperpolarization in the guinea-pig carotid artery. British Journal of Pharmacology. 2008;153(1):57–65. doi: 10.1038/sj.bjp.0707476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu ZG, Ge ZD, He GW. Difference in endothelium-derived hyperpolarizing factor-mediated hyperpolarization and nitric oxide release between human internal mammary artery and saphenous vein. Circulation. 2000;102(19):III296–III301. doi: 10.1161/01.cir.102.suppl_3.iii-296. [DOI] [PubMed] [Google Scholar]
- 22.Wang Y, Zhao X, Jin H, et al. Role of hydrogen sulfide in the development of atherosclerotic lesions in apolipoprotein E knockout mice. Arteriosclerosis, Thrombosis, and Vascular Biology. 2009;29(2):173–179. doi: 10.1161/ATVBAHA.108.179333. [DOI] [PubMed] [Google Scholar]
- 23.Han J, Chen ZW, He GW. Acetylcholine- and sodium hydrosulfide-induced endothelium-dependent relaxation and hyperpolarization in cerebral vessels of global cerebral ischemia-reperfusion rat. Journal of Pharmacological Sciences. 2013;121(4):318–326. doi: 10.1254/jphs.12277fp. [DOI] [PubMed] [Google Scholar]
- 24.Zhao W, Wang R. H2S-induced vasorelaxation and underlying cellular and molecular mechanisms. American Journal of Physiology: Heart and Circulatory Physiology. 2002;283(2):H474–H480. doi: 10.1152/ajpheart.00013.2002. [DOI] [PubMed] [Google Scholar]
- 25.Qu K, Chen CP, Halliwell B, Moore PK, Wong PT. Hydrogen sulfide is a mediator of cerebral ischemic damage. Stroke. 2006;37(3):889–893. doi: 10.1161/01.STR.0000204184.34946.41. [DOI] [PubMed] [Google Scholar]
- 26.Yang G, Wu L, Jiang B, et al. H2S as a physiologic vasorelaxant: hypertension in mice with deletion of cystathionine γ-lyase. Science. 2008;322(5901):587–590. doi: 10.1126/science.1162667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhao W, Zhang J, Lu Y, Wang R. The vasorelaxant effect of H2S as a novel endogenous gaseous KATP channel opener. EMBO Journal. 2001;20(21):6008–6016. doi: 10.1093/emboj/20.21.6008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fan YF, Chen ZW, Guo Y, Wang QH, Song B. Cellular mechanisms underlying Hyperin-induced relaxation of rat basilar artery. Fitoterapia. 2011;82(4):626–631. doi: 10.1016/j.fitote.2011.01.023. [DOI] [PubMed] [Google Scholar]
- 29.Cai SN, Fan YF, Chen ZW. The hyperpolorization produced by H2S in VSMC from middle cerebral artery of rat. Chinese Journal of Clinical Pharmacology and Therapeutics. 2011;16:155–159. [Google Scholar]
- 30.Shao ML, Ma XW, Wu Z, Yu WM. Rhododendron simsii Planch. In: Shao ML, Ma XW, Wu Z, Yu WM, editors. Pharmacopoeia of the People’s Republic of China. Beijing, China: China Medical Science & Technology Publishing; 2010. p. p. 339. [Google Scholar]
- 31.Yuan LP, Chen ZW, Li F, Dong LY, Chen FH. Protective effect of total flavones of rhododendra on ischemic myocardial injury in rabbits. The American Journal of Chinese Medicine. 2006;34(3):483–492. doi: 10.1142/S0192415X06004016. [DOI] [PubMed] [Google Scholar]
- 32.Zhao X. The research on chemical composition and extraction method of rhododendron dauricum. Acta Universitatis Medicinalis Anhui. 2013;48:580–582. [Google Scholar]
- 33.Chen Z, Ma C, Zhao W. Protective effect of hyperin against cerebral ischemia-reperfusion injury. Acta Pharmaceutica Sinica. 1998;33(1):14–17. [PubMed] [Google Scholar]
- 34.Chen Z, Zhang J, Ma C. Protective effect of hyperin on cerebral infarction in rats. China Journal of Chinese Materia Medica. 1998;23(10):626–628. [PubMed] [Google Scholar]
- 35.Cho JY, Kim IS, Jang YH, Kim AR, Lee SR. Protective effect of quercetin, a natural flavonoid against neuronal damage after transient global cerebral ischemia. Neuroscience Letters. 2006;404(3):330–335. doi: 10.1016/j.neulet.2006.06.010. [DOI] [PubMed] [Google Scholar]
- 36.Qing XJ, Hou XM, Li QS. The study on dilatation and its mechanism of matteucinol in the isolated rat aorta. Chinese Journal of Integrative Medicine on Cardio-/Cerebrovascular Disease. 2012;10(4):457–458. [Google Scholar]
- 37.Zhou XM, Yao H, Xia ML, Cao CM, Jiang HD, Xia Q. Comparison of vasodilatation effect between quercetin and rutin in the isolated rat thoracic aorta. Journal of Zhejiang University: Medical Sciences. 2006;35(1):29–33. doi: 10.3785/j.issn.1008-9292.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 38.Chen WM, Jin M, Wu W, Li JR, Zang BX, Pu YZ. Experimental study on inhibitory effect of rutin against platelet activation induced by platelet activating factor in rabbits. Chinese Journal of Integrated Traditional and Western Medicine. 2002;22(4):283–285. [PubMed] [Google Scholar]
- 39.Dai SJ, Chen RY, Yu DQ. Studies on the flavonoid compounds of Rhododendron anthopogonoides. Zhongguo Zhong Yao Za Zhi. 2004;29(1):44–47. [PubMed] [Google Scholar]
- 40.Guo Y, Chen ZW. Protective effects of total flavones of rhododendra on cerebral ischemia reperfusion injury. The American Journal of Chinese Medicine. 2008;36(2):343–354. doi: 10.1142/S0192415X08005813. [DOI] [PubMed] [Google Scholar]
- 41.Bryan RM, Jr., Eichler MY, Swafford MW, Johnson TD, Suresh MS, Childres WF. Stimulation of alpha 2 adrenoceptors dilates the rat middle cerebral artery. Anesthesiology. 1996;85(1):82–90. doi: 10.1097/00000542-199607000-00012. [DOI] [PubMed] [Google Scholar]
- 42.Zhao CX, Liang YZ, Li XN, Fang HZ. Chemical components in essential oils from tender branches and leaves of Rhododendron. Yao Xue Xue Bao. 2005;40(9):854–860. [PubMed] [Google Scholar]
- 43.Pulsinelli WA, Brierley JB. A new model of bilateral hemispheric ischemia in the unanesthetized rat. Stroke. 1979;10(3):267–272. doi: 10.1161/01.str.10.3.267. [DOI] [PubMed] [Google Scholar]
- 44.Marrelli SP, Childres WF, Goddard-Finegold J, Bryan RM., Jr. Potentiated EDHF-mediated dilations in the rat middle cerebral artery following ischemia/reperfusion. In: Vanhoutte PM, editor. EDHF 2000. London, UK: Taylor & Francis; 2001. [Google Scholar]
- 45.Xu SY, Bian RL, Chen X. Experimental Methodology of Pharmacology. 3rd edition. Beijing, China: People Health; 2002. [Google Scholar]
- 46.Bai XY, Zhang P, Yang Q, et al. Suxiao jiuxin pill induces potent relaxation and inhibition on contraction in human artery and the mechanism. Evidence-Based Complementary and Alternative Medicine. 2014;2014:11 pages. doi: 10.1155/2014/956924.956924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhang BQ, Sun J, Hu SJ, et al. The research of endothelium-dependent vasorelaxation and mechanism on mongolian milkvetch root. Chinese Journal of Pharmacology and Toxicology. 2005;19(1):442–481. [Google Scholar]
- 48.Markewitz BA, Michael JR, Kohan DE. Cytokine-induced expression of a nitric oxide synthase in rat renal tubule cells. Journal of Clinical Investigation. 1993;91(5):2138–2143. doi: 10.1172/JCI116439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Stipanuk MH, Beck PW. Characterization of the enzymic capacity for cysteine desulphhydration in liver and kidney of the rat. Biochemical Journal. 1982;206(2):267–277. doi: 10.1042/bj2060267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Félétou M, Vanhoutte PM. Endothelium-dependent hyperpolarizations: past beliefs and present facts. Annals of Medicine. 2007;39(7):495–516. doi: 10.1080/07853890701491000. [DOI] [PubMed] [Google Scholar]
- 51.Mori A, Suzuki S, Sakamoto K, Nakahara T, Ishii K. Role of calcium-activated potassium channels in acetylcholine-induced vasodilation of rat retinal arterioles in vivo . Naunyn-Schmiedeberg’s Archives of Pharmacology. 2011;383(1):27–34. doi: 10.1007/s00210-010-0570-1. [DOI] [PubMed] [Google Scholar]
- 52.Sandow SL, Haddock RE, Hill CE, et al. What’s where and why at a vascular myoendothelial microdomain signalling complex. Clinical and Experimental Pharmacology and Physiology. 2009;36(1):67–76. doi: 10.1111/j.1440-1681.2008.05076.x. [DOI] [PubMed] [Google Scholar]
- 53.Cheng Z, Jiang X, Kruger WD, et al. Hyperhomocysteinemia impairs endothelium-derived hyperpolarizing factor-mediated vasorelaxation in transgenic cystathionine beta synthase-deficient mice. Blood. 2011;118(7):1998–2006. doi: 10.1182/blood-2011-01-333310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhao P, Huang X, Wang ZY, et al. Dual effect of exogenous hydrogen sulfide on the spontaneous contraction of gastric smooth muscle in guinea-pig. European Journal of Pharmacology. 2009;616(1–3):223–228. doi: 10.1016/j.ejphar.2009.05.014. [DOI] [PubMed] [Google Scholar]
- 55.Oh GS, Pae HO, Lee BS, et al. Hydrogen sulfide inhibits nitric oxide production and nuclear factor-κB via heme oxygenase-1 expression in RAW264.7 macrophages stimulated with lipopolysaccharide. Free Radical Biology and Medicine. 2006;41(1):106–119. doi: 10.1016/j.freeradbiomed.2006.03.021. [DOI] [PubMed] [Google Scholar]


