Abstract
Background & Aim
Considerable progress has been made in developing anti-fibrotic agents and other strategies to treat liver fibrosis; however, significant long-term restoration of functional liver mass has not yet been achieved. Therefore, we investigated whether transplanted hepatic stem/progenitor cells can effectively repopulate the liver with advanced fibrosis/cirrhosis.
Methods
Stem/progenitor cells derived from fetal livers or mature hepatocytes from DPPIV+ F344 rats were transplanted into DPPIV− rats with thioacetamide (TAA)-induced fibrosis/cirrhosis; rats were sacrificed 1, 2, or 4 months later. Liver tissues were analyzed by histochemistry, hydroxyproline determination, RT-PCR, and immunohistochemistry.
Results
After chronic TAA administration, DPPIV− F344 rats exhibited progressive fibrosis, cirrhosis and severe hepatocyte damage. Besides stellate cell activation, increased numbers of stem/progenitor cells (Dlk-1+, AFP+, CD133+, Sox-9+, FoxJ1+) were observed. In conjunction with partial hepatectomy (PH), transplanted stem/progenitor cells engrafted, proliferated competitively compared to host hepatocytes, differentiated into hepatocytic and biliary epithelial cells, and generated new liver mass with extensive long-term liver repopulation (40.8 ± 10.3%). Remarkably, more than 20% liver repopulation was achieved in the absence of PH, associated with reduced fibrogenic activity (e.g., expression of α-SMA, PDGFRβ, desmin, vimentin, TIMP1) and fibrosis (reduced collagen). Furthermore, hepatocytes can also replace liver mass with advanced fibrosis/cirrhosis, but to a lesser extent than FLSPCs.
Conclusions
This study is a Proof of Principle demonstration that transplanted epithelial stem/progenitor cells can restore injured parenchyma in a liver environment with advanced fibrosis/cirrhosis and exhibit anti-fibrotic effects.
Keywords: Epithelial stem/progenitor cells, cell transplantation, thioacetamide induced fibrosis
Chronic liver disease with cirrhosis is the twelfth leading cause of death in the United States (1). Cirrhosis, the advanced stage of hepatic fibrosis, is mainly caused by viral infection or alcohol abuse; age over 50 is also recognized as a risk factor for cirrhosis (2). Liver transplantation is the only effective therapeutic option for these patients (3). Because of a shortage of donor organs (4) and a dramatic increase in the mortality rate of patients on the liver transplant waiting list during the past decade (5), an alternative strategy to restore liver mass before the end-stage would represent a major clinical advance.
Progressive hepatic fibrosis as a wound-healing response to chronic liver injury leads to accumulation of collagen surrounding liver nodules and further replacement of injured parenchyma by scar tissue, resulting in impaired hepatocyte function (2,6). Hepatic stellate cells are the main contributors to the pathogenesis of liver fibrosis (7,8). Therefore, these cells have represented the primary target to reduce or reverse fibrosis by developing specific anti-fibrotic strategies (9,10). At present, however, therapeutic options in humans are quite limited (7,11). Hepatic cell therapy could be an alternative strategy to generate new functional liver parenchyma in the cirrhotic liver. Stem/progenitor cells – characterized by their high proliferative capacity, ability to differentiate into different lineages, and ability to reconstitute tissue mass (12) – can be isolated from developing or adult liver, as well as from extra-hepatic tissues, and can be transplanted into normal or pre-conditioned recipient liver (13–17). To date, rat fetal stem/progenitor cells (FLSPCs) exhibit the most favorable characteristics for effective liver repopulation by cells transplanted into the (near-)normal liver (13,17–21). Liver repopulation by FLSPCs under non-selective conditions requires only partial hepatectomy (PH) (13,19). This cell type, therefore, may represent an excellent resource for restoring hepatocyte mass in a diseased liver environment.
In the present study, we transplanted FLSPCs and demonstrated that epithelial stem/progenitor cells can engraft, proliferate and differentiate into hepatocytes in the recipient liver with advanced fibrosis/cirrhosis. Surprisingly, transplantation of FLSPCs leads to considerable liver repopulation without the need for PH and reduces active fibrogenesis and net fibrosis. In comparison, mature hepatocytes also repopulate the thioacetamide (TAA)-induced fibrotic liver, but to a lesser extent than FLSPCs. Our model system, therefore, represents an excellent tool to study novel cell transplantation strategies and to elucidate basic mechanisms necessary for successful tissue replacement, critical for development of useful protocols to treat patients with advanced liver diseases.
EXPERIMENTAL PROCEDURES
Animals and TAA administration
Pregnant ED14 DPPIV+ F344 rats were purchased from Charles River. F344-Tg(EGFP) F455/Rrrc rats were obtained from the Rat Resource and Research Center of the University of Missouri-Columbia and used to provide time pregnant EGFP+ F344 rats. Male DPPIV− F344 rats were provided by the Liver Research Center, Albert Einstein College of Medicine (AECOM). 200 mg/kg b.w. TAA was injected i.p. into DPPIV− F344 rats (1.5 to 2 months of age) twice weekly for up to 3 months prior to cell transplantation, followed by 100 or 200 mg/kg b.w. TAA twice weekly after cell infusion. All animal studies were conducted under protocols approved by the Institutional Animal Care and Use Committees of AECOM and University of Pittsburgh in accordance with NIH guidelines.
Isolation of fetal liver cells and hepatocytes
Unfractionated fetal liver cells were isolated from ED14/15 fetal livers of pregnant DPPIV+ or DPPIV+/EGFP+ F344 rats, as described previously (18,19). Hepatocytes were isolated from livers of adult DPPIV+ F344 rats. Detailed information concerning the cell isolation procedures can be found in Supplemental Material & Methods of Ref. 21.
Cell transplantation and liver repopulation
Fetal liver cells (viability >95%) or adult hepatocytes (viability >80%) were transplanted through the portal vein into DPPIV− F344 rats (22) treated with TAA or untreated recipients with or without 2/3 PH (hepatectomized liver lobes were used to assess liver fibrosis and for other studies). After rats were sacrificed at different times following cell transplantation, liver repopulation was determined by enzyme histochemistry for DPPIV, as described previously (18,19). For engraftment studies, transplanted fetal liver cells were detected by immunohistochemistry for EGFP.
RT-PCR and qRT-PCR analysis
RT-PCR analysis
Total RNA was extracted from snap frozen liver tissue derived from TAA-treated DPPIV− F344 rats and untreated age-matched control rats. Qualitative RT-PCR analyses were performed at least twice. Quantitative real-time RT-PCR was performed in doublet/triplicate, as described in Supplemental Material & Methods of Ref. 21. A list of the primers is shown in Supplemental Table 1.
Histochemistry and Immunohistochemistry
Information concerning histochemical and immunohistochemical analyses can be found in Supplemental Material & Methods.
Hydroxyproline (HYP) determination
Using two different fragments per liver, the HYP content was determined biochemically, as described previously (23).
Microscopy and Imaging
Tissue slides were examined under an AxioObserver Z1 microscope. Images were obtained with an AxioCam ICc3, ICm1 or HRc camera and processed with AxioVision 4.8 or ZEN imaging software (Carl Zeiss MicroImaging).
Data analysis
Data were analyzed using SigmaStat 2.01 (SPSS Scientific), GraphPad Prism5 (GraphPad), and NIS-Elements D (Nikon) software and are reported as mean ± SEM.
RESULTS
Characterization of hepatic fibrosis/cirrhosis after TAA treatment
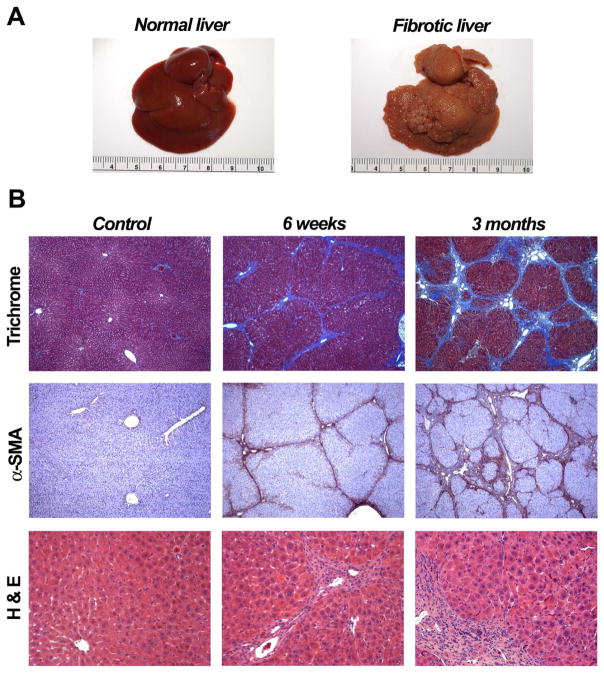
After chronic TAA administration (200 mg/kg b.w., twice weekly), liver fibrosis was assessed (Fig. 1) using the Laennec classification system (24). At 6 weeks, the progressive liver injury produces moderate fibrosis and mild cirrhosis, i.e., predominant nodularity caused by narrow fibrous septa bridging portal areas. By 3 months, more advanced fibrosis occurs, leading to moderate to severe cirrhosis in different parts of the liver. Previously narrow septa have thickened; many nodules are surrounded by broad septa and in some areas, extensive tracts of fibrous tissue contain a few larger nodules and small regions of fragmented nodules. In addition, immunohistochemistry for α-SMA in liver sections of TAA-treated rats, compared to non-treated control rats, showed increasing numbers of α-SMA-positive cells in intralobular septa in areas of fibrosis, indicating activation of hepatic stellate cells.
Figure 1.
Liver fibrosis in thioacetamide (TAA)-treated rats. 200 mg/kg TAA was injected i.p. into mutant DPPIV− F344 rats twice weekly for up to 3 months. (A) Normal rat liver and liver at 3 months after administration of TAA are shown. (B) Pathologic changes at 6 weeks and 3 months were determined using Masson’s trichrome staining, immunohistochemistry for α-SMA, and H&E staining. Tissue samples derived from non-treated rats were used as controls. Original magnification, x50 (upper and middle panels), x200 (lower panels).
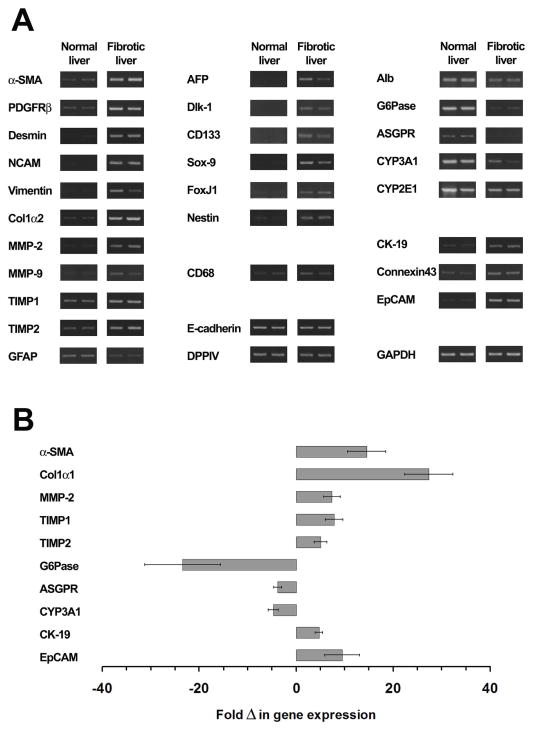
To determine the expression levels of genes relevant to advanced fibrosis/cirrhosis, we performed RT-PCR analysis in liver tissues 3 months after TAA administration compared to age-matched non-treated liver (Fig. 2A & B). We observed elevated expression of α-SMA, platelet-derived growth factor receptor β (PDGFRβ), desmin, neural cell adhesion molecule (N-CAM), and vimentin mRNAs (Fig. 2A). In addition, after induction of advanced fibrosis/cirrhosis, procollagen α2(I) (Col1α2), matrix metalloproteinase-2 (MMP-2), MMP-9, tissue inhibitor of metalloproteinase-1 (TIMP1), TIMP2 were up-regulated, and glial fibrillary acidic protein (GFAP) was down-regulated (Fig. 2A). The expression profiles of these genes clearly reflect activation of stellate cells and ongoing fibrogenesis. Furthermore, biochemical analysis of the relative HYP content in non-treated vs. TAA-treated liver (n=6/6 rats) increased from 0.23 ± 0.02 to 1.38 ± 0.18 mg HYP/g liver, indicating advanced fibrosis/cirrhosis at 3 months after TAA administration.
Figure 2.
Gene expression in TAA-induced liver fibrosis. (A) Liver RNA extracts from two TAA-treated rats at 3 months after starting TAA administration compared to two age-matched non-treated rats were analyzed for mRNA expression. These experiments were performed at least twice. (B) Quantitative RT-PCR analysis of selected genes in fibrotic liver. Values are mean ± SEM of liver samples from TAA-treated rats (n=4), expressed as fold differences in gene expression compared to age-matched non-treated rats (n=4). One representative experiment for each gene, from at least 2 replicate experiments, is shown. qRT-PCR analysis was also performed for AFP, Dlk-1, Sox-9 and FoxJ1, expressed in all 4 fibrotic liver samples (Ct values ranged from 29 to 33) were undetectable in normal liver.
We observed increased expression of AFP, Dlk-1, CD133, Sox-9, FoxJ1 and nestin mRNAs (Fig. 2A), indicating increased numbers of progenitor cells (14,16,25–28) after induction of advanced fibrosis/cirrhosis. Compared to normal hepatic tissue, liver samples with advanced fibrosis also showed down-regulation of glucose-6-phosphatase (G6Pase), asialoglycoprotein receptor (ASGPR), and cytochrome 3A1 (CYP3A1) mRNAs (all of which are related to hepatocyte-specific cell functions), indicating hepatocellular damage or loss. In contrast, biliary epithelial cell-specific genes (cytokeratin-19 [CK-19], connexin43, EpCAM) were upregulated in strongly fibrotic liver (Fig. 2A). These data were confirmed and fold changes quantified by qRT-PCR analysis for selected genes (Fig. 2B).
Repopulation studies in rats with TAA-induced liver fibrosis transplanted with hepatic cells
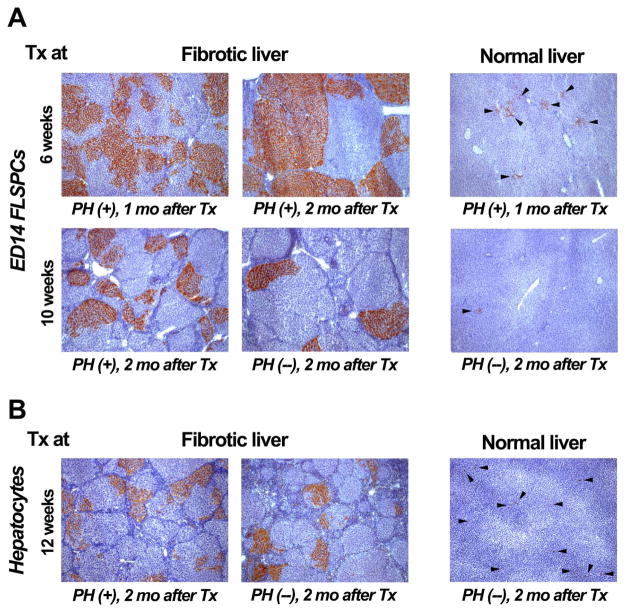
In a pilot experiment, we tested whether fetal liver cells are capable of repopulating the fibrotic liver. To induce moderate hepatic fibrosis, 200 mg/kg TAA was injected into DPPIV− F344 rats twice weekly for 6 weeks, followed by a maintenance dose of 100 mg/kg TAA after cell transplantation. Since repopulation of the normal liver by FLSPCs occurs only after two-thirds PH (13,19), PH was performed just prior to cell infusion (~1.5 × 107 ED14 unfractionated fetal liver cells, of which ~2.5% are AFP+/CK-19+ bipotential stem/progenitor cells; refs. 17,22). At both one and two months after cell transplantation, we observed extensive liver repopulation with more than 50% tissue replacement in many areas of TAA-treated recipient liver (n=3 rats) (Fig. 3A, upper left & middle panel). In contrast, only small cell clusters were observed in age-matched rats that did not receive TAA, with very little liver repopulation at 1 month (n=2) (Fig. 3A, upper right panel).
Figure 3.
Enhanced repopulation of fibrotic liver by transplanted ED14 FLSPCs and adult hepatocytes. DPPIV enzyme histochemistry showing repopulation by unfractionated wt. ED14 FLSPCs (~1.5 × 107, A) and adult hepatocytes (~2 × 106, B, left panel; ~5 × 106, B, middle & right panel) transplanted into DPPIV− TAA-treated rats at 6, 10 or 12 weeks after initiating TAA administration (200 mg/kg TAA, followed by a maintenance dose of 100 mg/kg TAA twice weekly after cell transplantation) vs. non-treated normal rats in conjunction with (+) or without PH (−). Rats were sacrificed at 1 or 2 months (mo) after cell infusion. Single cells/small cell clusters are highlighted by arrowheads (A & B, right panels). Original magnification, x50.
Having demonstrated that transplanted fetal hepatic cells can repopulate a liver with moderate fibrosis, we next tested whether cell transplantation is feasible in recipient rats with advanced fibrosis. After inducing advanced liver fibrosis in DPPIV− F344 rats (200 mg/kg TAA, twice weekly for 10 weeks; followed by 100 mg/kg TAA after cell transplantation), we infused ~1.5 × 107 ED14 fetal liver cells into TAA-treated rats in conjunction with PH. At 2 months after cell transplantation (n=3), we observed small and large DPPIV+ cell clusters in host livers with extensive fibrosis. Many repopulating cell clusters encompassed entire fibrotic lobules (Fig. 3A, lower left panel). Although many areas showed extensive liver repopulation with multiple adjacent DPPIV+ regenerating nodules, other areas showed only limited repopulation. The majority of transplanted FLSPCs differentiated into hepatocytic cells; however, substantial bile duct generation, mainly within the fibrotic bands, was also observed (Fig. 4B, below). Furthermore, we transplanted FLSPCs into TAA-treated rats w/o PH and normal rats w/o PH (n=4/2) and observed scattered repopulation clusters in the fibrotic rat livers. Some of theseclusters were of large size (Fig. 3A, lower middle panel), in contrast to normal rats w/o PH in which no liver repopulation was achieved by FLSPCs (Fig. 3A, lower right panel).
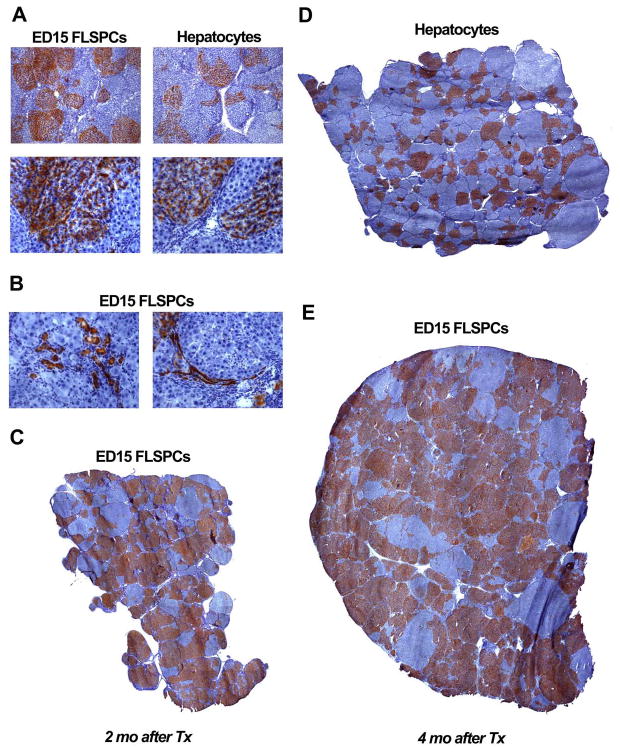
Figure 4.
Repopulation of transplanted cells in host liver with progressing fibrosis/cirrhosis. Donor cells (~8 × 107 fetal liver cells vs. ~2 × 106 hepatocytes) were infused in conjunction with PH into rats at 3 months after starting TAA administration, which was continued at the same dose, thereafter (200 mg/kg, twice weekly). Liver repopulation was analyzed at 2 (A,B,C) and 4 months (D,E) after cell transplantation. ED15 FLSPCs differentiated into hepatocytes (A, lower left panel) and bile ducts (B). Original magnification, x50 (A, upper panels), x200 (A, lower panels; B). (C,D,E) Whole liver sections with the highest repopulation level observed at 2 and 4 months after cell transplantation are shown. Images contain 30, 41 or 64 merged adjacent microscopic fields (original magnification, x50) and can be zoomed in online.
Although a limiting factor in liver repopulation might be the ability of hepatocytes, which are of large size, to engraft in the fibrotic liver tissue (29), we investigated the repopulation potential of differentiated mature hepatic cells in the TAA fibrosis model. Hepatocytes were infused into rats with advanced liver fibrosis/cirrhosis (produced by administration of 200 mg/kg TAA, twice weekly for 10–12 weeks; followed by 100 mg/kg TAA after cell transplantation). In two TAA-treated rats transplanted with ~1.5 or 2 × 106 hepatocytes in conjunction with PH, DPPIV+ hepatocytic clusters were observed in both rats at 2 months, remarkably with up to 10% liver repopulation in the rat transplanted with ~2 × 106 hepatocytes (Fig. 3B, left panel). In addition, we transplanted ~2 or 5 × 106 hepatocytes into TAA-treated rats w/o PH (n=5). Small and larger repopulating hepatocyte clusters were seen in all rats with advanced fibrosis/cirrhosis (Fig. 3B, middle panel). In contrast, normal untreated rats transplanted with similar numbers of hepatocytes w/o PH (~5 × 106 cells; n=3) showed only single cells in the parenchyma, without cluster formation or significant liver repopulation (Fig. 3B, right panel).
Comparative repopulation studies in rats with advanced fibrosis/cirrhosis transplanted with FLSPCs vs. mature hepatocytes
For definitive long-term repopulation studies under the most stringent fibrosis conditions, we infused cells into rats at 3 months after starting TAA administration (200 mg/kg) and continued with the same TAA dose after cell infusion. In these experiments, we attempted to inoculate equivalent numbers of hepatic epithelial lineage cells, based on our previous analysis showing that “bipotential” stem/progenitor cells (AFP+/CK-19+) represent ~2.5% of total unfractionated ED14 fetal liver cells (17). We therefore estimated that 8 × 107 unfractionated fetal liver cells contained ~2 × 106 “bipotential” FLSPCs, comparable to 2 × 106 mature hepatocytes. To obtain sufficient numbers of cells for these studies, we isolated unfractionated hepatic stem/progenitor cells from ED15 fetal livers.
While maintaining the TAA dose after cell transplantation into advanced fibrotic rat liver (Figs. 4 & 5), levels of 35.7 ± 6.4% and 40.8 ± 10.3% repopulation were achieved with FLSPCs at 2 and 4 months, respectively (n=4/4). FLSPCs differentiated into hepatocytes (Fig. 4A) and bile duct cells. The large DPPIV+ clusters of hepatocytes typically had DPPIV+ bile ducts along the edges of fibrous septae (see Fig. 4). In some cases, DPPIV+ bile ducts extended into surrounding DPPIV-negative regions (Fig. 4B), presumably resulting from a stimulus for bile duct proliferation in the injured liver. The cells formed large DPPIV+ clusters with extensive tissue replacement (Fig. 4C & E). In comparison, substantial numbers of transplanted mature hepatocytes engrafted in the cirrhotic liver, proliferated long-term, and replaced diseased liver mass (Fig. 4A, right panels, & 4D). However, liver repopulation levels with mature hepatocytes were lower at 2 and 4 months after cell transplantation (8.3 ± 2.0% and 10.5 ± 3.2%, respectively; n=3/4) compared to that obtained with FLSPCs (35.7 ± 6.4% and 40.8 ± 10.3%, respectively). Although there was higher repopulation with transplanted stem/progenitor cells, which indicates a higher engraftment or proliferation rate, our findings with mature hepatocytes also represent a significant new observation in the fibrotic liver.
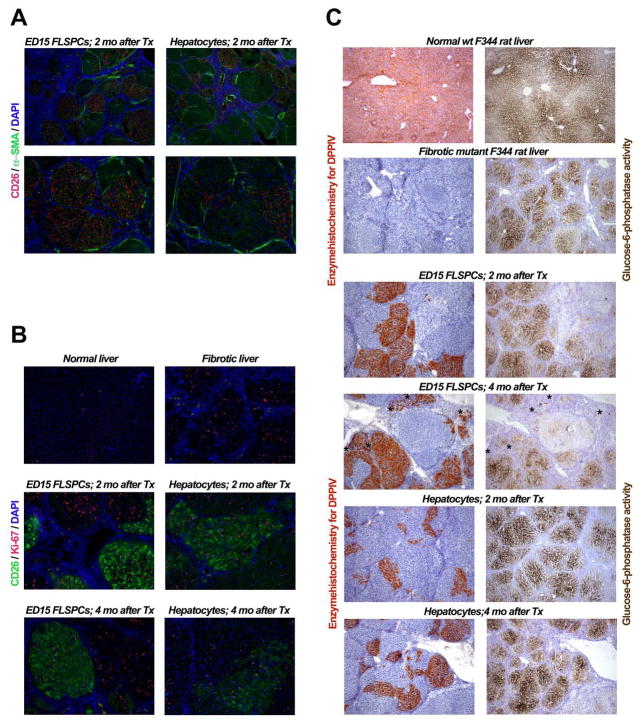
Figure 5.
Expansion of transplanted ED15 FLSPCs and hepatocytes transplanted in conjunction with PH into recipient rats with progressing fibrosis/cirrhosis. (A) Localization of transplanted cell clusters in recipient liver with advanced fibrosis/cirrhosis using simultaneous immunohistochemistry for DPPIV (CD26) and α-SMA. (B) Detection of proliferating cells in normal, fibrotic and recipient liver after cell transplantation, using co-staining for CD26 and Ki-67. (C) Functional integration of transplanted cells in cirrhotic liver. Normal lobular distribution and gradients of glucose 6-phosphatase expression (C, upper right panel) in untreated wt DPPIV+ F344 rat liver (C, upper left panel) compared to cirrhotic liver after 3 months TAA administration (C, right panel, 2nd row) in mutant F344 rats negative for DPPIV (C, left panel, 2nd row). The panels in C, 3rd to 6th row, compare DPPIV and G6Pase staining of nearby liver sections. Where transplanted cells have proliferated and generated new hepatocytes, large DPPIV+ cell clusters showed normal expression of glucose-6-phosphatase. Panels in the 4th row demonstrate that transplanted stem/progenitor cellsdifferentiate into both DPPIV+ bile ducts (see asterisks) and hepatocytes, but only hepatocytic clusters express glucose-6-phosphatase. Original magnification, x50 (A, upper panels; C), x100 (A, lower panels; B).
Simultaneous immunohistochemical analysis for DPPIV (CD26) and α-SMA (Fig. 5A) showed that DPPIV+ cell clusters derived from transplanted FLSPCs completely replaced host hepatocytes within liver nodules surrounded by fibrous host tissue containing α-SMA+ cells (Fig. 5A, left panels), a phenomenon also observed after hepatocyte transplantation (Fig. 5A, upper right panel). Double label immunohistochemistry for DPPIV (CD26) and Ki-67 (Fig. 5B) showed that FLSPC and hepatocyte-derived cell clusters contained actively proliferating cells for up to 4 months (Fig. 5B, middle and lower panels) and “competed” with proliferating host hepatocytes (Fig. 5B, upper right panel). Furthermore, DPPIV and G6Pase expressing hepatocytic cells were detected at 2 and 4 months after transplantation of FLSPCs or hepatocytes (Fig. 5C), demonstrating hepatocyte-specific metabolic activity of transplanted cells.
Engraftment and repopulation of the liver with advanced fibrosis/cirrhosis after transplantation of fetal liver cells in the absence of PH
Since we showed that FLSPCs can form cell clusters in the fibrotic liver without PH (Fig. 3A), we performed additional cell transplantations to determine whether substantial liver repopulation could be obtained under these conditions. These studies required infusion of ~1.5 × 108 donor cells into DPPIV− rats without PH at 3 months after TAA administration. At 4 months after cell transplantation, 23.8 ± 4.4% liver repopulation was achieved and G6Pase-expressing, differentiated cells were integrated in the cirrhotic liver environment. Compared to non-transplanted TAA-treated livers, analysis of mRNA showed that FLSPC transplantation up-regulated genes related to specific hepatocytic functions (G6P, 3.7-fold; CYP3A1, 3.5-fold; TAT, 1.8-fold; n=3/3) (Supplemental Figure 1).
The above findings suggested that a significant number of FLSPCs can engraft in the cirrhotic liver and substantial repopulation can be achieved in the absence of PH. FLSPC transplantation under these conditions is well tolerated and we observed a mortality of 11%.
We next studied the dynamics of donor cell engraftment and expansion immediately after FLSPC infusion. Since DPPIV is not expressed before ED18, ED15 FLSPCs were isolated from EGFP-marked transgenic F344 rats to identify engrafted cells. We infused ~1.5 × 108 EGFP-expressing fetal liver cells into 3 DPPIV− rats at 3 months after TAA administration without PH. At days 1 and 3 after cell infusion, single EGFP+ cells (Fig. 6A) and small groups of EGFP+ cells (Fig. 6B) were detected in the host liver parenchyma, respectively, demonstrating successful engraftment of transplanted stem/progenitor cells into the cirrhotic liver. By day 7, expanding fetal liver cells formed small cell clusters primarily along the border of fibrotic bands (Fig. 6C), demonstrating ongoing repopulation in the cirrhotic liver tissue environment.
Figure 6.
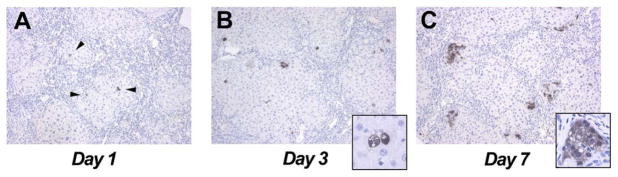
Engraftment and early expansion of ED15 FLSPCs transplanted into recipients with advanced fibrosis/cirrhosis without PH. Donor fetal liver cells (~1.5 × 108) were infused into rats at 3 months after starting TAA administration (200 mg/kg, twice weekly). Single EGFP+ cells were detected at day 1 (A; arrowheads), which divided at day 3 (B), and formed small cell clusters by day 7 (C). Original magnification, x100 (A–C), x630 (insets).
Effect of stem/progenitor cells on fibrogenesis
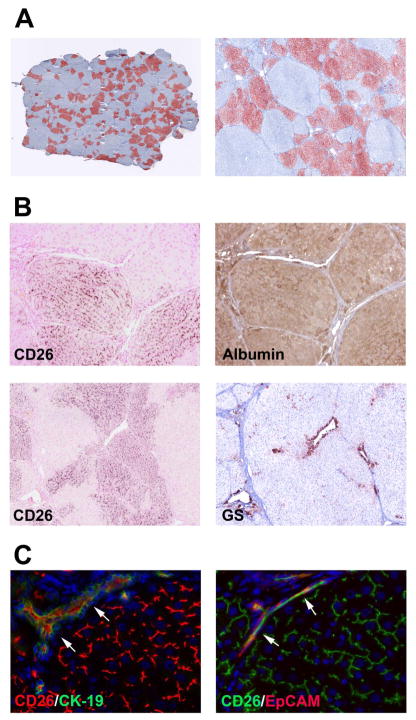
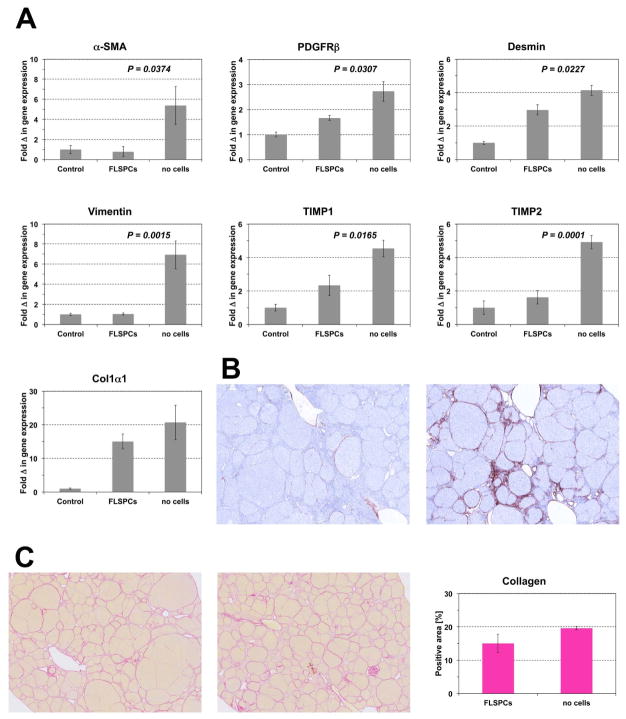
Having demonstrated that transplanted fetal hepatic cells can engraft and significantly repopulate the recipient liver with advanced fibrosis/cirrhosis, we next determined whether stem/progenitor cells can effect fibrogenesis and the extent of liver fibrosis. After inducing advanced fibrosis in DPPIV− rats (200 mg/kg TAA, twice weekly for 3 months), we infused ~1.8 × 108 unfractionated ED15 fetal liver cells into TAA-treated rats that had not undergone PH (n=6). Two months later, TAA administration was discontinued and rats were sacrificed 5 weeks later. Other rats received identical TAA-treatment without cell transplantation (n=6). Repopulation analysis of the cell transplant recipients showed that 26.9 ± 6.3% of the liver mass was repopulated by FLSPC-derived hepatocytes that expressed albumin at the same level as observed in adjacent host liver tissue (Figure 7A & B). Selective expression of glutamine synthetase in the centrilobular regions of engrafted liver tissue suggested complete zonal differentiation by repopulating FLSPC-derived hepatocytes (Figure 7B, lower panels). Double label immunohistochemistry for CD26 and CK-19/EpCAM demonstrated that transplanted stem/progenitor cells also differentiated into bile duct cells (Figure 7C). Furthermore, a marked decrease of α-SMA, PDGFRβ, desmin, vimentin, TIMP1, and TIMP2 mRNA was observed in the FLSPC-transplanted group (Figure 8A), indicating a dramatic reduction of the fibrinogenic process. This also correlated with a marked reduction in activated α-SMA-positive stellate cells (Figure 8B). Histological examination of Sirius Red-Stained sections showed more macronodules in FLSPC recipients, i.e., intact regions without internal fibrosis (Figure 8C). Quantification of Sirius Red-Stained collagen showed less collagen in the liver of FLSPC recipients compared to non-transplanted rats (15.4 ± 2.8% vs. 19.6 ± 0.5%, Figure 8C, right panel; see also Col1α1 mRNA levels in Figure 8A), although these changes were not statistically significant.
Figure 7.
Repopulation and functional incorporation of differentiated, donor-derived cells after transplantation without PH. ED15 fetal liver cells (~1.8 × 108 cells) were transplanted into DPPIV− rats 3 months after starting TAA administration (A). Images contain 65 or 25 tiled microscopic fields (original magnification, x50 and x100). (B) Patterns of DPPIV (CD26) and albumin expression, and DPPIV and glutamine synthetase (GS) expression in consecutive liver sections demonstrate that DPPIV+ cells differentiated into hepatocytes and become incorporated into morphologically normal hepatocyte plates. (C) Transplanted FLSPCs showed a canalicular pattern of CD26 staining characteristic of mature hepatocytes and also differentiated into bile ducts coexpressing CD26 with CK-19 or EpCAM (see arrows). Original magnification, x10 and x5 (B), x40 (C).
Figure 8.
Effect of FLSPC transplantation on hepatic fibrogenesis. ED15 fetal liver cells (~1.8 × 108 cells) were transplanted into DPPIV− rats 3 months after starting TAA administration, which was continued for 2 months thereafter. Five weeks later, rats were sacrificed. (A) Quantitative RT-PCR analysis for mRNA of genes for stellate cell activation and fibrogenesis. Values are mean ± SEM of liver samples from FLSPC-transplanted fibrotic rats (n=6) or non-cell transplanted fibrotic rats (n=6) compared to age-matched normal control rats, set at a value of 1 (n=3). One representative example from at least 3 replicate experiments is shown. (B) Immunohistochemical detection of α-SMA-positive cells in rat livers with (left) or without (right) cell transplantation. Images contain 25 adjacent microscopic fields (original magnification, x100). (C) Selected areas of Sirius Red-Stained tissue sections of rat livers with (left) or without (middle) cell transplantation. Quantification of Sirius Red-Stained collagen (right). Values are mean ± SEM of whole liver sections (n=6/6).
DISCUSSION
Several rodent models of cirrhosis in rodents have been established to study the mechanism of fibrosis progression or anti-fibrotic therapies (reviewed in ref. 30). To develop a cell transplantation model for epithelial stem/progenitor cells in a cirrhotic recipient background, we induced fibrosis/cirrhosis in the mutant DPPIV− F344 rat (31), an inbred strain originally utilized to follow the fate of transplanted wt. DPPIV+ hepatocytes in DPPIV− recipients (32). TAA-induced liver fibrosis was selected in preference to CCl4 and other known fibrosis models, because it produces more extensive and stable fibrosis and is most similar to human fibrosis in clinical progression (30,33,34). We demonstrated that advanced fibrosis/cirrhosis was established at 3 months after chronic TAA administration, indicated by characteristic hepatic lesions and collagen deposition (33). The cirrhotic liver showed increased HYP, α-SMA, PDGFRβ, procollagen, TIMP1, and MMP-2, indicating increased numbers of activated stellate cells and ongoing fibrogenesis (8,35,36), and decreased GFAP, which is down-regulated in activated stellate cells in advanced fibrosis (37). Advanced fibrosis/cirrhosis in the recipient liver was further supported by decreased levels of unique hepatocyte-specific mRNA transcripts (e.g., ASGPR, CYP3A1, and G6Pase mRNA) (see also Fig. 5C). Finally, an increased number/activation of cholangiocytes, which secrete fibrogenic growth factors and activate stellate cells in fibrotic/cirrhotic liver (30), was reflected by augmented CK-19, connexin43 and EpCAM levels in TAA-treated liver.
Using the TAA-induced experimental model of liver fibrosis/cirrhosis, we have made five major observations. First, we showed that rat fetal liver-derived epithelial stem/progenitor cells can engraft into the recipient liver with advanced fibrosis/cirrhosis and differentiate into hepatocytes, i.e., cells with hepatocyte-specific morphology and metabolic function. Second, the engrafted cells expand and replace failing liver mass within a short time after cell infusion. Third, efficient liver repopulation by transplanted epithelial stem/progenitor cells can be achieved in a densely fibrotic liver without an additional stimulus provided by liver regeneration. Fourth, the engrafted liver exhibited reduced fibrinogenic activity. Fifth, we also showed that transplanted mature hepatocytes proliferate and form large cell clusters with substantial tissue replacement.
Rodent fibrosis models are crucial to investigate the efficiency of anti-fibrotic agents (30). Since it is impossible to distinguish between the anti-inflammatory and anti-fibrotic effects of agents tested in hepatotoxin-induced fibrosis models, CCl4 or TAA is generally withdrawn during drug administration and the rate of fibrosis recovery is determined to assess the effectiveness of the tested treatment (30). Because the main focus of the present study was to assess whether transplanted epithelial stem/progenitor cells can restore hepatic parenchyma in a chronically injured liver environment during evolution of fibrosis/cirrhosis, we continued TAA administration after cell infusion. Then, to evaluate whether transplanted FLSPCs have an anti-fibrotic effect, in some studies, we discontinued the TAA administration after successful cell engraftment and repopulation.
Potential obstacles to effective repopulation of fibrotic tissue include infarction of the liver by infused cells or poor engraftment of transplanted cells. Indeed, fibrotic rats infused through the portal vein with 5 × 106 hepatocytes in conjunction with PH died within 48 hours (n=3). Infusion of 2 × 106 cells was better tolerated, although a noticeable mortality was still observed (data not shown). Rat FLSPCs are much smaller than adult hepatocytes (10–12 μm vs. 20–35 μm diameter, respectively, see ref. 13; for human fetal cells, see ref. 15), which allowed us to infuse high numbers of unfractionated fetal liver cells (8 × 107 or 1.5 × 108 cells, contains ~2 × 106 or 4 × 106 “bipotential” FLSPCs, respectively), with or without PH. Importantly, a preliminary study of FLSPCs enriched by immuno-magnetic bead cell sorting showed that we can significantly increase the number of FLSPCs transplanted without increasing the total cell number infused (see Supplemental Figure 2).
Previously, we have demonstrated that FLSPCs can effectively repopulate the (near-)normal liver, but only in conjunction with PH (13,19), suggesting that PH is required for their engraftment and/or expansion (19). However, the present study showed substantial early engraftment and efficient repopulation after FLSPC infusion into the TAA-treated recipient liver without PH. These results suggest that chronic injury during evolution of cirrhosis, or the altered cirrhotic liver microenvironment, favors engraftment and proliferation of transplanted epithelial stem/progenitor cells. However, to achieve long-term correction of cirrhosis after hepatic stem cell transplantation, additional modifications of the microenvironment may be necessary (38). During the past 2 decades, several model systems have been developed to study liver repopulation by transplanted hepatic cells (reviewed in ref. 17). In these models, substantial liver replacement by transplanted wt. hepatocytes was achieved by genetic modifications of the host liver causing massive hepatic injury (39,40) or by inhibition of the proliferative capacity of host hepatocytes through administration of DNA damaging agents or liver irradiation (41–43). Liver replacement was observed in the α1-antitrypsin deficient transgenic mouse, in which the proliferation of endogenous hepatocytes is impaired (44). These repopulation models are characterized by a strong growth advantage of transplanted cells compared to host hepatocytes. Although previous studies demonstrated increased survival of rats with decompensated liver cirrhosis after intrasplenic hepatocyte transplantation (45), to our knowledge, there is no previous report showing significant hepatic tissue replacement by transplanted epithelial stem/progenitor cells in an experimental model of advanced liver fibrosis/cirrhosis.
There are currently only a few pioneering human studies of mature or fetal hepatic cell transplantations in patients with chronic liver diseases (46–48). Nevertheless, animal studies must provide critical understanding of the basic requirements and mechanisms for effective liver repopulation. In the present study, using experimental conditions that reflect circumstances similar to human fibrosis/cirrhosis, we have demonstrated that transplanted progenitor cells can efficiently proliferate after their engraftment and are capable of differentiating into hepatic cell lineages. In conjunction with replacement of 20–30% of hepatocytic mass by FLSPCs, hepatic fibrogenesis was reduced, as evidenced by reduced stellate cell activation, decreased expression of fibrogenesis genes, and reduced collagen in the tissue. Thus, transplantation of epithelial stem/progenitor or FLSPC-like cells engineered via iPS cell technology, perhaps combined with targeted anti-fibrotic therapy, holds great promise for treatment of patients with end-stage liver diseases.
Supplementary Material
Repopulation and functional incorporation of differentiated donor cells transplanted without PH into rats with progressing fibrosis/cirrhosis. 1.4–1.6 × 108 ED15 fetal liver cells were transplanted into DPPIV− rats at 3 months after starting TAA administration, which was continued thereafter (200 mg/kg, twice weekly). (A,B) Patterns of DPPIV expression (A) and G6Pase activity (B) in two adjacent liver sections from one recipient rat at 4 months after cell transplantation. These liver sections demonstrate that DPPIV+ cells differentiated into hepatocytic cells, become incorporated into the fibrotic liver structure and exhibit G6Pase expression. (C) Quantitative RT-PCR analysis for mRNA levels of selected genes specific for hepatocytic functions in recipient livers at 4 months after FLSPC transplantation into rats with advanced fibrosis/cirrhosis. Values are mean ± SEM of liver samples from FLSPC-transplanted rats (n=3) and are expressed as fold differences in gene expression compared to liver samples derived from age-matched non-transplanted TAA-treated rats (n=3). One representative example from 3 replicate experiments is shown.
Liver tissue replacement without partial hepatectomy after cell infusion of enriched ED15 FLSPCs into a recipient rat with progressing fibrosis/cirrhosis. After isolation of unfractionated fetal liver cells, negative selection was performed by incubating ~3.5 × 108 cells with a monoclonal mouse antibody specific for rat erythrocytes and erythroid progenitors (BD Biosciences) at 4°C for 30 min. Cells were washed twice in Hanks’ balanced salt solution (HBSS) containing 0.3% BSA, 0.8 mM MgCl2, 10 mM HEPES and incubated with rat anti-mouse IgM bound to magnetic microbeads (Miltenyi) at 4°C for 30 min. The cell suspension was washed and passed through LS-columns placed in a MidiMACS™ Separator (Miltenyi). Erythroid cells were retained in the magnetic field (column); non-erythroid cells enriched with epithelial stem/progenitor cells passed through the columns. (A,B) Cytospins of unfractionated vs. cytospins of enriched ED15 FLSPCs were stained with mouse anti-E-cadherin antibody (BD Biosciences), followed by CyTm3-conjugated donkey anti-mouse IgG (Jackson) as secondary antibody. Unfractionated fetal liver cells contained 2.6% E-cadherin+ epithelial cells (A). After enrichment with immunomagnetic beads, this increased to 11.4% (4.4-fold enrichment) in the erythroid-depleted fraction (B). (C) Enriched fetal liver cells (~8 × 107) from DPPIV+ F344 rats were infused into a DPPIV− F344 rat with progressing fibrosis/cirrhosis without partial hepatectomy. At one month after cell infusion, the rat was sacrificed and liver sections were stained using enzyme histochemistry for DPPIV to detect expanding DPPIV+ cell clusters. The majority of the liver was successfully engrafted by transplanted fetal liver cells, which formed DPPIV+ cell clusters; some of them were already encompassing entire fibrotic lobules. The image contains 4 merged adjacent microscopic fields. Original magnification x400 (A,B) and x50 (C).
Acknowledgments
Financial support: Supported by NIH grant R01 DK090325 (to M.O.) and a Pilot and Feasibility Study (to M.O.) from P30 DK041296 (to D.A.S.), and R01 DK017609 (to D.A.S.).
The authors would like to thank Dr. Scott L. Friedman (Division of Liver Diseases, Mount Sinai School of Medicine, New York, NY) for his advice in using the TAA-induced fibrosis model. Furthermore, the excellent technical assistance of Ms. Amanda Franklin is gratefully acknowledged.
Footnotes
Disclosures: The authors indicate no potential conflicts of interest.
Contributions: M.I.Y. and Y.X carried out experiments and analyzed data. D.A.S. contributed to experimental design and data analyses. J.L. performed histological subclassification of fibrosis/cirrhosis. M.O. designed the studies and performed experiments, analyzed data and wrote the manuscript. All authors read and commented on the manuscript.
References
- 1.Xu J, Kochanek KD, Murphy SL, et al. Deaths: final data for 2007. Natl Vital Stat Rep. 2010;58:1–135. [PubMed] [Google Scholar]
- 2.Schuppan D, Afdhal NH. Liver cirrhosis. Lancet. 2008;371:838–851. doi: 10.1016/S0140-6736(08)60383-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cárdenas A, Ginès P. Management of patients with cirrhosis awaiting liver transplantation. Gut. 2011;60:412–421. doi: 10.1136/gut.2009.179937. [DOI] [PubMed] [Google Scholar]
- 4.Perera MT, Mirza DF, Elias E. Liver transplantation: Issues for the next 20 years. J Gastroenterol Hepatol. 2009;24:S124–131. doi: 10.1111/j.1440-1746.2009.06081.x. [DOI] [PubMed] [Google Scholar]
- 5.http://optn.transplant.hrsa.gov/latestData/rptData.asp
- 6.Hernandez-Gea V, Friedman SL. Pathogenesis of liver fibrosis. Annu Rev Pathol. 2011;6:425–456. doi: 10.1146/annurev-pathol-011110-130246. [DOI] [PubMed] [Google Scholar]
- 7.Henderson NC, Iredale JP. Liver fibrosis: cellular mechanisms of progression and resolution. Clin Sci (Lond) 2007;112:265–280. doi: 10.1042/CS20060242. [DOI] [PubMed] [Google Scholar]
- 8.Friedman SL. Mechanisms of hepatic fibrogenesis. Gastroenterology. 2008;134:1655–1669. doi: 10.1053/j.gastro.2008.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schuppan D, Popov Y. Rationale and targets for antifibrotic therapies. Gastroenterol Clin Biol. 2009;33:949–957. doi: 10.1016/j.gcb.2009.07.021. [DOI] [PubMed] [Google Scholar]
- 10.Kisseleva T, Brenner DA. Anti-fibrogenic strategies and the regression of fibrosis. Best Pract Res Clin Gastroenterol. 2011;25:305–317. doi: 10.1016/j.bpg.2011.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Friedman SL. Evolving challenges in hepatic fibrosis. Nat Rev Gastroenterol Hepatol. 2010;7:425–436. doi: 10.1038/nrgastro.2010.97. [DOI] [PubMed] [Google Scholar]
- 12.Loeffler M, Potten CS. Stem cells and cellular pedigrees - a conceptual introduction. In: Potten CS, editor. Stem cells. London: Academic Press; 1997. pp. 1–29. [Google Scholar]
- 13.Dabeva MD, Petkov PM, Sandhu J, et al. Proliferation and differentiation of fetal liver epithelial progenitor cells after transplantation into adult rat liver. Am J Pathol. 2000;156:2017–2031. doi: 10.1016/S0002-9440(10)65074-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Suzuki A, Sekiya S, Onishi M, et al. Flow cytometric isolation and clonal identification of self-renewing bipotent hepatic progenitor cells in adult mouse liver. Hepatology. 2008;48:1964–1978. doi: 10.1002/hep.22558. [DOI] [PubMed] [Google Scholar]
- 15.Schmelzer E, Zhang L, Bruce A, et al. Human hepatic stem cells from fetal and postnatal donors. J Exp Med. 2007;204:1973–1987. doi: 10.1084/jem.20061603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cardinale V, Wang Y, Carpino G, et al. Multipotent stem/progenitor cells in human biliary tree give rise to hepatocytes, cholangiocytes, and pancreatic islets. Hepatology. 2011;54:2159–2172. doi: 10.1002/hep.24590. [DOI] [PubMed] [Google Scholar]
- 17.Oertel M. Fetal liver cell transplantation as a potential alternative to whole liver transplantation? J Gastroenterology. 2011;46:953–965. doi: 10.1007/s00535-011-0427-5. [DOI] [PubMed] [Google Scholar]
- 18.Sandhu JS, Petkov PM, Dabeva MD, et al. Stem cell properties and repopulation of the rat liver by fetal liver epithelial progenitor cells. Am J Pathol. 2001;159:1323–1334. doi: 10.1016/S0002-9440(10)62519-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Oertel M, Menthena A, Dabeva MD, et al. Cell competition leads to high level of normal liver reconstitution by transplanted fetal liver stem/progenitor cells. Gastroenterology. 2006;130:507–520. doi: 10.1053/j.gastro.2005.10.049. [DOI] [PubMed] [Google Scholar]
- 20.Oertel M, Menthena A, Chen YQ, et al. Purification of fetal stem/progenitor cells containing all the repopulation potential for normal adult rat liver. Gastroenterology. 2008;134:823–832. doi: 10.1053/j.gastro.2008.01.007. [DOI] [PubMed] [Google Scholar]
- 21.Menthena A, Koehler C, Sandhu JS, et al. Activin A, p15INK4b signaling, and cell competition promote stem/progenitor cell repopulation of livers in aging rats. Gastroenterology. 2011;140:1009–1020. doi: 10.1053/j.gastro.2010.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Oertel M, Shafritz DA. Liver repopulation by fetal liver stem/progenitor cells. In: Soto-Gutierrez A, Navarro-Alvarez N, Fox IJ, editors. Methods in Bioengineering: Cell transplantation. Chapter 13. Artech House; 2011. pp. 201–217. [Google Scholar]
- 23.Gomes AT, Bastos CG, Afonso CL, et al. How variable are hydroxyproline determinations made in different samples of the same liver? Clin Biochem. 2006;39:1160–1163. doi: 10.1016/j.clinbiochem.2006.08.002. [DOI] [PubMed] [Google Scholar]
- 24.Kim MY, Cho MY, Baik SK, et al. Histological subclassification of cirrhosis using the Laennec fibrosis scoring system correlates with clinical stage and grade of portal hypertension. J Hepatol. 2011;55:1004–1009. doi: 10.1016/j.jhep.2011.02.012. [DOI] [PubMed] [Google Scholar]
- 25.Sell S. Distribution of alpha-fetoprotein- and albumin-containing cells in the livers of Fischer rats fed four cycles of N-2-fluorenylacetamide. Cancer Res. 1978;38:3107–3113. [PubMed] [Google Scholar]
- 26.Jensen CH, Jauho EI, Santoni-Rugiu E, et al. Transit-amplifying ductular (oval) cells and their hepatocytic progeny are characterized by a novel and distinctive expression of delta-like protein/preadipocyte factor 1/fetal antigen 1. Am J Pathol. 2004;164:1347–1359. doi: 10.1016/S0002-9440(10)63221-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yovchev MI, Grozdanov PN, Joseph B, et al. Novel hepatic progenitor cell surface markers in the adult rat liver. Hepatology. 2007;45:139–149. doi: 10.1002/hep.21448. [DOI] [PubMed] [Google Scholar]
- 28.Dorrell C, Erker L, Schug J, et al. Prospective isolation of a bipotential clonogenic liver progenitor cell in adult mice. Genes Dev. 2011;25:1193–1203. doi: 10.1101/gad.2029411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fox IJ, Roy-Chowdhury J. Hepatocyte transplantation. J Hepatol. 2004;40:878–886. doi: 10.1016/j.jhep.2004.04.009. [DOI] [PubMed] [Google Scholar]
- 30.Popov Y, Schuppan D. Targeting liver fibrosis: strategies for development and validation of antifibrotic therapies. Hepatology. 2009;50:1294–1306. doi: 10.1002/hep.23123. [DOI] [PubMed] [Google Scholar]
- 31.Thompson NL, Hixson DC, Callanan H, et al. A Fischer rat substrain deficient in dipeptidyl peptidase IV activity makes normal steady-state RNA levels and an altered protein. Use as a liver-cell transplantation model. Biochem J. 1991;237:497–502. doi: 10.1042/bj2730497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rajvanshi PA, Kerr A, Bhargava KK, et al. Studies of liver repopulation using the dipeptidyl peptidase IV deficient rat and other rodent recipients: cell size and structure relationships regulate capacity for increased transplanted hepatocytes mass in the liver lobule. Hepatology. 1996;23:482–496. doi: 10.1002/hep.510230313. [DOI] [PubMed] [Google Scholar]
- 33.Müller A, Machnik F, Zimmermann T, et al. Thioacetamide-induced cirrhosis-like liver lesions in rats--usefulness and reliability of this animal model. Exp Pathol. 1988;34:229–236. doi: 10.1016/s0232-1513(88)80155-5. [DOI] [PubMed] [Google Scholar]
- 34.Jeong DH, Jang JJ, Lee SJ, et al. Expression patterns of cell cycle-related proteins in a rat cirrhotic model induced by CCl4 or thioacetamide. J Gastroenterol. 2001;36:24–32. doi: 10.1007/s005350170150. [DOI] [PubMed] [Google Scholar]
- 35.Popov Y, Patsenker E, Bauer M, et al. Halofuginone induces matrix metalloproteinases in rat hepatic stellate cells via activation of p38 and NFkappaB. J Biol Chem. 2006;281:15090–15098. doi: 10.1074/jbc.M600030200. [DOI] [PubMed] [Google Scholar]
- 36.Safadi R, Ohta M, Alvarez CE, et al. Immune stimulation of hepatic fibrogenesis by CD8 cells and attenuation by transgenic interleukin-10 from hepatocytes. Gastroenterology. 2004;127:870–882. doi: 10.1053/j.gastro.2004.04.062. [DOI] [PubMed] [Google Scholar]
- 37.Niki T, De Bleser PJ, Xu G, et al. Comparison of glial fibrillary acidic protein and desmin staining in normal and CCl4-induced fibrotic rat livers. Hepatology. 1996;23:1538–1545. doi: 10.1002/hep.510230634. [DOI] [PubMed] [Google Scholar]
- 38.Liu L, Yannam GR, Nishikawa T, et al. The microenvironment in hepatocyte regeneration and function in rats with advanced cirrhosis. Hepatology. 2012;55:1529–1539. doi: 10.1002/hep.24815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rhim J, Sangren EP, Degen JL, et al. Replacement of diseased mouse liver by hepatic cell transplantation. Science. 1994;263:1149–1152. doi: 10.1126/science.8108734. [DOI] [PubMed] [Google Scholar]
- 40.Overturf K, Al-Dhalimy M, Tanguay R, et al. Hepatocytes corrected by gene therapy are selected in vivo in a murine model of hereditary tyrosinaemia type I. Nat Genet. 1996;12:266–273. doi: 10.1038/ng0396-266. [DOI] [PubMed] [Google Scholar]
- 41.Laconi E, Oren R, Mukhopadhay D, et al. Long-term, near total liver replacement by transplantation of isolated hepatocytes. Am J Pathol. 1998;153:319–329. doi: 10.1016/S0002-9440(10)65574-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Witek RP, Fisher SH, Petersen BE. Monocrotaline, an alternative to retrorsine-based hepatocyte transplantation in rodents. Cell Transplant. 2005;14:41–47. doi: 10.3727/000000005783983278. [DOI] [PubMed] [Google Scholar]
- 43.Guha C, Sharma A, Gupta S, Alfieri A, Gorla GR, Gagandeep S, et al. Amelioration of radiation-induced liver damage in partially hepatectomized rats by hepatocytes transplantation. Cancer Res. 1999;59:5871–5874. [PubMed] [Google Scholar]
- 44.Ding J, Yannam GR, Roy-Chowdhury N, et al. Spontaneous hepatic repopulation in transgenic mice expressing mutant human α1-antitrypsin by wild-type donor hepatocytes. J Clin Invest. 2011;121:1930–1934. doi: 10.1172/JCI45260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kobayashi N, Ito M, Nakamura J, et al. Hepatocyte transplantation in rats with decompensated cirrhosis. Hepatology. 2000;31:851–857. doi: 10.1053/he.2000.5636. [DOI] [PubMed] [Google Scholar]
- 46.Fisher RA, Strom SC. Human hepatocyte transplantation: worldwide results. Transplantation. 2006;82:441–449. doi: 10.1097/01.tp.0000231689.44266.ac. [DOI] [PubMed] [Google Scholar]
- 47.Fitzpatrick E, Mitry RR, Dhawan A. Human hepatocyte transplantation: state of the art. J Intern Med. 2009;266:339–357. doi: 10.1111/j.1365-2796.2009.02152.x. [DOI] [PubMed] [Google Scholar]
- 48.Khan AA, Shaik MV, Parveen N, et al. Human fetal liver-derived stem cell transplantation as supportive modality in the management of end-stage decompensated liver cirrhosis. Cell Transplant. 2010;19:409–418. doi: 10.3727/096368910X498241. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Repopulation and functional incorporation of differentiated donor cells transplanted without PH into rats with progressing fibrosis/cirrhosis. 1.4–1.6 × 108 ED15 fetal liver cells were transplanted into DPPIV− rats at 3 months after starting TAA administration, which was continued thereafter (200 mg/kg, twice weekly). (A,B) Patterns of DPPIV expression (A) and G6Pase activity (B) in two adjacent liver sections from one recipient rat at 4 months after cell transplantation. These liver sections demonstrate that DPPIV+ cells differentiated into hepatocytic cells, become incorporated into the fibrotic liver structure and exhibit G6Pase expression. (C) Quantitative RT-PCR analysis for mRNA levels of selected genes specific for hepatocytic functions in recipient livers at 4 months after FLSPC transplantation into rats with advanced fibrosis/cirrhosis. Values are mean ± SEM of liver samples from FLSPC-transplanted rats (n=3) and are expressed as fold differences in gene expression compared to liver samples derived from age-matched non-transplanted TAA-treated rats (n=3). One representative example from 3 replicate experiments is shown.
Liver tissue replacement without partial hepatectomy after cell infusion of enriched ED15 FLSPCs into a recipient rat with progressing fibrosis/cirrhosis. After isolation of unfractionated fetal liver cells, negative selection was performed by incubating ~3.5 × 108 cells with a monoclonal mouse antibody specific for rat erythrocytes and erythroid progenitors (BD Biosciences) at 4°C for 30 min. Cells were washed twice in Hanks’ balanced salt solution (HBSS) containing 0.3% BSA, 0.8 mM MgCl2, 10 mM HEPES and incubated with rat anti-mouse IgM bound to magnetic microbeads (Miltenyi) at 4°C for 30 min. The cell suspension was washed and passed through LS-columns placed in a MidiMACS™ Separator (Miltenyi). Erythroid cells were retained in the magnetic field (column); non-erythroid cells enriched with epithelial stem/progenitor cells passed through the columns. (A,B) Cytospins of unfractionated vs. cytospins of enriched ED15 FLSPCs were stained with mouse anti-E-cadherin antibody (BD Biosciences), followed by CyTm3-conjugated donkey anti-mouse IgG (Jackson) as secondary antibody. Unfractionated fetal liver cells contained 2.6% E-cadherin+ epithelial cells (A). After enrichment with immunomagnetic beads, this increased to 11.4% (4.4-fold enrichment) in the erythroid-depleted fraction (B). (C) Enriched fetal liver cells (~8 × 107) from DPPIV+ F344 rats were infused into a DPPIV− F344 rat with progressing fibrosis/cirrhosis without partial hepatectomy. At one month after cell infusion, the rat was sacrificed and liver sections were stained using enzyme histochemistry for DPPIV to detect expanding DPPIV+ cell clusters. The majority of the liver was successfully engrafted by transplanted fetal liver cells, which formed DPPIV+ cell clusters; some of them were already encompassing entire fibrotic lobules. The image contains 4 merged adjacent microscopic fields. Original magnification x400 (A,B) and x50 (C).