Abstract
In nearly all non-photosynthetic cells, pantothenate (vitamin B5) transport and utilization are prerequisites for the synthesis of the universal essential cofactor Coenzyme A (CoA). Early studies showed that human malaria parasites rely on the uptake of pantothenate across the parasite plasma membrane for survival within erythrocytes. Recently, a P. falciparum candidate pantothenate transporter (PAT) was characterized by functional complementation in yeast. These studies revealed that PfPAT mediated survival of yeast cells in low pantothenate concentrations and restored sensitivity of yeast cells lacking pantothenate uptake to fenpropimorph. In addition, PfPAT was refractory to deletion in P. falciparum in vitro, but nothing is known about the in vivo functions of PAT in Plasmodium life cycle stages. Herein, we used gene-targeting techniques to delete PAT in Plasmodium yoelii. Parasites lacking PAT displayed normal asexual and sexual blood stage development compared to wild-type (WT) and WT-like p230p(-) parasites. However, progression from the ookinete to the oocyst stage and sporozoite formation were completely abolished in pat(-) parasites. These studies provide the first evidence for an essential role of a candidate pantothenate transport in malaria transmission to Anopheles mosquitoes. This will set the stage for the development of PAT inhibitors against multiple parasite life cycle stages.
An estimated 3.3 billion people were at risk of malaria infection in 20101. There is as of yet no licensed malaria vaccine and malaria parasites are becoming increasingly resistant to anti-malarial drugs, including the arsenal of Artemisinin-Combination-Therapies2. Most anti-malarial drugs on the market are aimed at treating patient's symptoms and target asexual blood stage (BS) parasites, with no or limited activity against mature sexual stages3. Moreover, a frequent side-effect of antimalarial chemotherapies application is the dramatic increase in the number of gametocytes, which enhances the risk of future malaria transmission in endemic areas1,3. Therefore, the development of an anti-transmission drug component is important for the improvement of public health safety in areas that suffer high morbidity and mortality associated with malaria transmission.
Pantothenic acid (or vitamin B5, or when ionized as pantothenate) is a precursor of coenzyme A (CoA), which is an essential enzyme cofactor in all living organisms. CoA plays important roles in cellular metabolism and fatty acid biosynthesis4. Pantothenate acquisition from extracellular sources has been shown to be essential in nearly all non-photosynthetic cells for CoA biosynthesis. Early evidence for the essential role of pantothenate in the growth and survival of malaria parasites came from studies in chickens infected with P. gallinaceum and ducks infected with P. lophurae5,6. It was shown that birds fed on a diet lacking pantothenic acid or given pantothenic acid analogs orally showed little or no infection of the red blood cells compared to birds fed on pantothenic acid-rich diet5. Subsequently studies by Trager and colleagues following the establishment of the in vitro culture system for P. falciparum showed that pantothenate analogs block parasite proliferation in culture7. Similarly, Divo and colleagues showed that culture media lacking pantothenic acid do not allow growth of P. falciparum8,9. Supplementation of this vitamin B5-free medium with pantothenic acid alone was sufficient to restore normal parasite growth, suggesting that pantothenic acid is an essential vitamin transported by the parasite from host plasma and is absolutely required for parasite intraerythrocytic development. Since pantothenic acid requires a specialized transporter to move across cellular membranes, the transport of this essential precursor into P. falciparum-infected erythrocytes and its subsequent metabolism into CoA are therefore essential for this parasite's survival10. All CoA biosynthesis genes have been identified in the Plasmodium genomes except the pantothenate transporter4.
In a marked contrast with the findings in P. falciparum, no activity of CoA biosynthesis enzymes could be detected in Plasmodium lophurae isolated from duck red blood cells6,11,12. It has been suggested that in these cells pantothenate is taken up from host plasma via the endogenous vitamin transporter and transported across the red blood cell membrane into the host erythrocyte where it is converted by host enzymes into CoA7,13,14. This preformed CoA is subsequently transported into the intra erythrocytic parasite6,11,12. However, the mechanism by which CoA is transported across the parasite plasma membrane remains unknown.
In a recent study, a conserved putative transporter gene with multiple transmembrane domains was functionally characterized as a candidate pantothenate transporter (PAT) in Plasmodium falciparum15. PfPAT localized to the parasite plasma membrane of P. falciparum and its expression in a fen2Δ yeast mutant (lacking the only pantothenate transporter Fen2p) restored growth on low pantothenate concentrations and restored sensitivity to the antifungal drug, fenpropimorph, which is primarily transported into yeast cells via the Fen2 permease16. Moreover, genetic studies in P. falciparum indicated that PfPAT gene is not amenable to targeted deletion by classical genetic methods and down-regulation of its expression using morpholino-based oligonucleotides resulted in parasite death15. Together these data indicated that PfPAT might function as the primary pantothenate transporter of P. falciparum and its transport activity could be essential for BS development. Because of this phenotype the role of PAT in sexual stages development and parasite transmission could not be investigated.
In order to further characterize the role pantothenate transport plays during different life cycle stages of the malaria parasite, we have used the murine malaria parasite Plasmodium yoelii as a model system. Using reverse genetics approaches, we found that pantothenate transporter ortholog PyPAT is not essential for P. yoelii development in erythrocytes, thus making it possible to investigate the role pantothenate uptake and utilization play in sexual differentiation and parasite transmission to the mosquito. Our studies provide the first evidence that a candidate pantothenate transporter is required for oocyst development and sporozoite formation, and thus is essential for parasite transmission to the mosquito.
Results
Identification and Targeted Deletion of P. yoelii PAT
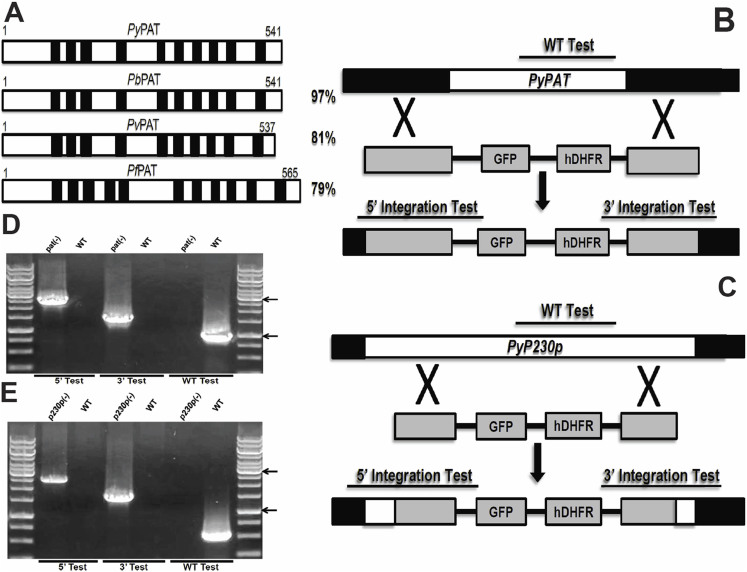
Using PfPAT protein sequence and chromosomal location we identified several orthologs in other human and murine parasite species (Fig. 1A) including a P. yoelii ortholog that we named PyPAT (PlasmoDB ID: PYYM_0304800), which shares 79% amino acid sequence identity with PfPAT (Figure 1A). To examine the importance of PyPAT during P. yoelii development in mouse erythrocytes, we used double genetic crossover technique to target PAT coding sequence for deletion in P. yoelii XNL (PyWT) non-lethal parasites (Figure 1B). The transfection plasmid that was used (AA20 plasmid) contains the enhanced GFP (eGFP) reporter cassette driven by the strong and constitutive PyHSP70_1 5′UTR promoter (PlasmoDB ID: PYYM_0712000) and the human DHFR resistance selectable marker cassette driven by PbEF1 alpha 5′ promoter (PlasmoDB ID: PBANKA_113330). As a control for the transfection procedures and the transfection plasmid, we applied the same knockout strategy to delete the P. yoelii P230p gene (Figure 1C), which has been previously shown to be dispensable for all malaria parasite life cycle stages in P. berghei and in P. yoelii17,18,19. Following the transfection, drug selection and cloning of transgenic parasites, genomic PCR analyses were performed to confirm the genetic replacement events and the deletion of PyPAT and PyP230p genes (Figure 1D and 1E).
Figure 1. Structure, conservation of PAT and Targeted deletion of PAT and P230p.
(A). Schematic representation of the putative PyPAT protein organization and alignment with PAT proteins from other Plasmodium species. The transmembrane domains are shown as black boxes. Total amino acid sequence identities to PyPAT are shown to the right. (B. and C.) Schematic representation of the replacement strategy to generate Pypat(-) parasites in (B) and Pyp230p(-) parasites in (C). The endogenous PyPAT and PyP230p genomic loci are targeted with replacement fragments containing the 5′ and 3′ PyPAT UTRs and PyP230p ORFs sequences, respectively, flanking the human DHFR positive selection marker and eGFP cassettes. Diagnostic WT-specific or integration-specific test amplicons are indicated by lines. (D. and E.) PCR genotyping confirms the integration of gene-replacement construct using oligonucleotide primer combinations that can only amplify from the recombinant loci (5′ Test and 3′Test). The WT-specific PCR reaction (WT) confirms the absence of WT parasites in Pypat(-) parasites in (D) and in Pyp230p(-) parasites in (E).The arrows showing the size of DNA ladder bands of 1000 and 3000 bps, respectively.
PAT deficient parasites undergo normal asexual replication in erythrocytes
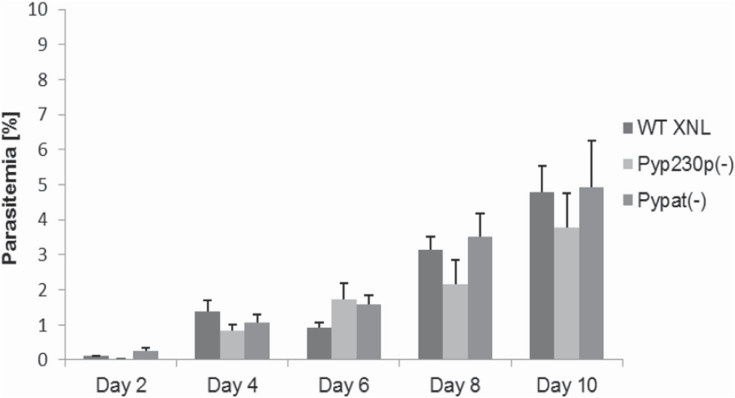
To determine if PyPAT gene disruption led to any deficiency in BS development in Pypat(-), we compared BS parasitemia between groups of Swiss Webster (SW) mice intravenously (IV) infected with 5000 BS Pypat(-), PyWT XNL or Pyp230p(-) parasites. Parasitemia was estimated from giemsa-stained thin blood smears until day 10 post infection (pi) and then monitored afterwards until clearance. No significant differences in BS parasitemia could be detected between all three strains on all days tested (Figure 2) and all days afterwards until clearance at days 17 or 18 pi. These findings demonstrate that PyPAT is not essential for P. yoelii development in mouse erythrocytes.
Figure 2. Targeted deletion of PAT does not affect Blood stage Growth.
Average BS parasitemia (evaluated as percentage of infected erythrocytes) in groups of SW mice after IV injection of 5000 BS per mouse with Pypat(-), Pyp230p(-) and WT parasites.
PAT does not function in sexual stages and ookinete development
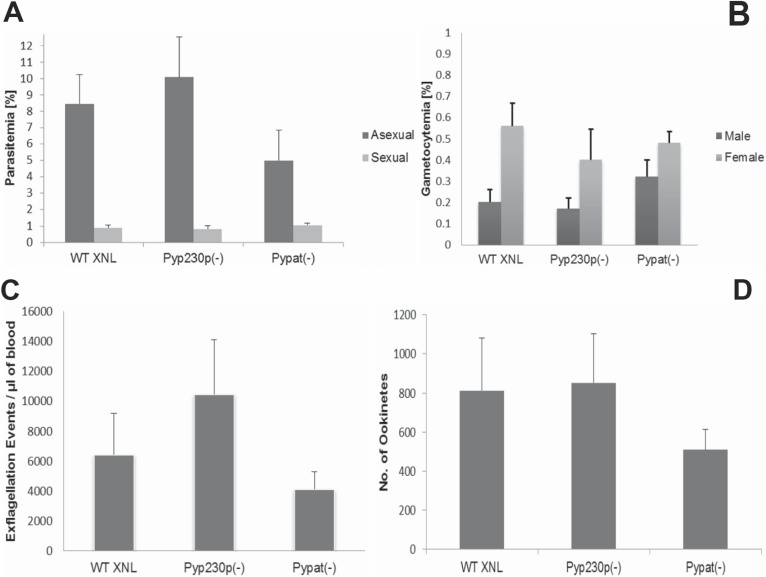
The viability of Pypat(-) parasites in mouse erythrocytes made it possible to assess the function of PyPAT in gametocyte differentiation, gametogenesis and ookinete production. Outbred SW mice were IV infected with 1 × 106 BS Pypat(-), PyWT XNL or Pyp230p(-) parasites. Blood samples were collected and smears were made simultaneously at day 3 pi to determine exflagellation rate, parasitemia of BS asexual and sexual stages and the number of male and female gametocytes for each parasite genotype infected mice groups. Most importantly in this experiment, all three parasite genotype clones used were Generation5 (G5) transfers (transferred in mice 5 times as BS from a G0 sporozoite induced BS infection that was used in the transfection culture) when injected into donor mice. We compared parasites with similar Generation number because several mechanical passages of BS parasites between mice without sporozoite transmission significantly decrease the mosquito transmission efficiency20,21. We used one-way analysis of variance (ANOVA) to determine significance in all experiments listed with the three genotype groups, with significance set at P value of <0.05. No significant differences in parasitemia percentage of asexual and sexual BS (Fig. 3A), average percentage of male vs. female gametocytes (Fig. 3B) could be detected between Pypat(-) parasites and the two control strains. P values were 0.06 and 0.69 for asexual and sexual parasitemia %, respectively, and for male and female gametocytes average % P values were 0.53 and 0.77, respectively. On the other hand, the average of male gamete exflagellation events was apparently slightly reduced in the Pypat(-) strain compared to WT and Pyp230p(-) parasite clones (Fig. 3C). However, we determined a P value of 0.06 indicating no significant difference between male gamete exflagellation in all three strains tested. Together these results suggest that PAT's function is not important as the parasite prepares for transition from intracellular survival inside host erythrocyte to extracellular survival inside the mosquito midgut as male and female gametes. To investigate the importance of this candidate transporter during the development of zygotes and ookinetes, female Anopheles stephensi mosquitoes were fed on infected mice displaying the highest male gamete exflagellation rate (to obtain the highest fertilization rate of female gametes, and therefore highest zygotes and ookinete numbers possible). After 20 hours post mosquito feeding (pmf), mosquito midguts from at least 20 blood-fed female Anopheles stephensi were dissected and mature ookinetes were quantified. The number of mature ookinetes detected in mosquitoes infected with Pypat(-) was also seemingly slightly reduced than that of ookinetes from mosquitoes infected with WT and p230p(-) parasites (Fig. 3D). However, we determined that there is no statistical significant difference between the mean ookinete numbers of all three genotype strains tested, with a P value of 0.41. Moreover, no differences in morphology (Figure 4A) was noted between mature ookinetes of Pypat(-) compared to WT and Pyp230p(-). The apparent slight insignificant reduction in the ratios of the male gamete exflagellation is similar to the apparent slight insignificant reduction in number of generated ookinetes in Pypat(-) compared to the other strains. These studies indicate that PAT does not have essential or important functions in male gamete development, fertilization, zygote development or mature ookinete formation.
Figure 3. PAT deficient parasites do not show significant reduction in the development of sexual stages and ookinetes.
(A. and B.) Graphs are showing the average BS parasitemia (evaluated as percentage of infected erythrocytes) for asexual and sexual stages in (A), and the average ratio of male and female mature gametocytes in (B). In each experiment, groups of SW mice were IV injected three days earlier with 1 million BS parasites per mouse from each parasite genotype. All parasite genotypes were G5 transfers at the time of donor mice infection. (C.) Graph is showing average number of male gamete exflagellation events per μl of mouse blood determined by a hemocytometer using 1:10 dilution of tail blood. (D.) Graph is showing the average number of ookinetes dissected out of mosquitos infected with each of the three genotypes 20 hours pmf. The median values for all three genotypes were analyzed with the one-way Analysis-of-Variance (ANOVA) and statistical significance was set at a P value of <0.05. The P values in all experiments shown here were >0.05.
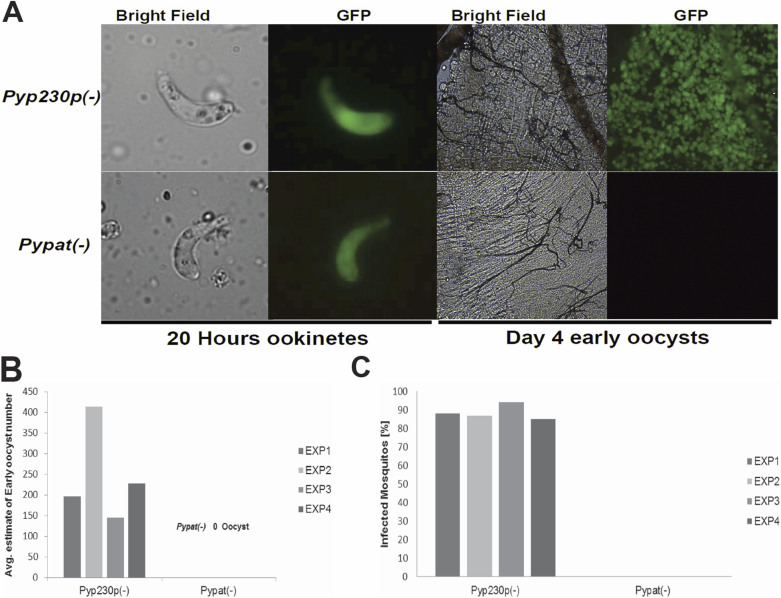
Figure 4. Ookinete to oocyst conversion is completely abolished in Pypat(-).
(A.) Images of live eGFP-expressing mature ookinetes (magnification 1000×) and early oocysts (magnification 100×) of Pypat(-) and Pyp230p(-) dissected out from infected mosquitoes midguts 20 hours and 4 days pmf, respectively. (B.) Percentage of mosquito midgut infection determined by the presence of early oocysts (day 4 pmf) for Pypat(-) and Pyp230p(-) infected mosquitoes in four different independent experiments using fluorescence microscopy. Midguts with at least one oocyst were counted as infected. (C.) Estimation of the number of early oocysts (day 4 pmf) from Pypat(-) and Pyp230p(-) infected mosquitoes midguts in four different independent experiments using fluorescence microscopy. Note that in (B) and (C) the value for Pypat(-) parasite is 0.
Pypat(-) parasites do not form oocysts
In the mosquito midgut, Plasmodium ookinetes traverse several midgut epithelial cells before reaching the basal lamina and transforming into a spherical oocyst 24–48 hours pmf22. The oocyst in turn undergoes multiple mitotic divisions that subsequently result in the formation of several hundred sporozoites from each oocyst. To investigate the importance of PyPAT in oocyst formation and sporozoite development, we took advantage of the fact that both Pypat(-) and Pyp230p(-) parasites were designed to constitutively express eGFP. Midguts of mosquitoes fed on Pypat(-) and Pyp230p(-) parasites were dissected out and the number of oocysts on their midguts were estimated using fluorescence microscopy at day 4 pmf. Results from four independent experiments revealed that 85 to 94% of dissected mosquitoes fed on Pyp230p(-) parasites were infected and contained between 145 and 414 oocysts (Figures 4B and 4C). Conversely, out of 106 dissected mosquitoes fed on Pypat(-) infected mice none contained any oocysts (Figure 4A, 4B and 4C). Consistent with the inability of parasites lacking PyPAT to produce oocysts, no oocyst sporozoites could be detected at day 10 pmf in mosquitoes fed on Pypat(-) parasites in 7 independent experiments. In contrast, the number of oocyst sporozoites in mosquitoes fed on mice infected with WT or Pyp230p(-) parasites ranged between 5,680 and 24,000 average sporozoites per mosquito (Table 1). Together these findings suggest an essential role for PAT in the ookinete to oocyst stage conversion and sporozoite formation and highlight the possible critical role of pantothenate uptake and CoA biosynthesis in Plasmodium development in mosquitoes and malaria transmission.
Table 1. Oocyst Sporozoites formation is completely abolished in Pypat(-).
| Pypat(-) | Pyp230p(-) | WT XNL | |
|---|---|---|---|
| Average number of Oocyst Sporozoites per Mosquito (Day 10 pmf) | |||
| Experiment 1 | 0 | 24,000 | (ND) |
| Experiment 2 | 0 | 5,680 | 19,451 |
| Experiment 3 | 0 | 10,000 | 18,150 |
| Experiment 4 | 0 | 15,680 | (ND) |
| Experiment 5 | 0 | 11,429 | 15,000 |
| Experiment 6 | 0 | 19,400 | 6,400 |
| Experiment 7 | 0 | 18,240 | 4,800 |
ND: Not Done.
Discussion
During their development in the vertebrate host or mosquito vector, Plasmodium parasites undergo rapid multiplication to produce large number of daughter parasites22,23. This rapid multiplication relies heavily on successful acquisition of essential nutrients such as purine nucleosides and nucleobases, amino acids, sugars and vitamins from the host23,24. One of these vitamins, pantothenic acid (Vitamin B5), is a precursor of the important enzyme cofactor CoA4. Because Plasmodium parasites cannot synthesize pantothenate de novo, the uptake and utilization of this precursor from the host is essential for survival4,13,14. Studies in P. falciparum and P. lophurae have indicated two different possible strategies used by malaria parasites in host erythrocytes to synthesize CoA. Whereas P. falciparum appears to use endogenous vitamin transporters to take up pantothenate from human plasma and a parasite-encoded transporter on the parasite plasma membrane to transport it from the erythrocyte cytoplasm into the parasite for subsequent utilization, P. lophurae uses pre-formed CoA in its nucleated erythrocyte cytoplasm4. The CoA transporter used by P. lophurae on its plasma membrane has not yet been identified.
Our studies in P. yoelii may imply that the murine malaria parasite might use a similar mechanism to P. lophurae for CoA acquisition during the intra-erythrocytic phase of its development but requires pantothenate uptake and utilization for de novo CoA biosynthesis for development in the mosquito. However, the fact that rodent erythrocytes are very similar to human erythrocytes and very different from the nucleated and metabolically active duck erythrocytes, could indicate that P. yoelii BS parasites are more similar in their nutritional needs to P. falciparum BS parasites than to P. lophurae. We found that knockout parasites lacking PyPAT undergo normal asexual replication and gametocyte differentiation during BS development in mice. Interestingly, PyPAT was found to play an essential function in oocyst progression and sporozoite formation but was not critical for gametogenesis, fertilization and ookinete development. Male gametocytes, in less than ten minutes, undergo up to three rounds of mitotic divisions and cytokinesis to produce up to eight highly motile gametes in the mosquito midgut22. This indicates that all the nutritional needs for mitotic division, cytokinesis and fertilization must be present in male gametocytes before transmission to the mosquito. Likewise, female gametocytes provide maternally the main cell structure, all cellular organelles and all nutritional needs required for female gametogenesis, fertilization and the conversion of the spherical zygote into pointed elongated invasive ookinete22. However, following maturation of ookinetes and traversal of midgut epithelial cells, ookinetes stop migrating and convert to oocysts, which undergo several mitotic divisions that result in the production of hundreds of sporozoites from each oocyst22. Because malaria parasites cannot synthesize the precursors of several essential macromolecules and co-factors, including CoA, their rapid development and active replication inside the mosquito vector is heavily dependent on the acquisition of these precursors from the host environment. Whereas major efforts have been made over the years to understand these processes during the parasite intraerythrocytic life cycle, little is known about parasite nutrient requirement, uptake and utilization in an extracellular environment in the mosquito vector. The inability of Pypat(-) knockout parasite to progress from ookinetes to oocysts or to form sporozoites suggest that this gene is required for these stages of parasite development. Apparently, active CoA biosynthesis from its primary precursor pantothenate at this specific stage of the life cycle, unlike BS parasites, seems essential for survival. However, we should not undermine the possibility of the presence of an alternative primary or a secondary pantothenate or pantetheine transporter that rescue the lack of PAT in BS, albeit this does not happen in mosquito stages.
Our study provides the first evidence for an essential role of a candidate pantothenate transporter in the arthropod phase of the life cycle of any protozoan parasite. The availability of Pypat(-) knockout parasites will make it possible to investigate the direct role of this molecule in the uptake of exogenous pantothenate. Due of the rapid and magnitude of multiplication of the parasite in hepatocytes (in which tens of thousands of merozoites develop from one liver stage), we predict that PAT function might also be essential for parasite pre-erythrocytic development.
Experimental Procedures
Experimental Animals, Parasites and Mosquitoes
Female Swiss Webster (SW) mice (6 to 8 weeks old) were purchased from Harlan Laboratories (Indianapolis, Indiana). Animal handling was conducted according to Institutional-Animal-Care-and-Use-Committee (IACUC) approved protocols. Wild-type (WT) P. yoelii XNL (non-lethal strain), Pyp230p(-) clone A5 and Pypat(-) clone E5 parasites were used to infect SW mice and Anopheles stephensi mosquitoes. We always used the same G-number (Generation number, which refers to the number of serial mouse blood transfers from sporozoite induced blood stage infection counted as G0) while comparing phenotypes of different clonal parasite populations. Mosquitoes were allowed to feed for at least 15 minutes on anesthetized infected mice on day 3 post intravenous injection (IV) of 1 million parasites in each parasite clone. Infected mosquitoes were maintained on 10% Sucrose solution +0.05% Para-Amino Benzoic Acid (PABA) wetted cotton pads at 24°C, 70% humidity and 16/8 hours light/dark cycles. Midguts were extracted from infected mosquitoes on days 1, 4 and 10 or 11 post mosquito infection (pmf) for counting ookinetes, early oocysts and oocyst sporozoites, respectively. Pyp230p(-) clone A5 and Pypat(-) clone E5 early oocyst detection was by counting green fluorescent oocysts via fluorescence microscopy. Sporozoites were freed from oocysts in RPMI medium by grinding the midguts with a pestle, sporozoites were counted using a hemocytometer.
Generation of transgenic parasites
Targeted deletions of PyPAT and PyP230p genes were done by double genetic crossover strategy driven by homologous recombination. P. yoelii XNL genomic DNA (gDNA) was used as a template to amplify fragments of the 5′UTR of PyPAT and of the 5′ end of PyP230p ORF (Open Reading Frame) using primers 31–32 and primers 41–42, respectively. The amplified fragments were inserted into the transfection plasmid AA20 between SacII and BamHI restriction enzyme sites. Fragments from the 3′UTR sequence of PyPAT and of the 3′ end of PyP230p ORF were amplified using primers 33–34 and primers 43–44, respectively, from P. yoelii XNL gDNA as a template, and inserted between the KpnI and HindIII restriction sites in the respective intermediate transfection plasmids. The fragments cloned for PyPAT targeted deletion were designed not to interfere with the coding sequences of any upstream or downstream neighboring genes and the makeup of those fragments was checked by sequencing to ensure that any unknown 5′ or 3′ promoter signals for the neighboring genes were not altered. The final plasmids were linearized with SacII and KpnI prior to the transfection. Transfection of P. yoelii XNL parasites with Lonza Nucleofector II device was performed on a parasite cultures that originated from donor rats that were infected with P. yoelii WT XNL sporozoites. The transfection procedures, resistant parasites selection and transgenic recombinant parasite cloning by serial dilutions were all conducted as described elsewhere17,25. To confirm the targeted deletion and the new genetic recombination, gDNAs were extracted from Pyp230p(-) clone A5 and Pypat(-) clone E5 and 5′ and 3′ integration-specific and WT specific PCR amplifications were conducted for 35 cycles (PCR cycle conditions: 94° for 30 seconds, 55° for 30 seconds and 60° for 3 minutes). 5′ Integration Tests were performed with primers 35–16 and primers 45–16, 3′ Integration Tests with primers 17–36 and primers 17–46 and WT coding sequence Tests with primers 37–38 and primers 47–48 for Pypat(-) and Pyp230p(-) gDNA, respectively.
Blood Stage Growth, Male gamete Exflagellation and Gametocytes Ratio Estimations
Donor mice were infected with the three genotypes with the same generation number by Intraperitoneal infection (IP) of frozen stocks. At day 2 pi, number of asexual BS per μl of donor mouse blood was determined by giemsa stained thin blood smears (to determine parasitemia %) and hemocytometer counts of donor mouse erythrocytes. Limited dilution with RPMI medium of donor mouse blood, when the parasitemia was between 0.1% and 1%, was used to aliquot the specific number of parasites to be injected into each mouse into separate micro-centrifuge tubes. For estimation of BS growth, recipient groups of 3 SW received IV injection of 5000 parasites from each genotype, and parasitemia % were counted daily from giemsa stained thin blood smears from day 2 until day 10 pi. At least 50 microscopic fields (magnification 1000×) were used for the parasitemia quantification on Nikon eclipse 50i microscope. Afterwards, giemsa stained thin blood smears were checked on days 12, 14, 16, 17 and 18 pi for clearance, which occurred at days 17 or 18 pi for all three genotypes. For the estimation of male gamete exflagellation and mosquito infections, recipient naïve mice were treated with phenylhydrazine (50 mg/kg in PBS) three days before the IV injection with 1 million BS parasites. Male gamete exflagellation was quantified three days after the IV infection by adding 5 μL blood from a tail puncture with a heparinized needle to complete ookinete medium in 1:10 dilution and counting the exflagellation events in a hemocytometer after 10 minutes incubation at room temperature (22–24°C). At the same time, a blood droplet was taken from the leftover blood at the site of tail puncture and was smeared and giemsa stained as a thin blood smear. From these smears BS sexual and asexual parasitemia % and average percentage of male and female gametocytes were estimated at the same recording time.
In vivo Ookinete and Early Oocysts Quantifications
The mice with the highest exflagellation rate from each genotype were fed to mosquitoes cages. 18–20 hours pmf, the midguts of at least 20 blood fed mosquitoes from each genotype were dissected out, ground with a pestle and diluted before loading onto a hemocytometer for counting of mature ookinetes. On day 4 pmf, mosquito midguts were extracted and placed on a microscope slide. We then counted the number of early oocysts and estimated the number of oocysts per infected mosquito.
Statistical Analysis
Statistical significant differences in all experiments was determined by the GraphPad InStat software between the median values for all three genotypes using one-way Analysis-of-Variance (ANOVA). P values of <0.05 were considered statistically significant.
Primers sequences (5′-3′)
31: GGCCGCGGTCATGTAATGTTATAGTGTCATGCAACTAAAAAT;
32: TCCGGATCCCGCTTACCTGTTTTGGGTATAGGAATCGGCATTCAT;
33: TGCCAAGCTTTCAAAATAGGATAATAATAGATACTTTTAGAAT;
34: TCCGGTACCTATATAGATATACATATTACTAAAAATGCAGTC;
35: ACCTTTTACTTACTATTTTTAATTAGCTATTTAA;
36: ATATCAAAAAAAAATATATAACCATATTTAACTA;
37: GGCCGCGGGATGAAATATTTTGGAACAATTCGAGGTTGGAG;
38: TCCGAATTCTGACATAACTTTATCTGCCATAGTCTCGTCATC;
41: GGCCGCGGATGATAAACCATATTTTGAAGAAATTATTAGTGA;
42: TCCGGATCCGCTTATTCTGGATTTTCAAATGAATAGTTAATAA;
43: GCCCAAGCTTCTTTAAGAAATTATGAACAATTCAAGCAATCCAA;
44: TCCGGTACCTTCCTAATATATATGATTTATAAATTAATTTGAT;
45: GAAGATTTTATCATTATTCGAGTCAGAGTACATCA;
46: ACACGAACCCAAAGAAATCGGCAAGCTAAAACTA;
47: ATTATATGGATGTGATTTTTCAGGTAATTCTAA;
48: CATACTCAAATATCTTTTTAGTAGTTCCTCAATA;
16: ATGTCCATTAACATCACCATCTAATTCAACAAG;
17: GTGTTCTTTCTGATGTTCAAGAAGAAAAAGGTA.
Author Contributions
A.A. and C.B.M. initiated the research, A.A. designed the experiments, A.A., R.H., L.L. and E.F. conducted the experiments, A.A. and R.H. analyzed the data and prepared the figures, A.A. and C.B.M. wrote the manuscript.
Acknowledgments
We like to thank Emily Molina and Fleur Porter for technical assistance to some of the experiments and Ashley Vaughn for critical reading of the manuscript. This work was supported by funds provided by Tulane University School of Public Health and Tropical Medicine.
References
- Alonso P. L. & Tanner M. Public health challenges and prospects for malaria control and elimination. Nat Med 19, 150–155, 10.1038/nm.3077 (2013). [DOI] [PubMed] [Google Scholar]
- Beshir K. B. et al. Residual Plasmodium falciparum parasitemia in Kenyan children after artemisinin-combination therapy is associated with increased transmission to mosquitoes and parasite recurrence. J Infect Dis 208, 2017–2024, 10.1093/infdis/jit431 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- mal E. R. A. C. G. o. D. A research agenda for malaria eradication: drugs. PLoS Med 8, e1000402, 10.1371/journal.pmed.1000402 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spry C., Kirk K. & Saliba K. J. Coenzyme A biosynthesis: an antimicrobial drug target. FEMS microbiology reviews 32, 56–106, 10.1111/j.1574-6976.2007.00093.x (2008). [DOI] [PubMed] [Google Scholar]
- Brackett S., Waletzky E. & Baker M. The relation between pantothenic acid and Plasmodium gallinaceum infections in the chicken and the antimalarial activity of analogues of pantothenic acid. J Parasitol 32, 453–462 (1946). [PubMed] [Google Scholar]
- Trager W. Further Studies on the Survival and Development in Vitro of a Malarial Parasite. The Journal of experimental medicine 77, 411–420 (1943). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trager W. Cofactors and vitamins in the metabolism of malarial parasites. Factors other than folates. Bulletin of the World Health Organization 55, 285–289 (1977). [PMC free article] [PubMed] [Google Scholar]
- Divo A. A., Geary T. G., Davis N. L. & Jensen J. B. Nutritional requirements of Plasmodium falciparum in culture. I. Exogenously supplied dialyzable components necessary for continuous growth. J Protozool 32, 59–64 (1985). [DOI] [PubMed] [Google Scholar]
- Saliba K. J., Ferru I. & Kirk K. Provitamin B5 (pantothenol) inhibits growth of the intraerythrocytic malaria parasite. Antimicrob Agents Chemother 49, 632–637 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saliba K. J. & Kirk K. H+-coupled pantothenate transport in the intracellular malaria parasite. J Biol Chem 276, 18115–18121, 10.1074/jbc.M010942200 (2001). [DOI] [PubMed] [Google Scholar]
- Trager W. Coenzyme A and the antimalarial action in vitro of antipantothenate against Plasmodium lophurae, P. coatneyi and P. falciparum. Trans N Y Acad Sci 28, 1094–1108 (1966). [DOI] [PubMed] [Google Scholar]
- Trager W. Studies on the extracellular cultivation of an intracellular parasite (avian malaria). II. The effects of malate and of coenzyme A concentrates. The Journal of experimental medicine 96, 465–476 (1952). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spry C. & Saliba K. J. The human malaria parasite Plasmodium falciparum is not dependent on host coenzyme A biosynthesis. J Biol Chem 284, 24904–24913, 10.1074/jbc.M109.025312 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spry C., van Schalkwyk D. A., Strauss E. & Saliba K. J. Pantothenate utilization by Plasmodium as a target for antimalarial chemotherapy. Infectious disorders drug targets 10, 200–216 (2010). [DOI] [PubMed] [Google Scholar]
- Augagneur Y. et al. Identification and functional analysis of the primary pantothenate transporter, PfPAT, of the human malaria parasite Plasmodium falciparum. J Biol Chem 288, 20558–20567, 10.1074/jbc.M113.482992 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stolz J., Caspari T., Carr A. M. & Sauer N. Cell division defects of Schizosaccharomyces pombe liz1- mutants are caused by defects in pantothenate uptake. Eukaryotic cell 3, 406–412 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janse C. J. et al. High efficiency transfection of Plasmodium berghei facilitates novel selection procedures. Mol Biochem Parasitol 145, 60–70, 10.1016/j.molbiopara.2005.09.007 (2006). [DOI] [PubMed] [Google Scholar]
- Lin J. W. et al. A novel ‘gene insertion/marker out' (GIMO) method for transgene expression and gene complementation in rodent malaria parasites. PLoS One 6, e29289, 10.1371/journal.pone.0029289 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Dijk M. R. et al. Three members of the 6-cys protein family of Plasmodium play a role in gamete fertility. PLoS pathogens 6, e1000853, 10.1371/journal.ppat.1000853 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha A. et al. A cascade of DNA-binding proteins for sexual commitment and development in Plasmodium. Nature 10.1038/nature12970 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janse C. J., Ponzi M., Sinden R. E. & Waters A. P. Chromosomes and sexual development of rodent malaria parasites. Memorias do Instituto Oswaldo Cruz 89 Suppl 2 43–46 (1994). [DOI] [PubMed] [Google Scholar]
- Aly A. S., Vaughan A. M. & Kappe S. H. Malaria parasite development in the mosquito and infection of the mammalian host. Annu Rev Microbiol 63, 195–221, 10.1146/annurev.micro.091208.073403 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saliba K. J. & Kirk K. Nutrient acquisition by intracellular apicomplexan parasites: staying in for dinner. Int J Parasitol 31, 1321–1330 (2001). [DOI] [PubMed] [Google Scholar]
- Kirk K. & Saliba K. J. Targeting nutrient uptake mechanisms in Plasmodium. Current drug targets 8, 75–88 (2007). [DOI] [PubMed] [Google Scholar]
- Philip N., Orr R. & Waters A. P. Transfection of rodent malaria parasites. Methods Mol Biol 923, 99–125, 10.1007/978-1-62703-026-7_7 (2013). [DOI] [PubMed] [Google Scholar]