Abstract
MicroRNAs have added a new dimension to our understanding of tumorigenesis and associated processes like epithelial-to-mesenchymal transition (EMT). Here, we show that miR-375 is elevated in epithelial-like breast cancer cells, and ectopic miR-375 expression suppresses EMT in mesenchymal-like breast cancer cells. We identified short stature homeobox 2 (SHOX2) as a miR-375 target, and miR-375–mediated suppression in EMT was reversed by forced SHOX2 expression. Ectopic SHOX2 expression can induce EMT in epithelial-like breast cancer cells, whereas SHOX2 knockdown diminishes EMT traits in mesenchymal-like breast cancer cells, demonstrating SHOX2 as an EMT inducer. We show that SHOX2 acts as a transcription factor to upregulate transforming growth factor β receptor I (TβR-I) expression, and TβR-I inhibitor LY364947 abolishes EMT elicited by ectopic SHOX2 expression, suggesting that transforming growth factor β signaling is essential for SHOX2-induced EMT. Manipulating SHOX2 abundance in breast cancer cells impact in vitro invasion and in vivo dissemination. Analysis of breast tumor microarray database revealed that high SHOX2 expression significantly correlates with poor patient survival. Our study supports a critical role of SHOX2 in breast tumorigenicity.
Introduction
The metastatic spread of epithelial cancer cells from the primary tumor to distant organs is enhanced with the gain of mesenchymal characteristics and the loss of epithelial features, a phenomenon known as the epithelial-to-mesenchymal transition (EMT) [1,2]. During EMT, epithelial cells lose their epithelial characteristics marked by the down-regulation of E-cadherin while acquiring a mesenchymal phenotype characterized by the up-regulation of mesenchymal proteins such as vimentin and N-cadherin (or cadherin 11) and mesenchymal-specific transcription factors including Snail, Slug, Twist, ZEB1, and ZEB2.
MicroRNAs (miRNAs) are 20- to 22-nucleotide noncoding RNAs that can posttranscriptionally silence the expression of target genes by base pairing mostly with their 3′-untranslated regions (3′-UTRs) [3]. Recent studies have demonstrated that miRNAs are involved in the processes of tumor progression and EMT-associated metastasis. For example, miR-205 and members of miR-200 family can suppress EMT by silencing the expression of ZEB1 and ZEB2 [4,5]. Using a panel of human breast cancer cell lines exhibiting both epithelial- and mesenchymal-like phenotypes, we revealed that miR-200c, miR-205, and miR-375 are the miRNAs most consistently upregulated in epithelial-like cells [6]. Despite the well-established role of miR-200c and miR-205 in EMT, whether miR-375 and its associated gene targets are involved in EMT process has not been answered. Nevertheless, a recent study showed that re-expressing miR-375 in tamoxifen-resistant breast cancer MCF7 cells induces epithelial-like properties resembling tamoxifen-nonresistant MCF7 cells [7], raising a possibility that miR-375 may play a role in EMT.
Short stature homeobox 2 (SHOX2) is a homolog to the short stature homeobox gene SHOX in humans. SHOX2 is the only SHOX gene present in mice, and ablation of SHOX2 causes embryonic lethality at midgestation due to cardiac and vascular defects [8]. Studies of SHOX2 conditional knockout mice further show that SHOX2 plays an indispensable role in the formation of the proximal portion of the limb skeleton and synovial joints [9,10]. Several recent studies reported that hypermethylation of the SHOX2 DNA locus could be a candidate biomarker for lung cancer [11]. These findings underscore the relevance of SHOX2 in tumorigenesis. A potential role of SHOX2 in tumorigenesis is also supported by the observations that its expression is associated with tumor recurrence in hepatocellular carcinoma (HCC) [12]. We recently showed that the expression of SHOX2 has an inverse correlation with miR-375 in breast cancer cell lines and is higher in mesenchymal-like breast cancer cells whereas lower in epithelial-like ones [6]. However, it is absolutely unknown whether SHOX2 plays a role in EMT or any other specific role in tumorigenic process.
The objective of this study is to determine the relationship of miR-375 and SHOX2 during EMT in breast cancer cells. With the aid of multiple breast cancer cell lines, we reconfirmed the inverse relationship between miR-375 and SHOX2 and showed that miR-375 silenced SHOX2 expression by directly targeting the 3′-UTR of SHOX2 mRNA. To determine the role of miR-375 in EMT, we found that enforced miR-375 expression induced the expression of E-cadherin while diminishing the expression of vimentin and blocking in vitro invasion of mesenchymal-like breast cancer cells. However, miR-375–mediated events were completely reverted by ectopic SHOX2 expression, suggesting that miR-375 is involved in EMT by regulating SHOX2 expression. In fact, knockdown of SHOX2 caused mesenchymal-like breast cancer cells to display an epithelial-like phenotype, whereas ectopic expression of SHOX2 in epithelial-like breast cancer cells led to EMT induction. These results consequently demonstrate SHOX2 as an EMT inducer in breast cancer cells. In an attempt to elucidate the underlying mechanism of SHOX2-induced EMT, we showed that this observed SHOX2-mediated event was dependent on transforming growth factor β (TGF β) signaling on the basis of the fact that TGFβ receptor I (TβR-I) inhibitor LY364947 reverted mesenchymal-like phenotype of SHOX2-overexpressing MCF7 and T47D cells back to their original epithelial-like phenotype. SHOX2 directly impacts TGFβ signaling by acting as a TβR-I–specific transcription factor. By analyzing publicly available microarray databases, we revealed that SHOX2 expression correlated with both poor overall and recurrence-free survival of patients with breast cancer. Moreover, we showed that depletion of SHOX2 blocked the ability of mesenchymal-like breast cancer cells to invade in vitro and to disseminate in vivo, whereas ectopic SHOX2 expression converted epithelial-like breast cancer cells to be invasive in vitro and disseminative in vivo. This study concludes that miR-375/SHOX2 relationship is a potent EMT regulator and plays a critical role in breast tumorigenicity.
Materials and Methods
Antibodies and Other Reagents
Detailed information on antibodies used for Western blot analysis, immunofluorescence staining, and chromatin immunoprecipitation (ChIP) were described in Supplementary Materials section. SHOX2 short hairpin RNA lentiviral constructs were purchased from Thermo Fisher Scientific (RHS4430-101105065, RHS4430-101105700, and RMM4431-99202942; Waltham, MA). LY364947 was obtained from the LC Laboratories (Woburn, MA).
Cell Migration and In Vitro Invasion
Cell migration and in vitro invasion were analyzed using Transwells (Corning Life Sciences, Tewksbury, MA) and Matrigel invasion chamber (Cell Biolabs, San Diego, CA), respectively, as previously described [13]. For cell migration, the undersurface of the upper chamber of the Transwell was coated with 10 μg/ml collagen I overnight at 4°C. Cells (1 × 105 cells per Transwell) were added into the upper chamber and allowed 4 hours for cell migration. For in vitro invasion, 2 × 105 cells were added into each upper chamber and allowed 24 hours for invasion. After the period of migration or invasion, cells on the undersurface of the upper units were stained and counted under a phase-contrast microscope.
Quantitative Reverse Transcription–Polymerase Chain Reaction
Total RNA was extracted from cells using TRIzol (Life Technologies, Carlsbad, CA) and used to measure the amount of SHOX2, E-cadherin, vimentin, TβR-I, TβR-II, TβR-III, and β-actin mRNA. The amount of mature miR-375 was analyzed using miR-375 TaqMan MicroRNA Assay kit (Life Technologies). The amount of β-actin mRNA was used as the internal standardization.
Luciferase Reporter Gene Constructs and Luciferase Assay
SHOX2 3′-UTR luciferase reporter gene plasmid was constructed by inserting the entire human SHOX2 3′-UTR sequence into the pMiR vector (Life Technologies). TβR-I promoter luciferase reporter gene plasmid was constructed by inserting 1504 bp of human TβR-I promoter sequence into the pGL2 vector (Promega, Madison, WI). To generate miR-375 targeting SHOX2 3′-UTR mutants and TβR-I promoter mutated in Homeobox (HOX)-binding sites, site-directed mutagenesis was performed on the respective wild-type plasmids using QuikChange Site-Directed Mutagenesis Kit (Agilent Technologies, Santa Clara, CA). To determine luciferase activity, cells were transfected with luciferase reporter gene plasmids for 2 days and then lysed for measurement of luciferase activity. To normalize transfection efficiency, pGK-RLuc plasmid was included in all transfection experiments and Renilla luciferase activity was used for standardization. Luciferase activities were determined using Dual-Luciferase Reporter Assay System (Promega).
Chromatin Immunoprecipitation
ChIP was performed as previously described [14,15]. Because there is no SHOX2 antibody suitable for ChIP, MDA-MB-231 cells were transfected with FLAG-tagged SHOX2 vector and subsequently subjected to ChIP using anti-FLAG monoclonal antibody (mAb).
Cell Proliferation Assay
Cell proliferation was analyzed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay as previously described [16]. Cells were seeded in 24-well plates and cultured for 1 to 3 days following with addition of MTT solution to the cells for 2 hours. After removing the medium, the remaining MTT formazan crystals were solubilized in DMSO and measured with a microplate reader at 560 nm.
Immunofluorescence Staining
Cells were cultured on coverslips overnight and fixed with 3% paraformaldehyde, followed by the treatment of 1% Triton X-100 (Thermo Fisher, Waltham, MA) for permeabilization. To visualize the E-cadherin and vimentin, we incubated the coverslips with the respective antibodies for 1 hour and then rhodamine-conjugated secondary antibody for another hour. The fluorescence staining was observed with the aid of a fluorescence microscope (Axiovert 200M; Carl Zeiss, Thornwood, NY). 4',6-diamidino-2-phenylindole (DAPI) was included during staining to visualize nuclei of cells.
Zebrafish Tumor Cell Dissemination Model
The dissemination capability of cells was evaluated by a recently established zebrafish metastasis model [17] and performed as previously described [18]. All experimental procedures were approved by the Experimental Animal Ethical Committee of Georgia Regents University. Briefly, cells were first labeled with fluorescent dye CM-Dil (Life Technologies), and approximately 200 labeled cells were microinjected into the perivitelline space of 48-hour postfertilization zebrafish embryos using a pressure microinjector. After confirmation of a visible cell mass at the injection site, the embryos were transferred to a 34°C incubator for 30 hours. To define the precise localization of metastatic cells within the zebrafish, living zebrafish embryos were imaged under anesthetic by confocal microscopy. Z-stack images were processed using ImageJ as previously described [19].
Statistical Analysis
Statistical analyses of in vitro invasion assays and luciferase activities were performed by the Student’s t test using Microsoft Excel software (Redmond, WA). For SHOX2 analysis with human breast tumor microarray data sets (Gene Expression Omnibus, GSE18229 and GSE7390, National Center for Biotechnology Information, Bethesda, MD), the cutoff value of high- and low-SHOX2 expression groups was based on the sample median value. Association between SHOX2 expression and clinical features was analyzed by the Pearson χ2 tests. The correlation between SHOX2 and TβR-I expression was determined by Pearson correlation coefficient. In univariate survival analyses, the Kaplan-Meier method and the log-rank test were used to compare recurrence-free (data sets GSE7390 and GSE18229) and overall survival curves (data set GSE18229) between high- and low-SHOX2 expression groups. In multivariate survival analysis, the Cox proportional hazards regression model was used to identify important factors on overall survival for breast cancer (data set GSE18229). This was performed by starting with an initial model in which only SHOX2 expression was considered and then adding a clinical feature one at a time to the model. By repeating this procedure, the final regression model with important factors was identified by a likelihood ratio test. Both positive and negative association between overall survival and each factor were tested in the Cox proportional hazards regression models (i.e., one-sided test). P < .05 was considered statistically significant.
Results
MiR-375 Inhibits SHOX2 Expression in Breast Cancer Cells
By analyzing the miRNA expression profile of a panel of established human breast cancer cell lines on microarray, we were previously able to identify miR-375 along with miR-200c and miR-205 as the miRNAs whose expression is consistently elevated in epithelial-like breast cancer cell lines. In the same study, microarray analysis also revealed that SHOX2, which contains the putative miR-375 targeting sites in its 3′-UTR of mRNA, is preferentially expressed in mesenchymal-like breast cancer cell lines. To confirm this inverse correlation between miR-375 and SHOX2 in breast cancer cells, we performed quantitative reverse transcription–polymerase chain reaction (qRT-PCR) to analyze their expression in nine breast cancer cell lines consisting of both epithelial-like and mesenchymal-like phenotypes. The amount of miR-375 was much lower in mesenchymal-like lines (characterized by vimentin expression) than epithelial-like lines (characterized by E-cadherin expression) (Figure 1, A and C). In contrast, SHOX2 mRNA and protein were highly expressed in mesenchymal-like lines but were either low or undetectable in epithelial-like lines (Figure 1, B and C). To determine the potential regulatory role of miR-375 on SHOX2, we lentivirally introduced miR-375 into MDA-MB-231 and Hs-578T cells. Western blot analysis of the transduced cells showed that ectopic miR-375 expression led to dramatic reduction in SHOX2 expression (Figure 1D). The effect of miR-375 appeared to be specific because its inhibitory outcome on SHOX2 expression was abolished by treating cells with miR-375 inhibitor (Figure 1D).
Figure 1.
SHOX2 is a miR-375 target. (A) qRT-PCR analysis of SHOX2 expression in breast cancer cell lines. Data are means ± SEM (n = 3). (B) qRT-PCR analysis of miR-375 expression in breast cancer cell lines. Data are means ± SEM (n = 3). (C) Western blot analyses of SHOX2, E-cadherin, and vimentin expression in breast cancer cell lines. β-Actin was used as control. (D) Western blot analysis was used to analyze the effect of miR-375 on SHOX2 expression. miR-375 was lentivirally introduced into MDA-MB-231 and Hs578T cells, followed by the treatment with or without anti–miR-375. β-Actin was used as control. (E) Representation of miR-375 binding sites in the 3′-UTR of SHOX2 mRNA and the mutations generated in this region. Alphabetical numbers are the relative nucleotide positions in the 3′-UTR of SHOX2 mRNA. (F) pMiR containing SHOX2 3′-UTR with or without mutation in miR-375 targeting site was cotransfected into miR-375–transduced MDA-MB-231 and Hs578T cells for 2 days, followed by the analysis of luciferase activity. Data are means ± SEM (n = 3). #P < .01. *P < .05 versus control.
The presence of putative miR-375 targeting sites in the 3′-UTR of SHOX2 mRNA prompted us to determine whether SHOX2 mRNA is a direct target of miR-375. We inserted SHOX2′s 3′-UTR sequence into the luciferase reporter plasmid pMiR and transfected this plasmid into miR-375–expressing MDA-MB-231 and Hs578T cells. The luciferase activity was more than 70% lower in miR-375–expressing cells when compared to the control (empty pMiR plasmid) (Figure 1, E and F), suggesting SHOX2 mRNA as a miR-375 target. To ascertain whether miR-375 targets SHOX2 mRNA through its predicted pairing sites (on the basis of TargetScan program, Whitehead Institute, Cambridge, MA) [20,21], we performed G/C→C/G and A/U→U/A mutations in one or both of these sites to disrupt the potential miR-375/SHOX2 mRNA interaction (Figure 1E). Transfection experiments with these constructs demonstrated that mutation in either site abolished the ability of miR-375 to reduce luciferase activity (Figure 1F). These results show that both of the putative miR-375 target sites in the 3′-UTR of SHOX2 mRNA are critical for miR-375–mediated reduction in SHOX2 expression in breast cancer cells.
miR-375/SHOX2 Interaction Regulates EMT in Breast Cancer Cells
The low abundance of miR-375 in mesenchymal-like breast cancer cells indicates that miR-375 could be a potential negative EMT regulator (Figure 1A). To test this possibility, we examined several EMT markers in MDA-MB-231 and Hs578T cells with ectopic miR-375 expression. Western blot analysis showed that miR-375 diminished vimentin expression while inducing E-cadherin expression (Figure 2A). Matrigel invasion assay further showed that miR-375 inhibited more than 70% of the in vitro invasion in both lines (Figure 2B). The effect of miR-375 was clearly specific because miR-375 inhibitor (anti–miR-375) abolished miR-375–induced events (Figure 2B). To determine whether SHOX2 expression is functionally linked to miR-375, we enforced SHOX2 expression in MDA-MB-231 and Hs578T cells with ectopic miR-375 expression. Forced SHOX2 expression restored vimentin expression and cell migration and suppressed E-cadherin expression in these miR-375–expressing cells (Figure 2, A and B). These results suggest that miR-375 suppresses EMT by blocking SHOX2 expression in breast cancer cells.
Figure 2.
miR-375 suppresses EMT by diminishing SHOX2 expression. (A) Western blot analysis was used to determine the effect of ectopic SHOX2 expression on E-cadherin and vimentin expression. β-Actin was used as control. (B) Matrigel invasion assay was used to analyze the effect of ectopic SHOX2 expression on in vitro invasion of miR-375–trasduced MDA-MB-231 and Hs578T cells. Data are means ± SEM (n = 3). #P < .05. *P < .05 versus miR-control. (C) qRT-PCR was used to determine the effect of SHOX2 knockdown on E-cadherin and vimentin expression. Data are means ± SEM (n = 3). *P < .05 versus shControl. (D) Western blot analysis was used to determine the effect of SHOX2 knockdown on E-cadherin and vimentin expression. β-Actin was used as control. (E) Morphologies of control and SHOX2 knockdown MDA-MB-231 and Hs578T cells. (F) Transwell migration assay was used to analyze the effect of SHOX2 knockdown on cell motility. Data are means ± SEM (n = 3). *P < .05 versus shControl.
Next, we investigated the importance of SHOX2 in EMT by lentivirally introducing SHOX2 shRNA into mesenchymal-like MDA-MB-231 and Hs578T cells. Knockdown of SHOX2 decreased the amount of both vimentin mRNA and protein while increasing the level of E-cadherin mRNA and protein (Figure 2, C and D). SHOX2 knockdown cells also displayed an epithelial morphology (Figure 2E) and impaired cell migratory capability (Figure 2F). In a parallel study, we lentivirally introduced SHOX2 into epithelial-like MCF7 and T47D cells. Ectopically expressing SHOX2 diminished E-cadherin expression and induced vimentin expression in both lines (Figure 3, A and B, and Figure W1). When these cells were subjected to microscopic observation, MCF7 and T47D cells with ectopic SHOX2 expression displayed a mesenchymal morphology, whereas control cells exhibited a typical epithelial cell phenotype (Figure 3C). Moreover, ectopic SHOX2 expression also enhanced the migratory capability of both MCF7 and T47D cells (Figure 3D) and triggered significant expression of mesenchymal-specific Slug, Twist, ZEB1, and ZEB2 (Figure W2). These results suggest that SHOX2 is a potent inducer of EMT in breast cancer cells.
Figure 3.
SHOX2 induces EMT traits in epithelial-like breast cancer cells. (A) qRT-PCR was used to determine the effect of ectopic SHOX2 expression on E-cadherin and vimentin expression. Data are means ± SEM (n = 3). #P < .01 versus Control. *P < .05 versus Control. (B) Western blot analysis was used to determine the effect of ectopic SHOX2 expression on E-cadherin and vimentin expression. β-Actin was used as control. (C) Morphologies of control and SHOX2-transduced MCF7 and T47D cells. (D) Transwell migration assay was used to analyze the effect of ectopic SHOX2 expression on cell motility. Data are means ± SEM (n = 3). *P < .05 versus Control.
SHOX2 Regulates TβR-I Expression in Breast Cancer Cells
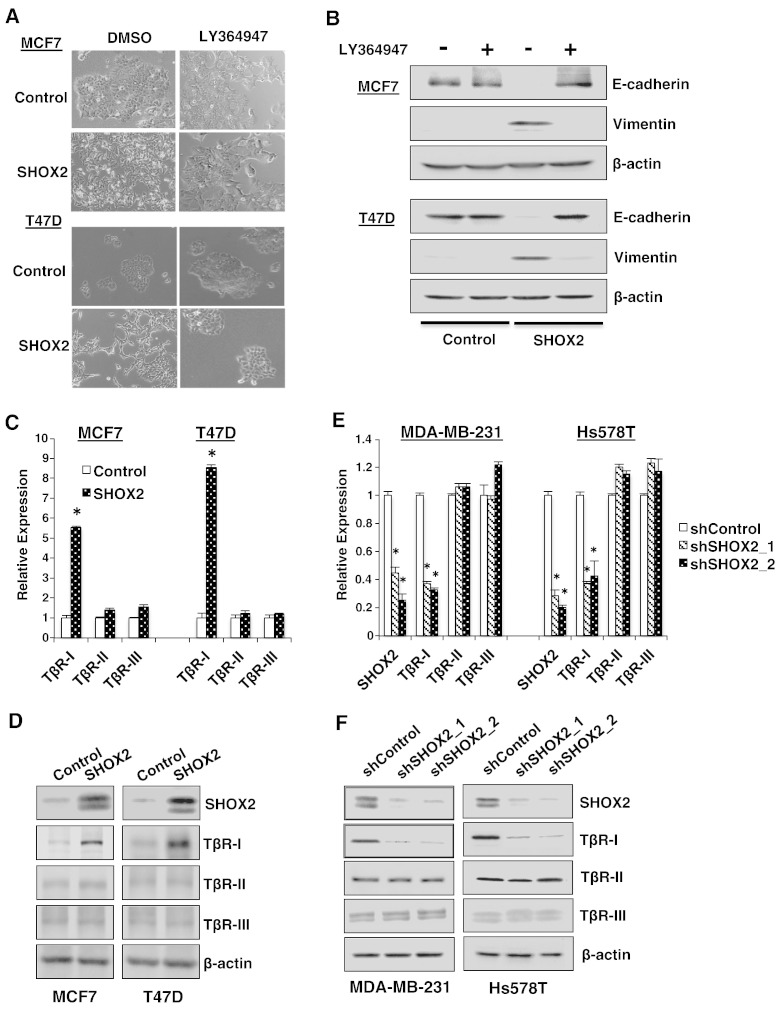
TGFβ signaling network plays an essential role in EMT of breast cancer cells. To determine whether SHOX2 regulation of EMT is functionally associated with the TGFβ signaling pathway, we overexpressed SHOX2 in MCF7 and T47D cells and then treated them with TβR-I inhibitor LY364947 or DMSO as vehicle. The morphology of both control lines treated with or without LY364947 remained unchanged (Figure 4A). In contrast, the mesenchymal-like morphology of SHOX2-overexpressing MCF7 and T47D cells was reverted back to epithelial-like on treatment with LY364947 (Figure 4A). Western blot analysis further showed that LY364947 treatment restored E-cadherin expression and abolished vimentin expression in SHOX2-overexpressing MCF7 and T47D cells (Figure 4B). qRT-PCR also revealed identical results at the mRNA level (Figure W3). These data suggest that SHOX2-induced EMT depends on the TGFβ signaling network.
Figure 4.
SHOX2 induces EMT through TGFβ signaling network. (A) Effect of LY364947 on morphologies of control and SHOX2-transduced MCF7 and T47D cells. (B) Western blot analysis was used to determine the effect of LY364947 on E-cadherin and vimentin expression in control and SHOX2-transduced MCF7 and T47D cells. β-Actin was used as control. (C) qRT-PCR was used to determine the effect of ectopic SHOX2 expression on TβR-I, TβR-II, and TβR-III expression in MCF7 and T47D cells. Data are means ± SEM (n = 3). *, P < .05 versus Control. (D) Western blot analysis was used to determine the effect of ectopic SHOX2 expression on TβR-I, TβR-II, and TβR-III expression in MCF7 and T47D cells. (E) qRT-PCR was used to determine the effect of SHOX2 knockdown on TβR-I, TβR-II, and TβR-III expression in MDA-MB-231 and Hs578T cells. Data are means ± SEM (n = 3). *P < .05 versus shControl. (F) Western blot analysis was used to determine the effect of SHOX2 knockdown on TβR-I, TβR-II, and TβR-III expression in MDA-MB-231 and Hs578T cells.
To identify the TGFβ signaling network component that is important for SHOX2-mediated EMT, we analyzed the mRNA levels of TGFβ1, TGFβ2, TGFβ3, TβR-I, TβR-II, and TβR-III in MCF7 and T47D cells with ectopic SHOX2 expression. qRT-PCR showed similar levels of TGFβ1, TGFβ2, and TGFβ3 mRNA between control cells and cells with ectopic SHOX2 expression (Figure W4). Among the TβRs, the amount of TβR-I increased more than five-fold in MCF7 and eight-fold in T47D cells on ectopic SHOX2 expression, though little change was detected in TβR-II or TβR-III (Figure 4, C and D). In a subsequent experiment, we examined the abundance of TβRs in control and SHOX2 knockdown MDA-MB-231 and Hs578T cells. Knockdown of SHOX2 diminished the expression of TβR-I but not that of TβR-II and TβR-III mRNA and protein (Figure 4, E and F). Pearson correlation coefficient analysis with publicly available microarray data sets of human breast cancer tissues also indicated a strong positive correlation between SHOX2 and TβR-I expression (ρ = 0.374; P < .001). These results clearly show that SHOX2 regulates TβR-I expression in breast cancer cells.
SHOX2 Activates TβR-I Promoter through the Consensus HOX-Binding Site in TβR-I Promoter
To understand how modulating the abundance of SHOX2 alters the level of TβR-I mRNA in breast cancer cells (Figure 4, C and E), we determined the effect of SHOX2 knockdown on TβR-I promoter activity in mesenchymal-like MDA-MB-231 and Hs578T cells. Luciferase activity assay showed that TβR-I promoter activity was at least 75% lower when compared to their respective control cells (Figure 5A). In parallel, we also measured TβR-I promoter activity in epithelial-like MCF7 and T47D cells. Whereas TβR-I promoter activity was inherently low, ectopic expression of SHOX2 induced a more than four-fold increase in TβR-I activity (Figure 5B). These results show that SHOX2 promotes TβR-I transcription in breast cancer cells.
Figure 5.
SHOX2 transcriptionally regulates TβR-I expression. (A) Effect of SHOX2 knockdown on TβR-I promoter activity in MDA-MB-231 and Hs578T cells. Data are means ± SEM (n = 3). #P < .01 versus shControl. (B) Effect of ectopic SHOX2 expression on TβR-I promoter activity in MCF7 and T47D cells. Data are means ± SEM (n = 3). #P < .01 versus Control. (C) Effect of mutation in putative SHOX2-binding site on TβR-I promoter activity in MDA-MB-231 and Hs578T cells or SHOX2-induced TβR-I promoter activity in MCF7 and T47D cells. Data are means ± SEM (n = 3). #P < .01 versus wild-type (WT) or SHOX2 (−). (D) FLAG-SHOX2-transduced MDA-MB-231 cells were subjected to ChIP with FLAG mAb or mouse IgG. Quantitative PCR (qPCR) was performed using a primer set that amplifies various regions in TβR-I promoter. Data are means ± SEM (n = 3). *P < .05 versus Control (mouse IgG).
Because SHOX2 contains the DNA-binding HOX domain, we hypothesized that SHOX2 regulated TβR-I expression by acting as a TβR-I–specific transcription factor in breast cancer cells. To test this hypothesis, we analyzed the TβR-I promoter for HOX-binding consensus sequence ATTA(N)nTAAT and identified two such sites located at nucleotides − 1238 to − 1197 and − 943 to − 906 relative to TβR-I transcription start site. We performed site-directed mutagenesis at nucleotides − 1238 to − 1237 and − 943 to − 942 in the TβR-I promoter by switching ATTA to GCTA. Luciferase activity assay showed that mutation at nucleotides − 1238 to − 1237, but not − 943 to − 942, abolished more than 70% of TβR-I promoter activity in both MDA-MB-231 and Hs578T cells (Figure 5C). The mutagenesis at nucleotides − 1238 to − 1237 also incapacitated SHOX2 to activate TβR-I promoter in MCF7 and T47D cells (Figure 5D), suggesting that nucleotides − 1238 to − 1237 are the sites for SHOX2 binding. In a parallel experiment, we performed ChIP experiment in MDA-MB-231 cells transiently transfected with FLAG-tagged SHOX2 using FLAG mAb or mouse IgG. PCR with primers amplifying various regions of the TβR-I promoter showed that there was an almost 12- and 18-fold greater amplification at the regions spanning nucleotides − 1341 to − 1247 and − 1223 to − 1110, respectively, (Figure 5E). Because HOX consensus sequence, located at nucleotides − 1238 to − 1197, is within these two regions, these results further support our finding that SHOX2 interacts with TβR-I promoter through the HOX-binding site at nucleotides − 1238 to − 1237.
SHOX2 Expression Correlates with Poor Survival of Patients with Breast Cancer and Is Required for Breast Tumorigenicity
The ability of SHOX2 to regulate the EMT process indicates that SHOX2 potentially plays a critical role in breast tumor progression. To substantiate this claim, we first assessed the correlation between SHOX2 expression and clinical features of patients with breast cancer by analyzing two breast-tumor gene expression microarray data sets available from Gene Expression Omnibus. In these data sets, high SHOX2 expression correlated with positive lymph node status and estrogen receptor (ER) negativity (P = .001 and .010, respectively; Table 1). Because ER-negative breast tumors generally show less differentiated (mesenchymal-like) phenotype, high expression of SHOX2 in the ER-negative subtype of breast tumors is consistent with its role as a facilitator of the EMT process.
Table 1.
| Clinical Features | SHOX2 Expression⁎ |
P Value⁎⁎ | |
|---|---|---|---|
| Low (%) | High (%) | ||
| Histologic grade | |||
| 1 | 31 | 24 | .339 |
| 2-3 | 186 | 198 | |
| Tumor diameter | |||
| < 2 cm | 87 | 75 | .176 |
| ≥ 2 cm | 137 | 157 | |
| Lymph node status | |||
| Negative | 177 | 152 | .001 |
| Positive | 47 | 84 | |
| ER receptor | |||
| Negative | 71 | 103 | .010 |
| Positive | 150 | 129 | |
Low/high by the sample median.
Pearson χ2 test.
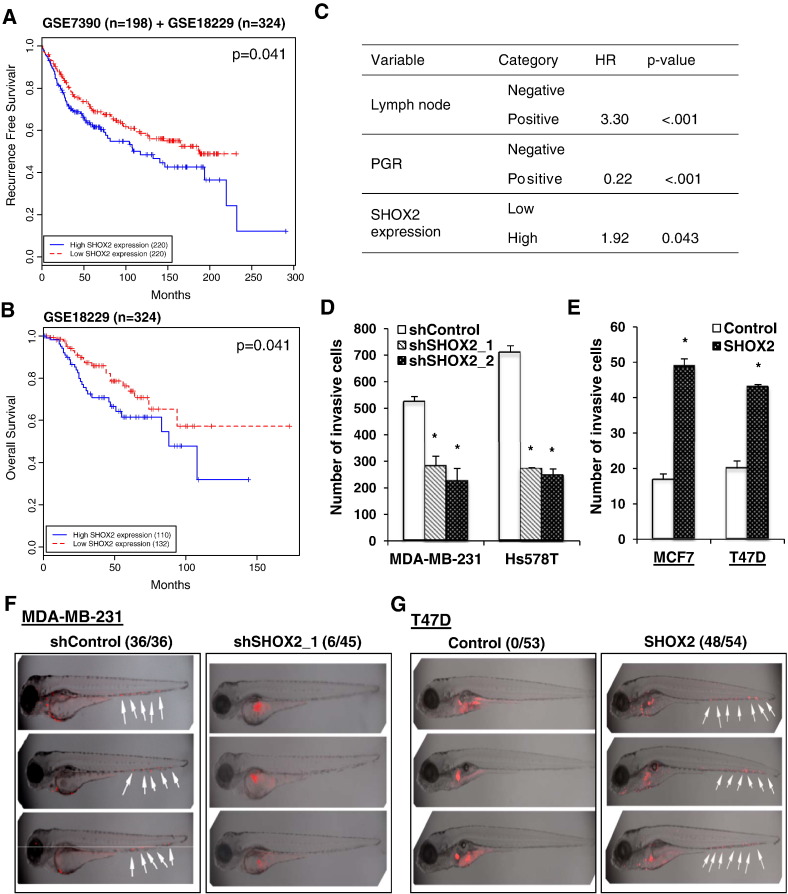
Next, we evaluated the correlation between SHOX2 expression and survival of patients with breast cancer. Univariate survival analysis (Kaplan-Meier method and log-rank test) showed that low SHOX2 expression was associated with both high recurrence-free survival and overall survival rates of patients (P = .041 for both sets; Figure 6, A and B). Multivariate analysis (proportional hazards method) further showed that SHOX2 expression was a bad prognostic factor along with lymph node status and progesterone receptor (PGR) status (Figure 6C).
Figure 6.
SHOX2 is functionally associated with breast tumorigenicity. (A) Univariate survival method (Kaplan-Meier method) of patients with breast cancer indicates a strong correlation between SHOX expression and recurrence-free survival (P = .041; log-rank test). (B) Univariate survival method (Kaplan-Meier method) of patients with breast cancer indicates a strong correlation between SHOX expression and overall survival (P = .041; log-rank test). (C) Multivariate survival analysis (proportional hazards method) shows a positive, independent prognostic importance of SHOX2 expression (P < .05; likelihood ratio test), in addition to the independent prognostic impact of lymph node status and PGR positivity. HR, hazard ratio. (D) Effect of ectopic SHOX2 expression on in vitro invasion of MCF7 and T47D cells. Data are means ± SEM (n = 3). *P < .05 versus Control. (E) Effect of SHOX2 knockdown on in vitro invasion of MDA-MB-231 and Hs578T cells. Data are means ± SEM (n = 3). *P < .05 versus shControl. (F) Effect of SHOX2 knockdown on in vivo dissemination of MDA-MB-231 in zebrafish. (G) Effect of ectopic SHOX2 expression on in vivo dissemination of T47D in zebrafish.
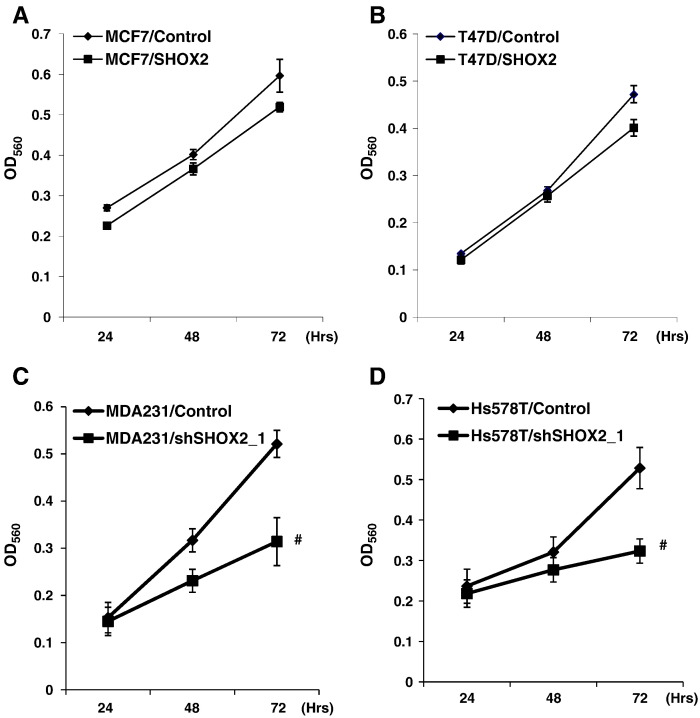
To investigate the importance of SHOX2 in breast tumorigenicity, we investigated how modulating SHOX2 expression affected cell growth and in vitro invasion. Ectopic SHOX2 expression did not affect cell growth in both MCF7 and T47D cells (Figure W5A). However, knockdown of SHOX2 inhibited the growth rate of MDA-MB-231 and Hs578T cells (Figure W5B), further suggesting its importance in maintaining mesenchymal phenotype. However, ectopic SHOX2 expression conferred MCF7 and T47D cells with the ability to invade the Matrigel (Figure 6E), whereas knockdown of SHOX2 led to a greater than 50% reduction in in vitro invasion in both MDA-MB-231 and Hs578T cells (Figure 6D). In subsequent experiments, a well-established zebrafish model was employed to determine the effect of SHOX2 on breast cancer cell in vivo dissemination. Fluorescence dye–labeled cells were microinjected into the perivitelline space of 48-hour postfertilization zebrafish embryos. Thirty hours after injection, dissemination of control MDA-MB-231 cells was seen in all zebrafishes (36 of 36), whereas SHOX2 knockdown MDA-MB-231 cells were only disseminated in 13.3% of zebrafish (6 of 45) (Figure 6F). In contrast, control T47D cells were not disseminated at all (Figure 6G); however, dissemination of T47D cells with forced SHOX2 expression was observed in 88.9% of zebrafishes (48 of 54) (Figure 6G). Together, these results strongly suggest that SHOX2 plays a critical role in breast tumor cell dissemination by facilitating the EMT process.
Discussion
Accumulating evidences have demonstrated a dynamic involvement of the miRNA system in the process of EMT. In our previous attempt to identify miRNAs specifically expressed in epithelial-like breast cancer cells, we revealed that miR-200c, miR-205, and miR-375 are most consistently expressed in epithelial-like breast cancer cells [6]. The amount of miR-375 is elevated in ER-positive breast cancer cells compared with ER-negative cells [22]. Analysis of primary breast tumor tissues reveals that high miR-375 expression is mainly detected in more differentiated tumors [23]. These previous findings are in agreement with our observation that miR-375 is specifically expressed in epithelial-like breast cancer cells that are mostly ER positive (Figure 1). The ability of the miR-200 family and miR-205 to suppress EMT is well established [4,5]. As one of the three miRNAs consistently expressed in epithelial-like breast cancer cells, it is unknown whether miR-375 can also suppress EMT. In this study, we show that ectopic miR-375 expression in mesenchymal-like breast cancer cells suppressed EMT traits (Figure 2), demonstrating that, similar to well-characterized miR-200c and miR-205, miR-375 is also an EMT-suppressive miRNA. Low level of miR-375 expression is associated with poor outcome and metastasis of head and neck squamous cell carcinoma (HNSCC) [24]. Forced miR-375 expression can deter tumorigenicity of HNSCC [24] and HCC [25]. The tumor-suppressing role of miR-375 in HNSCC and HCC is linked to its ability to target astrocyte elevated gene-1 (AEG-1) [25,26]. However, we show that miR-375 suppresses EMT by diminishing SHOX2 expression because forced SHOX2 expression is sufficient to restore EMT traits blocked by miR-375 (Figure 2). Opposite to miR-375, SHOX2 is preferentially expressed in mesenchymal-like breast cancer cells (Figure 1). The inverse correlation between miR-375 and SHOX2 expression is consistent with our finding that miR-375 suppresses EMT by diminishing SHOX2 expression.
The involvement of SHOX2 in tumorigenesis has been suggested since the discovery that hypermethylation of SHOX2 in bronchial aspirates and blood plasma is a clinically useful tumor marker for diagnosis of lung cancer [11,27]. Its expression is recently shown to correlate with tumor recurrence in HCC [12]. In this study, we demonstrate that depletion of SHOX2 is sufficient to abrogate EMT traits in mesenchymal-like breast cancer cells (Figure 2). Conversely, ectopic expression of SHOX2 in epithelial-like breast cancer cells caused EMT (Figure 3), suggesting that SHOX2 is an EMT inducer in breast cancer cells. Consistent with its role in regulating EMT, SHOX2 expression is very low in epithelial-like breast cancer cells (Figure 1). Knockdown of SHOX2 abolished both in vitro invasion and in vivo dissemination of mesenchymal-like breast cancer cells, whereas forced SHOX2 expression confers epithelial-like breast cancer cells with the ability to invade Matrigel and to disseminate in zebrafish (Figure 6). Moreover, high expression of SHOX2 correlates with poor survival of patients with breast cancer and hence is a poor prognostic factor in patients with breast cancer. These data strongly suggest that concurrent loss of miR-375 and gain of SHOX2 expression may be an important step in breast tumor progression and metastasis.
SHOX2 contains the HOX domain and is thus likely to act as a transcription factor. We show that SHOX2 directly facilitates TβR-I expression by activating TβR-I gene transcription (Figure 5), indicating that activation of TGFβ signaling network may be related to SHOX2-induced EMT. This is clearly supported by the observation that SHOX2-induced EMT in epithelial-like breast cancer cells was reversed by the TβR-I inhibitor LY364947 (Figure 4). TGFβ signaling is generally elevated in basal-like breast cancer cells (they are generally mesenchymal-like), whereas it is poorly represented among luminal cells (they are generally epithelial-like) [28]. Our studies raise the possibility that SHOX2 is a critical molecule linking mesenchymal-like breast cancer cells to their TGFβ signature. Several other HOX domain–containing proteins, including Six1, HOXB9, and Prrx1, have been shown capable of inducing EMT in breast cancer cells [29-32]. Especially, both Six1 and HOXB9 facilitate EMT by activating TGFβ signaling [30,33]. We reason that activation of TGFβ signaling may be a common pathway leading to EMT of breast cancer cells.
Our study was performed with established breast cancer cell lines that may not fully simulate clinical setting. However, the consistency seen in the in vivo model and the excellent correlation between SHOX2 expression and survival of patients with breast cancer in breast tumors support the role of SHOX2 in breast tumorigenicity established by our experimental studies. Our studies conclude that miR-375/SHOX2 functional relationship regulates breast tumorigenesis by controlling the process of EMT.
Footnotes
This work was supported by funding from National Institute of Health (NIH) (HL083335), Shanghai Eastern Scholar Fund, and E-Institutes of Shanghai Municipal Education Commission projectE03008.
This article refers to supplementary materials, which are designated by Figures W1 to W5 and are available online at www.neoplasia.com.
Supplementary Materials.
Antibodies
Antibodies used for Western blot analysis were all used at dilution of 1:1000. They include SHOX2 polyclonal antibody (kindly provided by Dr Yi-Ping Chen at Tulane University, New Orleans, LA), Vimentin mAb (Santa Cruz Biotechnology, Santa Cruz, CA, Catalog No. sc-6260), E-cadherin monolyclonal antibody (BD Biosciences, San Jose, CA, Catalog No. 610405), β-Actin mAb (Santa Cruz Biotechnology, Catalog No. sc-47778), TβR-I polyclonal antibody (Cell Signaling Technology, Catalog No. 3712), TβR-II polyclonal antibody (Cell Signaling Technology, Catalog No. 3713), and TβR-III polyclonal antibody (Cell Signaling Technology, Danvers, MA, Catalog No. 2519). Antibodies used for immunofluorescence staining were used at the dilution of 1:200. They include E-cadherin monolyclonal antibody (BD Biosciences, Catalog No. 610405) and Vimentin mAb (BD Biosciences, Catalog No. 550513). FLAG mAb (Sigma, St. Louis, MO, Catalog No. A2220) was used for ChIP (2 μg per reaction).
Figure W1.
Ectopic SHOX2 expression induces EMT characteristics in epithelial-like breast cancer cells. Control and SHOX2-expressing MCF7 cells were subjected to immunofluoresence staining with E-cadherin (A) or vimentin mAb (B). DAPI was used to visualize nuclei.
Figure W2.
Effect of ectopic SHOX2 expression on the expression of mesenchymal-specific transcription factors. qRT-PCR to determine the levels of SLUG, SNAIL, TWIST, ZEB1, and ZEB2 mRNA in both control and SHOX2-overexpressing MCF7 (A) and T47D cells (B). Data are means ± SEM (n = 3). *P < .01 versus Control.
Figure W3.
TβR-I inhibitor LY364947 restores EMT traits in SHOX2-overexpressing epithelial-like breast cancer cells. qRT-PCR was used to determine the effect of LY364947 on E-cadherin (A) and vimentin expression (B) in control or SHOX2-overexpressing MCF7 and T47D cells. Data are means ± SEM (n = 3). #P < .01 and *P < .05 versus SHOX2/vehicle.
Figure W4.
Effect of manipulating SHOX2 level on TGFβ1,TGFβ 2, and TGFβ3 mRNA. qRT-PCR was used to analyze the level of TGFβ1, TGFβ2, and TGFβ3 mRNA in control and SHOX2 knockdown MDA-MB-231 (A), SHOX2 knockdown Hs578T (B), SHOX2-overexpressing MCF7 (C), and SHOX2-overexpressing T47D cells (D). Data are means ± SEM (n = 3).
Figure W5.
Effect of manipulating SHOX2 level on cell proliferation. MTT assay was performed on cell proliferation of SHOX2-transduced MCF7 (A), SHOX2-transduced T47D (B), SHOX2 knockdown MDA-MB-231 (C), and SHOX2 knockdown Hs578T (D) cells. Data are means ± SEM (n = 3). #P < .01 versus Control.
References
- 1.Thiery J.P. Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002;2:442–454. doi: 10.1038/nrc822. [DOI] [PubMed] [Google Scholar]
- 2.Thiery J.P., Acloque H., Huang R.Y., Nieto M.A. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139:871–890. doi: 10.1016/j.cell.2009.11.007. [DOI] [PubMed] [Google Scholar]
- 3.Bartel D.P. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 4.Gregory P.A., Bert A.G., Paterson E.L., Barry S.C., Tsykin A., Farshid G., Vadas M.A., Khew-Goodall Y., Goodall G.J. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol. 2008;10:593–601. doi: 10.1038/ncb1722. [DOI] [PubMed] [Google Scholar]
- 5.Park S.M., Gaur A.B., Lengyel E., Peter M.E. The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev. 2008;22:894–907. doi: 10.1101/gad.1640608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Luo D., Wilson J.M., Harvel N., Liu J., Pei L., Huang S., Hawthorn L., Shi H. A systematic evaluation of miRNA:mRNA interactions involved in the migration and invasion of breast cancer cells. J Transl Med. 2013;11:57. doi: 10.1186/1479-5876-11-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ward A., Balwierz A., Zhang J.D., Küblbeck M., Pawitan Y., Hielscher T., Wiemann S., Sahin Ö. Re-expression of microRNA-375 reverses both tamoxifen resistance and accompanying EMT-like properties in breast cancer. Oncogene. 2013;32:1173–1182. doi: 10.1038/onc.2012.128. [DOI] [PubMed] [Google Scholar]
- 8.Espinoza-Lewis R.A., Yu L., He F., Liu H., Tang R., Shi J., Sun X., Martin J.F., Wang D., Yang J. Shox2 is essential for the differentiation of cardiac pacemaker cells by repressing Nkx2-5. Dev Biol. 2009;327:376–385. doi: 10.1016/j.ydbio.2008.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cobb J., Dierich A., Huss-Garcia Y., Duboule D. A mouse model for human short-stature syndromes identifies Shox2 as an upstream regulator of Runx2 during long-bone development. Proc Natl Acad Sci U S A. 2006;103:4511–4515. doi: 10.1073/pnas.0510544103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gu S., Wei N., Yu L., Fei J., Chen Y. Shox2-deficiency leads to dysplasia and ankylosis of the temporomandibular joint in mice. Mech Dev. 2008;125:729–742. doi: 10.1016/j.mod.2008.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schmidt B., Liebenberg V., Dietrich D., Schlegel T., Kneip C., Seegebarth A., Flemming N., Seemann S., Distler J., Lewin J. SHOX2 DNA methylation is a biomarker for the diagnosis of lung cancer based on bronchial aspirates. BMC Cancer. 2010;10:600. doi: 10.1186/1471-2407-10-600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang T., Zhang H., Cai S.Y., Shen Y.N., Yuan S.X., Yang G.S., Wu M.C., Lu J.H., Shen F. Elevated SHOX2 expression is associated with tumor recurrence of hepatocellular carcinoma. Ann Surg Oncol. 2013;20(Suppl 3):S644–S649. doi: 10.1245/s10434-013-3132-1. [DOI] [PubMed] [Google Scholar]
- 13.Chen H., Zhu G., Li Y., Padia R.N., Dong Z., Pan Z.K., Liu K., Huang S. Extracellular signal–regulated kinase signaling pathway regulates breast cancer cell migration by maintaining slug expression. Cancer Res. 2009;69:9228–9235. doi: 10.1158/0008-5472.CAN-09-1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li Y., Kimura T., Huyck R.W., Laity J.H., Andrews G.K. Zinc-induced formation of a coactivator complex containing the zinc-sensing transcription factor MTF-1, p300/CBP, and Sp1. Mol Cell Biol. 2008;28:4275–4284. doi: 10.1128/MCB.00369-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hu Q., Lu Y.Y., Noh H., Hong S., Dong Z., Ding H.F., Su S.B., Huang S. Interleukin enhancer-binding factor 3 promotes breast tumor progression by regulating sustained urokinase-type plasminogen activator expression. Oncogene. 2013;32:3933–3943. doi: 10.1038/onc.2012.414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huang S., Chakrabarty S. Expression of antisense fibronectin RNA in human colon carcinoma cells disrupts the regulation of carcinoembryonic antigen by transforming growth factor β1. J Biol Chem. 1994;269:28764–28768. [PubMed] [Google Scholar]
- 17.Rouhi P., Jensen L.D., Cao Z., Hosaka K., Länne T., Wahlberg E., Steffensen J.F., Cao Y. Hypoxia-induced metastasis model in embryonic zebrafish. Nat Protoc. 2010;5:1911–1918. doi: 10.1038/nprot.2010.150. [DOI] [PubMed] [Google Scholar]
- 18.Shao J., Teng Y., Padia R., Hong S., Noh H., Xie X., Mumm J.S., Dong Z., Ding H.F., Cowell J. COP1 and GSK3β cooperate to promote c-Jun degradation and inhibit breast cancer cell tumorigenesis. Neoplasia. 2013;15:1075–1085. doi: 10.1593/neo.13966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Teng Y., Xie X., Walker S., White D.T., Mumm J.S., Cowell J.K. Evaluating human cancer cell metastasis in zebrafish. BMC Cancer. 2013;13:453. doi: 10.1186/1471-2407-13-453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lewis B.P., Shih I.H., Jones-Rhoades M.W., Bartel D.P., Burge C.B. Prediction of mammalian microRNA targets. Cell. 2003;115:787–798. doi: 10.1016/s0092-8674(03)01018-3. [DOI] [PubMed] [Google Scholar]
- 21.Friedman R.C., Farh K.K., Burge C.B., Bartel D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009;19:92–105. doi: 10.1101/gr.082701.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.de Souza Rocha Simonini P., Breiling A., Gupta N., Malekpour M., Youns M., Omranipour R., Malekpour F., Volinia S., Croce C.M., Najmabadi H. Epigenetically deregulated microRNA-375 is involved in a positive feedback loop with estrogen receptor α in breast cancer cells. Cancer Res. 2010;70:9175–9184. doi: 10.1158/0008-5472.CAN-10-1318. [DOI] [PubMed] [Google Scholar]
- 23.Jonsdottir K., Janssen S.R., Da Rosa F.C., Gudlaugsson E., Skaland I., Baak J.P., Janssen E.A. Validation of expression patterns for nine miRNAs in 204 lymph-node negative breast cancers. PLoS One. 2012;7:e48692. doi: 10.1371/journal.pone.0048692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Harris T., Jimenez L., Kawachi N., Fan J.B., Chen J., Belbin T., Ramnauth A., Loudig O., Keller C.E., Smith R. Low-level expression of miR-375 correlates with poor outcome and metastasis while altering the invasive properties of head and neck squamous cell carcinomas. Am J Pathol. 2012;180:917–928. doi: 10.1016/j.ajpath.2011.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.He X.X., Chang Y., Meng F.Y., Wang M.Y., Xie Q.H., Tang F., Li P.Y., Song Y.H., Lin J.S. MicroRNA-375 targets AEG-1 in hepatocellular carcinoma and suppresses liver cancer cell growth in vitro and in vivo. Oncogene. 2012;31:3357–3369. doi: 10.1038/onc.2011.500. [DOI] [PubMed] [Google Scholar]
- 26.Nohata N., Hanazawa T., Kikkawa N., Mutallip M., Sakurai D., Fujimura L., Kawakami K., Chiyomaru T., Yoshino H., Enokida H. Tumor suppressive microRNA-375 regulates oncogene AEG-1/MTDH in head and neck squamous cell carcinoma (HNSCC) J Hum Genet. 2011;56:595–601. doi: 10.1038/jhg.2011.66. [DOI] [PubMed] [Google Scholar]
- 27.Kneip C., Schmidt B., Seegebarth A., Weickmann S., Fleischhacker M., Liebenberg V., Field J.K., Dietrich D. SHOX2 DNA methylation is a biomarker for the diagnosis of lung cancer in plasma. J Thorac Oncol. 2011;6:1632–1638. doi: 10.1097/JTO.0b013e318220ef9a. [DOI] [PubMed] [Google Scholar]
- 28.Bhola N.E., Balko J.M., Dugger T.C., Kuba M.G., Sánchez V., Sanders M., Stanford J., Cook R.S., Arteaga C.L. TGF-β inhibition enhances chemotherapy action against triple-negative breast cancer. J Clin Invest. 2013;123:1348–1358. doi: 10.1172/JCI65416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McCoy E.L., Iwanaga R., Jedlicka P., Abbey N.S., Chodosh L.A., Heichman K.A., Welm A.L., Ford H.L. Six1 expands the mouse mammary epithelial stem/progenitor cell pool and induces mammary tumors that undergo epithelial-mesenchymal transition. J Clin Invest. 2009;119:2663–2677. doi: 10.1172/JCI37691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Micalizzi D.S., Christensen K.L., Jedlicka P., Coletta R.D., Barón A.E., Harrell J.C., Horwitz K.B., Billheimer D., Heichman K.A., Welm A.L. The Six1 homeoprotein induces human mammary carcinoma cells to undergo epithelial-mesenchymal transition and metastasis in mice through increasing TGF-β signaling. J Clin Invest. 2009;119:2678–2690. doi: 10.1172/JCI37815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hayashida T., Takahashi F., Chiba N., Brachtel E., Takahashi M., Godin-Heymann N., Gross K.W., Vivanco M., Wijendran V., Shioda T. HOXB9, a gene overexpressed in breast cancer, promotes tumorigenicity and lung metastasis. Proc Natl Acad Sci U S A. 2010;107:1100–1105. doi: 10.1073/pnas.0912710107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ocaña O.H., Córcoles R., Fabra A., Moreno-Bueno G., Acloque H., Vega S., Barrallo-Gimeno A., Cano A., Nieto M.A. Metastatic colonization requires the repression of the epithelial-mesenchymal transition inducer Prrx1. Cancer Cell. 2012;22:709–724. doi: 10.1016/j.ccr.2012.10.012. [DOI] [PubMed] [Google Scholar]
- 33.Chiba N., Comaills V., Shiotani B., Takahashi F., Shimada T., Tajima K., Winokur D., Hayashida T., Willers H., Brachtel E. Homeobox B9 induces epithelial-to-mesenchymal transition-associated radioresistance by accelerating DNA damage responses. Proc Natl Acad Sci U S A. 2012;109:2760–2765. doi: 10.1073/pnas.1018867108. [DOI] [PMC free article] [PubMed] [Google Scholar]