Abstract
Calciphylaxis occurs rarely in the absence of end stage renal disease. Predisposing factors for nonuremic calciphylaxis (NUC) include hyperparathyroidism, coagulopathies, connective tissue disease, liver disease, glucocorticoid use, and malignancy. Warfarin can facilitate vascular calcification by reducing vitamin K-dependent carboxylation of matrix-Gla proteins. An 86-year-old Caucasian woman with a history of polymyalgia rheumatica, two spontaneous deep venous thromboses (DVTs) and multiple fractures was treated with calcium, vitamin D, prednisone, and warfarin. The patient’s low bone density was treated initially with estrogen, then oral bisphosphonate, which was discontinued due to upper gastrointestinal symptoms. Nasal calcitonin was initiated. After 10 years of calcitonin treatment, she was changed to teriparatide. Two months after initiating teriparatide, she developed lower extremity edema and painful erythematous nodular lesions on her calves bilaterally, that progressed to necrotic ulcers despite antibiotic therapy. Biopsy of the lesions showed calcification in the media of small blood vessels and subcutaneous fat with fat necrosis, consistent with calciphylaxis. Teriparatide was discontinued. Aggressive wound care, antibiotics, and intravenous zoledronic acid were initiated. With cessation of teriparatide therapy and intensive wound care, the patient’s lesions resolved over 8 months. We report the first case of NUC precipitated by teriparatide therapy. Our patient had multiple underlying predisposing factors including a connective tissue disorder, glucocorticoid therapy, warfarin use, and possible underlying coagulopathy given her history of multiple DVTs. In such patients, alternative osteoporosis therapies may be preferred.
Keywords: Calciphylaxis, Fracture, Osteoporosis, Teriparatide
Introduction
Calciphylaxis is an uncommon disorder, most typically occurring in patients with end stage renal disease (ESRD) and in renal transplant patients. Calciphylaxis is characterized as a small-vessel calcification and tissue necrosis, presenting as painful subcutaneous nodules that progress to ulcerated skin lesions.
Calciphylaxis has been also reported in patients without renal disease, a condition known as nonuremic calciphylaxis (NUC). Primary hyperparathyroidism, malignancy, alcoholic liver disease, and connective tissue diseases are the most common underlying comorbidities seen in patients with NUC [1].
The findings associated with calciphylaxis were first described in 1898 [2], but it was not until 1962 when the term “calciphylaxis” was introduced by Selye, whose studies demonstrated that parathyroid hormone was able to sensitize animals and precipitate calciphylaxis [3].
Teriparatide [rhPTH(1–34)] is the only anabolic agent approved for the treatment of osteoporosis in the USA. PTH(1–84) is available in Europe. Teriparatide is FDA-approved for the treatment of osteoporosis in men, postmenopausal women, and glucocorticoid-treated individuals at high risk for fracture [4]. Hypercalcemia and hypercalciuria are known side effects [4]; however, non-skeletal calcium deposition has not been reported.
In this article, we report a case of NUC in a patient who received teriparatide and we provide a review of the physiology and treatment options for this rare disorder.
Case presentation
An 86-year-old Caucasian woman with a history of polymyalgia rheumatica (PMR), two spontaneous deep venous thrombosis (DVT) and low bone density was referred for evaluation of lower extremity ulcers. The patient’s first DVT occurred 5 years prior to presentation and was treated with warfarin for 8 months. Warfarin was discontinued and the patient experienced a second DVT within 2 months. Evaluation for underlying coagulopathy was negative and the patient was placed on chronic warfarin therapy. The patient had been diagnosed with PMR and was on chronic glucocorticoid therapy with prednisone 5 mg per day. The patient had had a wrist fracture in her 20s and multiple vertebral compression fractures seen on an abdominal CT scan obtained for abdominal pain 2 years prior to presentation.
The patient was found to have low bone density approximately 18 years prior to presentation based on a dual x-ray absorptiometry scan. Femoral neck bone mineral density (BMD) was 0.570 g/cm2 (T-score −2.5); lumbar spine BMD (L1–L2) was 0.725 g/cm2 (T-score −2.3). The patient was treated with estrogen for 8 years with calcium and vitamin D. Approximately 10 years prior to presentation, the estrogen was discontinued and the patient was placed on oral bisphosphonate therapy which was discontinued 1 year later due to upper gastrointestinal side effects. The patient was then treated with calcitonin until 4 months prior to presentation. Bone density testing demonstrated a 4.7 % decrease in bone density at the femoral neck with a T-score of −2.8. Calcitonin was discontinued and the patient was started on 20 mcg/day of teriparatide subcutaneously.
Within 2 months after initiating teriparatide therapy, she developed painful erythematous nodular lesions on her calves bilaterally. She was diagnosed initially with cellulitis and treated with first oral, then intravenous antibiotics. The lesion on the left calf progressed and became ulcerated. Lower extremity MRI was obtained which showed circumferential subcutaneous edema, felt to be consistent with cellulitis. C-reactive protein was elevated at 1.7 (normal range 0.3–0.9 mg/dl).
Due to lack of clinical improvement despite antibiotics, biopsy was performed. Histologic examination showed calcification of the subcutaneous fat and fat necrosis as well as medial calcification of small vessels, consistent with calciphylaxis (Fig. 1, arrow points to the calcification of the small vessel). Teriparatide was discontinued and the patient was referred to our Metabolic Bone Center. At our center, laboratory studies (Table 1) demonstrated a serum creatinine of 1.1 (0.6–1.3 mg/dl), an eGFR of 46 ml/min/1.73 m2, calcium 9.4 (8.5–10 mg/dl), ionized calcium 5.4 (4.8–5.6 mg/dl), phosphorus 3.5 (2.5–4.6 mg/dl), and iPTH 47 (12–65 pg/ml). Liver enzymes and alkaline phosphatase were normal. ANA was positive at 1:320. Coagulopathy screen off warfarin did not show any abnormalities. As the patient’s d-dimers were elevated, she was felt to be at high risk for recurrent thrombosis and warfarin therapy was resumed. Aggressive wound care was continued and 5-mg intravenous zoledronic acid was administered. The patient received the zoledronic acid infusion approximately 3 months after discontinuation of teriparatide. The patient’s lesions began improving upon discontinuation of the teriparatide and continued to improve, resolving over the ensuing 8 months.
Fig. 1.
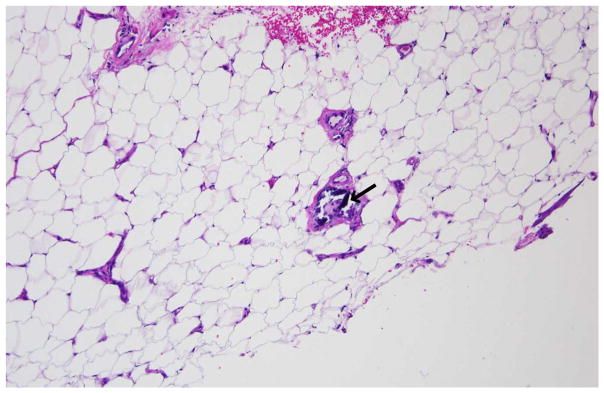
Biopsy of the patient’s ulcerated lesion demonstrating subcutaneous fat and fat necrosis as well as medial calcification of small vessels, consistent with calciphylaxis. The arrow points to the calcification of the small vessel
Table 1.
Laboratory data
| GLU | Na | K | Cl | CO2 | BUN |
| 117 mg/dl | 143 mEq/L | 3.8 mEq/L | 107 mEq/L | 26 mEq/L | 24 mg/dL |
| ALP | AST | ALT | WBC | Hgb | Ht |
| 111 U/L | 20 U/L | 17 IU/mL | 6,800 mm3 | 9.4 g/dL | 30.1 % |
| Cr | Ca | Ionized Ca | PO4 | iPTH | ANA |
| 1.1 mg/dl | 9.4 mg/dl | 5.4 mg/dl | 3.5 mg/dl | 47 pg/dl | 1:320 |
Discussion
Calciphylaxis occurs mainly in patients with ESRD and in renal transplant patients with a prevalence of 1–4 % [5, 6]. The pathogenesis of calciphylaxis remains incompletely understood with many potential factors thought to be contributors. Conditions that are associated with uremia, such as hyperphosphatemia, hyperparathyroidism, and calcium-based phosphate binders are thought to be predisposing factors [6]. A calcium phosphate product above 70 mg2/dl2 has been observed in some dialysis patients with calciphylaxis; however, this is not a consistent finding [7]. Aluminum excess, obesity, alcoholic liver disease, and systemic glucocorticoids also represent risk factors [8].
NUC is a rare disease involving subcutaneous vascular calcification of the small vessels that leads to necrosis of the dermis, subcutaneous tissue, fascia, muscle, or even of the internal organs. The histology of calciphylaxis involves intra-vascular calcium deposition in the media of dermal and subcutaneous arterioles, fibrin thrombi formation, intimal proliferation, and tissue ischemia with subsequent necrosis [1, 9, 10]. Iron deposition has been detected in areas of microvascular calcification, where extravascular calcification has been described between and within the lipocyte [8].
In early experiments studying the pathophysiology of calciphylaxis, animals were initially exposed to what were felt to be sensitizing agents, such as dihydrotachysterol, vitamin D2, vitamin D3, and PTH [2]. The animals were further exposed to a challenger, such as iron, trauma, aluminum, or egg albumin. After this “2-hit” approach, animals developed soft tissue calcification, suggesting that a complex interplay of factors with exposure to specific precipitants, including PTH, could lead to the development of calciphylaxis [3]. Even in the absence of other precipitating factors, parathyroid hormone infusion in rats is associated with increased expression of RANK ligand and down regulation of the expression of osteoprotegerin, inducing calciphylaxis [11].
In NUC, primary hyperparathyroidism, malignancy, alcoholic liver disease, diabetes, protein C and S deficiencies and connective tissue diseases are the most common contributing factors [1]. Warfarin use has also been described as a risk factor [12]. Warfarin is thought to induce calciphylaxis by inhibiting vitamin K carboxylation of matrix-Gla protein, a protein that can inhibit local calcification [13]. Matrix-Gla protein consists of 84 amino acids that are activated by carboxylation of glutamate residues in a vitamin K-dependent fashion. Disruption of matrix-Gla in rats results in extensive vascular calcification [14]. Deficiencies in other vascular calcification inhibitors such as fetuin-A as well as derangements of RANK ligand and osteoprotegerin have been proposed in the pathogenesis of calciphylaxis and are influenced by PTH [15, 16].
Our patient had multiple long standing risk factors for calciphylaxis including a history of connective tissue disorder, glucocorticoid therapy, warfarin use, and a possible underlying coagulopathy. Within 2 months after the initiation of teriparatide, she developed calciphylaxis. It appears that in our patient with predisposing factors, teriparatide, recombinant human parathyroid hormone (1–34), was the triggering factor that induced calciphylaxis, similar to the “2-hit” approach that resulted in calciphylaxis in animal models [3, 11].
The mortality rate in patients with calciphylaxis is approximately 50–60 % with sepsis accounting for nearly half of deaths [1]. Aggressive wound care and antibiotics, sodium thiosulfate, hyperbaric oxygen, and biphosphonates have been used with varying degrees of success in ESRD patients with calciphylaxis [17]. Although glucocorticoids have been identified as a predisposing factor of calciphylaxis, some authors have recommended them as a treatment option in patients without ulcerations or evidence of infection [18]. In this case series, 30–60 mg/day of prednisone appeared to induce a more rapid improvement in disease, but it is not clear that there was long-term benefit. Notably however, in five reports of documented NUC, glucocorticoid administration resulted in four deaths [10]. As increased levels of PTH have been thought to be involved in the pathogenesis of calciphylaxis, cinacalcet, and parathyroidectomy have been also tried as treatment options, with parathyroidectomy leading to reduced mortality in at least one series [19, 20].
The experience with the treatment of NUC is even more limited. Similar strategies to those employed for ESRD have been utilized with inconsistent results [1]. Aggressive wound care is critical, but other modalities have such limited data that recommendations for any one specific therapy cannot be made. In our patient, teriparatide was discontinued as it appeared to be the triggering factor for the development of calciphylaxis. We also used, in addition to aggressive wound care, intravenous zoledronic acid. Biphosphonates have been utilized in calciphylaxis due to anti-inflammatory properties, as they can promote macrophage inhibition and suppression of inflammatory cytokines [21]. Pamidronate treatment has previously been utilized successfully in one patient with NUC [22]. In our patient, utilizing zoledronic acid provided both osteoporosis treatment as well as therapy for calciphylaxis. Consideration was given to changing from warfarin to low molecular weight heparin; however, the patient’s lesions began healing promptly after discontinuation of the teriparatide and continued to heal after administration of zoledronic acid, completely resolving despite continuing on warfarin.
Conclusions
We report the first case of NUC precipitated by teriparatide therapy. Our patient had multiple underlying predisposing factors including glucocorticoid therapy, warfarin use, a connective tissue disorder, and possible underlying coagulopathy. This case of NUC was managed with aggressive wound care, antibiotics, and intravenous zoledronic acid, in addition to discontinuation of teriparatide. NUC is a rare disorder with high mortality. Multiple potential treatment modalities have been described, none of which has been studied in a systematic fashion. The prompt onset of disease with exposure to teriparatide and the prompt initiation of healing after teriparatide was withdrawn despite continuation of all other potential contributing factors, underscores the pathophysiologic importance of parathyroid hormone in the development of calciphylaxis. In osteoporosis patients with multiple underlying predisposing factors for calciphylaxis, alternative osteoporosis therapies may be preferred.
Acknowledgments
This work was partially supported by the NIH T32 training grant (#101776).
Footnotes
Conflicts of interest
None.
Contributor Information
E. K. Spanakis, Division of Endocrinology, Johns Hopkins Hospital, Johns Hopkins University, Baltimore, MD, USA
D. E. Sellmeyer, Email: dsellme1@jhmi.edu, Division of Endocrinology, Johns Hopkins Bayview Hospital, Johns Hopkins University, 5200 Eastern Ave, Mason F Lord Center Tower, Suite 4300, Baltimore, MD 21224, USA
References
- 1.Nigwekar SU, Wolf M, Sterns RH, Hix JK. Calciphylaxis from nonuremic causes: a systematic review. Clin J Am Soc of Nephrol: CJASN. 2008;3:1139–1143. doi: 10.2215/CJN.00530108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bryant JHWW. A case of calcification of the arteries and obliterative endarteritis associated with hydronephrosis in a child aged six months. Guy’s Hosp Rep. 1898;55:17–20. [Google Scholar]
- 3.Selye H, Gabbiani G, Strebel R. Sensitization to calciphylaxis by endogenous parathyroid hormone. Endocrinology. 1962;71:554–558. doi: 10.1210/endo-71-4-554. [DOI] [PubMed] [Google Scholar]
- 4.Neer RM, Arnaud CD, Zanchetta JR, et al. Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med. 2001;344:1434–1441. doi: 10.1056/NEJM200105103441904. [DOI] [PubMed] [Google Scholar]
- 5.Angelis M, Wong LL, Myers SA, Wong LM. Calciphylaxis in patients on hemodialysis: a prevalence study. Surgery. 1997;122:1083–1089. doi: 10.1016/s0039-6060(97)90212-9. discussion 1089–1090. [DOI] [PubMed] [Google Scholar]
- 6.Budisavljevic MN, Cheek D, Ploth DW. Calciphylaxis in chronic renal failure. J Am Soc Nephrol: JASN. 1996;7:978–982. doi: 10.1681/ASN.V77978. [DOI] [PubMed] [Google Scholar]
- 7.Weenig RH. Pathogenesis of calciphylaxis: Hans Selye to nuclear factor kappa-B. J Am Acad Dermatol. 2008;58:458–471. doi: 10.1016/j.jaad.2007.12.006. [DOI] [PubMed] [Google Scholar]
- 8.Weenig RH, Sewell LD, Davis MD, McCarthy JT, Pittelkow MR. Calciphylaxis: natural history, risk factor analysis, and outcome. J Am Acad Dermatol. 2007;56:569–579. doi: 10.1016/j.jaad.2006.08.065. [DOI] [PubMed] [Google Scholar]
- 9.Rogers NM, Teubner DJ, Coates PT. Calcific uremic arteriolopathy: advances in pathogenesis and treatment. Semin Dialysis. 2007;20:150–157. doi: 10.1111/j.1525-139X.2007.00263.x. [DOI] [PubMed] [Google Scholar]
- 10.Vedvyas C, Winterfield LS, Vleugels RA. Calciphylaxis: a systematic review of existing and emerging therapies. J Am Acad Dermatol. 2012;67:e253–e260. doi: 10.1016/j.jaad.2011.06.009. [DOI] [PubMed] [Google Scholar]
- 11.Ma YL, Cain RL, Halladay DL, Yang X, Zeng Q, Miles RR, Chandrasekhar S, Martin TJ, Onyia JE. Catabolic effects of continuous human PTH (1–38) in vivo is associated with sustained stimulation of RANKL and inhibition of osteoprotegerin and gene-associated bone formation. Endocrinology. 2001;142:4047–4054. doi: 10.1210/endo.142.9.8356. [DOI] [PubMed] [Google Scholar]
- 12.Asobie N, Wong E, Cook MG. Calciphylaxis in a diabetic patient provoked by warfarin therapy. Clin Exp Dermatol. 2008;33:342–344. doi: 10.1111/j.1365-2230.2007.02626.x. [DOI] [PubMed] [Google Scholar]
- 13.Wallin R, Cain D, Sane DC. Matrix Gla protein synthesis and gamma-carboxylation in the aortic vessel wall and proliferating vascular smooth muscle cells—a cell system which resembles the system in bone cells. Thromb Haemost. 1999;82:1764–1767. [PubMed] [Google Scholar]
- 14.Howe AM, Webster WS. Warfarin exposure and calcification of the arterial system in the rat. Int J Exp Pathol. 2000;81:51–56. doi: 10.1046/j.1365-2613.2000.00140.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schafer C, Heiss A, Schwarz A, Westenfeld R, Ketteler M, Floege J, Muller-Esterl W, Schinke T, Jahnen-Dechent W. The serum protein alpha 2-Heremans-Schmid glycoprotein/fetuin-A is a systemically acting inhibitor of ectopic calcification. J Clin Investig. 2003;112:357–366. doi: 10.1172/JCI17202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Luo G, Ducy P, McKee MD, Pinero GJ, Loyer E, Behringer RR, Karsenty G. Spontaneous calcification of arteries and cartilage in mice lacking matrix GLA protein. Nature. 1997;386:78–81. doi: 10.1038/386078a0. [DOI] [PubMed] [Google Scholar]
- 17.Vedvyas C, Winterfield LS, Vleugels RA. Calciphylaxis: a systematic review of existing and emerging therapies. J Am Acad Dermatol. 2011;67(6):e253–e260. doi: 10.1016/j.jaad.2011.06.009. [DOI] [PubMed] [Google Scholar]
- 18.Fine A, Zacharias J. Calciphylaxis is usually non-ulcerating: risk factors, outcome and therapy. Kidney Int. 2002;61:2210–2217. doi: 10.1046/j.1523-1755.2002.00375.x. [DOI] [PubMed] [Google Scholar]
- 19.Mohammed IA, Sekar V, Bubtana AJ, Mitra S, Hutchison AJ. Proximal calciphylaxis treated with calcimimetic ‘Cinacalcet’. Nephrol Dial Transplant. 2008;23:387–389. doi: 10.1093/ndt/gfm676. [DOI] [PubMed] [Google Scholar]
- 20.Kang AS, McCarthy JT, Rowland C, Farley DR, van Heerden JA. Is calciphylaxis best treated surgically or medically? Surgery. 2000;128:967–971. doi: 10.1067/msy.2000.110429. discussion 971–962. [DOI] [PubMed] [Google Scholar]
- 21.Cecchini MG, Felix R, Fleisch H, Cooper PH. Effect of bisphosphonates on proliferation and viability of mouse bone marrow-derived macrophages. J Bone Miner Res: Off J Am Soc Bone Miner Res. 1987;2:135–142. doi: 10.1002/jbmr.5650020209. [DOI] [PubMed] [Google Scholar]
- 22.Schliep S, Schuler G, Kiesewetter F. Successful treatment of calciphylaxis with pamidronate. Eur J Dermatol: EJD. 2008;18:554–556. doi: 10.1684/ejd.2008.0499. [DOI] [PubMed] [Google Scholar]


