The protein tyrosine phosphatase (PTP) family of enzymes play critical roles in controlling cellular signaling, in concert with the protein tyrosine kinases (PTKs).[1] Misregulated tyrosine phosphorylation can lead to inflammation, cancer, metabolic and autoimmune disorders.[2–4] As a result, the enzymes that control tyrosine phosphorylation are intriguing therapeutic targets for the treatment of a host of human diseases. While the PTKs have successfully been targeted for drug design,[3, 5] PTP inhibitors have not yet made it to the clinic.[6, 7] One PTP whose inhibition is therapeutically attractive is the lymphoid tyrosine phosphatase (LYP or PTPN22). LYP is expressed exclusively in hematopoietic cells and exercises an inhibitory effect on T cell receptor signaling.[8, 9] It has been proposed that LYP inhibition could be therapeutic in the treatment of human autoimmunity.[10–16] In addition, LYP has been implicated in mast cell signaling and the anaphylactic response and it has been shown that LYP inhibitors attenuate this response.[17, 18]
A number of small molecule inhibitors of LYP have been reported to date and can largely be divided among three classes: salicylic acids[17, 19–21], auranofin analogs[22–24] and allosteric inhibitors.[25] The salicylic acid-based compounds are mimics of phosphotyrosine and behave as reversible competitive inhibitors of LYP with IC50 potencies ranging from nanomolar to low micromolar range and up to 9-fold selectivity for LYP over other PTPs.[17] The auranofin analogs are covalent but reversible competitive inhibitors of LYP with low micromolar IC50 values and notable (~7-fold) selectivity over other PTPs in vitro, with significant activity in cells and animal models.[18, 23] A series of allosteric inhibitors has been obtained with moderate potency (IC50 values in the 8 – 20 µM range) and little selectivity.[25] More recently, epigallocatechin-3,5-digallate has been reported as the most potent LYP inhibitor to date, with an IC50 value of 50 nM[26] and a series of thiuram disulfides have emerged as covalent inhibitors of LYP activity.[27]
We have become interested in developing covalent inhibitors of LYP for use as chemical probes for investigating the roles of this critical enzyme in physiological and pathological cellular signaling pathways. Several covalent, irreversible inhibitors of PTPs have been reported as activity-based probes for proteomic profiling, including non-hydrolyzable α-bromobenzyl phosphonates[28, 29] and phenyl vinyl sulfones and sulfonates.[30] While covalent inhibitors have significant advantages as mechanism-based probes for proteomic profiling[31, 32] and several successful drugs are known to function through a covalent mechanism, irreversible inhibitors have largely been avoided in drug development due to the disadvantage of potential off-target reactivity.[33] However, covalent inhibitors confer the advantage of having reactivity that can be tuned for selectivity towards their target and can exert a prolonged duration of action compared to conventional reversible inhibitors.[33] Interestingly, in recent years there has been a conceptual shift to strategically design covalent inhibitors to intentionally exploit non-catalytic nucleophilic residues as targets for adduct formation as a means to achieve selectivity. As an example, afatinib (Boehringer Ingelheim) is a covalent tyrosine kinase inhibitor that has recently been approved for use in the treatment of metastatic non-small cell lung cancer.[34] Afatinib contains an acrylamide moiety that targets a non-catalytic cysteine residue in the human epidermal growth factor receptor 2 (Her2) and epidermal growth factor receptor (EGFR) kinases, forming a covalent Michael adduct. Considering that LYP has an active site Cys involved in catalysis and two non-conserved cysteine residues in the catalytic site[35] we hypothesized that soft electrophiles could also be used in the development of covalent inhibitors of LYP.
In a recent high-throughput screen of the National Institutes of Health Molecular Libraries for compounds capable of inhibiting LYP activity (PubChem AID:1779),[28] we identified a few compounds that contained electrophilic moieties and could, potentially, inhibit LYP activity through a covalent, irreversible inhibition mechanism. One compound in particular, compound 1 (Figure 1), was identified for further study. This compound contains both a phosphotyrosine mimietic benzoic acid moiety and also a soft, electrophilic acrylonitrile moiety. It seemed reasonable to propose that the benzoic acid moiety could interact with the phosphotyrosine binding pocket and that an appropriately spaced nucleophile might react with the electrophilic acrylonitrile. Indeed, the kinetics of LYP inhibition by compound 1 were indicative of a time-dependent interaction. Therefore, we carried out a detailed kinetic analysis of the interaction between compound 1 and the catalytic domain of LYP. We observed a mono-exponential decrease in the first order rate constant, kobs, which is consistent with the model shown in Scheme 1. This model involves a two-step inactivation mechanism, in which the inhibitor first forms a non-covalent interaction with the enzyme (E•I), followed by irreversible covalent modification of the enzyme (E–I).[36, 37] By fitting the kobs values as a function of inhibitor concentration we observed saturation kinetics as shown in Figure 2, and calculated the kinetic constants KI = 0.25 ± 0.06 µM and kinact = 0.009 ± 0.002 s−1 for compound 1, taking into account the effect of substrate protection. Taken together, these kinetic results suggest an active site-directed irreversible (or pseudo-irreversible) mode of inhibition.
Figure 1.
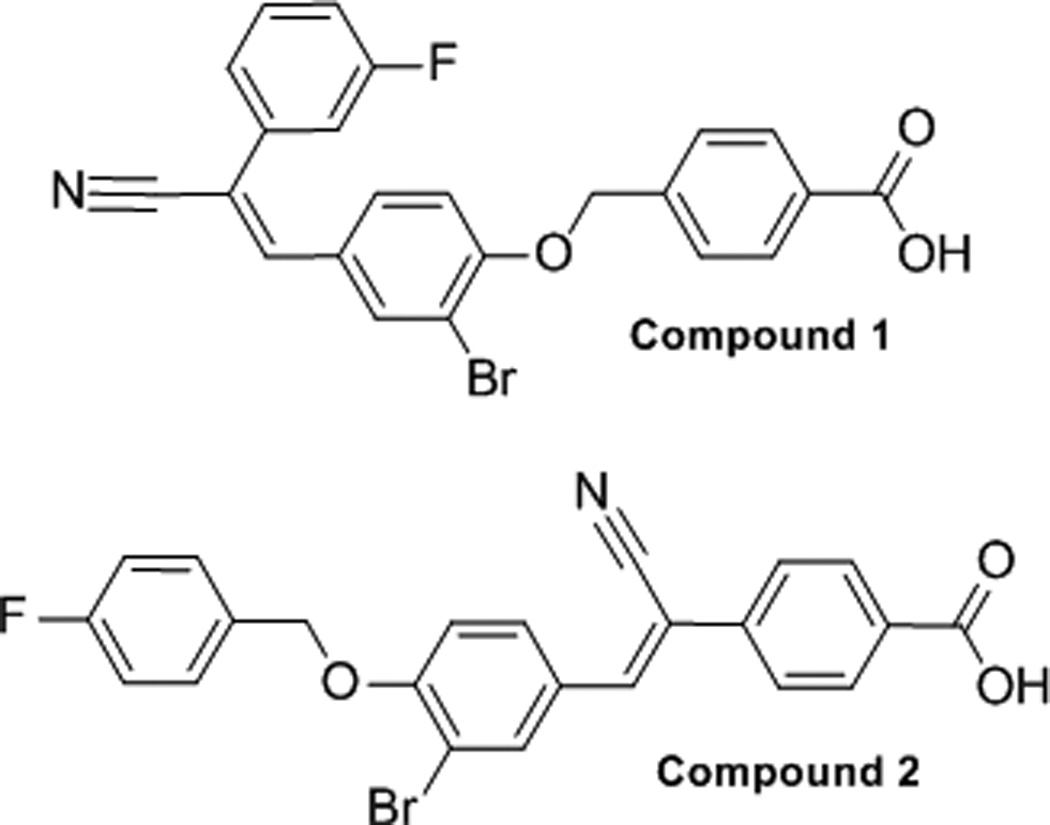
Structures of two electrophilic compounds with activity as covalent LYP inhibitors.
Scheme 1.
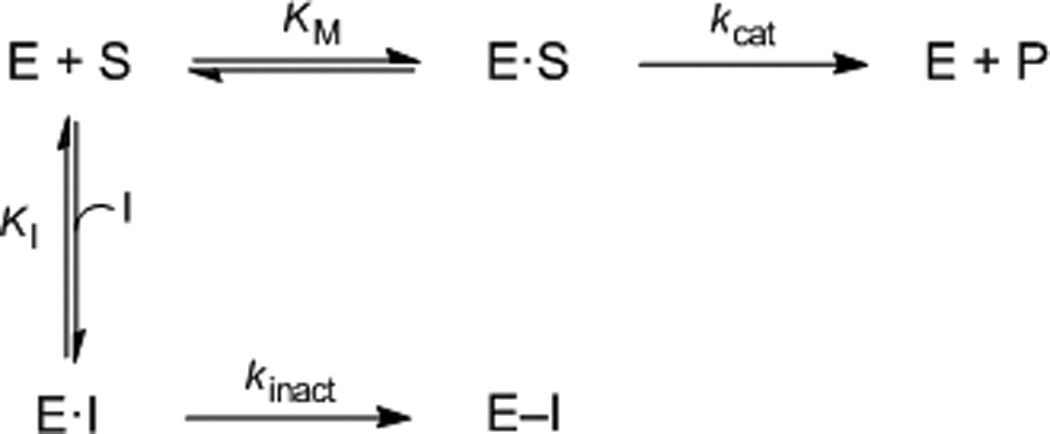
Equation illustrating the irreversible inhibition of an enzyme.
Figure 2.
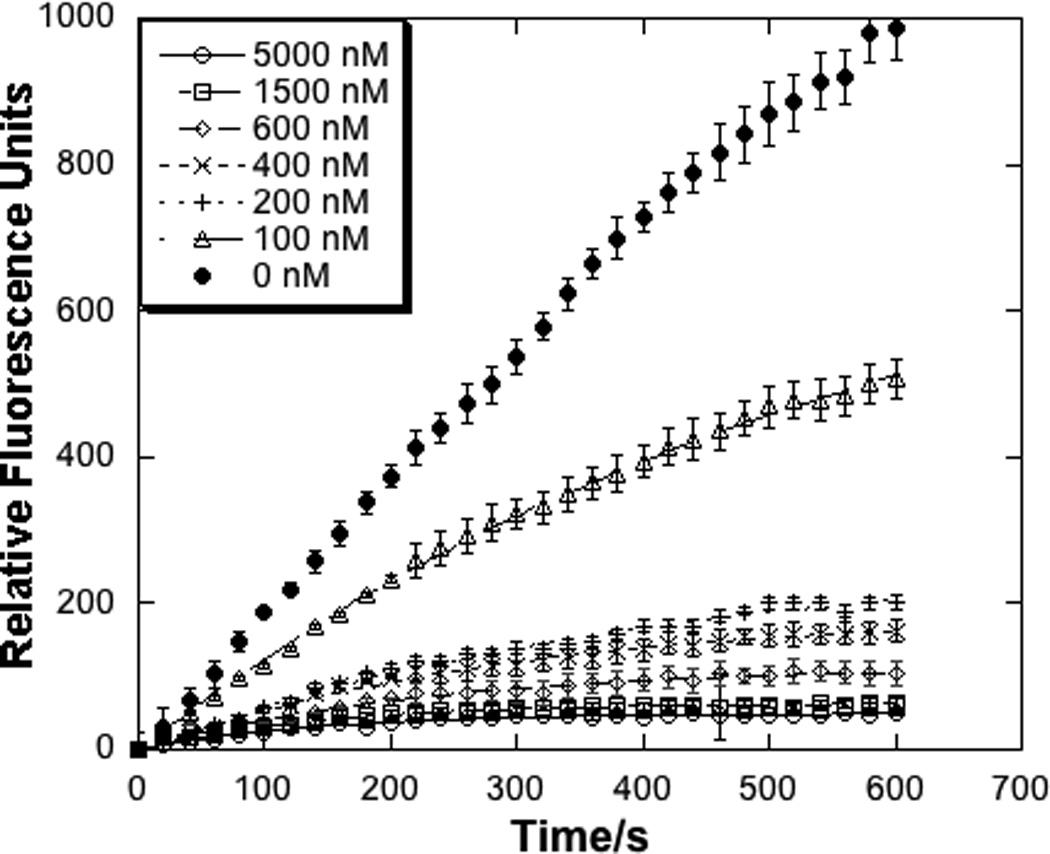
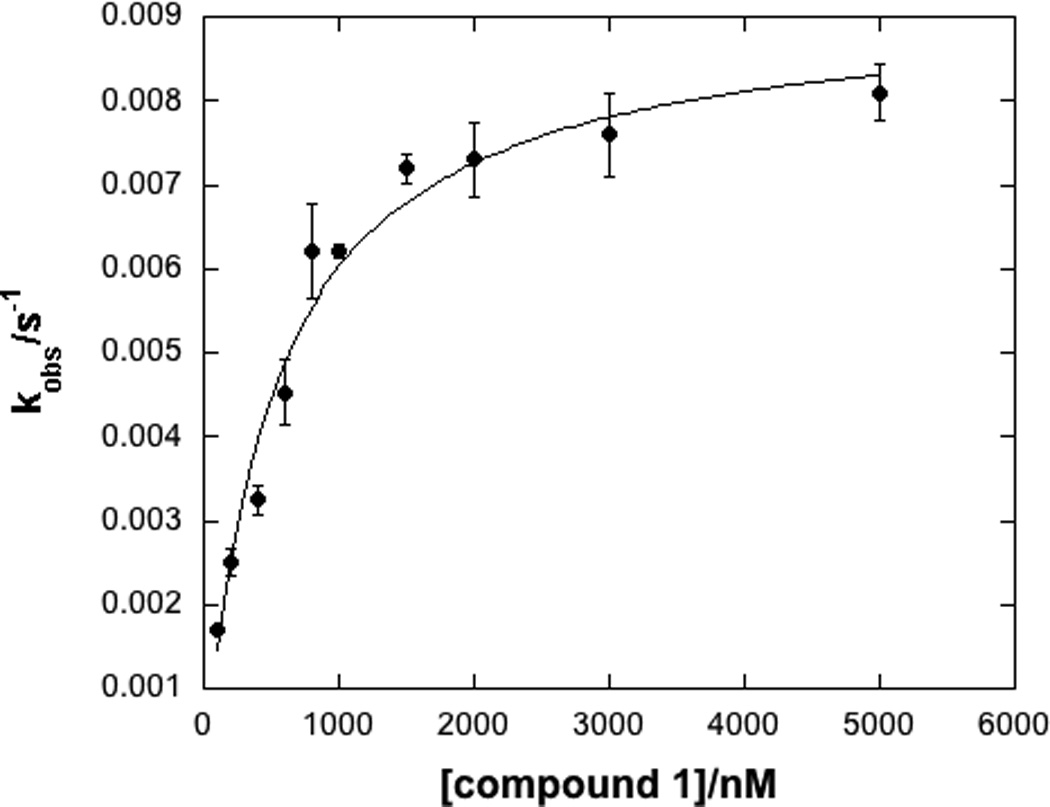
Time dependent inhibition of LYP by compound 1 (selected progress curves, left panel) and a fit of the kobs data to obtain KI and kinact (right panel).
We then obtained a series of analogs of compound 1 from ChemBridge (San Diego, CA). Although all of the compounds tested displayed time-dependent inhibition of LYP activity, only compound 2 showed more potent inhibition of LYP than the parent compound, with a KI = 0.05 ± 0.01 µM and kinact = 0.008 ± 0.002 s−1 (Figure 3). To our knowledge, the KI values, kinact, and efficiency ratios (kinact/KI) observed for compounds 1 and 2 represent the best kinetic parameters reported in the literature to date for covalent inhibitors of PTPs (see Table 1). For example, the phenyl vinyl sulfone (PVS) and phenyl vinyl sulfonate (PVSN) active site-directed inhibitors have KI values of 290 and 350 µM with YopH,[30] while a α-bromobenzyl phosphonate (BBP) biotinylated probe had a KI value of 4100 µM with YopH.[29] The efficiency ratios of compounds 1 and 2 are more than four orders of magnitude better than those observed for the PVS and PVSN compounds.[30] The efficiency ratios of compounds 1 and 2 are approx. five orders of magnitude better than that of the BBP-biotinylated probe.[29] Although the PVS, PVSN and BBP probes were tested on YopH rather than LYP, making direct comparisons of efficiency ratios difficult, it is clear that compounds 1 and 2 are significantly more potent.
Figure 3.

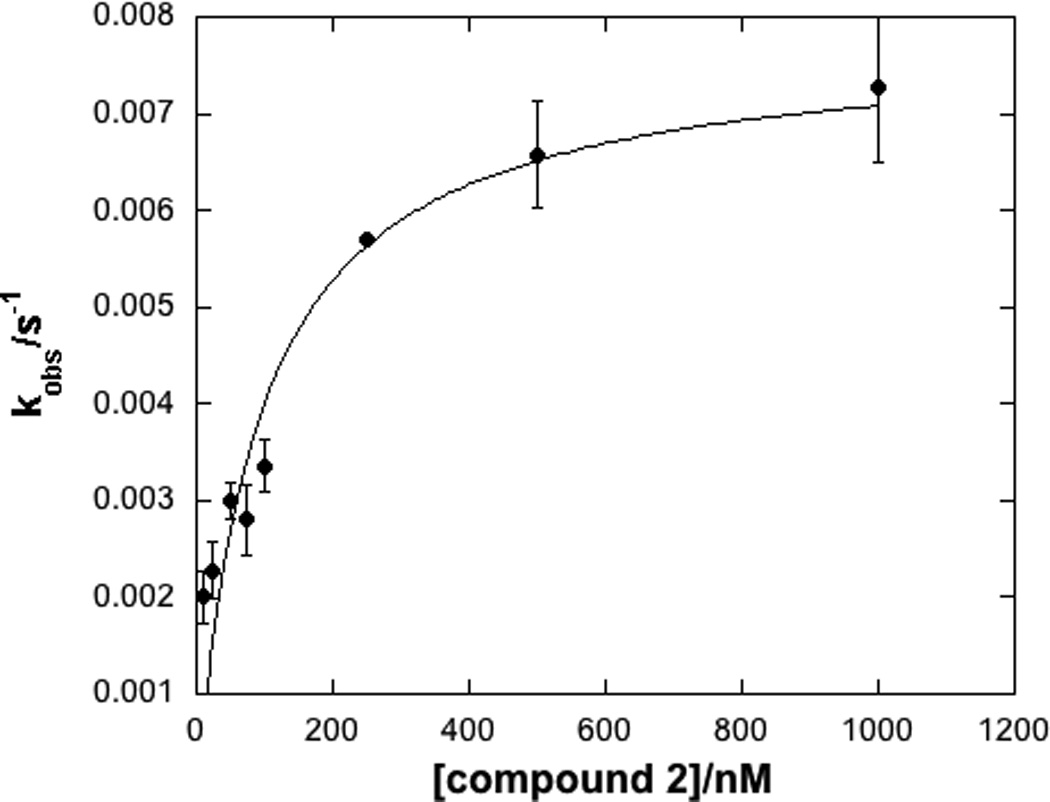
Time dependent inhibition of LYP by compound 2 (selected progress curves, left panel) and a fit of the kobs data to obtain KI and kinact (right panel).
Table 1.
Kinetic parameters for a series of covalent PTP inhibitors
| Inhibitor (enzyme) | kinact (s−1) | KI (µM) | kinact/KI (µM−1s−1) |
|---|---|---|---|
| Compound 1 (LYP) | 0.009 | 0.25 | 0.036 |
| Compound 2 (LYP) | 0.008 | 0.05 | 0.17 |
| BBP (YopH) | 0.002 | 4100 | 0.00000049 |
| PVS (YopH) | 0.001 | 350 | 0.0000029 |
| PVSN (YopH) | 0.01 | 290 | 0.0000034 |
Notably, the spacing between the phosphotyrosine mimetic benzoic acid moiety and the electrophilic acrylonitrile moiety is significantly different in compound 1 and compound 2. While it seems reasonable that the initial interaction between LYP and the compounds would be mediated by the benzoic acid moiety binding in the phosphotyrosine binding pocket, it is not clear whether the covalent interaction arises from nucleophilic attack at the acrylonitrile by a nucleophile outside of the catalytic site, or subsequent rearrangement and nucleophilic attack by one of the cysteine residues in the catalytic site. Repeated attempts to characterize the adducts by mass spectrometry failed, as is common with similar electrophiles.[38] However, we see no evidence of adduct reversibility after extensive dialysis, indicating that the adduct is, indeed, pseudo-irreversible. Future work will be aimed at identifying the nucleophile responsible for adduct formation, with the ultimate goal of selectively targeting LYP by targeting nonconserved residues in the enzyme.
Experimental Section
A detailed description of the materials and methods employed in this work can be found in the Supporting Information section along with structures of all of the analogs of compound 1 and compound 2 tested as covalent LYP inhibitors.
Acknowledgements
This work was supported in part by funding from the National Institutes of Health: grant NS056945 to NB and DK080165 to AMB and NB. The MLSMR screen completed by the Conrad Prebys Center for Chemical Genomics at Sanford|Burnham Medical Research Institute was funded through NIH Roadmap grant: U54 HG005033-02 with Dr. John C. Reed as PI. The authors also wish to thank the following members of the Prebys Center: Dr. Eduard Sergienko and Dr. Thomas D.Y. Chung for critical reading and editing of this manuscript (ES also guided initial assay development). Dr. Ekatarina V. Bobkova (presently at Allostere, Inc. San Diego, CA), Sharon Colayco, and Justin Rascon for implementation and execution of the screen and hit validation (Summary AID: 1784), and Dr. Ying Su for initial compound analysis and ordering during the hit validation. We also thank Dr. Michael Kay for helpful discussions about the kinetic data.
Abbreviations
- PTP
protein tyrosine phosphatase
- PTK
protein tyrosine kinase
- LYP
lymphoid tyrosine phosphatase
- LYPcat
catalytic subunit of LYP
- PVS
phenyl vinyl sulfone
- PVSN
phenyl vinyl sulfonate
- BBP
α-bromobenzyl phosphonate
- YopH
Yersinia tyrosine phosphatase
- DMSO
dimethylsulfoxide
- DiFMUP
6,8-difluoro-4-methylumbelliferyl phosphate
- DiFMU
6,8-difluoro-4-methylumbelliferone.
References
- 1.Hunter T. Curr. Opin. Cell Biol. 2009;21:140–146. doi: 10.1016/j.ceb.2009.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alonso A, Sasin J, Bottini N, Friedberg I, Friedberg I, Osterman A, Godzik A, Hunter T, Dixon J, Mustelin T. Cell. 2004;117:699–711. doi: 10.1016/j.cell.2004.05.018. [DOI] [PubMed] [Google Scholar]
- 3.Bialy L, Waldmann H. Angew. Chem. Int. Ed. 2005;44:2–27. doi: 10.1002/anie.200461517. [DOI] [PubMed] [Google Scholar]
- 4.Zhang Z-Y. Curr. Opin. Chem. Biol. 2001;5:416–423. doi: 10.1016/s1367-5931(00)00223-4. [DOI] [PubMed] [Google Scholar]
- 5.Zhang Z-Y. Annu. Rev. Pharmacol. Toxicol. 2002;42:209–234. doi: 10.1146/annurev.pharmtox.42.083001.144616. [DOI] [PubMed] [Google Scholar]
- 6.Blaskovich M. Curr Med Chem. 2009;16:2095–2176. doi: 10.2174/092986709788612693. [DOI] [PubMed] [Google Scholar]
- 7.Heneberg P. Curr. Med. Chem. 2009;16:706–733. doi: 10.2174/092986709787458407. [DOI] [PubMed] [Google Scholar]
- 8.Gjörloff-Wingren A, Saxena M, Williams S, Hammi D, Mustelin T. Eur. J. Immunol. 1999;29:3845–3854. doi: 10.1002/(SICI)1521-4141(199912)29:12<3845::AID-IMMU3845>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 9.Wu J, Katrekar A, Honigberg LA, Smith AM, Conn MT, Tang J, Jeffery D, Mortara K, Sampang J, Williams SR, Buggy J, Clark JM. J. Biol. Chem. 2006;281:11002–11010. doi: 10.1074/jbc.M600498200. [DOI] [PubMed] [Google Scholar]
- 10.Bottini N, Musumeci L, Alonso A, Rahmouni S, Nika K, Rostamkhani M, MacMurray J, Pellecchia M, Eisenbarth GS, Comings D, Mustelin TA. Nat. Genet. 2004;36:337–338. doi: 10.1038/ng1323. [DOI] [PubMed] [Google Scholar]
- 11.Begovich AB, Carlton VE, Honigberg LA, Schrodi SJ, Chokkalingam AP, Alexander HC, Ardlie KG, Huang Q, Smith AM, Spoerke JM, Conn MT, Chang M, Chang SY, Saiki RK, Catanese JJ, Leong DU, Garcia VE, McAllister LB, Jeffery DA, Lee AT, Batliwalla F, Remmers E, Criswell LA, Seldin MF, Kastner DL, Amos CI, Sninsky JJ, Gregersen PK. Am. J. Hum. Genet. 2004;75:330–337. doi: 10.1086/422827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Carlton VE, Hu X, Chokkalingam AP, Schrodi SJ, Brandon R, Alexander HC, Chang M, Catanese JJ, Leong DU, Ardlie KG, Kastner DL, Seldin MF, Criswell LA, Gregersen PK, Beasley E, Thomson G, Amos CI, Begovich AB. Am. J. Hum. Genet. 2005;77 doi: 10.1086/468189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Smyth D, Cooper JD, Colline JE, Heward JM, Franklyn JA, Howson JM, Vella A, Nutland S, Rance HE, Maier L, Barratt BJ, Guja C, Ionescu-Tirgoviste C, Savage DA, Dunger DB, Widmer B, Strachan DP, Ring SM, Walker N, Clayton DG, Twells RC, Gough SC, Todd JA. Diabetes. 2004;53:3020–3023. doi: 10.2337/diabetes.53.11.3020. [DOI] [PubMed] [Google Scholar]
- 14.Kyogoku C, Langefeld CD, Ortmann WA, Lee A, Selby S, Carlton VE, Chang M, Ramos P, Baechler EC, Batliwalla F, Novitzke J, Williams AH, Gillett C, Rhodine P, Graham RR, Ardlie KG, Gaffney PM, Moser KL, Petri M, Begovich AB, Gregersen PK, Behrens TW. Am. J. Hum. Genet. 2004;75:504–507. doi: 10.1086/423790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Orru V, Tsai S, Rueda B, Fiorillo E, Stanford SM, Dasgupta J, Hartiala J, Zhao L, Ortego-Centeno N, D'Alfonso S, Group IC, Arnett F, Wu H, Gonzalez-Gay M, Tsao B, Pons-Estel B, Alarcon-Riquelme M, He Y, Zhang Z-Y, Allayee H, Chen X, Martin J, Bottini N. Hum. Mol. Genet. 2009;18:569–579. doi: 10.1093/hmg/ddn363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rodriguez-Rodriguez L, Taib W, Topless R, Steer S, Gonzalez-Escribano M, Balsa A, Pascual-Salcedo D, Gonzalez-Gay M, Raya E, Fernandez-Gutierrez B, Gonzalez-Alvaro I, Bottini N, Witte T, Viken M, Coenen M, van Riel P, Franke B, den Heijer M, Radstake T, Wordsworth P, Lie B, Merriman T, Martin J. Arthritis Rhem. 2011;63:365–372. doi: 10.1002/art.30145. [DOI] [PubMed] [Google Scholar]
- 17.He Y, Liu S, Menon A, Stanford S, Oppong E, Gunawan A, Wu L, Wu D, Barrios AM, Bottini N, Cato A, Zhang Z-Y. J. Med. Chem. 2013;56:4990–5008. doi: 10.1021/jm400248c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Obiri D, Flink N, Maier J, Neeb A, Maddalo D, Thiele W, Menon A, Stassen M, Kulkarni RA, Garabedian M, Barrios AM, Cato A. Allergy. 2012;67:175–182. doi: 10.1111/j.1398-9995.2011.02731.x. [DOI] [PubMed] [Google Scholar]
- 19.Vang T, Xie Y, Liu W, Vidovic D, Liu Y, Wu S, Smith D, Rinderspacher A, Chung C, Gong G, Mustelin T, Landry DW, Rickert RC, Schürer S, Deng S-X, Tautz L. J. Med. Chem. 2011;54:562–571. doi: 10.1021/jm101004d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xie Y, Liu Y, Gong G, Rinderspacher A, Deng S-X, Smith DH, Toebben U, Tzilianos E, Branden L, Vidovic D, Chung C, Schurer S, Tautz L, Landry DW. Bioorg. Med. Chem. Lett. 2008;18:2840–2844. doi: 10.1016/j.bmcl.2008.03.079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yu X, Sun J-P, He Y, Guo X, Liu S, Zhou B, Hudmon A, Zhang Z-Y. Proc. Nat. Aca. Sci. 2007;104:19767–19772. doi: 10.1073/pnas.0706233104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Karver MR, Krishnamurthy D, Bottini N, Barrios AM. J. Inorg. Biochem. 2010;104:268–273. doi: 10.1016/j.jinorgbio.2009.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Karver MR, Krishnamurthy D, Kulkarni RA, Bottini N, Barrios AM. J. Med. Chem. 2009;52:6912–6918. doi: 10.1021/jm901220m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Krishnamurthy D, Karver MR, Fiorillo E, Orru V, Stanford SM, Bottini N, Barrios AM. J. Med. Chem. 2008;51:4790–4795. doi: 10.1021/jm800101w. [DOI] [PubMed] [Google Scholar]
- 25.Stanford SM, Krishnamurthy D, MD F, Messina R, Debnath B, Li S, Liu T, Kazemi R, Dahl R, He Y, Yu X, Chan AC, Zhang Z-Y, Barrios AM, Woods VL, Jr, Neamati N, Bottini N. J. Med. Chem. 2011;54:1640–1654. doi: 10.1021/jm101202j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kulkarni RA, Vellore NA, Bliss M, Stanford S, Falk M, Bottini N, Baron R, Barrios AM. ChemBioChem. 2013;13:1640–1647. doi: 10.1002/cbic.201300273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kulkarni RA, Stanford S, Vellore NA, Krishnamurthy D, Bliss M, Baron R, Bottini N, Barrios AM. ChemMedChem. 2013;8:1561–1568. doi: 10.1002/cmdc.201300215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Stanford S, Krishnamurthy D, Kulkarni RA, Karver C, Bruenger E, Walker L, Ma C, Chung T, Sergienko E, Bottini N, Barrios AM. Methods. 2013 doi: 10.1016/j.ymeth.2013.07.022. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kumar S, Zhou B, Liang F, Wang W, Zhang Z-Y. PNAS. 2004;101:7943–7948. doi: 10.1073/pnas.0402323101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu S, Zhou B, Yang H, He Y, Jiang Z, Kumar S, Wu L, Zhang Z-Y. J. Am. Chem. Soc. 2008;130:8251–8260. doi: 10.1021/ja711125p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cravatt B, Wright A, Kozarich J. Annu. Rev. Biochem. 2008;77:383–414. doi: 10.1146/annurev.biochem.75.101304.124125. [DOI] [PubMed] [Google Scholar]
- 32.Karisch R, Fernandez M, Taylor P, Virtanen C, St-Germain J, Jin L, Harris I, Mori J, Mak T, Senis Y, Ostman A, Moran M, Neel B. Cell. 2011;146:826–840. doi: 10.1016/j.cell.2011.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Singh J, Petter R, Baillie T, Whitty A. Nat. Rev. Drug Discov. 2011;10:307–317. doi: 10.1038/nrd3410. [DOI] [PubMed] [Google Scholar]
- 34.Chen X, Zhu Q, Zhu L, Pei D, Liu Y, Yin Y, Schuler M, Shu Y. Lung Cancer. 2013;81:155–161. doi: 10.1016/j.lungcan.2013.02.021. [DOI] [PubMed] [Google Scholar]
- 35.Tsai S, Sen U, Zhao L, Greenleaf W, Dasgupta J, Fiorillo E, Orru V, Bottini N, Chen X. Biochem. 2009;48:4838–4845. doi: 10.1021/bi900166y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tian W-X, Tsou C-L. Biochem. 1982;21:1028–1032. doi: 10.1021/bi00534a031. [DOI] [PubMed] [Google Scholar]
- 37.Walker B, Elmore D. Biochem. J. 1984;221:277–280. doi: 10.1042/bj2210277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Serafimova I, Pufall M, Krishnan S, Duda K, Cohen M, Maglathlin R, McFarland J, Miller R, Frödin M, Taunton J. Nat Chem Biol. 2012;8:471–476. doi: 10.1038/nchembio.925. [DOI] [PMC free article] [PubMed] [Google Scholar]


