Abstract
Background
Based on ethnographic investigations and mathematical models, older sexual partners are often considered a major risk factor for HIV for young women in sub-Saharan Africa. Numerous public health campaigns have been conducted to discourage young women from relationships with older men. However, longitudinal evidence relating sex-partner age-disparity on HIV acquisition in women is limited.
Methods
Using data from a population-based, open cohort in rural KwaZulu-Natal, South Africa, we studied 15–29 year old women who were HIV seronegative at first interview between January 2003 and June 2012 (n=2,444). Using proportional hazards models, we analysed whether the age-disparity of each woman’s most recent sexual partner at each annual round of HIV testing was associated with subsequent HIV acquisition.
Results
458 HIV seroconversions occurred over 5,913 person-years of follow-up (incidence rate: 7.75 per 100 person-years). The age-disparity of women’s partners was not associated with HIV acquisition when measured either continuously (hazard ratio [HR] for a one-year increase in partner’s age: 1.00, 95% confidence interval [CI] 0.97–1.03) or categorically (man ≥5 years older: HR 0.98, 95%CI 0.81–1.20; man ≥10 years older: HR 0.98, 95%CI 0.67–1.43). These results were robust to adjustment for known socio-demographic and behavioural HIV risk factors, and did not vary significantly by women’s age, marital status, education attainment, or household wealth.
Conclusions
In this rural KwaZulu-Natal setting with very high HIV incidence, partner age-disparity did not predict HIV acquisition amongst young women. Campaigns to reduce age-disparate sexual relationships may not be a cost-effective use of HIV-prevention resources in this community.
Keywords: HIV, sexual behaviour, South Africa, women
Introduction
In recent years, there has been much concern in sub-Saharan Africa regarding age-disparate relationships. Most current National Strategic HIV Plans in countries with generalized epidemics name age-disparate relationships as a driver of the HIV epidemic (see Table 1). The World Health Organization considers sexual partnerships between young women and substantially older men an important contributor to young people’s vulnerability for HIV.1
Table 1.
National Strategic Plans containing references to age-disparate sexual relationships
| % HIV prevalence (ages 15–49)* | NSP period | Age-disparity or “sugar daddy” cited as risk factor | Justification given | Policies mandated | Explicit reduction target set | |
|---|---|---|---|---|---|---|
| Swaziland | 26.0 | 2009–14 | Yes | Prevalence of inter-generational sex, especially amongst out-of-school young women | Mandated, no specific policy | |
| Botswana | 23.4 | 2010–16 | Yes | Older men have longer sexual history; Young women having intergenerational sex have lower condom use | Mandated, no specific policy | |
| Lesotho | 23.3 | 2012–16 | Yes | None | Yes† | Increase BCC coverage† |
| South Africa | 17.3 | 2012–16 | Yes | Intergenerational relationships increase risk of HIV exposure | Yes | |
| Zimbabwe | 14.9 | 2011–15 | Yes | Inter-generational sex is a factor that makes women and girls more vulnerable | Mandated, no specific policy | |
| Namibia | 13.4 | 2011–16 | Yes | “Sugar daddy” phenomenon well-known in Namibia. Inter-generational sex associated with STIs, multiple and concurrent partnerships and introduction of HIV into younger cohort | Yes † | |
| Zambia | 12.5 | 2011–15 | Yes | - | Yes | |
| Mozambique | 11.3 | 2010–14 | Yes | Sexual relations between individuals from different generations associated with transactional and less-safe sex | ||
| Malawi | 10.0 | 2009–13† | Yes | - | Yes | |
| Uganda | 7.2 | 2011/12–14/15 | Yes | Prevalence of cross-generational sex (15–19 year-old women with men at least 10 years older) † | Yes | Reduce cross-generational sex by 50% by 2015† |
| Kenya | 6.2 | 2008/09–12/13 | No | Transactional relationships may be important but “sugar daddy” relationships are possibly less frequent than generally thought | ||
| Tanzania | 5.8 | 2008–12 | Yes | Prevalence of cross-generational sex, which is linked to transactional sex and multiple concurrent partnerships | Yes | Reduce cross-generational sex by 50% for teenage girls by 2025 |
| Cameroon | 4.6 | 2011–15 | Yes | - | No | |
| Central African Republic | 4.6 | 2006–10 | No | |||
| Nigeria | 3.7 | 2010–15 | Yes | - | Yes | |
| Togo | 3.4 | 2012–15 | Yes | Prevalence of intergenerational relationships is increasing, and is a source of infections among young women | No | |
| Congo | 3.3 | 2009–13 | No | |||
| Chad | 3.1 | 2007–11 | Yes | - | No | |
| Côte d’Ivoire | 3.0 | 2011–15 | Yes† | Young people in sexual relationships with older partners lack negotiating power | Mandated, no specific policy | 80% of youth adopt lower risk sexual behaviours |
| Rwanda | 2.9 | 2009–12 | Yes | Disparity in HIV prevalence amongst 20–24 year old men and women attributed to cross-generational sex | Yes |
Based on the most recent NSP available for countries with adults HIV prevalence over 2% in 2011. No NSPs published since 2002 were available for Angola, Equatorial Guinea, Gabon or Guinea-Bissau. BCC: Behaviour change communication. NSP: National Strategic Plan.
HIV prevalence for 2011 from UNAIDS Report on the global AIDS epidemic 2012. 3
From an accompanying National Prevention Strategy rather than National Strategic Plan.
This concern appears to have arisen from a number of observed ecological associations. First, the distribution of HIV incidence and prevalence in predominantly heterosexual, sexually transmitted epidemics is consistently shifted towards younger ages for women compared to men. One explanation for this would be that relationships in which HIV is transmitted are typified by an older male and a younger female partner. Second, sub-Saharan Africa has both the largest average relationship age disparities,2 and the highest HIV prevalence,3 in the world. Third, population-level variation in age-disparity and HIV prevalence also appears to positively co-vary within regions of sub-Saharan Africa.4
As a result, numerous public health campaigns have been implemented, aiming to discourage sexual relationships between widely age-disparate partners or “sugar daddies”.5 Population Services International (PSI), an international non-governmental organization, has run health messaging programmes targeting age-disparate relationships in at least eight sub-Saharan countries.6 In 2012, the KwaZulu-Natal (KZN) provincial Department of Health in South Africa began a campaign under the title ‘“Sugar Daddies” Destroy Lives’, aiming to “create a taboo against cross-generational sex” with 14–25 year old women in order to reduce HIV infection.7
Despite these considerable investments, quantitative evidence for a causal relationship between age disparities and HIV remains very limited. Cross-sectional analyses in Rakai, Uganda (1994–1998), Manicaland, Zimbabwe (1998–2000), and nationwide in South Africa (2003) and Kenya (2007) found positive associations between having an older partner and prevalent HIV infection.8–11 Cross-sectional studies cannot, however, rule out many alternative explanations for an association between age-disparity and HIV infection, since measurement of the exposure does not precede the outcome. For example, women could preferentially seek out relationships with older men after they have become HIV-infected – for instance, because older men are better able to support them economically;5,12 and HIV-infected women who have older partners may live longer because they are economically better off and thus have better access to antiretroviral treatment (ART).
Nevertheless, there are plausible mechanisms through which age-disparate relationships might be a driver of the HIV epidemic in sub-Saharan Africa.5 First, HIV incidence rates rise rapidly from youth through middle age amongst men in sub-Saharan Africa.3 As a result, young women in age-disparate relationships are, ceteris paribus, at higher risk of HIV exposure than if they were in a relationship with someone their own age.
Second, sexual behaviours may vary with the level of age-disparity in sexual relationships; specifically, fewer preventative measures may be taken. This risky behaviour may arise because the man perceives younger women to be less likely to be HIV infected, or because the economic and social power differentials arising from the age disparity may make it difficult for women to negotiate safe sex with their older partners.13,14 For example, there is some evidence that men are less likely to use condoms in sexual relationships with younger women.15,16 In particular, the subset of age-disparate relationships which are non-marital relationships between a young woman and a substantially older man which involve both age and economic asymmetries – the typical definition of a “sugar daddy” relationship – are expected to exhibit riskier behaviours.
Additionally, theoretical mathematical models have investigated the potential impact of age-mixing in relationships on population HIV dynamics, and on individual infection risk.17–20 Predictions from such models have varied in their estimation of the extent to which age disparities are required for the transmission of the epidemic through the generations. Early studies suggested that the impact of age-mixing on epidemics would be considerable,20,21 however recent analysis has suggested that age-disparate relationships may have only limited effects on community HIV prevalence, and that HIV can reach high endemic levels without highly age-disparate partnerships.22
Given the limited quantitative evidence and the mixed predictions from modelling studies, an empirical longitudinal analysis would represent a significant improvement on existing evidence. We therefore test the hypothesis that age-disparate relationships increase the risk of HIV infection amongst young women, using one of Africa’s largest HIV incidence cohorts. The cohort is located in a rural community in KZN where HIV incidence in young women is extremely high,23 and male HIV prevalence rises from 8% amongst 20–24 year olds to a peak of over 40% between the ages of 30 and 34.24,25
Methods
We conducted survival analysis using data from population-based, longitudinal surveillance conducted by the Africa Centre for Health and Population Studies (hereafter Africa Centre) in a predominantly rural community in the uMkhanyakude district of KZN. The district is one of the most deprived in the country and is characterized by high levels of circular migration and HIV infection. The Africa Centre has been collecting household demographic data since 2000.26 In addition, since 2003 adults have annually been invited to participate in anonymised HIV testing, when they are also asked questions relating to their sexual history and behaviours over the past 12 months. These questions are asked face-to-face by fieldworkers recruited from the local community.23 HIV test results are linked anonymously to other information in the database.
Data were available from January 2003 until June 2012. Inclusion criteria for our analysis were that respondents were (i) female; (ii) were aged between 15 and 30 in the study period; (iii) were HIV seronegative at first participation in the HIV surveillance; (iv) had at least one more valid HIV test result recorded; and (v) participated at least once in the annual General Health surveillance questionnaire which elicits information on sexual behaviour and sexual partners’ ages. Individuals entered the cohort at the date of the first interview at which they reported a sexual partnership. They were right-censored at either their thirtieth birthday or on the date of their most recent interview at which they tested HIV seronegative. We further excluded any person-time during which a respondent indicated not having had sex (e.g. the respondent reported no sexual partners in the past year), under the assumption that such a respondent was not at risk of sexually contracting HIV in that period.
The primary outcome was HIV seroconversion; we assumed the date of HIV seroconversion to be midway between the date of an individual’s last negative and first positive HIV test (results were insensitive to selecting a random date between the two test – analysis not shown). The exposure of interest was the age-disparity of each woman’s most recent sexual partner (in only 0.8% of interviews did women report more than one sexual partner in the past year). This exposure was time-varying, and updated based on women’s reports of the age of their most recent sexual partner in the General Health interview immediately prior to the HIV test that began a period of observation (e.g. if a woman completed General Health interviews on 1 July 2008 and 1 July 2009, and had a negative HIV test on 1 September 2009 and a positive one on 1 September 2010, the most recent partner reported in July 2008 would be the relevant partner for the period in which seroconversion occurred). For our main analysis individuals could not enter the cohort until they had reported the age of at least one partner.
We considered multiple functional forms of age-disparity. Our primary approach was to use age-disparity as a continuous variable; this reflected a theoretical orientation towards a continuum of risk, the empirical finding that there was no sharp change in risk at any cut-off, and the statistical reality that it is more flexible than dichotomous measures. We additionally considered step functions to capture age-disparities that are larger than 5 and larger than 10 years, corresponding to common definitions of age-disparate relationships,5,8,12,16 and larger than 20 years as an extreme case.
We considered as potential socio-demographic time-varying confounders: current completed education (none or primary, 0–7 years; secondary, 8–12 years; tertiary, >12 years); household wealth (quintiles of the first component identified by principal-components analysis of 28 household assets, toilet type, and sources of water, electricity, and energy – defined for all households in the study area); and marital status (never married, engaged, married, previously married). We considered as potential behavioural confounders or mediators: age at sexual debut and three time-varying measures of sexual behaviour in the past 12 months: number of partners (1 vs. >1); any casual partner (yes vs. no); and lowest level of condom use with any partner (never, sometimes, always).
We used Cox proportional hazards models, verifying the proportional-hazards assumption using the Schoenfeld residuals from each regression. The primary model included the woman’s age (centred at age 15) and their relationship age-disparity. We included linear, quadratic and cubic terms in age, but only a linear term in age-disparity, since tests of functional form showed that this combination generated the best model fit based on Akaike’s Information Criteria (see Table, Supplemental Digital Content 1). Our model also included indicator variables for the year of observation, which controls for unmeasured confounders that change over time. Notably, these time period controls account for the potentially important confounding by differential availability of HIV prevention and treatment services over time.
We then considered whether any effect of age-disparity varied by women’s age in three categories (15–19, 20–24, and 25–29 years old) using indicator variables for disparate relationships in each age-group. Finally, we added socio-demographic and sexual behaviour covariates to the model. We reran our analyses after multiple imputation of missing variables in the dataset (see Table, Supplemental Digital Content 2).
Ethical approval for Africa Centre surveillance was granted by the Biomedical Research Ethics Committee, University of KwaZulu-Natal. Informed consent is required separately for the main and sexual behaviour questionnaires, and for anonymised HIV sero-testing. This analysis was exempted from additional ethical review by the Harvard School of Public Health Institutional Review Board due to its use of anonymised secondary data.
Results
Between January 2003 and June 2012, 2,444 women, contributing 5,913 years of person-time, met the inclusion criteria and had full covariate information. Each woman was tested for HIV between two and eight times (41.6% twice, 25.1% three times; 16.0% four times; 12.6% 5–8 times). The median gap between tests was 365 days (interquartile range 343–421 days).
Baseline characteristics of the respondents are provided in Table 2 divided into five-year age cohorts. A large majority of the young women had never been married, although 22% were engaged by ages 25–29. Very few women reported multiple partners or a casual partner in the past year – the latter declined strongly with age.
Table 2.
Baseline characteristics of the study sample of 15–29 year old women
| All | 15–19 | 20–24 | 25–29 | |
|---|---|---|---|---|
| Sample size | 2,444 | 1,112 | 982 | 350 |
| Number of subsequent seroconversions | 458 | 136 | 251 | 71 |
| Age at baseline (years) | 20 (18 to 23) | 18 (17 to 19) | 21 (20 to 23) | 27 (25 to 28) |
| Partner age-disparity in most recent relationship (years) | 3 (2 to 5) | 3 (2 to 5) | 3 (1 to 5) | 4 (2 to 6) |
| Highest educational attainment | ||||
| None or Primary (0–7 years) | 9.6 | 13.8 | 4.3 | 11.1 |
| Secondary (8–12 years) | 86.5 | 84.7 | 92.0 | 77.1 |
| Tertiary | 3.8 | 1.4 | 3.8 | 11.7 |
| Household wealth quintile | ||||
| Lowest | 19.5 | 19.1 | 19.5 | 20.9 |
| 2nd lowest | 25.7 | 23.9 | 26.6 | 29.1 |
| Middle | 24.9 | 24.4 | 25.7 | 24.6 |
| 2nd highest | 18.0 | 19.6 | 17.2 | 15.1 |
| Highest | 11.9 | 13.0 | 11.1 | 10.3 |
| Marital status | ||||
| Never Married | 87.6 | 93.5 | 88.4 | 66.3 |
| Engaged | 10.1 | 6.2 | 10.2 | 22.0 |
| Married | 2.3 | 0.3 | 1.4 | 11.4 |
| Divorced/Separated/Widowed | 0.0 | 0.0 | 0.0 | 0.3 |
| Age at sexual debut (years) | 17 (16 to 18) | 16 (15 to 17) | 18 (16 to 19) | 18 (16 to 19) |
| Multiple partners in past 12 months | 1.3 | 1.2 | 1.8 | 0.3 |
| Casual partner in past 12 months | 4.3 | 5.8 | 3.4 | 2.0 |
| Lowest condom use level in relationships in past 12 months | ||||
| Never | 49.3 | 45.1 | 50.7 | 59.1 |
| Sometimes | 27.1 | 25.0 | 29.0 | 28.3 |
| Always | 23.6 | 29.9 | 20.3 | 12.6 |
Figures for categorical data are percentages; figures for continuous data are medians and (Interquartile ranges)
The typical sexual partnership involved a man who was three years older than the woman, with a range from 10 years younger to 47 years older. Overall, 922 (37.7%) respondents reported having had a partner five or more years older than themselves at some point in the study period, and 222 (9.1%) reported one 10 or more years older.
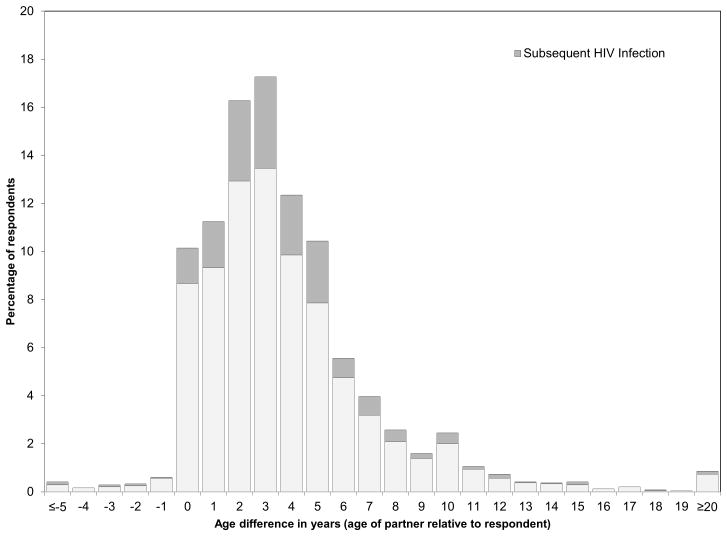
During follow-up 458 HIV seroconversions were observed (Kaplan-Meier curve shown in Figure, Supplemental Digital Content 3). The overall incidence rate was 7.75 per 100 person-years (95% confidence interval (CI): 7.07–8.49). The incidence rate per 100 person-years rose from 7.79 (95% CI: 6.59–9.22) amongst those aged under 20 to 8.63 (95% CI: 7.63–9.77) for those aged 20–24, before dropping to 5.63 (95% CI: 4.46–7.11) for those aged 25–29. A crude comparison of the age-disparity of each woman’s most recent sexual partner at baseline and their subsequent risk of seroconverting whilst under observation (Figure 1), suggests no obvious correlation.
Figure 1.
Age-disparity between female respondent and most recent male sexual partner at baselines
In survival analysis, we find no significant relationship between age-disparity and HIV acquisition, a result that is robust to model specification and to our approach to account for missing data. In a model containing only respondent’s age and relational age-disparity (Model 1, Table 3), there was (Hazard ratio (HR) for a one-year increase in partner’s age: 1.00, 95% CI 0.97–1.03). This result appeared to vary only very slightly – and non-significantly – by five-year age categories (Model 2). The addition of socioeconomic covariates (Model 3) and behavioural covariates (Model 4) had almost no effect on this result. Analyses using categorical measures of age-disparity found similar results (man ≥5 years older: HR 0.98, 95% CI 0.81–1.20; man ≥10 years older: HR 0.98, 95% CI 0.67–1.43; man≥20 years older: HR 0.61, 95% CI 0.15–2.46).
Table 3.
Multivariable Cox proportional hazards models of HIV acquisition
| Model 1 | Model 2 b | Model 3 | Model 4 | |||||
|---|---|---|---|---|---|---|---|---|
| Age-disparity (one year increase in partner’s age) | 1.00 | (0.97 – 1.03) | 1.00 | (0.98 – 1.03) | 1.00 | (0.98 – 1.03) | ||
| Respondent aged 15–19 | 1.03 | (0.98 – 1.07) | ||||||
| Respondent aged 20–24 | 0.96 | (0.90 – 1.01) | ||||||
| Respondent aged 25–29 | 0.98 | (0.92 – 1.05) | ||||||
| Age of respondent (years, centred at 15 years old)a | ||||||||
| Age | 1.18 | (1.07 – 1.29) | 1.09 | (0.95 – 1.24) | 1.19 | (1.08 – 1.31) | 1.21 | (1.09 – 1.33) |
| Age squared | 0.73 | (0.67 – 0.80) | 0.73 | (0.67 – 0.81) | 0.73 | (0.67 – 0.81) | 0.74 | (0.67 – 0.81) |
| Age cubed | 1.11 | (1.05 – 1.17) | 1.13 | (1.06 – 1.21) | 1.11 | (1.05 – 1.17) | 1.10 | (1.05 – 1.17) |
| Highest educational attainment | ||||||||
| None or Primary (0–7 years) | 1.36 | (0.98 – 1.89) | 1.29 | (0.93 – 1.81) | ||||
| Secondary (8–12 years) | 1.00 | 1.00 | ||||||
| Tertiary | 0.81 | (0.50 – 1.32) | 0.82 | (0.51 – 1.33) | ||||
| Household wealth quintile | ||||||||
| Lowest | 1.38 | (0.95 – 2.00) | 1.38 | (0.94 – 2.01) | ||||
| 2nd lowest | 1.34 | (0.94 – 1.91) | 1.36 | (0.95 – 1.94) | ||||
| Middle | 1.54 | (1.09 – 2.18) | 1.56 | (1.10 – 2.22) | ||||
| 2nd highest | 1.36 | (0.94 – 1.95) | 1.36 | (0.94 – 1.96) | ||||
| Highest | 1.00 | 1.00 | ||||||
| Current marital status of respondent | ||||||||
| Never Married | 1.00 | 1.00 | ||||||
| Engaged | 0.96 | (0.69 – 1.32) | 0.97 | (0.71 – 1.34) | ||||
| Married | 0.10 | (0.01 – 0.72) | 0.10 | (0.01 – 0.73) | ||||
| Divorced/Separated/Widowed | 13.41 | (1.84 – 98.0) | 12.34 | (1.68 – 90.7) | ||||
| Age at sexual debut (one-year increment) | 0.98 | (0.92 – 1.04) | ||||||
| Any casual partner in past 12 months | 1.12 | (0.72 – 1.74) | ||||||
| Multiple partners in past 12 months | 2.10 | (1.16 – 3.81) | ||||||
| Lowest condom use level in relationships in past 12 months | ||||||||
| Never | 1.00 | |||||||
| Sometimes | 0.89 | (0.71 – 1.11) | ||||||
| Always | 1.08 | (0.86 – 1.37) | ||||||
| Akaike Information Criteria | 6,482 | 6,484 | 6,472 | 6,474 | ||||
Values are hazard ratios and 95% confidence intervals. For all models, n=2,444, time at risk = 5,913 person-years and there were 458 seroconversions. All models contain indicator variables for year of observation (not shown).
The coefficient on Age squared represents a 10-unit change in this variable; the coefficient on Age cubed represents a 100-unit change in this variable.
This model also contains indicator variables for age categories. A joint test for equality on the three age by age-disparity interaction terms was not statistically significant (χ2 = 2.26, p-value: 0.324).
After multiple imputation of missing values, our results remained essentially unchanged (Table, Supplemental Digital Content 4). Interactions of age-disparity with woman’s age did not change our findings (Table, Supplemental Digital Content 5). No significant effect-modification of the relationship between age-disparities and HIV infection was seen for marital status or educational attainment (Table, Supplemental Digital Content 6). For wealth, there may be an effect such that those in the richest and poorest quintiles are protected by greater age-disparities while those in the middle quintile are placed at greater risk by them, but this was only significant for a binary measure of age-disparity with a cut-point of ≥5 years.
Discussion
We tested whether age-disparities affect HIV incidence in young women using data from one of Africa’s largest population-based HIV incidence cohorts. In a high HIV prevalence and incidence context, we find no evidence that having an older male partner increases the risk of infection in young women. Our result does not appear to be due to low statistical power, as indicated by the very tight confidence intervals. We obtain this finding even though age-disparate relationships are common in this community - over the study period, more than one-third of young women had a partner five or more years older than themselves – higher than the South African national figure (32.6%)10 – and almost one in ten had a partner at least ten years older – comparable to rural Uganda (16.3%).8 Our results are robust to the functional form of age-disparity and to the inclusion of several potentially confounding covariates.
There are several plausible explanations for the absence of significant associations between age-disparate relationships and HIV acquisition risk in this community. First, young women in age-disparate relationships may be more careful in selecting their partners so as to offset their risk. There is evidence that while men have more control over partnership sexual practices, including condom use, women have control over partnership formation and dissolution.16,27 This can be seen in the effectiveness of interventions aimed at changing young women’s partners using information or financial incentives.28,29 Careful partner selection could explain our findings if young women select lower-risk partners as relationship age-disparity increases. If young women are aware that older partners are likely to be more risky, they may use knowledge drawn from their social networks to identify lower-risk partners from amongst the pool of older men available to them. Such a strategy is most likely to succeed where dense social networks allow successful identification of risky partners, such as in the cohesive rural community studied here.30
Second, age-disparities are likely to be more weakly linked to economic disparities in this setting than elsewhere, reducing or eliminating the infection risk that has been hypothesized to exist within age-disparate relationships due to steep differentials in sexual negotiation power.12,27 Socioeconomic differentials in this poor rural community are far less pronounced than in many other settings, in particular urban areas, limiting the potential for resource transfers from older men to younger women. In fact, the majority of men living in this community were unemployed over the observation period.31
Third, ART is increasingly widely available in this community. As a result, while more older men may be infected with HIV, relatively few may have high viral loads and therefore be likely to infect their partners. This context is in contrast to earlier studies, which took place in the pre-ART era, perhaps explaining the discrepant results between our work and prior cross-sectional studies. However, when we stratified our analyses by year of observation time (Table 4), we found no change in the association between age-disparity and HIV over time, suggesting that ART provision alone does not explain our null finding.
Table 4.
Sensitivity analysis using stratification by year of observation
| Year of observation | Number of HIV seroconversions | Hazard ratio for a one-year increase in age disparity | 95% confidence interval |
|---|---|---|---|
| 2003–5 | 29 | 1.00 | 0.92 to 1.08 |
| 2006 | 80 | 1.03 | 0.99 to 1.07 |
| 2007 | 88 | 0.98 | 0.94 to 1.03 |
| 2008 | 99 | 1.00 | 0.95 to 1.04 |
| 2009 | 75 | 1.00 | 0.95 to 1.05 |
| 2010 | 62 | 1.03 | 0.98 to 1.09 |
| 2011–12 | 24 | 0.99 | 0.90 to 1.08 |
Years 2003–05 and January 2011–June 2012 grouped together due to limited observation-time and observed seroconversions. Each row refers to a separate covariate in a single regression model containing the relationship age-disparity in years if the observation time was in the year of interest, and zero otherwise. The regression model also contains age of respondent (linear, quadratic and cubic terms centred at age 15) and all socio-demographic and behavioural covariates from the main analysis.
Much of the debate regarding age-disparate relationships and HIV has focused on the subset which is also extra-marital and feature economic disparities and transfers of cash or in-kind gifts from the men, i.e. “sugar daddies”, to the woman.7,12,16,32,33 Since we do not have information on the male partner’s economic situation, or on whether relationships involved the transfer of money or in-kind gifts, we cannot directly address whether economically asymmetrical, age-disparate relationships specifically are a risk factor for HIV. “Sugar daddy” relationships are sometimes conceived as fulfilling basic needs, such as providing funds for food, shelter or school uniforms, in which case might expect the effects of age-disparities to be greatest for those women who are least educated and poorest, and thus in greatest need of material support and least able to refuse older men’s advances. However, “sugar daddies” have also been described amongst women of high socioeconomic status, who gain social standing or luxury goods from their relationships.12,34,35 Our finding of no effect-modification by educational attainment, and reduced risk of HIV related to age-disparities for the poorest women, suggests that socio-economic inequalities are unlikely to be playing a substantial role in causing HIV acquisition risk in age-disparate relationships in this community.
One concern with our findings might be how the sex-specific HIV prevalence pattern, peaking for women several years before men, could be generated other than through age-disparate relationships. While this study cannot answer this empirical question, we note that while relationships with large age-disparities are sufficient to propagate an epidemic through generations, they are not necessary. As recently shown in a theoretical modelling study,22 it is also possible to maintain a severe HIV epidemic through multiple relationships with relatively small age-disparities, or through a core population of high-risk young men having multiple peer-aged relationships.
This study has several strengths, notably the longitudinal nature of the data, collected over almost a decade, and the rich set of covariates, allowing us to rule out many confounding and reverse causation relationships. One benefit of this longitudinal data is that we can capture the exposure of partner age-disparity in a time-varying manner, updating each woman’s age-disparity information as her sexual relationships change. We also allow for time-varying socio-demographic variables – marital status, educational attainment and household wealth – to confound or effect-modify any age-disparity effect, and account for any unmeasured, time-varying confounders through the inclusion of year of observation indicator variables. This ensures that the results shown could not have been driven by the differential availability of HIV treatment and prevention interventions over time. Furthermore, the data constitute one of the largest HIV incidence cohorts in young women in Africa, providing very high power to detect significant effects.
We also note some limitations. As with any long-term community-based study, the cohort suffered from attrition and non-response. However, non-response was limited and our results did not change after accounting for data missingness through multiple imputation. Additionally, relationship age-disparities are reported by women. Evidence from Malawi suggests that women may underestimate their partner’s age in relationships with large age-disparities, which would lead to an attenuation of any true effect.36 However, in order to generate the exactly null finding we present here would require women to differentially underestimate the age of men from whom they acquired HIV, relative to non-infecting men. We are unaware of a mechanism that would generate such a bias, especially since women will often not know the HIV status of their partner.
Caution is needed in generalizing our findings. Age-disparate relationships may place young women at particularly high risk when they need economic support for survival.5,12 Such survival needs may be greater in other settings: while this setting is one of the poorest in South Africa, it is considerably wealthier than many high-prevalence regions of Africa. Additionally, this community contains relatively few older, rich men who might act as “sugar daddies”, since the community is relatively economically homogeneous, and HIV-related mortality over the past 20 years has substantially reduced the number of older men.37 The dense social networks in this area may also allow women to differentiate higher and lower risk older men more easily than in more urban or less settled areas, reducing the risk from age-disparate relationships.
In this analysis we have focused primarily on relationships between older men and younger women. This reflects the overwhelming proportion of relationships in this community (only 1.8% of baseline relationships were between older women and younger men (see Figure 1). Nevertheless, by using a continuous measure of the difference between male and female age as our primary exposure, we also include relationships in which the woman is older than the man, assuming that these relationships are increasingly less risky. This reflects the reality that over the age range we examine, male HIV prevalence is uniformly increasing with age – and thus younger men might be expected to be less risky.
However, an alternative hypothesis might be that any age-disparity (either older women or older men) will increase risk if risky behaviours arise in any unequal relationship and any age-disparate relationship contains power differentials. This might be particularly pertinent in settings where older women commonly partner with younger men. To test this hypothesis we conducted a sensitivity analysis replacing “age-of-male minus age-of-female” with the absolute difference in ages of the partners. The primary relationship remained null in this analysis: HR 1.01, 95% CI 0.98–1.03.
That age-disparate sexual relations are a primary driver of HIV incidence for young women in sub-Saharan Africa has often been taken as fact, despite limited evidence. Our analysis strongly suggests that in this rural South African community, sexual relationship age-disparities do not predict HIV risk for young women. Campaigns warning women about the risks of sexual partnerships with older men may well provide social benefits, particularly if they reduce unwanted pregnancies and increase sexual risk awareness. However, further research into how the relationship between age-disparities and HIV risk varies by geographic and social context is needed to justify the continuation of such campaigns. If replicated, our findings suggest that investing in this area specifically to reduce HIV incidence is unlikely to be an optimal use of scarce HIV prevention resources.
Supplementary Material
Acknowledgments
This analysis is based on data collected as part of the Africa Centre Demographic Information System and would not have been possible without the kind contributions of all respondents and the support of many staff at the Africa Centre for Health and Population Studies, for which the authors are extremely grateful.
Sources of Funding: GH acknowledges support from the Harvard University Committee on African Studies for travel to the research site for this study. The Wellcome Trust, UK, provides core funding to the Africa Centre, including for the surveillance on which this work is based (grant 082384/Z/07/Z). TB and FT received financial support through grant 1R01-HD058482-01 from the National Institute of Child Health and Human Development, National Institutes of Health. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnotes
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- 1.WHO. National AIDS programmes: a guide to indicators for monitoring and evaluating national HIV/AIDS prevention programmes for young people. Geneva, Switzerland: World Health Organization; 2004. [Google Scholar]
- 2.Wellings K, Collumbien M, Slaymaker E, et al. Sexual behaviour in context: a global perspective. Lancet. 2006;368(9548):1706–1728. doi: 10.1016/S0140-6736(06)69479-8. [DOI] [PubMed] [Google Scholar]
- 3.UNAIDS. 2012 Report on the Global AIDS Epidemic. Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS; 2012. [Google Scholar]
- 4.Chapman R, White RG, Shafer LA, et al. Do behavioural differences help to explain variations in HIV prevalence in adolescents in sub-Saharan Africa? Trop Med Int Health. 2010;15(5):554–566. doi: 10.1111/j.1365-3156.2010.02483.x. [DOI] [PubMed] [Google Scholar]
- 5.Hope R. Addressing Cross-Generational Sex: A desk review of research and programs. Washington, DC: Population Reference Bureau; 2007. [Google Scholar]
- 6.PSI. [Accessed February 4, 2014.];Cross-Generational Sex. http://www.psi.org/our-work/healthy-lives/interventions/cross-generational-sex.
- 7.KwaZulu-Natal Department of Health. [Accessed Febraury 4, 2014.];Sugar Daddy Campaign. 2012 http://www.kznhealth.gov.za/sugardaddy.htm.
- 8.Kelly RJ, Gray RH, Sewankambo NK, et al. Age differences in sexual partners and risk of HIV-1 infection in rural Uganda. J Acquir Immune Defic Syndr. 2003;32(4):446–451. doi: 10.1097/00126334-200304010-00016. [DOI] [PubMed] [Google Scholar]
- 9.Gregson S, Nyamukapa CA, Garnett GP, et al. Sexual mixing patterns and sex-differentials in teenage exposure to HIV infection in rural Zimbabwe. Lancet. 2002;359(9321):1896–1903. doi: 10.1016/S0140-6736(02)08780-9. [DOI] [PubMed] [Google Scholar]
- 10.Pettifor AE, Rees HV, Kleinschmidt I, et al. Young people’s sexual health in South Africa: HIV prevalence and sexual behaviors from a nationally representative household survey. AIDS. 2005;19(14):1525–1534. doi: 10.1097/01.aids.0000183129.16830.06. [DOI] [PubMed] [Google Scholar]
- 11.Kaiser R, Bunnell R, Hightower A, et al. Factors associated with HIV infection in married or cohabitating couples in Kenya: results from a nationally representative study. PLoS ONE. 2011;6(3):e17842. doi: 10.1371/journal.pone.0017842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Leclerc-Madlala S. Age-disparate and intergenerational sex in southern Africa: the dynamics of hypervulnerability. AIDS. 2008;22(Suppl 4):S17–25. doi: 10.1097/01.aids.0000341774.86500.53. [DOI] [PubMed] [Google Scholar]
- 13.Woolf SE, Maisto SA. Gender differences in condom use behavior? The role of power and partner-type. Sex Roles. 2008;9–10(58):689–701. [Google Scholar]
- 14.Wingood GM, DiClemente RJ. Application of the theory of gender and power to examine HIV-related exposures, risk factors, and effective interventions for women. Health Educ Behav. 2000;27(5):539–565. doi: 10.1177/109019810002700502. [DOI] [PubMed] [Google Scholar]
- 15.Bankole A, Biddlecom A, Guiella G, Singh S, Zulu E. Sexual behavior, knowledge and information sources of very young adolescents in four sub-Saharan African countries. Afr J Reprod Health. 2007;11(3):28–43. [PMC free article] [PubMed] [Google Scholar]
- 16.Luke N. Confronting the ‘sugar daddy’ stereotype: age and economic asymmetries and risky sexual behavior in urban Kenya. Int Fam Plan Perspect. 2005;31(1):6–14. doi: 10.1363/3100605. [DOI] [PubMed] [Google Scholar]
- 17.Bershteyn A, Klein DJ, Wenger E, Eckho PA. Description of the EMOD-HIV Model v0:7. arXiv.1206.3720v1. [Google Scholar]
- 18.Johnson LF, Hallett TB, Rehle TM, Dorrington RE. The effect of changes in condom usage and antiretroviral treatment coverage on human immunodeficiency virus incidence in South Africa: a model-based analysis. J R Soc Interface. 2012;9(72):1544–1554. doi: 10.1098/rsif.2011.0826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Korenromp EL, van Vliet C, Bakker R, de Vlas SJ, Habbema JDF. HIV spread and partnership reduction for different patterns of sexual behaviour - a study with the microsimulation model STDSIM. Math Popul Stud. 2000;8(2):135–173. [Google Scholar]
- 20.Garnett GP, Anderson RM. Factors controlling the spread of HIV in heterosexual communities in developing countries: patterns of mixing between different age and sexual activity classes. Philos Trans R Soc Lond B Biol Sci. 1993;342(1300):137–159. doi: 10.1098/rstb.1993.0143. [DOI] [PubMed] [Google Scholar]
- 21.Garnett GP, Anderson RM. Sexually transmitted diseases and sexual behavior: insights from mathematical models. J Infect Dis. 1996;174(Suppl 2):S150–161. doi: 10.1093/infdis/174.supplement_2.s150. [DOI] [PubMed] [Google Scholar]
- 22.Hallett TB, Gregson S, Lewis JJC, Lopman BA, Garnett GP. Behaviour change in generalised HIV epidemics: impact of reducing cross-generational sex and delaying age at sexual debut. Sex Transm Infect. 2007;83(Suppl 1):i50–54. doi: 10.1136/sti.2006.023606. [DOI] [PubMed] [Google Scholar]
- 23.Bärnighausen T, Tanser F, Newell M-L. Lack of a decline in HIV incidence in a rural community with high HIV prevalence in South Africa, 2003–2007. AIDS Res Hum Retroviruses. 2009;25(4):405–409. doi: 10.1089/aid.2008.0211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Welz T, Hosegood V, Jaffar S, Bätzing-Feigenbaum J, Herbst K, Newell M-L. Continued very high prevalence of HIV infection in rural KwaZulu-Natal, South Africa: a population-based longitudinal study. AIDS. 2007;21(11):1467–1472. doi: 10.1097/QAD.0b013e3280ef6af2. [DOI] [PubMed] [Google Scholar]
- 25.Zaidi J, Grapsa E, Tanser F, Newell M-L, Bärnighausen T. Dramatic increase in HIV prevalence after scale-up of antiretroviral treatment. AIDS. 2013;27(10):2301–2305. doi: 10.1097/QAD.0b013e328362e832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tanser F, Hosegood V, Bärnighausen T, et al. Cohort Profile: Africa Centre Demographic Information System (ACDIS) and population-based HIV survey. Int J Epidemiol. 2008;37(5):956–962. doi: 10.1093/ije/dym211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Luke N. Age and economic asymmetries in the sexual relationships of adolescent girls in sub-Saharan Africa. Stud Fam Plann. 2003;34(2):67–86. doi: 10.1111/j.1728-4465.2003.00067.x. [DOI] [PubMed] [Google Scholar]
- 28.Baird SJ, Garfein RS, McIntosh CT, Ozler B. Effect of a cash transfer programme for schooling on prevalence of HIV and herpes simplex type 2 in Malawi: a cluster randomised trial. Lancet. 2012;379(9823):1320–1329. doi: 10.1016/S0140-6736(11)61709-1. [DOI] [PubMed] [Google Scholar]
- 29.Dupas P. Do Teenagers Respond to HIV Risk Information? Evidence from a Field Experiment in Kenya. Am Econ J Appl Econ. 2011;3(1):1–34. [Google Scholar]
- 30.Reynolds L, Cousins T, Newell ML, Imrie J. The social dynamics of consent and refusal in HIV surveillance in rural South Africa. Soc Sci Med. 2013;77:118–125. doi: 10.1016/j.socscimed.2012.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ardington C, Case A, Hosegood V. Labor supply responses to large social transfers: longitudinal evidence from South Africa. Am Econ J Appl Econ. 2009;1:22–48. doi: 10.1257/app.1.1.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wyrod R, Fritz K, Woelk G, et al. Beyond sugar daddies: intergenerational sex and AIDS in urban Zimbabwe. AIDS Behav. 2011;15(6):1275–1282. doi: 10.1007/s10461-010-9800-2. [DOI] [PubMed] [Google Scholar]
- 33.Ott MQ, Bärnighausen T, Tanser F, Lurie MN, Newell M-L. Age-gaps in sexual partnerships: seeing beyond ‘sugar daddies’. AIDS. 2011;25(6):861–863. doi: 10.1097/QAD.0b013e32834344c9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Masvawure T. ‘I just need to be flashy on campus’: female students and transactional sex at a university in Zimbabwe. Cult Health Sex. 2010;12(8):857–870. doi: 10.1080/13691050903471441. [DOI] [PubMed] [Google Scholar]
- 35.Nkosana J, Rosenthal D. The dynamics of intergenerational sexual relationships: the experience of schoolgirls in Botswana. Sex Health. 2007;4(3):181–187. doi: 10.1071/sh06070. [DOI] [PubMed] [Google Scholar]
- 36.Helleringer S, Kohler HP, Mkandawire J. Women underestimate the age of their partners during survey interviews: implications for HIV risk associated with age mixing in northern Malawi. Sex Transm Dis. 2011;38(11):1030–1035. doi: 10.1097/OLQ.0b013e318227a486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bor J, Herbst AJ, Newell M-L, Bärnighausen T. Increases in adult life expectancy in rural South Africa: valuing the scale-up of HIV treatment. Science. 2013;339(6122):961–965. doi: 10.1126/science.1230413. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.