Abstract
Aim
Clinical cardiovascular disease is a major risk factor for cognitive impairment and dementia. However, less is known about the association of subclinical myocardial damage with cognition and dementia. We sought to examine the associations of high-sensitivity cardiac troponin T (hs-cTnT) with cognition and dementia.
Methods and results
Cross-sectional analysis of cognition (baseline 1996–98) and prospective analysis of dementia (follow-up through 2010) in 9472 participants in the Atherosclerosis Risk in Communities study. High-sensitivity cardiac troponin T was measured using a novel highly sensitive assay with a lower limit of the blank of 3 ng/L. Cognitive function was assessed by three tests: the delayed word recall test (DWRT), the digit symbol substitution test (DSST), and the word fluency test (WFT). Dementia was defined using ICD-9 codes. Linear regression and Cox models were adjusted for traditional cardiovascular risk factors. The mean age of participants was 63 years, 59% were female, 21% were black, and 66% had hs-cTnT ≥3 ng/L. In cross-sectional analyses, higher hs-cTnT was associated with lower scores on the DSST (P-trend < 0.001) and the WFT (P-trend = 0.002), but not on the DWRT (P-trend = 0.089). Over a median of 13 years, there were 455 incident dementia hospitalizations. In prospective analyses, higher baseline concentrations of hs-cTnT were associated with an increased risk for dementia hospitalizations overall (P-trend < 0.001) and for vascular dementia (P-trend = 0.029), but not for Alzheimer's dementia (P-trend = 0.212).
Conclusion
Elevations in baseline concentrations of hs-cTnT were associated with lower cognitive test scores at baseline and increased dementia hospitalization risk during the follow-up. Our results suggest that subclinical myocardial injury is associated with cognition and dementia.
Keywords: High-sensitivity Troponin T, Cognition, Dementia
See page 1779 for the editorial comment on this article (doi:10.1093/eurheartj/ehu198)
Introduction
Cognitive impairment and dementia affect ∼10 and 14% of elderly persons in the USA, respectively,1,2 and the number of persons with cognitive impairment and dementia are expected to triple by the year 2030.3 A history of clinical cardiovascular disease has been shown to be a major risk factor for cognitive impairment and dementia,4–6 but the association of subclinical myocardial injury with cognition and dementia has not been fully characterized.
Novel highly sensitive assays for cardiac troponin can detect troponin concentrations ∼10 times lower than the standard cardiac troponin assays.7 These novel highly sensitive assays are approved and recommended for clinical use in parts of Europe,8 but are not yet approved for clinical use in the USA. These highly sensitive assays are also of increasing interest for risk stratification purposes. Troponin concentrations measured using highly sensitive assays have been shown to improve the prediction of coronary heart disease and mortality in community-based populations without clinically evident cardiovascular disease.9,10 It has been hypothesized that hs-cTnT is a novel marker for subclinical myocardial injury,11,12 but the full-clinical significance of minor elevations in circulating troponin is unclear. For example, it has also been suggested that minute elevations in hs-cTnT may be a marker for diffuse subclinical small vessel disease rather than specific for myocardial damage.13 It is possible that subclinical small vessel disease in the brain may contribute to associations of hs-cTnT with cognition and dementia. Alternatively, elevations in hs-cTnT may reflect subclinical ischaemia due to atherosclerosis. Subclinical atherosclerosis (e.g. elevated carotid intima medial thickness) has previously been associated with cognitive impairment and dementia risk.14 Recent studies have shown that hs-cTnT is positively associated with risk of incident stroke, especially cardioembolic stroke.15,16 However, there are scant data on the possible association of hs-cTnT with cognitive function and risk of dementia.
We sought to characterize the relationship of hs-cTnT with cognitive function and future dementia risk in a community-based population. We hypothesized that higher concentrations of hs-cTnT would be associated cross-sectionally with lower cognitive test scores and prospectively with an increased risk of incident hospitalization with an ICD-9 code for dementia.
Methods
Study population
The Atherosclerosis Risk in Communities (ARIC) study is an ongoing, community-based prospective cohort of 15 792 middle-aged adults from four US communities: Washington County, MD; Forsyth County, NC; suburbs of Minneapolis, MN; and Jackson, MS. The ARIC study participants were initially seen at four in-person visits which occurred ∼3 years apart, from 1987–89 for visit 1, to 1996–98 for visit 4. A fifth visit was conducted from 2011 to 2013. High-sensitivity cardiac troponin T was measured in blood samples from ARIC participants who attended visit 4 (baseline for the present study). Of the 11 625 black and white participants who attended visit 4, we excluded those with a history of coronary heart disease (1151), myocardial infarction (n = 134), heart failure (n = 100), or stroke (n = 100) at or before visit 4, those with a hospitalization with an ICD-9 code for dementia before visit 4 (n = 25), those missing cognitive test (n = 246) or hs-cTnT data (n = 339), and those missing covariates included in statistical models (n = 60), leaving 9472 participants included in the present analysis.
The ARIC study has been approved by the Institutional Review Boards of all participating institutions, including the University of Minnesota, Johns Hopkins University, University of North Carolina, and University of Mississippi Medical Center. All the participants gave written informed consent at each study visit.
Measurement of high-sensitivity cardiac troponin T
High-sensitivity cardiac troponin T was measured in 2010–11 from plasma samples that were stored at –70°C since collection during visit 4 (1996–98). The Elecys troponin T, a novel high-sensitivity assay, was performed using a Cobas e411 analyzer (Roche Diagnostics, Indianapolis, IN). As reported by the manufacturer, this assay has a limit of blank of 3 ng/L and a limit of detection (LOD) of 5 ng/L.9,17 The reliability coefficient was 0.99 and the inter-assay coefficient of variation was 15%.18 Our samples were analysed using a reagent lot numbers that were not impacted by a calibration issue disclosed by Roche.
Measures of cognitive function
Cognitive functioning was assessed at visit 4 (1996–98) using three standardized tests: the delayed word recall test (DWRT),19 the digit symbol substitution test (DSST) of the Wechsler adult intelligence scale-revised (WAIS-R),20 and the word fluency test (WFT), also known as the controlled oral word association test of the multilingual aphasia examination.21 Protocols for the tests were standardized, and trained examiners administered the cognitive tests in a fixed order during one session in a quiet room. Examiner performance was monitored by audio tape recording, and recordings were reviewed locally and shared across centres to ensure consistency with testing procedures.
The DWRT is a test of verbal learning and recent memory. In this test, participants were given 10 common nouns that they were asked to learn by using each word in one or two sentences. After a 5-min delay, participants were given 60-s to recall the words. The score for the DWRT is the number of words correctly recalled.
The DSST is a test of executive function and processing speed, where participants were asked to translate numbers to symbols using a key. The score is the total number of numbers correctly translated to symbols within 90-s and the range of possible scores is 0–93.
The WFT is a test of executive function and language, and tests the ability to spontaneously generate words beginning with a particular letter, excluding proper names or places. Participants were given 60-s for each of three trials for the letters ‘F’, ‘A’, and ‘S’. The word fluency score is the total number of words generated across the three trials.
In addition to the raw cognitive scores, we calculated standardized z-scores for each test by subtracting the test mean and dividing by its standard deviation; means and standard deviations were calculated separately by race. We then calculated a composite global score by calculating the mean of the individual z-scores. We standardized the global score by subtracting the average global mean and dividing by its standard deviation.
Incident hospitalization with an ICD-9 code for dementia
The ARIC study obtains hospitalization information from annual telephone contact with study participants and through active surveillance of all hospitalizations in the study communities. For the present study, follow-up was available through 31 December 2010. We defined time to first hospitalization with an ICD-9 code for dementia using the following ICD-9 codes (listed anywhere in the hospital discharge record): Alzheimer's disease (331.0), vascular dementia (290.4), or dementia of other aetiology (290.0, 290.1, 290.2, 290.3, 290.9, 294.1, 294.2, 294.8, 294.9, 331.1, 331.2, 331.8, and 331.9). This definition for incident hospitalization with an ICD-9 code for dementia has been used previously,22,23 and scores on the DWRT, DSST, and WFT have previously been shown to be associated with this definition.23
Covariates
All covariates used in the regression models were assessed at visit 4 (1996–98), except education, which was assessed at visit 1 (1987–89). Covariates included age (years), gender, race/centre (Maryland whites; Minnesota whites; North Carolina whites; North Carolina blacks; and Mississippi blacks), education (<high school; high school or equivalent; college, graduate or professional school), income (<$35 000/year; ≥$35 000/year; not reported), body mass index (kg/m2), physical activity (score range 1–5, based on replies to questions on type, frequency, intensity, and duration of physical activity from a modified Baecke Physical Activity questionnaire24,25), alcohol consumption [current high (>7 drinks/week for women; >14 drinks/week for men); current low (≤7 drinks/week for women; ≤14 drinks/week for men); former; never], cigarette smoking (current; former; never), total and HDL cholesterol (mg/dL), diabetes (self-report, medication use, or fasting blood glucose ≥126 mg/dL), systolic and diastolic blood pressure (mmHg, defined as the mean of the second and third of three measurements), and use of hypertension medication.
Statistical analysis
We analysed hs-cTnT both continuously and categorically. For the categorical analysis, our reference group was the 34% of the ARIC population with hs-cTnT concentrations <3 ng/L. The remaining population was divided into categories as follows (based on prior publications9,10): 3–5 ng/L, 6–8 ng/L, 9–13 ng/L, and ≥14 ng/L. In a sensitivity analysis, we repeated our analyses using <5 ng/L (LOD) as the reference group. Baseline characteristics of our population are shown by hs-cTnT category and are adjusted for age and sex due to the differences in hs-cTnT distribution by these factors.9,26 We also consider two binary definitions of hs-cTnT: ≥3 vs. 3 ng/L, elevated (≥14 ng/L) vs. non-elevated (<14 ng/L).
Linear regression models were used to assess the cross-sectional association between hs-cTnT and cognitive test scores. For the prospective association between hs-cTnT and incident hospitalization with an ICD-9 code for dementia, we estimated hazard ratios (95% confidence intervals) using Cox proportional-hazard models. The proportional-hazards assumptions were checked with the use of Schoenfeld residuals and graphic methods. We tested for linear trend across the median of hs-cTnT concentration categories. We also modelled the association of hs-cTnT with incident dementia using a restricted cubic spline model with knots at the 10th, 50th, and 90th percentiles. We performed the spline analyses among those with hs-cTnT concentrations within the measuring range only (n = 6221). Models were adjusted for demographic factors (age, race/field centre, sex, education level, and income), lifestyle/behavioural factors (body mass index, physical activity, alcohol consumption, and smoking), and cardiovascular risk factors (total cholesterol, HDL cholesterol, diabetes, systolic and diastolic blood pressure, and blood pressure-lowering medication). We formally tested for interaction by gender. We calculated Harrell's C-statistic to assess the discriminatory ability of our models.27
In exploratory analyses, we used Cox proportional-hazards models to examine the prospective association between hs-cTnT and two subcategories of incident hospitalization with an ICD-9 code for dementia: incident Alzheimer's dementia (n = 112) and incident vascular dementia (n = 34).
Associations between hs-cTnT and hospitalization with an ICD-9 diagnosis of dementia may reflect differences in dementia or may reflect differences in hospitalization rates, as persons without a hospitalization could not receive a diagnosis of dementia. Therefore, we performed a sensitivity analysis where we restricted our analysis to those individuals who had at least one hospitalization during the follow-up (n = 7395).
We also examined the past trajectory of cognitive function by examining the association of hs-cTnT with the 6-year change in cognitive test scores from visit 2 (1990–92) to visit 4 (1996–98) among the subset of participants who had cognitive function measured at both visits (n = 9311). This past trajectory of cognitive function change analysis is less likely to be biased by cultural confounding factors and less affected by measurement error compared with our main cross-sectional analysis.28 Therefore, we performed this analysis to attempt to account for potential residual confounding and measurement error that may be present in our cross-sectional analyses.
We also performed analyses excluding participants with atrial fibrillation or atrial flutter at or before visit 4 (n = 143). Atrial fibrillation diagnoses were obtained from ECGs performed at all study visits and hospital discharge records (ICD-9 codes: 427.31, 427.32). In our prospective analysis, we conducted additional analyses censoring participants who had a diagnosis of atrial fibrillation or atrial flutter prior to a dementia diagnosis (n = 37), stroke occurring prior to a dementia diagnosis (n = 47), or heart failure occurring prior to a dementia diagnosis (n = 60). We performed two additional analyses where we adjusted for left ventricular hypertrophy (defined using Cornell voltage criteria,29 n = 9472) and where we adjusted for carotid intima media thickness (n = 5498) to assess for possible mediation.
All reported P-values are two-sided and P < 0.05 was considered statistically significant. All analyses were performed using Stata Version 12 (StataCorp, College Station, TX, USA).
Results
Baseline (1996–98) characteristics of the study population overall and by hs-cTnT categories (<3, 3–5, 6–8 , 9–13, and ≥14 ng/L) are shown in Table 1. The mean age of participants was 63 years old, 59% of participants were female, 21% of participants were black, and 66% of participants had hs-cTnT concentrations within the measuring range. Compared with those with hs-cTnT ≥14 ng/L (elevated), those with hs-cTnT <3 ng/L (below the measuring range) were younger (61 vs. 65 years, P < 0.001) and were more likely to be female (79 vs. 25%, P < 0.001). After adjustment for age and sex, those with hs-cTnT <3 ng/L (compared with those with hs-cTnT ≥14 ng/L) were less likely to be black (18 vs. 31%, P < 0.001), were less likely to have less than high school education (16 vs. 21%, P = 0.002), and were less likely have diabetes (9 vs. 35%, P < 0.001). Characteristics of the study population using the categories of <5, 5–8, 9–13, and ≥14 are shown in Supplementary material online, Table S1.
Table 1.
Baseline characteristics (1996–98) by category of high-sensitivity cardiac troponin T adjusted for age and sex
| All participants (n = 9472) | <3 ng/L (n = 3251) | 3–5 ng/L (n = 2452) | 6–8 ng/L (n = 1926) | 9–13 ng/L (n = 1207) | ≥14 ng/L (elevated) (n = 636) | P-value <3 ng/L vs. ≥14 ng/L | |
|---|---|---|---|---|---|---|---|
| Age (years), mean | 62.5 | 60.7 | 62.3 | 63.5 | 64.9 | 65.2 | <0.001 |
| Female, % | 59.3 | 79.4 | 62.4 | 47.4 | 36.5 | 24.7 | <0.001 |
| Black, % | 20.8 | 17.5 | 18.0 | 22.9 | 26.6 | 31.3 | <0.001 |
| Education, % | |||||||
| <High school | 17.1 | 15.7 | 16.7 | 17.1 | 19.3 | 21.1 | 0.002 |
| High school or vocational school | 42.6 | 44.0 | 42.8 | 42.5 | 40.9 | 38.5 | 0.002 |
| College, graduate, or professional school | 40.3 | 40.3 | 40.5 | 40.4 | 39.7 | 40.4 | 0.951 |
| Income, % | |||||||
| <$35 000/year | 46.8 | 45.2 | 46.3 | 46.5 | 49.7 | 52.0 | 0.002 |
| ≥$35 000/year | 47.9 | 49.0 | 49.7 | 48.4 | 44.0 | 41.4 | 0.001 |
| Not reported | 5.3 | 5.8 | 4.0 | 5.1 | 6.3 | 6.6 | 0.448 |
| Cigarette smoking status, % | |||||||
| Current smoker | 14.4 | 20.8 | 12.8 | 9.6 | 9.9 | 11.1 | <0.001 |
| Former smoker | 42.0 | 41.3 | 42.1 | 42.5 | 43.1 | 41.8 | 0.794 |
| Never smoker | 43.6 | 37.9 | 45.1 | 47.8 | 47.0 | 47.1 | <0.001 |
| Alcohol use status, % | |||||||
| Current alcohol use—high a | 39.8 | 41.8 | 42.8 | 38.0 | 35.2 | 31.8 | <0.001 |
| Current alcohol use—low b | 11.2 | 13.7 | 10.8 | 9.9 | 8.8 | 8.7 | 0.006 |
| Former alcohol use | 28.3 | 27.2 | 26.3 | 29.1 | 29.6 | 36.7 | <0.001 |
| Never alcohol use | 20.7 | 17.3 | 20.0 | 23.1 | 26.4 | 22.8 | 0.002 |
| Physical activity index, mean | 2.5 | 2.5 | 2.5 | 2.5 | 2.4 | 2.3 | <0.001 |
| Body mass index (kg/m2), mean | 28.7 | 27.7 | 28.5 | 29.4 | 30.0 | 30.5 | <0.001 |
| Total cholesterol (mg/dL), mean | 202 | 204 | 202 | 201 | 200 | 197 | <0.001 |
| HDL cholesterol (mg/dL), mean | 50.9 | 51.7 | 51.1 | 50.4 | 49.7 | 49.1 | 0.002 |
| Diabetes, % | 14.6 | 9.00 | 11.8 | 16.8 | 21.4 | 34.8 | <0.001 |
| Systolic BP (mmHg), mean | 127 | 125 | 126 | 128 | 131 | 132 | <0.001 |
| Diastolic BP (mmHg), mean | 71.1 | 70.4 | 70.9 | 71.6 | 72.1 | 71.8 | 0.033 |
| Hypertension medication, % | 38.5 | 31.5 | 35.1 | 42.3 | 47.3 | 59.5 | <0.001 |
| Delayed word recall test (words), mean | 6.6 | 6.7 | 6.7 | 6.6 | 6.6 | 6.4 | <0.001 |
| Digit symbol substitution test (points), mean | 44.4 | 45.3 | 45.3 | 44.0 | 42.8 | 40.4 | <0.001 |
| Word fluency test (words), mean | 38.9 | 34.9 | 34.0 | 33.3 | 33.1 | 31.5 | <0.001 |
| Global z-score, mean | 0.00 | 0.05 | 0.03 | −0.03 | −0.05 | −0.18 | <0.001 |
aCurrent high alcohol consumption was defined as >7 drinks/week for women and >14 drinks/week for men.
bCurrent low alcohol consumption was defined as ≤7 drinks/week for women and ≤14 drinks/week for men.
Table 2 shows β-coefficients (95% confidence intervals) for the cross-sectional association of hs-cTnT with cognitive tests scores. Higher concentrations of hs-cTnT were associated with lower cognitive test scores on the DSST (P-trend < 0.001) and WFT (P-trend = 0.002), but not on the DWRT (P-trend = 0.089). Compared with participants with hs-cTnT concentrations <3 ng/L, participants with hs-cTnT concentrations ≥14 ng/L translated 1.83 (95% CI: 0.96, 2.71) fewer numbers to symbols on the DSST and generated 1.63 (95% CI: 0.62, 2.64) fewer words on the WFT. Higher concentrations of hs-cTnT were also associated with a lower global z-score (P-trend < 0.001). Results of analyses using our binary definitions of hs-cTnT ≥3 vs. 3 ng/L and ≥14 vs. <14 ng/L were similar to our main categorical analyses. We found no evidence of interaction by gender for DWRT and DSST scores (P-for-interaction = 0.625 and 0.132, respectively). Associations of hs-cTnT with lower WFT score tended to be stronger for women than men (P-for-interaction = 0.007). Similar results were found when the analyses were repeated using <5 ng/L as the reference group (Supplementary material online, Table S2).
Table 2.
Adjusted β-coefficients (95% confidence intervals) for the cross-sectional associations of categories of high-sensitivity cardiac troponin T with cognitive test scores
| Categories of hs-cTnT | Delayed word recall test | Digit symbol substitution test | Word fluency test | Global Z-score |
|---|---|---|---|---|
| hs-cTnT <3 ng/L | Reference | Reference | Reference | Reference |
| hs-cTnT 3–5 ng/L | 0.03 (−0.05, 0.11) | −0.04 (−0.55, 0.47) | −0.80 (−1.38, −0.21) | −0.02 (−0.06, 0.02) |
| hs-cTnT 6–8 ng/L | −0.06 (−0.15, 0.02) | −0.50 (−1.06, 0.07) | −1.10 (−1.76, −0.45) | −0.08 (−0.12, −0.03) |
| hs-cTnT 9–13 ng/L | −0.02 (−0.12, 0.08) | −0.64 (−1.33, 0.04) | −0.59 (−1.38, 0.19) | −0.05 (−0.11, 0.01) |
| hs-cTnT ≥14 ng/L | −0.10 (−0.23, 0.04) | −1.83 (−2.71, −0.96) | −1.63 (−2.64, −0.62) | −0.14 (−0.22, −0.07) |
| P-value for trend | 0.089 | <0.001 | 0.002 | <0.001 |
Model is adjusted for age (continuous), race/field centre (Washington County, Maryland whites; suburbs of Minneapolis, Minnesota whites; Forsyth County, North Carolina whites; Forsyth County, North Carolina blacks; Jackson, Mississippi blacks), sex (male; female), education level (<high school; high school or equivalent; college, graduate or professional school), income (<$35 000/year; ≥$35 000/year; not reported), body mass index (continuous), physical activity (continuous), alcohol consumption (current high; current low; former; never), smoking (current; former; never), total cholesterol (continuous), HDL cholesterol (continuous), diabetes (yes; no), systolic and diastolic blood pressure (continuous), and blood pressure-lowering medication (yes; no).
Bolded data represent P<0.05.
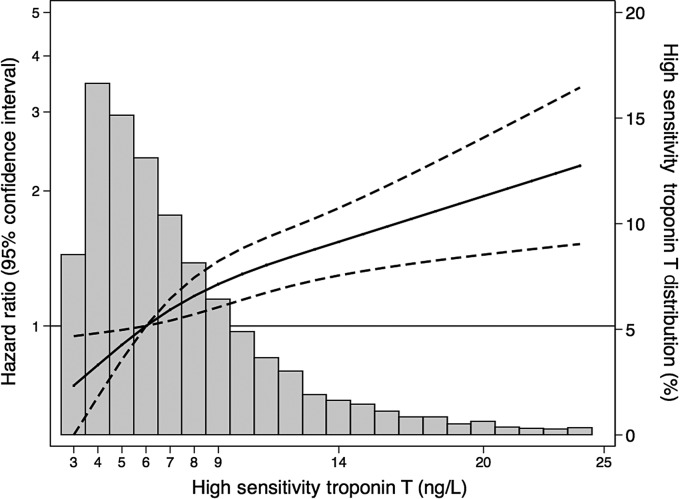
Among our population of 9472 persons without a history of cardiovascular disease at baseline, there were 455 incident hospitalizations with an ICD-9 code for dementia that occurred during a median of 13 years of follow-up. Table 3 shows the hazard ratios (95% confidence intervals) for the association of hs-cTnT at baseline with risk for incident hospitalization with an ICD-9 code for dementia. Higher concentrations of hs-cTnT were associated with an increased risk for incident hospitalization with an ICD-9 code for dementia (P-trend < 0.001). Compared with participants with hs-cTnT concentrations <3 ng/L, participants with hs-cTnT concentrations between 6 and 8 ng/L had 1.43 (95% CI: 1.07, 1.91) times increased risk, participants with hs-cTnT concentrations between 9 and 13 ng/L had 1.82 (95% CI: 1.33, 2.48) times increased risk, and participants with hs-cTnT concentrations ≥14 ng/L had 2.69 (95% CI: 1.87, 3.84) times increased risk for hospitalization with an ICD-9 code for dementia during the follow-up. Results of analyses using our binary definitions of hs-cTnT ≥3 vs. 3 ng/L and ≥14 vs. <14 ng/L were similar to our main categorical analyses. We found no evidence of interaction by gender (P-for-interaction = 0.240). Results from our sensitivity analysis restricted to individuals who had at least one hospitalization over follow-up (n = 7395) were slightly attenuated, but remained statistically significant (P-trend <0.001). Comparing models with and without hs-cTnT, Harrell's C-statistics were 0.798 and 0.790, respectively (P-for-difference = 0.004), indicating that the addition of hs-cTnT to our model improves the discriminatory ability of our model. Similar results were found when the analysis was repeated using <5 ng/L as the reference group (Supplementary material online, Table S3). Figure 1 shows the continuous association of hs-cTnT with incident dementia-related hospitalization among the 6321 participants with hs-cTnT concentrations ≥3 ng/L using a restricted cubic spline model. Similar results were seen among the 4690 participants with hs-cTnT concentrations ≥5 ng/L (Supplementary material online, Figure S1).
Table 3.
Adjusted hazard ratios (95% confidence intervals) for the prospective association between categories of baseline high-sensitivity cardiac troponin T and incident dementia hospitalization
| Category of hs-cTnT | Hazard ratio (95% CI) |
|---|---|
| hs-cTnT <3 ng/L | 1.00 (reference) |
| hs-cTnT 3–5 ng/L | 1.09 (0.82, 1.45) |
| hs-cTnT 6–8 ng/L | 1.43 (1.07, 1.91) |
| hs-cTnT 9–13 ng/L | 1.82 (1.33, 2.48) |
| hs-cTnT ≥14 ng/L | 2.68 (1.87, 3.84) |
| P-value for trend | <0.001 |
Model is adjusted for age (continuous), race/field centre (Washington County, Maryland whites; suburbs of Minneapolis, Minnesota whites; Forsyth County, North Carolina whites; Forsyth County, North Carolina blacks; Jackson, Mississippi blacks), sex (male; female), education level (<high school; high school or equivalent; college, graduate or professional school), income (<$35 000/year; ≥$35 000/year; not reported), body mass index (continuous), physical activity index (continuous), alcohol consumption (current high; current low; former; never), smoking (current; former; never), total cholesterol (continuous), HDL cholesterol (continuous), diabetes (yes; no), systolic and diastolic blood pressure (continuous), and blood pressure-lowering medication (yes; no).
Bolded data represent P < 0.05.
Figure 1.
Adjusted restricted cubic spline model showing the hazard ratios (95% confidence intervals) for the association of high-sensitivity cardiac troponin T with incident dementia [among the 6321 participants with high-sensitivity cardiac troponin T concentrations within the measuring range (≥3 ng/L)]. Knots at 4, 6, and 14 ng/L (10th, 50th, and 90th percentiles). Histogram shows the distribution of concentrations of high-sensitivity cardiac troponin T within the measuring range. Data truncated at 97.5 percentile of high-sensitivity cardiac troponin T. Model is adjusted for age (continuous), race/field centre (Washington County, Maryland whites; suburbs of Minneapolis, Minnesota whites; Forsyth County, North Carolina whites; Forsyth County, North Carolina blacks; Jackson, Mississippi blacks), sex (male; female), education level (<high school; high school or equivalent; college, graduate or professional school), income (<$35 000/year; ≥$35 000/year; not reported), body mass index (continuous), physical activity (continuous), alcohol consumption (current high; current low; former; never), smoking (current; former; never), total cholesterol (continuous), HDL cholesterol (continuous), diabetes (yes; no), systolic and diastolic blood pressure (continuous), and blood pressure-lowering medication (yes; no).
In our exploratory analysis examining the association of hs-cTnT with two subcategories of incident hospitalization with an ICD-9 code for dementia, there were 112 incident cases of hospitalization with an ICD-9 code for Alzheimer's dementia and 34 incident cases of hospitalization with an ICD-9 code for vascular dementia. Three individuals who had ICD-9 codes for both Alzheimer's dementia and vascular dementia. High-sensitivity cardiac troponin T was not associated with incident hospitalization with an ICD-9 code for Alzheimer's dementia (P-trend = 0.212), but higher concentrations of hs-cTnT were significantly associated with incident hospitalization with an ICD-9 code for vascular dementia (P-trend = 0.029).
In our analysis examining the past trajectory of cognitive function (n = 9311), higher concentrations of hs-cTnT remained significantly associated with more cognitive decline over the prior 6 years on the DSST (P-trend = 0.001) and WFT (P-trend = 0.027), but not on the DWRT (P-trend = 0.753).
In the analyses excluding participants with atrial fibrillation or atrial flutter at or before visit 4 (n = 9331 included in this analysis), higher concentrations of hs-cTnT remained significantly associated with lower DSST (P-trend < 0.001) and WFT (P-trend = 0.003) scores. In the analyses censoring participants who had a diagnosis of atrial fibrillation or atrial flutter, stroke, or heart failure occurring prior to a dementia diagnosis, the association of hs-cTnT with risk for dementia was slightly attenuated but remained statistically significant (Supplementary material online, Table S4). Similarly, when left ventricular hypertrophy (n = 9472) and carotid intima media thickness (n = 5498) were added to the model, the results were not appreciably altered.
Discussion
In this large, community-based population without coronary heart disease, myocardial infarction, heart failure, or stroke at baseline, elevations in cardiac troponin T detected using a highly sensitive assay were cross-sectionally associated with lower scores on the DSST and the WFT, as well as with lower global z-scores, independent of traditional risk factors. High-sensitivity cardiac troponin T concentrations were not associated with DWRT scores. Elevations in cardiac troponin T were also significantly associated with an increased risk of incident hospitalization with an ICD-9 code for dementia, particularly incident hospitalization with an ICD-9 code for vascular dementia, independent of traditional risk factors.
Potential mechanisms whereby hs-cTnT could contribute to cognitive dysfunction and dementia include shared risk factors,30 subclinical or clinical cardioembolic stroke,15,16 hypoperfusion from inadequate left ventricular function,9,10,31,32 or subclinical ischaemia due to atherosclerosis.14 Although it remains unclear whether hs-cTnT is a marker of subclinical small vessel disease,13 it has been hypothesized that the same persons with subclinical myocardial injury may also have subclinical small vessel disease in the brain as a result of shared risk factors.30 Though we tried to account for many of these factors in our analyses, residual confounding remains a possibility. The subclinical small vessel disease in the brain may manifest as subtle impairment on standardized cognitive tests and may place the person at a higher risk for dementia later in life.
The association of baseline hs-cTnT concentration with risk of incident hospitalization with an ICD-9 code for dementia remained after accounting for the intervening occurrence (mediation effect) of incident atrial fibrillation or atrial flutter, stroke, or heart failure prior to the hospitalization. Previous reports have suggested that hs-cTnT may be a marker of atrial fibrillation resulting from subclinical myocardial injury,33 which might lead to either clinical or subclinical cardioembolic stroke. Indeed, hs-cTnT been associated with cardioembolic stroke,15,16 and stroke is a known cause of cognitive impairment and dementia.34,35 High-sensitivity cardiac troponin T may also be a marker for subclinical myocardial damage causing inadequate left ventricular function, which may lead to hypoperfusion in the brain.9,10,31,32 Cerebral hypoperfusion has been noted to be associated with cognitive impairment and dementia.36,37 Alternatively, elevations in hs-cTnT may reflect subclinical ischaemia due to atherosclerosis, which has been associated with cognitive function.14 However, after adding left ventricular hypertrophy and carotid intima media thickness to our model to assess for possible mediation effects, our results remained significant, suggesting that these factors do not mediate the associations.
Our results suggest that hs-cTnT is more associated with vascular pathology in the brain than with Alzheimer's disease pathology. Higher concentrations of hs-cTnT were significantly associated with lower scores on the DSST and the WFT, but not on the DWRT. The DSST and WFT are tests of executive function that are more associated with vascular pathology and vascular dementia,34,38,39 whereas the DWRT is a test of memory and is more associated with Alzheimer's dementia.19 The results of our analyses of the subcategories of incident hospitalization with an ICD-9 code for subcategories of dementia also support this notion; higher concentrations of hs-cTnT were associated with an increased risk for incident hospitalization with an ICD-9 code for vascular dementia, but not for incident hospitalization with an ICD-9 code for Alzheimer's dementia.
Limitations of this study should be taken into consideration when interpreting the results. We used ICD-9 hospitalization codes to define incident dementia and we did not have information to determine whether dementia was the primary reason for hospitalization. Although hospitalization with an ICD-9 code for dementia is a relatively specific marker of dementia, it likely disproportionately occurs with the most severe cases of dementia and dementia in the presence of other diseases, suggesting that our results may be attenuated. A prior analysis of ARIC data22 showed that age-specific incidence rates of hospitalization with an ICD-9 code for dementia were lower than age-specific incidence rates of dementia in other studies.40,41 Additionally, in our main analysis, we had only single measurements of hs-cTnT and cognitive test performance, which have been shown to vary over time within individuals and can be subject to measurement error.42,43 However, the results of our sensitivity analysis examining the association of hs-cTnT with the past trajectory of 6-year change in cognitive test scores remained significant, suggesting that the cross-sectional association of hs-cTnT with cognition is not due to residual confounding or measurement errors.28 Our exploratory analysis of the subcategories of hospitalization with an ICD-9 code for dementia should be interpreted cautiously because we did not have the ability to validate the ICD-9 codes, leaving open the possibility of misclassification. The ability of ICD-9 codes to differentiate subtypes of dementia is limited; the majority of our dementia cases were identified by non-specific dementia ICD-9 codes, leaving small numbers of participants in these subcategory analyses.
Our study also had a number of important strengths, including the large sample size, a median of 13 years of follow-up, and comprehensive measurement of confounders. We also had data on three cognitive tests, which allowed us to investigate the association of hs-cTnT with different cognitive domains, as well as to investigate the association of hs-cTnT with a global cognitive assessment.
In conclusion, our study suggests that subclinical myocardial injury, as assessed by hs-cTnT, is associated with lower cognitive performance. Our results also suggest that higher concentrations of hs-cTnT are associated with an increased risk for incident hospitalization for dementia, particularly vascular dementia. These associations may be the result of shared risk factors for myocardial and cerebral injury or the result of subclinical small vessel disease affecting both the heart and the brain. Our results suggest that in addition to the prediction of hard cardiovascular events, minor elevations in cardiac troponin may provide risk information regarding dementia outcomes.
Supplementary material
Supplementary material is available at European Heart Journal online.
Funding
A.L.C.S and A.M.R were supported by NIH/NHLBI grant T32 HL007024. E.S was supported by NIH/NIDDK grant R01 DK089174. The Atherosclerosis Risk in Communities Study is carried out as a collaborative study supported by National Heart, Lung, and Blood Institute contracts (HHSN268201100005C, HHSN268201100006C, HHSN268201100007C, HHSN268201100008C, HHSN268201100009C, HHSN268201100010C, HHSN268201100011C, and HHSN268201100012C).
Conflict of interest: R.C.H and C.M.B have received grant support from Roche Diagnostics (and the NIH). The other authors declare no commercial conflicts of interest (but receive NIH grant funding). C.M.B and R.C.H. are co-investigators on a provisional patent filed by Roche for use of biomarkers in heart failure prediction. Roche Diagnostics provided reagents and loan of an instrument to conduct the highly sensitive cardiac troponin T assay. Roche had no role in design, analysis, or manuscript preparation.
Supplementary Material
Acknowledgements
The authors thank the staff and participants of the ARIC study for their important contributions.
References
- 1.DeCarli C. Mild cognitive impairment: prevalence, prognosis, aetiology, and treatment. Lancet Neurol. 2003;2:15–21. doi: 10.1016/s1474-4422(03)00262-x. [DOI] [PubMed] [Google Scholar]
- 2.Plassman BL, Langa KM, Fisher GG, Heeringa SG, Weir DR, Ofstedal MB, Burke JR, Hurd MD, Potter GG, Rodgers WL, Steffens DC, Willis RJ, Wallace RB. Prevalence of dementia in the United States: the aging, demographics, and memory study. Neuroepidemiology. 2007;29:125–132. doi: 10.1159/000109998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hebert LE, Scherr PA, Bienias JL, Bennett DA, Evans DA. Alzheimer disease in the US population: prevalence estimates using the 2000 census. Arch Neurol. 2003;60:1119–1122. doi: 10.1001/archneur.60.8.1119. [DOI] [PubMed] [Google Scholar]
- 4.Kovacic JC, Castellano JM, Fuster V. The links between complex coronary disease, cerebrovascular disease, and degenerative brain disease. Ann N Y Acad Sci. 2012;1254:99–105. doi: 10.1111/j.1749-6632.2012.06482.x. [DOI] [PubMed] [Google Scholar]
- 5.Emanuele E, Martinelli V, Abbiati V, Ricevuti G. Linking atherosclerosis to Alzheimer’s disease: focus on biomarkers. Front Biosci. 2012;4:700–710. doi: 10.2741/e411. [DOI] [PubMed] [Google Scholar]
- 6.Sahathevan R, Brodtmann A, Donnan GA. Dementia, stroke, and vascular risk factors: a review. Int J Stroke. 2012;7:61–73. doi: 10.1111/j.1747-4949.2011.00731.x. [DOI] [PubMed] [Google Scholar]
- 7.Tate JR. Troponin revisited 2008: assay performance. Clin Chem Lab Med. 2008;46:1489–1500. doi: 10.1515/CCLM.2008.292. [DOI] [PubMed] [Google Scholar]
- 8.Hamm CW, Bassand JP, Agewall S, Bax J, Boersma E, Bueno H, Caso P, Dudek D, Gielen S, Huber K, Ohman M, Petrie MC, Sonntag F, Uva MS, Storey RF, Wijns W, Zahger D Guidelines ESCCfP. ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: the Task Force for the management of acute coronary syndromes (ACS) in patients presenting without persistent ST-segment elevation of the European Society of Cardiology (ESC) Eur Heart J. 2011;32:2999–3054. doi: 10.1093/eurheartj/ehr236. [DOI] [PubMed] [Google Scholar]
- 9.Saunders JT, Nambi V, de Lemos JA, Chambless LE, Virani SS, Boerwinkle E, Hoogeveen RC, Liu X, Astor BC, Mosley TH, Folsom AR, Heiss G, Coresh J, Ballantyne CM. Cardiac troponin T measured by a highly sensitive assay predicts coronary heart disease, heart failure, and mortality in the Atherosclerosis Risk in Communities Study. Circulation. 2011;123:1367–1376. doi: 10.1161/CIRCULATIONAHA.110.005264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.de Lemos JA, Drazner MH, Omland T, Ayers CR, Khera A, Rohatgi A, Hashim I, Berry JD, Das SR, Morrow DA, McGuire DK. Association of troponin T detected with a highly sensitive assay and cardiac structure and mortality risk in the general population. JAMA. 2010;304:2503–2512. doi: 10.1001/jama.2010.1768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rubin J, Matsushita K, Ballantyne CM, Hoogeveen R, Coresh J, Selvin E. Chronic hyperglycemia and subclinical myocardial injury. J Am Coll Cardiol. 2012;59:484–489. doi: 10.1016/j.jacc.2011.10.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.de Lemos JA, Grundy SM. Low levels of circulating troponin as an intermediate phenotype in the pathway to heart failure. J Am Coll Cardiol. 2012;59:490–492. doi: 10.1016/j.jacc.2011.10.874. [DOI] [PubMed] [Google Scholar]
- 13.Moreno V, Hernandez-Romero D, Vilchez JA, Garcia-Honrubia A, Cambronero F, Casas T, Gonzalez J, Martinez P, Climent V, de la Morena G, Valdes M, Marin F. Serum levels of high-sensitivity troponin T: a novel marker for cardiac remodeling in hypertrophic cardiomyopathy. J Card Fail. 2010;16:950–956. doi: 10.1016/j.cardfail.2010.07.245. [DOI] [PubMed] [Google Scholar]
- 14.Zhong W, Cruickshanks KJ, Schubert CR, Acher CW, Carlsson CM, Klein BE, Klein R, Chappell RJ. Carotid atherosclerosis and 10-year changes in cognitive function. Atherosclerosis. 2012;224:506–510. doi: 10.1016/j.atherosclerosis.2012.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hijazi Z, Oldgren J, Andersson U, Connolly SJ, Ezekowitz MD, Hohnloser SH, Reilly PA, Vinereanu D, Siegbahn A, Yusuf S, Wallentin L. Cardiac biomarkers are associated with an increased risk of stroke and death in patients with atrial fibrillation: a Randomized Evaluation of Long-term Anticoagulation Therapy (RE-LY) substudy. Circulation. 2012;125:1605–1616. doi: 10.1161/CIRCULATIONAHA.111.038729. [DOI] [PubMed] [Google Scholar]
- 16.Folsom AR, Nambi V, Bell EJ, Oluleye OW, Gottesman RF, Lutsey PL, Huxley RR, Ballantyne CM. Troponin T, N-terminal pro-B-type natriuretic peptide, and incidence of stroke: the atherosclerosis risk in communities study. Stroke. 2013;44:961–967. doi: 10.1161/STROKEAHA.111.000173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Giannitsis E, Kurz K, Hallermayer K, Jarausch J, Jaffe AS, Katus HA. Analytical validation of a high-sensitivity cardiac troponin T assay. Clin Chem. 2010;56:254–261. doi: 10.1373/clinchem.2009.132654. [DOI] [PubMed] [Google Scholar]
- 18.Nambi V, Liu X, Chambless LE, de Lemos JA, Virani SS, Agarwal S, Boerwinkle E, Hoogeveen RC, Aguilar D, Astor BC, Srinivas PR, Deswal A, Mosley TH, Coresh J, Folsom AR, Heiss G, Ballantyne CM. Troponin T and N-terminal pro-B-type natriuretic peptide: a biomarker approach to predict heart failure risk: the atherosclerosis risk in communities study. Clin Chem. 2013;59:1802–1810. doi: 10.1373/clinchem.2013.203638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Knopman DS, Ryberg S. A verbal memory test with high predictive accuracy for dementia of the Alzheimer type. Arch Neurol. 1989;46:141–145. doi: 10.1001/archneur.1989.00520380041011. [DOI] [PubMed] [Google Scholar]
- 20.Wechsler D. Wechsler Adult Intelligence Scale Revised Manual. Psychological Corp: New York, NY; 1981. [Google Scholar]
- 21.Benton AL, Hamsher K. Multilingual Aphasia Examination. 2nd Edn. AJA Associates: Iowa City, IA; 1989. [Google Scholar]
- 22.Alonso A, Mosley TH, Jr, Gottesman RF, Catellier D, Sharrett AR, Coresh J. Risk of dementia hospitalisation associated with cardiovascular risk factors in midlife and older age: the Atherosclerosis Risk in Communities (ARIC) study. J Neurol Neurosurg Psychiatry. 2009;80:1194–1201. doi: 10.1136/jnnp.2009.176818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schneider AL, Gottesman RF, Mosley T, Alonso A, Knopman DS, Coresh J, Sharrett AR, Selvin E. Cognition and incident dementia hospitalization: results from the atherosclerosis risk in communities study. Neuroepidemiology. 2013;40:117–124. doi: 10.1159/000342308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Baecke JA, Burema J, Frijters JE. A short questionnaire for the measurement of habitual physical activity in epidemiological studies. Am J Clin Nutr. 1982;36:936–942. doi: 10.1093/ajcn/36.5.936. [DOI] [PubMed] [Google Scholar]
- 25.Richardson MT, Ainsworth BE, Wu HC, Jacobs DR, Jr, Leon AS. Ability of the atherosclerosis risk in communities (ARIC)/Baecke questionnaire to assess leisure-time physical activity. Int J Epidemiol. 1995;24:685–693. doi: 10.1093/ije/24.4.685. [DOI] [PubMed] [Google Scholar]
- 26.Salton CJ, Chuang ML, O'Donnell CJ, Kupka MJ, Larson MG, Kissinger KV, Edelman RR, Levy D, Manning WJ. Gender differences and normal left ventricular anatomy in an adult population free of hypertension. A cardiovascular magnetic resonance study of the Framingham Heart Study Offspring cohort. J Am Coll Cardiol. 2002;39:1055–1060. doi: 10.1016/s0735-1097(02)01712-6. [DOI] [PubMed] [Google Scholar]
- 27.Harrell FE, Jr, Lee KL, Mark DB. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996;15:361–387. doi: 10.1002/(SICI)1097-0258(19960229)15:4<361::AID-SIM168>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 28.Glymour MM, Weuve J, Berkman LF, Kawachi I, Robins JM. When is baseline adjustment useful in analyses of change? An example with education and cognitive change. Am J Epidemiol. 2005;162:267–278. doi: 10.1093/aje/kwi187. [DOI] [PubMed] [Google Scholar]
- 29.Casale PN, Devereux RB, Alonso DR, Campo E, Kligfield P. Improved sex-specific criteria of left ventricular hypertrophy for clinical and computer interpretation of electrocardiograms: validation with autopsy findings. Circulation. 1987;75:565–572. doi: 10.1161/01.cir.75.3.565. [DOI] [PubMed] [Google Scholar]
- 30.Moran A, Degennaro V, Ferrante D, Coxson PG, Palmas W, Mejia R, Perez-Stable EJ, Goldman L. Coronary heart disease and stroke attributable to major risk factors is similar in Argentina and the United States: the Coronary Heart Disease Policy Model. Int J Cardiol. 2011;150:332–337. doi: 10.1016/j.ijcard.2011.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Omland T, de Lemos JA, Sabatine MS, Christophi CA, Rice MM, Jablonski KA, Tjora S, Domanski MJ, Gersh BJ, Rouleau JL, Pfeffer MA, Braunwald E. A sensitive cardiac troponin T assay in stable coronary artery disease. N Engl J Med. 2009;361:2538–2547. doi: 10.1056/NEJMoa0805299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.deFilippi CR, de Lemos JA, Christenson RH, Gottdiener JS, Kop WJ, Zhan M, Seliger SL. Association of serial measures of cardiac troponin T using a sensitive assay with incident heart failure and cardiovascular mortality in older adults. JAMA. 2010;304:2494–2502. doi: 10.1001/jama.2010.1708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hijazi Z, Oldgren J, Siegbahn A, Granger CB, Wallentin L. Biomarkers in atrial fibrillation: a clinical review. Eur Heart J. 2013;34:1475–1480. doi: 10.1093/eurheartj/eht024. [DOI] [PubMed] [Google Scholar]
- 34.Mosley TH, Jr, Knopman DS, Catellier DJ, Bryan N, Hutchinson RG, Grothues CA, Folsom AR, Cooper LS, Burke GL, Liao D, Szklo M. Cerebral MRI findings and cognitive functioning: the Atherosclerosis Risk in Communities study. Neurology. 2005;64:2056–2062. doi: 10.1212/01.WNL.0000165985.97397.88. [DOI] [PubMed] [Google Scholar]
- 35.Kuller LH, Shemanski L, Manolio T, Haan M, Fried L, Bryan N, Burke GL, Tracy R, Bhadelia R. Relationship between ApoE, MRI findings, and cognitive function in the Cardiovascular Health Study. Stroke. 1998;29:388–398. doi: 10.1161/01.str.29.2.388. [DOI] [PubMed] [Google Scholar]
- 36.Liu H, Zhang J. Cerebral hypoperfusion and cognitive impairment: the pathogenic role of vascular oxidative stress. Int J Neurosci. 2012;122:494–499. doi: 10.3109/00207454.2012.686543. [DOI] [PubMed] [Google Scholar]
- 37.Kitagawa K, Oku N, Kimura Y, Yagita Y, Sakaguchi M, Hatazawa J, Sakoda S. Relationship between cerebral blood flow and later cognitive decline in hypertensive patients with cerebral small vessel disease. Hypertens Res. 2009;32:816–820. doi: 10.1038/hr.2009.100. [DOI] [PubMed] [Google Scholar]
- 38.van de Pol LA, Korf ES, van der Flier WM, Brashear HR, Fox NC, Barkhof F, Scheltens P. Magnetic resonance imaging predictors of cognition in mild cognitive impairment. Arch Neurol. 2007;64:1023–1028. doi: 10.1001/archneur.64.7.1023. [DOI] [PubMed] [Google Scholar]
- 39.Jokinen H, Kalska H, Ylikoski R, Madureira S, Verdelho A, van der Flier WM, Scheltens P, Barkhof F, Visser MC, Fazekas F, Schmidt R, O'Brien J, Waldemar G, Wallin A, Chabriat H, Pantoni L, Inzitari D, Erkinjuntti T. Longitudinal cognitive decline in subcortical ischemic vascular disease: the LADIS Study. Cerebrovasc Dis. 2009;27:384–391. doi: 10.1159/000207442. [DOI] [PubMed] [Google Scholar]
- 40.Fitzpatrick AL, Kuller LH, Ives DG, Lopez OL, Jagust W, Breitner JC, Jones B, Lyketsos C, Dulberg C. Incidence and prevalence of dementia in the Cardiovascular Health Study. J Am Geriatr Soc. 2004;52:195–204. doi: 10.1111/j.1532-5415.2004.52058.x. [DOI] [PubMed] [Google Scholar]
- 41.Tang MX, Cross P, Andrews H, Jacobs DM, Small S, Bell K, Merchant C, Lantigua R, Costa R, Stern Y, Mayeux R. Incidence of AD in African-Americans, Caribbean Hispanics, and Caucasians in northern Manhattan. Neurology. 2001;56:49–56. doi: 10.1212/wnl.56.1.49. [DOI] [PubMed] [Google Scholar]
- 42.Agarwal SK, Avery CL, Ballantyne CM, Catellier D, Nambi V, Saunders J, Sharrett AR, Coresh J, Heiss G, Hoogeveen RC. Sources of variability in measurements of cardiac troponin T in a community-based sample: the atherosclerosis risk in communities study. Clin Chem. 2011;57:891–897. doi: 10.1373/clinchem.2010.159350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pavlik VN, de Moraes SA, Szklo M, Knopman DS, Mosley TH, Jr, Hyman DJ. Relation between cognitive function and mortality in middle-aged adults: the atherosclerosis risk in communities study. Am J Epidemiol. 2003;157:327–334. doi: 10.1093/aje/kwf209. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.