Significance
Premature leaf senescence is known to decrease rice yield severely, but the molecular mechanism underlying this relationship remains largely unknown. Similarly, although abscisic acid (ABA)-induced leaf senescence has long been observed, the mechanism of this pathway has yet to be determined. In this study we identified and characterized a dominant premature leaf senescence mutant, prematurely senile 1 (ps1-D). The data demonstrated both that PS1/Oryza sativa NAC (no apical meristem, Arabidopsis ATAF1/2, and cup-shaped cotyledon2)-like, activated by apetala3/pistillata (OsNAP) is an ideal marker of natural senescence onset and that it functions as an important link between ABA and leaf senescence in rice. Furthermore, reduced OsNAP expression led to extended grain filling and an improved seed-setting rate, which significantly enhanced the grain yield. Thus, fine-tuning OsNAP expression should be a means of improving rice yield.
Keywords: hormones, nutrition remobilization, programmed cell death
Abstract
It has long been established that premature leaf senescence negatively impacts the yield stability of rice, but the underlying molecular mechanism driving this relationship remains largely unknown. Here, we identified a dominant premature leaf senescence mutant, prematurely senile 1 (ps1-D). PS1 encodes a plant-specific NAC (no apical meristem, Arabidopsis ATAF1/2, and cup-shaped cotyledon2) transcriptional activator, Oryza sativa NAC-like, activated by apetala3/pistillata (OsNAP). Overexpression of OsNAP significantly promoted senescence, whereas knockdown of OsNAP produced a marked delay of senescence, confirming the role of this gene in the development of rice senescence. OsNAP expression was tightly linked with the onset of leaf senescence in an age-dependent manner. Similarly, ChIP-PCR and yeast one-hybrid assays demonstrated that OsNAP positively regulates leaf senescence by directly targeting genes related to chlorophyll degradation and nutrient transport and other genes associated with senescence, suggesting that OsNAP is an ideal marker of senescence onset in rice. Further analysis determined that OsNAP is induced specifically by abscisic acid (ABA), whereas its expression is repressed in both aba1 and aba2, two ABA biosynthetic mutants. Moreover, ABA content is reduced significantly in ps1-D mutants, indicating a feedback repression of OsNAP on ABA biosynthesis. Our data suggest that OsNAP serves as an important link between ABA and leaf senescence. Additionally, reduced OsNAP expression leads to delayed leaf senescence and an extended grain-filling period, resulting in a 6.3% and 10.3% increase in the grain yield of two independent representative RNAi lines, respectively. Thus, fine-tuning OsNAP expression should be a useful strategy for improving rice yield in the future.
Leaf senescence is an integral part of the final stages of plant development and is controlled by a fine-tuned, complex regulatory network (1). During senescence, leaf cells undergo dramatic changes in cellular metabolism, structure, and gene expression (2, 3). The most striking feature of these changes is the yellowing of the leaves caused by the breakdown of chlorophyll during chloroplast degeneration, followed by the hydrolysis of macromolecules such as lipids, proteins, and nucleic acids, which, in turn, results in mitochondria and nuclei dissociation and cell death (4, 5). This process facilitates both hydrolysis and the recycling of nutrients from source to sink tissues to increase reproductive success (6). Thus, senescence is not a passive process but rather is a developmentally programmed procedure that has a strong adaptive advantage (7, 8). Although leaf senescence is controlled primarily by developmental age, the onset and progression of this process also is influenced by a number of endogenous and external factors (1, 9, 10). For example, abscisic acid (ABA) is thought to be one of the phytohormones that promote leaf senescence (11, 12). Specifically, both an up-regulation of genes associated with ABA signaling and a dramatic increase in endogenous ABA levels can be observed in many plants during leaf senescence (13). Furthermore, exogenously applied ABA has been shown to induce the expression of several senescence-associated genes (SAGs) known to accelerate leaf senescence (14), indicating the presence of a link between ABA signaling and leaf senescence. Moreover, a variety of biotic and abiotic stresses both elevate ABA levels and activate signaling pathways leading to senescence (1). Thus, it seems clear that ABA acts as a key positive regulator of leaf senescence. To date, however, the mechanistic evidence of ABA’s regulatory role in leaf senescence has been based exclusively on studies of RPK1 and SAG113 (11, 15). Consequently, both the molecular mechanism driving ABA-mediated leaf senescence and the specificity of ABA signaling in this process remain largely undetermined.
NAC [no apical meristem (NAM), Arabidopsis ATAF1/2, and cup-shaped cotyledon2] transcription factors comprise one of the largest plant-specific transcription factor classes and are involved in various plant processes, including plant development, leaf senescence, cell division, wood formation, and biotic/abiotic stress responses (16–18). Recently, high-resolution temporal profiling of these transcripts has revealed that the expression of 30 of 117 NAC genes was altered notably during the various stages of natural senescence in Arabidopsis (12), suggesting that NAC transcription factors play a crucial role in regulating the leaf senescence process. Despite this evidence, however, to date only a few NAC transcription factors have been shown to regulate senescence specifically. Prior research has identified AtNAP (Arabidopsis thaliana NAC-like, activated by apetala3/pistillata, At1g69490; also called “ANAC029”) (18), Oresara1 (ORE1, At5g39610; also called “ANAC092” or “ATNAC2”) (19), Oresara sister1 (ORS1, At3g29035; also called “ANAC059”) (20), Jungbrunnen1 (JUB1, At2g43000; also called “ANAC042”) (21), and Vascular-related NAC-domain interacting (VNI2, At5g13180; also called “ANAC083”) as playing significant regulatory roles in leaf senescence (6). For example, overexpression of AtNAP, ORE1, and ORS1 triggers precocious senescence, and blocking the function of these transcription factors delays senescence significantly, thus suggesting that AtNAP, ORE1, and ORS1 act as nonredundant positive regulators of senescence in Arabidopsis. In contrast, JUS1 and VNI2 regulate leaf senescence negatively. Although there are 151 known NAC genes in rice, to date only a few members have been identified as playing a regulatory role in leaf senescence (22, 23).
Premature leaf senescence is one of the primary factors influencing yield stability in rice (24) and particularly in hybrid rice. Compared with the model plant Arabidopsis, the identification of key SAGs and their cognate molecular regulatory mechanisms in rice has begun only recently. Similarly, the majority of the SAGs identified to date have been shown only to be involved in chlorophyll breakdown and degradation (25–27). Because there is likely to be a significant level of gene redundancy in the complex pathways that are integral to the senescence process, identifying the key regulators of leaf senescence using loss-of-function mutants has proved difficult. To uncover key genes controlling leaf senescence in rice, we screened our transferred-DNA (T-DNA) population for gain-of-function mutants (28). Over the course of this process, more than 250 independent lines with altered senescence phenotypes were identified. One of the gain-of-function mutants, prematurely senile 1 (ps1-D), which demonstrated significant premature leaf senescence, was selected for this study.
Results
ps1-D Mutants Exhibit Premature Leaf Senescence Phenotype.
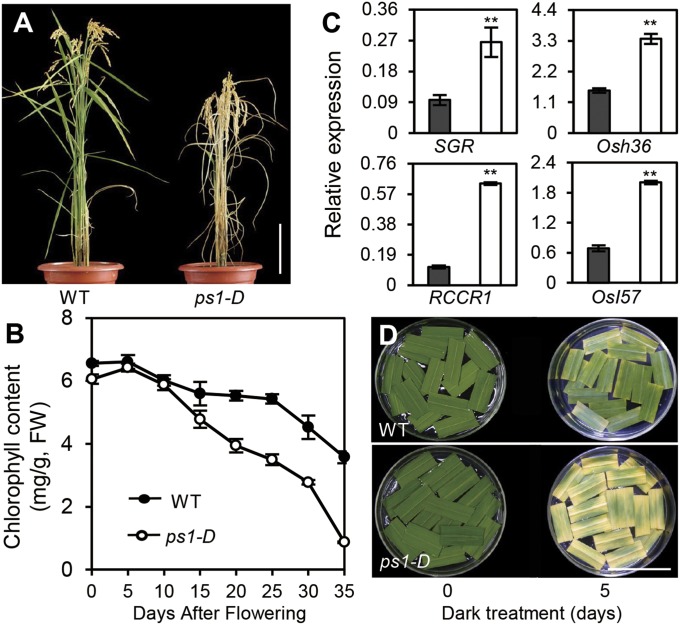
ps1-D mutants did not show any phenotypic differences as compared with wild-type plants before the four-leaf stage of development. When the plant progressed to the tillering stage, however, leaf senescence was initiated (Fig. S1A), and all five upper leaves of the ps1-D mutant exhibited significantly accelerated leaf senescence at the heading stage (Fig. S1 B and C). Similarly, after grain filling, ps1-D mutants had an accelerated yellowing phenotype, displaying senescence 7–10 d earlier than the wild-type plants (Fig. 1A). In addition, after flowering, ps1-D mutants also exhibited faster chlorophyll degradation than the wild-type plants. The most pronounced difference between the ps1-D mutants and wild-type plants was that 25 d after flowering (DAF) the chlorophyll content of the mutants was only two-thirds that of wild-type plants. Moreover, this disparity in chlorophyll content became more dramatic with time. For example, at 35 DAF the chlorophyll content of the mutants was only 20% of that found in the wild-type plants, demonstrating that chlorophyll degradation proceeded at a significantly more rapid pace in the ps1-D mutant (Fig. 1B) than in wild-type plants. Similarly, two chlorophyll degradation-related genes (CDGs), stay-green (SGR) and red chlorophyll catabolite reductase 1 (RCCR1) (29, 30), and two other SAGs, Osh36 and OsI57 (31), were expressed at higher levels in the fully expanded leaves of ps1-D mutants than in wild-type plants (Fig. 1C). Overall, the leaf senescence process was accelerated significantly in the ps1-D mutants. Darkness is one of the most powerful known external stimuli of leaf senescence. Consequently, it is used frequently as an effective method of simulating synchronous senescence (5, 25). As demonstrated by the rapid reduction in chlorophyll content and elevated expression levels of two senescence marker genes, Osh36 and OsI57 in the mutant plants (Fig. S1 D–F), the ps1-D mutation significantly accelerated dark-induced leaf senescence (Fig. 1D). Overall, our results clearly demonstrated that mutation of PS1 influences a variety of processes associated with the induction of senescence.
Fig. 1.
ps1-D mutant phenotypes. (A) Phenotypes of the ps1-D mutant 40 DAF. (Scale bar: 20 cm.) (B) Chlorophyll content of flag leaves in the ps1-D mutant after flowering. Values are means ± SD of 20 measurements. FW, fresh weight. (C) Expression of CDGs (SGR and RCCR1) (Left) and other SAGs (Osh36 and OsI57) (Right) in wild-type plants and ps1-D mutants. The black bar represents wild-type plants, and the white bar represents ps1-D mutants. **P ≤ 0.01; Student t test. (D) ps1-D promoted dark-induced leaf senescence. Detached flag leaves from wild-type plants and ps1-D mutants at the heading stage were incubated with water for 5 d in darkness.
PS1 Encodes a Plant-Specific NAC Transcription Activator.
The genetic analysis of the heterologous ps1-D offspring indicated that the leaf senescence phenotype was cosegregated with the T-DNA insertion at a ratio of ∼3:1, suggesting that ps1-D is a dominant mutation caused by the T-DNA insertion. The flanking region of T-DNA was obtained by SiteFinding PCR, and sequence analysis revealed that the T-DNA insertion site was 214-bp upstream of the translation initiation site of LOC_Os03g21060 (Fig. S2 A and B). LOC_Os03g21060 was the only gene within the vicinity of the T-DNA insertion site whose expression was elevated 20-fold in ps1-D seedlings (Fig. S2C). Overexpression of LOC_Os03g21060 in the wild-type plants led to a phenotype demonstrating varying degrees of premature leaf senescence (Fig. S2D). Similarly, the magnitude of this change in senescence correlated with the expression level of LOC_Os03g21060 (Fig. S2E). These results confirm both that LOC_Os03g21060 is PS1 and that its activation is the cause of premature senescence phenotype of ps1-D mutants.
PS1 contains a typical NAC structure at the N terminus (Fig. S2F) and shares about 69% of its amino acids in this area with AtNAP. It belongs to the Va (1)/NAP subfamily of NAC proteins, which has three members in Arabidopsis, six members in rice, and five members in maize (Fig. S3A). Previously PS1 was named “Oryza sativa NAC-like, activated by apetala3/pistillata” (OsNAP), which could complement the atnap-null mutant, confirming that PS1 is the functional ortholog of AtNAP (18, 22). In vitro transactivation activity assays in yeast confirmed that OsNAP is a functional transcriptional activator (Fig. S3B). A detailed domain analysis showed that the C-terminal region (amino acids 181–392) possessed high transcriptional activation activity, whereas the N-terminal region of the NAC domain (amino acids 1–190) did not display any activity. Interestingly, the C-terminal region of OsNAP (amino acids 18–392) was found to have stronger transcriptional activation activity than the full-length gene, suggesting that a repression domain may exist in the N-terminal region. To map these potential transcriptional repression domains further, vectors expressing various lengths of truncated OsNAP were tested in a yeast system. Among these fragments, the fragment containing subdomains 3 (amino acids 64–100) and 4 (amino acids 100–142) of the NAC motif showed stronger repression activity, whereas subdomains 1 and 2 (amino acids 1–64) and 5 (amino acids 157–181) exhibited weaker repression activity (Fig. S3B). These results indicate that OsNAP functions as a transcriptional activator. Specifically, OsNAP’s C terminus appears to act as an activation domain, whereas the NAC subdomains 3 (amino acids 64–100) and 4 (amino acids 100–142) function as a repressor used to tune C-terminal activity.
OsNAP Is Highly Expressed in Senescing Tissues.
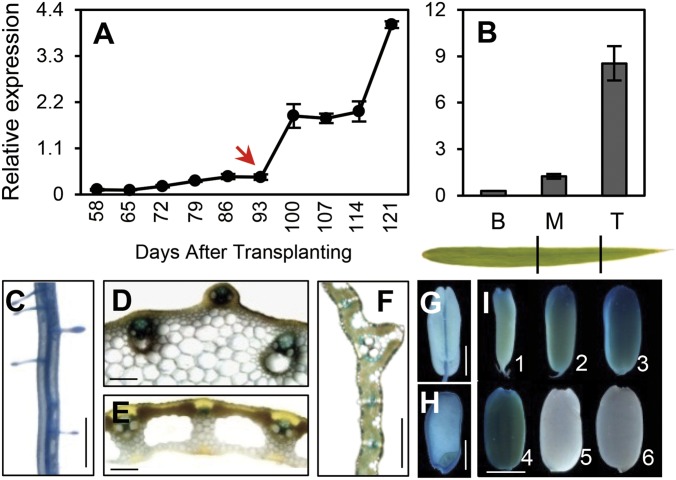
Temporal and spatial expression analysis showed that OsNAP is expressed preferentially in the leaf blade, leaf sheath, and endosperm with a low, but still detectable, level of expression in the root, culm, and young panicle (Fig. S4A). Therefore we examined OsNAP expression in the leaves at different developmental stages. The OsNAP transcripts were higher in old, senescing leaves than in young, green leaves (Fig. S4 B and D). Similarly, a kinetic analysis of OsNAP expression in flag leaves showed that, although the number of OsNAP transcripts increased gradually before heading, it increased dramatically with the beginning of grain filling (Fig. 2A). Consistent with this observation, OsNAP expression decreased gradually from the tip to the base of a fully expanded leaf (Fig. 2B). Strikingly, the expression of OsNAP in the endosperm increased gradually over the course of the grain-filling process, reaching its transcriptional peak at ∼25 DAF (Fig. S4C). This finding suggests that OsNAP plays an important role in the endosperm maturation, another programmed cell-death process in plants. Overall, these data indicate that OsNAP is an ideal marker for the natural senescence process in rice.
Fig. 2.
Analysis of OsNAP expression. (A) Change over time in the OsNAP transcription levels of flag leaves. The red arrow indicates the flowering time. (B) OsNAP expression in different parts of the fully expanded leaf. B, base; M, middle; T, tip. (C–I) Histochemical staining of OsNAPPro::GUS transgenic lines. Roots (C), culms (D), leaf sheathes (E), leaf blades (F), stamen (G), and seeds (H and I). (Scale bars: 1 mm in C and G; 2.5 mm in H and I; 100 µm in D, E, and F.)
Examination of transgenic plants harboring an OsNAPPro::β-glucuronidase (GUS) construct revealed that OsNAP was expressed throughout a number of different organs over the course of plant development. For example, in young seedlings, GUS activity was detected in both the primary and lateral roots, particularly in the vascular tissues (Fig. 2C). GUS also was expressed specifically in the primary phloem of the culm and leaf sheath (Fig. 2 D and E). Finally, consistent with the results of quantitative RT-PCR (qRT-PCR), GUS expression was strongest in old leaves or in the senescent regions of leaves. Further examination of the tissue sections revealed that OsNAP was expressed principally in the primary phloem and in the peripheral zone of the leaf vascular bundles (Fig. 2F), although GUS expression also was observed in the floral tissues (Fig. 2G) and endosperm (Fig. 2 H and I). In the endosperm, GUS expression was congruent with the endogenous OsNAP expression pattern suggested by qRT-PCR, further confirming our hypothesis that OsNAP plays a role in regulating both leaf senescence and endosperm maturation.
ABA Participates in Leaf Senescence by Modulating OsNAP Expression.
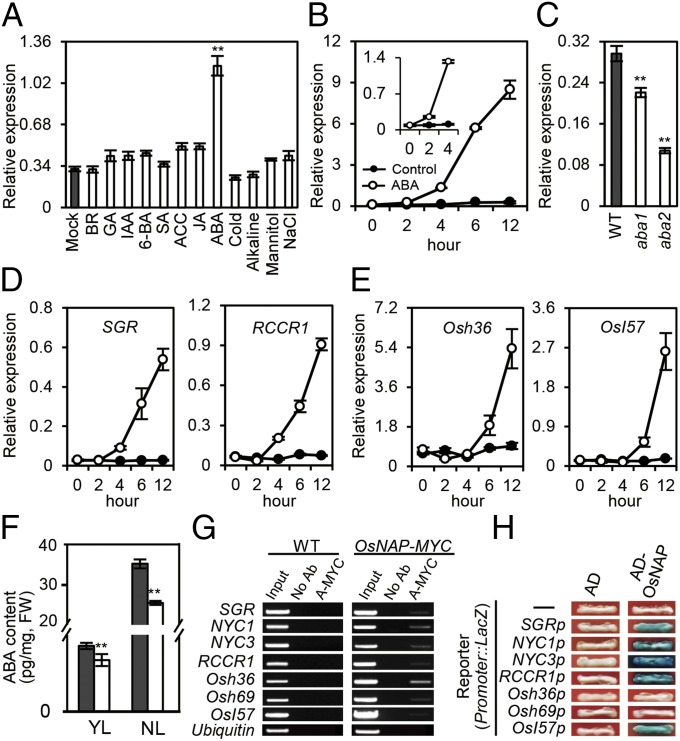
Leaf senescence is a genetically controlled developmental process that can be modulated by a variety of phytohormones and environmental factors (1). Expression profiling of OsNAP was performed with RNAs derived from different phytohormones (including brassinosteroid, gibberellin, auxin, 6-benzylaminopurine, salicylic acid, 1-aminocyclo-propane-1-carboxylic acid, jasmonic acid, and ABA) and abiotic stress treatments (cold, alkaline, mannitol, and NaCl). The results demonstrate that OsNAP is induced exclusively by ABA, with expression levels increasing approximately threefold after 2 h of ABA treatment (Fig. 3A). OsNAP transcription increased rapidly up to 16-fold after 4 h, by 68-fold after 6 h, and by 102-fold after 12 h of treatment (Fig. 3B). Conversely, OsNAP transcription is reduced significantly in two ABA biosynthetic mutants, aba1 and aba2 (Fig. 3C), confirming our theory that OsNAP is positively regulated by ABA. We also examined the kinetic expression of CDGs and other SAGs following ABA treatment. No increase in SGR and/or RCCR1 expression was observed 2 h after ABA treatment, but a marginal increase was observed after 4 h (Fig. 3D). Similarly, induced expression of Osh36 and OsI57 was observed only at 4 h after ABA treatment (Fig. 3E). This lag in expression implies that ABA-mediated leaf senescence might be dependent on the modulation of OsNAP expression, which, in turn, either directly or indirectly regulates the expression of SAGs. Thus, it seems that OsNAP functions as an important link between ABA signaling and leaf senescence.
Fig. 3.
OsNAP connects ABA and SAGs by directly targeting promoters of SAGs in rice. (A) Effects of phytohormones and abiotic stresses on OsNAP expression. 6-BA, 6-benzylaminopurine; ACC, 1-aminocyclo-propane-1-carboxylic acid; BR, brassinosteroid; GA3, gibberellic acid; IAA, indole-3-acetic acid; JA, jasmonic acid; SA, salicylic acid. **P ≤ 0.01; Student t test. (B) Kinetic analysis by qRT-PCR of OsNAP expression after treatment with 50 µm ABA. (Inset) Enlarged view of time points 0, 2, and 4. (C) Decreased expression of OsNAP in aba1 and aba2 mutants. Age-matched fully expanded flag leaves were used for the analysis. **P ≤ 0.01; Student t test. (D and E) Kinetic expression analysis of two CDGs, SGR and RCCR1, (D) and two other SAGs, Osh36 and OsI57, (E) after treatment with 50 µm ABA. (F) ABA content in wild-type plants and ps1-D mutants. YL, young leaf approximately half the size of a fully expanded leaf. NL, fully-expanded, non-senescent flag leaves. The black bar represents wild-type plants, and the white bar represents for ps1-D mutants. FW, fresh weight. **P ≤ 0.01; Student t test. (G) ChIP-PCR analysis of the promoter regions of SAGs. Immunoprecipitation was performed with anti-MYC antibody (A-MYC) or without antibody (No Ab). The promoter of Ubiquitin was used as a negative control. (H) AD-OsNAP activates the expression of the LacZ reporter genes driven by the promoters of respective SAGs in yeast. Representative data are shown from one of the three biological replicates, which yielded similar results.
We next examined the ABA level in young leaves and fully expanded leaves of ps1-D and wild-type plants. The ABA content was significantly lower in ps1-D mutants than in wild-type plants (Fig. 3F). Further examination revealed that the transcription levels of key ABA biosynthesis genes, including OsNCED1, OsNCED3, OsNCED4, and OsZEP, were significantly down-regulated in the ps1-D mutant (Fig. S5A), but no alteration was detected in any ABA-inactivation genes, including OsABA8ox1, OsABA8ox2, and OsABA8ox3 (Fig. S5B). Consequently, it appears that the high level of OsNAP transcripts in the ps1-D mutant may regulate ABA biosynthesis further via a feedback mechanism.
OsNAP Functions Upstream of SAGs in ABA-induced Leaf Senescence.
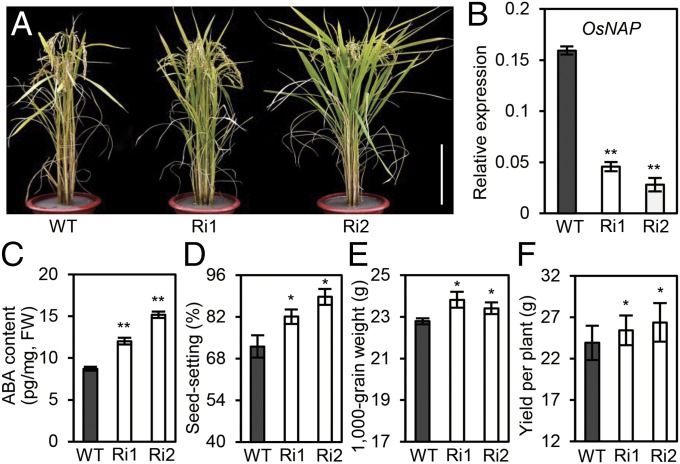
To explore further the intrinsic functions of OsNAP, RNA interference (RNAi) technology was used to suppress OsNAP expression in wild-type plants. The RNAi construct was targeted specifically to the nonconserved 3′ end of OsNAP outside the NAC domain to avoid interference with other NAC proteins. Expression analysis revealed no significant alteration in the five orthologous genes between the RNAi lines and wild-type plants (Fig. S6A), confirming the specificity of the OsNAP RNAi targeting. Our RNAi transgenic lines displayed distinctly delayed leaf senescence (Fig. 4A), which was consistent with the observed decline in OsNAP expression (Fig. 4B). As expected, the expression levels of representative CDGs, including SGR, NYC1, NYC3, and RCCR1, and other SAGs, including Osh36, OsI57, Osh69, and OsI85, were significantly lower in the fully expanded leaves of the RNAi lines than in the wild-type plants (Fig. S6 B and C).
Fig. 4.
Agronomic traits of OsNAP RNAi transgenic lines. (A) Gross morphology of the OsNAP RNAi lines in field conditions. (Scale bar: 20 cm.) (B) Expression of OsNAP in RNAi plants. (C) ABA content in RNAi transgenic lines. Fully expanded and nonsenescent flag leaves were used for ABA analysis. FW, fresh weight. (D–F) Seed-setting rate (D), 1,000-grain weight (E), and grain yield per plant (F) in the RNAi lines. *P ≤ 0.05; **P ≤ 0.01; Student t test.
We further examined the senescence symptoms of detached leaves after 10-d incubation in darkness or under ABA treatment. Detached leaves of OsNAP RNAi transgenic plants exhibited distinctly delayed senescence (Fig. S6D), as was consistent with the slower chlorophyll degradation (Fig. S6E) and delayed expression of OsNAP and other physiological senescence-marker genes, such as Osh36 and Os157 (Fig. S6 F–H), observed in the mutant plants. ChIP assays further confirmed that OsNAP indeed bound to the promoters of SGR, NYC1, NYC3, RCCR1, Osh36, OsI57, and Osh69 (Fig. 3G). A yeast one-hybrid assay also showed that the GAL4 transcriptional activation domain-OsNAP (AD-OsNAP) fusion protein activates the LacZ reporter gene driven by the promoters of SGR, NYC1, NYC3, RCCR1, and OsI57, respectively (Fig. 3H). However, OsNAP did not bind directly to the promoters of Osh36 and Osh69, indicating that OsNAP-interaction proteins may exist that bind to the promoters of Osh36 and Osh69. Thus, OsNAP, as a transcription factor, regulates the expression levels of SAGs by directly or indirectly binding to their promoter regions in vivo.
OsNAP Regulates Nutrition Remobilization.
A genome-wide expression analysis of the ps1-D mutant using an Affymetrix whole-genome microarray showed 599 differently expressed genes (DEGs) in the mutant with log2 ratios greater than 1.00 or less than −1.00 compared with the wild-type plants (Table S1). Notably, a large proportion of these DEGs were involved in macromolecule degradation and nutrient mobilization. About 13.5% of the up- regulated transcripts and 19.6% of the down-regulated transcripts were involved in the metabolism of amino acids, lipids, nucleic acids, and carbohydrates (Table S1). A number of proteases, including cysteine and aspartyl proteases, lipases, and nucleases, also were identified according to their putative Gene Ontology function. These data suggest that the accumulation of OsNAP transcripts leads to enhanced metabolism and remobilization of the nutrients synthesized during the vegetative phase. Previous studies have shown that remobilization of nutrients from senescing cells to developing tissues, such as seeds was mediated primarily by phloem transport (32). We found that the expression of 30 genes encoding ABC transporters, permeases, and transports of amino acids, peptides, sugars, lipids, metals were all notably up- or down-regulated in the ps1-D mutant (Table S2). Genes encoding peptide transporter 2 (OsPTR2), proton-dependent oligopeptide transporter (OsPOT), high-affinity K+ transporter 5 (OsHAK5), and natural resistance-associated macrophage protein 6 (OsNramp6) were all up-regulated in the mutant, whereas high-affinity potassium transporter 6 (OsHKT6), peptide transporter 3 (OsPTR3), and phosphate transporter1 (OsPHT1) were all down-regulated in the mutant. ChIP-PCR further proved that OsNAP actually bound to the promoters of the genes encoding OsHKT6, heavy metal transport/detoxification protein, and calcium-transporting ATPase (Fig. S7), implying that OsNAP plays an important role in the augmented translocation of nutrients during the senescing process.
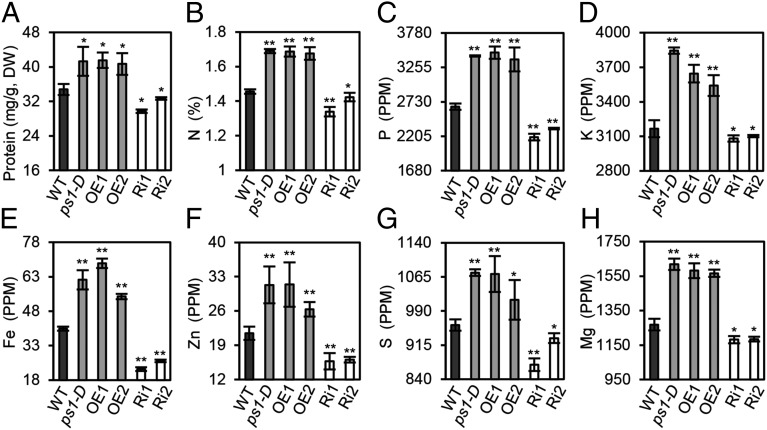
To determine whether the transporters that exhibited altered expression levels in the mutant were functionally involved in nutrient remobilization, the total grain protein content (GPC) and nutrient elements of mature seeds from ps1-D mutants, OsNAP over-expressing (OE) lines, RNAi lines, and wild-type plants were all quantified. As shown in Fig. 5, significant increases in the total grain protein and in N, P, K, S, Fe, Zn, and Mg concentrations were observed in the rice grains originating from the ps1-D and OsNAP OE lines. In contrast, the grain protein and mineral micronutrient concentrations found in the OsNAP RNAi lines were 10% lower than their counterparts in the wild-type plants. Further analysis of the N, P, K, S, Fe, Zn, and Mg content in flag leaves likewise showed that ps1-D and OsNAP OE transgenic plants demonstrated a reduction of more than 7.8% in N, 12.6% in P, 14.8% in S, 1.9% in K, 10.5% in Mg, 26.6% in Fe, and 18.7% in Zn concentrations in comparison with wild-type plants. In contrast, the OsNAP RNAi transgenic lines retain more nutrient elements than wild-type plants (Table S3). Overall, these data demonstrate that the transcription level of OsNAP is linked directly to the nutrient remobilization associated with senescence.
Fig. 5.
Protein and nutrient content in rice grain. Total grain protein (A) and N (B), P (C), K (D), Fe (E), Zn (F), S (G), and Mg (H) concentrations in mature seeds. Values are means ± SD of 10 biological replicates. DW, dry weight. *P ≤ 0.05; **P ≤ 0.01; Student t test.
Down-Regulation of OsNAP Leads to Increased Grain Yield.
Because the OsNAP RNAi transgenic plants showed a 5- to 7-d delay in senescence, we next investigated whether this mutation would improve grain yield in plants grown in the field. As expected, OsNAP-knockdown transgenic plants showed both a significantly slower decrease in functional photosynthetic capacity and an extended grain-filling period than wild-type plants (Table S4). Furthermore, an examination of the agronomic traits related to grain yield in two OsNAP RNAi lines, Ri1 and Ri2, showed a 13.7% and 23.0% increase in seed-setting ratio and a 4.4% and 3.1% increase in 1,000-grain weight, respectively, compared with our nontransgenic controls. These changes resulted in increased grain yields of 6.3% and 10.3% in the Ri1 and Ri2 lines, respectively (Fig. 4 D–F). However, grain size did not differ in the wild-type and RNAi plants, suggesting that reduced OsNAP expression should extend the functional period of photosynthesis and improve the grain yield in rice.
Discussion
Although ABA was characterized as a senescence-promoting hormone several decades ago, the mechanism underlying ABA-mediated senescence still is poorly understood. In our study, the leaf-senescence process was accelerated significantly in the activation mutant ps1-D during the tillering and grain-filling stages of development but not before plants reached the four-leaf stage. This finding suggests that OsNAP mediates leaf senescence in an age-dependent manner, making OsNAP an ideal marker for the onset of senescence in rice. Although the expression of OsNAP can be regulated positively by either exogenous or endogenous ABA, it cannot be regulated by any other phytohormones or abiotic stresses. Similarly, although ABA is known to promote the senescence process of detached leaves, this response is markedly delayed in OsNAP RNAi transgenic plants. The kinetic expressions of two different types of senescence-marker genes were observed to lag significantly behind OsNAP expression under ABA treatment. Similarly, the knockdown of OsNAP expression also decreased the expression of these senescence-marker genes. ChIP-PCR and yeast one-hybrid assays also demonstrated that OsNAP bound directly to the promoter regions of these genes, further showing that OsNAP functions upstream of these senescence markers. These observations led us to conclude that ABA-mediated leaf senescence is dependent primarily on the modulation of OsNAP expression and that OsNAP acts as a key component linking ABA signaling and leaf senescence. Intriguingly, the accumulation of OsNAP transcripts also reduced ABA content by inhibiting ABA biosynthesis, whereas the ABA content was significantly higher in the two RNAi transgenic plants than in the wild-type plants (Fig. 4C). This result indicates that OsNAP also controls ABA synthesis via a feedback mechanism. Other than being slightly shorter than the wild-type plants, no obvious developmental defect was observed in the ps1-D mutant, indicating that ABA-mediated senescence may be an independent physiological event mediated by a specific, OsNAP-mediated signaling pathway.
Senescence is a highly complex but finely regulated developmental process that is tightly linked to crop yield, biomass production, and nutritional quality. Although recent research has greatly improved our understanding of leaf senescence in the model plant Arabidopsis, the molecular mechanism driving leaf senescence in monocot crop plants such as rice, maize, and wheat remains largely unknown. In rice, only a limited number of the leaf-senescence regulators have been identified so far, including CCCH-tandem zinc finger protein 1 (OsTZF1) (33), rapid leaf senescence 1 (RLS1) (34), alkaline a-galactosidase (OsAkaGal, also called “Osh69”) (35), NYC1 (26), NYC3 (36), NYC4 (27), SGR (29, 37), and delay of the onset of senescence (OsDOS) (25). However, most of the regulators identified to date are involved almost exclusively in processes of chlorophyll breakdown and degradation. Because the mutation of these regulators also resulted in defective photosynthetic capacity, the delayed senescence observed in the leaves of these mutants did not have any positive impact on their yield. In this study, we identified and characterized OsNAP, which acts as an important regulator in natural, dark-, and ABA-induced senescence. Knockdown of OsNAP delayed leaf senescence significantly, extended photosynthetic capabilities, and led to higher grain yields. Previous reports have shown that a NAC transcription factor, NAM-B1, accelerates the translocation of nutrients from vegetative tissues to grains, leading to the senescence process in wheat (38). In our microarray data, several classes of nutrient transport-related genes (NTGs), including eight amino acid or peptide transporters, four potassium transporters, two phosphate transporters, one sulfate transporter, three metal transporters, and 12 other transporters involved in nutrient translocation, were obviously up- or down-regulated. OsPTR2 (AK060510), for example, is the homolog to Arabidopsis NRT1.7, which is responsible for the phloem loading of nitrates in the source leaf to facilitate the rapid delivery of nitrates from the older leaves to nitrogen-demanding tissues (32). The function of phloem is linked closely with the remobilization of nutrients from senescing cells to reproductive and other organs. Coincidentally, our GUS staining clearly indicated that OsNAP was expressed preferentially in the primary phloem of blades, culms, and leaf sheaths. These results indicate that OsNAP also may regulate leaf senescence directly or indirectly through regulating genes involved in nutrient remobilization.
Overall, we propose a functional model of OsNAP’s role in rice leaf senescence (Fig. 6). The leaf-aging signal triggers the accumulation of OsNAP transcripts, and this accumulation initiates the onset of senescence. The validity of this mechanism is supported by our observations that OsNAP directly or indirectly regulates the expression of genes known to control senescence in an age-dependent manner, including CDGs, nutrient transport-related genes, and other SAGs. In addition, ABA participates in leaf senescence by modulating OsNAP expression, although high OsNAP expression levels also could regulate ABA biosynthesis via a feedback mechanism. Thus, OsNAP appears to act as a key regulator linking the ABA-signaling and leaf-senescence processes.
Fig. 6.
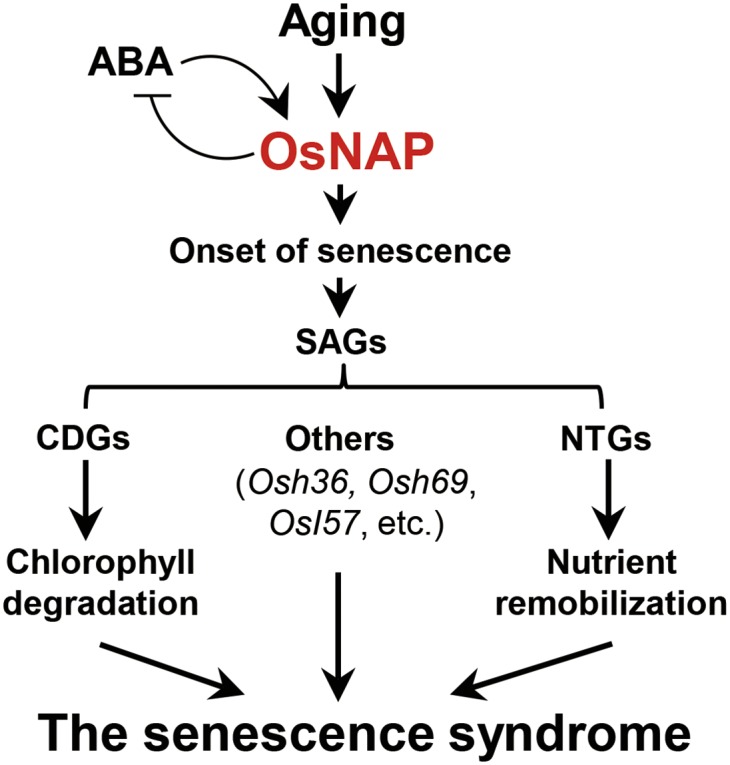
Proposed model for OsNAP’s function in rice leaf senescence.
Materials and Methods
The ps1-D mutant was identified by screening our T-DNA population in japonica Nipponbare background from our laboratory (28). Details of experimental procedures, such as ABA analysis, ChIP-PCR assay, yeast one-hybrid assay, element and protein measurements, microarray hybridization, and field cultivation of rice, are described in SI Materials and Methods. See Tables S5 and S6 for the primers used in this study.
Supplementary Material
Acknowledgments
We thank Abigail Coplin (Columbia University) and Prof. Gary J. Loake (Edinburgh University) for critically reading the manuscript. This work was supported by Grant 2014ZX0800933B from the Ministry of Agriculture, Grants 31171514 and 90817008 from the National Natural Science Foundation of China, and Grant XDA08010401 from the Chinese Academy of Sciences.
Footnotes
The authors declare no conflict of interest.
*This Direct Submission article had a prearranged editor.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1321568111/-/DCSupplemental.
References
- 1.Lim PO, Kim HJ, Nam HG. Leaf senescence. Annu Rev Plant Biol. 2007;58:115–136. doi: 10.1146/annurev.arplant.57.032905.105316. [DOI] [PubMed] [Google Scholar]
- 2.Quirino BF, Noh Y-S, Himelblau E, Amasino RM. Molecular aspects of leaf senescence. Trends Plant Sci. 2000;5(7):278–282. doi: 10.1016/s1360-1385(00)01655-1. [DOI] [PubMed] [Google Scholar]
- 3.Buchanan-Wollaston V, et al. Comparative transcriptome analysis reveals significant differences in gene expression and signalling pathways between developmental and dark/starvation-induced senescence in Arabidopsis. Plant J. 2005;42(4):567–585. doi: 10.1111/j.1365-313X.2005.02399.x. [DOI] [PubMed] [Google Scholar]
- 4.Woo HR, et al. ORE9, an F-box protein that regulates leaf senescence in Arabidopsis. Plant Cell. 2001;13(8):1779–1790. doi: 10.1105/TPC.010061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kim HJ, et al. Cytokinin-mediated control of leaf longevity by AHK3 through phosphorylation of ARR2 in Arabidopsis. Proc Natl Acad Sci USA. 2006;103(3):814–819. doi: 10.1073/pnas.0505150103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yang SD, Seo PJ, Yoon HK, Park CM. The Arabidopsis NAC transcription factor VNI2 integrates abscisic acid signals into leaf senescence via the COR/RD genes. Plant Cell. 2011;23(6):2155–2168. doi: 10.1105/tpc.111.084913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gan S, Amasino RM. Inhibition of leaf senescence by autoregulated production of cytokinin. Science. 1995;270(5244):1986–1988. doi: 10.1126/science.270.5244.1986. [DOI] [PubMed] [Google Scholar]
- 8.Woo HR, Kim HJ, Nam HG, Lim PO. Plant leaf senescence and death - regulation by multiple layers of control and implications for aging in general. J Cell Sci. 2013;126(Pt 21):4823–4833. doi: 10.1242/jcs.109116. [DOI] [PubMed] [Google Scholar]
- 9.Jing HC, Schippers JH, Hille J, Dijkwel PP. Ethylene-induced leaf senescence depends on age-related changes and OLD genes in Arabidopsis. J Exp Bot. 2005;56(421):2915–2923. doi: 10.1093/jxb/eri287. [DOI] [PubMed] [Google Scholar]
- 10.Zhang K, Halitschke R, Yin C, Liu CJ, Gan SS. Salicylic acid 3-hydroxylase regulates Arabidopsis leaf longevity by mediating salicylic acid catabolism. Proc Natl Acad Sci USA. 2013;110(36):14807–14812. doi: 10.1073/pnas.1302702110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee IC, et al. Age-dependent action of an ABA-inducible receptor kinase, RPK1, as a positive regulator of senescence in Arabidopsis leaves. Plant Cell Physiol. 2011;52(4):651–662. doi: 10.1093/pcp/pcr026. [DOI] [PubMed] [Google Scholar]
- 12.Breeze E, et al. High-resolution temporal profiling of transcripts during Arabidopsis leaf senescence reveals a distinct chronology of processes and regulation. Plant Cell. 2011;23(3):873–894. doi: 10.1105/tpc.111.083345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tan BC, et al. Molecular characterization of the Arabidopsis 9-cis epoxycarotenoid dioxygenase gene family. Plant J. 2003;35(1):44–56. doi: 10.1046/j.1365-313x.2003.01786.x. [DOI] [PubMed] [Google Scholar]
- 14.Quiles MJ, García C, Cuello J. Differential effects of abscisic acid and methyl jasmonate on endoproteinases in senescing barley leaves. Plant Growth Regul. 1995;16(2):197–204. [Google Scholar]
- 15.Zhang K, Gan SS. An abscisic acid-AtNAP transcription factor-SAG113 protein phosphatase 2C regulatory chain for controlling dehydration in senescing Arabidopsis leaves. Plant Physiol. 2012;158(2):961–969. doi: 10.1104/pp.111.190876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fujita M, et al. A dehydration-induced NAC protein, RD26, is involved in a novel ABA-dependent stress-signaling pathway. Plant J. 2004;39(6):863–876. doi: 10.1111/j.1365-313X.2004.02171.x. [DOI] [PubMed] [Google Scholar]
- 17.Xie Q, Frugis G, Colgan D, Chua NH. Arabidopsis NAC1 transduces auxin signal downstream of TIR1 to promote lateral root development. Genes Dev. 2000;14(23):3024–3036. doi: 10.1101/gad.852200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guo Y, Gan S. AtNAP, a NAC family transcription factor, has an important role in leaf senescence. Plant J. 2006;46(4):601–612. doi: 10.1111/j.1365-313X.2006.02723.x. [DOI] [PubMed] [Google Scholar]
- 19.Kim JH, et al. Trifurcate feed-forward regulation of age-dependent cell death involving miR164 in Arabidopsis. Science. 2009;323(5917):1053–1057. doi: 10.1126/science.1166386. [DOI] [PubMed] [Google Scholar]
- 20.Balazadeh S, et al. ORS1, an H2O2-responsive NAC transcription factor, controls senescence in Arabidopsis thaliana. Mol Plant. 2011;4(2):346–360. doi: 10.1093/mp/ssq080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu A, et al. JUNGBRUNNEN1, a reactive oxygen species-responsive NAC transcription factor, regulates longevity in Arabidopsis. Plant Cell. 2012;24(2):482–506. doi: 10.1105/tpc.111.090894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhou Y, et al. Identification and functional characterization of a rice NAC gene involved in the regulation of leaf senescence. BMC Plant Biol. 2013;13(1):132. doi: 10.1186/1471-2229-13-132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sperotto RA, et al. Identification of up-regulated genes in flag leaves during rice grain filling and characterization of OsNAC5, a new ABA-dependent transcription factor. Planta. 2009;230(5):985–1002. doi: 10.1007/s00425-009-1000-9. [DOI] [PubMed] [Google Scholar]
- 24.Jing HC, Nam HG. Leaf senescence in plants: From model plants to crops, still so many unknowns. J Integr Plant Biol. 2012;54(8):514–515. doi: 10.1111/j.1744-7909.2012.01148.x. [DOI] [PubMed] [Google Scholar]
- 25.Kong Z, Li M, Yang W, Xu W, Xue Y. A novel nuclear-localized CCCH-type zinc finger protein, OsDOS, is involved in delaying leaf senescence in rice. Plant Physiol. 2006;141(4):1376–1388. doi: 10.1104/pp.106.082941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kusaba M, et al. Rice NON-YELLOW COLORING1 is involved in light-harvesting complex II and grana degradation during leaf senescence. Plant Cell. 2007;19(4):1362–1375. doi: 10.1105/tpc.106.042911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yamatani H, et al. NYC4, the rice ortholog of Arabidopsis THF1, is involved in the degradation of chlorophyll - protein complexes during leaf senescence. Plant J. 2013;74(4):652–662. doi: 10.1111/tpj.12154. [DOI] [PubMed] [Google Scholar]
- 28.Ma Y, et al. Molecular analysis of rice plants harboring a multi-functional T-DNA tagging system. J Genet Genomics. 2009;36(5):267–276. doi: 10.1016/S1673-8527(08)60114-9. [DOI] [PubMed] [Google Scholar]
- 29.Jiang H, et al. Molecular cloning and function analysis of the stay green gene in rice. Plant J. 2007;52(2):197–209. doi: 10.1111/j.1365-313X.2007.03221.x. [DOI] [PubMed] [Google Scholar]
- 30.Tang Y, et al. Knockdown of OsPAO and OsRCCR1 cause different plant death phenotypes in rice. J Plant Physiol. 2011;168(16):1952–1959. doi: 10.1016/j.jplph.2011.05.026. [DOI] [PubMed] [Google Scholar]
- 31.Lee RH, Wang CH, Huang LT, Chen SC. Leaf senescence in rice plants: Cloning and characterization of senescence up-regulated genes. J Exp Bot. 2001;52(358):1117–1121. doi: 10.1093/jexbot/52.358.1117. [DOI] [PubMed] [Google Scholar]
- 32.Fan SC, Lin CS, Hsu PK, Lin SH, Tsay YF. The Arabidopsis nitrate transporter NRT1.7, expressed in phloem, is responsible for source-to-sink remobilization of nitrate. Plant Cell. 2009;21(9):2750–2761. doi: 10.1105/tpc.109.067603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jan A, et al. OsTZF1, a CCCH-tandem zinc finger protein, confers delayed senescence and stress tolerance in rice by regulating stress-related genes. Plant Physiol. 2013;161(3):1202–1216. doi: 10.1104/pp.112.205385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jiao BB, et al. A novel protein RLS1 with NB-ARM domains is involved in chloroplast degradation during leaf senescence in rice. Mol Plant. 2012;5(1):205–217. doi: 10.1093/mp/ssr081. [DOI] [PubMed] [Google Scholar]
- 35.Lee RH, Hsu JH, Huang HJ, Lo SF, Chen SC. Alkaline alpha-galactosidase degrades thylakoid membranes in the chloroplast during leaf senescence in rice. New Phytol. 2009;184(3):596–606. doi: 10.1111/j.1469-8137.2009.02999.x. [DOI] [PubMed] [Google Scholar]
- 36.Morita R, Sato Y, Masuda Y, Nishimura M, Kusaba M. Defect in non-yellow coloring 3, an alpha/beta hydrolase-fold family protein, causes a stay-green phenotype during leaf senescence in rice. Plant J. 2009;59(6):940–952. doi: 10.1111/j.1365-313X.2009.03919.x. [DOI] [PubMed] [Google Scholar]
- 37.Park SY, et al. The senescence-induced staygreen protein regulates chlorophyll degradation. Plant Cell. 2007;19(5):1649–1664. doi: 10.1105/tpc.106.044891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Uauy C, Distelfeld A, Fahima T, Blechl A, Dubcovsky J. A NAC Gene regulating senescence improves grain protein, zinc, and iron content in wheat. Science. 2006;314(5803):1298–1301. doi: 10.1126/science.1133649. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.