Abstract
Background
The aim of this study was to investigate the relationship between salivary concentration of the soluble fragment of the HER2 (human epidermal growth factor receptor) protein and its status in mammary tissues.
Methods
This case-control study was done in 27 breast cancer patients with no visible metastatic disease treated at the gynecology service, Maternity Souissi Hospital, Rabat, Morocco. Two groups were selected, ie, patients with positive and negative HER2 status in mammary tissue. The salivary HER2 protein concentration was assessed by enzyme-linked immunosorbent assay. The salivary HER2 concentration was compared between the HER2-positive and HER2-negative groups using the Mann-Whitney U test. A P-value <0.05 was considered to be statistically significant.
Results
No statistically significant difference in salivary HER2 protein expression was found between the case and control groups. There was also no significant difference in clinical characteristics according to positive and negative HER2 status (P>0.05), except for the progesterone hormone receptor which was statistically significant in both the case and control groups (P=0.047).
Conclusion
According to our data, salivary expression of the HER2 receptor may not be a reliable alternative to tissue assessment.
Keywords: breast cancer, HER2, saliva, diagnosis
Introduction
HER2 (human epidermal growth factor receptor 2) is a transmembrane growth receptor protein encoded by a proto-oncogene on chromosome 17q21.1 HER2 consists of an extracellular binding domain rich in cysteines, a lipophilic transmembrane domain, and an intracellular domain with kinase activity.2,3 Studying this protein is important for many reasons. HER2 is overexpressed in 20%–25% of breast cancers,4 gene amplification has been observed in 30% of metastatic breast cancers,5 and it is associated with a poor prognosis and resistance to certain chemotherapeutic agents.6,7 Moreover, targeting the HER2 protein could reduce the pathogenicity caused by overexpression of the gene.8,9 Trastuzumab (a recombinant humanized monoclonal antibody directed against the extracellular domain of HER2) and lapatinib are the two agents approved to treat invasive breast cancer with positive HER2 status.5,10 When selecting patients for targeted anti-HER2 therapy, the status of the protein can be determined by two methods, ie, immunohistochemistry (to assess the HER2 protein level in the tumor) and fluorescence in situ hybridization (for HER2 gene amplification).11 The high cost of targeted therapy and the risk of toxicity mean that unequivocal determination of positive HER2 status is necessary. Further, there has been considerable research interest in the HER2 extracellular domain found in biological fluids such as serum and saliva, with studies suggesting that its use may be promising for monitoring metastatic breast cancer.12,13 The extracellular domain of HER2 is released in blood after cleavage by ADAM (disintegrin and metalloproteinase) and can be determined using immunoenzymatic reaction techniques such as enzyme-linked immunosorbent assay (ELISA). Using this technique, a strong association was observed between the serum concentration of the soluble fragment of HER2 and its status in mammary tissues.14,15 The potential use of saliva as a diagnostic medium for breast cancer has also been discussed by some authors,13 on the basis of a positive correlation between saliva and serum in HER2 quantification.13 Consequently, assessment of the HER2 soluble fragment in saliva may be an alternative to invasive tissue analysis.16
The aim of this study was to assess the relationship between the saliva concentration of the soluble fragment of HER2 and its status in mammary tissues by anatomopathological methods.
Patients and methods
Study design
This case-control study was done in 27 patients diagnosed with breast cancer but no visible metastatic disease at the gynecology service, Maternity Souissi Hospital, Rabat, Morocco. The study protocol was approved by the local ethics committee for biomedical research in Morocco. Before entry to the study, all patients were required to sign a consent form and asked to answer a brief patient questionnaire concerning risk factors for breast cancer, eg, age of menarche, age at first birth, number of children, breastfeeding, tobacco use and alcohol intake, menopausal status, family history, and use of hormone replacement therapy and oral contraception.
Selection criteria
Patients with breast cancer were included if they were candidates for mastectomy, if they had undergone mastectomy, if they were receiving their first session of chemotherapy, and if they had no metastatic disease. Selection of cases and controls was based on HER2 status determined by immunohistochemistry as documented in medical records. Two groups were selected, ie, ten patients with HER2-positive status (score 3+) and 17 patients with HER2-negative status (scores 0 and 1+).17
Patients treated with trastuzumab or/and lapatinib were excluded, as were those with undetermined HER2 status or an equivocal HER2 score (2+).
Saliva collection
Saliva stimulated by chewing flavored sugar-free gum purchased from a local store was collected in the morning. All patients were asked not to eat, drink, or smoke for at least 2 hours before the test.18,19 Each patient was required to rinse her mouth several times and to sit for 5 minutes before collection of 5 mL of stimulated saliva in a plastic cup. The saliva samples were centrifuged at 2,000 rpm for 10 minutes; the supernatant was then stored at −80°C for later determination of HER2 concentration.
Determination of HER2 concentration
The salivary HER2 protein concentration was assessed using an ELISA kit (RayBiotech, Norcross GA, USA) designed to quantify serum HER2 levels. The protein was detected by sandwich reaction, whereby protein is trapped between two antibodies, the first one is attached to ELISA solid phase and the second recognizes the HER2 extracellular domain. After incubation, the ELISA plate was washed and antibodies HRP-conjugated streptavidin is added to each well. Then the plate was washed after incubation and TMB substrate solution is pipetted to wells. The intensity of the color developed is proportional to the amount of bound HER2. The reaction was then stopped and absorbance of each well was measured at 450 nm. The unknown HER2 concentration in the saliva samples was determined from the standard curve run after each test.
Statistical analysis
The statistical analysis was performed using Statistical Package for the Social Sciences version 13.0 software (SPSS Inc., Chicago, IL, USA). The HER2 protein concentration is expressed as the median and interquartile range, and age and body mass index are expressed as the mean and standard deviation. Demographic data, ie, age of menarche, age at the first birth, number of children, breastfeeding, adverse lifestyle habits, menopausal status, and hormone use are expressed as numbers and percentages. The clinical data, including histological type, tumor size, lymph node involvement, Scarff–Bloom–Richardson (SBR) grade, hormone receptor status, and history of vascular emboli are also expressed as numbers and percentages. The cases and controls were compared using the Mann–Whitney U test. A P-value <0.05 was considered to be statistically significant.
Results
The patient demographic data are summarized in Table 1, and show that 63% of patients started menstruating after the age of 13 years, 44.4% gave birth before the age of 25 years, 37.03% had 3–4 children, and 81.4% had breastfed their children. None of the patients smoked or drank alcohol, 25.9% were postmenopausal, and 11.1% had breast cancer antecedents. None of the patients used hormone replacement therapy during menopause; however, 70.4% had previously used oral contraceptives.
Table 1.
Demographic data based on participant questionnaire
| Patients (n=27) | |
|---|---|
| Age of menarche, years, n (%) | |
| <12 | 1 (3.7) |
| 12–13 | 9 (33.3) |
| >13 | 17 (63.0) |
| Age of first birth, years n (%) | |
| >25 | 12 (44.4) |
| 25–29 | 8 (29.6) |
| >30 | 2 (7.4) |
| Children, n (%) | |
| 0 | 5 (18.5) |
| 1–2 | 8 (29.6) |
| 3–4 | 10 (37.03) |
| >4 | 4 (14.8) |
| Breastfeeding, n (%) | 22 (81.4) |
| Adverse lifestyle habits, n (%) | |
| Smoking | 0 |
| Alcoholism | 0 |
| Menopause status, n (%) | 7 (25.9) |
| Family history, n (%) | 3 (11.1) |
| Hormone use, n (%) | |
| Oral contraception | 19 (70.4) |
| Hormone replacement therapy | 0 |
The clinical data are summarized in Table 2. No statistically significant differences were found between patients with positive and negative HER2 status with regard to age, body mass index, histological type of cancer, tumor size, lymph node status, SBR grade, estrogen receptor status, or history of vascular emboli (P>0.05). However, a statistically significant difference in the progesterone receptor was found between the two groups of patients (P=0.047).
Table 2.
Clinicopathological features of patients
| Patients with HER2(+) status (n=10) | Patients with HER2(−) status (n=17) | P-value | |
|---|---|---|---|
| Age, years (mean ± SD) | 43±5.79 | 46.82±10.05 | 0.28 |
| Body mass index (mean ± SD) | 27.46±4.99 | 25.53±4.69 | 0.32 |
| Histological type, n (%) | 1.00 | ||
| Invasive ductal carcinoma | 10 (100) | 15 (88.2) | |
| Papillary carcinoma | – | 1 (5.9) | |
| In situ papillary carcinoma | – | 1 (5.9) | |
| Tumor size, n (%) | 0.235 | ||
| T1 | 4 (40.0) | 2 (11.8) | |
| T2 | 3 (30.0) | 10 (58.8) | |
| T3 | 3 (30.0) | 5 (29.4) | |
| Regional lymph node involvement, n (%) | 0.921 | ||
| N0 | 4 (44.4) | 9 (56.3) | |
| N1 | 3 (33.3) | 4 (25.0) | |
| N2 | 1 (11.1) | 2 (12.5) | |
| N3 | 1 (11.1) | 1 (6.3) | |
| SBR grade, n (%) | 1.00 | ||
| I | 0 | 1 (5.9) | |
| II | 6 (60.0) | 10 (58.8) | |
| III | 4 (40.0) | 6 (35.3) | |
| Hormone receptors, n (%) | |||
| Progesterone receptor (+) | 6 (60.0) | 16 (94.1) | 0.047 |
| Estrogen receptor (+) | 5 (50.0) | 14 (82.4) | 0.102 |
| Vascular emboli (+) n (%) | 1 (12.5) | 4 (36.4) | 0.338 |
Abbreviations: SD, standard deviations; SBR, Scarff–Bloom–Richardson.
Comparison of the salivary HER2 concentration between the HER2-positive and HER2-negative breast cancer patients was not statistically significant (P=0.45, Figure 1).
Figure 1.
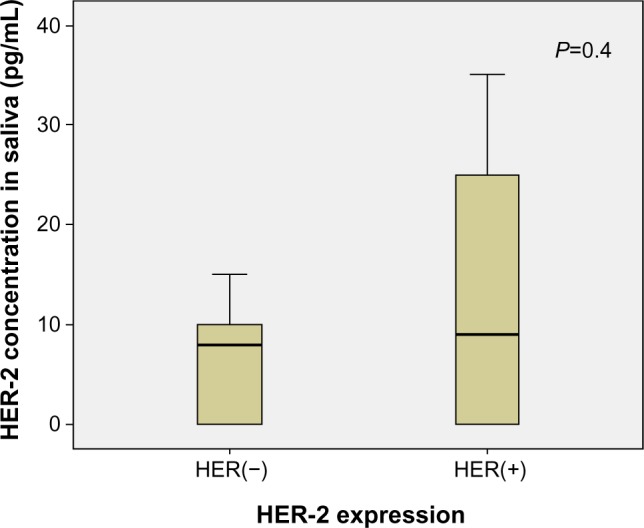
Salivary HER2 concentration according to positive and negative HER2 expression in mammary tissues.
Discussion
Our findings did not reveal a statistically significant difference in salivary HER2 protein concentration between cases and controls. Clinical characteristics collected from medical records (ie, age, body mass index, histological type of cancer, tumor size, lymph node involvement, SBR grade, estrogen and progesterone hormone receptor status, and history of vascular emboli) were compared between patients with positive and negative HER2 status, and no statistically significant difference was found between cases and controls, except for progesterone receptor expression. Many authors have reported a negative association between progesterone receptor expression and HER2 protein expression. In one report, overexpression of HER2 protein was strongly associated with negative estrogen and progesterone receptor status.20 Konecny et al and Mostafa et al also found an inverse association between steroid (estrogen and progesterone) receptor expression and HER2 expression,21,22 and Badzek et al reported an association between serum HER2 and certain clinical features, including hormone receptor status. However, no relationship with histological overexpression has been observed.23 Otherwise, there are many studies in the literature reporting reliable use of the soluble fragment of the HER2 protein in breast cancer monitoring.24 Assessment of serum HER2 using immunoenzymatic reaction methods such as ELISA is relatively rapid, and can be repeated many times during treatment.24 It can also be very useful for selecting patients eligible for targeted therapy and monitoring their response to this targeted therapy (trastuzumab) or response to chemotherapy.24–28 Further, comparison of serum HER2 concentration according to receptor expression in mammary tissues has shown a significant association between HER2 status in tumor tissue and serum HER2 concentrations in many studies.29–31 In addition, some authors have reported a strong association between serum HER2 concentration and receptor expression in mammary tissues.14,15
Developing a noninvasive sampling method for soluble HER2 protein in saliva would be highly desirable. Whole saliva contains a mixture of secretions from the salivary gland along with other constituents from the gingival crevicular fluid,32,33 which is essentially serum exudate.33 Thus, whole saliva reflects the constituents of serum and could have diagnostic potential in the detection of a number of biomarkers.34 Saliva is a perfect diagnostic medium, and this assay has many advantages.35,36 Sampling of saliva is technically easier than collection of blood, particularly when volunteers are invited for repeat sample collection.35,36 Otherwise, quantifying the soluble fragment of HER2 protein in saliva is possible.13 Streckfus et al suggested the diagnostic potential of HER2 and the possible use of saliva as a diagnostic medium based on the finding of positive and moderate correlations.13 However, the groups compared in their study were different from the groups chosen for our study, ie, patients with positive and negative HER2 status in mammary tissue. Their study was conducted in three groups, ie, healthy women, patients with benign breast tumors, and patients with breast cancer. Moreover, the comparison was based on biological fluids, ie, serum and saliva, and the tissue status of HER2 was not considered in patients with breast cancer in their study.13
Studies in patients with oral squamous cell carcinoma have yielded results consistent with our data. Bernardes et al reported that salivary HER2 levels were not elevated in patients with oral squamous cell carcinoma.37 Moreover the clinicopathological data, such as patient age, smoking habit, histological grading, T status, or nodal involvement of the tumor, the difference between patients with positive and negative HER2 overexpression in tumors was not statistically significant.37 No differences in salivary levels was remarked in patients with positive or negative HER2 status in tumors.37 Similar data were reported by Pardis et al, ie, salivary HER2 levels in patients with head and neck squamous cell carcinoma were not significantly higher than in healthy subjects, and there was no association between salivary HER2 levels and clinicopathological features such as age, sex, tumor grade, tumor size, or nodal status.38
The main limitation of our study is its small sample size, and this is attributed to the tissue determination of HER2 status, ie, many patients had an equivocal score (+2) by immunohistochemistry, so could not be included. This should encourage other studies with important sample size. In addition, the salivary concentrations should be compared to the assessment of the receptor HER2 in tumors.
In conclusion, our findings indicate that salivary expression of the HER2 receptor may not be a reliable alternative to tissue assessment because no association between salivary and tissue expression was found.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Coussens L, Yang-Feng TL, Lioa YC, et al. Tyrosine kinase receptor with extensive homology to EGF receptor shares chromosomal location with neu oncogene. Science. 1985;230(4730):1132–1139. doi: 10.1126/science.2999974. [DOI] [PubMed] [Google Scholar]
- 2.Nunes RA, Harris LN. The HER-2 extracellular domain as a prognostic and predictive factor in breast cancer. Clin Breast Cancer. 2002;3(2):125–135. doi: 10.3816/cbc.2002.n.017. [DOI] [PubMed] [Google Scholar]
- 3.Calzada V, Garcia F, Fernández M, et al. Labeling and biological evaluation of (99m)Tc-HYNIC-trastuzumab as a potential radiopharmaceutical for in vivo evaluation of HER2 expression in breast cancer. World J Nucl Med. 2013;12(1):27–32. doi: 10.4103/1450-1147.113953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neuoncogene. Science. 1987;235(4785):177–182. doi: 10.1126/science.3798106. [DOI] [PubMed] [Google Scholar]
- 5.Tsé C. sHER-2: a novel marker? Med Nucl. 2010;34(1):66–71. French. [Google Scholar]
- 6.Pellikainen J, Naukkarinen A, Ropponen K, et al. Expression of HER2 and its association with AP-2 in breast cancer. Eur J Cancer. 2004;40(10):1485–1495. doi: 10.1016/j.ejca.2004.02.020. [DOI] [PubMed] [Google Scholar]
- 7.Muss HB, Thor AD, Berry DA, et al. c-erbB-2 expression and response to adjuvant therapy in women with node-positive early breast cancer. N Engl J Med. 1994;330(18):1260–1266. doi: 10.1056/NEJM199405053301802. [DOI] [PubMed] [Google Scholar]
- 8.Piccart-Gebhart M, Procter M, Leyland-Jones B, et al. Trastuzumab after adjuvant chemotherapy in HER-2-positive breast cancer. N Engl J Med. 2005;353(16):1659–1672. doi: 10.1056/NEJMoa052306. [DOI] [PubMed] [Google Scholar]
- 9.Belkacemi Y, Gligorov J, Mauriac L, Azria D. HER-2/neu positive breast cancer: how to prescribe adjuvant trastuzumab (Herceptin)? Bull Cancer. 2006;93(10):991–999. [PubMed] [Google Scholar]
- 10.Nahta R, Yu D, Hung MC, Hortobagyi GN, Esteva FJ. Mechanisms of disease: understanding resistance to HER-2 targeted therapy in human breast cancer. Nat Clin Pract Oncol. 2006;3(5):269–280. doi: 10.1038/ncponc0509. [DOI] [PubMed] [Google Scholar]
- 11.Sauter G, Lee J, Barlett JMS, Slamon DJ, Press MF. Guidelines for human epidermal growth factor receptor 2 testing: biologic and methodologic considerations. J Clin Oncol. 2009;27(8):1323–1333. doi: 10.1200/JCO.2007.14.8197. [DOI] [PubMed] [Google Scholar]
- 12.McIntyre R, Bigler L, Dellinger T, Pfeifer M, Mannery T, Streckfus C. Oral contraceptive usage and the expression of CA 15-3 and c-erbB-2 in the saliva of healthy women. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88(6):687–690. doi: 10.1016/s1079-2104(99)70011-9. [DOI] [PubMed] [Google Scholar]
- 13.Streckfus C, Bigler L, Dellinger T, Dai X, Kingman A, Thigpen JT. The presence of soluble c-erbB-2 in saliva and serum among women with breast carcinoma: a preliminary study. Clin Cancer Res. 2000;6(6):2363–2370. [PubMed] [Google Scholar]
- 14.Kong Y, Dai S, Xie X, et al. High serum HER2 extracellular domain levels: correlation with a worse disease-free survival and overall survival in primary operable breast cancer patients. J Cancer Res Clin Oncol. 2012;138(2):275–284. doi: 10.1007/s00432-011-1095-9. [DOI] [PubMed] [Google Scholar]
- 15.Di Gioia D, Dresse M, Mayr D, et al. Serum HER2 supports HER2-testing in tissue at the time of primary diagnosis of breast cancer. Clin Chim Acta. 2014;430:86–91. doi: 10.1016/j.cca.2013.12.036. [DOI] [PubMed] [Google Scholar]
- 16.Fehm T, Becker S, Duerr-Stoerzer S, et al. Determination of HER2 status using both serum HER2 levels and circulating tumor cells in patients with recurrent breast cancer whose primary tumor was HER2 negative or of unknown HER2 status. Breast Cancer Res. 2007;9(5):1–8. doi: 10.1186/bcr1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dako . HercepTest working procedure. 18th edition. 2013. [Accessed June 6, 2014]. Available from: http://www.dako.com/dist/download.pdf?objectid=120856006. [Google Scholar]
- 18.Navazesh M, Christensen CM. A comparison of whole mouth resting and stimulated salivary measurement procedures. J Dent Res. 1982;61(10):1158–1162. doi: 10.1177/00220345820610100901. [DOI] [PubMed] [Google Scholar]
- 19.Nogourani MC, Janghorbani M, Isfahan RK, Beheshti MH. Effects of chewing different flavored gums on salivary flow rate and pH. Int J Dent. 2012;2012:569327. doi: 10.1155/2012/569327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Krishnaswamy U, Balachandran P, Rajakumar I, Balachandar K, Mansoor M. Correlation of HER-2 neu over-expression with clinicopathological features of carcinoma breast. Apollo Med. 2013;10(4):313–317. [Google Scholar]
- 21.Konecny G, Pauletti G, Pegram M, et al. Quantitative association between HER-2/neu and steroid hormone receptors in hormone receptor-positive primary breast cancer. J Natl Cancer Inst. 2003;95(2):142–153. doi: 10.1093/jnci/95.2.142. [DOI] [PubMed] [Google Scholar]
- 22.Mostafa NA, Eissa SS, Belal DM, Shoman SH. Assessment of Her-2/neu gene amplification status in breast carcinoma with equivocal 2+ Her-2/neu immunostaining. J Egypt Natl Canc Inst. 2011;23(1):41–46. doi: 10.1016/j.jnci.2011.07.006. [DOI] [PubMed] [Google Scholar]
- 23.Badzek S, Kelovic VL, Plestina S, Humar I, Veir Z, Mihaljevic Z. Serum HER2/ECD value in stage I and II early breast cancer: need of a lower cut-off? Wien Klin Wochenschr. 2011;123(23–24):726–731. doi: 10.1007/s00508-011-0099-4. [DOI] [PubMed] [Google Scholar]
- 24.Leyland-Jones B, Smith BS. Serum HER2 testing in patients with HER2-positive breast cancer: the death knell tolls. Lancet Oncol. 2011;12(3):286–295. doi: 10.1016/S1470-2045(10)70297-7. [DOI] [PubMed] [Google Scholar]
- 25.Köstler WJ, Schwab B, Singer CF, et al. Monitoring of serum Her-2/neu predicts response and progression-free survival to trastuzumab-based treatment in patients with metastatic breast cancer. Clin Cancer Res. 2004;10(5):1618–1624. doi: 10.1158/1078-0432.ccr-0385-3. [DOI] [PubMed] [Google Scholar]
- 26.Müller V, Witzel I, Lück HJ, et al. Prognostic and predictive impact of the HER-2/neu extracellular domain (ECD) in the serum of patients treated with chemotherapy for metastatic breast cancer. Breast Cancer Res Treat. 2004;86(1):9–18. doi: 10.1023/B:BREA.0000032919.83803.48. [DOI] [PubMed] [Google Scholar]
- 27.Colomer R, Montero S, Lluch A, et al. Circulating HER2 extracellular domain and resistance to chemotherapy in advanced breast cancer. Clin Cancer Res. 2000;6(6):2356–2362. [PubMed] [Google Scholar]
- 28.Fornier MN, Seidman AD, Schwartz MK, et al. Serum HER2 extracellular domain in metastatic breast cancer patients treated with weekly trastuzumab and paclitaxel: association with HER2 status by immunochemistry and fluorescence in situ hybridization and with response rate. Ann Oncol. 2005;16(2):234–239. doi: 10.1093/annonc/mdi059. [DOI] [PubMed] [Google Scholar]
- 29.Kong SY, Nam BH, Lee KS, et al. Predicting tissue HER2 status using serum HER2 levels in patients with metastatic breast cancer. Clin Chem. 2006;52(8):1510–1515. doi: 10.1373/clinchem.2006.067512. [DOI] [PubMed] [Google Scholar]
- 30.Tse C, Brault D, Gligorov J, et al. Evaluation of the quantitative analytical methods real-time PCR for HER-2 gene quantification and ELISA of serum HER-2 protein and comparison with fluorescence in situ hybridization and immunohistochemistry for determining HER-2 status in breast cancer patients. Clin Chem. 2005;51(7):1093–1101. doi: 10.1373/clinchem.2004.044305. [DOI] [PubMed] [Google Scholar]
- 31.Krainer M, Brodowicz T, Zellinger R, et al. Tissue expression and serum levels of HER-2/neu in patients with breast cancer. Oncology. 1997;54(6):475–481. doi: 10.1159/000227606. [DOI] [PubMed] [Google Scholar]
- 32.Al Kawas S, Rahim ZH, Ferguson DB. Potential uses of human salivary protein and peptide analysis in the diagnosis of disease. Arch Oral Biol. 2012;57(1):1–9. doi: 10.1016/j.archoralbio.2011.06.013. [DOI] [PubMed] [Google Scholar]
- 33.Lamster IB, Ahlo JK. Analysis of gingival crevicular fluid as applied to the diagnosis of oral and systemic diseases. Ann N Y Acad Sci. 2007;1098:216–229. doi: 10.1196/annals.1384.027. [DOI] [PubMed] [Google Scholar]
- 34.Wong DT. Salivary diagnostics powered by nanotechnologies, proteomics and genomics. J Am Dent Assoc. 2006;137(3):313–321. doi: 10.14219/jada.archive.2006.0180. [DOI] [PubMed] [Google Scholar]
- 35.Lac G. Intérêt et champs d’application des dosages salivaires [Relevance and usefulness of saliva assays] Sci Sports. 1998;13:55–63. French. [Google Scholar]
- 36.Hofman LF. Human saliva as a diagnostic specimen. J Nutr. 2001;131(5):1621S–1625S. doi: 10.1093/jn/131.5.1621S. [DOI] [PubMed] [Google Scholar]
- 37.Bernardes VF, Gleber-Netto FO, Sousa SF, Silva TA, Aguiar MC. Clinical significance of EGFR, Her-2 and EGF in oral squamous cell carcinoma: a case control study. J Exp Clin Cancer Res. 2010;29(40):1–7. doi: 10.1186/1756-9966-29-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pardis S, Sardari Y, Ashraf MJ, et al. Evaluation of tissue expression and salivary levels of HER2/neu in patients with head and neck squamous cell carcinoma. Iran J Otorhinolaryngol. 2012;24(69):161–170. [PMC free article] [PubMed] [Google Scholar]


