Abstract
The extracellular matrix polysaccharide hyaluronan (HA) exerts size-dependent effects on leukocyte behavior. Low-molecular weight HA is abundant at sites of active tissue catabolism and promotes inflammation via effects on Toll-like receptor signaling. Conversely, high-molecular weight HA is prevalent in uninjured tissues and is anti-inflammatory. We propose that the ability of high-molecular weight but not low-molecular weight HA to cross-link CD44 functions as a novel form of pattern recognition that recognizes intact tissues and communicates “tissue integrity signals” that promote resolution of local immune responses.
Keywords: Hyaluronan, Danger signals, DAMP, Integrity signal, CD44, ECM
Hyaluronan and the tissue response to injury
Hyaluronan (HA) is an extracellular matrix (ECM) glycos-aminoglycan that is abundant in the ECM of inflamed tissues. HA is a long, non-branching disaccharide made of glucuronic acid and N-acetyl-glucosamine with diverse effects on tissue structure and function (reviewed in [1–3]).
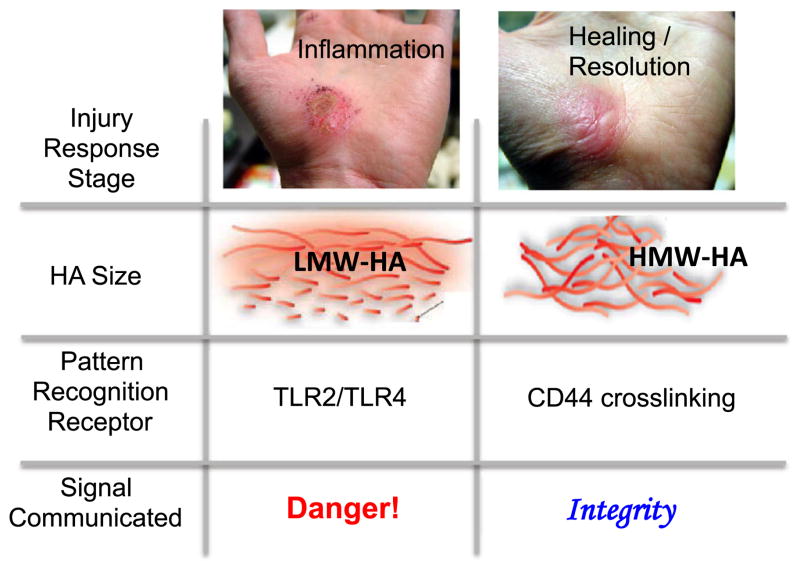
Both the size and the amount of HA are tightly regulated during progression through the stages of an injury response. Immediately upon injury, local HA production increases substantially [2, 3]. The three HA synthases responsible for this production generate predominantly high-molecular weight HA (HMW-HA) (defined here as >5 × 105 Da) [1, 4–6]. During inflammation, this HA is rapidly catabolized by a diverse group of host and (if infection is present) microbial hyaluronidases (HA’ases), mechanical forces, and oxidation [7, 8], resulting in fragmentary, low-molecular weight HA (defined here as <200 kDa) that are cleared via CD44-mediated endocytosis. Upon the resolution of inflammation, both the amount and size of HA return to basal levels. However, in chronically inflamed tissues, shorter HA polymers predominate. In light of these associations, HA size has been termed a natural biosensor for the state of tissue integrity [9, 10].
Here, we propose that the receptors that discriminate between HMW-HA and low-molecular weight hyaluronan (LMW-HA) together constitute an integrated system of pattern recognition capable of communicating the presence of either intact or fragmented ECM and, furthermore, that the resulting contextual cues are relevant for integrating wound healing with the local immune response to injury.
LMW-HA-mediated danger signals
Pattern recognition allows for efficient, choreographed responses to environmental stimuli. During infection, pathogen-associated molecular patterns (PAMPs) such as lipopolysaccharide (LPS), instigate rapid, programmatic responses that engender appropriately polarized immunologic responses. Endogenous markers of inflammation, termed danger-associated molecular patterns (DAMPs), function in an analogous manner to microbial PAMPs and trigger many of the same receptors [11, 12]. DAMPs share with PAMPs the properties of being small, structurally repetitive molecules. However, unlike PAMPs, DAMPs are also present in sterile inflammation. Examples of DAMPs include heat-shock proteins [13, 14], urate crystals [4, 15], and fragmentary components of the ECM [16, 17].
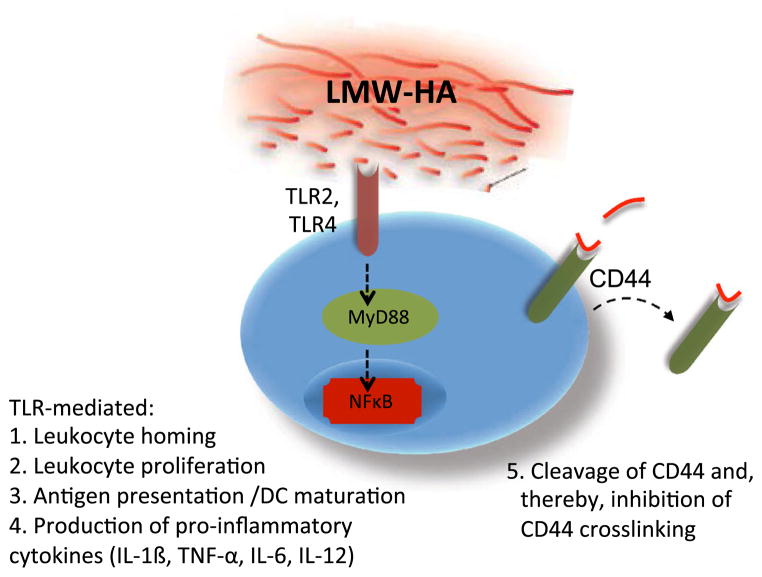
LMW-HA is an ECM molecule that functions as a pro-inflammatory DAMP [3, 18–24]. LMW-HA promotes the activation and maturation of dendritic cells (DC) [1, 25], drives the release of pro-inflammatory cytokines such as IL-1β, TNF-α, IL-6, and IL-12 by multiple cell types [6, 26–28], drives chemokine expression and cell trafficking [29–31], and promotes proliferation [32–34] (Fig. 1). These signals may be particularly relevant in settings of sterile inflammation.
Fig. 1.
Pro-inflammatory actions of LMW-HA and TLR signaling. LMW-HA characterizes inflamed tissues with active matrix catabolism. LMW-HA is an agonist of TLR signaling through interactions with TLR2 and/or TLR4 and communicates “danger signals” to infiltrating leukocytes. LMW-HA promotes leukocyte homing, proliferation and production of pro-inflammatory cytokine production as well as DC maturation and antigen presentation. LMW-HA also induces turnover of CD44, potentially limiting the likelihood of CD44 cross-linking by intact ECM
Many of the pro-inflammatory effects of LMW-HA are attributed to interactions with the pattern recognition receptors Toll-like receptor 2 (TLR2) or Toll-like receptor 4 (TLR4). LMW-HA promotes TLR-mediated phosphorylation of MAPK, nuclear translocation of NF-κB, and TNF-α production (reviewed in [3, 35]). While HA molecules of all sizes share the same repeating disaccharide structure, only LMW-HA can signal through TLR2 or TLR4 [1, 4, 6, 36]. Therefore, only products of HA catabolism, indicative of active inflammation, promote TLR signaling.
HMW-HA-mediated tissue integrity signals
HMW-HA predominates in healthy tissues and typically inhibits inflammation. Specifically, HMW-HA prevents cell growth and differentiation [7, 37], diminishes the production of inflammatory cytokines by multiple cell types [9, 38], and impairs phagocytosis by macrophages [11, 39]. Recently, HMW-HA has been implicated in the inhibition of tumor progression [13, 25]. Administration of HMW-HA is anti-inflammatory in lung injury models [4, 40], collagen-induced arthritis [16, 41], and a variety of other in vivo model systems [18, 20, 22–24, 42].
Most of these anti-inflammatory properties are attributable to interactions of HMW-HA and CD44, the major cell-surface HA-binding transmembrane glycoprotein. CD44 is thought to translate cues from the ECM, including HA, into signals that may influence growth, survival, activation, and differentiation [25, 43, 44]. Consistent with this, CD44−/−mice are unable to efficiently resolve inflammation. Initially, this was demonstrated in a bleomycin-induced lung injury model where CD44−/− mice have impaired clearance of apoptotic neutrophils, persistent accumulation of LMW-HA, and impaired activation of TGF-β1. Upon reconstitution with CD44+ leukocytes, the inflammation is resolved [6, 40, 43]. Similar defects were subsequently demonstrated in other injury models including bacterial pneumonia [29, 39]. Myocardial infarcts in CD44−/− mice are associated with reduced collagen deposition and fewer fibroblasts [32, 45], suggesting that the loss of CD44 impairs the effectiveness and efficiency of wound repair.
In addition to these anti-inflammatory effects, CD44 is reported to contribute to immune homeostasis via the maintenance of type 1 helper T (Th1) memory cells. Baaten and colleagues find that CD44 can counteract Fas-mediated apoptosis of these cells [35, 46]. These data suggest that despite the multiple roles for CD44 in leukocyte activation, survival, and costimulation, CD44 primarily promotes homeostasis and the resolution, rather than the propagation, of inflammation.
However, CD44 is not exclusively anti-inflammatory. CD44 interacts with a large number of ECM components, growth factors, and cytokines, and many of these factors, including osteopontin, are pro-inflammatory [36, 47]. Indeed, CD44 contributes to leukocyte trafficking [37, 48] and antibodies against CD44 that inhibit this trafficking have been proposed as a therapy for inflammatory and autoimmune diseases [38, 49]. CD44 is also implicated in TLR signaling in certain contexts [39, 50, 51]. We propose that CD44 is anti-inflammatory in those contexts in which it is cross-linked by HMW-HA and potentially other ECM ligands.
How HMW-HA and CD44 inhibit inflammation
Most downstream effects of CD44-binding HA and other ligands are mediated either via cellular re-organization (i.e., cytoskeleton or lipid raft architecture) or through receptor complexes that CD44 partners with (reviewed in [25, 52]). Several such mechanisms are highlighted in Fig. 2.
Fig. 2.
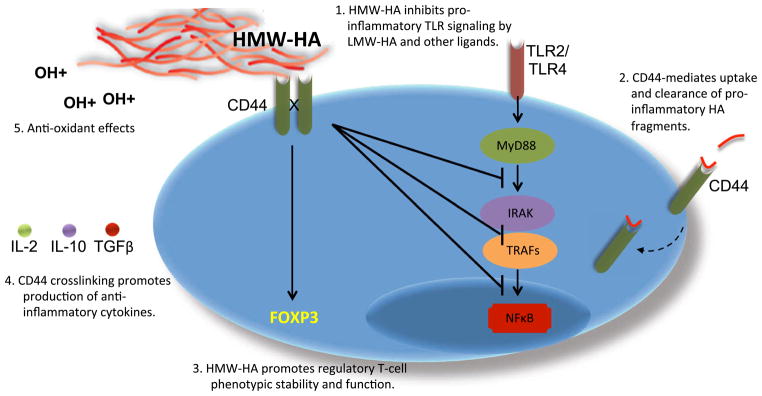
Anti-inflammatory actions of HMW-HA and CD44 cross-linking. Several mechanisms of anti-inflammatory activities propagated via HA and CD44 have been reported. 1. HMW-HA and CD44 negatively regulate pro-inflammatory TLR signaling at multiple levels. 2. CD44 is thought to play an important role in clearance of pro-inflammatory LMW-HA. 3. HMW-HA promotes the function and phenotypic stability of regulatory T-cell populations, including Foxp3+ Treg, TR1, and NKT cells. 4. CD44 cross-linking promotes production of anti-inflammatory cytokines. 5. HA is a potent antioxidant that limits the damage caused by free radicals generated at sites of inflammation. HA length probably does not impact this anti-oxidant property but is likely to contribute to its longevity. In light of these anti-inflammatory roles, we propose that HMW-HA functions as a tissue integrity signal that dampens inflammation at sites of intact tissues
HMW-HA negatively regulates pro-inflammatory TLR signaling. Mice treated with HMW-HA prior to LPS exposure have greatly decreased serum IL-6 and TNFα levels and are protected from symptoms of sepsis [36, 40]. In other studies, oral administration of HMW-HA likewise suppresses inflammation in a TLR4-dependent manner [41, 53] and actively downregulates LPS-induced NF-κB translocation [42, 54]. Consistent with these findings, both MyD88 expression levels and nuclear translocation of NF-κB are increased in CD44−/− mice [43, 44, 55, 56]. These mice also have diminished levels of several negative regulators of TLR signaling, including A20 and IRAK-M [35, 40, 43], suggesting that CD44 could govern expression of these molecules. In addition to inducing effects on TLR signaling downstream, CD44 physically associates with TLR4 and co-expressed MD-2 and facilitates the binding of LMW-HA to this complex [39, 57], presumably in the absence of HMW-HA. Together, these data suggest that HMW-HA and CD44 modulate TLR signaling at multiple points.
CD44 cross-linking may disproportionately impact cell types that are important for immune suppression, such as regulatory T cells (Tregs). The suppressive capacity of Tregs correlates with expression levels of the transcription factor, Foxp3 [45]. Foxp3+ Tregs are a specialized sub-population of CD4+ T cells that maintain immune homeostasis via production of IL-10, an anti-inflammatory cytokine critical for both immune regulation and for wound healing [58]. Depletion of Tregs leads to multi-systemic autoimmunity in both mice and humans [46], but can be prevented or ablated upon Treg adoptive transfer [47, 48].
HMW-HA promotes the function and phenotypic stability of Tregs. Firan and colleagues were the first to demonstrate an association between HMW-HA binding and Foxp3 expression [49, 59]. We subsequently reported that HMW-HA enhanced the function and viability of Tregs, particularly in low IL-2 environments [50, 51, 60]. Moreover, HMW-HA promotes Treg function via increased Foxp3 expression and production of IL-10 [36, 51, 52, 61]. Along similar lines, we find that HMW-HA, in the context of a TCR signal, induces conventional T cells to produce IL-10 and behave like type 1 regulatory cells (TR1), a subset of Tregs [36, 52]. Similarly, stimulation of CD44 through Ab cross-linking or HMW-HA causes immunoregulatory natural killer T cells (NKT) to secrete cytokines, up-regulate activation markers, and resist activation-induced cell death [50, 51, 53].
Another way CD44 may impact regulatory cell populations is through effects on antigenic responses. CD44 cross-linking is known to impact formation of TCR complexes [53, 54] and immune synapse function [55, 56, 62]. Therefore, CD44 may magnify low-level antigenic responses and provide tonic signals to these cells. As Treg and NKT cells both constitutively express high levels of CD44 receptor, they are poised to respond to such signals. Memory T cells are another group that is constitutively CD44 high and have impaired homeostasis in the absence of CD44 [35, 63].
CD44 cross-linking by HMW-HA as a novel form of pattern recognition
The ability of HA to bind to CD44 is dependent upon interactions with multiple CD44 molecules. This is because the interaction between HA and CD44 is mediated by low-affinity hydrogen bonds [25, 57] such that interactions with multiple receptors or increasing amounts of HA are required for efficient binding. In a study in which HA molecules bound to lipid nanoparticles were used to probe a CD44-coated surface, it was demonstrated that only HA molecules of >700 kDa could bind CD44 reliably [6, 59]. HA binding is further influenced by the density of CD44 receptors on the cell surface and their relative affinity for HA [60, 64]. While the precise size cutoffs vary between model systems, these data suggest that efficient HA binding to CD44 is predicated upon CD44 cross-linking.
The ability of HMW-HA to cross-link spatially isolated CD44 molecules and the inability of LMW-HA to do so may explain how leukocytes discriminate between HMW-HA and shorter HA polymers. Consistent with this, the anti-inflammatory effects attributed to HMW-HA in multiple systems can be recapitulated using antibody-mediated CD44 cross-linking [36, 51, 52, 61, 65]. We propose that CD44 cross-linking functions as a form of pattern recognition that distinguishes between tissue microenvironments that are actively inflamed, characterized by LMW-HA, and those that are healing or uninjured, characterized by HMW-HA. This model does not exclude other roles for CD44 but attributes the homeostatic and anti-inflammatory properties of HMW-HA to CD44 cross-linking. Consistent with this view, cross-linking is implicated in CD44-mediated effects on IL-10 production [36, 52, 66], Treg homeostasis [50, 51, 67], NKT cell function [53], neutrophil inhibition [62], and cell survival [63]. CD44 interacts with multiple other cell-surface receptors whose signaling influences leukocyte proliferation, maturation, activation, and trafficking [25], and we speculate that the contribution of HMW-HA and CD44 cross-linking to these pathways will prove to promote homeostasis and the resolution of inflammation.
Of note, HMW-HA and CD44 also reduce inflammation in ways that do not necessitate cross-linking. CD44 mediates clearance of HA fragments such that, in the absence of CD44, these fragments accumulate and drive inflammation through interactions with TLR [6]. Further, HA acts as an antioxidant and the greater the longevity of HMW-HA could translate into a prolonged ability to mitigate oxidative damage.
The integration of HA contextual cues
We propose a model whereby HA size-specific interactions with TLR2/TLR4 and CD44 together constitute an integrated system of pattern recognition that discriminates between actively inflamed and healing tissues based on the predominant size of HA. In this model, LMW-HA, typical of actively inflamed tissues, communicates pro-inflammatory “danger” signals via TLR signaling while HMW-HA, typical of post-inflammatory or uninflamed tissues, communicates “tissue integrity” signals via CD44 cross-linking. Together, these HA-mediated contextual signals constitute an integrated system for sensing changes in the inflammatory milieu and coordinating appropriate responses (Fig. 3). By “contextual signals,” we mean the information gained from the immune environment, specifically in the form of factors that engage co-stimulatory receptors.
Fig. 3.
Model for how the local size of HA may provide contextual signals in immune regulation at sites of sterile inflammation. We propose that the ability of different HA receptors to discriminate between polymers of different size functions as an integrated system of pattern recognition. LMW-HA, typical of inflamed wounds, promotes TLR signaling and functions as a pro-inflammatory “danger” signal. HMW-HA, typical of healing or uninjured tissues, cross-links CD44 and functions as an anti-inflammatory “tissue integrity” signal. Together, these HA-mediated contextual signals constitute an integrated system for sensing changes in the inflammatory milieu
Mechanisms must exist for the integration of these signals, given that HA molecules of disparate size are likely to coexist in injured tissues. Since HA molecules of different sizes may compete for binding to CD44, another variable that might impact competitive binding interactions is the molar predominance of LMW-HA generated from a single HMW-HA molecule. The relative expression of HA receptors may also be impacted by the HA molecules themselves. For example, LMW-HA is known to induce CD44 cleavage [64], and this could make less CD44 available for cross-linking. Additionally, HA may regulate its own catabolism; the enzymatic activity of the primary extracellular hyaluronidase, HYAL-2, is dependent on CD44 [65] along with other factors [66].
Other ECM components are likely to influence both HA integrity as well as interactions with CD44. In vivo, a diverse group of HA-binding molecules, called hyaladherins, contribute to particular HA structural architectures and may also contribute to CD44 cross-linking [67]. Hyaladherins and the nature and extent of HA superstructures may be essential to the anti-inflammatory properties of HMW-HA, an idea first proposed by Day and De La Motte [68].
An example of a hyaladherin with established roles in the cross-linking of CD44 is tumor necrosis α-stimulated gene 6 (TSG-6). TSG-6 catalyzes the covalent transfer of heavy chains of inter-α-inhibitor (IαI) present in serum to HA polymers, forming complex, cross-linked HA networks, promoting further interactions of HA with CD44 [69]. Such structures form the basis of provisional wound matrix, the scaffold that is a crucial early component of healing tissue. Additionally, CD44 binds heavy chain bound HA with greater avidity than unbound HA, promoting more efficient wound healing [70]. TSG-6 prevents HA degradation and inhibits enzymes involved in ECM catabolism, including HA’ases [68] [71]. TSG-6 levels are diminished at sites of chronic inflammation [72, 73], and, indeed, treatment with TSG-6 has been shown to be of benefit in some inflammatory disorders, particularly in the context of autoimmunity [74, 75]. Additionally, there are other hyaladherins, such as pro-inflammatory versican [76], that can inhibit HA–CD44 interactions.
Conclusions
The integration of wound healing and immune regulation is critical for the resolution of inflammatory responses but is poorly understood. Here, we have proposed that tissue repair and the immune response to tissue damage are synchronized by contextual cues that reflect the local integrity of the ECM, in particular the size of HA. Moreover, we have proposed that the receptors that discriminate between HA molecules of different sizes together constitute an integrated system of pattern recognition. This system of HA size-specific pattern recognition offers a powerful model for how tissue repair and immune regulation are integrated in injured and healing tissues.
This model also suggests that catabolic HA at sites of sterile inflammation may drive autoimmunity and predicts that it may be possible to impact HA integrity in vivo in ways that promote wound healing and immune tolerance. Indeed, delivery of exogenous HMW-HA is beneficial in both the amelioration of airway inflammation [77] and in limiting scar formation in dermatologic wounds [78]. Supplementation with TSG-6 to promote HA integrity is likewise a strategy for immunotherapy with known benefit in autoimmune arthritis [74] and other inflammatory settings.
However, many questions remain. For example, are there additional fragmentary ECM components, that are also CD44 ligands, that function in an analogous manner to HA? How are CD44 and TLR-mediated signals integrated? How do other HA receptors (e.g., RHAMM, LYVE-1, HARE) and the various sizes of HA polymers fit within this model of pattern recognition? Do clinical preparations of HMW-HA used to prevent abdominal adhesions (e.g., Seprafilm) or reduce joint inflammation (e.g., Synvisc) in fact provide tissue integrity signals? These questions and others warrant continued investigation into the role of HA in immune regulation.
Acknowledgments
This work was supported by National Institutes of Health grants T32 AI07290 (to SMR); R01 DK096087-01, R01 HL113294-01A1, and U01 AI101984 (to PLB); and HL018645 and a BIRT supplement AR037296 (to TNW).
Abbreviations
- HMW-HA
High-molecular weight hyaluronan
- LMW-HA
Low-molecular weight hyaluronan
- HA
Hyaluronan
- PAMPs
Pathogen-associated molecular patterns
- DAMPs
Damage-associated molecular patterns
- TLR
Toll-like receptor
- APC
Antigen-presenting cell
- DC
Dendritic cell
Footnotes
The authors declare that they have no conflict of interest.
Contributor Information
S. M. Ruppert, Division of Infectious Diseases, Stanford University School of Medicine, 300 Pasteur Drive, Rm. L-133, Stanford, CA 94305-5107, USA
T. R. Hawn, Division of Allergy and Infectious Diseases, University of Washington Medical Center, 1959 NE Pacific Ave, Seattle, WA 98195, USA
A. Arrigoni, Division of Infectious Diseases, Stanford University School of Medicine, 300 Pasteur Drive, Rm. L-133, Stanford, CA 94305-5107, USA
T. N. Wight, Matrix Biology Division, Benaroya Research Institute, 1201 9th Ave, Seattle, WA 98101, USA
P. L. Bollyky, Email: pbollyky@stanford.edu, Division of Infectious Diseases, Stanford University School of Medicine, 300 Pasteur Drive, Rm. L-133, Stanford, CA 94305-5107, USA
References
- 1.Termeer C, et al. Oligosaccharides of hyaluronan activate dendritic cells via toll-like receptor 4. J Exp Med. 2002;195:99–111. doi: 10.1084/jem.20001858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Laurent TC, et al. The structure and function of hyaluronan: an overview. Immunol Cell Biol. 1996;74:A1–7. doi: 10.1038/icb.1996.32. [DOI] [PubMed] [Google Scholar]
- 3.Jiang D, et al. Hyaluronan as an immune regulator in human diseases. Physiol Rev. 2011;91:221–64. doi: 10.1152/physrev.00052.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jiang D, et al. Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat Med. 2005;11:1173–9. doi: 10.1038/nm1315. [DOI] [PubMed] [Google Scholar]
- 5.Itano N, Kimata K. Mammalian hyaluronan synthases. IUBMB Life. 2002;54:195–9. doi: 10.1080/15216540214929. [DOI] [PubMed] [Google Scholar]
- 6.Teder P. Resolution of lung inflammation by CD44. Science. 2002;296:155–8. doi: 10.1126/science.1069659. [DOI] [PubMed] [Google Scholar]
- 7.Delmage JM, et al. The selective suppression of immunogenicity by hyaluronic acid. Ann Clin Lab Sci. 1986;16:303–10. [PubMed] [Google Scholar]
- 8.Stern R, Jedrzejas MJ. Hyaluronidases: their genomics, structures, and mechanisms of action. Chem Rev. 2006;106:818–39. doi: 10.1021/cr050247k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Neumann A, et al. High molecular weight hyaluronic acid inhibits advanced glycation endproduct-induced NF-kappaB activation and cytokine expression. FEBS Lett. 1999;453:283–7. doi: 10.1016/s0014-5793(99)00731-0. [DOI] [PubMed] [Google Scholar]
- 10.Stern R, et al. Hyaluronan fragments: an information-rich system. Eur J Cell Biol. 2006;85:699–715. doi: 10.1016/j.ejcb.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 11.Forrester JV, Balazs EA. Inhibition of phagocytosis by high molecular weight hyaluronate. Immunology. 1980;40:435–46. [PMC free article] [PubMed] [Google Scholar]
- 12.Suresh R, Mosser DM. Pattern recognition receptors in innate immunity, host defense, and immunopathology. Adv Physiol Educ. 2013;37:284–91. doi: 10.1152/advan.00058.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tian X, et al. High-molecular-mass hyaluronan mediates the cancer resistance of the naked mole rat. Nature. 2013 doi: 10.1038/nature12234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tsan M-F, Gao B. Heat shock proteins and immune system. J Leukoc Biol. 2009;85:905–10. doi: 10.1189/jlb.0109005. [DOI] [PubMed] [Google Scholar]
- 15.Gasse P, et al. Uric acid is a danger signal activating NALP3 inflammasome in lung injury inflammation and fibrosis. Am J Respir Crit Care Med. 2009;179:903–13. doi: 10.1164/rccm.200808-1274OC. [DOI] [PubMed] [Google Scholar]
- 16.Campo GM, et al. Hyaluronan reduces inflammation in experimental arthritis by modulating TLR-2 and TLR-4 cartilage expression. Biochim Biophys Acta. 2011;1812:1170–81. doi: 10.1016/j.bbadis.2011.06.006. [DOI] [PubMed] [Google Scholar]
- 17.Morwood SR, Nicholson LB. Modulation of the immune response by extracellular matrix proteins. Arch Immunol Ther Exp. 2006;54:367–74. doi: 10.1007/s00005-006-0043-x. [DOI] [PubMed] [Google Scholar]
- 18.Galeano M, et al. Systemic administration of high-molecular weight hyaluronan stimulates wound healing in genetically diabetic mice. Biochim Biophys Acta. 2011;1812:752–9. doi: 10.1016/j.bbadis.2011.03.012. [DOI] [PubMed] [Google Scholar]
- 19.Powell JD, Horton MR. Threat matrix: low-molecular-weight hyaluronan (HA) as a danger signal. Immunol Res. 2005;31:207–18. doi: 10.1385/IR:31:3:207. [DOI] [PubMed] [Google Scholar]
- 20.Hašová M, et al. Hyaluronan minimizes effects of UV irradiation on human keratinocytes. Arch Dermatol Res. 2011;303:277–84. doi: 10.1007/s00403-011-1146-8. [DOI] [PubMed] [Google Scholar]
- 21.Tesar BM, et al. The role of hyaluronan degradation products as innate alloimmune agonists. Am J Transpl. 2006;6:2622–35. doi: 10.1111/j.1600-6143.2006.01537.x. [DOI] [PubMed] [Google Scholar]
- 22.Huang PM, et al. High MW hyaluronan inhibits smoke inhalation-induced lung injury and improves survival. Respirology. 2010;15:1131–9. doi: 10.1111/j.1440-1843.2010.01829.x. [DOI] [PubMed] [Google Scholar]
- 23.Ye J, et al. High molecular weight hyaluronan decreases oxidative DNA damage induced by EDTA in human corneal epithelial cells. Eye (Lond) 2012;26:1012–20. doi: 10.1038/eye.2012.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Austin JW, et al. High molecular weight hyaluronan reduces lipopolysaccharide mediated microglial activation. J Neurochem. 2012;122:344–55. doi: 10.1111/j.1471-4159.2012.07789.x. [DOI] [PubMed] [Google Scholar]
- 25.Ponta H, et al. CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol. 2003;4:33–45. doi: 10.1038/nrm1004. [DOI] [PubMed] [Google Scholar]
- 26.Yamawaki H, et al. Hyaluronan receptors involved in cytokine induction in monocytes. Glycobiology. 2008;19:83–92. doi: 10.1093/glycob/cwn109. [DOI] [PubMed] [Google Scholar]
- 27.De la Motte C, et al. Platelet-derived hyaluronidase 2 cleaves hyaluronan into fragments that trigger monocyte-mediated production of proinflammatory cytokines. Am J Pathol. 2009;174:2254–64. doi: 10.2353/ajpath.2009.080831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Campo GM, et al. Small hyaluronan oligosaccharides induce inflammation by engaging both toll-like-4 and CD44 receptors in human chondrocytes. Biochem Pharmacol. 2010;80:480–90. doi: 10.1016/j.bcp.2010.04.024. [DOI] [PubMed] [Google Scholar]
- 29.van der Windt GJW, et al. The role of CD44 in the acute and resolution phase of the host response during pneumococcal pneumonia. Lab Invest. 2011;91:588–97. doi: 10.1038/labinvest.2010.206. [DOI] [PubMed] [Google Scholar]
- 30.McKee CM, et al. Hyaluronan (HA) fragments induce chemokine gene expression in alveolar macrophages. The role of HA size and CD44. J Clin Invest. 1996;98:2403–13. doi: 10.1172/JCI119054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Horton MR, et al. Regulation of hyaluronan-induced chemokine gene expression by IL-10 and IFN-gamma in mouse macrophages. J Immunol. 1998;160:3023–30. [PubMed] [Google Scholar]
- 32.Huebener P, et al. CD44 is critically involved in infarct healing by regulating the inflammatory and fibrotic response. J Immunol. 2008;180:2625–33. doi: 10.4049/jimmunol.180.4.2625. [DOI] [PubMed] [Google Scholar]
- 33.Scheibner KA, et al. Hyaluronan fragments act as an endogenous danger signal by engaging TLR2. J Immunol. 2006;177:1272–81. doi: 10.4049/jimmunol.177.2.1272. [DOI] [PubMed] [Google Scholar]
- 34.Zheng L, et al. Regulation of colonic epithelial repair in mice by toll-like receptors and hyaluronic acid. YGAST. 2009;137:2041–51. doi: 10.1053/j.gastro.2009.08.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Baaten BJG, et al. CD44 regulates survival and memory development in Th1 cells. Immunity. 2010;32:104–15. doi: 10.1016/j.immuni.2009.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bollyky PL, et al. ECM components guide IL-10 producing regulatory T-cell (TR1) induction from effector memory T-cell precursors. Proc Natl Acad Sci USA. 2011;108:7938–43. doi: 10.1073/pnas.1017360108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cuff CA, et al. The adhesion receptor CD44 promotes atherosclerosis by mediating inflammatory cell recruitment and vascular cell activation. J Clin Invest. 2001;108:1031–40. doi: 10.1172/JCI12455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Naor D, et al. CD44 involvement in autoimmune inflammations: the lesson to be learned from CD44-targeting by antibody or from knockout mice. Ann NY Acad Sci. 2007;1110:233–47. doi: 10.1196/annals.1423.025. [DOI] [PubMed] [Google Scholar]
- 39.Taylor KR, et al. Recognition of hyaluronan released in sterile injury involves a unique receptor complex dependent on Toll-like receptor 4, CD44, and MD-2. J Biol Chem. 2007;282:18265–75. doi: 10.1074/jbc.M606352200. [DOI] [PubMed] [Google Scholar]
- 40.Muto J, et al. Engagement of CD44 by hyaluronan suppresses TLR4 signaling and the septic response to LPS. Mol Immunol. 2009;47:449–56. doi: 10.1016/j.molimm.2009.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Asari A, et al. Oral administration of high molecular weight hyaluronan (900 kDa) controls immune system via Toll-like receptor 4 in the intestinal epithelium. J Biol Chem. 2010;285:24751–8. doi: 10.1074/jbc.M110.104950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yasuda T. Hyaluronan inhibits Akt, leading to nuclear factor-κB down-regulation in lipopolysaccharide-stimulated U937 macrophages. J Pharmacol Sci. 2011;115:509–15. doi: 10.1254/jphs.10244fp. [DOI] [PubMed] [Google Scholar]
- 43.Liang J, et al. CD44 is a negative regulator of acute pulmonary inflammation and lipopolysaccharide-TLR signaling in mouse macrophages. J Immunol. 2007;178:2469–75. doi: 10.4049/jimmunol.178.4.2469. [DOI] [PubMed] [Google Scholar]
- 44.Kawana H, et al. CD44 suppresses TLR-mediated inflammation. J Immunol. 2008;180:4235–45. doi: 10.4049/jimmunol.180.6.4235. [DOI] [PubMed] [Google Scholar]
- 45.Sakaguchi S, et al. FOXP3 + regulatory T cells in the human immune system. Nat Rev Immunol. 2010;10:490–500. doi: 10.1038/nri2785. [DOI] [PubMed] [Google Scholar]
- 46.Wildin RS, et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat Genet. 2001;27:18–20. doi: 10.1038/83707. [DOI] [PubMed] [Google Scholar]
- 47.Huter EN, et al. TGF-β-induced Foxp3 +regulatory T cells rescue scurfy mice. Eur J Immunol. 2008;38:1814–21. doi: 10.1002/eji.200838346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tang Q, et al. CD4(+)Foxp3(+) regulatory T cell therapy in transplantation. J Mol Cell Biol. 2012;4:11–21. doi: 10.1093/jmcb/mjr047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Firan M, et al. Suppressor activity and potency among regulatory T cells is discriminated by functionally active CD44. Blood. 2006;107:619–27. doi: 10.1182/blood-2005-06-2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bollyky PL, et al. Cutting edge: high molecular weight hyaluronan promotes the suppressive effects of CD4 + CD25 + regulatory T cells. J Immunol. 2007;179:744–7. doi: 10.4049/jimmunol.179.2.744. [DOI] [PubMed] [Google Scholar]
- 51.Bollyky PL, et al. Intact extracellular matrix and the maintenance of immune tolerance: high molecular weight hyaluronan promotes persistence of induced CD4 + CD25 + regulatory T cells. J Leukoc Biol. 2009;86:567–72. doi: 10.1189/jlb.0109001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bollyky PL, et al. CD44 costimulation promotes FoxP3 + regulatory T cell persistence and function via production of IL-2, IL-10, and TGF-beta. J Immunol. 2009;183:2232–41. doi: 10.4049/jimmunol.0900191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Larkin J, et al. CD44 differentially activates mouse NK T cells and conventional T cells. J Immunol. 2006;177:268–79. doi: 10.4049/jimmunol.177.1.268. [DOI] [PubMed] [Google Scholar]
- 54.Föger N, et al. CD44 supports T cell proliferation and apoptosis by apposition of protein kinases. Eur J Immunol. 2000;30:2888–99. doi: 10.1002/1521-4141(200010)30:10<2888::AID-IMMU2888>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 55.Bollyky PL, et al. Th1 cytokines promote T-cell binding to antigen-presenting cells via enhanced hyaluronan production and accumulation at the immune synapse. Cell Mol Immunol. 2010;7:211–20. doi: 10.1038/cmi.2010.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hegde VL, et al. CD44 mobilization in allogeneic dendritic cell-T cell immunological synapse plays a key role in T cell activation. J Leukoc Biol. 2008;84:134–42. doi: 10.1189/jlb.1107752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Banerji S, et al. Structures of the Cd44-hyaluronan complex provide insight into a fundamental carbohydrate-protein interaction. Nat Struct Mol Biol. 2007;14:234–9. doi: 10.1038/nsmb1201. [DOI] [PubMed] [Google Scholar]
- 58.King A, et al. Interleukin-10 regulates the fetal hyaluronan-rich extracellular matrix via a STAT3-dependent mechanism. J Surg Res. 2013;184:671–7. doi: 10.1016/j.jss.2013.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Mizrahy S, et al. Hyaluronan-coated nanoparticles: the influence of the molecular weight on CD44-hyaluronan interactions and on the immune response. J Control Release. 2011;156:231–8. doi: 10.1016/j.jconrel.2011.06.031. [DOI] [PubMed] [Google Scholar]
- 60.Wolny PM, et al. Analysis of CD44-hyaluronan interactions in an artificial membrane system: insights into the distinct binding properties of high and low molecular weight hyaluronan. J Biol Chem. 2010;285:30170–80. doi: 10.1074/jbc.M110.137562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fujii Y, et al. Crosslinking of CD44 on human osteoblastic cells upregulates ICAM-1 and VCAM-1. FEBS Lett. 2003;539:45–50. doi: 10.1016/s0014-5793(03)00182-0. [DOI] [PubMed] [Google Scholar]
- 62.Hutás G, et al. CD44-specific antibody treatment and CD44 deficiency exert distinct effects on leukocyte recruitment in experimental arthritis. Blood. 2008;112:4999–5006. doi: 10.1182/blood-2008-04-150383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Marhaba R, et al. CD44v6 promotes proliferation by persisting activation of MAP kinases. Cell Signal. 2005;17:961–73. doi: 10.1016/j.cellsig.2004.11.017. [DOI] [PubMed] [Google Scholar]
- 64.Sugahara KN, et al. Hyaluronan oligosaccharides induce CD44 cleavage and promote cell migration in CD44-expressing tumor cells. J Biol Chem. 2003;278:32259–65. doi: 10.1074/jbc.M300347200. [DOI] [PubMed] [Google Scholar]
- 65.Harada H, Takahashi M. CD44-dependent intracellular and extracellular catabolism of hyaluronic acid by hyaluronidase-1 and -2. J Biol Chem. 2007;282:5597–607. doi: 10.1074/jbc.M608358200. [DOI] [PubMed] [Google Scholar]
- 66.Bourguignon LYW, et al. CD44 interaction with Na +-H + exchanger (NHE1) creates acidic microenvironments leading to hyaluronidase-2 and cathepsin B activation and breast tumor cell invasion. J Biol Chem. 2004;279:26991–7007. doi: 10.1074/jbc.M311838200. [DOI] [PubMed] [Google Scholar]
- 67.Day AJ, Prestwich GD. Hyaluronan-binding proteins: tying up the giant. J Biol Chem. 2002;277:4585–8. doi: 10.1074/jbc.R100036200. [DOI] [PubMed] [Google Scholar]
- 68.Day AJ, De la Motte CA. Hyaluronan cross-linking: a protective mechanism in inflammation? Trends Immunol. 2005;26:637–43. doi: 10.1016/j.it.2005.09.009. [DOI] [PubMed] [Google Scholar]
- 69.Baranova NS, et al. The inflammation-associated protein TSG-6 cross-links hyaluronan via hyaluronan-induced TSG-6 oligomers. J Biol Chem. 2011;286:25675–86. doi: 10.1074/jbc.M111.247395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhuo L, et al. SHAP potentiates the CD44-mediated leukocyte adhesion to the hyaluronan substratum. J Biol Chem. 2006;281:20303–14. doi: 10.1074/jbc.M506703200. [DOI] [PubMed] [Google Scholar]
- 71.Lesley J, et al. TSG-6 modulates the interaction between hyaluronan and cell surface CD44. J Biol Chem. 2004;279:25745–54. doi: 10.1074/jbc.M313319200. [DOI] [PubMed] [Google Scholar]
- 72.Tan KT, et al. Characterization of hyaluronan and TSG-6 in skin scarring: differential distribution in keloid scars, normal scars and unscarred skin. J Eur Acad Dermatol Venereol. 2011;25:317–27. doi: 10.1111/j.1468-3083.2010.03792.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kvezereli M, et al. TSG-6 protein expression in the pancreatic islets of NOD mice. J Mol Histol. 2008;39:585–93. doi: 10.1007/s10735-008-9199-5. [DOI] [PubMed] [Google Scholar]
- 74.Bárdos T, et al. Anti-inflammatory and chondroprotective effect of TSG-6 (tumor necrosis factor-alpha-stimulated gene-6) in murine models of experimental arthritis. Am J Pathol. 2001;159:1711–21. doi: 10.1016/s0002-9440(10)63018-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kota DJ, et al. TSG-6 produced by hMSCs delays the onset of autoimmune diabetes by suppressing Th1 development and enhancing tolerogenicity. Diabetes. 2013;62:2048–58. doi: 10.2337/db12-0931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Evanko SP, et al. Hyaluronan and versican in the control of human T-lymphocyte adhesion and migration. Matrix Biol. 2012;31:90–100. doi: 10.1016/j.matbio.2011.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Liu Y, et al. High-molecular-weight hyaluronan—a possible new treatment for sepsis-induced lung injury: a preclinical study in mechanically ventilated rats. Crit Care. 2008;12:R102. doi: 10.1186/cc6982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Voigt J, Driver VR. Hyaluronic acid derivatives and their healing effect on burns, epithelial surgical wounds, and chronic wounds: a systematic review and meta-analysis of randomized controlled trials. Wound Repair Regen. 2012;20:317–31. doi: 10.1111/j.1524-475X.2012.00777.x. [DOI] [PubMed] [Google Scholar]