Abstract
Post-translational modification of proteins by members of the small ubiquitin-like modifier (SUMO) is involved in diverse cellular functions. Many viral proteins are SUMO targets and also interact with the cellular SUMOylation system. During human cytomegalovirus (HCMV) infection, the immediate-early (IE) proteins IE1 and IE2 are covalently modified by SUMO. IE2 SUMOylation promotes its transactivation activity, whereas the role of IE1 SUMOylation is not clear. We performed in silico, genome-wide analysis to identify possible SUMOylation sites in HCMV-encoded proteins and evaluated their modification using the E. coli SUMOylation system and in vitro assays. We found that only IE1 and IE2 are substantially modified by SUMO in E. coli, although US34A was also identified as a possible SUMO target in vitro. We also found that SUMOylation of IE1 and IE2 is temporally regulated during viral infection. Levels of SUMO-modified form of IE1 were increased during the early phase of infection, but decreased in the late phase when IE2 and its SUMO-modified forms were expressed at high levels. IE2 expression inhibited IE1 SUMOylation in cotransfection assays. As in IE2 SUMOylation, PIAS1, a SUMO E3 ligase, interacted with IE1 and enhanced IE1 SUMOylation. In in vitro assays, an IE2 fragment that lacked covalent and non-covalent SUMO attachment sites, but was sufficient for PIAS1 binding, effectively inhibited PIAS1-mediated SUMOylation of IE1, indicating that IE2 expression negatively regulates IE1 SUMOylation. We also found that the IE2-mediated downregulation of IE1 SUMOylation correlates with the IE1 activity to repress the promoter containing the interferon stimulated response elements. Taken together, our data demonstrate that IE1 and IE2 are the main viral SUMO targets in HCMV infection and that temporal regulation of their SUMOylation may be important in the progression of this infection.
Introduction
Small ubiquitin-like modifier (SUMO) proteins are members of the ubiquitin-like protein family. Covalent modification of proteins by SUMO (SUMOylation) affects their activity, intracellular localization, stability, and interaction with other proteins and DNA. The cellular SUMOylation pathway, which is largely analogous to the ubiquitin modification pathway, regulates many important cellular processes [1], [2]. In brief, SUMO precursors are C-terminally processed to create an active form, which is activated by the formation of a thioester bond between the C-terminal glycine residue of SUMO and the active cysteine reside of a heterodimeric E1 activation enzyme, which comprises SAE1 and SAE2. SUMO is then transferred to the E2 conjugation enzyme, Ubc9, via an analogous thioester bond, and finally to the lysine residue of a substrate. SUMO E3 ligases, such as PIAS proteins, RanBP2, and Pc2, help transfer SUMO from Ubc9 to the substrate [3]–[5]. On most substrates, SUMO is conjugated to a lysine residue through an isopeptide linkage within the consensus sequence ΨKxE/D (where Ψ is a bulky hydrophobic residue and x is any amino acid), which is often found in the disordered region of proteins [6]–[9]. Both Ubc9 and the E3 ligases appear to control the substrate specificity of SUMOylation. SUMO can be released from a substrate through cleavage by proteases called SENP; therefore, SUMOylation is reversible [10]–[12]. Proteins also can interact with SUMO non-covalently through a SUMO-interacting motif (SIM), which is characterized by a stretch of hydrophobic residues, often flanked by acidic residues [13]–[16].
Evidence is accumulating that the cellular SUMOylation pathway plays a regulatory role in infection by many different viruses, including human cytomegalovirus (HCMV) [17], [18]. HCMV is an opportunistic pathogen that can cause congenital disease and produces serious disease complications in immunocompromised individuals. During the lytic cycle of HCMV infection, viral genes are expressed in a cascade fashion with immediate-early (IE), early, and late phases. The 72-kDa IE1 (also known as IE1-p71 or IE72) and 86-kDa IE2 (IE2-p86 or IE86) proteins are the major IE proteins that regulate activation of viral genes and modulate host cell functions [19]. Both IE1 and IE2 are modified by SUMO during HCMV infection.
IE2 is a strong transactivator that interacts with numerous cellular transactivators and is essential for early and late viral gene expression. IE2 is modified by SUMO at two lysine residues, K175 and K180. In transfection assays, SUMOylation of IE2 enhances the transactivation of diverse cellular and viral promoters by IE2 [20], [21]. Consistently, transactivation activity of IE2 has been correlated with its degree of SUMOylation [22]. IE2 directly binds to Ubc9 [20], [21] and PIAS1 [23]. Mutation of both K175 and K180 in a laboratory strain and a clinical isolate caused a modest decrease in virus replication, indicating that IE2 SUMOylation promotes the virus lytic cycle in the context of virus infection [24]. However, the effect of IE2 SUMOylation on viral growth appears to depend on the virus strains and infection conditions, since similar mutations in another laboratory strain did not significantly affect viral growth [25]. IE2 also non-covalently interacts with SUMO through a SIM adjacent to the SUMO conjugation sites. This SIM is necessary for efficient SUMOylation and transactivation activity of IE2, thereby promoting viral growth [24], [26]. The IE2 SIM promotes transactivation by IE2 by recruiting other SUMO-modified transcription cofactors, such as TAF12 [26].
IE1 is required for efficient viral gene expression, particularly at a low multiplicity of infection [27], [28]. IE1 also plays a key role in disarming host intrinsic and innate antiviral responses. IE1 disrupts PML nuclear bodies (NBs), also known as nuclear domain 10 (ND10) [29]–[32]. This activity correlates with the loss of SUMOylated PML NB components, such as PML and Sp100, which repress incoming viral genomes [33]–[35]. IE1 also interferes with type I interferon (IFN) signaling by directly targeting STAT2 using its near C-terminal region, and, to a lesser extent, by binding to STAT1 [36]–[38]. IE1 is modified by SUMO at K450 within the acidic domain [39], [40]. The role of IE1 SUMOylation in virus infection is unclear. IE1 SUMOylation has been reported to promote viral growth, while other studies have found a lack of significant impact [40]–[42]. We previously found that the SUMO-modified form of IE1 failed to interact with STAT2, suggesting that SUMOylation of IE1 may inhibit the ability of IE1 to downregulate type I IFN signaling [37]. The SUMOylation site of IE1 is close to its C-terminal chromatin-tethering domain; however, IE1 SUMOylation did not affect IE1 accumulation at mitotic chromosomes [43]. Phosphorylation of IE1 has been reported to decrease its SUMOylation [44].
In this study, we performed an in silico genome-wide analysis to identify HCMV-encoded SUMO targets. We found that viral IE1 and IE2 proteins might be the main SUMO targets. We also investigated whether SUMOylation of IE1 and IE2 is regulated during HCMV infection. Our results showed that high-level expression of IE2 and its SUMO-modified forms at the late stage of infection downregulates IE1 SUMOylation via competing PIAS1 binding, potentiating IE1 repression of interferon-stimulated gene (ISG) activation.
Materials and Methods
Plasmids
pSG5 [45]-based expression plasmids for IE1 (pJHA303), IE2 (pJHA124), GST-IE2(346–542) (pHJK13), flag-SUMO-1 (pJHA312), and flag-SUMO-2 (pJHA342) were previously described [21], [23], [42]. Plasmids for HA-IE1 (pDJK170), HA-UL53 (pMK56), and GST-IE1 (pDJK175) were produced by moving the cDNAs from pENTR vectors (Invitrogen) to pSG5-HA and pGEX-3-based destination vectors, respectively, using LR Clonase (Invitrogen). Similarly, plasmid for His-IE1 (pSHJ9) was produced with the pDEST17 (with a 6His tag) destination vector (Invitrogen), and plasmids for HA-PIAS1 (pHJK1), SRT-PIAS1 (pSAN22), myc-PIAS1 (RYK595), and myc-IE2(346–542) (pRYK593) were produced with the pSG5-HA, pSG5-SRT or pCS3-MT (with a 6myc tag) [46]-based destination vectors. pCMV-Flag-PIAS1 was kindly provided by Ke Shuai (UCLA, Los Angeles, CA, USA). Plasmids for GST-SAE2/SAE1, in which GST-SAE2 and SAE1 are translationally linked via a ribosome binding site, His-Ubc9, and GST-SUMO-1GG were previously described [47], and the plasmid for His-SUMO-1GG was produced with the pDEST17 destination vector using LR Clonase. pT-E1E2S1, which encodes the E1 and E2 enzymes for SUMO conjugation as well as the active form of SUMO-1 [48], was used to introduce a synthetic SUMO-1 conjugation pathway into E. coli.
Cloning HCMV ORFs
HCMV open reading frames (ORFs) were cloned as previously described [49]. HCMV ORFs were PCR amplified using primers based on the GenBank sequences AY446894, GU937742, and FJ616285. Bacterial artificial chromosomes Toledo-BAC [50] and Towne-BAC [51] were used as templates (gifts from H. Zhu, UMDNJ-New Jersey Medical School, Newark, New Jersey, USA). The 5′ primers contained the attB1 recombination site, and the 3′ primers contained the attB2 recombination site (attB1, 5′-GGGGACAAGTTTGTACAAAAAAGCAGGCTCC-3′; attB2, 5′-GGGGACCACTTTGTACAAGAAAGCTGGGTC-3′) (Invitrogen). Some long ORFs were amplified in segments. For some ORFs that encode spliced products, cDNAs prepared form virus-infected cells were used for PCR amplification. PCR products of the correct size were recombined into the gateway vector pDONR201 (to make pENTR clones) using BP Clonase (Invitrogen). E. coli that had been transformed with the reaction products (pENTR clones) were selected, and the DNA inserts were analyzed by digestion with BsrGI and sequencing. Yeast cells expressing plasmids encoding GAL4-activation domain (AD)-ORF fusions were produced by transferring the ORFs from pENTR vectors to a pACTII [21]-based destination vector using LR Clonase.
Transfection
293T cells were transfected via the N,N-bis-(2-hydroxyethyl)-2-aminoethanesulfonic acid-buffered saline (BBS) version of the calcium phosphate method, as described previously [23].
Immunoblot analysis
Samples were prepared by boiling in loading buffer, separated by SDS-PAGE, and transferred to a nitrocellulose membrane (Schleicher & Schuell, Dassel, Germany). The membrane was blocked for 1 h in PBS-T [PBS plus 0.1% Tween-20 (Sigma)] containing 5% skim milk and then washed with PBS-T. After incubation with the appropriate antibody, the proteins were visualized by the standard procedure using an enhanced chemiluminescence system (Roche). For SUMOylation assays in transfected cells, cells were washed with PBS containing 5 mM NEM, and the samples were prepared by boiling in SDS loading buffer.
Coimmunoprecipitation (CoIP) assays
293T (8×105 in 100-mm dish) cells were harvested and sonicated in 1 ml CoIP buffer (50 mM Tris-Cl [pH 7.4], 50 mM NaF, 5 mM sodium phosphate, 0.1% Triton X-100, containing protease inhibitors [Sigma]) using a microtip probe (Vibra cell; Sonics and Materials, Inc., USA) for 10 sec (pulse on: l sec, pulse off: 3 sec). Clarified cell lysates were incubated for 16 h with appropriate antibodies at 4°C. Thirty microliters of a 50% slurry of protein A- and G-Sepharose (Amersham) was then added. After a 2 h incubation at 4°C, the mixture was pelleted and washed several times with CoIP buffer. The beads were resuspended and boiled for 5 min in loading buffer. Each sample was analyzed by SDS-PAGE, and immunoblotting was performed.
In vitro binding assays with GST fusion proteins
The GST and GST-IE1 fusion proteins were prepared in E. coli by standard procedures. The [35S]Met-labeled PIAS1 was produced from a pSG5-derived template using the TNT Quick Coupled Transcription/Translation System (Promega) as specified by the manufacturer. The standard procedure for the GST pull-down assays was described previously [21].
In vitro SUMOylation assays
Recombinant GST fusion proteins were expressed in E. coli, and purified on glutathione-agarose 4B (Peptron) according to the manufacturer's instructions. His-tagged proteins were also produced in E. coli and purified on Ni-NTA beads (Invitrogen) according to the manufacturer's guidelines. Typical SUMOylation reactions were conducted in a 30 µl volume containing 70 nM GST-SAE2/SAE1, 1 µM His-Ubc9, and 9 µM His-SUMO-1GG or GST-SUMO-1GG in buffer (50 mM Tris-HCl [pH 7.5], 10 mM MgCl2, 1 mM DTT, and 5 mM ATP). To prepare flag-PIAS1 protein, 293T cells in a 150-mm dish were transfected with 30 µg of flag-PIAS1-expressing plasmid, followed by immunoprecipitation of total cell lysates with 50 µl of anti-flag M2 antibody. SUMOylation reaction mixes were incubated for 1 h at 37°C. After terminating the reaction with SDS sample buffer containing β-mercaptoethanol, the reaction products were fractionated by SDS-PAGE.
Antibodies
Anti-His (H-3) mouse monoclonal antibody (MAb) conjugated with horseradish peroxidase (HRP) and anti-GST MAb (B-14) were purchased from Santa Cruz. Anti-HA rat MAb (3F10) and anti-myc mouse MAb (9E10) conjugated with HRP were purchased from Roche. Anti-flag mouse MAb M2 was obtained from Sigma. Anti-IE1 polyclonal antibody (PAb) was raised in rabbits using the purified IE1 protein. Mouse MAb 8131, which detects epitopes present in both IE1 and IE2 (exons-2 and -3), was purchased from Chemicon (Temecula, CA, USA). Mouse MAbs specific for IE1 (6E1) and IE2 (12E2) were purchased from Vancouver Biotech and mouse MAb against β-actin was purchased from Sigma. Mouse MAb against SRT epitope was previously described [23].
Luciferase reporter assay
Cells were collected and lysed by three freeze-thaw steps in 200 µl of 0.25 M Tris-HCl (pH 7.9) plus 1 mM dithiothreitol. Cells extracts were clarified in a microcentrifuge and 30 µl of extracts were incubated with 350 µl of reaction buffer A (25 mM glycyl-glycine [pH 7.8], 15 mM ATP, and 4 mM EGTA) and then mixed with 100 µl of 0.25 mM luciferin (Sigma-Aldrich) in reaction buffer A. A TD-20/20 luminometer (Turner Designs) was used for a 10-s assay of the photons produced (measured in relative light units).
Results
In silico analysis of SUMOylation sites in HCMV-encoded proteins and evaluation of SUMOylation
To identify SUMO targets in the entire HCMV genome, we used the SUMOplot Analysis Program (http://www.abgent.com/sumoplot) and SUMOsp program (http://sumosp.biocuckoo.org/online.php) [52] to predict and score SUMO modification sites in proteins. We tested 165 HCMV ORFs from the HCMV Towne and Toledo strains. From this in silico analysis, 24 ORFs, including UL122 (IE2) and UL123 (IE1), which were previously identified as SUMO targets, were predicted with high probability to contain SUMO modification sites by both programs (Table 1).
Table 1. HCMV ORFs that contain possible SUMOylation sites.
| ORFa | Template | Amino acids | Lys position | Consensus sequence | SUMOsp score | SUMOplot score | pEXP-GST clone | Lys position (in Merlin ORF) |
| US34A | Towne-BAC | 64 | 38 | VKQE | 5.18 | 0.93 | pETK283 | 38 |
| UL123(Ex2/3/4) | Towne cDNA | 471 | 450 | VKSE | 4.95 | 0.93 | pDJK175 | 450 |
| UL122(Ex2/3/5) | Towne cDNA | 579 | 175, 180 | IKQE,IKPE | 3.97, 3.32 | 0.94, 0.94 | pDJK183 | 175, 180 |
| UL150 | Toledo cDNA | 640 | 124 | AKSD | 3.59 | 0.79 | pETK279 | 124 |
| UL84 | Towne-BAC | 587 | 73, 163, 464 | LKTP,KKKE,LKMP | 3.40, 3.39, 3.70 | 0.8, 0.48, 0.8 | pETK271 | 73, 163, 464 |
| UL35 | Towne-BAC | 640 | 137 | VKPE | 2.18 | 0.93 | pETK263 | 137 |
| RL10 | Towne-BAC | 168 | 35 | VKAE | 2.13 | 0.93 | pETK259 | 35 |
| UL27 | Towne-BAC | 608 | 431, 496 | FKGE,IKRE | 0.52, 2.2 | 0.85, 0.94 | pETK262 | 431, 496 |
| UL46 | Towne-BAC | 290 | 27, 159 | AKRE,LKTE | 1.11, 2.05 | 0.79, 0.91 | pETK265 | 27, 159 |
| UL89B(Ex2)b | Towne-BAC | 378 | 19 | IKKE | 2.04 | 0.94 | pETK272 | 19 |
| UL111A(Ex1/2/3) | Towne cDNA | 176 | 108 | LKTE | 1.94 | 0.91 | pETK276 | 108 |
| UL83 | Towne-BAC | 561 | 457 | LKAE | 1.69 | 0.91 | pETK270 | 457 |
| UL57 | Towne-BAC | 1235 | 417 | LKDE | 1.64 | 0.91 | pETK268 | 417 |
| US27 | Towne-BAC | 364 | 320 | VKQE | 1.54 | 0.93 | pETK282 | 320 |
| UL94 | Towne-BAC | 345 | 324 | VKVE | 1.46 | 0.93 | pETK273 | 324 |
| UL98 | Towne-BAC | 584 | 214 | IKHE | 1.41 | 0.94 | pETK274 | 214 |
| UL72 | Towne-BAC | 388 | 35 | MKEE | 1.34 | 0.8 | pETK269 | 35 |
| UL105 | Towne-BAC | 956 | 29, 377 | AKIE,LKEE | 0.99, 1.30 | 0.79, 0.91 | pETK275 | 29, 377 |
| UL48 | Towne-BAC | 2240 | 604, 1032 | AKQE,VKGE | 1, 1.01 | 0.79, 0.93 | pSE45 | 605, 1033 |
| UL54 | Towne-BAC | 1242 | 947 | VKLE | 0.95 | 0.93 | pETK267 | 947 |
| UL43 | Towne-BAC | 423 | 278 | MKRE | 0.94 | 0.8 | pETK264 | 278 |
| UL49 | Towne-BAC | 470 | 359 | LKCE | 0.77 | 0.91 | pETK266 | 359 |
| UL44 | Towne-BAC | 433 | 410 | AKEE | 0.7 | 0.79 | pRYK18 | 410 |
| UL148 | Towne-BAC | 316 | 275 | AKAE | 0.67 | 0.79 | pETK278 | 275 |
ORFs are listed by score with the SUMOsp program.
UL89 exon 2. For the full-length UL89 protein, one lysine residue (K315) was predicted as a SUMO attachment site with the same score from both SUMOplot and SUMOsp.
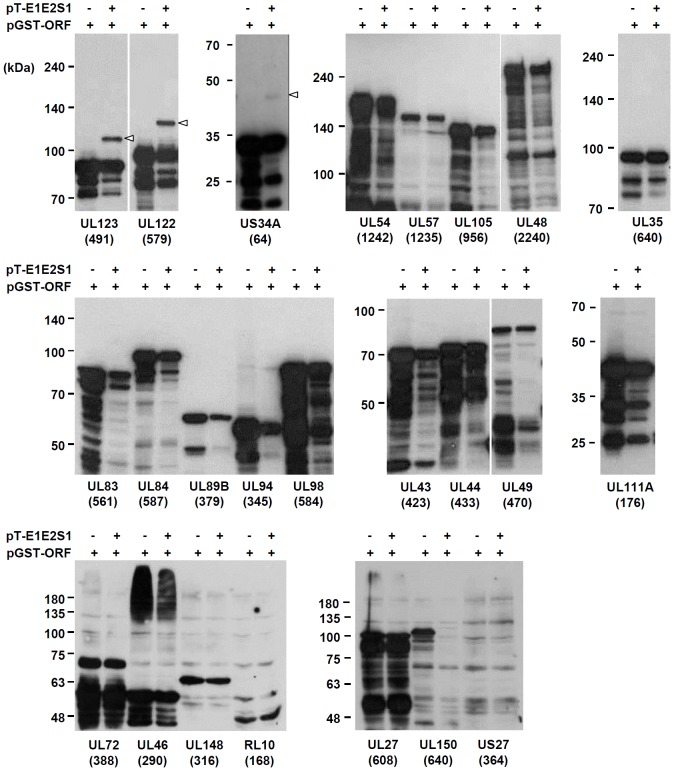
We next investigated whether the predicted 24 proteins are covalently modified by SUMO. The HCMV ORF library was produced in the pENTR vector (Invitrogen) and pGEX-3-derived plasmids expressing GST-ORF fusion proteins were produced (Table 1) (see Material and Methods). E. coli BL21 cells were transformed with pGST-ORF or cotransformed with pGST-ORF and pT-E1E2S1, which encodes a SAE2/SAE1 fusion (E1), Ubc9 (E2), and SUMO-1GG, an active form of SUMO-1. After the cells were grown, expression of GST-fusion proteins was induced with IPTG, and total cell lysates were prepared and immunoblotted with anti-GST antibody. The results showed that UL123 (IE2) and UL122 (IE1) were substantially modified by SUMO, and US34A was weakly modified by SUMO in this E. coli SUMOylation system (Fig. 1). We could not detect SUMOylated bands for 19 proteins (UL84, UL35, RL10, UL27, UL46, UL89 exon 2, UL111A, UL83, UL57, UL27, UL98, UL72, UL105, UL48, UL54, UL43, UL49, UL44, and UL148) in these assays (Fig. 1). We could not evaluate SUMOylation of UL150 and US27 in E. coli, since GST-UL150 became undetectable in E. coli cells that received both pGST-UL150 and pT-E1E2S1 probably due to change of protein stability, and GST-US27 was not expressed or expressed as several week bands, making detection of SUMOylated forms difficult (Fig. 1).
Figure 1. SUMOylation analysis of HCMV proteins in bacteria.
E. coli (BL21) cells were transformed with plasmids expressing GST-HCMV ORF (ampicillin-resistant) or cotransformed with plasmids expressing GST-HCMV ORF and pT-E1E2S1 (chloramphenicol-resistant). One milliliter of bacterial cell culture was induced with 0.4 mM IPTG for 5 h at 30°C. Total cell lysates were prepared by boiling the cell pellet in 200 µl of 1× protein loading dye. Clarified cell lysates were separated by SDS-PAGE, and immunoblot analysis was performed with anti-GST antibody. HCMV ORFs fused to GST and the ORF sizes (number of amino acids in parenthesis) are indicated. The SUMO-modified forms of UL123 (IE1), UL122 (IE2), and US34A are indicated as open arrowheads.
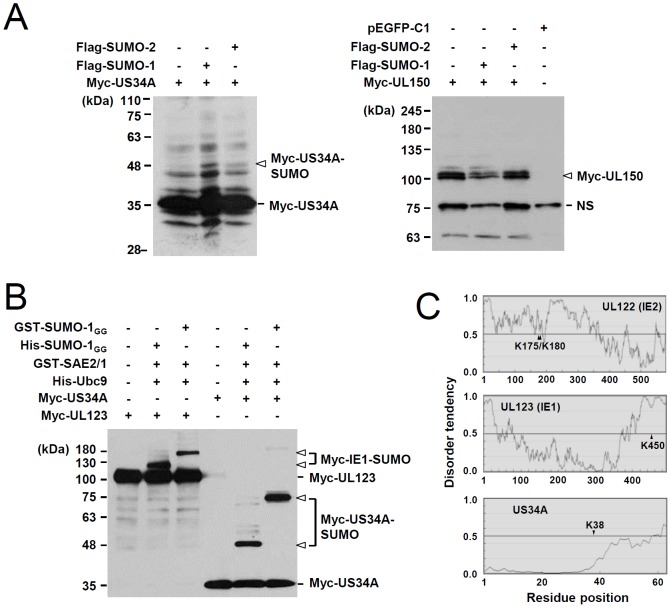
We further tested SUMOylation of US34A, UL150, and US27 using cotransfection assays. 293T cells were cotransfected with plasmids expressing a viral protein and SUMO-1, and immunoblotting was performed. We detected a small amount of SUMOylated US34A, but did not detect SUMOylated UL150 (Fig. 2A). US27 SUMOylation could not be evaluated because US27 migrated as a smear in cotransfected cells (data not shown), as previously described [53], [54]. US34A SUMOylation was further investigated in vitro using purified bacterial GST-SAE2/SAE1 (E1), His-Ubc9 (E2), and His- or GST-tagged SUMO-1GG, an active form of SUMO-1. The results showed that US34A was modified by SUMO-1 as efficiently as UL123 (IE1) in vitro, suggesting that US34A may be another SUMO target encoded by HCMV (Fig. 2B). However, unlike UL122 (IE2) and UL123 (IE1), the region of US34A containing the predicted SUMOylation site, K38, did not have a tendency to be highly disordered (Fig. 2C). Overall, our in silico genome-wide analysis of HCMV-encoded SUMO targets and subsequent cotransfection and in vitro assays demonstrated that IE1 and IE2 might be main SUMO targets in HCMV. These experiments also suggested that UL34A may be a potential SUMO target.
Figure 2. Evaluation of SUMOylation in cotransfection and in vitro assays.
(A) 293T cells in six-well plates were cotransfected with 0.5 µg of plasmid expressing myc-US34A, myc-UL150, flag-SUMO-1, or flag-SUMO-2 as indicated. At 48 h, total cell lysates were prepared and immunoblotted with an anti-myc antibody. The bands corresponding to unmodified and SUMO-modified forms of myc-US34A and unmodified myc-UL150 are indicated. NS, non-specific bands. (B) In vitro SUMOylation reactions. Myc-UL123(IE1) and myc-US34A produced by in vitro transcription/translation were incubated with GST-SAE2/1, His-Ubc9, and His-SUMO-1GG or GST-SUMO-1GG as indicated. The reaction products were analyzed by SDS-PAGE (8%) and immunoblot assays with a myc-IE1 antibody. Unmodified and SUMO-modified forms of IE1 and US34A are indicated. (C) The disorder in UL122 (IE2), UL123 (IE1), and US34A was predicted with the IUPred program (http://iupred.enzim.hu). The lysine residues modified by SUMO (for IE1 and IE2) or predicted to be SUMOylation sites (for US34A) are indicated.
SUMOylation patterns of IE1 and IE2 during HCMV infection
We next investigated the change in SUMOylation patterns of IE1 and IE2 during HCMV infection. Total cell lysates prepared at different time points after HCMV infection were immunoblotted with antibodies specific for IE1, IE2, or both. We found that IE1 SUMOylation peaked 24 h after infection and then declined at 48 h when the level of IE2 and its SUMOylation was drastically increasing (Fig. 3A). This result suggested that IE1 SUMOylation is temporally regulated during virus infection and that this change depends on the IE2 level. The effect of IE2 expression on IE1 SUMOylation was further examined in cotransfection assays. Immunoblots showed that the level of SUMOylated IE1 was reduced when IE2 was overexpressed, suggesting an inhibitory effect of IE2 on IE1 SUMOylation (Fig. 3B).
Figure 3. SUMOylation patterns of IE1 and IE2 during HCMV infection.
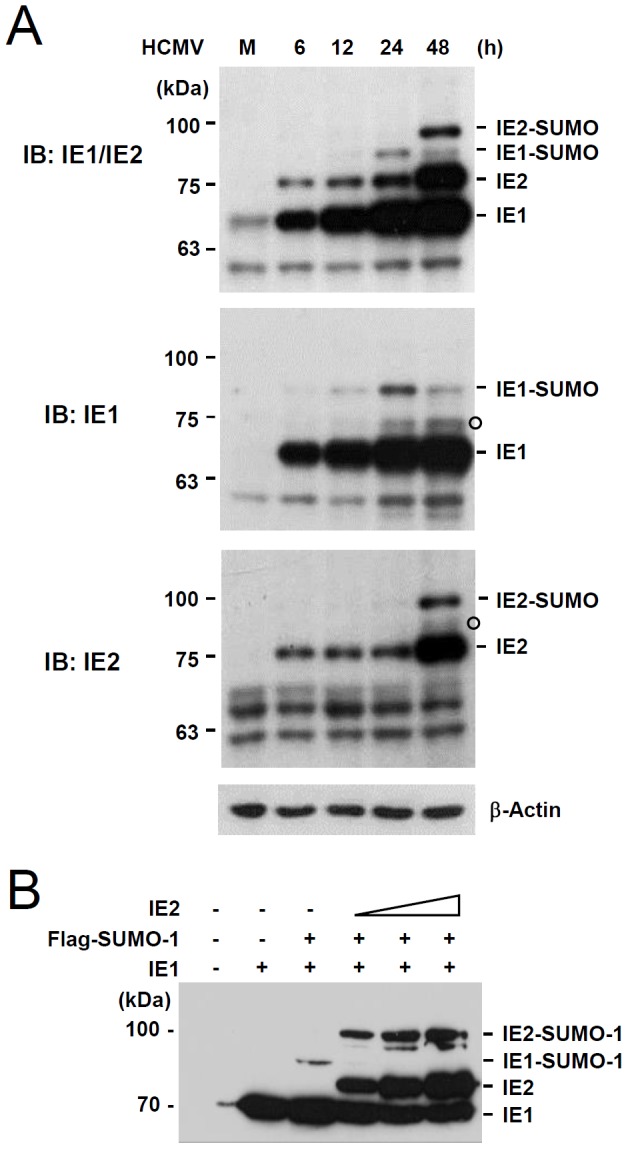
(A) HF cells were mock-infected or infected with HCMV at an MOI of 5. Total cell lysates were prepared at indicated time points and immunoblotting was performed with antibodies that recognize IE1 (6E1), IE2 (12E2), or both IE1 and IE2 (8131). The β-actin levels are shown as a loading control. The bands indicated as open circles appear to be non-specific or represent other modified forms of IE1 and IE2. (B) 293T cells in six-well plates were cotransfected with plasmids expressing IE1 (1 µg), flag-SUMO-1 (1 µg), and increasing amounts of IE2 (0.3, 1, and 3 µg), as indicated. At 48 h, total cell lysates were prepared and immunoblot assays were performed with anti-IE1/IE2 antibody.
PIAS1 interacts with IE1 and acts as a SUMO E3 ligase
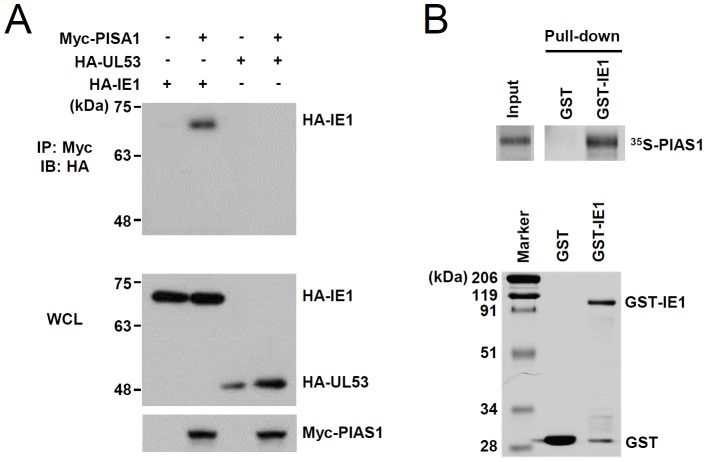
We hypothesized that increased IE2 expression might compete with IE1 for the cellular SUMOylation machinery. To address this question, we first tested whether IE1 SUMOylation requires PIAS1, a SUMO E3 ligase that acts as an E3 for IE2 SUMOylation [23]. In cotransfection assays, PIAS1 was coimmunoprecipitated with IE1 but not with UL53 (a negative control), suggesting that PIAS1 specifically interacts with IE1 (Fig. 4A). Furthermore, in GST pull-down assays, bacterial GST-IE1protein effectively interacted with PIAS1 produced by in vitro transcription/translation (Fig. 4B). These results indicated that IE1 indeed interacts with PIAS1.
Figure 4. Interaction of IE1 with PIAS1.
(A) 293T cells in a 100-mm dish were cotransfected with 5 µg of plasmids expressing myc-PIAS1 and HA-IE1 or HA-UL53, as indicated. At 48 h, total cell lysates were prepared and immunoprecipitated with an anti-myc antibody, followed by immunoblotting with an anti-HA antibody. The levels of HA-IE1, HA-UL53, and myc-PIAS1 in whole cell lysates (WCL) were also shown by immunoblotting. (B) The GST and GST-IE1 proteins purified from bacteria were used in GST pull-down assays. Five micrograms of GST and GST-IE1 proteins were immobilized on glutathione-Sepharose beads and were incubated with in vitro-translated and [35S]-methionine–labeled PIAS1. Input PIAS1 (5%) and the GST pull-down samples were separated by SDS-PAGE and visualized by autoradiography (upper panels). The purified GST and GST-IE1 used in pull-down assays are shown by SDS-PAGE and Coomassie Brilliant Blue staining (lower panel).
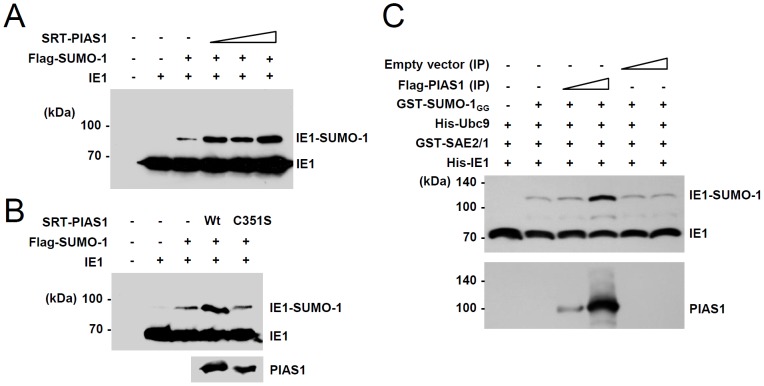
We next tested whether PIAS1 enhances IE1 SUMOylation. In cotransfection assays, PIAS1 increased SUMOylation of IE1 in a dose-dependent manner (Fig. 5A). A catalytically inactive PIAS1 mutant (C351S), in which the active site cysteine at amino acid 351 was replaced with serine [23], did not increase IE1 SUMOylation. This result suggests that PIAS1 may act as an E3 ligase for IE1 SUMOylation (Fig. 5B). To confirm the role of PIAS1 in IE1 SUMOylation, we performed in vitro SUMOylation assays. We used PIAS1 protein that was immunoprecipitated from transfected cells, because PIAS1 is not easy to produce in a soluble fraction in E. coli. Consistent with the results of cotransfection assays, we found that immunoprecipitated PIAS1 increased IE1 SUMOylation in a dose-dependent manner in vitro. These data indicate that PIAS1 acts as a SUMO E3 ligase for IE1 SUMOylation (Fig. 5C).
Figure 5. Enhancement of IE1 SUMOylation by PIAS1.
(A) 293T cells in six-well plates were cotransfected with plasmids expressing IE1 (1 µg), flag-SUMO-1 (1 µg), and increasing amounts of SRT-PIAS1 (0.3, 1, and 3 µg), as indicated. At 48 h, total cell lysates were prepared and immunoblotted with an anti-IE1 antibody. (B) 293T cells were cotransfected with plasmids expressing IE1 (1 µg), flag-SUMO-1 (1 µg), and wild-type or C351S mutant SRT-PIAS1 (0.5 µg), as indicated. At 48 h, total cell lysates were prepared and immunoblotted with anti-IE1 or anti-SRT antibodies. (C) In vitro SUMOylation reactions were conducted with bacterially purified His-IE1, GST-SAE2/1, His-Ubc9, and GST-SUMO-1GG proteins, and immunoprecipitated flag-PIAS1 proteins (see Materials and Methods). The reaction products were analyzed by SDS-PAGE (8%) and immunoblot assays with anti-IE1 antibody. The amounts of flag-PIAS1 protein used were also shown by immunoblotting with anti-flag antibody.
IE2 inhibits PIAS1-mediated SUMOylation of IE1
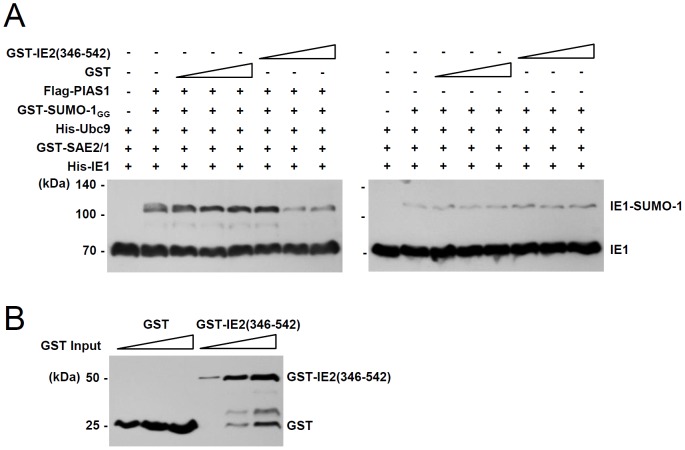
To address whether IE2 competes with IE1 for PIAS1 in SUMOylation reactions, we examined the effect of the IE2(346–542) fragment on IE1 SUMOylation in vitro. IE2(346–542) contains the PIAS1 binding region [23], but not sites for covalent or non-covalent SUMO attachment [26]. The in vitro SUMOylation assays showed that the level of IE1 SUMOylation produced in reactions containing SAE2/SAE1 (E1) and Ubc9 was increased in the presence of PIAS1, but this PIAS1-mediated IE1 SUMOylation was inhibited by IE2(346–542) (Fig. 6). In a control experiment, IE1 SUMOylation without PIAS1 was not affected by IE2(346–542) (Fig. 6). These results demonstrate that IE1 SUMOylation was negatively regulated by the PIAS1-binding activity of IE2. The moderate inhibitory effect of IE2(346–542) on IE2 SUMOylation was also observed in cotransfection assays (data not shown).
Figure 6. Inhibition of the PIAS1-mediated IE1 SUMOylation by IE2 in vitro.
(A) In vitro SUMOylation reactions for IE1 were conducted as in Fig. 5C using His-IE1, GST-SAE2/1, His-Ubc9, GST-SUMO-1GG, and increasing amounts (0.1, 0.5, and 1 µg) of GST or GST-IE2(346–542) with (left panel) or without (right panel) immunoprecipitated flag-PIAS1. The reaction products were analyzed by SDS-PAGE (8%) and immunoblot assays with anti-IE1 antibody. (B) The GST and GST-IE2(346–542) proteins added to in vitro SUMOylation reactions were detected by immunoblotting with an anti-GST antibody.
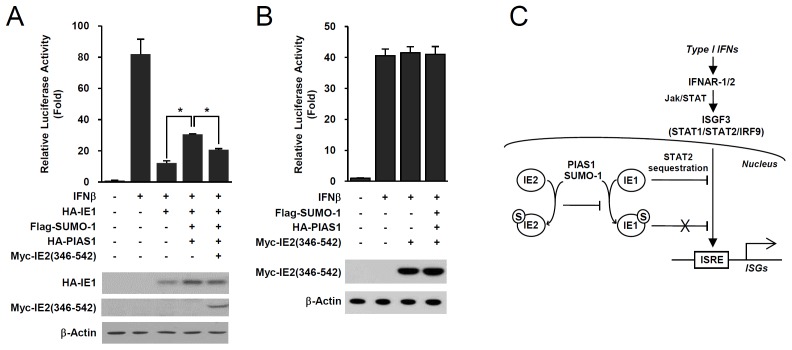
We further investigated whether IE2 inhibiting IE1 SUMOylation affects the ability of IE1 to downregulate the promoter containing the IFN stimulated response element (ISRE). In luciferase reporter assays using the ISG54 ISRE-luciferase reporter gene, coexpressing SUMO-1 and PIAS1 inhibited the ability of IE1 to suppress ISRE promoter induction by IFNβ. However, adding IE2(346–542) reversed this effect (Fig. 7A). IE2(346–542) does not contain the transactivation domains (codons 25–85 and 544–579) [55]. Consistently, in a control experiment, IE2(346–542) did not affect the induction of ISRE promoter by IFNβ (Fig. 7B). This result suggests that IE2 expression and its PIAS1-binding activity can interfere with PIAS1-mediated IE1 SUMOylation, resulting in unmodified IE1 more efficiently suppressing type I IFN-mediated ISG expression (Fig. 7C).
Figure 7. IE2 reverses the SUMOylation-dependent inhibition of IE1 activity to downregulate ISRE activation.
(A and B) The reporter assays using the ISG54 ISRE-luciferase construct. 293T cells in 12-well plates were cotransfected with 0.5 µg of the ISG54 ISRE-luciferase reporter construct and plasmids expressing HA-IE1, flag-SUMO-1, HA-PIAS1, and myc-IE2(346–542) as indicated. The total amount of plasmid was adjusted with empty vectors. At 24 h, cells were untreated or treated with IFNβ (1,000 units/ml) for 8 h, and luciferase reporter assays were performed. The results shown are the mean values and standard errors of three independent experiments. Statistical significance between samples was determined using Student's t-test (values of *P<0.0005). The expression levels of IE1, IE2, and β-actin proteins in cell lysates were determined by immunoblotting with specific antibodies. (C) A hypothetical model showing that expression of IE2 and its SUMOylation regulates the PIAS1-mediated IE1 SUMOylation, enhancing IE1 activity to downregulate type I IFN-stimulated gene (ISG) expression. ISRE, interferon stimulated response element.
Discussion
In this study, we performed in silico analysis to predict possible SUMO modification sites in all HCMV ORFs. Among 24 ORFs that were predicted to have a consensus sequence with relatively high scores, only UL123 (IE2), UL123 (IE1), and US34A, which received the highest scores using the SUMOsp program, were SUMOylated in E. coli SUMOylation assays. The SUMOylation levels of US34A in E. coli and in cotransfected cells were much lower those of IE1 and IE2, although US34A was SUMOylated as efficiently as IE1 in vitro. Unlike IE1 and IE2, the predicted SUMOylation site in US34A was not in the disordered region. Therefore, whether US34A SUMOylation occurs during virus infection needs to be addressed. Given that SUMOylation of IE1 and IE2 was easily detectable in virus-infected cells [20], [39], [40], [44](this study), the data from our in silico genome-wide analysis suggest that these two IE proteins may be the main HCMV-encoded targets for SUMO. Nevertheless, we cannot exclude the possibility that SUMOylation of other HCMV proteins predicted in this study occurs in virus-infected cells. An example is UL44. Although we could not detect SUMOylation of UL44 in E. coli assays, SUMOylated UL44 was detected in cotransfected cells, in vitro SUMOylation reactions, and virus-infected cells [56]. We also observed SUMOylation of UL44 in in vitro assays (data not shown).
We and others have found SUMO in viral replication compartments (RCs) in HCMV-infected cells [25], [57], suggesting that viral or cellular SUMO substrates may accumulate at viral RCs. Although IE2 is recruited to viral RCs [58], SUMO is also found in viral RCs in cells infected with a virus encoding a mutant IE2 protein that lacks both the SUMOylation sites and the SIM [57]. Thus, other viral proteins implicated in viral DNA replication have been suggested to be SUMO targets or recruit SUMO via the SIM-mediated intercation. SUMOylation of UL44 (polymerase percessicity factor) might explain the presence of SUMO species in viral RCs. In addition, UL54 (DNA polymerase), UL57 (single-stranded DNA-binding protein), and UL105 (DNA helicase) have been suggested to have SUMO modification sites [57]. Our in silico analysis also predicted these viral replication proteins to have possible SUMOylation sites; however, none were SUMOylated in our E. coli SUMOylation assays.
In this study, we demonstrated that SUMOylation of IE1 and IE2 is temporally regulated during HCMV infection. SUMOylated IE1 levels were increased at the early phase of infection and decreased at the late phase when the expression of IE2 and its SUMO-modified forms drastically increased. The increase of IE2 SUMOylation at the late stage of infection is consistent with a general role of IE2 SUMOylation in increasing viral gene expression [20]–[24], [26]. The biphasic regulation of IE1 SUMOylation is intriguing. The role of IE1 SUMOylation in viral infection is not clear. A mutant virus encoding SUMOylation-defective IE1 grew less efficiently than normal virus, suggesting a positive role of IE1 SUMOylation in virus infection [41]. However, a similar mutant virus did not have a significant growth defect [42], and the lack of IE1 SUMOylation did not affect the ability of IE1 to complement the growth defect of the IE1-deleted mutant virus [40]. Further studies are necessary to address whether IE1 SUMOylation plays a role at early steps of the viral replication cycle or whether IE1 SUMOylation is just a consequence of IE1 targeting to PML nuclear bodies, where the components of SUMOylation machinery are enriched. Recently, we found that IE1 SUMOylation inhibited the interaction between IE1 and STAT2 and that the SUMO-modified form of IE1 failed to inhibit IFNβ-mediated activation of the ISRE-containing promoter [37]. These findings suggested that IE1 SUMOylation may be detrimental for viruses trying to evade cellular innate immune responses, although the overall effect of IE1 SUMOylation on viral replication could be different. In this regard, IE2, by inhibiting IE1 SUMOylation, may assist in immune escape by the virus. The interplay between SUMOylation of two viral proteins has been shown in Epstein-Barr virus. The SIM-containing BGLF4 protein inhibits BZLF1 SUMOylation through its SUMO-binding activity and also reduces overall SUMOylation, which enhances EBV lytic infection [59], [60].
Several viral proteins have been shown to reduce cellular SUMOylation by directly targeting SUMOylation machinery. The Gam1 protein of avian adenovirus CELO (chicken embryo lethal orphan) reduces cellular SUMOylation by interacting with and destabilizing the SAE1-SAE2 complex [61], [62]. Human papillomavirus E6 induces degradation of Ubc9 [63]. Our finding that IE2 expression inhibits IE1 SUMOylation by binding to PIAS1 raises a question whether IE2 has a general role in regulating the cellular SUMO pathway. We observed that IE2 overexpression slightly reduces the level of cellular SUMO conjugates (data not shown). This intriguing hypothesis remains to be addressed.
Acknowledgments
We thank Ke Shuai, Ronald T. Hay and Hisato Saitoh for providing expression plasmids. We also thank Hua Zhu for the Toldeo-BAC and Towne-BAC clones. We also thank Chuang-Jiun Chiou for technical assistance in preparing the HCMV ORF library.
Data Availability
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper and its Supporting Information files.
Funding Statement
This research was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (2005-2001163 and 2012R1A2A2A01002551). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Hay RT (2005) SUMO: a history of modification. Mol Cell 18: 1–12. [DOI] [PubMed] [Google Scholar]
- 2. Gareau JR, Lima CD (2010) The SUMO pathway: emerging mechanisms that shape specificity, conjugation and recognition. Nat Rev Mol Cell Biol 11: 861–871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Kahyo T, Nishida T, Yasuda H (2001) Involvement of PIAS1 in the sumoylation of tumor suppressor p53. Mol Cell 8: 713–718. [DOI] [PubMed] [Google Scholar]
- 4. Pichler A, Gast A, Seeler JS, Dejean A, Melchior F (2002) The nucleoporin RanBP2 has SUMO1 E3 ligase activity. Cell 108: 109–120. [DOI] [PubMed] [Google Scholar]
- 5. Kagey MH, Melhuish TA, Wotton D (2003) The polycomb protein Pc2 is a SUMO E3. Cell 113: 127–137 118: Sternsdorf T et al. Sumo….[PMID: 12676099]Related Articles, Links. [DOI] [PubMed] [Google Scholar]
- 6. Bernier-Villamor V, Sampson DA, Matunis MJ, Lima CD (2002) Structural basis for E2-mediated SUMO conjugation revealed by a complex between ubiquitin-conjugating enzyme Ubc9 and RanGAP1. Cell 108: 345–356. [DOI] [PubMed] [Google Scholar]
- 7. Lin D, Tatham MH, Yu B, Kim S, Hay RT, et al. (2002) Identification of a substrate recognition site on ubc9. J Biol Chem 277: 21740–21748. [DOI] [PubMed] [Google Scholar]
- 8. Rodriguez MS, Dargemont C, Hay RT (2001) SUMO-1 conjugation in vivo requires both a consensus modification motif and nuclear targeting. J Biol Chem 276: 12654–12659. [DOI] [PubMed] [Google Scholar]
- 9. Sampson DA, Wang M, Matunis MJ (2001) The small ubiquitin-like modifier-1 (SUMO-1) consensus sequence mediates Ubc9 binding and is essential for SUMO-1 modification. J Biol Chem 276: 21664–21669. [DOI] [PubMed] [Google Scholar]
- 10. Hay RT (2007) SUMO-specific proteases: a twist in the tail. Trends Cell Biol 17: 370–376. [DOI] [PubMed] [Google Scholar]
- 11. Yeh ET (2009) SUMOylation and De-SUMOylation: wrestling with life's processes. J Biol Chem 284: 8223–8227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Hickey CM, Wilson NR, Hochstrasser M (2012) Function and regulation of SUMO proteases. Nat Rev Mol Cell Biol 13: 755–766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Minty A, Dumont X, Kaghad M, Caput D (2000) Covalent modification of p73alpha by SUMO-1. Two-hybrid screening with p73 identifies novel SUMO-1-interacting proteins and a SUMO-1 interaction motif. J Biol Chem 275: 36316–36323. [DOI] [PubMed] [Google Scholar]
- 14. Song J, Durrin LK, Wilkinson TA, Krontiris TG, Chen Y (2004) Identification of a SUMO-binding motif that recognizes SUMO-modified proteins. Proc Natl Acad Sci U S A 101: 14373–14378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hannich JT, Lewis A, Kroetz MB, Li SJ, Heide H, et al. (2005) Defining the SUMO-modified proteome by multiple approaches in Saccharomyces cerevisiae. J Biol Chem 280: 4102–4110. [DOI] [PubMed] [Google Scholar]
- 16. Kerscher O (2007) SUMO junction-what's your function? New insights through SUMO-interacting motifs. EMBO Rep 8: 550–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Everett RD, Boutell C, Hale BG (2013) Interplay between viruses and host sumoylation pathways. Nat Rev Microbiol 11: 400–411. [DOI] [PubMed] [Google Scholar]
- 18. Wimmer P, Schreiner S, Dobner T (2012) Human pathogens and the host cell SUMOylation system. J Virol 86: 642–654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mocarski ES, Shenk T, Griffiths PD, Pass RF (2013) Cytomegaloviruses, p. 1960–2014. In D. MKnipe, P. MHowley, J. ICohen, D. EGriffin, R. ALamb, M. AMartin, V. RRacaniello, and BRoizman (ed.), Fields virology. Lippincott Williams & Wilkins. Philadelphia, PA.
- 20. Hofmann H, Floss S, Stamminger T (2000) Covalent modification of the transactivator protein IE2-p86 of human cytomegalovirus by conjugation to the ubiquitin-homologous proteins SUMO-1 and hSMT3b. J Virol 74: 2510–2524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ahn JH, Xu Y, Jang WJ, Matunis MJ, Hayward GS (2001) Evaluation of interactions of human cytomegalovirus immediate-early IE2 regulatory protein with small ubiquitin-like modifiers and their conjugation enzyme Ubc9. J Virol 75: 3859–3872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Barrasa MI, Harel N, Yu Y, Alwine JC (2003) Strain variations in single amino acids of the 86-kilodalton human cytomegalovirus major immediate-early protein (IE2) affect its functional and biochemical properties: implications of dynamic protein conformation. J Virol 77: 4760–4772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Lee JM, Kang HJ, Lee HR, Choi CY, Jang WJ, et al. (2003) PIAS1 enhances SUMO-1 modification and the transactivation activity of the major immediate-early IE2 protein of human cytomegalovirus. FEBS Lett 555: 322–328. [DOI] [PubMed] [Google Scholar]
- 24. Berndt A, Hofmann-Winkler H, Tavalai N, Hahn G, Stamminger T (2009) Importance of covalent and noncovalent SUMO interactions with the major human cytomegalovirus transactivator IE2p86 for viral infection. J Virol 83: 12881–12894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Lee HR, Ahn JH (2004) Sumoylation of the major immediate-early IE2 protein of human cytomegalovirus Towne strain is not required for virus growth in cultured human fibroblasts. J Gen Virol 85: 2149–2154. [DOI] [PubMed] [Google Scholar]
- 26. Kim ET, Kim YE, Huh YH, Ahn JH (2010) Role of noncovalent SUMO binding by the human cytomegalovirus IE2 transactivator in lytic growth. J Virol 84: 8111–8123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Greaves RF, Mocarski ES (1998) Defective growth correlates with reduced accumulation of a viral DNA replication protein after low-multiplicity infection by a human cytomegalovirus ie1 mutant. J Virol 72: 366–379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Mocarski ES, Kemble GW, Lyle JM, Greaves RF (1996) A deletion mutant in the human cytomegalovirus gene encoding IE1(491aa) is replication defective due to a failure in autoregulation. Proc Natl Acad Sci U S A 93: 11321–11326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ahn JH, Hayward GS (1997) The major immediate-early proteins IE1 and IE2 of human cytomegalovirus colocalize with and disrupt PML-associated nuclear bodies at very early times in infected permissive cells. J Virol 71: 4599–4613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Ahn JH, Brignole EJ 3rd, Hayward GS (1998) Disruption of PML subnuclear domains by the acidic IE1 protein of human cytomegalovirus is mediated through interaction with PML and may modulate a RING finger-dependent cryptic transactivator function of PML. Mol Cell Biol 18: 4899–4913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Korioth F, Maul GG, Plachter B, Stamminger T, Frey J (1996) The nuclear domain 10 (ND10) is disrupted by the human cytomegalovirus gene product IE1. Exp Cell Res 229: 155–158. [DOI] [PubMed] [Google Scholar]
- 32. Wilkinson GW, Kelly C, Sinclair JH, Rickards C (1998) Disruption of PML-associated nuclear bodies mediated by the human cytomegalovirus major immediate early gene product. J Gen Virol 79 (Pt 5) 1233–1245. [DOI] [PubMed] [Google Scholar]
- 33. Kim YE, Lee JH, Kim ET, Shin HJ, Gu SY, et al. (2011) Human cytomegalovirus infection causes degradation of Sp100 proteins that suppress viral gene expression. J Virol 85: 11928–11937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Tavalai N, Adler M, Scherer M, Riedl Y, Stamminger T (2011) Evidence for a Dual Antiviral Role of the Major Nuclear Domain 10 Component Sp100 during the Immediate-Early and Late Phases of the Human Cytomegalovirus Replication Cycle. J Virol 85: 9447–9458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Tavalai N, Papior P, Rechter S, Leis M, Stamminger T (2006) Evidence for a role of the cellular ND10 protein PML in mediating intrinsic immunity against human cytomegalovirus infections. J Virol 80: 8006–8018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Paulus C, Krauss S, Nevels M (2006) A human cytomegalovirus antagonist of type I IFN-dependent signal transducer and activator of transcription signaling. Proc Natl Acad Sci U S A 103: 3840–3845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Huh YH, Kim YE, Kim ET, Park JJ, Song MJ, et al. (2008) Binding STAT2 by the acidic domain of human cytomegalovirus IE1 promotes viral growth and is negatively regulated by SUMO. J Virol 82: 10444–10454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Krauss S, Kaps J, Czech N, Paulus C, Nevels M (2009) Physical requirements and functional consequences of complex formation between the cytomegalovirus IE1 protein and human STAT2. J Virol 83: 12854–12870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Xu Y, Ahn JH, Cheng M, apRhys CM, Chiou CJ, et al. (2001) Proteasome-independent disruption of PML oncogenic domains (PODs), but not covalent modification by SUMO-1, is required for human cytomegalovirus immediate-early protein IE1 to inhibit PML-mediated transcriptional repression. J Virol 75: 10683–10695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Spengler ML, Kurapatwinski K, Black AR, Azizkhan-Clifford J (2002) SUMO-1 modification of human cytomegalovirus IE1/IE72. J Virol 76: 2990–2996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Nevels M, Brune W, Shenk T (2004) SUMOylation of the human cytomegalovirus 72-kilodalton IE1 protein facilitates expression of the 86-kilodalton IE2 protein and promotes viral replication. J Virol 78: 7803–7812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Lee HR, Kim DJ, Lee JM, Choi CY, Ahn BY, et al. (2004) Ability of the human cytomegalovirus IE1 protein to modulate sumoylation of PML correlates with its functional activities in transcriptional regulation and infectivity in cultured fibroblast cells. J Virol 78: 6527–6542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Shin HJ, Kim YE, Kim ET, Ahn JH (2012) The chromatin-tethering domain of human cytomegalovirus immediate-early (IE) 1 mediates associations of IE1, PML and STAT2 with mitotic chromosomes, but is not essential for viral replication. J Gen Virol 93: 716–721. [DOI] [PubMed] [Google Scholar]
- 44. Sadanari H, Yamada R, Ohnishi K, Matsubara K, Tanaka J (2005) SUMO-1 modification of the major immediate-early (IE) 1 and 2 proteins of human cytomegalovirus is regulated by different mechanisms and modulates the intracellular localization of the IE1, but not IE2, protein. Arch Virol 150: 1763–1782. [DOI] [PubMed] [Google Scholar]
- 45. Green S, Issemann I, Sheer E (1988) A versatile in vivo and in vitro eukaryotic expression vector for protein engineering. Nucleic Acids Res 16: 369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Turner DL, Weintraub H (1994) Expression of achaete-scute homolog 3 in Xenopus embryos converts ectodermal cells to a neural fate. Genes Dev 8: 1434–1447. [DOI] [PubMed] [Google Scholar]
- 47. Kang H, Kim ET, Lee HR, Park JJ, Go YY, et al. (2006) Inhibition of SUMO-independent PML oligomerization by the human cytomegalovirus IE1 protein. J Gen Virol 87: 2181–2190. [DOI] [PubMed] [Google Scholar]
- 48. Uchimura Y, Nakamura M, Sugasawa K, Nakao M, Saitoh H (2004) Overproduction of eukaryotic SUMO-1- and SUMO-2-conjugated proteins in Escherichia coli. Anal Biochem 331: 204–206. [DOI] [PubMed] [Google Scholar]
- 49. Zhu J, Liao G, Shan L, Zhang J, Chen MR, et al. (2009) Protein array identification of substrates of the Epstein-Barr virus protein kinase BGLF4. J Virol 83: 5219–5231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Murphy E, Yu D, Grimwood J, Schmutz J, Dickson M, et al. (2003) Coding potential of laboratory and clinical strains of human cytomegalovirus. Proc Natl Acad Sci U S A 100: 14976–14981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Marchini A, Liu H, Zhu H (2001) Human cytomegalovirus with IE-2 (UL122) deleted fails to express early lytic genes. J Virol 75: 1870–1878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Ren J, Gao X, Jin C, Zhu M, Wang X, et al. (2009) Systematic study of protein sumoylation: Development of a site-specific predictor of SUMOsp 2.0. Proteomics 9: 3409–3412. [DOI] [PubMed] [Google Scholar]
- 53. O'Connor CM, Shenk T (2011) Human cytomegalovirus pUS27 G protein-coupled receptor homologue is required for efficient spread by the extracellular route but not for direct cell-to-cell spread. J Virol 85: 3700–3707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Margulies BJ, Gibson W (2007) The chemokine receptor homologue encoded by US27 of human cytomegalovirus is heavily glycosylated and is present in infected human foreskin fibroblasts and enveloped virus particles. Virus Res 123: 57–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Pizzorno MC, Mullen MA, Chang YN, Hayward GS (1991) The functionally active IE2 immediate-early regulatory protein of human cytomegalovirus is an 80-kilodalton polypeptide that contains two distinct activator domains and a duplicated nuclear localization signal. J Virol 65: 3839–3852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Sinigalia E, Alvisi G, Segre CV, Mercorelli B, Muratore G, et al. (2012) The human cytomegalovirus DNA polymerase processivity factor UL44 is modified by SUMO in a DNA-dependent manner. PLoS One 7: e49630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Scherer M, Reuter N, Wagenknecht N, Otto V, Sticht H, et al. (2013) Small ubiquitin-related modifier (SUMO) pathway-mediated enhancement of human cytomegalovirus replication correlates with a recruitment of SUMO-1/3 proteins to viral replication compartments. J Gen Virol 94: 1373–1384. [DOI] [PubMed] [Google Scholar]
- 58. Ahn JH, Jang WJ, Hayward GS (1999) The human cytomegalovirus IE2 and UL112-113 proteins accumulate in viral DNA replication compartments that initiate from the periphery of promyelocytic leukemia protein-associated nuclear bodies (PODs or ND10). J Virol 73: 10458–10471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Hagemeier SR, Dickerson SJ, Meng Q, Yu X, Mertz JE, et al. (2010) Sumoylation of the Epstein-Barr virus BZLF1 protein inhibits its transcriptional activity and is regulated by the virus-encoded protein kinase. J Virol 84: 4383–4394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Li R, Wang L, Liao G, Guzzo CM, Matunis MJ, et al. (2012) SUMO binding by the Epstein-Barr virus protein kinase BGLF4 is crucial for BGLF4 function. J Virol 86: 5412–5421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Boggio R, Colombo R, Hay RT, Draetta GF, Chiocca S (2004) A mechanism for inhibiting the SUMO pathway. Mol Cell 16: 549–561. [DOI] [PubMed] [Google Scholar]
- 62. Boggio R, Passafaro A, Chiocca S (2007) Targeting SUMO E1 to ubiquitin ligases: a viral strategy to counteract sumoylation. J Biol Chem 282: 15376–15382. [DOI] [PubMed] [Google Scholar]
- 63. Heaton PR, Deyrieux AF, Bian XL, Wilson VG (2011) HPV E6 proteins target Ubc9, the SUMO conjugating enzyme. Virus Res 158: 199–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper and its Supporting Information files.