Abstract
Many targets have been identified in solid tumors for antibody therapy but it is less clear what surface antigens may be most commonly expressed on disseminated tumor cells. Using malignant pleural effusions as a source of disseminated tumor cells, we compared a panel of 35 antigens for their cancer specificity, antigen abundance and functional significance. These antigens have been previously implicated in cancer metastasis and fall into four categories: (i) cancer stem cell, (ii) epithelial-mesenchymal transition, (iii) metastatic signature of in vivo selection and (iv) tyrosine kinase receptors. We determined the antigen density of all 35 antigens on the cell surface by flow cytometry, which ranges from 3 × 103−7 3 106 copies per cell. Comparison between the malignant and benign pleural effusions enabled us to determine the antigens specific for cancer. We further chose six antigens and examined the correlation between their expression levels and tumor formation in immunocompromised mice. We concluded that CD24 is one of the few antigens that could simultaneously meet all three criteria of an ideal target. It was specifically and abundantly expressed in malignant pleural effusions; CD24high tumor cells formed tumors in mice at a faster rate than CD24low tumor cells, and shRNA-mediated knockdown of CD24 in HT29 cells confirmed a functional requirement for CD24 in the colonization of the lung. Concomitant consideration of antigen abundance, specificity and functional importance can help identify potentially useful markers for disseminated tumor cells.
Keywords: surface biomarker, disseminated tumor cells, pleural effusions, CD24, flow cytometry
The discovery and validation of antigen targets is the first step in the development of an efficacious antibody therapy.1 Several successful targets have been identified in solid tumors, but it is unclear whether the same targets are equally applicable to disseminated tumor cells, which are detached from the primary site and often possess greater tumorigenic potential than cells in the primary tumor.2 We wished to evaluate a panel of antigens for their suitability to target disseminated tumor cells.
In terms of antigen characterization, an ideal antibody target should fulfill three criteria: (i) cancer specificity, (ii) abundant expression and (iii) functional importance.1 One of the most successful antibody targets, ErbB2, is an antigen that meets all three criteria. ErbB2 is overexpressed in 82% of breast cancer samples.3 However, antibody therapy against ErbB2 with Trastuzumab is only effective in patients expressing the highest levels of ErbB2 at around 5 × 105 receptors per cell but ineffective in patients expressing 2 × 105 receptors per cell, indicating that abundant expression of the antigen is an essential determinant of therapeutic outcome.4 Functionally, ErbB2 plays an important role in tumor growth as it can activate survival and mitogenic pathways.5
Although many targets have been evaluated according to the aforementioned guidelines in solid tumors, few antigens have been assessed in disseminated tumor cells. On the basis of recent evidence, we identified candidate antigens that are particularly involved in the molecular pathways and cellular mechanisms of metastasis. They fall under four general categories. First, cancer stem cell (CSC) markers have been shown to delineate subpopulations with markedly increased tumor-initiating potential.6,7 Second, the loss of epithelial markers and the gain of mesenchymal markers during epithelial-mesenchymal transition (EMT) are features of a more invasive cell type.8 Third, in vivo selection experiments have identified gene signatures responsible for the organ tropism of metastasis9 or metastasis in general.10 The fourth class is the family of receptor tyrosine kinases important for tumor growth, and monoclonal antibodies targeting these are used to treat solid tumors.11
What’s new?
In order to develop effective antibody therapies, researchers must first identify cancer-specific antigens that are important for tumor growth or metastasis. Because few such antigens have been assessed in disseminated tumor cells, the authors examined a broad panel of surface markers on cells from malignant pleural effusions. They found that CD24 has three qualities that make it a promising target: specificity, abundance, and functional importance for tumorigenesis. CD24 may thus be well suited to both diagnosis of circulating tumor cells and therapeutic targeting.
Acquisition of biological materials presents a challenge for target discovery in disseminated tumor cells1. Circulating tumor cells are too rare (~1 cell/ml blood) for direct screening of surface antigens12. In contrast, malignant pleural effusions are a large reservoir of highly malignant tumor cells in suspension (104 −106 cells/L), and with higher tumorigenic potential than that of primary tumors2. In addition to cancer, congestive heart, renal and liver diseases can also cause the buildup of fluids in the pleural cavity, forming benign effusions that can be used as negative controls in the measurement of cancer specificity. Soluble factors have been evaluated as tumor markers for malignant pleural effusion13,14 but to our knowledge there has not been a previous screening effort of cell-surface antigens. Cell-surface antigens are desirable for antibody targeting because they are directly accessible and localized to the cells of interest.
We evaluated a panel of 35 surface antigens in 12 malignant and 8 benign pleural effusions. We determined the average receptor number per cell for each of the 35 antigens using flow cytometry, and validated the tumorigenicity of six targets using tumor formation studies in immunocompromised mice. We identified CD24 as a potential target for malignant pleural effusion because it satisfied all three criteria of an ideal antibody target-it is preferentially expressed in malignant but not in benign pleural effusion; it is abundantly expressed at 6.8 × 105 receptors/cell and is functionally required for the colonization of the lung by HT29 cells.
Material and Methods
Collection of patient samples
Pleural effusions were obtained from consenting patients as part of an Institutional Review Board-approved protocol at the Lahey Clinic and Use of Humans as Experimental Subjects- approved study at MIT. Samples were collected from patients with biopsy-proven cancer and patients with no cancer as determined by clinical presentation and histology. A total of 400 ml of pleural fluid was set aside for routine cytologic evaluation and the rest was sent to MIT on ice within 24 hr after collection, and analyzed for surface expression within 48 hr after collection. A total of 12 malignant pleural effusions and 8 benign effusions were used for surface antigen analysis. Refer to Supporting Information Table S1 for detailed patient information.
Surface marker expression analysis using flow cytometry
A total of 30 ml of pleural effusions were passed through a 40 µm cell strainer (BD Biosciences), incubated with human FcR block (eBioscience) in 1:20 dilution and then stained with antibodies (see Supporting Information experimental procedures) for 30 min on ice. DAPI is added immediately before flow cytometry analysis to gate out the dead cells. Cells were analyzed on LSRII flow cytometer (BD Biosciences) with four different lasers (355 nm, 488 nm, 561 nm, and 635 nm) in five different channels: DAPI, FITC (marker 1), PE (marker 2), EpCAM-APC and CD45-PECY7 using HTS plate reader (Supporting Information Fig. S2B). Antibodies were purchased directly conjugated, or labeled with Alexa Fluor 488 dye (Invitrogen) or PE (Dojindo). The fluorophores were chosen to be those with minimal bleed through. Compensations were performed with beads (Bangs Laboratory) coated with individual clone of antibodies, and the compensation matrix was applied to all the patient samples.
Quantification of surface receptors expression
The number of receptors per cell is determined by calibrating the fluorescence intensity using microspheres with known Antibody Binding Capacities (Bangs Laboratory). The Quantum Simply Cellular micropheres come with one blank and four standards with increasing levels of Fc-specific capture antibodies. The beads were labeled to saturation with the same antibodies used to stain the cells and analyzed on the flow cytometer with the same settings used to analyze the cells. The standards were then used to construct a calibration curve (Supporting Information Fig. S2A). The number of antigen was determined by converting the mean fluorescence level to antibody binding capacities. As we only used monoclonal antibodies to stain the cells (except cadherin-11), each antibody only binds to one antigen and the antibody binding capacities of the microspheres is equivalent to the number of antigens. The mean fluorescence level is tabulated by Flowjo (Tree Star) and compiled using a Matlab (Mathworks) program. Monoclonal antibody for Cadherin-11 was not commercially available, so we used a goat polyclonal antibody. The calibration kit against goat Fc was also not available, so we estimated the number of Cadherin-11 based on the fluorescence intensity of N-cadherin antibody.
Sorting and animal studies
All experimental protocols were approved by the Animal Care and Use Committee of MIT. Tumor from patient LC52’s pleural effusion was formed after injecting the cells pelleted from 30 ml of pleural effusion subcutaneously into NOD/SCID/IL2γR−/− with 200 µl Matrigel (BD Biosciences). LC52 was propagated in vivo by serial transplantation. The tumors were cut up with razor blades, and digested in 1 mg/ ml collagenase A (Roche) and 100 units/ml DNAse I (Roche) in RMPI at 37°C for 20 min. Single cell suspensions were stained with antibodies for 30 min on ice and sorted on Aria (BD) by fluorescence-activated cell sorting (FACS). Sorted cells were injected with Matrigel and observed for tumor formation for up to 1 year.
Cell lines and shRNA knockdown
HT29 cells (ATCC) were maintained in DMEM (Cellgro) and 10% FBS (PAA Laboratories). shRNA lentiviral particles were purchased from Sigma-Aldrich (clone1-TRCN0000057 675, 5′-CCGG-TCTTCTGCATCTCTACTCTTA-CTCGAG-TAAGAGTAGAGATGCAGAAGA-TTTTTG-3′ and clone 2-TRCN000007677, 5Ȳ-CCGG-CGCAGATTTATTCCAGTGAA A-CTCGAG-TTTCACTGGAATAAATCTGCG-TTTTTG-3′). The nontargeted control uses an shRNA sequence targeting no known mammalian genes (Sigma-Aldrich SHC002V). Cells were plated at 1 × 104/well in a 96-well plate, transduced with lentiviral particles, and selected with puromycin for 10 days. After antibiotic selection, cells were further FACS-sorted for CD24 low-expressing cells.
Lung colonization study
About 1 × 105 HT29 cells, either transduced with nontargeted shRNA or CD24 shRNA, were injected into mice through the tail vein. The lung tissue was fixed with 4% paraformaldehyde for 15 min and rinsed with PBS. The lungs were embedded in paraffin, sectioned 100 µm apart and stained by hematoxylin and eosin (H&E). The number of metastases observed was counted and averaged over five sections per mouse.
Statistical analysis
ANOVA tests in Matlab were used to determine p-values of the heatmap. Kaplan-Meier plots were created and p-value calculated by a log-rank test in GraphPad Prism.
Sample size determination for statistical powering of Type II error is computed by this formula:
where n = sample size, d = effect or the difference between the means of two populations, s = standard deviation and C = 7.85 when significance level (α) = 0.05 and power (1–β) = 0.08.
Results
Only the EpCAM+ population of cells in pleural effusions is tumorigenic
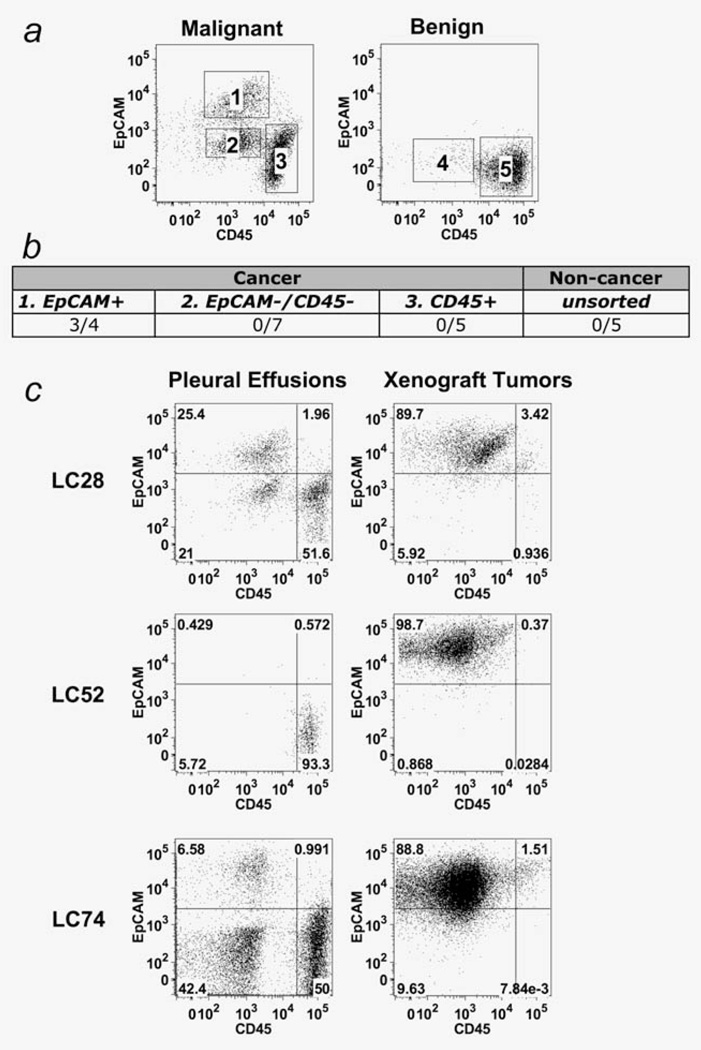
The pleural effusion is a complex mixture of immune cells, mesothelial cells, fibroblasts, and cancer cells. As the proportion of stromal cells can be >90% of the total population, taking the global average across different cell types would skew the result toward the more abundant cell types and preclude antigen discovery in rare tumor-forming cells. Therefore, we used lineage markers—CD45 for hematopoietic cells and EpCAM for epithelial cells—to provisionally separate the pleural effusions into different subsets. We observed three distinct populations in malignant pleural effusions: (i) EpCAM+, (ii) EpCAM− /CD45− and (iii) CD45+ (Fig. 1a). The subsets were morphologically distinct, with the EpCAM+ cells being round and the EpCAM−/CD45− cells spindle-shaped (Supporting Information Fig. S1A). The EpCAM+ subset was present in 50% of the malignant pleural effusions and was completely absent in the benign effusions (Fig. 1a).
Figure 1. The EpCAM+ subset is tumorigenic.
(a) Cells from pleural effusion samples were separated into 5 different subsets based on their EpCAM and CD45 expression, and whether obtained from a cancer patient (malignant) or otherwise (benign). The EpCAM+ subset was only present in the malignant effusions whereas the EpCAM−/CD45− and CD45+ subsets were found in both the malignant and benign effusions. (b) Malignant pleural effusions were sorted into the EpCAM+, EpCAM−/CD45− and CD45+ subsets by flow cytometry and injected subcutaneously into NOD/SCID/ IL2γR−/− mice and monitored for tumor formation for up to a year. The table shows the ratio of the number of tumors formed to the total number of clinical samples injected. Only the EpCAM+ subset formed tumors. (c) The pleural effusions and their corresponding xenograft tumors were analyzed for their EpCAM and CD45 expression. The xenograft tumors were pre-gated with HLA-ABC antibody to remove contaminating mouse cells.
Before we could evaluate the antigens for cancer specificity, we need to establish which subset is tumorigenic. Therefore, we FACS sorted seven malignant effusions into EpCAM+ , EpCAM−/CD45− and CD45+ subsets and injected the sorted cells subcutaneously into NOD/SCID/IL2γR−/− mice, monitoring for tumor growth for up to a year. The sorting results showed that only the EpCAM+ subset formed tumors in mice, with a 75% efficiency (Supporting Information Fig. 1b). Four of the seven effusions sorted contained EpCAM+ cells and tumor formation was restricted to these EpCAM+ samples. No tumor formation was observed with the EpCAM−/CD45− subset (Fig. 1b). All cells obtained from sorting were injected without normalization to a common cell number, to maximize each subset’s probability of tumor formation (Refer to Supporting Information Table S2 for numbers of cells injected). When pleural effusions were injected into mice without prior sorting, the formed tumors were entirely EpCAM+ (Fig. 1c). For example, the percentage of EpCAM+ cells in the LC52 sample (Refer Supporting Information Fig. S1B for histology of LC52) increased from 0.4% to 99% during tumor growth.
CD24 is one of the several surface markers abundantly expressed in malignant pleural effusions
After establishing that the EpCAM+ subset is tumorigenic, we proceeded to screen surface antigens for their abundance and cancer specificity. We analyzed 12 malignant and 8 benign pleural effusions using flow cytometry (Supporting Information Table S1) and stained the effusions with 35 directly conjugated antibodies. Because the antibodies were labeled with different fluorophores and with different labeling efficiency, we calibrated the raw fluorescence signal to the absolute number of surface receptor molecules using beads with predetermined antibody binding capability (Supporting Information Fig. S2A and Table S3). We used monoclonal antibodies to calibrate the absolute number of receptors because each monoclonal antibody should theoretically bind to a single receptor, allowing us to equate the number of receptors to the number of monoclonal antibodies, with the assumption that all the receptors are fully saturated with antibodies. Polyclonal antibodies are not suitable for this application because multiple polyclonal antibodies may bind to a single receptor, thus overestimating the number of receptors. We included carcinomas of various tissue types including lung, endometrial or colorectal so as to identify ubiquitously expressed markers. The benign effusions came from patients with congestive heart failure or liver diseases.
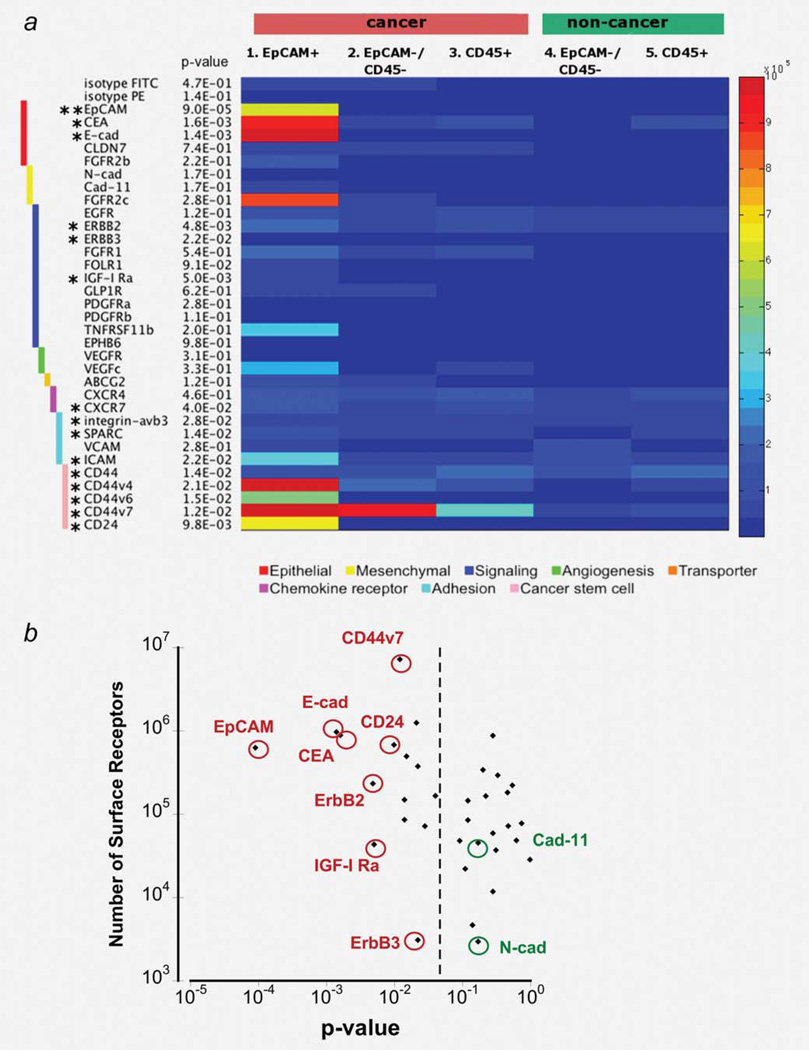
Expression levels of 35 surface markers for each of the five cell subsets described in Figure 1a (malignant EpCAM+, EpCAM−/CD45− and CD45+; benign EpCAM−/CD45− and CD45+) (Refer Supporting Information Fig. S2B for gating strategy) is shown in the heatmap (Fig. 2a), which was obtained by averaging all the patient samples for each of the subsets (Refer to Supporting Information Fig. S2C for individual patient surface antigen expression). An analysis of variance (ANOVA) was applied to each marker and a small p-value indicates significant deviation of any one subset from others (most commonly the cancer patient EpCAM+ subset). A preferred cancer marker was defined as one that is both consistently overexpressed across multiple cancer patients and found abundantly on the cell surface (Fig. 2b).
Figure 2. Surface marker expression in malignant and benign pleural effusions.
(a) Each of the 5 FACS-defined subsets was stained with a panel of 35 surface markers by flow cytometry. We determined the absolute number of receptors on the cell surface with fluorescence calibration beads (exact values given in table S3). Shown are the average numbers of receptors averaged across all patients from each subset. Range shown is 1×105 to 1×106 receptors/cell. Markers with * are differentially expressed in at least one subset with p-value <0.05 (** p-value < 0.001) based on the ANOVA test. (b) For the same surface marker expression in (a), the p-values are plotted against the absolute number of surface markers for the EpCAM+ subset to illustrate the point that an ideal marker is both consistently overexpressed across different patients (p-value < 0.05) and highly expressed (>105 receptors/cell). The dotted line separates the markers with p-value < 0.05 from those with p-value > 0.05. Some example markers are circled in red, representing the statistically significant markers, and in green representing the non-statistically significant markers.
Epithelial markers such as EpCAM, CEA and E-cadherin (E-cad) were among the most highly expressed surface proteins (Figs. 2a and 2b), at close to 1 × 106 receptors/cell in the EpCAM+ subset. In contrast, we did not observe high expression of mesenchymal markers such as N-cadherin (N-cad) and cadherin-11 (Cad-11) in any of the subsets. These mesenchymal markers were expressed at 1 × 103−1 × 104 receptors/cell, approaching background staining. The mesenchymal marker FGFR2c was sometimes overexpressed, but its expression was inconsistent across different patient samples. The expression of signaling receptors predominantly fell between 1 × 104 and 2 × 105 receptors/cell. ErbB2, ErbB3, IGF-I Ra were significantly overexpressed in the EpCAM+ subset although they were much less abundant than the epithelial markers. Integrin αvβ3, SPARC and ICAM1 were the adhesion molecules overexpressed in the EpCAM+ subset. Interestingly, CD44 variants and CD24 but not CD44 were significantly overexpressed in the EpCAM+ subset.
We also compared the marker expression of the EpCAM−/CD45− and CD45+ subsets between the malignant and benign effusions by t-test. There was no significant difference between cancer and non-cancer EpCAM−/CD45− cells. However, the CD45+ cells of cancer samples showed a somewhat higher expression of signaling receptors, namely ErbB2, ErbB3 and IGF-1Ra (Supporting Information Table S4).
CD24 predicts increased tumor growth in xenograft tumors of pleural effusions
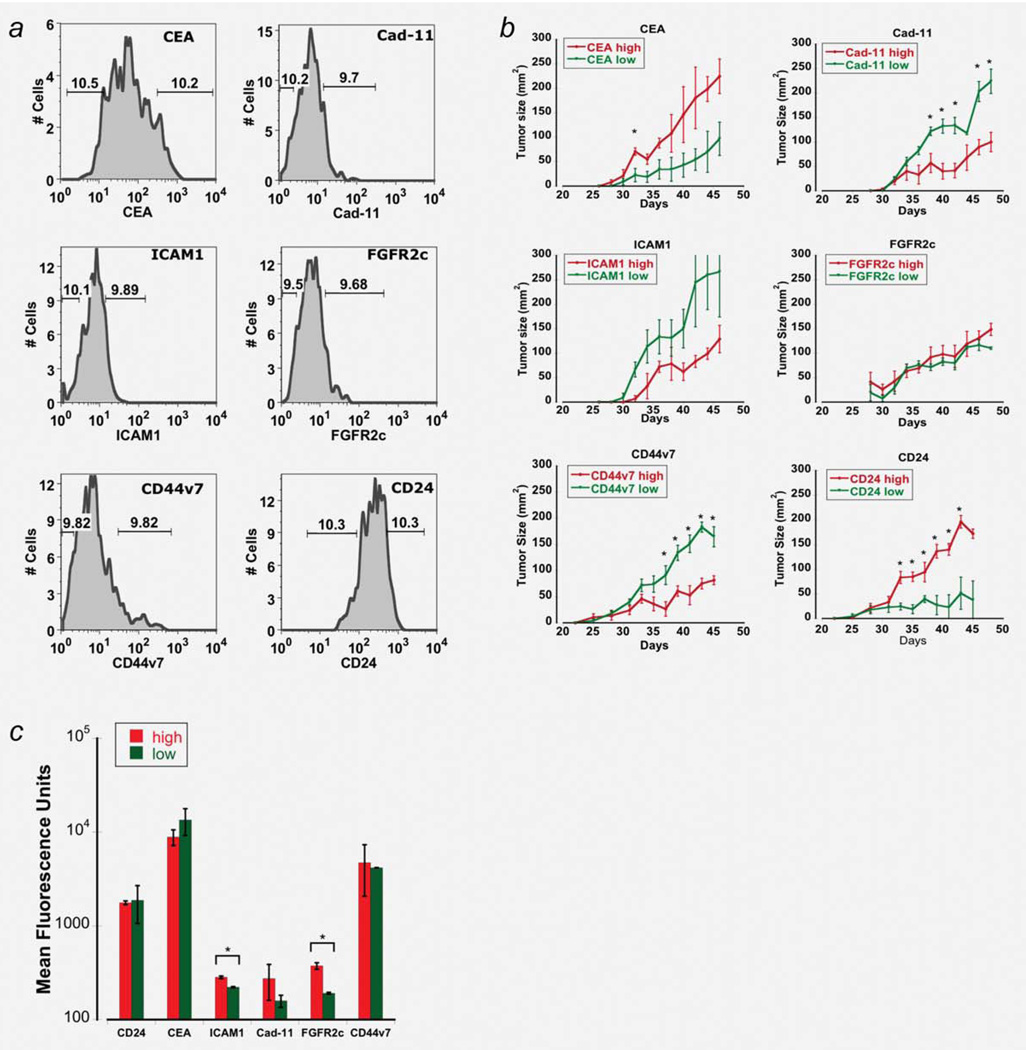
We further explored whether antigens overexpressed in the EpCAM+ population were correlated with tumor growth. To address this question, tumor cells propagated in mice from the pleural effusion of a colorectal cancer patient (LC52) were sorted based on surface marker expression. This xenograft tumor was used because it retained the complex tissue morphology of its origin (Supporting Information Fig. S1B), and could be propagated in the shortest amount of time (around 2 months for each passage) and could grow to a reasonable size yielding sufficient cells for subsequent experiments. A total of six antigens overexpressed in malignant pleural effusions and representing the major categories of surface antigens were screened for their ability to predict tumorigenesis: CEA (epithelial marker), ICAM1 (adhesion marker), CD44v7 and CD24 (stem cell markers), and Cadherin-11 and FGFR2c (mesenchymal markers). After gating on the human cells with human leukocyte antigen class I (HLA-A, B, C) staining, LC52 tumor cells were sorted into the top and bottom 10% expressing each of the six antigens (Fig. 3a), reinjected subcutaneously into NOD/SCID/IL2γR−/− mice, and monitored for tumor growth (Fig. 3b). CD24high, CEAhigh, CD44v7low, and Cadherin-11low populations formed statistically significantly faster-growing tumors. The faster tumor growth observed with cell populations expressing low levels of certain cancer markers suggests that overexpression does not always correlate with faster tumor growth.
Figure 3. CD24high cells are tumorigenic.
(a) To investigate which surface marker predicts tumorigenicity, we used tumor formed from xeno-transplants of the LC52 effusion, FACS-sorted the highest and lowest 10% of cells expressing each of the 6 markers. (b) Sorted cells were injected subcutaneously into NOD/SCID/IL2γR−/− mice and monitored for tumor growth (n=4 per group). Red curves represent the average tumor growth from cells expressing the highest 10% of the indicated markers whereas the green curves represent the tumor growth from cells expressing the lowest 10% of the markers. Error bars shown are the standard errors of the mean. Overexpression of CD24 and CEA correlates with faster tumor growth (*p-value < 0.05 for each day). (c) Tumors formed from high (red bars) and low (green bars) starting populations were analyzed for surface marker expression at the end of the tumor growth studies shown in (b) by flow cytometry (*p-value < 0.05).
To investigate whether tumors retained the same level of surface expression as their starting population, we compared the marker expression in tumors formed from high and low populations by flow cytometry at the end of the tumor induction study (Fig. 3c). Of note, tumors from CD24low and CEAlow cells regained their CD24 and CEA expression, reaching the same level of expression as CD24high and CEAhigh tumors. This implies that marker expression is a dynamic process and that selected markers, in this case CD24 and CEA, are upregulated as tumors form.
CD24 is critical for tumorigenic lung colonization
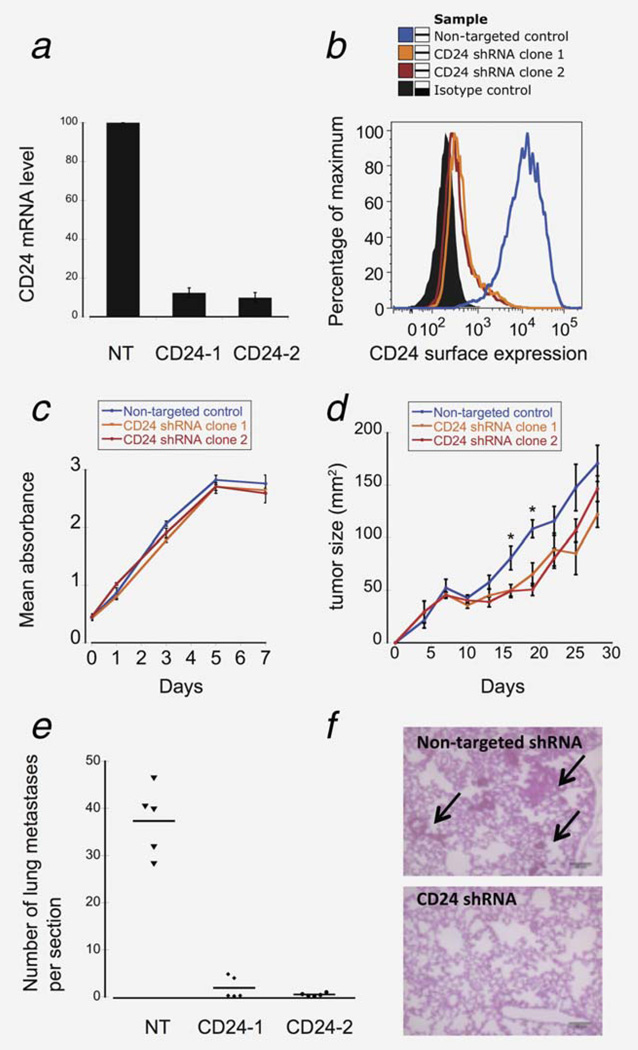
To investigate a possible causal role for CD24 in tumorigenicity, we knocked down CD24 expression by shRNA in the HT29 colorectal adenocarcinoma line (Figs. 4a and 4b). A colorectal line was chosen because LC52 was of rectal origin but LC52 tumor cells were refractory to lentiviral transduction. HT29 was chosen because it has a marker expression profile similar to LC52 and other EpCAM+ pleural effusions (Supporting Information Table S5); high in EpCAM, CEA and CD24.
Figure 4. CD24 is critical for tumor colonization.
To investigate the functional role of CD24, we knocked down the expression of CD24 in HT29 cells. Both the mRNA levels (a) and the surface protein expression levels (b) of CD24 were reduced by 90% in the shRNA clones. NT: non-targeted control; CD24-1: CD24 shRNA clone 1; CD24-2 shRNA clone 2. (c) HT29 cells were plated and monitored for in vitro proliferation. The knockdown of CD24 had little effect on the in vitro proliferation of HT29. (d) HT29 cells were injected subcutaneously and monitored for tumor formation (n=4 per group). (*p-value < 0.05). Error bars shown are the standard errors of the mean. (e) HT29 cells were injected intravenously and the numbers of lung nodules formed were counted one month post injection (n=5 per group). Scale bar = 100 µm. p-value < 0.001 for both shRNA clones. On the H&E section, black arrows indicate some examples of lung nodules formed from HT29 cells with non-targeted shRNA control.
We found that HT29 cells transduced with CD24 shRNA grew at a similar rate in vitro as did the nontargeted control (Fig. 4c). When HT29 cells were injected subcutaneously, CD24 imparted a small but statistically significant growth advantage to tumors during the linear phase (p < 0.05) (Fig. 4d). Both the in vitro and in vivo growth curves are consistent with previous observations (15). Interestingly, whereas CD24high HT29 cells formed numerous tumor nodules in the lungs of mice when injected intravenously, very few nodules were observed when CD24 was knocked down. This result demonstrates that CD24 expression by tumor cells is required for efficient lung colonization (Fig. 4e).
Discussion
Identifying a surface antigen commonly expressed on disseminated tumor cells could help improve therapeutic outcome in metastatic disease. With that objective, we evaluated 35 previously known cancer antigens based on three criteria: cancer specificity, abundance and functional requirement for tumor growth. We used malignant pleural effusions as the source of disseminated tumor cells because tumor cells found in the effusions are highly tumorigenic and much more abundant than other sources of disseminated tumor cells such as circulating tumor cells in blood or cytokeratin positive cells in bone marrow aspirates.
CD24 emerged as a candidate antigen that fulfilled all three criteria simultaneously—it is abundantly and specifically expressed in malignant pleural effusions, and is required for the colonization of the lung. In contrast, not all antigens can meet this tripartite assessment. The CD44 splice variants were previously shown to mediate metastasis of rat carcinoma,15 but in our screening, even though CD44v7 was specifically and abundantly expressed, it did not seem to be required for tumor formation (Fig. 3b). Another example is ErbB3, despite being cancer specific (Fig. 2a) and functionally important,16,17 is expressed at a relatively low level of 3.1 × 103 copies per cell on average. Targeting this low-abundance antigen can be challenging and may require high-affinity antibodies. Lastly, we examined the mesenchymal markers because tumor cells undergoing EMT are highly invasive and anchorage-independent,8 so we expected the highly malignant cells in pleural effusions to exhibit similar expression profile. Contrary to our hypothesis, we found that N-cad and Cad-11, were neither abundant nor cancer specific. The tumor formation experiment also did not suggest any requirement for Cad-11. The low expression of mesenchymal markers in our screening indicates that tumor cells in pleural effusions are not mesenchymal but remain predominately epithelial.
The use of pleural effusions as a source of highly malignant tumor cells can help identify targets unique to metastasis. It is interesting to note that CD24 mediates the lung colonization of HT29 cells without contributing significantly to tumor growth as seen in the subcutaneous tumor growth experiment. It is known that some genes important for metastasis do not actually have a direct effect on tumor growth, such as tumor-derived endothelin-1.18
CD24 is a heavily glycosylated, GPI-anchored peptide expressed in naïve B cells and cancer cells.19 Antibody against CD24 has achieved therapeutic efficacy in preclinical models,20,21 especially in the prevention of metastatic formation in the lungs. Earlier studies ascribed the role of CD24 in tumorigenesis and metastasis as twofold: (i) proliferation and (ii) cell motility. CD24-siRNA knockdowns of HT29 cells showed a reduced exponential growth and lower saturation density, with effects seen more prominently in serum-starved conditions.20 Network analysis using gene expression arrays revealed that downregulation of CD24 affected members in the mitogen-activated protein kinase (MAPK) pathway downstream of Ras. Other pathways affected also included phospholipase C signaling, vascular endothelial growth factor, hypoxia and angiogenesis.20 Another group has shown that CD24 is regulated by Ral GTPases, which mediate cell transformation through Ras.22 Second, the involvement of CD24 in cell motility was observed when CD24-siRNA clones of HT29 cells failed to transpass transwell assays.20 Indeed, studies have shown that CD24 promotes binding to extracellular matrix including fibronectin, laminin, collagen I and collagen V through α3β1 and α4β1 and is important for cell spreading.23
The importance of CD24 may seem contrary to the observation that CD44+/CD24− cells were of increased tumorigenic potential24 and potential cancer stem cells25 in breast cancer. However, the CD44+/CD24− phenotype is not always seen in other cancer types. For example, it is the CD44+/ CD24+ cells that take on the properties of cancer stem cells in pancreatic cancer. Second, despite the evidence of CD24− cells being the cancer stem cells in breast cancer, overexpression of CD24 has a poor prognosis in breast carcinomas including increased staging, tumor grade and lymph node positivity.26 In human bladder cancer, CD24 expression was observed in 75% of the primary tumors and 93% of the matched metastases, suggesting a more advanced tumor staging with higher CD24 expression.21
One improvement that can be made to our study is to include a larger patient size with a defined cancer type. In general, a sample size of 8–12 will only be powered to find effects of a rather large size. This means that only highly overexpressed markers can be identified by this sample size. A sample size of 8 is powered to find effects 1.5 times that of the standard deviation of the population (see Methods). The difference seen for a few markers including CD24 between the EpCAM+ population of the malignant effusions and the other benign populations exceeds this magnitude (Supporting Information Table S6), and thus, our sample size is sufficiently powered to detect the large effects. With our small sample size, we observed considerable patient heterogeneity, indicating the need for patient stratification. We did not limit our patients to lung cancer because effusions from rectal (LC52) and endometrial (LC74) cancers were both tumorigenic in mice. Another limitation of our study is that we could not obtain matched primary tumors because the resection of the primary tumor took place years before the development of pleural effusions and was not always performed in the same hospital. Therefore, it is unclear whether the antigens overexpressed in pleural effusions were present in the primary tumor or in the adjacent healthy tissue. Tumor tissue was formalin-fixed, paraffin-embedded (FFPE); membrane antigens are sensitive to crosslinking, and not all the antibodies are developed for in FFPE slides. Future study would involve the analysis of fresh tumor samples, and waiting 3–5 years for pleural effusions to develop in a subset of patients. Nevertheless, an antigen expressed in normal tissue may still be a useful cancer target when it was found in an unusual location. EpCAM, a marker of both normal and tumor epithelia, indicates metastatic disease when present in pleural effusions, blood or bone marrow. Antibody against EpCAM can completely eliminate EpCAM+ cells in the bone marrow aspirates of some patients.
Lastly, in the quantification of surface antigen abundance, we assume that all the antibodies are equally accessible to the antigens and all antigens are labeled to saturation. It might be more valid to verify the antigen expression by a different antibody targeting another epitope. A more accurate quantification of surface antigen can be accomplished if the Kd of the antibodies is known.
where yeq = fraction of saturation of surface antigens at equilibrium, [Ab] = concentration of antibody, and Kd = affinity of the antibody. However, an estimate of Kd was not available for each of the 35 antigens in this study.
In conclusion, we have applied a general guideline to evaluate the suitability of targets for disseminated tumor cells: the concomitant consideration of antigen abundance, specificity and functional importance, along with the use of tumor cells disseminated from the primary site. CD24 is an antigen that has satisfied all three criteria, especially its functional importance in the colonization of the lung and thus a potential relevance to metastasis. In the future, this screening method can be extended to a greater panel of antibodies for the discovery of targets in disseminated tumor cells.
Supplementary Material
Acknowledgements
The authors thank Dr. Nathan Reticker-Flynn, Dr. Monte Winslow, Dr. Sangeeta Bhatia and Dr. Frank Gertler for their intellectual discussion and Patti Wisniewski for her assistance with flow cytometry and Dr. Eliza Vasile for her assistance with microscopy.
Footnotes
Additional Supporting Information may be found in the online version of this article.
References
- 1.Carter P, Smith L, Ryan M. Identification and validation of cell surface antigens for antibody targeting in oncology. Endocrine-Related Cancer. 2004;11:659–687. doi: 10.1677/erc.1.00766. [DOI] [PubMed] [Google Scholar]
- 2.Verschraegen CF, Hu W, Du Y, et al. Establishment and characterization of cancer cell cultures and xenografts derived from primary or meta-static Mullerian cancers. Clin Cancer Res. 2003;9:845–852. [PubMed] [Google Scholar]
- 3.Camp RL, Dolled-Filhart M, King BL, et al. Quantitative analysis of breast cancer tissue microarrays shows that both high and normal levels of HER2 expression are associated with poor outcome. Cancer Res. 2003;63:1445–1448. [PubMed] [Google Scholar]
- 4.Vogel CL, Cobleigh MA, Tripathy D, et al. Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. J Clin Oncol. 2002;20:719–726. doi: 10.1200/JCO.2002.20.3.719. [DOI] [PubMed] [Google Scholar]
- 5.Harari D, Yarden Y. Molecular mechanisms underlying ErbB2/HER2 action in breast cancer. Oncogene. 2000;19:6102–6114. doi: 10.1038/sj.onc.1203973. [DOI] [PubMed] [Google Scholar]
- 6.Visvader JE, Lindeman GJ. Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008;8:755–768. doi: 10.1038/nrc2499. [DOI] [PubMed] [Google Scholar]
- 7.Hermann PC, Huber SL, Herrler T, et al. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2007;1:313–323. doi: 10.1016/j.stem.2007.06.002. [DOI] [PubMed] [Google Scholar]
- 8.Yang J, Mani SA, Donaher JL, et al. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell. 2004;117:927–939. doi: 10.1016/j.cell.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 9.Minn AJ, Gupta GP, Siegel PM, et al. Genes that mediate breast cancer metastasis to lung. Nature. 2005;436:518–524. doi: 10.1038/nature03799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Winslow MM, Dayton TL, Verhaak RGW, et al. Suppression of lung adenocarcinoma progression by Nkx2-1. Nature. 2011;473:U101–U120. doi: 10.1038/nature09881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Adams GP, Weiner LM. Monoclonal antibody therapy of cancer. Nat Biotechnol. 2005;23:1147–1157. doi: 10.1038/nbt1137. [DOI] [PubMed] [Google Scholar]
- 12.Allard WJ, Matera J, Miller MC, et al. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res. 2004;10:6897–6904. doi: 10.1158/1078-0432.CCR-04-0378. [DOI] [PubMed] [Google Scholar]
- 13.Liang QL, Shi HZ, Qin XJ, et al. Diagnostic accuracy of tumour markers for malignant pleural effusion: a meta-analysis. Thorax. 2008;63:35–41. doi: 10.1136/thx.2007.077958. [DOI] [PubMed] [Google Scholar]
- 14.Lombardi G, Zustovich F, Nicoletto MO, et al. Diagnosis and treatment of malignant pleural effusion: a systematic literature review and new approaches. Am J Clin Oncol. 2010;33:420–423. doi: 10.1097/COC.0b013e3181aacbbf. [DOI] [PubMed] [Google Scholar]
- 15.Hofmann M, Rudy W, Zoller M, et al. CD44 splice variants confer metastatic behavior in rats: homologous sequences are expressed in human tumor cell lines. Cancer Res. 1991;51:5292–5297. [PubMed] [Google Scholar]
- 16.Alimandi M, Romano A, Curia MC, et al. Cooperative signaling of Erbb3 and Erbb2 in neoplastic transformation and human mammary carcinomas. Oncogene. 1995;10:1813–1821. [PubMed] [Google Scholar]
- 17.Schoeberl B, Pace EA, Fitzgerald JB, et al. Therapeutically Targeting ErbB3: a key node in ligand-induced activation of the ErbB Receptor-PI3K Axis. Sci Signal. 2009:2. doi: 10.1126/scisignal.2000352. [DOI] [PubMed] [Google Scholar]
- 18.Said N, Smith S, Sanchez-Carbayo M, et al. Tumor endothelin-1 enhances metastatic colonization of the lung in mouse xenograft models of bladder cancer. J Clin Invest. 2011;121:132–147. doi: 10.1172/JCI42912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kristiansen G, Sammar M, Altevogt P. Tumour biological aspects of CD24, a mucinlike adhesion molecule. J Mol Histol. 2004;35:255–262. doi: 10.1023/b:hijo.0000032357.16261.c5. [DOI] [PubMed] [Google Scholar]
- 20.Sagiv E, Starr A, Rozovski U, et al. Targeting CD24 for treatment of colorectal and pancreatic cancer by monoclonal antibodies or small interfering RNA. Cancer Res. 2008;68:2803–2812. doi: 10.1158/0008-5472.CAN-07-6463. [DOI] [PubMed] [Google Scholar]
- 21.Overdevest JB, Thomas S, Kristiansen G, et al. CD24 offers a therapeutic target for control of bladder cancer metastasis based on a requirement for lung colonization. Cancer Res. 2011;71:3802–3811. doi: 10.1158/0008-5472.CAN-11-0519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Smith SC, Oxford G, Wu Z, et al. The metastasis-associated gene CD24 is regulated by Ral GTPase and is a mediator of cell proliferation and survival in human cancer. Cancer Res. 2006;66:1917–1922. doi: 10.1158/0008-5472.CAN-05-3855. [DOI] [PubMed] [Google Scholar]
- 23.Braun S, Hepp F, Kentenich CRM, et al. Monoclonal antibody therapy with Edrecolomab in breast cancer patients: monitoring of elimination of disseminated cytokeratin-positive tumor cells in bone marrow. Clin Cancer Res. 1999;5:3999–4004. [PubMed] [Google Scholar]
- 24.Al-Hajj M, Wicha MS, Benito-Hernandez A, et al. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA. 2003;100:3983–3988. doi: 10.1073/pnas.0530291100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mani SA, Guo W, Liao MJ, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133:704–715. doi: 10.1016/j.cell.2008.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Athanassiadou P, Grapsa D, Gonidi M, et al. CD24 expression has a prognostic impact in breast carcinoma. Pathol Res Prac. 2009;205:524–533. doi: 10.1016/j.prp.2009.01.008. [DOI] [PubMed] [Google Scholar]
- 27.Ponta H, Sherman L, Herrlich PA. CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol. 2003;4:33–45. doi: 10.1038/nrm1004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.